Porous polymer microneedles with interconnecting microchannels for rapid fluid transport†
Liming Liu,
Hiroyuki Kai*,
Kuniaki Nagamine,
Yudai Ogawa and
Matsuhiko Nishizawa*
Department of Bioengineering and Robotics, Graduate School of Engineering, Tohoku University, 6-6-01 Aramaki Aoba, Aoba-ku, Sendai 980-8579, Japan. E-mail: nishizawa@biomems.mech.tohoku.ac.jp; kai@biomems.mech.tohoku.ac.jp
First published on 12th May 2016
Abstract
A porous polymer microneedle array with an average pore diameter of ∼1 μm was fabricated by photopolymerization of an acrylate monomer in the presence of a porogen within a mold. Porosity measurement and water absorption testing revealed that the pores are interconnected throughout the microneedle, which enabled rapid wetting of the microneedle by capillary action. The porosity of the polymer microneedles can be modulated by varying porogen content, and this allowed balancing fast water absorption speed and sufficient mechanical strength to penetrate a skin. The developed porous polymer microneedle array is a potentially versatile tool for rapid fluid transport from and into a body through the skin.
Introduction
The microneedle array, a two-dimensional array of submillimeter needles, has been studied as a minimally invasive tool for drug delivery.1–5 Microneedles penetrate skin to bypass the diffusion barrier of the stratum corneum, the outermost layer of skin, without pain or bleeding owing to their small height and diameter.6 In addition to drug delivery, microneedle arrays have also been used for sampling of interstitial fluid for the analysis of clinically relevant molecules.7–10 Transdermal monitoring of interstitial fluid below the skin could be a less invasive replacement for the monitoring of molecules in blood, as the concentration of molecules in interstitial fluid is often correlated with the concentration in blood.11–13Microneedles used for sampling interstitial fluid can be mainly divided into three types: hollow microneedles, hydrogel microneedles, and porous microneedles. Hollow microneedles have a cylindrical channel in the body of the microneedle. They are mostly made by photolithographic fabrication on silicon, and they have been applied to interstitial fluid sensing and drug delivery.14,15 Hydrogel microneedles swell upon insertion into the skin,16 and their uses for interstitial fluid sampling have been reported recently.17 Porous microneedles have a large volume of randomly distributed pores, and they are advantageous for fast wetting inside the microneedle body and sampling of interstitial fluid by capillary action. They are, however, intrinsically fragile because of a large volume of the void, and hard inorganic materials have been needed for making porous microneedles. Porous microneedles with continuous pores that can penetrate skin to sample interstitial fluid were made by sintering alumina particles.18 Other porous microneedles were made of partly porous structures to ensure mechanical strength, such as silicon microneedles with a porous tip19 and microneedles coated with porous calcium phosphate.20 Making porous microneedles with polymer materials such as hydrogels and plastics have various advantages e.g. biocompatibility, tunability of molecular structures, simple fabrication process by molding, etc. Nonetheless, fabrication of porous microneedles with polymer materials has been a challenge. Porous polymer microneedles were made by ultrasonic welding of poly(lactic acid) microparticles, but the fabricated microneedles were fragile and unable to penetrate the skin.21 Microneedles with a porous polymer were tried to fabricate by polymerization in the presence of a porogen (pore template).22 Although the synthesized polymer had high mechanical strength, the resultant microneedles had inconsistent geometry, which made them unsuitable for further applications.
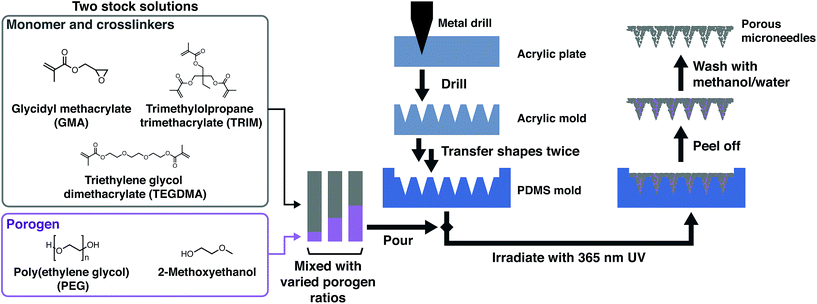
In this study, we developed a porous polymer microneedle array with continuous micropores that combines fast water absorption and well-defined geometry and high mechanical strength to penetrate skin (Fig. 1). We took advantage of continuously porous structures of a polymer monolith of poly(glycidyl methacrylate) (PGMA).23 The porous microneedle array made of a PGMA polymer monolith is synthesized by photopolymerization in the presence of poly(ethylene glycol) (PEG) solution in 2-methoxyethanol as a porogen within a mold (Fig. 2). By varying a porogen ratio, water absorption speed and mechanical strength were modulated to balance the trade-off between fast water absorption and skin penetration capability by sharp tips and sufficient mechanical strength.
Experimental
General methods
Polydimethylsiloxane (PDMS) pre-polymer and curing agent (Sylpot 194) were purchased from Dow Corning. Irgacure 184 was purchased from BASF. Trypan blue was purchased from Dojindo. All other chemicals were purchased from Sigma-Aldrich. All chemicals were used without further purification. Pig skin samples were purchased from Funakoshi.Fabrication of the mold of a microneedle array
An acrylic plate was drilled by a 5-axis CNC machine (PRODIA M45, Modia systems) with a tapered end mill (custom-made by DoCraft) and a NC file generated by Rhinoceros software to make a female mold. The dimensions of the microneedles on the array were designed with a height of 700 μm and a diameter of 350 μm, and the array was composed of 6 × 6 conical needles with an interval of 1 mm. These dimensions were chosen for the sake of minimal invasiveness according to the previous reports. A male polydimethylsiloxane (PDMS) mold was made by pouring a PDMS solution (Sylpot 194, Dow Corning, pre-polymer/curing agent with 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 w/w ratio, vacuum degassed for 1 hour at room temperature) into the acrylic mold, degassed under vacuum three times, and cured at 70 °C for 30 minutes or more. The male PDMS mold was then gently peeled off from the female acrylic mold. The surface of the male PDMS mold was treated with 1H,1H,2H,2H-perfluorooctyltrichlorosilane for more than 6 hours. PDMS solution was poured into the surface-treated male mold, degassed at room temperature, cured at 70 °C, and peeled off from the male mold to obtain a female PDMS mold.
1 w/w ratio, vacuum degassed for 1 hour at room temperature) into the acrylic mold, degassed under vacuum three times, and cured at 70 °C for 30 minutes or more. The male PDMS mold was then gently peeled off from the female acrylic mold. The surface of the male PDMS mold was treated with 1H,1H,2H,2H-perfluorooctyltrichlorosilane for more than 6 hours. PDMS solution was poured into the surface-treated male mold, degassed at room temperature, cured at 70 °C, and peeled off from the male mold to obtain a female PDMS mold.
Fabrication of porous microneedles
A porous microneedle array made of a polymer monolith was synthesized according to the literature23 in the mold fabricated above. Trimethylolpropane trimethacrylate (TRIM) and triethylene glycol dimethacrylate (TEGDMA) were used as a crosslinker for PGMA. Polyethylene glycol (PEG) solution in 2-methoxyethanol is used as a porogen that can be easily removed afterward. First, two stock solutions were prepared for the easier control of precise porogen ratios. A porogen stock solution was prepared by dissolving PEG (20 g, 10 kDa, 2.0 mmol) in 2-methoxyethanol (100 g) at 50 °C. It is important to ensure the solution is transparent before use. If not so, the solution was slightly heated until the solution became clear. A monomer stock solution was prepared by mixing the monomer glycidyl methacrylate (GMA, 10 mL; 73.3 mmol, 1 equiv.), crosslinker TRIM (6.18 mL; 19.4 mmol, 0.26 equiv.), and crosslinker TEGDMA (15.7 mL; 57.6 mmol, 0.79 equiv.). This formulation made the quasi-equimolar mixture of a monomer and crosslinkers, and crosslinkers have 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 TRIM/TEGDMA molar ratio. The monomer stock solution was stored at −15 °C. Prior to synthesis, the solution was quickly heated to room temperature with a 37 °C water bath and mixed evenly. Next, a photoinitiator Irgacure 184 (0.10 g, 1 wt% to the monomer) was put into the mixture of the monomer and porogen stock solutions with a desired porogen ratio (Table S1†), where a porogen ratio refers to the volume ratio of a porogen stock solution in the entire mixture. After putting the mixture into a microneedle mold, the mold with the mixture was put under vacuum for 1 hour to make sure that bubbles were removed and all the cavities of the microneedle mold were filled with the solution. The mold was put under 365 nm UV light for 1 hour to polymerize the solution to make a microneedle array. After dissolving the porogen PEG in the microneedles with methanol/water (1
3 TRIM/TEGDMA molar ratio. The monomer stock solution was stored at −15 °C. Prior to synthesis, the solution was quickly heated to room temperature with a 37 °C water bath and mixed evenly. Next, a photoinitiator Irgacure 184 (0.10 g, 1 wt% to the monomer) was put into the mixture of the monomer and porogen stock solutions with a desired porogen ratio (Table S1†), where a porogen ratio refers to the volume ratio of a porogen stock solution in the entire mixture. After putting the mixture into a microneedle mold, the mold with the mixture was put under vacuum for 1 hour to make sure that bubbles were removed and all the cavities of the microneedle mold were filled with the solution. The mold was put under 365 nm UV light for 1 hour to polymerize the solution to make a microneedle array. After dissolving the porogen PEG in the microneedles with methanol/water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 volume), microneedles with porous structures were obtained (Fig. 3).
1 volume), microneedles with porous structures were obtained (Fig. 3).
Scanning electron microscopy
A specimen of a microneedle array was coated with gold by a sputtering system (350S-C, Anelva). Scanning electron microscopy (SEM) was carried out on Keyence VE 9800 microscope operated at 1 kV. Images were recorded from randomly chosen areas.Porosity measurement
For calculating the porosity (pore volume ratio), the pore volume was determined according to the method previously reported.24 The porous microneedles were immersed into a fluorescein solution (10 μg mL−1) for 1 hour. Subsequently, the microneedles were taken out from the solution and immersed into the buffer solution with shaking for 4 hours. The fluorescein concentration in the buffer was determined on a microplate reader (Gemini XPS, Molecular Devices), from which the pore volume (Vpore) was calculated. The porosity was calculated by eqn (1)| (Porosity) = Vpore/(WPMN/ρcontr. + Vpore) | (1) |
Water absorption speed
To investigate how fast the porous microneedles can transport fluid from a skin, a piece of water sensor paper (Sweat Checker, Life Care Giken) was placed on the microneedle array. The water sensor paper turns blue when it absorbs water and dissolves blue dye stored inside. The porous microneedle array with the water sensor on the top was gently inserted to a 0.75% agarose gel colored with water-based blue ink. Water absorption speed was evaluated by the time it takes for the sweat checker to change its color. Water absorption speed of porous microneedle arrays with varied porogen ratios was measured.Fracture strength
To study the mechanical strength of the porous microneedles, fracture forces were evaluated with forces of the two orthogonal directions, compression force and transverse force (Fig. S2†). For fracture testing by compression force, a metal jig was pushed from above the tip of a microneedle at the rate of 4 mm min−1. Displacement and force on a metal jig were simultaneously recorded by a force gauge (FSA-1KE-5N, Imada), and fracture force was determined as the maximum force before the sharp drop of force. For the transverse force, the microneedle array (6 × 1) was attached vertically on a metal base, and a metal jig was pushed from the side at the rate of 4 mm min−1.Pig skin penetration
The porous microneedles were attached to the tip of a force gauge (ZTS-200N, Imada), and pushed onto a piece of the back skin of a porker with the vertical force of 0.5 N per needle and a contact speed of 40 mm min−1 using a motorized test stand (EMX-1000N-FA, Imada). The force was continuously measured during the contact, and when the force reached 0.5 N per needle, it was kept constant by adjusting the position of the force gauge for 1 minute. The pig skin was immediately stained with trypan blue (0.4 wt% solution in water) for more than 3 hours, followed by washing residual trypan blue on the skin surface with water. The pig skin sample was imaged by an optical microscope (KH-1300, HIROX).Results and discussion
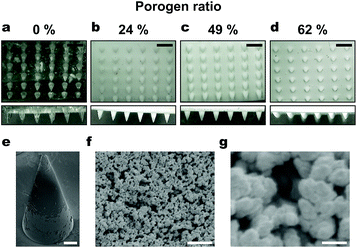
Porous microneedle arrays with different porogen ratios were fabricated (Table S1†). The microneedles made with no porogen were transparent, while those with a porogen are opaque, which is due to light scattering caused by the pores. The fabricated porous microneedle array (6 × 6, porogen ratio 39%) has a center to center spacing of ∼950 μm, a base diameter ∼350 μm, a tip diameter ∼30 μm, a height ∼700 μm, measured by optical microscopy (Fig. 3(a)–(d)). These are common dimensions for transdermal microneedle arrays, and known to be minimally invasive. SEM revealed that the pore size was ∼1 μm (Fig. 3(f) and (g)), and the pore size was constant for varied porogen content, which is in agreement with the previous report.16 We found that when the porogen ratio is higher, microneedles tend to have blunter tips (Fig. 3(a) and S1†). A hypothesis to explain this trend could be that PGMA and porogen undergo macroscopic phase separation at the interface with a PDMS mold when the porogen ratio is high. In addition, microneedles made with a porogen ratio no less than 76% are so fragile that they broke when peeled off from the mold.We calculated the porosity of the porous microneedle arrays with different porogen ratios (eqn (1)). Fig. 4 shows the theoretical and experimental porosity of the porous microneedles with different porogen ratios. Theoretical porosity is defined as the volumetric ratio of the porogen (PEG in 2-methoxyethanol) based on the assumptions that all PEG and 2-methoxyethanol coexist as a porogen in a solution phase after polymerization and all the porogen is removed by washing after polymerization. The ratio between experimental and theoretical porosity corresponds to a ratio of pores that are continuous to the microneedle surface, where porogen can be removed by washing. For the porogen ratio no less than 56%, the ratio between experimental and theoretical porosity was close to unity, whereas below the porogen ratio of 36%, the experimental value became significantly lower than the theoretical value as a porogen ratio was decreased. This result suggests that for lower porogen ratios, the part of porogen phases exists as isolated regions inside the microneedle body. Above a certain threshold, the porogen phases are more likely to be connected to each other. Appearance of the threshold is reasonable because the formation of continuous pores can be thought as a percolation phenomenon.25
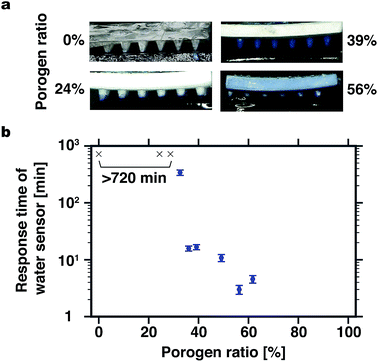
Speed of sampling of fluid through the microneedle is an important parameter, as it determines the response time of the whole sensing system. We used an agarose gel containing water-based ink as a simple model system that gives a controlled environment for testing water absorption speed of the microneedles. When the porogen ratio was no more than 29%, the water sensor did not change its color even after 12 hours (Fig. 5(b)). When the porogen ratio was gradually increased, the water absorption speed suddenly increased at the porogen ratio of 36%, and this result corresponded well with the emergence of continuous pores as the porosity measurement above suggested. Mass transfer in the porous microneedles when they are applied to a skin can be thought as a two-step process: initial wetting of the micropores by capillary action, followed by diffusion of molecules inside the micropores filled with liquid. A high pore volume ratio with a large number of interconnected micropores of the developed porous microneedles should be beneficial for both of wetting and diffusion. In addition to the response of the water sensor, we also observed that water-based ink can be pulled up to the root of the microneedles within a few seconds (ESI Video†). This suggests that it might be even possible for the developed porous microneedles to sample interstitial fluid almost instantaneously due to fast capillary action. It should be noted that the transport of interstitial fluid from the actual skin may occur differently, because the water content in the skin tissue, fluid pressure, viscosity, etc. are different. A preliminary experiment with the porous microneedles with the same water sensor that was applied to a piece of pig skin did not yield a reproducible response of the water sensor. Experiment with live animal models is currently planned with more sensitive probes for water and biological molecules.
Skin penetration capability of a microneedle array is essential for sampling interstitial fluid from inside the skin. There are a variety of factors that influence the penetration efficiency, such as the insertion force, the insertion speed, the age of the skin, etc.26 The insertion force was determined by a preliminary experiment on the microneedles with no porogen with the constant insertion speed of 40 mm min−1 but with different insertion forces (Fig. S3†). The penetration efficiency increased with the increasing insertion force, and when the insertion force was higher than 0.5 N per needle, the penetration efficiency was higher than 80%. We used an aged pig skin that requires high insertion force than a young pig skin (similarly to the human skin),26 and hairs on the pig skin prevent the insertion of microneedles. From these considerations, we adopted the total applied force of 18 N (1.8 kgf, which is easy to apply by human fingers) for the microneedle array with 36 microneedles, and measured the penetration efficiency for different porogen ratios (Fig. 6). When the porogen ratio was no more than 49%, the penetration efficiency was more than 80%, which means that the porous microneedles with a porogen ratio no more than 49% had enough strength to penetrate skin. Moreover, no visible breakage of the microneedles of those porogen ratios was observed after penetration. When the porogen ratio was larger than 49%, however, a significant decrease in the penetration efficiency was observed. This can be attributed to blunt tips (Fig. 3(d)) and the low transverse fracture force of the microneedles made with high porogen ratios (Fig. S2†).
Conclusions
We developed the porous polymer microneedles with a simple fabrication method. The porous microneedles have an average pore diameter about 1 μm. With the optimized porogen ratio, the fabricated microneedles combine the fast water absorption speed and sufficient strength to penetrate the skin. The fabricated microneedle array is a potentially versatile tool for sampling of interstitial fluid as well as drug delivery. While PGMA has the unique capability for the surface modification on an epoxide group,27 a wide range of monomers that can form porous polymer monoliths28 can be used to tailor the properties of microneedles. The large surface area inside the microneedles is advantageous for sensing of interstitial fluid by surface modification with sensor molecules.Acknowledgements
We thank Taro Kondo for his assistance for porosity measurement. This work was partly supported by Center of Innovation Program (COI), Creation of Innovation Centers for Advanced Interdisciplinary Research Area Program from Japan Science and Technology Agency, JST, Regional Innovation Strategy Support Program “Knowledge-based Medical Device Cluster/Miyagi Area”, and by Grand-in-Aid for Scientific Research A (25246016) and Challenging Exploratory Research (K15K13315) from the Ministry of Education, Culture, Sports, Science and Technology, Japan.References
- M. R. Prausnitz, Adv. Drug Delivery Rev., 2004, 56, 581–587 CrossRef CAS PubMed
.
- K. van der Maaden, W. Jiskoot and J. Bouwstra, J. Controlled Release, 2012, 161, 645–655 CrossRef CAS PubMed
.
- T.-M. Tuan-Mahmood, M. T. C. McCrudden, B. M. Torrisi, E. McAlister, M. J. Garland, T. R. R. Singh and R. F. Donnelly, Eur. J. Pharm. Sci., 2013, 50, 623–637 CrossRef CAS PubMed
.
- D. V. McAllister, P. M. Wang, S. P. Davis, J.-H. Park, P. J. Canatella, M. G. Allen and M. R. Prausnitz, Proc. Natl. Acad. Sci. U. S. A., 2003, 100, 13755–13760 CrossRef CAS PubMed
.
- S. P. Sullivan, D. G. Koutsonanos, M. del Pilar Martin, J. W. Lee, V. Zarnitsyn, S.-O. Choi, N. Murthy, R. W. Compans, I. Skountzou and M. R. Prausnitz, Nat. Med., 2010, 16, 915–920 CrossRef CAS PubMed
.
- S. Kaushik, A. H. Hord, D. D. Denson, D. V. McAllister, S. Smitra, M. G. Allen and M. R. Prausnitz, Anesth. Analg., 2001, 92, 502–504 CAS
.
- S. Zimmermann, D. Fienbork, B. Stoeber, A. W. Flounders and D. Liepmann, in TRANSDUCERS, Solid-State Sensors, Actuators and Microsystems, 12th International Conference on, 2003, vol. 1, pp. 99–102 Search PubMed
.
- P. M. Wang, M. Cornwell and M. R. Prausnitz, Diabetes Technol. Ther., 2005, 7, 131–141 CrossRef CAS PubMed
.
- D. A. Muller, S. R. Corrie, J. Coffey, P. R. Young and M. A. Kendall, Anal. Chem., 2012, 84, 3262–3268 CrossRef CAS PubMed
.
- B. Chua, S. P. Desai, M. J. Tierney, J. A. Tamada and A. N. Jina, Sens. Actuators, A, 2013, 203, 373–381 CrossRef CAS
.
- M. Brunner and H. Derendorf, Trends Anal. Chem., 2006, 25, 674–680 CrossRef CAS
.
- E. Cengiz and W. V. Tamborlane, Diabetes Technol. Ther., 2009, 11, S–11–S–16 CrossRef PubMed
.
- T. K. L. Kiang, U. O. Häfeli and M. H. H. Ensom, Clin Pharmacokinet, 2014, 53, 695–730 CrossRef CAS PubMed
.
- E. V. Mukerjee, S. D. Collins, R. R. Isseroff and R. L. Smith, Sens. Actuators, A, 2004, 114, 267–275 CrossRef CAS
.
- S. P. Davis, W. Martanto, M. G. Allen and M. R. Prausnitz, IEEE Trans. Biomed. Eng., 2005, 52, 909–915 CrossRef PubMed
.
- R. F. Donnelly, T. R. R. Singh, M. J. Garland, K. Migalska, R. Majithiya, C. M. McCrudden, P. L. Kole, T. M. T. Mahmood, H. O. McCarthy and A. D. Woolfson, Adv. Funct. Mater., 2012, 22, 4879–4890 CrossRef CAS PubMed
.
- E. Caffarel-Salvador, A. J. Brady, E. Eltayib, T. Meng, A. Alonso-Vicente, P. Gonzalez-Vazquez, B. M. Torrisi, E. M. Vicente-Perez, K. Mooney, D. S. Jones, S. E. J. Bell, C. P. McCoy, H. O. McCarthy, J. C. McElnay and R. F. Donnelly, PLoS One, 2015, 10, e0145644 Search PubMed
.
- M. Verhoeven, S. Bystrova, L. Winnubst, H. Qureshi, T. D. de Gruijl, R. J. Scheper and R. Luttge, Microelectron. Eng., 2012, 98, 659–662 CrossRef CAS
.
- J. Ji, F. E. H. Tay, J. Miao and C. Iliescu, J. Micromech. Microeng., 2006, 16, 958 CrossRef CAS
.
- M. Shirkhanzadeh, J. Mater. Sci.: Mater. Med., 2005, 16, 37–45 CrossRef CAS PubMed
.
- J.-H. Park, S.-O. Choi, R. Kamath, Y.-K. Yoon, M. G. Allen and M. R. Prausnitz, Biomed. Microdevices, 2006, 9, 223–234 CrossRef PubMed
.
- L. Humrez, M. Ramos, A. Al-Jumaily, M. Petchu and J. Ingram, J. Polym. Res., 2010, 18, 1043–1052 CrossRef
.
- J. Courtois, E. Byström and K. Irgum, Polymer, 2006, 47, 2603–2611 CrossRef CAS
.
- K. van der Maaden, R. Luttge, P. J. Vos, J. Bouwstra, G. Kersten and I. Ploemen, Drug Delivery Transl. Res., 2015, 5, 397–406 CrossRef CAS PubMed
.
- S. A. Saba, M. P. S. Mousavi, P. Bühlmann and M. A. Hillmyer, J. Am. Chem. Soc., 2015, 137, 8896–8899 CrossRef CAS PubMed
.
- O. Olatunji, D. B. Das, M. J. Garland, L. Belaid and R. F. Donnelly, J. Pharm. Sci., 2013, 102, 1209–1221 CrossRef CAS PubMed
.
- M. Benaglia, A. Alberti, L. Giorgini, F. Magnoni and S. Tozzi, Polym. Chem., 2013, 4, 124–132 RSC
.
- F. Svec, J. Chromatogr. A, 2010, 1217, 902–924 CrossRef CAS PubMed
.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra07882f |
| This journal is © The Royal Society of Chemistry 2016 |