Three-dimensional nanoarchitecture SnSbZn–C composite nanofibers as anode materials for lithium-ion batteries†
Linping Liu,
Peixin Zhang*,
Yongliang Li*,
Xiangzhong Ren and
Libo Deng
College of Chemistry and Environmental Engineering, Shenzhen University, Shenzhen, Guangdong 518060, PR China. E-mail: pxzhang@szu.edu.cn; liyli@szu.edu.cn; Fax: +86-755-26733136; Fax: +86-755-26536627; Tel: +86-755-26733136 Tel: +86-755-26536627
First published on 25th May 2016
Abstract
SnSbZn–carbon (SnSbZn–C)-based hybrid composite nanofibers are synthesized by electrospinning. The zero-dimensional alloy nanoparticles, SnSb and SbZn, are enclosed by one-dimensional carbon nanofibers, therefore they exhibit improved electrochemical performance as anode materials for lithium-ion batteries. For the 200th cycle, the discharge capacity remains at 663 mA h g−1 and the capacity retention is 84%, the high cycling stability can be attributed to the unique one-dimensional nanofiber structure which can accommodate the volume expansion generated during cycling, and prevent the particles from aggregating.
1. Introduction
Lithium-ion batteries (LIBs) are one of the most widely used rechargeable power sources for portable electronic products due to their high energy densities, fast rate capabilities and safety. They are also promising choices for large-scale applications such as electric vehicles and energy storage for utility grids.1 The search for anodic materials to replace graphite is being examined as a substantial issue in improving the energy density of the battery. Many possible alternatives, such as Si, Sn, Sb, Ni, Mg, Al and Zn, and their alloys have attracted much attention due to their ability to reversibly react with lithium.2–6 However, the large volume change upon charge/discharge processes is a serious limitation of anode materials, which leads to mechanical disintegration of the electrodes and rapid capacity fading.In order to reduce the significant capacity fading, many anodes have been employed, such as SnSb–M (M = Ag, Co, Cu, Ti, Ni) alloy, in which various active elements will react with lithium at different stages in the charge and discharge cycles.7–11 Further efforts have been focused on nanocomposites formed with carbonaceous materials, such as graphite,12 carbon nanotube (CNT),13 carbon microbeads14 and amorphous carbon15 because carbon materials prevent the agglomeration of nanomaterials and buffer the volume change during cycling.
Zn displays a low operating voltage (about 0.2 V) which could play an important role in supporting lithium insertion/extraction to keep electric contact between the current collector and electrode materials. Moreover, Zn is an abundant and inexpensive element, which can be widely used as anode material in LIBs. Additional, it was proved that SbZn alloy as anode material having excellent electrochemical properties.16–18 In order to design nanostructured electrodes, electrospinning as a simple technique for fabricating one-dimensional (1D) nanostructures has been widely used in fabrication of anode and cathode materials for LIBs.19–22 In this study, Zn was introduced to the binary SnSb materials to form SnSb and SbZn alloys, which were encapsulated in carbon nanofiber network by electrospinning and heat-treatment. It was demonstrated that SnSbZn–C composites displayed improved electrochemical performance compared with the pristine SnSb materials.
2. Experimental section
2.1 Chemicals
Polyvinylpyrrolidone (PVP, average Mw = 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00
00![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000, Aladdin), stannic chloride (SnCl2·H2O, 98%, Aladdin), antimony trichloride (SbCl3, 99%, Aladdin), zinc nitrate hexahydrate (Zn(NO3)2·6H2O, 99%, Aladdin), N,N-dimethylformamide (DMF, 99.5%, Aladdin), ethanol (99.7%).
000, Aladdin), stannic chloride (SnCl2·H2O, 98%, Aladdin), antimony trichloride (SbCl3, 99%, Aladdin), zinc nitrate hexahydrate (Zn(NO3)2·6H2O, 99%, Aladdin), N,N-dimethylformamide (DMF, 99.5%, Aladdin), ethanol (99.7%).
2.2 Synthesis of anode materials
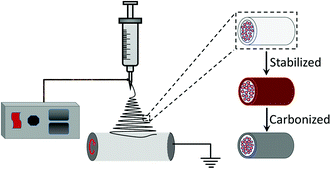
The SnSbZn–C composite nanofibers were prepared by electrospinning process (Scheme 1). The starting materials, SnCl2·H2O, SbCl3 and Zn(NO3)2·6H2O with the molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) x (x = 0, 0.2, 0.4, 0.6, 0.8) were completely dissolved in dimethylformamide (DMF) solution (12 wt%). A polymer solution with a concentration of 13 wt% was prepared by dissolving of polyvinylpyrrolidone (PVP) in ethanol. Both solutions were vigorously stirred for at least 12 h, and then the SnSbZn solution was added slowly to the PVP solution, which was stirred vigorously for another 12 h to obtain the precursor for electrospinning. The prepared solution was loaded into a plastic syringe equipped at a flow rate of 1 mL h−1 with a 21-gaguge stainless steel nozzle. The electrospun nanofibers were collected on the aluminum foil with a needle-to-collector distance of 15 cm. The applied voltage between the collector and the needle was 15 kV. The electrospun nanofibers were stabilized at 280 °C for 6 h with a heating rate of 5 °C min−1 in air atmosphere. Subsequently, the material was carbonized in N2/H2 (95
x (x = 0, 0.2, 0.4, 0.6, 0.8) were completely dissolved in dimethylformamide (DMF) solution (12 wt%). A polymer solution with a concentration of 13 wt% was prepared by dissolving of polyvinylpyrrolidone (PVP) in ethanol. Both solutions were vigorously stirred for at least 12 h, and then the SnSbZn solution was added slowly to the PVP solution, which was stirred vigorously for another 12 h to obtain the precursor for electrospinning. The prepared solution was loaded into a plastic syringe equipped at a flow rate of 1 mL h−1 with a 21-gaguge stainless steel nozzle. The electrospun nanofibers were collected on the aluminum foil with a needle-to-collector distance of 15 cm. The applied voltage between the collector and the needle was 15 kV. The electrospun nanofibers were stabilized at 280 °C for 6 h with a heating rate of 5 °C min−1 in air atmosphere. Subsequently, the material was carbonized in N2/H2 (95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v) at 600 °C with a heating rate of 5 °C min−1 and cooled to obtain the SnSbZn nanoparticles embedded in carbon nanofibers. SnSb alloy nanoparticles were synthesized as reported.16
5, v/v) at 600 °C with a heating rate of 5 °C min−1 and cooled to obtain the SnSbZn nanoparticles embedded in carbon nanofibers. SnSb alloy nanoparticles were synthesized as reported.16
2.3 Physical characterizations
X-ray diffraction (XRD) (Bruker D8 Advance) with Cu Kα radiation operated at 40 kV and 40 mA was employed to identify the crystal structure (2θ = 20–80°). Scanning electron microscope (SEM) (Hitachi, S-3400N) and high resolution transmission electron microscope (HRTEM) (Tecnai G2 F30) were used to observe the morphologies and structures. Power X-ray photoelectron spectroscopy (XPS) (ESCALAB 250Xi) was performed to analyze the chemical valence states. Analysis of data was performed using the XPSPEAK 4.1 program. All data were calibrated using adventitious C1s peak with a fixed value of 284.4 eV. Thermogravimetric analysis (TGA) (STA409PC) measurement was performed from room temperature to 1000 °C with the heating rate of 10 °C min−1 in air atmosphere.2.4 Electrochemical characterizations
Electrodes were prepared by active materials, acetylene black and carboxyl methyl cellulose (CMC) and styrene butadiene rubber (SBR) with a weight ratio of 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5
5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 to form a slurry. The slurry was pasted on copper foil and dried at 120 °C for 10 h under vacuum. The areal loading of the active materials was about 1.2 mg cm−2. The coin cells were assembled in an glove box (MBRAUN, UNILab2000), lithium metal foil was used as counter electrode, Celgard 2400 as separator, 1 M LiPF6 in ethylene carbonate (EC)–dimethyl carbonate (DMC) (1
10 to form a slurry. The slurry was pasted on copper foil and dried at 120 °C for 10 h under vacuum. The areal loading of the active materials was about 1.2 mg cm−2. The coin cells were assembled in an glove box (MBRAUN, UNILab2000), lithium metal foil was used as counter electrode, Celgard 2400 as separator, 1 M LiPF6 in ethylene carbonate (EC)–dimethyl carbonate (DMC) (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v) as the electrolyte (about 60 μL), the synthesized composite material as working electrode. The cyclic performance and rate capability were assessed using a CT2001A battery tester from 0.01 to 2.0 V (vs. Li/Li+). Cyclic voltammetry (CV) from 0.01 to 2.0 V (vs. Li/Li+) at 0.1 mV s−1 and electrochemical impedance spectroscopy (EIS) in the frequency range 10−2 to 105 Hz and with an amplitude of 5 mV were performed using an electrochemical testing system (CHI660A). All electrochemical tests were measurement at room temperature.
1 v/v) as the electrolyte (about 60 μL), the synthesized composite material as working electrode. The cyclic performance and rate capability were assessed using a CT2001A battery tester from 0.01 to 2.0 V (vs. Li/Li+). Cyclic voltammetry (CV) from 0.01 to 2.0 V (vs. Li/Li+) at 0.1 mV s−1 and electrochemical impedance spectroscopy (EIS) in the frequency range 10−2 to 105 Hz and with an amplitude of 5 mV were performed using an electrochemical testing system (CHI660A). All electrochemical tests were measurement at room temperature.
3. Results and discussion
Fig. 1 shows the XRD patterns of the SnSbZn–C composite nanofibers. For the SnSb sample, all the peaks correspond to single β-SnSb phase (JCPDS #33-0118), which belongs to the R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m space group with a rhombohedral structure.19,23 There is no impurity peaks of Sn and Sb were founded. In contrast, when Zn was introduced to the SnSb system, all samples contained two intermetallic phases, the peaks of β-SnSb phase were still observed and some peaks corresponding to hexagonal SbZn phase appeared (JCPDS #18-0140; a = b = 4.304 Å, c = 11.07 Å).17,24
m space group with a rhombohedral structure.19,23 There is no impurity peaks of Sn and Sb were founded. In contrast, when Zn was introduced to the SnSb system, all samples contained two intermetallic phases, the peaks of β-SnSb phase were still observed and some peaks corresponding to hexagonal SbZn phase appeared (JCPDS #18-0140; a = b = 4.304 Å, c = 11.07 Å).17,24
 | ||
| Fig. 1 XRD patterns of (a) SnSb–C, (b) SnSbZn0.2–C, (c) SnSbZn0.4–C and (d) SnSbZn0.8–C composite nanofibers. | ||
However, followed with increased Zn amount, the diffraction peak intensities of SbZn alloys are stronger than that of SnSb alloys (Fig. 2). The data were collected in the 2theta range of 28.4°to 29.4° (indicated by the dotted square in Fig. 1), similar situation happened as reported.25 Therefore, dual alloys of SnSb and SbZn encapsulated in composite nanofibers by electrospinning and carbonization were obtained.
 | ||
| Fig. 2 XRD patterns of (a) SnSb–C, (b) SnSbZn0.2–C, (c) SnSbZn0.4–C and (d) SnSbZn0.8–C composite nanofibers in 2theta range of 28.4°to 29.4°. | ||
The encapsulation of SnSb and SbZn alloy nanoparticles was confirmed further by SEM and TEM characterizations (Fig. 3). As show in Fig. 3a–c, the samples produced from electrospinning and carbonization exhibit 1D fibrous morphologies with diameter of 170 ± 30 nm. Moreover, it was showed that alloy nanoparticles are indeed encapsulated in carbon nanofibers after heat-treatment. The structures are also illustrated in Fig. 3d and e. The TEM images further confirmed that the alloy particles were not agglomerated and predominantly confined inside the carbon nanofibers. The size distribution of embedded alloy nanoparticles is shown in Fig. 3h and the size was measured to be about 20 nm.
To further confirm the crystallographic structures of the alloy nanoparticles, the HRTEM images (Fig. 3f and g) clearly reveal that the lattice fringe is about 0.37 nm, which corresponds to the (100) plane of SbZn crystal. In addition, a pair of lattice fringes with d-spacing of about 0.31 and 0.22 nm correspond to (101) and (012) planes of SnSb, respectively.26,27 These are consistent with the XRD results. Furthermore, the TEM elemental mapping images confirmed the uniform distribution of Sn, Sb, Zn and C (Fig. 4). It was also demonstrated that the molar ratio in the product is about 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.08
1.08![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.42, which was close to the starting amounts of the Sn, Sb and Zn precursors (1
0.42, which was close to the starting amounts of the Sn, Sb and Zn precursors (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.4). Furthermore, the SEM elemental analysis of SnSb–C, SnSbZn0.2–C, SnSbZn0.4–C and SnSbZn0.8–C composite nanofibers has also showed that the molar ratios of Sn, Sb, and Zn in various samples were very close to the starting materials, respectively (Fig. S1 and Table S1†). The zero-dimensional (0D) nanoparticles were encapsulated in 1D nanofibers and formed a 3D network structure, which can provide a good structural buffering effect to accommodate the huge volume changes during the prolonged charge/discharge cycles. In addition, the conductive network can keep the structural integrity and substantially improving the transport pathways for both electrons and lithium ions.28,29
0.4). Furthermore, the SEM elemental analysis of SnSb–C, SnSbZn0.2–C, SnSbZn0.4–C and SnSbZn0.8–C composite nanofibers has also showed that the molar ratios of Sn, Sb, and Zn in various samples were very close to the starting materials, respectively (Fig. S1 and Table S1†). The zero-dimensional (0D) nanoparticles were encapsulated in 1D nanofibers and formed a 3D network structure, which can provide a good structural buffering effect to accommodate the huge volume changes during the prolonged charge/discharge cycles. In addition, the conductive network can keep the structural integrity and substantially improving the transport pathways for both electrons and lithium ions.28,29
Fig. 5a shows the CV curves for the first five cycles of the SnSbZn–C composite nanofibers. The observed redox peaks in the charge and discharge cycles were involved in three kinds of electrochemical reactions, which are in well agreement with those of previous report on SnSb19,27,30 and ZnSb.17,18,25,31,32 During the first cathodic scan, a broad reduction peak appeared at around 1.3 V, this corresponded to the formation of the solid electrolyte interphase (SEI) layer on the electrode surface and the decomposition reactions of the electrolyte.33 The first differential discharge profile showed a major reduction peak at around 0.75 V derived from Li3Sb formation when Sb phase reacts with Li+. The multistep alloying reactions between Sn and Li which result in the formation of LixSn were shown by the broad peaks at around 0.4 V. However, when the potential is from 0.4 to 0.08 V, the alloying reaction peaks between Zn and Li to form the LiZn phase were not obvious as previous reports.18,32 This should be attribute to its low content. The salient broad peak from 0.7 to 0.0 V can be attributed to an irreversible reaction between Li+ and defects or buckled layers of carbon nanofibers, because this peak disappeared in subsequent cycles. During the anodic scan, the LiZn dealloying process was from 0.2 to 0.58 V, these peaks were not obvious similar as previous report on ZnSb.31 The oxidation peaks in the range of 0.6–0.8 V in the first anodic cycle can be assigned to dealloying reaction of LixSn, while the oxidative peak at 1.1 V is due to the LiSb dealloying reaction. While during the second cycle, it was obvious that two reduction peaks at around 0.8 V were distinct from the first cycle, which can be attribute to the electrochemical reaction mechanism of the ZnSb is somewhat different from the first alloying reaction. The two peaks can be assigned to the lithium alloys with Sb and Li2SbZn. In order to verify the existence of Li2SbZn, the XRD patterns of the anode materials during the second cathodic scan at 0.8, 0.7 and 0.6 V were shown in Fig. S6.† Therefore, the reaction mechanism of the SnSbZn–C composite during the first and second cycles can be summarized as follows:18,30–32
 | ||
| Fig. 5 (a and b) CV curves and (c and d) discharge–charge profiles of (a and c) SnSbZn–C and (b and d) SnSb–C composite nanofibers. | ||
During the first cycle:
| 1st discharge: SnSb + SbZn → Li3Sb + Sn + Zn → Li3Sb + LiZn + Li4.4Sn |
| 1st charge: Li3Sb + LiZn + Li4.4Sn → Li3Sb + Sn + Zn → Li2SbZn + LixSnSb (0 < x < 1.6) → SnSb + Li2−ySbZn (0 < y < 2) |
During the second cycle:
| 2nd discharge: SnSb + Li2−ySbZn → Li2SbZn + LixSnSb → Li3Sb + Sn + Zn → Li3Sb + LiZn + Li4.4Sn |
| 2nd charge: Li3Sb + LiZn + Li4.4Sn → Li3Sb + Sn + Zn → Li2SbZn + LixSnSb (0 < x < 1.6) → SnSb + Li2−ySbZn (0 < y < 2) |
The formation of Li2SbZn is direct evidence of Zn alloying with Li in SnSbZn–C composite, and the unique structure of SbZn facilitates in relieving the mechanical strains on SnSb. Generally, the dissociation reaction of intermetallic XaYb (X, Y = Sn, Sb, Zn, P; a, b = 1, 2) compounds into the Li–X and Li–Y phases during discharge and recombination reaction of XaYb nanocrystallites during charge showed advantages in terms of the electrochemical performance of Li alloy-based anodes. Fig. 5c shows the voltage profiles of the SnSbZn–C composite electrode in the voltage range of 0.01–2.00 V at a current density of 200 mA g−1. The discharge–charge plateaus are in good agreement with the CV. The initial discharge and charge capacities were 1288 and 813 mA h g−1, respectively, corresponding to an initial coulombic efficiency of 63%, which quickly improves after a few cycles. Although the composite electrode shows a poor initial coulombic efficiency, it shows high charge capacity, which is two times larger compared with commercial graphite anode (372 mA h g−1). The irreversible capacity is mainly related to the electrolyte decomposition and the formation of SEI layers on the carbon surface.28,29,34 However, because of the volume change of the alloy particles during cycling, the SEI layers formation on alloy is ruptured and reorganization of the dynamic process.11,13,34 During the initial several cycles, the more stable SEI layers gradually formed. Hence, a rapid discharge capacity loss was observed during this time; but the stability of SEI layers is beneficial for cycling performance. In the second cycle, the discharge and charge capacities of 795 and 786 mA h g−1 were obtained with a coulombic efficiency of 98.8%. And it was also exhibited a quite stable capacity of approximately 663 mA h g−1 over 200 cycles.
The cycling performances of the SnSbZn0.4–C, SnSb–C and SnSb samples at the current density of 0.2C are shown in Fig. 6a. Compare with SnSb sample, it is obvious that the cycling performance of SnSb–C sample is improved significantly. For the 200th cycle, the discharge capacities of SnSbZn0.4–C and SnSb–C are 663 and 550 mA h g−1, respectively, and their corresponding capacity retentions were 84% and 76%, as calculated from the second cycle. The highly stable cyclability can be attributed to the geometric configuration of alloy nanoparticles encapsulated in the carbon nanofiber, and the unique structure to accommodate the volume expansion generated during cycling, and prevent the particles from aggregating (Fig. S2†).19,29,35 As shown in Fig. 6b, compare with SnSb–C sample, the results showed that the Zn also plays an important role in improving the electrochemical properties of the SnSnZnx–C composite.16 On one hand, the addition of Zn can effectively alleviate the influence of volume changes by acting as a buffer matrix. On the other hand, the unique structure of SbZn facilitates in relieving the mechanical strains on SnSb. However, compared with the SnSb, the theoretical capacity of Zn is relatively lower, therefore, introducing appropriate amount of Zn will benefit for the capacity, and otherwise the capacity will decrease.
The rate performances of the SnSb–C and SnSbZn0.4–C composite was also tested, as shown in Fig. 6c. It can be observed that the current density increased progressively from 0.1 to 6C, with 10 cycles being performed at each step. The specific discharge capacities of SnSbZn0.4–C are respectively 843, 742, 629, 570, 487, 408 and 312 mA h g−1 at 0.1, 0.2, 0.5, 1, 2, 3, and 6C. Following 70 cycles, the discharge capacity of the composite recovered well to 710 mA h g−1 as the current density returned to 0.2C. The good rate capability of the SnSbZn0.4–C composite is due to the increased lithium ion diffusion rate into the SnSb and SbZn nanocrystallites and the combined effects of homogeneous distribution of SnSb and SbZn nanoparticles inside the carbon nanofiber.19,29,36
To investigate the interface reactions, the electrochemical impedance behaviors of SnSb–C and SnSbZn–C were measured at first and 100th cycles as shown in Fig. 7. The impedance spectra exhibited a depressed semicircle at high frequency and an inclined line at lower frequency, which were fitted by the equivalent circuit in the insets of Fig. 7. In the equivalent circuit, Re is the resistance of the electrolyte and Rct is the resistance of the surface film and the charge transfer, CPE represents the constant phase angle elements, Ws refers to Warburg impedance which relates to the Li+ bulk diffusion resistance.37 The fitting values of Re and Rct were summarized in Table 1. Compared with the impedance spectra of SnSb–C and SnSbZn–C before cycling (Fig. 7a), the value of Re increased from 5.65 to 7.14 Ω, showing little change, but there is a huge reduction of Rct from 170.8 to 125.1 Ω. The reduction of Rct can be attributed to the more numerous electrochemical active sites and the large contact area with the electrolyte for charge transfer reactions.38 While compared with the impedance spectra of SnSbZn–C before and after 100 cycles (Fig. 7b), the values of Re and Rct increased from 7.14 and 125.1 Ω to 16.33 and 180.7 Ω, respectively. There is a small change which can be attributed to the stable SEI formation throughout cycling.31 Therefore the excellent electrochemical performance of SnSbZn–C sample is related both to the addition of the hybrid composite and the construction of a stable SEI film, which increase the active specific surface area and buffer the volume expansion.
 | ||
| Fig. 7 EIS of (a) SnSb–C and SnSbZn–C electrodes before cycling, (b) SnSbZn–C after 100th cycles. Inserts of (a) and (b) are the equivalent circuits. | ||
| SnSb–C | SnSbZn–C | ||
|---|---|---|---|
| 1st | 1st | 100th | |
| Re (Ω) | 5.65 | 7.14 | 16.33 |
| Rct (Ω) | 170.80 | 125.10 | 180.70 |
4. Conclusions
The SnSbZn–C composite nanofibers were successfully synthesized by electrospinning and carbonization. SnSb and SbZn nanoparticles were encapsulated in nanofibers and formed a network structure, which displayed remarkable performance through stable cyclability and high storage of lithium. For the 200th cycle, the discharge capacity remained at 663 mA h g−1 and the capacity retention was 84%, the highly cycling stability could be attributed to the unique structure to accommodate the volume expansion generated during cycling, and it also prevented the particles from aggregating. Furthermore, this unique structure is potential for a wide range of applications, especially for energy storage and conversion systems.Acknowledgements
We are grateful for the financial support provided by the National Natural Science Foundation of China (Grant #51374146, 50874074, 51502177), Natural Science Foundation of Guangdong (Grant #2014A030310323, 2015A030310087), Shenzhen Government's Plan of Science and Technology (Grant #JCYJ20120613173950029, JCYJ20140418095735619, JCYJ20140418182819158, JCYJ20150324141711596), Natural Science Foundation of SZU (Grant #201445, 827-000039).References
- N. Nitta and G. Yushin, Part. Part. Syst. Charact., 2014, 31, 317–336 CrossRef CAS.
- C. M. Park and K. J. Jeon, Chem. Commun., 2011, 47, 2122–2124 RSC.
- J.-T. Li, J. Światowska, V. Maurice, A. Seyeux, L. Huang, S.-G. Sun and P. Marcus, J. Phys. Chem. C, 2011, 115, 7012–7018 CAS.
- M. Li, Z. Yu and M. Qu, J. Alloys Compd., 2010, 491, 643–648 CrossRef CAS.
- J.-H. Choi, C.-W. Ha, H.-Y. Choi, H.-C. Shin and S.-M. Lee, Mater. Lett., 2015, 159, 349–352 CrossRef CAS.
- C.-M. Park and H.-J. Sohn, Chem. Mater., 2008, 20, 6319–6324 CrossRef CAS.
- H. Guo, H. Zhao, X. Jia, W. Qiu and F. Cui, Mater. Res. Bull., 2007, 42, 836–843 CrossRef CAS.
- Q. Jiang, D. Hu, M. Jia and R. Xue, Appl. Surf. Sci., 2014, 321, 109–115 CrossRef CAS.
- C. Marino, M. T. Sougrati, B. Gerke, R. Pöttgen, H. Huo, M. Ménétrier, C. P. Grey and L. Monconduit, Chem. Mater., 2012, 24, 4735–4743 CrossRef CAS.
- T. Tabuchi, N. Hochgatterer, Z. Ogumi and M. Winter, J. Power Sources, 2009, 188, 552–557 CrossRef CAS.
- P. Zhang, Y. Wang, J. Wang, D. Zhang, X. Ren and Q. Yuan, Electrochim. Acta, 2014, 137, 121–130 CrossRef CAS.
- J.-W. Jung, W.-H. Ryu, J. Shin, K. Park and I.-D. Kim, ACS Nano, 2015, 9, 6717–6727 CrossRef CAS PubMed.
- P. Zhang, Y. Wang, Y. Wang, X. Ren, K. Liu and S. Chen, J. Power Sources, 2013, 233, 166–173 CrossRef CAS.
- M. Xu, W. Li and B. L. Lucht, J. Power Sources, 2009, 193, 804–809 CrossRef CAS.
- X. Niu, H. Zhou, Z. Li, X. Shan and X. Xia, J. Alloys Compd., 2015, 620, 308–314 CrossRef CAS.
- Y. Li, W. Zhang, H. Cai, J. Wang, X. Ren and P. Zhang, RSC Adv., 2015, 5, 105643–105650 RSC.
- S. Saadat, J. Zhu, M. M. Shahjamali, S. Maleksaeedi, Y. Y. Tay, B. Y. Tay, H. H. Hng, J. Ma and Q. Yan, Chem. Commun., 2011, 47, 9849–9851 RSC.
- C. M. Park and H. J. Sohn, Adv. Mater., 2010, 22, 47–52 CrossRef CAS PubMed.
- K. Shiva, H. B. Rajendra and A. J. Bhattacharyya, ChemPlusChem, 2015, 80, 516–521 CrossRef CAS.
- D. Ma, P. Zhang, Y. Li and X. Ren, Sci. Rep., 2015, 5, 11257 CrossRef PubMed.
- J.-W. Jung, C.-L. Lee, S. Yu and I.-D. Kim, J. Mater. Chem. A, 2016, 4, 703–750 CAS.
- C. Chen, K. Fu, Y. Lu, J. Zhu, L. Xue, Y. Hu and X. Zhang, RSC Adv., 2015, 5, 30793–30800 RSC.
- L. Baggetto, H.-Y. Hah, J.-C. Jumas, C. E. Johnson, J. A. Johnson, J. K. Keum, C. A. Bridges and G. M. Veith, J. Power Sources, 2014, 267, 329–336 CrossRef CAS.
- C. Wang, Y. Xu, S. Yang, H. Jiang, J. Li, J. Zhu, S. Yang and X. Liu, J. Phase Equilib. Diffus., 2015, 36, 350–356 CrossRef.
- M. Zhong, X. Meng and J. Li, Appl. Surf. Sci., 2015, 332, 76–79 CrossRef CAS.
- J.-C. Kim and D.-W. Kim, Electrochem. Commun., 2014, 46, 124–127 CrossRef CAS.
- L. Xue, X. Xia, T. Tucker, K. Fu, S. Zhang, S. Li and X. Zhang, J. Mater. Chem. A, 2013, 1, 13807 CAS.
- X. Xia, X. Wang, H. Zhou, X. Niu, L. Xue, X. Zhang and Q. Wei, Electrochim. Acta, 2014, 121, 345–351 CrossRef CAS.
- D. Zhou, W. L. Song and L. Z. Fan, ACS Appl. Mater. Interfaces, 2015, 7, 21472–21478 CAS.
- M.-S. Park, S. A. Needham, G.-X. Wang, Y.-M. Kang, J.-S. Park, S.-X. Dou and H.-K. Liu, Chem. Mater., 2007, 19, 2406–2410 CrossRef CAS.
- M.-G. Park, C. K. Lee and C.-M. Park, RSC Adv., 2014, 4, 5830 RSC.
- Y. Hwa, J. H. Sung, B. Wang, C.-M. Park and H.-J. Sohn, J. Mater. Chem., 2012, 22, 12767 RSC.
- J.-G. Wang, Y. Yang, Z.-H. Huang and F. Kang, Electrochim. Acta, 2015, 170, 164–170 CrossRef CAS.
- T. H. Hwang, Y. M. Lee, B. S. Kong, J. S. Seo and J. W. Choi, Nano Lett., 2012, 12, 802–807 CrossRef CAS PubMed.
- Y. Yu, L. Gu, C. Wang, A. Dhanabalan, P. A. van Aken and J. Maier, Angew. Chem., 2009, 48, 6485–6489 CrossRef CAS PubMed.
- B.-O. Jang, S.-H. Park and W.-J. Lee, J. Alloys Compd., 2013, 574, 325–330 CrossRef CAS.
- Y. Hu, Q.-R. Yang, J. Ma, S.-L. Chou, M. Zhu and Y. Li, Electrochim. Acta, 2015, 186, 271–276 CrossRef CAS.
- S. Y. Kim and B.-H. Kim, Synth. Met., 2015, 210, 386–391 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra09661a |
| This journal is © The Royal Society of Chemistry 2016 |