NiO/Ag heterostructure: enhanced UV emission intensity, exchange interaction and photocatalytic activity†
Santanab Majumder,
Swarupananda Bhattacharjee and
Chandan Kr. Ghosh*
School of Materials Science and Nanotechnology, Jadavpur University, Jadavpur, Kolkata – 700032, India. E-mail: chandu_ju@yahoo.co.in
First published on 1st June 2016
Abstract
The effect of direct Ag coating on the optical and catalytic properties of NiO flake-like structures has been investigated in detail in this work. Ag nanoparticle decorated NiO flakes were synthesized using a facile two-step process. The phases and crystal structure were examined using an X-ray diffractometer, whereas the morphologies of the synthesized nanostructures were investigated using FESEM and HRTEM. Interfacial interaction of the synthesized heterostructures was examined using Fourier transformation infrared and Raman spectroscopy. Importantly the LO phonon and magnon scattering mode of NiO are significantly affected by the Ag coating. The band gap of NiO is found to be lowered (direct band gap: 3.52 to 2.78 eV and indirect band gap: 2.72 to 0.50 eV) due to the change in valence band energy originating from the charge transfer process between NiO and Ag. A UV emission that depends on the crystal field splitting of the Ni d orbitals is observed to possess a blue shift along with their enhancement of intensity. Variation of the magnon scattering mode also reveals that the enhancement of exchange interaction with NiO nanoflakes causes the blue shift of the UV emission. The nonradiative part corresponding to this UV emission which is parameterized using the Huang-Rhys factor gets enhanced due to the lowering of the electron–phonon interaction. The photocatalytic activity of the synthesized heterostructures was evaluated using the catalytic decomposition of the dye methylene blue in aqueous solution. The results show that the NiO/Ag heterostructures exhibit a much higher degradation rate than virgin NiO flakes that might be from improved separation of the photogenerated electrons and holes. In particular, the pseudo-first-order degradation rate constant of NiO/Ag is found to be 10 times greater than bare NiO.
Introduction
Nanostructures of transition metal oxides are some of the most nurtured oxides in recent times due to their exotic physical and chemical properties, completely different from bulk, and their application aspects in various optoelectronic applications.1–3 In this context, it may be stated that NiO being an antiferromagnetic p-type semiconducting material has also proven it’s potential use in the fields of catalysis,4 battery cathodes,5 gas sensors,6 fuel cell electrodes,7 photoelectrochemical water splitting,8 dye sensitized solar cell9 etc. due to its suitable electrical, optical and magnetic properties. Various morphologies of NiO have been synthesized previously by many researchers in order to tune their electronic structure and related properties. As an example, NiO nanofibers and nanorods are found to exhibit effective glucose sensitivity in alkaline medium due to their high surface to volume ratio.10,11 Recently, heterostructure formation using organic as well as inorganic materials has been found to be another effective method by which various surface related properties of nanostructures are able to be modified.12–19Like ZnO and TiO2, a few heterostructures have already been synthesized with NiO and their related properties have been explored. For example, the processes of photogeneration, separation and recombination of charge carriers have been investigated at the heterointerfaces of NiO/P3HT, NiO/CH3NH3PbI3, NiO/acetonitrile, NiO/water etc. and their effect on photoelectrochemical activity has been examined.20 The coaxial carbon nanofibers/NiO core–shell structure is found to be a potential anode material for lithium ion batteries.21 Pt–NiO nanoplate array/reduced graphene oxide nanocomposite exhibits non-enzymatic glucose sensing ability.22 The enhanced HCHO sensing properties of α-Fe2O3@NiO nanofibers have been investigated by Cao et al.23 Compared to ZnO, much less effort has been made to investigate noble metal–NiO heterostructures. In this regard, Yu Ding et al. synthesized NiO/Ag heterostructures for blood–glucose sensing.24 In this context it may be stated that Raman scattering, luminescence properties, and the catalytic activity of ZnO are significantly tuned by Ag, Au and Pt coatings.
Till now, no effort has been made to explore the optical and catalytic properties of NiO/Ag heterostructures. In this paper, we have investigated the effect of a NiO/Ag heterostructure on the optical and catalytic properties of a NiO flake. Here, the heterostructure is synthesized using a two-step process. The interfacial interaction is studied using Fourier transform infrared and Raman spectroscopy. The charge transfer process between NiO and Ag has been proposed from optical characterizations. To explore the effect of charge transfer on optoelectronic properties, the photocatalytic activities of the as prepared heterostructures have been tested using methylene blue. In summary, it may be mentioned that though there exist several methods to modify the optical and magnetic properties of NiO, here we have first illustrated that heterostructure formation with a noble metal may be another method to modify the optical properties of NiO, but magnetic exchange interaction may also be tuned.
Experimental
Synthesis of NiO/Ag heterostructure
Synthesis of the NiO nanoflakes
It is a typical synthesis procedure where 1.57 g of Ni(NO3)2·6H2O, 1.57 g of urea and 0.785 g of HMTA (the molar ratio is 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5) were added to 90 mL of DI water. The green mixture was stirred for 1 hour at room temperature to make it homogenous. Then the mixture was transferred into a 100 mL Schott bottle and was kept in a hot air oven, maintained at 90 °C. After 6 hours of reaction, a dense, light green precipitation appeared in the bottle which was filtered and washed repeatedly using distilled water and ethanol. Light green powder was obtained after drying at 40 °C for 4 hours. The green powder was calcined at 450 °C for 2 hours in a furnace (marked as NFO).
0.5) were added to 90 mL of DI water. The green mixture was stirred for 1 hour at room temperature to make it homogenous. Then the mixture was transferred into a 100 mL Schott bottle and was kept in a hot air oven, maintained at 90 °C. After 6 hours of reaction, a dense, light green precipitation appeared in the bottle which was filtered and washed repeatedly using distilled water and ethanol. Light green powder was obtained after drying at 40 °C for 4 hours. The green powder was calcined at 450 °C for 2 hours in a furnace (marked as NFO).
Formation of the hetero-structure
In the next step, the NFO (1.0 mmol) sample was used as a seed layer for the synthesis of silver nanoparticles where AgNO3 was reduced to metallic silver with the help of 5.8 mL of oleyamine in 80 mL of toluene.25 Two samples were prepared by fixing NiO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) AgNO3 with molar ratio 1
AgNO3 with molar ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (1.0 mmol, named as NAG1) and 1
1 (1.0 mmol, named as NAG1) and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (2.0 mmol, named as NAG2). The mixtures were taken into a 100 mL Schott bottle, and then with the help of an oil bath, the solution was heated up to 85 °C for 8 hours, under vigorous stirring. The samples were collected using centrifugation, followed by drying at room temperature. The NAG1 sample appeared grey and for the NAG2 the grey color became paler. The synthesized products were preserved for further characterizations.
2 (2.0 mmol, named as NAG2). The mixtures were taken into a 100 mL Schott bottle, and then with the help of an oil bath, the solution was heated up to 85 °C for 8 hours, under vigorous stirring. The samples were collected using centrifugation, followed by drying at room temperature. The NAG1 sample appeared grey and for the NAG2 the grey color became paler. The synthesized products were preserved for further characterizations.
Characterization
The crystalline phases of the synthesized samples were investigated using X-ray diffraction (XRD) using the CuKα emission (λ = 1.54 Å, Rigaku-Ultima-III). The morphology and elemental analysis of the samples were examined with FESEM (S – 4800, Hitachi) and EDX (JEOL JSM 6300, Oxford-7582) respectively. High resolution transmission electron microscopy (HRTEM, JEOL) was used to take a high magnified image, selected area electron diffraction (SAED) and lattice fringes. Attachment processes of the silver on NiO flakes were studied using Fourier transform infrared (FTIR, SHIMADZU IR Prestige 21) and Raman spectroscopy (Raman spectrometer alpha 300, Witec, laser excitation wavelength 530 nm). Optical properties of the synthesized samples were investigated using a JASCO V650 spectrophotometer whereas room-temperature photoluminescence (PL) spectra of the samples were recorded using a JASCO-S8200 spectrofluorometer.Photocatalytic study
Methylene Blue (MB, aqueous stain solution, Merck) was used as a representative dye pollutant to evaluate the photocatalytic activity of the as prepared samples. For the catalytic investigation, a stock solution was prepared by dissolving 52 μL of MB into 100 mL of DI water. 20 mL of the as prepared stock solution was taken into a glass beaker and 20 mg of NFO, NAG1 and NAG2 were added to the solutions. Before degradation in the presence of UV light, the mixture solution was kept under dark conditions for 1 hour without further agitation to achieve an adsorption–desorption equilibrium. To monitor the degradation process, the mixture solution was stirred at different time intervals viz. 30, 90, 150, 210 minutes under radiation. The decanted solution was collected via centrifugation. The degradation process was monitored using absorption spectroscopy, measured with the help of a JASCO V650 spectrophotometer within the range of 250–750 nm. The powdered samples were dried and were kept for phase analysis if there are any phase changes.Results and discussion
Crystal structure and microstructure of NiO/Ag heterostructure nanocrystals
The diffraction patterns, obtained from the X-ray diffractometer, of NFO, NAG1 and NAG2 are shown in Fig. 1. The peaks (marked by #), identified at 2θ values of 37.28, 43.30, 62.92, 75.44 and 79.39 correspond to the reflections from the (111), (200), (220), (311), and (222) crystal planes of cubic NiO [JCPDS data card no. 78-0643], and are observed for all synthesized samples.For NAG1 and NAG2 four additional peaks (marked by *), measured at 2θ values of 38.12, 44.28, 64.43 and 77.47 are attributed to the reflections from the (111), (200), (220) and (311) planes of the fcc Ag peaks [JCPDS data card 040783]. No peaks other than Ag and NiO are present in the diffraction patterns, indicating the purity of the synthesized materials. In addition, no significant shift in the diffraction patterns of NAG1 and NAG2 i.e. changes in the lattice parameter are noticed, indicating no formation of a AgxNi1−xO solid solution. The existence of peaks corresponding to both NiO and Ag confirms the coexistence of NiO and Ag within the synthesized nanohybrids.26 The average crystallite size of the NiO flakes, calculated from the well-known Scherrer’s relation (0.9λ/β![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos
cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ), where β represents the FWHM of the corresponding XRD peak, are found to be 25 nm. The crystallite sizes of the Ag nanoparticles were found to be 35 and 36 nm for NAG1 and NAG2 respectively indicating that the particle size is getting enhanced by the introduction of Ag.26–28 The detailed reaction mechanism has been discussed in the ESI.†
θ), where β represents the FWHM of the corresponding XRD peak, are found to be 25 nm. The crystallite sizes of the Ag nanoparticles were found to be 35 and 36 nm for NAG1 and NAG2 respectively indicating that the particle size is getting enhanced by the introduction of Ag.26–28 The detailed reaction mechanism has been discussed in the ESI.†
In order to obtain more insight into the microstructure and morphology of the as-prepared samples, FESEM and TEM were employed. As investigated using FESEM (shown in Fig. 2(a)), the morphology of NFO is found to be a well-defined 3D flake-like structure hierarchical in nature with good monodispersity between flakes. From the high magnification FESEM image of NFO (Fig. S1 in ESI†), it is clear that the widths and thicknesses of the flakes are ∼600 and 4.38 nm respectively. Selected areas for the elemental mapping of NAG1 and NAG2 using EDS are presented in Fig. S2(a) and (b) in the ESI† respectively and Fig. S2(c) and (d) in the ESI† show their oxygen, nickel and silver mappings. EDS results, obtained at various parts of the synthesized samples, indicate that the prepared samples are mainly composed of nickel, oxygen and silver.
 | ||
| Fig. 2 (a): FESEM image of NFO, (b): TEM image (c) lattice image and (d): SAED pattern of the NAG2 sample. | ||
It can be observed from the figures that the NiO/Ag systems remain in the 3D flake-like structure the same as NFO and varied amounts of the silver nanoparticles with diameters ∼32 nm are found on the surfaces of the NiO flakes. Ag percentage measured at different positions of the samples is not found to vary significantly i.e. the as prepared samples are found to be homogeneous. Ag concentration, measured using EDS, is found to be 3.83 and 9.34 (atomic%) for NAG1 and NAG2 respectively i.e. it may be concluded that with increasing percentage of Ag, deposition of Ag on the NiO flake increases. Careful analyses of the images predict that with increasing Ag content coarsening of the nanoflake surfaces increases. In addition, uniform distribution of Ag (blue dots in Fig. S2(c) and (d)†) on the EDS also supports the homogeneity of the synthesized samples.
The structure of the synthesized samples was investigated further using HRTEM. Fig. 2(b) shows the typical HRTEM image of the as prepared NAG2 sample. The image depicts the unique flake-like morphology of NiO and the homogeneous decoration of Ag nanoparticles on the NiO flakes. It may be also inferred that individual Ag nanoparticles are well adhered onto the surface of the flake. In addition, a few aggregations of Ag nanoparticles are also observed. One typical high magnification TEM image of an individual Ag on the NiO nanoflake is presented in Fig. 2(c), it illustrates a well defined lattice fringe having interplanar spacing (d) ∼ 2.39 Å corresponding to the (111) plane of fcc Ag.29,30 Therefore it may be concluded that the TEM images of NAG2 have full consistency with the obtained SEM results. The SAED pattern recorded at the interface of NiO and Ag corresponding to sample NAG2, is shown in Fig. 2(d) it illustrates a mixture of the NiO [220] and [200] zone diffraction spots and the Ag [111] and [311] zone diffraction spots. The mixed patterns originating separately from cubic NiO and fcc Ag are attributed to the Ag nanoparticles attached on the surface of the NiO flake-like structure in agreement with the XRD data. Similar types of SAED pattern were obtained previously by Warule et al. for ZnO/Ag heterostructures.31
As represented in the FTIR spectra (Fig. S3, ESI†), no characteristic peak corresponding to the Ag–O bond is observed, while the band, appearing at 569,32 has been observed for NFO and is assigned to the stretching vibration of the tetrahedral Ni–O cluster within an anisotropic environment.33,34 Presence of this peak in all samples suggests that there is no structural change within the NiO. But the stretching vibrational energy gets significantly reduced with increasing silver concentration i.e. there exists a strong coordination interaction between the NiO and Ag nanoparticles that favours adsorption of Ag on NiO.35 Reduction of the vibrational energy may be attributed to the charge transfer between them.36
Fig. 3 illustrates the Raman spectra of the synthesized heterostructures. As depicted in the figure, the Raman spectrum of NFO consists of three peaks located at 505, 1142 and 1538 cm−1 which are assigned to the longitudinal optical (LO) one-phonon (1P) mode, the longitudinal optical (2LO) two-phonon (2P) mode37–39 and the two-zone boundary magnons (2M) scattering mode.39,40 In this context, it may be stated that first order Raman scattering could not be observed as k = 0 phonons in the cubic phase transform as Γ4−.41 Thus the observed first order LO mode might be originating from the parity-breaking imperfections near the flake-surfaces.39,42 It is further noticed that the intensity of the LO peak increases in the presence of Ag.
This enhancement may be assigned to the enhanced parity-breaking of phonons due to the local electric field that acts on the flakes originating from the electrons of the Ag nanoparticles. Analysis of the LO mode illustrates that the Raman shift corresponding to the LO mode increases from NFO to NAG1 to NAG2 (shown in Fig. S4, ESI†) i.e. the strength of the LO mode gets enhanced. Such enhancement may be ascribed to the charge transfer from Ag to NiO (discussed later). Careful analysis of the magnon scattering reveals that the Raman shift for 2M scattering increases with respect to Ag decoration on the NiO flake-surfaces (shown in Fig. S4, ESI†). It was established earlier that the next-nearest-neighbour antiferromagnetic exchange (J) interaction (180°) within NiO possesses a much higher value than the nearest-neighbour (90°) ferromagnetic interaction and is responsible for type-II antiferromagnetism with spin S = 1.43 Raman shift (γ2M) for 2M scattering is related to exchange interaction by the following equation44,45
| γ2M = 4JZS − 2J |
Optical properties and electron–phonon interactions within the NiO/Ag heterostructure
Optical properties of the NiO/Ag heterostructure were examined using UV-Vis spectroscopy and photoluminescence (PL) spectroscopy taking identical amounts of the samples. As depicted in Fig. 4, absorption curves of the NiO/Ag nano-flake heterostructures are similar in nature to those of bare NiO nano-flake structures. The direct band gap (Eg) of the NiO/Ag heterostructure was calculated using the well-known equation| α = A(hν − Eg)1/2/hν (A(hν − Eg)2/hν, for indirect band gap), |
The luminescence properties of NiO were investigated previously by many researchers. In this context it may be stated that two bands (361 and 392 nm) in the UV region due to the near band edge emission, two bands (436 and 467 nm) in the visible region due to the surface oxygen vacancy and green emission (560 nm) due to the cation vacancy, interstitial oxygen trapping etc.51,57 were observed in NiO. As revealed by Qi et al., the luminescence spectrum of NiO nanostructures also consists of a single peak at 391 nm (3.17 eV) and one peak at 467 nm (2.66 eV).58 Activation energy and lifetime corresponding to these transitions were examined by Guerra et al.59 Interestingly, NiO nanowires exhibit a strong orange peak (660 nm) and relatively weak red peak (680 nm), though a UV emission peak corresponding to the near band edge emission has been found to be absent.60 In the present paper, two luminescence peaks, measured at 360 nm in the UV region and 429 nm in the visible region, have been observed for NiO (shown in Fig. 6(a)).
 | ||
| Fig. 6 Photoluminescence spectra of (a) NFO (b) NAG1 (c) NAG2 sample, (d) variation of the FWHM of the UV emission. | ||
The former one is assigned to the near band edge emission, whereas the later one is attributed to the surface oxygen vacancy.51,57 As presented in Fig. 6(b) and (c), the emission spectra of NAG1 and NAG2 also consist of two peaks in the UV and visible region respectively. Careful analysis of the spectra reveals that the UV emission peak shifts towards a lower wavelength i.e. blue shift in the UV emission after Ag decoration (plotted in Fig. S6, ESI†). In contrast to red shift of the band gap of NiO/Ag heterostructure, we have observed blue shift in this emission spectrum. Therefore, the present emission in contrast to the p–d charge transfer is referred to as the intersite d–d crystal field transitions within the Ni2+ ions.61–63 Charge transfer from Ag to oxygen (as revealed from a previous study) increases crystal field splitting due to electrostatic coulomb interaction between the O 2p-orbitals and Ni 3d-orbitals according to molecular orbital theory i.e. blue shift of UV emission peak may be attributed to the charge transfer from Ag to oxygen as discussed earlier. This phenomenon is supported from 2M Raman scattering. According to discussion by Anderson, the crystal field splitting (10Dq) is related to J for NiO by the following equation64,65
Our previous Raman study reveals that J increases in the presence of Ag decoration i.e. it may be concluded that crystal field splitting gets enhanced and causes blue shift of the UV emission. In contrast to the UV emission, visible emission exhibits non-linearity, initially its emission energy decreases and finally gets enhanced. The initial red shift may be assigned to the p–d charge transfer mechanism as described earlier. The later one is still to be investigated. For better clarity of the shifts in the luminescence spectrum, an enlarged version of it is represented in Fig. S6, ESI.† The striking point to mention here is that the full width at half maxima (FWHM) corresponding to the UV emission monotonically decreases with respect to Ag decoration (shown in Fig. 6(d)). It was realized earlier that the FWHM of the emission spectra significantly depends on the nonradiative transition which by consequence is determined by the electron–LO phonon interaction. In this context, the Huang-Rhys factor (S) is defined to attribute the electron–LO phonon interaction and was established to be proportional to the square of FWHM corresponding to the luminescence spectra.66 Mathematically, the S-factor is expressed by the following expression:
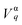 , ħωLO and
, ħωLO and 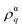 represent the electron–LO phonon interaction strength, energy of the LO vibrational mode and electron density respectively. Our previous Raman study indicates that the strength corresponding to the LO vibrational energy increases with Ag incorporation, thus the reduction of FWHM i.e. S-factor may be ascribed to the enhancement of electron–LO phonon within NiO due to the transfer of charge as discussed earlier. Therefore the shrinkage of FWHM leads to a decrement of the nonradiative transition; hence enhancement in the intensity corresponding to UV emission is caused by increased radiative transition.
represent the electron–LO phonon interaction strength, energy of the LO vibrational mode and electron density respectively. Our previous Raman study indicates that the strength corresponding to the LO vibrational energy increases with Ag incorporation, thus the reduction of FWHM i.e. S-factor may be ascribed to the enhancement of electron–LO phonon within NiO due to the transfer of charge as discussed earlier. Therefore the shrinkage of FWHM leads to a decrement of the nonradiative transition; hence enhancement in the intensity corresponding to UV emission is caused by increased radiative transition.
Photocatalytic activity within the NiO/Ag heterostructure
The photocatalytic activity of the NiO/Ag heterostructure was examined by monitoring the photodegradation of MB under UV light irradiation. Fig. 7 illustrates the variations of MB concentration with UV light irradiation time using NFO, NAG1 and NAG2 as photocatalysts. The concentration of MB was determined from absorbance spectra and the degradation efficiency was calculated via the following equation| η = (C0 − C)/C0 × 100 |
 | ||
| Fig. 7 MB degradation under UV irradiation (a) NFO (c) NAG1 (d) NAG2, (b) showing the magnified image of the NFO graph. | ||
The efficiency of the NFO, NAG1, and NAG2 is found to be 2.75%, 41% and 70% (after 210 minutes) respectively. Fig. S7(b)† shows the linear relationship between the ln(C/C0) versus ‘t’ justifying the fact that the reactions followed pseudo 1st order kinetics. Now according to the Langmuir–Hinshelwood kinetic model, pseudo 1st order rate constant (Kapp) may be calculated using the following equation56
| ln(C − C0) = Kapp × t |
The values of Kapp are presented in Fig. S7(c).† It is evident from the above data that the Kapp monotonically increases from the NFO to NAG2 samples implying that the addition of Ag on NiO surfaces has significantly increased the catalytic properties of NiO. The basic mechanism behind the observed photocatalytic process involves the excited electrons in the conduction band getting trapped by the electron absorbing agents such as the adsorbed O2 and generating superoxide anion radicals (O2−). At the same time, holes in the valance band produce hydroxyl radicals (˙OH).30 Our previous optical studies revealed that the electrons tunnel through the heterostructure formed between the metallic Ag and semiconducting NiO due to the difference in their work function. When such heterostructures are irradiated using UV light with an energy greater than the band gap energy of NiO, electrons are excited from the valence band to the conduction band of NiO. Since the energy of the conduction band of NiO, as presented in Fig. 5, is higher than that of the Fermi energy of Ag, excited electrons migrate from the conduction band of NiO into the Fermi energy of Ag through an interfacial charge transfer. Thus the enhanced value of Kapp is attributed to the electron–hole separation within the heterostructure. In this context it may be stated that photocatalytic degradation of methyl orange (MO) was studied using NiO/TiO2 and Ag/TiO2 heterostructures. It is interesting to note that the Kapp, obtained for our sample (NAG2) is approximately three times smaller than the maximum Kapp obtained by them, but our synthesized sample possesses comparable Kapp with that of P25.67 It is also noted that 3% Ag coated TiO2 nanoparticles degrade almost 90% MO within 200 minutes.12 But our synthesized NAG2 sample degrades 70% MB within 210 minutes. The difference in the rate constant may be attributed to either the difference in the degradation rate for MO and MB or the difference in degree of charge separation by the synthesized nanostructures.
Conclusions
In summary, flake-like NiO/Ag heterostructures have been fabricated by a two-step method. NiO flakes were initially synthesized using a hydrothermal process. Highly dense Ag nanoparticles were then grown on the top of the NiO flakes using a chemical method. FESEM and HRTEM studies confirm the formation of such heterostructures. Strong interfacial interactions between the NiO flakes and Ag nanoparticles are also supported by an FTIR spectroscopic investigation. LO optical phonon and magnon scattering modes within NiO flakes were examined using Raman spectroscopy. It has been observed that Ag attachment has a strong influence on these scattering modes due to charge transfer between oxygen and silver atoms. Specifically, it may be stated that Ag attachment significantly alters the exchange interaction within NiO due to the transfer of charge. It is also noted that the band edge gets red shifted in NiO/Ag heterostructures. UV emission is mostly determined by d–d crystal field splitting of Ni d that gets blue shifted originating from the variation of exchange interaction. Photocatalytic degradation of methylene blue in aqueous solution has been investigated in detail by employing pristine NiO and Ag decorated NiO nanostructures. Apparent photocatalysis reaction rates for different heterostructures have been found to follow the order of NAG2 > NAG1 > NFO implying the significant photostability-dependent photocatalysis behavior of the NiO/Ag heterostructures.Acknowledgements
One of us (SM) wishes to thank the Council for Scientific and Industrial Research (CSIR), Government of India, for awarding him a Senior Research Fellowship during execution of the work. We also acknowledge the University Grant Commission, Govt of India for university with potential scheme (UPE - II).References
- M. Hassan, E. Haque, K. R. Reddy, A. I. Minett, J. Chen and V. G. Gomes, Nanoscale, 2014, 6, 11988–11994 RSC.
- X. Zhang, L. Zhang, T. Xie and D. Wang, J. Phys. Chem. C, 2009, 113, 7371–7378 CAS.
- G. Williams and P. V. Kamat, Langmuir, 2009, 25, 13869–13873 CrossRef CAS PubMed.
- R. Moreno-Tost, J. Santamaría-González, P. Maireles-Torres, E. Rodríguez-Castellón and A. Jiménez-López, J. Mater. Chem., 2002, 12, 3331–3336 RSC.
- B. Huang, Q.-C. Yu, H.-M. Wang, G. Chen and K.-A. Hu, J. Power Sources, 2004, 137, 163–174 CrossRef CAS.
- I. Hotovy, V. Rehacek, P. Siciliano, S. Capone and L. Spiess, Thin Solid Films, 2002, 418, 9–15 CrossRef CAS.
- R. Makkus, K. Hemmes and J. De Wit, J. Electrochem. Soc., 1994, 141, 3429–3438 CrossRef CAS.
- K. Fominykh, J. M. Feckl, J. Sicklinger, M. Döblinger, S. Böcklein, J. Ziegler, L. Peter, J. Rathousky, E. W. Scheidt and T. Bein, Adv. Funct. Mater., 2014, 24, 3123–3129 CrossRef CAS.
- G. Natu, P. Hasin, Z. Huang, Z. Ji, M. He and Y. Wu, ACS Appl. Mater. Interfaces, 2012, 4, 5922–5929 CAS.
- N. Senthilkumar, K. J. Babu, G. Gnana kumar, A. R. Kim and D. J. Yoo, Ind. Eng. Chem. Res., 2014, 53, 10347–10357 CrossRef CAS.
- P. Gibson, H. Schreuder-Gibson and D. Rivin, Colloids Surf., A, 2001, 187, 469–481 CrossRef.
- K. R. Reddy, K. Nakata, T. Ochiai, T. Murakami, D. A. Tryk and A. Fujishima, J. Nanosci. Nanotechnol., 2011, 11, 3692–3695 CrossRef CAS PubMed.
- K. R. Reddy, K.-P. Lee and A. I. Gopalan, J. Nanosci. Nanotechnol., 2007, 7, 3117–3125 CrossRef CAS PubMed.
- K. R. Reddy, K. Nakata, T. Ochiai, T. Murakami, D. A. Tryk and A. Fujishima, J. Nanosci. Nanotechnol., 2010, 10, 7951–7957 CrossRef CAS PubMed.
- K. R. Reddy, M. Hassan and V. G. Gomes, Appl. Catal., A, 2015, 489, 1–16 CrossRef CAS.
- K. R. Reddy, K. P. Lee, A. I. Gopalan, M. S. Kim, A. M. Showkat and Y. C. Nho, J. Polym. Sci., Part A: Polym. Chem., 2006, 44, 3355–3364 CrossRef CAS.
- A. M. Showkat, Y.-p. Zhang, M. S. Kim, A. I. Gopalan, K. R. Reddy and K. Lee, Bull. Korean Chem. Soc., 2007, 28, 1985 CrossRef CAS.
- K. R. Reddy, V. G. Gomes and M. Hassan, Mater. Res. Express, 2014, 1, 015012 CrossRef.
- K. R. Reddy, B. C. Sin, C. H. Yoo, W. Park, K. S. Ryu, J.-S. Lee, D. Sohn and Y. Lee, Scr. Mater., 2008, 58, 1010–1013 CrossRef CAS.
- Q. Liu, L. Wei, S. Yuan, X. Ren, Y. Zhao, Z. Wang, M. Zhang, L. Shi, D. Li and A. Li, RSC Adv., 2015, 5, 71778–71784 RSC.
- S.-H. Park and W.-J. Lee, RSC Adv., 2015, 5, 23548–23555 RSC.
- L. Wang, X. Lu, C. Wen, Y. Xie, L. Miao, S. Chen, H. Li, P. Li and Y. Song, J. Mater. Chem. A, 2015, 3, 608–616 CAS.
- J. Cao, Z. Wang, R. Wang, S. Liu, T. Fei, L. Wang and T. Zhang, J. Mater. Chem. A, 2015, 3, 5635–5641 CAS.
- Y. Ding, Y. Wang, L. Su, H. Zhang and Y. Lei, J. Mater. Chem., 2010, 20, 9918–9926 RSC.
- L. Zhang, Y.-H. Dou and H.-C. Gu, J. Colloid Interface Sci., 2006, 297, 660–664 CrossRef CAS PubMed.
- H. Wang, X. Liu and S. Han, CrystEngComm, 2016, 18, 1933–1943 RSC.
- M. Sasidharan, N. Gunawardhana, C. Senthil and M. Yoshio, J. Mater. Chem. A, 2014, 2, 7337–7344 CAS.
- K. R. Reddy, B. C. Sin, K. S. Ryu, J.-C. Kim, H. Chung and Y. Lee, Synth. Met., 2009, 159, 595–603 CrossRef CAS.
- Z. Li, F. Zhang, A. Meng, C. Xie and J. Xing, RSC Adv., 2015, 5, 612–620 RSC.
- Y. Zheng, L. Zheng, Y. Zhan, X. Lin, Q. Zheng and K. Wei, Inorg. Chem., 2007, 46, 6980–6986 CrossRef CAS PubMed.
- S. S. Warule, N. S. Chaudhari, R. T. Khare, J. D. Ambekar, B. B. Kale and M. A. More, CrystEngComm, 2013, 15, 7475–7483 RSC.
- G. Zhou, D.-W. Wang, L.-C. Yin, N. Li, F. Li and H.-M. Cheng, ACS Nano, 2012, 6, 3214–3223 CrossRef CAS PubMed.
- A. Citra, G. V. Chertihin, L. Andrews and M. Neurock, J. Phys. Chem. A, 1997, 101, 3109–3118 CrossRef CAS.
- W. Liu, X. Wang, L. Zhang and J. Lian, RSC Adv., 2015, 5, 54138–54147 RSC.
- Z. W. Tong, D. Yang, Y. Y. Sun, Y. Tian and Z. Y. Jiang, Phys. Chem. Chem. Phys., 2015, 17, 12199–12206 RSC.
- R. Tang, H. Su, S. Duan, Y. Sun, L. Li, X. Zhang, S. Zeng and D. Sun, RSC Adv., 2015, 5, 41949–41960 RSC.
- X. Ni, Q. Zhao, F. Zhou, H. Zheng, J. Cheng and B. Li, J. Cryst. Growth, 2006, 289, 299–302 CrossRef CAS.
- B. Wang, J. Cheng, Y. Wu, D. Wang and D. He, Electrochem. Commun., 2012, 23, 5–8 CrossRef CAS.
- G. Zhu, C. Xi, H. Xu, D. Zheng, Y. Liu, X. Xu and X. Shen, RSC Adv., 2012, 2, 4236–4241 RSC.
- N. Mironova-Ulmane, A. Kuzmin, I. Sildos and M. Pärs, Cent. Eur. J. Phys., 2011, 9, 1096–1099 CAS.
- R. Dietz, G. Parisot and A. Meixner, Phys. Rev. B: Solid State, 1971, 4, 2302 CrossRef.
- W. Wang, Y. Liu, C. Xu, C. Zheng and G. Wang, Chem. Phys. Lett., 2002, 362, 119–122 CrossRef CAS.
- M. T. Hutchings and E. Samuelsen, Phys. Rev. B: Solid State, 1972, 6, 3447 CrossRef CAS.
- E. Funkenbusch and B. Cornilsen, Solid State Commun., 1981, 40, 707–710 CrossRef CAS.
- G. Ghiringhelli, A. Piazzalunga, C. Dallera, T. Schmitt, V. Strocov, J. Schlappa, L. Patthey, X. Wang, H. Berger and M. Grioni, Phys. Rev. Lett., 2009, 102, 027401 CrossRef CAS PubMed.
- M. A. Sliem, T. Hikov, Z.-A. Li, M. Spasova, M. Farle, D. A. Schmidt, M. Havenith-Newen and R. A. Fischer, Phys. Chem. Chem. Phys., 2010, 12, 9858–9866 RSC.
- H. Zeng, W. Cai, B. Cao, J. Hu, Y. Li and P. Liu, Appl. Phys. Lett., 2006, 88, 181905 CrossRef.
- C. Chen, Y. Zheng, Y. Zhan, X. Lin, Q. Zheng and K. Wei, Dalton Trans., 2011, 40, 9566–9570 RSC.
- R. Udayabhaskar, B. Karthikeyan, P. Sreekanth and R. Philip, RSC Adv., 2015, 5, 13590–13597 RSC.
- J. Li, F. Zhao, L. Zhang, M. Zhang, H. Jiang, S. Li and J. Li, RSC Adv., 2015, 5, 67610–67616 RSC.
- B. Karthikeyan, T. Pandiyarajan, S. Hariharan and M. S. Ollakkan, CrystEngComm, 2016, 18, 601–607 RSC.
- H. Liu, G. Shao, J. Zhao, Z. Zhang, Y. Zhang, J. Liang, X. Liu, H. Jia and B. Xu, J. Phys. Chem. C, 2012, 116, 16182–16190 CAS.
- S. Singh, V. Nandanwar, H. Srivastava, A. Yadav, A. Bhakar, P. Sagdeo, A. Sinha and T. Ganguli, Dalton Trans., 2015, 44, 14793–14798 RSC.
- M. T. Greiner, M. G. Helander, Z.-B. Wang, W.-M. Tang and Z.-H. Lu, J. Phys. Chem. C, 2010, 114, 19777–19781 CAS.
- M. Butler and D. Ginley, J. Electrochem. Soc., 1978, 125, 228–232 CrossRef CAS.
- K. Dai, L. Lu, C. Liang, G. Zhu and L. Geng, RSC Adv., 2014, 4, 62751–62758 RSC.
- A. A. Al-Ghamdi, M. S. Abdel-wahab, A. Farghali and P. Hasan, Mater. Res. Bull., 2016, 75, 71–77 CrossRef CAS.
- Y. Qi, H. Qi, C. Lu, Y. Yang and Y. Zhao, J. Mater. Sci.: Mater. Electron., 2009, 20, 479–483 CrossRef CAS.
- C. Díaz-Guerra, A. Remon, J. Garcia and J. Piqueras, Phys. Status Solidi A, 1997, 163, 497–503 CrossRef.
- S. Lee, S. Park, C.-W. Kim, D. Lee, C. Lee and C. Jin, Thin Solid Films, 2016, 598, 33–38 CrossRef CAS.
- B. Brandow, Adv. Phys., 1977, 26, 651–808 CrossRef CAS.
- S. Hüfner, Adv. Phys., 1994, 43, 183–356 CrossRef.
- V. Sokolov, V. Pustovarov, V. Churmanov, V. Y. Ivanov, N. Gruzdev, P. Sokolov, A. Baranov and A. Moskvin, Phys. Rev. B: Condens. Matter Mater. Phys., 2012, 86, 115128 CrossRef.
- P. W. Anderson, Phys. Rev., 1959, 115, 2 CrossRef CAS.
- P. W. Anderson, Solid State Phys., 1963, 14, 99–214 CAS.
- S. Das, C. K. Ghosh, R. Dey and M. Pal, RSC Adv., 2016, 6, 236–244 RSC.
- T. Sreethawong, S. Ngamsinlapasathian and S. Yoshikawa, Chem. Eng. J., 2012, 192, 292–300 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra09432e |
| This journal is © The Royal Society of Chemistry 2016 |