Novel wide band gap polymers based on dithienobenzoxadiazole for polymer solar cells with high open circuit voltages over 1 V
Dakang Ding†
ab,
Jiuxing Wang†bd,
Weiye Chena,
Meng Qiub,
Junzhen Rena,
Huilin Zhenga,
Deyu Liub,
Mingliang Sun*ac and
Renqiang Yang*b
aInstitute of Material Science and Engineering, Ocean University of China, Qingdao 266100, People's Republic of China. E-mail: mlsun@ouc.edu.cn
bCAS Key Laboratory of Bio-based Materials, Qingdao Institute of Bioenergy and Bioprocess Technology, Chinese Academy of Sciences, Qingdao 266101, People's Republic of China. E-mail: yangrq@qibebt.ac.cn
cState Key Laboratory of Luminescent Materials and Devices, South China University of Technology, Guangzhou 510640, China
dUniversity of Chinese Academy of Sciences, Beijing 100049, China
First published on 17th May 2016
Abstract
A new monomer dithieno-[3′,2′:3,4;2′′,3′′:5,6]benzo[1,2-c]xadiazole (fDTBO) is first used as an electron-deficient acceptor to build D–A copolymers in a photovoltaic field. Two polymers PBDTT-fDTBO and PBDTO-fDTBO consist of fDTBO with thienyl-substituted-benzodithiophene (BDTT) or alkoxy-substituted benzodithiophene (BDTO). Both polymers show a deep HOMO around −5.5 eV with a wide band gap of over 1.9 eV. The polymer solar cells (PSCs) based on two polymers both show over 1 V high open circuit voltage (Voc) independent of polymer/PCBM ratios and solvent additives content in the PSCs active layer. The power conversion efficiency (PCE) based on PBDTT-fDTBO devices is 4.5% for single junction PSCs, and these polymers can be applied in tandem PSCs due to their wide band gap (up to 1.99 eV). This work demonstrates that the fDTBO unit is a promising building block to design wide band gap photovoltaic polymers with high Voc.
1 Introduction
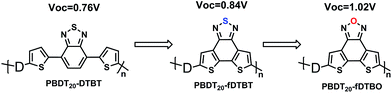
The bulk heterojunction polymer solar cells based on blending of polymer and PCBM have exhibited high PCE of over 10% for single polymer solar cells (PSCs) in recent years.1–4 In order to achieve higher PCE, the tandem solar cells based on wide band gap (WBG) polymers were needed5–7 and it may be the most promising route to reach PCE up to 15%.8,9 Thus, more and more WBG polymers were synthesized.10–13 Generally, the PSCs based on WBG polymers cannot provide very high power conversion efficiency due to limited light absorption.14–18 The PCE of PSCs is determined by short-circuit current density (Jsc), fill factor (FF) and Voc.19–21Increasing Voc of PSCs is usually as a simple and feasible method to increase the PCE. The Voc is proportional to the energy difference between the lowest unoccupied molecular orbital (LUMO) of the fullerene derivatives and the highest occupied molecular orbital (HOMO) of polymers.9 Higher Voc may be achieved by deeper HOMO of polymer as the fullerene derivatives remain the same.22 Deep HOMO of polymer also may improve the oxidative stability and decrease energy loss of PSCs devices.23,24 Typically, there are two molecular structure design strategies to obtain deep HOMO polymer. One is introducing the electron-withdrawing atom (fluorine, sulfur or oxygen) to the conjugated polymer backbone.25,26 The other is decreasing the effective conjugate degree by incorporating large side groups or increasing the sinuosity of conjugated backbone.27,28 Dithienyl-2,1,3-benzothiadiazole (DTBT) is a well-known electron-withdrawing acceptor, and it was first used for D–A copolymer by Andersson et al. in 2003.29 As shown in Chart 1, Wei You et al. synthesized a new monomer 4,7-bis(5-bromothiophen-2-yl)-5,6-diuorobenzo-[c][1,2,5]thiadiazole (DTffBT) by introducing F to DTBT for D–A co-polymer with a deeper HOMO.13 Li et al. used fusion strategy to synthesis a new monomer dithieno-[3′,2′:3,4;2′′,3′′:5,6]benzo[1,2-c][1,2,5]thiadiazole (fDTBT) by fusing two thienyl units on the benzothiadiazole (BT), and they got a high Voc of 0.84 based on polymer with fDTBT and benzodithiophene (BDT) derivative.30
The band gap is an important parameter for PSCs and it is tuned by electron-rich donor and electron-deficient acceptor unit.31 BDT is a good donor unit for high photovoltaic performance, and some WBG polymers based on BDT unit were synthesized and showed high PCEs.32–34 N-Alkylthieno[3,4-c]pyrrole-4,6-dione(TPD)33,35–37 derivatives, 2-alkyl-benzo[d][1,2,3]triazole(TAZ)38 derivatives, thiazolo[5,4d]thiazole(TTz)39,40 derivatives and 1,3-bis(thiophen-2-yl)-5,7-bis(2-ethyl-hexyl)benzo-[1,2-c:4,5-c]dithiophene-4,8-dione (BDD)41 et al. were main acceptor donors for BDT-based WBG polymers. To the best of our knowledge, the PSCs of wide band gap polymers based on BDT unit rarely show the Voc over 1 V with high PCE.
Hou and coworkers synthesized a new polymer with BDT-F and BDD, and the Voc based on the new polymer was 1.02 V which was reduced to 0.96 V after adding the solvent additives.
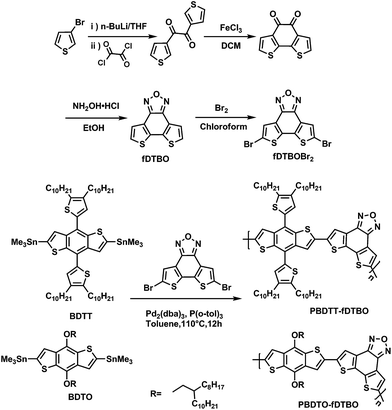
The fDTBT was a new promising electron-accepting moiety with good π-electron delocalization and planarity, and we can further get dithieno-[3′,2′:3,4;2′′,3′′:5,6]benzo[1,2c]xadiazole (fDTBO) with stronger intermolecular interactions and electron-withdrawing ability by replacing sulfur with oxygen (Chart 1). The fDTBO unit is first used in photovoltaic field in this work. Compared with sulfur atom, the oxygen had higher electronegativity and electron withdrawing ability, so higher Voc exceeding 1 V can be achieved. In this work, we use fDTBO to build two D–A copolymers: PBDTT-fDTBO and PBDTO-fDTBO. As shown in Scheme 1, two polymers were synthesized by stille coupling with fDTBO and BDTT or BDTO. As a result, PBDTT-fDTBO and PBDTO-fDTBO show high Voc with 1.04 V and 1.02 V, with wide band gap of 1.99 eV and 1.91 eV, respectively. What's more, the Voc of PSCs devices can maintain above 1 V during different polymers/PCBM ratios and solvent additives contents optimization. BHJ PSCs based on PBDTT-fDTBO obtained Voc of 1.04 V with decent PCE around 4.52%. We believe the fDTBO unit will be a promising building block to achieve high photovoltaic performances due to its good planarity and strong electron withdrawing.
2 Results and discussion
2.1 Synthesis and characterization
The synthetic routes of fDTBO, PBDTT-fDTBO and PBDTO-fDTBO are shown in Scheme 1. The detailed synthetic routes were described in experimental section and the monomer fDTBO was synthesized according to reported method.42 Polymer PBDTT-fDTBO and PBDTO-fDTBO were synthesized by Stille coupling reaction using Pd2(dba)3/P(o-tol)3 as catalysts and 5 ml toluene as the solvent. The color of two polymers was both deep red. Both of polymers could only be slowly dissolved in DCB at 90 °C because of their poor solubility. The number-average molecular weights of PBDTT-fDTBO and PBDTO-fDTBO were 88.4 kDa and 89.3 kDa, with a polydispersity index of 1.40 and 1.93, respectively.2.2 Thermal analysis
The thermal properties of PBDTT-fDTBO and PBDTO-fDTBO were analyzed by thermogravimetric analysis (TGA). As is shown in Fig. 1, the decomposition temperatures (5% weight loss) of PBDTT-fDTBO and PBDTO-fDTBO are 394 °C and 310 °C, respectively. This result indicts that both polymers show high thermal stability for PSCs applications.2.3 Optical properties
The ultraviolet-visible absorption spectra (UV-vis) of these two polymers in thin films and dilute DCB solution is shown in Fig. 2. The corresponding optical data is listed in Table 1. In DCB solution, the absorption peak wavelength of PBDTT-fDTBO and PBDTO-fDTBO is at 536 and 530 nm, respectively, which is attributed to intramolecular charge transfer (ICT)43 between BDT unit and fDTBO unit. The sharp shoulder peak of PBDTT-fDTBO and PBDTO-fDTBO at 580 nm and 568 nm is due to the aggregations of the polymer chains. Compared with the absorption spectra in DCB solution, two polymers in thin film have stronger intermolecular force, and show about 13 nm red-shifted. In addition, the localized π–π* transitions can bring about absorption peak in short-wavelength area. The absorption spectra of PBDTT-fDTBO showed red-shifted compared with the spectra of PBDTO-fDTBO which may be ascribed to the 2D conjugated structures of the polymer PBDTT-fDTBO. The optical band gap (Eoptg) of PBDTT-fDTBO and PBDTO-fDTBO are 1.91 eV and 1.99 eV respectively, which were calculated from their absorption onset of thin film.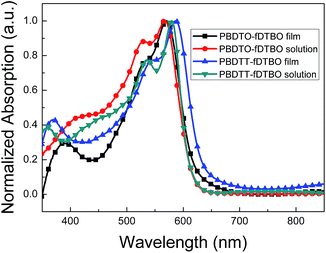 | ||
| Fig. 2 UV-vis absorption spectra of PBDTT-fDTBO and PBDTO-fDTBO in dilute DCB solution and in thin films. | ||
| Polymers | λmax film (nm) | λmax solution (nm) | λonset film (eV) | Eoptg (eV) | Eox (V) | HOMOcv (eV) | LUMOcv (eV) | EDFTg (eV) | HOMODFT (eV) | LUMODFT (eV) |
|---|---|---|---|---|---|---|---|---|---|---|
| PBDTT-fDTBO | 369, 546, 596 | 361, 534, 580 | 648 | 1.91 | 1.23 | −5.64 | −3.73 | 2.63 | −5.48 | −2.85 |
| PBDTO-fDTBO | 392, 532, 571 | 403, 530, 564 | 623 | 1.99 | 1.15 | −5.56 | −3.57 | 2.74 | −5.39 | −2.65 |
2.4 Electrochemical properties
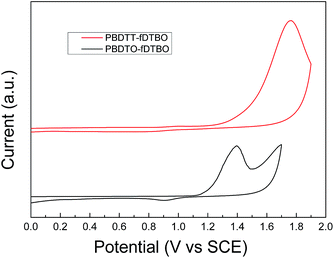
The electrochemical properties of two polymers were measured by cyclic voltammetry (CV). The CV curves are shown in Fig. 3 and detailed onset oxidation potentials (Eox) and HOMO, LUMO energy levels are shown in Table 1. The Eox of PBDTT-fDTBO and PBDTO-fDTBO were 1.23 V and 1.15 V vs. saturated calomel electrode (SCE), respectively. Half-wave potential of the ferrocene/ferrocenium (Fc/Fc+) redox couple was as the internal standard, and the potential (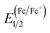 ) vs. SCE was measured to be 0.39 V. The HOMO of PBDTT-fDTBO and PBDTO-fDTBO were calculated by EHOMO = −e(Eox + 4.8 −
) vs. SCE was measured to be 0.39 V. The HOMO of PBDTT-fDTBO and PBDTO-fDTBO were calculated by EHOMO = −e(Eox + 4.8 − 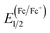 ) and the corresponding LUMO were calculated by HOMO and Eoptg.44 These two polymers both had deep HOMO energy levels regardless of the donor unit, which were mainly attributed to the strong electron-withdrawing group fDTBO unit. Thus high Voc of PSCs could be expected when polymers based on fDTBO unit were blended with PCBM.
) and the corresponding LUMO were calculated by HOMO and Eoptg.44 These two polymers both had deep HOMO energy levels regardless of the donor unit, which were mainly attributed to the strong electron-withdrawing group fDTBO unit. Thus high Voc of PSCs could be expected when polymers based on fDTBO unit were blended with PCBM.
2.5 Theoretical calculations
In order to understand the optoelectronic properties of two polymers, density functional theory (DFT) calculations were carried out using Gaussian 09 program. The optimized molecular geometries were recognized as the minimum energy conformations when calculating vibrational frequencies at the same level. Some methods were implemented to simplify the calculations, such as choosing one repeating unit and replacing alky side chains by methyl groups. The optimized molecular geometry and frontier molecular orbitals are shown in Fig. 4 and detailed energy levels are depicted in Table 1. The simulated data from the DFT calculations coincided well with experimental results estimated from the cyclic voltammograms. Two polymers showed similar distributions of electron density. The LUMO was mainly delocalized in the fDTBO unit, while the HOMO was located both in donor unit and acceptor unit. | ||
| Fig. 4 Optimized molecular geometries and frontier molecular orbitals from DFT calculations on PBDTT-fDTBO and PBDTO-fDTBO. | ||
2.6 Hole mobility
Hole mobility is an another import parameter influencing the PSCs performances. It was measured by space-charge-limited-current (SCLC) with polymer and PC71BM blend film. As shown in Fig. 5, the hole mobility of PBDTT-fDTBO and PBDTO-fDTBO is 3.11 × 10−4 and 2.90 × 10−5 cm2 V−1 s−1, respectively. The polymer PBDTT-fDTBO has higher hole mobility than PBDTO-fDTBO, which may induce the PSCs device based on PBDTT-fDTBO showing higher performances. The hole mobilities values of two polymers make a great difference, which should be partly attributed to the difference of their structures. Previous reports have revealed that 2D-structure can enhance the hole mobility effectively.48,49 The polymer PBDTT-fDTBO is based on alkylthienyl-substituted BDT (2D structure) and PBDTO-fDTBO is based on alkoxyl-substituted BDT, thus PBDTT-fDTBO exhibits higher hole mobility.2.7 Photovoltaic properties
The bulk heterojunction(BHJ) polymer solar cells were fabricated with the traditional structure of ITO/PEDOT (poly(3,4-ethylenedioxythiophene)):PSS(poly(styrenesulfonate))/PBDTT-fDTBO(PBDTO-fDTBO):PC71BM/Ca/Al to investigate the photovoltaic properties of two polymers. These polymers blended with PC71BM were heated to 80 °C for 1 hour before spin-coated as the active layer due to their poor solubility. As shown in Tables 2 and 3 and Fig. 6, different ratios of polymer/PC71BM and different proportions of solvent additives (CN)45 had obvious influences on the photovoltaic properties of PSCs devices. With different D/A ratios and the proportion of CN devices performances were tested, and the optimal D/A ratios of two polymers were both probed to be 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and the optimal proportion of CN of PBDTT-fDTBO and PBDTO-fDTBO was 3% and 0%, respectively. In addition, the Voc of PSCs based on two polymers both showed over 1 V with different polymer/PC71BM ratios and CN proportions.
2 and the optimal proportion of CN of PBDTT-fDTBO and PBDTO-fDTBO was 3% and 0%, respectively. In addition, the Voc of PSCs based on two polymers both showed over 1 V with different polymer/PC71BM ratios and CN proportions.
| Polymer | D/A | Voc (V) | Jsc (mA cm−2) | FF (%) | PCEmax/PCEave (%) |
|---|---|---|---|---|---|
| PBDTT-fDTBO | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
1.07 | 2.72 | 40.13 | 1.16/1.10 |
| PBDTT-fDTBO | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
1.05 | 5.53 | 49.57 | 2.88/2.75 |
| PBDTT-fDTBO | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3 |
1.06 | 3.56 | 53.59 | 2.02/1.87 |
| PBDTO-fDTBO | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
1.03 | 3.78 | 43.66 | 1.69/1.55 |
| PBDTO-fDTBO | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
1.02 | 5.41 | 50.67 | 2.80/2.75 |
| PBDTO-fDTBO | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3 |
1.00 | 3.30 | 41.38 | 1.36/1.25 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) with different CN amounts
2) with different CN amounts
| Polymer | CN (%) | Voc (V) | Jsc (mA cm−2) | FF (%) | PCEmax/PCEave (%) |
|---|---|---|---|---|---|
| PBDTT-fDTBO | 0 | 1.05 | 5.53 | 49.57 | 2.88/2.75 |
| PBDTT-fDTBO | 1 | 1.02 | 5.32 | 40.12 | 2.18/2.13 |
| PBDTT-fDTBO | 2 | 1.03 | 5.33 | 48.38 | 2.66/2.51 |
| PBDTT-fDTBO | 3 | 1.04 | 7.95 | 54.59 | 4.52/4.36 |
| PBDTT-fDTBO | 4 | 1.04 | 7.57 | 55.02 | 4.33/4.23 |
| PBDTO-fDTBO | 0 | 1.02 | 5.41 | 50.67 | 2.80/2.75 |
| PBDTO-fDTBO | 1 | 1.01 | 3.33 | 45.15 | 1.52/1.48 |
| PBDTO-fDTBO | 2 | 1.00 | 3.30 | 44.26 | 1.46/1.40 |
| PBDTO-fDTBO | 3 | 1.01 | 3.31 | 46.65 | 1.48/1.39 |
The best photovoltaic performance of PSCs device based on polymer PBDTT-fDTBO was 4.52% with a Voc of 1.04 V, a Jsc of 7.95 mA cm−2 and FF of 54.59%, while the PCE of PBDTO-fDTBO based PSCs device reached maximum value of 2.88%, with a Voc of 1.05 V, Jsc of 5.53 mA cm−2 and FF of 49.57%. Wei-Shi Li30 group synthesized several polymers based on BDT and DTBT (fDTBT) and the polymer PBDT16-fDTBT achieved the best photovoltaic performance. The PSCs based on PBDT20-DTBT showed maximum PCE of 3.98% with Voc of 0.76 V, and the PSCs based on PBDT16-fDTBT showed maximum PCE of 4.50% with Voc of 0.85 V. Thus, the Voc of our polymer PBDTT-fDTBO or PBDTO-fDTBO based device was nearly 0.2 V higher than Voc of PBDT16-fDTBT, which agreed well with our expectation. Furthermore, the higher Voc of PBDTT-fDTBO based PSCs agreed well with CV curves.
The external quantum efficiency (EQE) spectra of the optimized polymer solar cells based on PBDTT-fDTBO and PBDTO-fDTBO are shown in Fig. 7. The EQE curves exhibited absorption region from 300 to 640 nm. The maximum EQE of PBDTT-fDTBO based PSCs devices was 58.63% at 480 nm, while the maximum EQE of PBDTO-fDTBO based PSCs devices was 43.76% at 530 nm. The calculated current densities from the EQE curves were 8.02 and 5.40 mA cm−2 of PBDTT-fDTBO and PBDTO-fDTBO respectively, which were consistent with the Jsc obtained by J–V curves.
2.8 Morphology characterization
The influences of CN on the morphology characterizations of the blended films were investigated by atomic force microscopy (AFM) and transmission electron microscopy (TEM).As shown in Fig. 8(a, b, e and f), the surface of PBDTT-fDTBO/PC71BM blended film without CN was smooth with the root-mean-square (RMS) roughness value of 0.7 nm. While 3% CN was added, the RMS value of the blended film was 1.35 nm due to larger domains. Meanwhile, the surface of PBDTO-fDTBO/PC71BM blended film without CN and with 3% CN showed similar RMS value of 0.91 nm and 0.89 nm, respectively. As shown in Fig. 8(c, d, g and h), the aggregations of PC71BM and PBDTO-fDTBO without CN was more evident than those with CN, while the aggregations of polymer PBDTT-fDTBO and PC71BM became evident after adding 3% CN (Fig. 8(a, b, e and f)). What's more, the better surface morphology and phase separation of PBDTT-fDTBO was another reason for its higher hole mobility.
3 Conclusion
In summary, new monomer fDTBO is first used as acceptor to build D–A copolymers in photovoltaic polymer field. The PSCs based on two polymers both showed high Voc of above 1 V, and the PSCs devices can remained 1 V high Voc during polymer/PCBM ratios and solvent additives content optimization. Compared with the polymer PBDT16-fDTBT, the Voc of our polymer increased nearly 0.2 V. Therefore, this may provide us a new strategy by replacing the sulfur atom with oxygen for achieving stable higher Voc photovoltaic devices.4 Experimental section
4.1 Materials
All solvents and chemicals were purchased by reagent companies, and all of the solvents except toluene and tetrahydrofuran (THF) were used directly. Toluene and THF were used for water sensitive reactions after dried by sodium under argon atmosphere. Compound BDTT, BDTO, fDTBO were synthesized according to reported methods.42,46,474.2 Measures
1H NMR (nuclear magnetic resonance) and 13C NMR spectra were tested by the Bruker DRX with 600MHZ spectrometer with tetramethylsilane (TMS) as zero for a standard. Elemental analysis data was recorded on a Vario El Cube elementar analyzer. Under a nitrogen atmosphere, TGA curves were recorded by a SDT Q600 with a heating rate of 10 °C min−1. The molecular weights (Mn) of two polymers were performed by GPC with polystyrene as the standard and THF as the solvent at 35 °C. UV-vis absorption spectra were got with solvents and thin films using the Hitachi U-4100 spectrophotometer. CV curves were recorded by a CHI 660D electrochemical workstation using the standard three-electrode electrochemical cell system with a solution of tetrabutylammonium phosphorus hexafluoride (0.1 M) in acetonitrile (under an argon atmosphere) at a scan rate of 100 mV s−1. In the three-electrode system, Pt electrode with the polymer film was as the working electrode, SCE was as the reference electrode and Pt wire was as the auxiliary electrode at the ambient temperature. The DFT calculations were measured by Gaussian 09 program at the B3LYP/6-31G(d,p) levels at gas phase. AFM pictures were exhibited by an Alilgent 5400 at ambient conditions. Transmission electron microscopy (TEM) images were recorded by a JEOL JEM-1011 transmission electron microscope at the high voltage of 100 kV.4.3 Fabrication and characterization of PSCs
Conventional polymer solar cells devices were fabricated with the structure of ITO/PEDOT:PSS/PBDTT-fDTBO(PBDTO-fDTBO):PC71BM/Ca/Al. The ITO-coated glasses were cleaned in the ultrasonic bath with water, and washed with acetone, toluene, methanol and isopropyl alcohol subsequently. After about 20 min oxygen plasma treatment, the layer of PEDOT:PSS (30 nm) was spin-coated onto the ITO glasses and then dried at 120 °C under argon atmosphere for 20 min. The polymer blended with PC71BM (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and 1
2 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) were dissolved in CN/DCB (0%, 1%, 2% or 3%) solution. The solution was maintained at 90 °C for 1 hour before being spin-coated as the active layer (90 nm) onto the ITO/PEDOT:PSS substrate. At last Ca (10 nm) and Al (100 nm) layers were in sequence thermal evaporated onto the active layer with a pressure of 3 × 10−4 Pa. The active area of the PSCs device in this work was about 0.1 cm2. The current density–voltage characteristics were measured by a Keithley 2420 source measurement under illumination (AM 1.5 G, 100 mW cm−2) from a Newport solar simulator. A standard silicon solar cell was used to calibrate the light intensity. The external quantum efficiency (EQE) of the PSCs were recorded by a certified Newport incident photon conversion efficiency (IPCE) measurement system.
3) were dissolved in CN/DCB (0%, 1%, 2% or 3%) solution. The solution was maintained at 90 °C for 1 hour before being spin-coated as the active layer (90 nm) onto the ITO/PEDOT:PSS substrate. At last Ca (10 nm) and Al (100 nm) layers were in sequence thermal evaporated onto the active layer with a pressure of 3 × 10−4 Pa. The active area of the PSCs device in this work was about 0.1 cm2. The current density–voltage characteristics were measured by a Keithley 2420 source measurement under illumination (AM 1.5 G, 100 mW cm−2) from a Newport solar simulator. A standard silicon solar cell was used to calibrate the light intensity. The external quantum efficiency (EQE) of the PSCs were recorded by a certified Newport incident photon conversion efficiency (IPCE) measurement system.
Acknowledgements
The authors gratefully acknowledge financial support from the NSFC (21274134, 51573205), and State Key Laboratory of Luminescent Materials and Devices (2016-skllmd-01).References
- L. Lu, T. Zheng, Q. Wu, A. M. Schneider, D. Zhao and L. Yu, Chem. Rev., 2015, 115, 12666 CrossRef CAS PubMed.
- Z. Wu, C. Sun, S. Dong, X. F. Jiang, S. Wu, H. Wu, H. L. Yip, F. Huang and Y. Cao, J. Am. Chem. Soc., 2016, 138, 2004 CrossRef CAS PubMed.
- Y. Liu, J. Zhao, Z. Li, C. Mu, W. Ma, H. Hu, K. Jiang, H. Lin, H. Ade and H. Yan, Nat. Commun., 2014, 5, 5293 CrossRef CAS PubMed.
- S. H. Liao, H. J. Jhuo, P. N. Yeh, Y. S. Cheng, Y. L. Li, Y. H. Lee, S. Sharma and S. A. Chen, Sci. Rep., 2014, 4, 6813 CrossRef CAS PubMed.
- A. R. b. M. Yusoff, D. Kim, H. P. Kim, F. K. Shneider, W. J. da Silva and J. Jang, Energy Environ. Sci., 2015, 8, 303 CAS.
- J. You, L. Dou, K. Yoshimura, T. Kato, K. Ohya, T. Moriarty, K. Emery, C. C. Chen, J. Gao, G. Li and Y. Yang, Nat. Commun., 2013, 4, 1446 CrossRef PubMed.
- V. Vohra, K. Kawashima, T. Kakara, T. Koganezawa, I. Osaka, K. Takimiya and H. Murata, Nat. Photonics, 2015, 9, 403 CrossRef CAS.
- M. Lenes, G.-J. A. H. Wetzelaer, F. B. Kooistra, S. C. Veenstra, J. C. Hummelen and P. W. M. Blom, Adv. Mater., 2008, 20, 2116 CrossRef CAS.
- M. C. Scharber, D. Mühlbacher, M. Koppe, P. Denk, C. Waldauf, A. J. Heeger and C. J. Brabec, Adv. Mater., 2006, 18, 789 CrossRef CAS.
- Q. Liu, X. Bao, Y. Yan, Z. Du, V. A. L. Roy, D. Zhu, M. Sun, C. S. Lee and R. Yang, J. Mater. Sci., 2014, 50, 2263 CrossRef.
- V. Tamilavan, J. Lee, R. Agneeswari, D. Y. Lee, S. Cho, Y. Jin, S. H. Park and M. H. Hyun, Polymer, 2015, 80, 95 CrossRef CAS.
- W. Li, A. Furlan, W. S. Roelofs, K. H. Hendriks, G. W. van Pruissen, M. M. Wienk and R. A. Janssen, Chem. Commun., 2014, 50, 679 RSC.
- S. C. Price, A. C. Stuart, L. Yang, H. Zhou and W. You, J. Am. Chem. Soc., 2011, 133, 4625 CrossRef CAS PubMed.
- J. Huang, X. Wang, C. Zhan, Y. Zhao, Y. Sun, Q. Pei, Y. Liu and J. Yao, Polym. Chem., 2013, 4, 2174 RSC.
- M. M. Wienk, J. M. Kroon, W. J. H. Verhees, J. Knol, J. C. Hummelen, P. A. van Hal and R. A. J. Janssen, Angew. Chem., 2003, 115, 3493 CrossRef.
- S. Subramaniyan, H. Xin, F. S. Kim, N. M. Murari, B. A. E. Courtright and S. A. Jenekhe, Macromolecules, 2014, 47, 4199 CrossRef CAS.
- J. Gilot, M. M. Wienk and R. A. Janssen, Adv. Mater., 2010, 22, E67 CrossRef CAS PubMed.
- V. Tamilavan, K. H. Roh, R. Agneeswari, D. Y. Lee, S. Cho, Y. Jin, S. H. Park and M. H. Hyun, J. Polym. Sci., Polym. Chem. Ed., 2014, 52, 3564 CAS.
- L. Dou, J. You, Z. Hong, Z. Xu, G. Li, R. A. Street and Y. Yang, Adv. Mater., 2013, 25, 6642 CrossRef CAS PubMed.
- L. Ye, S. Zhang, L. Huo, M. Zhang and J. Hou, Acc. Chem. Res., 2014, 47, 1595 CrossRef CAS PubMed.
- J. Ren, X. Bao, L. Han, J. Wang, M. Qiu, Q. Zhu, T. Hu, R. Sheng, M. Sun and R. Yang, Polym. Chem., 2015, 6, 4415 RSC.
- J. Wang, M. Xiao, W. Chen, M. Qiu, Z. Du, W. Zhu, S. Wen, N. Wang and R. Yang, Macromolecules, 2014, 47, 7823 CrossRef CAS.
- D. Veldman, S. C. J. Meskers and R. A. J. Janssen, Adv. Funct. Mater., 2009, 19, 1939 CrossRef CAS.
- K. Vandewal, Z. Ma, J. Bergqvist, Z. Tang, E. Wang, P. Henriksson, K. Tvingstedt, M. R. Andersson, F. Zhang and O. Inganäs, Adv. Funct. Mater., 2012, 22, 3480 CrossRef CAS.
- H.-Y. Chen, J. Hou, S. Zhang, Y. Liang, G. Yang, Y. Yang, L. Yu, Y. Wu and G. Li, Nat. Photonics, 2009, 3, 649 CrossRef CAS.
- Y. Huang, L. Huo, S. Zhang, X. Guo, C. C. Han, Y. Li and J. Hou, Chem. Commun., 2011, 47, 8904 RSC.
- D. Liu, C. Gu, M. Xiao, M. Qiu, M. Sun and R. Yang, Polym. Chem., 2015, 6, 3398 RSC.
- R. S. Kularatne, F. J. Taenzler, H. D. Magurudeniya, J. Du, J. W. Murphy, E. E. Sheina, B. E. Gnade, M. C. Biewer and M. C. Stefan, J. Mater. Chem., 2013, 1, 15535 RSC.
- M. Z. F. Svensson, S. C. Veenstra, W. J. H. Verhees, J. C. K. Hummelen, J. M. O. Inganas and M. R. Andersson, Adv. Mater., 2003, 15, 988 CrossRef CAS.
- C.-Y. Mei, L. Liang, F.-G. Zhao, J.-T. Wang, L.-F. Yu, Y.-X. Li and W.-S. Li, Macromolecules, 2013, 46, 7920 CrossRef CAS.
- L. Huo and J. Hou, Polym. Chem., 2011, 2, 2453 RSC.
- J. Yuan, Y. Zou, R. Cui, Y.-H. Chao, Z. Wang, M. Ma, Y. He, Y. Li, A. Rindgen, W. Ma, D. Xiao, Z. Bo, X. Xu, L. Li and C.-S. Hsu, Macromolecules, 2015, 48, 4347 CrossRef CAS.
- J. Yuan, H. Dong, M. Li, X. Huang, J. Zhong, Y. Li and W. Ma, Adv. Mater., 2014, 26, 3624 CrossRef CAS PubMed.
- J. Wolf, F. Cruciani, A. El Labban and P. M. Beaujuge, Chem. Mater., 2015, 27, 4184 CrossRef CAS.
- Y. Zhang, S. K. Hau, H.-L. Yip, Y. Sun, O. Acton and A. K. Y. Jen, Chem. Mater., 2010, 22, 2696 CrossRef CAS.
- A. N. Y. Zou, P. Berrouard, B. B. R. d. A. c. S. Beaupré, Y. Tao and M. Leclerc, J. Am. Chem. Soc., 2010, 132, 5330 CrossRef PubMed.
- E. T. Hoke, K. Vandewal, J. A. Bartelt, W. R. Mateker, J. D. Douglas, R. Noriega, K. R. Graham, J. M. J. Fréchet, A. Salleo and M. D. McGehee, Adv. Energy Mater., 2013, 3, 220 CrossRef CAS.
- S. C. Price, A. C. Stuart, L. Yang, H. Zhou and W. You, J. Am. Chem. Soc., 2011, 133, 8057 CrossRef CAS.
- L. Huo, X. Guo, S. Zhang, Y. Li and J. Hou, Macromolecules, 2011, 44, 4035 CrossRef CAS.
- Z. G. Zhang, J. Min, S. Zhang, J. Zhang, M. Zhang and Y. Li, Chem. Commun., 2011, 47, 9474 RSC.
- M. Zhang, X. Guo, W. Ma, H. Ade and J. Hou, Adv. Mater., 2015, 27, 4655 CrossRef CAS PubMed.
- C. A. R. Frank, A. Arroyave and J. R. Reynolds, Org. Lett., 2012, 14, 6138 CrossRef PubMed.
- Y.-J. Cheng, J.-S. Wu, P.-I. Shih, C.-Y. Chang, P.-C. Jwo, W.-S. Kao and C.-S. Hsu, Chem. Mater., 2011, 23, 2361 CrossRef CAS.
- Q. Liu, X. Bao, S. Wen, Z. Du, L. Han, D. Zhu, Y. Chen, M. Sun and R. Yang, Polym. Chem., 2014, 5, 2076 RSC.
- W. Nie, C. M. Macneill, Y. Li, R. E. Noftle, D. L. Carroll and R. C. Coffin, Macromol. Rapid Commun., 2011, 32, 1163 CrossRef CAS PubMed.
- J. Hou, S. Zhang, Y. Yao, Li-M. Chen, J.-H. Li and Y. Yang, Macromolecules, 2008, 41, 6012 CrossRef CAS.
- C. Duan, A. Furlan, J. J. van Franeker, R. E. Willems, M. M. Wienk and R. A. Janssen, Adv. Mater., 2015, 27, 4461 CrossRef CAS PubMed.
- Y. F. Li and Y. P. Zou, Adv. Mater., 2008, 20, 2952 CrossRef CAS.
- L. J. Huo, S. Q. Zhang, X. Guo, F. Xu, Y. F. Li and J. H. Hou, Angew. Chem., Int. Ed., 2011, 50, 9697 CrossRef CAS PubMed.
Footnote |
| † Dakang Ding and Jiuxing Wang contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2016 |