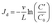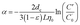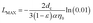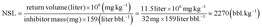DOI:
10.1039/C6RA09973D
(Paper)
RSC Adv., 2016,
6, 59195-59205
Phosphino-polycarboxylic acid modified inhibitor nanomaterial for oilfield scale control: transport and inhibitor return in formation media†
Received
18th April 2016
, Accepted 5th June 2016
First published on 6th June 2016
Abstract
Mineral scale deposition is a major threat to the oil and gas industry. Scale threat is conventionally controlled by injecting a scale inhibitor chemical into oil production systems. Scale inhibitor nanomaterials are an alternative to the conventional inhibitor products. This study evaluates the transport and inhibitor return properties of the phosphino-polycarboxylic acid (PPCA) modified scale inhibitor nanomaterial with both PPCA and phosphonate constituents. The objective is to expand the use of this type of inhibitor nanomaterial in the delivery of scale inhibitor into formation for oilfield scale control. Electrolyte content and the sonication method have been investigated for their roles in nanomaterial transport in formation medium. It has been shown that the nanofluid electrolyte content can considerably impact the nanomaterial transport in terms of breakthrough level, deposition towards formation surfaces and inhibitor solid phase distribution. It is evident that an increase in electrolyte content can reduce the transportability of the nanomaterial. Consequently, the deposition tendency and also inhibitor solid phase distribution can be influenced as well. Furthermore, it shows that sonication treatment can result in a change in particle size with varied transportability. Laboratory squeeze simulation studies have been conducted to compare the squeeze lifetime of inhibitor nanomaterial against conventional pills. It is evident that the nanomaterial outperformed the conventional pills, with an extended squeeze lifetime. Overall, the transport studies and squeeze simulations suggest that the prepared inhibitor nanomaterial has the potential for application in the oilfield to control the scale deposition threat.
Introduction
Soluble ionic species are ubiquitous in natural waters and industrial processed waters. Changes in the water conditions, such as temperature and pressure, can lead to the precipitation of inorganic mineral solids.1,2 The precipitated minerals are also called mineral scales (or scales). Scale deposition is a result of local oversaturation, with respect to the minerals. Scale deposition is a common phenomenon in various commercial and industrial processes, such as desalination, heating, cooling and oil production. Scale solids can form on the surfaces of water boilers, heating media, pipe walls, and tubings, etc. The deposited scale solids can block the pipeline throughout, leading to detrimental outcomes such as pipe corrosion, impedance of heat transfer and increased operational risks and costs.1,2 Particularly, in the oil and gas industry, scale deposition and blockage is one of the most commonly observed operational issues.3–5 Scale can deposit on any oilfield facility surface from downhole to gathering facilities, and further to processing facilities. Scale deposition can lead to formation blockage, narrowed tubing diameters, malfunctioning valves, etc. The most efficient strategy to control scale deposition is to inhibit scale formation rather than mitigating scale damage.5–7 The most common technique for scale inhibition is to inject scale inhibitor (inhibitor) into the oil production system. Scale inhibitors are water soluble chemicals and they need to be dissolved in the produced water in order to inhibit scale deposition. Among various types of inhibitors, phosphonates are the most widely used in the oilfield.5,6 The inhibitor is normally delivered into the production systems in a continuous injection mode through an injection mandrel located on the production system. Alternatively, the inhibitor can be delivered in batch mode by injecting (squeezing) a volume of inhibitor solution (pill solution) into the formation.
The additional benefit of scale squeeze treatment is to protect near wellbore formation and downhole production tubing. Typically, a scale squeeze treatment is initiated by injecting a volume of solution through a production well into the reservoir to clean up the formation (preflush). Next, an inhibitor pill is injected into the reservoir, followed by another volume of solution to push the injected inhibitor deeper into the formation (overflush). The inhibitor pill is basically a concentrated aqueous solution of inhibitor. The benefits of overflush treatment include enhanced inhibitor adsorption to formation surfaces and final tubing volume displacement.8 Subsequently, the well will be shut-in for a few hours to allow the injected inhibitor to attach to formation material surfaces. Following the shut-in period, the well will be put back on to allow formation fluids to flow into the production well. The delivered inhibitor will be desorbed or dissolved into produced water to control scale deposition (inhibitor return). An enhanced attachment of the injected inhibitor to formation surfaces is preferred, since the unattached free inhibitor will be rapidly flushed out of the formation with a high return concentration shortly after the resumed production. As is commonly observed in the field, as much as one third of the injected inhibitor will return within a few days of resumed production;6 however, it is desirable to be able to control the long-term return behavior of the inhibitor. A longer squeeze lifetime is always preferred, which translates to a less frequent squeeze treatment and a reduced operational cost. The inhibitor placement and squeeze lifetime are dictated by the inhibitor-formation interaction, which includes adsorption and precipitation.9–11 Kan et al.11 proposed that the return phosphonate concentration is determined by the desorption of the adsorbed inhibitor from formation surfaces at low phosphonate concentrations, while at high phosphonate concentrations, the inhibitor will form precipitates with divalent metals in the formation and the return concentration is governed by the dissolution of the metal–phosphonate precipitate. Consequently, precipitation squeeze and adsorption squeeze can be carried out in the field to extend squeeze lifetime.12–15 For instance, if an acidic pill is injected into the formation, the acid can dissolve the formation material, leading to an increase in solution pH and metal–phosphonate precipitation (precipitation squeeze); if a neutralized pill is injected, the inhibitor will attach to the formation surfaces by chemical adsorption (adsorption squeeze). The drawback of precipitation squeeze is that the inhibitor tends to have a limited transport distance inside the formation, resulting in a reduced surface area of formation to be treated; the neutralized pill has an enhanced inhibitor migration capacity, but a shorter squeeze lifetime, due to weak adsorption to formation surfaces.15
The idea of preparing a scale inhibitor nanomaterial (SINM) has been proposed and experimentally tested in the past few years.16–21 Commonly, the SINM particles were prepared by combining a divalent metal solution with a phosphonate solution. The obtained SINM aqueous solution (nanofluid) is stabilized by various methods, including ultrasonication, surfactant coating, silica templating, etc. These researchers typically followed the scheme of testing the properties of SINM as a carrier to deliver inhibitor into deep formation. A key property of SINM is the transportability in the formation medium.19,20 Laboratory transport experiments of SINM follow the approach of investigating transport behaviors of SINM particles in porous media.22–25 Obviously, it is desirable to be able to deliver these nanomaterials deeper into the formation, so as to treat a larger formation surface area. However, a limited transportability suggests that the majority of the injected nanomaterials will be accumulated in the vicinity of the wellbore, potentially leading to formation blockage. Another key property of SINM is the long-term inhibitor return behavior. As reported previously,19 SINM exhibits an enhanced return property with a prolonged squeeze lifetime. This is believed to be due to the slow dissolution of the pre-precipitated metal–phosphonate complex in produced water.19
Recently, we reported the synthesis and characterization of a polymer modified Ca–phosphonate SINM and its nanofluid.26 The prepared SINM combines both phosphonate and phosphino polycarboxylic acid (PPCA) polymer. A more effective scale inhibition is expected by employing such a SINM with both phosphonate and phosphino polymer. However, through an acid/base and metal complexation chemistry investigation, it has been reported that calcium (Ca) ions can form a strong complexation with PPCA molecules.27 Therefore, it can be argued that the complexation of PPCA with mobile Ca ions can impact the migration of the PPCA modified SINM, leading to a reduced transportability in the formation medium. Furthermore, one might argue that the Ca–PPCA complexation reaction can impede the dissolution of phosphonate from the PPCA modified SINM, resulting in a reduced return behavior, compared with other SINMs. In this study, we systematically evaluated the transportability of the PPCA modified SINM via laboratory column transport experiments. The impacts of electrolyte content and sonication treatment on SINM transport at various experimental conditions were investigated, and contrary to the above speculations, the PPCA modified SINM was found to be transportable through the formation medium. A laboratory squeeze simulation test was conducted to examine the return behavior of this nanomaterial. The novelty of this study is the evaluation of the transportability and return behavior of the PPCA and phosphonate combined SINM in the formation medium. This study suggests that the SINM of interest has the potential to be applied for oilfield scale control. The investigations in this study can further the understanding about the transport and return properties of the PPCA and phosphonate combined SINM to expand its use in the delivery of inhibitors into formation for scale control.
Experimental
Chemicals
Commercial grade diethylenetriaminepentakis(methylene phosphonic acid) (DTPMP) with 50% activity was used as the scale inhibitor (Solutia Inc.). Phosphino polycarboxylic acid (PPCA) with 50% activity (wt/wt) was purchased from BWA Water Additives. Chemicals such as calcium chloride, sodium chloride, potassium chloride, sodium hydroxide and hydrochloric acid were reagent grade and purchased from Fisher Scientific. Tritiated water was purchased from Sigma-Aldrich. Deionized water (DI water) was prepared by a reverse osmosis and ion exchange water purification process.
PPCA modified Ca–DTPMP nanofluid synthesis
As detailed previously,26 PPCA modified Ca–DTPMP SINM was prepared via a two-step synthesis approach. Initially, a Ca–DTPMP suspension was prepared by combining CaCl2 solution with a neutralized DTPMP solution (11.3 pH) at 90 °C. Then, a white Ca–DTPMP precipitate was obtained by centrifuging the suspension. Subsequently, the resultant white precipitate was dispersed into a PPCA solution (0.2% wt/wt) with the assistance of ultrasonication treatment. Both bath and probe sonicators were employed in the nanofluid preparation (details in the ESI†). The DTPMP loading in the nanofluid was about 0.8% (wt/wt) and the pH of the prepared nanofluid was ca. 7.2. A number of nanofluids were prepared with varying KCl content and different sonication treatments. The synthesis conditions of the nanofluids of interest in this study are tabulated in Table 1. The particle sizes of different SINM were determined by dynamic light scattering (DLS) (Zetasizer Nano, Malvern Instruments). The uncertainty of the DLS measurement is generally about 5%.
Table 1 Conditions of Ca–DTPMP nanomaterial sample preparationa
| Experiment # |
KCl (% wt/wt) |
PPCA (% wt/wt) |
Sonication treatment |
Sonication time (min) |
Particle size (nm)b |
| The synthesis conditions for nanofluid preparation were reported in a previous study, ref. 26. The particle size of SINM particles was measured by DLS for Exp. #1 to #4. The particle size of Exp. #5 particles was estimated based upon the transport experimental results. It shows that Sample #5 demonstrated a similar breakthrough level to that of Sample #2, as shown in Fig. 3. Thus, a particle size of 190 nm was assigned to Sample #5 to facilitate the transport parameter calculations. The particle sizes of Exp. #6 and #7 were not measured by DLS or evaluated via transport experiments. However, squeeze tests showed that they were transportable in Frio sandstone medium with a desirable solid phase distribution, immediately after overflush treatment, as shown in Fig. 4. |
| 1 |
0 |
0.2 |
Bath |
25 |
180 |
| 2 |
0.2 |
0.2 |
Bath |
25 |
190 |
| 3 |
0.5 |
0.2 |
Bath |
25 |
200 |
| 4 |
1 |
0.2 |
Bath |
25 |
420 |
| 5 |
1 |
0.2 |
Probe |
25 |
190c |
| 6 |
2 |
0.3 |
Probe |
75 |
N.A.d |
| 7 |
1 |
0.4 |
Probe |
40 |
N.A.d |
Laboratory column transport experiments
Transport behavior of the prepared Ca–DTPMP nanomaterial was evaluated via a number of column transport experiments in calcite medium. As detailed in the previous studies,19,26 calcium carbonate (calcite, Creel Chihuahua, Mexico) was packed into a stainless column of 1.09 cm inner diameter (ID) and 8 cm length. The grain size distribution of the calcite material was 106–180 μm. Initially, a non-reactive tracer (tritiated water, 3H2O) test was conducted to measure the pore volume (PV) and dispersion coefficient of the packed calcite medium. PV is essentially the empty space inside the calcite column and can be calculated as the total column volume minus the volume occupied by calcite particles. Following the tracer test, the calcite column was flushed with a NaCl solution (1 M) for ca. 4 PV. Subsequently, the nanofluid transport experiment was performed by injecting the prepared nanofluid into the column at 70 °C at a flow rate of 60 mL h−1, corresponding to a linear pore velocity of 2 cm min−1 (Table 2). By the end of each transport experiment, the calcite material was extruded out of the column and separated into segments from one end to the other end of the column. A fraction of the each segment was dissolved in HCl solution (1 M) for DTPMP content measurement, in order to establish a DTPMP solid phase distribution.
Table 2 Conditions of nanomaterial column transport experiments
| Transport Experiment (TE) #a |
KCl (% wt/wt) |
PPCA (% wt/wt) |
Sonication treatment |
Formation medium |
Flow rate (mL h−1) |
Pore velocity (cm min−1) |
Experimental temperature (°C) |
Dispersion coefficient, D (cm2 min−1) |
| The samples used in each transport experiment listed in this table were prepared at the synthesis conditions, as tabulated in Table 1, with the same sample #. The transport studies of TE #1 and #2 were presented in the previous study at ref. 26. |
| TE-1b |
0 |
0.2 |
Bath for 25 min |
Calcite for all cases |
60 mL h−1 for all cases |
2 cm min−1 for all cases |
70 °C for all cases |
0.065 cm2 min−1 for all cases |
| TE-2b |
0.2 |
0.2 |
Bath for 25 min |
| TE-3 |
0.5 |
0.2 |
Bath for 25 min |
| TE-4 |
1 |
0.2 |
Bath for 25 min |
| TE-5 |
1 |
0.2 |
Probe for 25 min |
Laboratory squeeze simulation tests
In this study, totally contained squeeze simulation apparatus was adopted for laboratory squeeze simulation studies of the prepared inhibitor nanofluid in a sandstone medium.19,20 A stainless steel column (1.09 cm ID and 16 cm length) was packed with Frio sandstone material. Frio sandstone was collected from Frio formation located in Galveston County, TX, at a depth of 9107–9110 ft. The sandstone was ground into fine particles with a grain size distribution of 106–180 μm. Nanofluids prepared under the conditions of Exp. 6 and Exp. 7 in Table 1 were selected for the squeeze simulation tests. About 30 min before the onset of the squeeze experiment, the inhibitor nanofluid was sonicated with a probe sonicator for 25 min. The squeeze simulation test was initiated by flushing the sandstone column with a KCl solution (1% wt/wt) for a few PV to saturate the sandstone medium. Following the KCl solution injection, half of a PV of the sonicated nanofluid was injected into the sandstone column. Immediately after the nanofluid injection, another half a PV of NaCl solution (1 M) was injected into the sandstone column to push the previously injected nanofluid into the second half of the column (overflush). Subsequently, the sandstone column was shut-in for 18 h to allow the injected inhibitor nanomaterial to affix to the surface of the sandstone medium. Afterwards, the column was eluted with synthetic brine (0.025 M CaCl2, 0.015 M NaHCO3 and 1 M NaCl, sparged with 100% CO2) from the opposite direction at 70 °C and 75 psi back pressure, to simulate the field brine production. The flow rate of the synthetic brine was 60 mL h−1, corresponding to a linear pore velocity of 2 cm min−1. The eluted brine out of the sandstone column was collected and analyzed for DTPMP concentration, so as to establish a DTPMP return concentration vs. PV of brine flushing (return curve). As a comparison, the above squeeze simulation experiment was repeated with a neutralized pill and an acidic pill instead of nanofluid, under the same experimental conditions (70 °C, 75 psi and 60 mL h−1). The neutralized pill was an aqueous solution of neutralized DTPMP (0.8% wt/wt, pH 6.7, neutralized with 5 M NaOH); the acidic pill was an aqueous solution of acidic DTPMP (0.8% wt/wt, pH 1).
Analytical methods
Calcium and phosphorus concentrations were analyzed by inductively coupled plasma-optical emission spectrometry (ICP-OES) (Optima 4300 Dv, Perkin Elmer). Measurement of low concentrations of DTPMP from the column eluted samples was realized by a spectrophotometric method (Hach Co.); as little as 0.12 mg L−1 of DTPMP can be measured by this method (details in the ESI†).
Results and discussion
Transport of inhibitor nanomaterial in calcite medium column
The previous SINM characterization study suggested that both electrolyte (KCl) content and sonication method can greatly impact the properties of the prepared SINM, including particle size and particle stability.26 As detailed in Table 1, the increase in KCl wt% from 0 to 1% (Exp. #1 to #4) resulted in an increase in particle size from 180 nm to 420 nm. It also shows that the increase in KCl content led to a reduction in particle stability, characterized by an increase in the measured zeta potential (details in the ESI†). Since DLS measures the hydrodynamic radius, the particle size values in Table 1 should be interpreted only as the relative sizes of the particles. Because of the electrolyte impact, the previous column transport study using Sample #1 (0% KCl) resulted in an enhanced transportability in terms of final breakthrough, compared with that using Sample #2 (0.2% KCl). In this study, we further evaluate the influences of nanofluid electrolyte content, coupled with the sonication method, on SINM transport behaviour in calcite medium. As shown in Table 1, seven nanofluids were prepared with varying KCl content and sonication treatment. The objective of the transport study is to explore the synthesis conditions, to prepare SINM and its nanofluid with an improved transportability in formation medium for oilfield scale control.
Theories to explain SINM transport in formation medium
A one-dimensional advection dispersive equation (1-D ADE) is commonly adopted to explain the transport behavior of particles in porous medium. The expression of 1-D ADE is as follows:28–30| |
 | (1) |
where C (mg L−1) is the SINM concentration in the effluent at a given time; t (min) is the time; x (cm) is the SINM migration distance inside the column; D (cm2 min−1) is the hydrodynamic dispersion coefficient; v (cm min−1) is the linear pore velocity. R is the retardation factor and Jd (min−1) is the deposition rate coefficient of SINM particles to calcite medium surfaces. PV and D values are determined from the tracer test (details of 1-D ADE are explained in the ESI†). R describes the sorption of SINM particles to calcite medium surfaces during flow, and Jd signifies the deposition of SINM particles to calcite surfaces. Furthermore, the mathematical solution to eqn (1) can be obtained, assuming a constant inlet concentration28,30 (details in the ESI†). Table 2 tabulates the experimental conditions of five of the prepared SINM in column transport studies. As shown in Table 3, the calculated final breakthrough levels based on 1-D ADE are very similar to the experimentally determined ones for all the transport experiments evaluated in this study. Additionally, according to the filtration theory, the Jd value can be calculated as follows:29–31| |
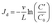 | (2) |
where L (cm) is the length of the stainless steel column (8 cm in this study) and C′/C′0 is the measured final breakthrough level. As shown in Table 3, the calculated Jd values for TE #1 to #5 based on eqn (1) and (2) are very similar, suggesting the agreement on Jd value calculation and also the validity of these two modeling approaches.
Table 3 Transport experimental results from the standpoint of advection and diffusion
| Transport Experiment (TE) #a |
Preflush fluid |
Observed final C′/C′0 (%) |
Calculated final C′/C′0 (%) |
Removal coefficient, Jd (min−1)c |
Removal coefficient, Jd (min−1)d |
Retardation factor, R |
| The samples used in each transport experiment listed in this table were prepared under the synthesis conditions as tabulated in Table 1, with the same sample #. The transport studies of TE #1 and #2 were presented in the previous study at ref. 26. The Jd values in this column are calculated according to the mathematical solution to eqn (1) with consideration of 1-D ADE.28–30 The Jd values in this column are calculated according to eqn (2), with consideration of filtration theory.29,30 The calculated parameters for TE #4 are not available, since quite a few observed breakthrough levels were zero, making the calculation unable to converge. |
| TE-1b |
1 M NaCl for all the cases |
99.8 |
99.9 |
0.0006 |
0.0005 |
1.28 |
| TE-2b |
95.8 |
95.8 |
0.010 |
0.010 |
1.25 |
| TE-3 |
94.3 |
92.7 |
0.019 |
0.016 |
1.47 |
| TE-4 |
2.0 |
N.A.e |
N.A.e |
N.A.e |
N.A.e |
| TE-5 |
98.7 |
95.5 |
0.012 |
0.012 |
1.26 |
According to the theories related to particle filtration and attachment, transport of submicron sized particles in porous medium can be viewed as a process of these particles being continuously removed by the medium by means of collection and attachment to the medium. Thus, SINM transport in formation medium can be elaborated from the standpoint of particle collection and attachment. Collection efficiency (η0) and attachment efficiency (α) were adopted to illustrate the collection and attachment of SINM particles to the medium, respectively. Collection of SINM by formation is a result of Brownian diffusion, interception and sedimentation (details in the ESI†). α can be calculated as follows:29,32
| |
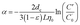 | (3) |
where
dc is the calcite medium diameter (143 μm in this study) and
ε is the porosity of the calcite medium (0.53 in this study). Normally, a higher
η0 value (or
α value) suggests a higher tendency for the particles to be removed by the medium surfaces. Rearranging the above
eqn (3) can generate the expression of
L (cm) as a function of
η0,
α and
C′/
C′0. Assuming a final breakthrough level of 1% (
i.e.,
C′/
C′0 = 0.01), a maximum transport distance (
LMAX) for SINM particles can be defined.
29 Therefore, according to
eqn (3),
LMAX can be calculated as follows:
| |
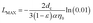 | (4) |
According to Table 2, the transport study encompasses five individual experiments of SINM with different compositions and/or sonication treatment. Of particular interest is the impact of nanofluid KCl content on SINM transport. The previous studies on inhibitor nanomaterials typically utilized calcite and sandstone materials as the formation media,19–21 because among many formation materials, calcite is the primary one responsible for phosphonate retention. Sandstone material is typically obtained from an oilfield and can appropriately represent the reservoir formation of interest. In this study, calcite was employed for SINM transport experiments; sandstone was used for squeeze simulation studies, as reported in the next section. All transport experiments considered in this study were conducted at the same flow rate of 60 mL h−1, which corresponds to a linear pore velocity of 2 cm min−1 at 70 °C (Table 2).
Impact of electrolyte content on SINM transport
Tables 1 and 2 tabulate the SINM synthesis and transport experiment (TE) conditions. Tables 3 and 4 detail the TE results from the viewpoints of 1-D ADE and particle filtration and attachment, respectively. TE #1 to #4 were deliberately designed to evaluate the impact of KCl content on SINM transport with bath sonication treatment. By evaluating the measured breakthrough levels in TE #1 to #4 (Table 3 and Fig. 1), the increase in KCl content obviously led to a decrease in SINM breakthrough level. Among TE #1 to TE #3, breakthrough level fell from almost 100% to ca. 94%. TE #4 encountered a more dramatic reduction to only 2% breakthrough. As elaborated in the previous study,19,26 KCl content can substantially impact the SINM particle size and particle stability by suppressing the diffuse double layer. One can argue that the observed breakthrough level reduction is a result of the increase in SINM particle size and reduction in particle stability. Particularly, the fact that the breakthrough level for TE #4 was almost zero, suggests that SINM with 1% KCl was unstable and not transportable through calcite medium. Correspondingly, from the standpoint of 1-D ADE, the calculated R and Jd values are also impacted by KCl content. According to Table 3, with the increase of KCl content from TE #1 to TE #4, the R value is calculated to change insignificantly from 1.28 to 1.47, while the Jd value experienced a more marked increase from 0.0006 to 0.019 (over 30 times). As for TE #4, R and Jd values are not available; since most of the measured breakthrough levels were zero, the calculation was unable to converge. Since R value is an evaluation of the sorption of SINM to the formation medium,28 the insignificant change in R values indicates that the SINM sorption to the calcite surface is less sensitive to electrolyte content. Jd value, on the other hand, is a measure of the deposition tendency of SINM towards calcite surfaces.28,29 The substantial increase in Jd value suggests that the increase in KCl content could drive SINM particles to deposit onto the calcite surfaces, as a result of a shrinking, diffuse double layer. From the standpoint of particle filtration and attachment, the increase in KCl led to a minor decrease in the calculated η0 value and a considerable increase in α value (Table 4). The change in η0 in this study is mainly a result of variation in SINM particle size. The α value is dependent on both η0 and final breakthrough level. The high α value for TE #4 is calculated based on only 2% breakthrough. These calculation results suggest that the increase in KCl content resulted in an increase in SINM particle size, which accounts for a slight decrease in the SINM particle collection by calcite medium (η0 value) and a considerable increase in the SINM particle attachment to calcite surfaces (α value). In addition, the KCl impact can be reflected by the calculated LMAX (Table 4). Considering the experimental conditions in this study, LMAX is mainly influenced by the α value. The increase in α value reduced the calculated LMAX from 184 m in TE #1 to only 0.1 m in TE #4, suggesting a significant reduction in SINM transportability in calcite. Finally, the impact of KCl content can be understood by examining the DTPMP distribution in the calcite packed column (DTPMP solid phase distribution) post the TE. As discussed in the previous study,26 DTPMP solid phase distribution can reveal the SINM transportability in calcite medium: SINM particles with an enhanced transportability will distribute more uniformly throughout the length of the calcite column; the ones with a less desirable transportability will tend to accumulate in the first half of the column. According to Fig. 2, DTPMP distributions of TE #1 to #3 show a relatively uniform distribution inside the column, with an average DTPMP loading calculated as 2.9 ± 0.6 mg g−1 (solid line in Fig. 2). Distribution of TE #4 exhibits a quite different pattern, with the majority of DTPMP accumulated in the first half of the column and little DTPMP detected in the second half. Evidently, the observed limited transportability of SINM in TE #4 is responsible for the uneven DTPMP solid phase distribution inside the column. Based on the above discussions, one can argue that the lower the electrolyte content, the better the transportability of SINM in calcite formation medium. However, it should be noted that electrolyte in the inhibitor pill is a key component in preventing formation damage related to clay swelling and particle migration.6,33 Thus, the electrolyte content in the inhibitor nanofluid should be carefully evaluated in order to understand the impact of electrolyte on the formation material of interest.
 |
| | Fig. 1 Scale inhibitor nanomaterial breakthrough curves in calcite medium for TE #1 to TE #4. The markers (diamond, square, triangle and dot) are experimentally obtained breakthrough levels from TE #1 to TE #4; the dashed lines are the calculated breakthrough levels according to the mathematical solution to eqn (1). Breakthrough curves in TE #1 and TE #2 were reported in the previous study.26 | |
Table 4 Transport experimental results from the standpoint of filtration and attachment
| Transport Experiment (TE) #a |
Observed final C′/C′0 (%) |
Calculated final C′/C′0 (%) |
η0 (× 10−4) |
α (× 10−2) |
LMAX (m) |
| The samples used in each transport experiment listed in this table were prepared under the synthesis conditions as tabulated in Table 1, with the same sample #. The calculated C′/C′0 for TE #4 is not available, since quite a few observed breakthrough levels were zero, making the calculation unable to converge. η0, α and LMAX values for TE #4 were calculated based upon the measured final breakthrough level of 2% in TE #4. |
| TE-1 |
99.8 |
99.9 |
124.7 |
0.04 |
184.0 |
| TE-2 |
95.8 |
95.8 |
120.6 |
0.91 |
8.6 |
| TE-3 |
94.3 |
92.7 |
117.0 |
1.28 |
6.3 |
| TE-4 |
2.0 |
N.A.b |
97.4c |
102.70c |
0.1c |
| TE-5 |
98.7 |
95.5 |
120.6 |
0.28 |
28.2 |
 |
| | Fig. 2 DTPMP solid phase distribution after column transport experiments for TE #1 to TE #4. The dashed line represents 2.9 mg g−1, which is the calculated average DTPMP loading for TE #1 to #3. DTPMP distribution after TE #1 was reported in the previous study.26 | |
Impact of sonication treatment
As discussed in a previous study, a probe sonicator can provide more robust fragmentation energy to break SINM into smaller particles, compared with a bath sonicator.34 Comparing Exp. #4 and #5 in Table 1, a probe sonicator can significantly reduce the particle size of SINM with the same electrolyte composition (1% KCl) and sonication time (25 min). Such a difference can be further illustrated by the SINM particle breakthrough curves for both scenarios (TE #4 and #5 in Fig. 3). It shows that the probe sonicated SINM in TE #5 demonstrated a considerably enhanced transportability, compared with bath sonicated SINM in TE #4. TE #5 exhibited a breakthrough pattern with over 95% breakthrough, similar to TE #2 (bath sonication, low KCl) (Table 3). The calculated Jd value of TE #5 was similar to that of TE #2 as well, suggesting a low tendency to deposit onto calcite surfaces. The calculated α value for TE #5 was lower than that in TE #2 or TE #4, accounting for a prolonged LMAX in calcite medium (Table 4).
 |
| | Fig. 3 Inhibitor nanomaterial breakthrough curves for TE #5, compared with curves of TE #2 and TE #4. The markers (square, dot and triangle) are experimentally obtained breakthrough levels of TE #2, #4 and #5; the dashed lines are the calculated breakthrough levels based on the mathematical solution to eqn (1). | |
Laboratory inhibitor nanofluid squeeze simulation
Understanding DTPMP return from SINM. The laboratory squeeze simulation experiment (SE) is a laboratory testing approach to evaluate the performance of a scale inhibitor product in terms of its return property and squeeze lifetime. Squeeze simulation testing includes the same component procedures of preflush, pill injection, overflush, shut-in and resumed production, as in the field operation. The synthetic brine used in this study was in equilibrium with respect to calcite, so that no calcite solid would be dissolved or formed during the test, as expected for reservoir flowback. In this study, two Ca–DTPMP nanofluids (Sample #6 and Sample #7 in Table 1) were selected to evaluate their return behaviors. These two samples were prepared at a high KCl content, due to the considerations of maintaining formation integrity and controlling clay swelling.33 Both samples were sonicated with a probe sonicator 30 min prior to the test to enhance their transportability inside the formation. In one set of SE, the sandstone column was flushed with a KCl solution (1%) to simulate the preflush treatment, since KCl is a common preflush fluid in field squeeze operation. Subsequently, half of a PV of the nanofluid was injected into the formation, followed by another half of a PV of 1 M NaCl, a common field overflush fluid, to push nanofluid to the other end of the column (overflush treatment). Immediately after the overflush treatment, the sandstone material inside the column was extracted to measure the DTPMP solid phase distribution (Fig. 4). Evidently, for both nanofluids tested, the majority of the injected SINM was pushed to the second half of the column, signified by the fact that DTPMP solid phase loading in the second half of column was much higher than in the first half. This result suggests that the SINM of interest is transportable inside the sandstone column and they can be pushed deeper into the formation for scale control. The significance of this study is that in an oilfield squeeze treatment, the nanofluid can be deployed deep into formation via pill injection and subsequent overflush treatment. In a separate study, full scale squeeze simulation tests in Frio sandstone columns were carried out (SE #1 and #2 in Table 5) using nanofluids from Sample #6 and Sample #7 in Table 1. After the overflush treatment, the column was shut-in for 18 h, to simulate the similar treatment in the field for the objective of allowing the inhibitor to attach to formation surfaces. Afterwards, the sandstone column was eluted with the calcite-equilibrated synthetic brine from a reverse direction to simulate the resumed production in the field. As a comparison, two additional squeeze simulation tests (SE #3 and #4 in Table 5) were performed using an acidic DTPMP pill (1.5 pH) and a neutralized DTPMP pill (6.7 pH), respectively. The conditions for the four SE were the same at 70 °C, 75 psi back pressure and a synthetic brine flow velocity of 2 cm min−1 (Table 5). By evaluating the return curves obtained from these four SE studies, one can argue that the return curve patterns of SE #1 and #2 (nanofluid) are relatively similar. DTPMP return concentrations drop to 10 ppm (V10ppm) between 100 and 130 PV and to 1 ppm (V1ppm) at 540 PV and further to 0.5 ppm (V0.5ppm) at ca. 700 PV (Fig. 5 and Table 6). SE #3 (acidic pill) exhibited a reduced squeeze lifetime with V1ppm and V0.5ppm of 240 PV and 450 PV, respectively. As for SE #4 (neutralized pill), return concentrations rapidly drop to 10 ppm at only 10 PV and to 1 ppm at 50 PV. Generally speaking, the total squeeze lifetime of these four studies is in the sequence of neutralized pill (SE #4) of 230 PV < acidic pill (SE #3) of 870 PV < SINM with 1% KCl (SE #2) of 1080 PV < SINM with 2% KCl (SE #1) of 1440 PV. As discussed above, the neutralized pill has a weak adsorption to the formation surface, compared with the acidic pill, because a fraction of the acidic pill forms a divalent metal (mainly Ca2+)–phosphonate complex to attach to the formation surfaces. Phosphonate return from the nanomaterial is governed by the dissolution of the Ca–DTPMP complex, instead of desorption of DTPMP from formation surfaces.19 The reason that SINM showed a longer squeeze lifetime compared with the acidic pill is that only a fraction of the DTPMP in the acidic pill formed complexes with divalent metals and the rest of the inhibitor was still in the form of free phosphonate. Finally, comparing the two SINM returns (SE #1 and #2), both exhibited a very similar pattern within the first 1000 PV. The extended squeeze lifetime beyond 1000 PV for SE #1 might be attributed to a higher KCl content and a slightly lower PPCA content in SE #1 nanofluid. This suggests that the squeeze lifetime and return behavior of such inhibitor nanomaterials might be tunable by adjusting the nanofluid composition.
 |
| | Fig. 4 DTPMP solid phase distribution in Frio sandstone medium immediately after the overflush treatment. | |
Table 5 Summary of physiochemical conditions of each squeeze simulation test
| Squeeze Experiment (SE) # |
Form of inhibitor added |
pH of inhibitor solutionc |
Formation medium |
Synthetic brine composition |
Synthetic brine pH |
Squeeze temperature (°C) |
Flow rate (mL h−1) |
Pore velocity (cm min−1) |
PV (mL) |
| SE #1 utilized scale inhibitor nanomaterial Sample #6 list in Table 1. Sample 6 contains 2% KCl. SE #2 utilized scale inhibitor nanomaterial Sample #7 list in Table 1. Sample 7 contains 1% KCl. pH values shown in this column are the pH of either the aqueous inhibitor pill solution or the scale inhibitor nanofluid. |
| SE-1a |
SINM (Sample #6) |
7.2 |
Frio sandstone for all cases |
0.025 M CaCl2, 0.015 M NaHCO3 1 M NaCl and bubbled with 100% CO2 |
5.54 for all cases |
70 °C for all cases |
60 mL h−1 for all cases |
2.0 cm min−1 for all cases |
8 mL for all cases |
| SE-2b |
SINM (Sample #7) |
7.2 |
| SE-3 |
Acid pill |
1.5 |
| SE-4 |
Neutralized pill |
6.7 |
 |
| | Fig. 5 Laboratory squeeze simulation results using scale inhibitor nanomaterials in Frio sandstone medium. Four DTPMP return profiles were included: SE #1 using inhibitor nanofluid with 2% KCl; SE #2 using inhibitor nanofluid with 1% KCl; SE #3 using a conventional acidic pill; SE #4 using a conventional neutralized pill. Figure (a) is the full return profile and Figure (b) is a part (up to 100 PV) of the main return profile. | |
Table 6 Summary of the results of squeeze simulation experiments
| Squeeze Experiment (SE) |
Form of inhibitor added |
DTPMP injected (mg)c |
Total volume returned (PV)d |
V10ppm (PV)e |
V1ppm (PV)e |
V0.5ppm (PV)e |
Initial pIP |
Final pIP |
NSL (bbl kg−1) |
| SE #1 utilized scale inhibitor nanomaterial Sample #6 list in Table 1. Sample 6 contains 2% KCl. SE #2 utilized scale inhibitor nanomaterial Sample #7 list in Table 1. Sample 7 contains 1% KCl. “DTPMP Injected” stands for the amount of DTPMP (in unit of mg) injected prior to the squeeze simulation test. “Total Volume Returned” corresponds to the total volume of synthetic brine squeezed during the SE test, measured by the number of PV. V10ppm, V1ppm and V0.5ppm correspond to the return volumes when the return concentrations drop below 10 ppm, 1 ppm and 0.5 ppm, respectively. |
| SE-1a |
SINM (Sample #6) |
32 |
1440 |
130 |
540 |
670 |
50.2 |
54.1 |
2270 |
| SE-2b |
SINM (Sample #7) |
32 |
1080 |
100 |
540 |
700 |
50.1 |
53.9 |
1700 |
| SE-3 |
Acid pill |
32 |
870 |
60 |
240 |
450 |
50.4 |
53.9 |
1370 |
| SE-4 |
Neutralized pill |
32 |
230 |
10 |
50 |
95 |
50.1 |
53.9 |
360 |
Ca–DTPMP acid/base complexation and NSL. Additionally, the inhibitor return profile can be evaluated from the standpoint of acid–base and calcium complex solution chemistry of DTPMP. A speciation model has been proposed to calculate the acid–base and calcium–DTPMP complex stability constants via a polymer type model.35 The negative logarithm of the ion activity product (pIP) of the return data can be calculated and compared against the proposed stability constants. The pIP of the Ca–DTPMP complex can be calculated based on the formula of Ca3H4DTPMP, and is in the form of the following:| | |
pIP = −log10[(Ca2+)3{H+}4(DTPMP10−)]
| (5) |
where parentheses account for molar concentration and braces for activity. The concentrations of free Ca2+ and DTPMP10− species were calculated based upon the speciation model.35 According to Table 6 and Fig. 6, the calculated pIP values for the four SE increased from initially 50 to a final value of ca. 54. The previous speciation study showed that the pIP of the amorphous Ca–DTPMP complex was determined to be 50 and a developed crystalline complex was 54.35 Thus, it can be argued that the initially formed Ca–DTPMP complexes in both nanofluid and the pill solutions were in an amorphous phase and during the course of SE, these amorphous materials gradually developed into a crystalline solid. Compared with the amorphous solid, crystalline Ca–DTPMP has a much lower solubility, which is responsible for the low return concentrations in the later SE.
 |
| | Fig. 6 The calculated negative logarithm of ion activity product of the four return profiles. The smaller inserted figure at the lower right corner is a part (up to 100 PV) of the main figure. The dashed line represents pIP of 54, which is close to the calculated final pIP. | |
The potential field squeeze application of the prepared inhibitor nanomaterial is depicted in Fig. 7. As elaborated in the above discussions, a volume of inhibitor nanofluid can be injected through the production well into deep formation and after the shut-in period, these delivered nanomaterials can flow back with the produced water to control scale deposition. Such a process can be assessed by calculating the “normalized squeeze lifetime” (NSL) for the four different inhibitor materials considered. As defined previously, NSL examines the squeeze lifetime on the same inhibitor mass basis in terms of barrels of brine treated per kilogram of inhibitor added.36 As for NSL for SE #1, the total return PV is 1440 and each PV is 8 mL (Tables 5 and 6), yielding a total synthetic brine return volume of 11.5 L. Recognizing that the mass of DTPMP injected was 32 mg (Table 6), NSL for SE #1 can be calculated as follows:
 |
| | Fig. 7 Schematic of the field scale squeeze operation employing inhibitor nanomaterial. (a) The process of injecting nanomaterial into formation; (b) the process of returning inhibitor after the shut-in period. | |
In other words, each batch of the inhibitor nanomaterial containing 1 kg of DTPMP is expected to treat produced water with a volume of ca. 2270 bbl. Similarly, NSL for the other three SE can be calculated as 1700, 1370 and 360 bbl kg−1, respectively (Table 6). It further reinforces the above conclusion that SINM outperformed the conventional inhibitor pills in terms of extending the squeeze lifetime. This is mainly due to the slow dissolution of the Ca–DTPMP complex in the nanofluid during synthetic brine flushing. It should be noted that based on the previous investigations,10,13,37,38 the actual inhibitor squeeze lifetime observed in the field is influenced by many factors, including production rate, reservoir geology, formation mineralogy, produced water chemistry, etc. Thus, the actual field squeeze lifetime can be different from the laboratory-determined lifetime. Additionally, the experimentally obtained return volume and corresponding NSL in this study using Frio sandstone can be different under other experimental conditions using a different type of formation material. Thus, the squeeze simulation test should be conducted under representative experimental conditions, using formation material obtained from the reservoir formation of interest.
Conclusions
In the present study, PPCA modified Ca–DTPMP SINM was experimentally evaluated for its transport and inhibitor return performance in formation media. Electrolyte content and the sonication method were investigated for their roles in nanomaterial transport and return performance. An increase in the electrolyte content can lead to a reduced breakthrough level and also an elevation in the tendency of SINM to be removed by calcite medium. This is believed to be due to the electrolyte impact on suppressing the nanomaterial particle diffuse double layer. The removal of SINM particles by formation medium can be characterized by the calculated attachment efficiency. In addition, a reduced breakthrough level at higher electrolyte content can result in nanomaterial accumulation in the first half of the column, instead of a more uniform distribution. A probe sonicator is more robust in breaking SINM into smaller particles, leading to an enhanced transportability in calcite medium. Laboratory squeeze simulation studies suggest that compared with the conventional pills, inhibitor nanomaterials show an improved inhibitor return performance with an extended squeeze lifetime. It seems that the nanofluid composition can impact the inhibitor return performance. During the course of the squeeze tests, the amorphous Ca–DTPMP complex in the nanofluid developed into a crystalline phase with a much lower solubility, accounting for an extended squeeze lifetime. Furthermore, the potential field application of such nanomaterial was assessed by calculating the normalized squeeze lifetime. It shows that this material has the potential to be applied in the field to outperform the conventional pills for oilfield scale control.
Acknowledgements
The authors would like to acknowledge the financial support by Brine Chemistry Consortium including Baker Hughes, BWA, CARBO, Cenovus, Chevron, ConocoPhillips, Dow, EOG Resources, GE, Hess, Halliburton, Italmatch, Kemira, Kinder Morgan, Lubrizol, Marathon Oil, NALCO Champion, Occidental, Petrobras, RSI, Saudi Aramco, Schlumberger, Shell, SNF, Statoil and Total. This work was supported by the NSF Nanosystems Engineering Research Center for Nanotechnology-Enabled Water Treatment (ERC-1449500).
Notes and references
- J. C. Cowan and D. J. Weintritt, Water-Formed Scale Deposits, Gulf Publishing Co., Houston, TX, 1976 Search PubMed.
- Mineral Scales and Deposits: Scientific and Technological Approaches, ed. Z. Amjad and K. Demadis, Elsevier Publishing, 2015 Search PubMed.
- J. Fink, Petroleum Engineer's Guide to Oil Field Chemicals and Fluids, Waltham, MA, 2012 Search PubMed.
- A. T. Kan and M. B. Tomson, Scale Prediction for Oil and Gas Production, SPE J., 2012, 17, 362–378 CrossRef CAS.
- M. A. Kelland, Production Chemicals for the Oil and Gas Industry, CRC Press, Boca Raton, FL, 2nd edn, 2014 Search PubMed.
- W. W. Frenier and M. Ziauddin, Formation, Removal, and Inhibition of Inorganic Scale in the Oilfield Environment, Richardson, TX, USA, 2008 Search PubMed.
- J. B. Becker, Corrosion and Scale Handbook, PennWell, Oklahoma, 1999 Search PubMed.
- M. M. Jordan, E. J. Mackay and O. Vazquez, The Influence Of Overflush Fluid Type On Scale Squeeze Life Time - Field Examples And Placement Simulation Evaluation, Presented in 2008 NACE International, New Orleans, Louisiana Search PubMed.
- P. Zhang, K. Allan and H. Bourne, Selection of Calcium Carbonate Scale Critical Values for Deepwater Production, SPE 173747, in SPE International Symposium on Oilfield Chemistry, The Woodlands, Texas, USA Search PubMed.
- M. M. Jordan, K. S. Sorbie, P. Griffin, S. Hennessey, K. E. Hourston and P. Waterhouse, Scale inhibitor adsorption/desorption vs. precipitation: the potential for extending squeeze life while minimizing formation damage, in SPE European Formation Damage Conference, The Hague, Netherlands Search PubMed.
- A. T. Kan, G. Fu and M. B. Tomson, Adsorption and precipitation of an aminoalkylphosphonate onto calcite, J. Colloid Interface Sci., 2005, 281, 275–284 CrossRef CAS PubMed.
- M. M. Jordan, K. S. Sorbie, G. M. Graham, K. Taylor, K. E. Hourston and S. Hennessey, The correct selection and application methods for adsorption and precipitation scale inhibitors for squeeze treatments in North Sea oilfields, in 1996 SPE Formation Damage Control Symposium, Lafayette, Louisiana Search PubMed.
- M. M. Jordan, K. S. Sorbie, P. Chen, P. Armitage, P. Hammond and K. Taylor, The design of polymer and phosphonate scale inhibitor precipitation treatments and the Importance of precipitate solubility in extending squeeze lifetime, in 1997 SPE International Symposium on Oilfield Chemistry, Houston, Texas Search PubMed.
- M. B. Tomson, G. Fu, M. A. Watson and A. T. Kan, Mechanisms of mineral scale inhibition, SPE J., 2002, 18, 192–199 Search PubMed.
- M. B. Tomson, A. T. Kan and G. Fu, Control of inhibitor squeeze via mechanistic understanding of inhibitor chemistry, SPE J., 2006, 11, 283–293 CrossRef CAS.
- Z. Kiaei and A. Haghtalab, Experimental study of using Ca-DTPMP nanoparticles in inhibition of CaCO3 scaling in a bulk water process, Desalination, 2014, 338, 84–92 CrossRef CAS.
- B. P. H. Do, B. Dung Nguyen, H. Duy Nguyen and P. T. Nguyen, Synthesis of magnetic composite nanoparticles enveloped in copolymers specified for scale inhibition application, Adv. Nat. Sci.: Nanosci. Nanotechnol., 2013, 4, 045016 Search PubMed.
- J. K. Y. Teck, R. H. Binti Abu and S. U. Binti Masuri, Numerical Study of Adsorption Enhancement by Nanoparticles Scale Inhibitor, Adv. Mater. Res., 2015, 1119, 43–48 CrossRef.
- P. Zhang, A. T. Kan and M. B. Tomson, Enhanced transport of novel crystalline calcium phosphonate scale inhibitor nanomaterials and their long term flow back performance in laboratory squeeze simulation tests, RSC Adv., 2016, 6, 5259–5269 RSC.
- P. Zhang, D. Shen, A. T. Kan and M. B. Tomson, Synthesis and laboratory testing of novel calcium-phosphonate reverse micelle nanofluid for oilfield mineral scale control, RSC Adv., 2016, 6, 39883–39895 RSC.
- P. Zhang, G. Ruan, A. T. Kan and M. B. Tomson, Functional scale inhibitor nanoparticle capsule delivery vehicles for oilfield mineral scale control, RSC Adv., 2016, 6, 43016–43027 RSC.
- R. J. Hunter, Foundations of colloid science, Oxford University Press, New York, 2001 Search PubMed.
- E. Rodier and J. A. Dodds, An experimental study of the transport and capture of colloids in porous media by a chromatographic technique, Colloids Surf., A, 1993, 73, 77–87 CrossRef CAS.
- C. Shani, N. Weisbrod and A. Yakirevich, Colloid transport through saturated sand columns: Influence of physical and chemical surface properties on deposition, Colloids Surf., A, 2008, 316, 142–150 CrossRef CAS.
- F. He, M. Zhang, T. Qian and D. Zhao, Transport of carboxymethyl cellulose stabilized iron nanoparticles in porous media: Column experiments and modeling, J. Colloid Interface Sci., 2009, 334, 96–102 CrossRef CAS PubMed.
- P. Zhang, D. Shen, G. Ruan, A. T. Kan and M. B. Tomson, Phosphino-polycarboxylic acid modified inhibitor nanomaterials for oilfield scale control: Synthesis, characterization and migration, submitted in May 2016 Search PubMed.
- J. A. Xiao, A. T. Kan and M. B. Tomson, Acid-Base and Metal Complexation Chemistry of Phosphino-polycarboxylic Acid under High Ionic Strength and High Temperature, Langmuir, 2001, 17, 4661–4667 CrossRef CAS.
- R. J. Charbeneau, Groundwater Hydraulics and Pollutant Transport, Waveland Press, Long Grove, IL, 1st edn, 2006 Search PubMed.
- M. M. Clark, Transport Modeling for Environmental Engineers and Scientists, John Wiley & Sons, Hoboken, NJ, 2nd edn, 2009 Search PubMed.
- J. N. Ryan and M. Elimelech, Colloid mobilization and transport in groundwater, Colloids Surf., A, 1996, 107, 1–56 CrossRef CAS.
- L. W. de Jonge, C. Kjaergaard and P. Moldrup, Colloids and colloid-facilitated transport of contaminants in soils: An introduction, Vadose Zone J., 2004, 3, 321–3258 CAS.
- P. B. Bedient, H. S. Rifai and C. J. Newell, Ground Water Contamination: Transport and Remediation, Prentice Hall, 2nd edn, 1999 Search PubMed.
- W. Stumm and J. J. Morgan, Aquatic Chemistry, Wiley-Interscience, 3rd edn, 1996 Search PubMed.
- J. S. Taurozzi, V. A. Hackley and M. R. Wiesner, Ultrasonic dispersion of nanoparticles for environmental, health and safety assessment – issues and recommendations, Nanotoxicology, 2011, 5, 711–729 CrossRef CAS PubMed.
- M. B. Tomson, A. T. Kan and J. E. Oddo, Acid/Base and Metal Complex Solution Chemistry of the Polyphosphonate DTPMP versus Temperature and Ionic Strength, Langmuir, 1994, 10, 1442–1449 CrossRef CAS.
- A. T. Kan, G. Fu, D. Shen, H. Al-Saiari and M. B. Tomson, Enhanced Scale-Inhibitor Treatments With the Addition of Zinc, SPE J., 2009, 14, 617–626 CrossRef CAS.
- E. J. Mackay, M. M. Jordan and F. Torabi, Predicting brine mixing deep within the reservoir, and the impact on scale control in marginal and deepwater developments, in 2002 SPE International Symposium and Exhibition on Formation Damage Control, Lafayette, Louisiana Search PubMed.
- K. S. Sorbie, E. M. Wesselingh, M. D. Yuan, R. Z. Lemanczyk and A. C. Todd, Scale inhibitor squeeze strategies in horizontal wells, J. Can. Pet. Technol., 1997, 36, 27–35 Search PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra09973d |
| ‡ Presently with Baker Hughes, Inc. |
|
| This journal is © The Royal Society of Chemistry 2016 |
Click here to see how this site uses Cookies. View our privacy policy here.