Co nanoparticles embedded in a 3D CoO matrix for electrocatalytic hydrogen evolution†
Xiangye Liua,
Chenlong Donga,
Wujie Donga,
Xin Wanga,
Xiaotao Yuana and
Fuqiang Huang*ab
aBeijing National Laboratory for Molecular Sciences and State Key Laboratory of Rare Earth Materials Chemistry and Applications, College of Chemistry and Molecular Engineering, Peking University, Beijing 100871, P. R. China. E-mail: huangfq@pku.edu.cn
bCAS Key Laboratory of Materials for Energy Conversion and State Key Laboratory of High Performance Ceramics and Superfine Microstructures, Shanghai Institute of Ceramics, Chinese Academy of Sciences, Shanghai 200050, P. R. China
First published on 13th April 2016
Abstract
Earth-abundant and highly efficient electrocatalysts for the hydrogen evolution reaction (HER) are desired for hydrogen production from water-splitting. Here, Co nanoparticles were embedded in the 3D CoO matrix via a template-free method, including cobalt hydroxy-carbonate nanowire arrays grown on Ni foam and the following non-contact Al-reduction process. The as-prepared 3D hierarchical structured Co/CoO nanowires possess good charge transfer and mass transport properties, and a synergistic effect at the Co/CoO interface can hugely facilitate the HER kinetics. A suitable balance between Co and CoO in the catalyst is crucial for high catalytic activity. And the optimal Co/CoO array exhibited outstanding HER activities in 1 M NaOH, achieving nearly zero onset potential, and a current density of 100 mA cm−2 with a small overpotential of 167 mV. They also showed good long-term stabilities. This hybrid Co/CoO nanowire array is a promising material for large-scale hydrogen production from water-splitting.
Introduction
Hydrogen generation from electrochemical water splitting has aroused intense attention for clean and sustainable energy applications. Efficient electrocatalysts are required to reduce the overpotential of the hydrogen evolution reaction (HER).1,2 So far, the most active HER catalysts are Pt-based metals, which can generate large cathodic current densities for HER at low overpotentials.3,4 But their scarcity and high cost impede their large-scale practical application. Although many kinds of earth-abundant HER catalysts have exhibited high activities approaching Pt in acid solutions, including transition metal chalcogenides,5–8 phosphide,9–12 carbides13–15 and nitrides,13,15,16 all of them cannot operate satisfactorily in an alkaline electrolyte.3 The alkaline HER electrocatalysts are more significant for the industrial water-alkali and chlor-alkali electrolyzers.Ni, Co and their alloys are the typical alkaline electrocatalysts, but they are still far from optimization for their high HER overpotentials and large Tafel slopes.1,17–20 Previous works have demonstrated that enhanced HER activity can be achieved by metal–metal oxide/carbon hybrids (Ni–NiO or Co–CoOx nano-particles supported on carbon nanotube or N-doped carbon), owing to an synergistic effect that HO–H bonds are first efficiently cleaved by oxide species and the derived hydrogen intermediates (H*) are then proficiently converted to H2 by the nearby metallic nano-particles.21–25 For electrochemical measurements and practical applications, these powder catalysts have to be immobilized on electrode surfaces by polymer binders. But the polymer binders usually has many disadvantages, such as increasing the series resistance, blocking active sites, inhibiting diffusion, and showing unreliable stabilities.11 To solve this problem, binder-free electrodes with active phases directly grown on current collectors have been developed, such as Ni/NiO and Co/Co3O4 core/shell nanosheet on Ni foams.26,27 While these three dimensional (3D) structured binder-free electrodes have not outperformed the powder analogues, which may result from their undesirable microstructures with the nanosheets of Ni/NiO or Co/Co3O4 randomly stacked on the Ni foams. That leads to unefficient mass transport and charge transfer. So a 3D aligned porous metal/metal oxide array may be an optimal structure for water-alkaline HER, because it affords large metal/metal oxide interface, excellent bubble management and charge transfer properties. Our recent work has demonstrated that a well-aligned porous Ni/NiO nanosheet array can exhibit excellent HER activities approaching to the Pt/C.28 Compared to the nanosheet structure, the one-dimensional nanowire should be more favorable considering its vectorial electron-transport property. However, it remains a challenge to fabricate such 3D metal/metal oxide nanowire arrays for HER.
Here, we demonstrated the non-contact Al reduction method can exquisitely convert the cobalt hydroxy-carbonate precursors to desired hybrid Co/CoO nanowire arrays. Co nanoparticles were embedded in the CoO matrix to form a 3D hierarchical structure. The Co/CoO arrays exhibit enhanced HER activities compared to the metallic Co and CoO analogues. They achieved nearly zero onset potential, and a current density of 100 mA cm−2 with a small overpotential of 167 mV, which is just 95 mV larger than Pt/C at the same current density. They also exhibited good durability.
Experimental
Synthesis of the hybrid Co/CoO nanowire array electrode
All chemicals used in this work were analytical reagent grade and commercially available, and used without further purification.Firstly, the Co2(OH)2CO3·nH2O nanowire arrays were grown on nickel foams by a facile hydrothermal method according to the previous work.29 Briefly, 4 mmol Co(NO3)2·6H2O, 20 mmol urea and 8 mmol NH4F were completed dissolved in 80 mL deionized water under magnetic stirring. A nickel foam substrate was vertically immersed in the solution after chemically cleaned with acetone, methanol, and deionized water. The resulting solution with nickel foams were then sealed in a 100 mL Teflon-lined stainless steel autoclave, and maintained at 120 °C for 6 h, followed by rinsing the nickel foams with deionized water and dried at 60 °C for 12 h. Powder products in the solution were collected by centrifugation. Secondly, the as-grown samples both on the nickel foams and powder from the solution were treated with a special Al reduction process to obtain the Co/CoO electrode and Co/CoO powder, according to the previous work of our group.30 Specifically, the as-grown samples and aluminum powder or aluminum ingot were separately placed in a two zone tube furnace and then evacuated to a base pressure ∼ 4 Pa, then the aluminum was heated at 850 °C and samples were heated at 300 °C or 400 °C, for 2 h.
Synthesis of the CoO and Co electrodes
Firstly, the Co2(OH)2CO3·nH2O nanowire arrays were grown on nickel foams and Co2(OH)2CO3·nH2O powder was collected with the same method described above. Then they were annealed in Ar at 350 °C for 2 h to obtain the CoO electrode and the CoO powder. Similarly, the samples were annealed in H2/Ar (H2 10%) at 260 °C for 2 h to obtain the Co electrode and the Co powder.Material characterization
The samples were characterized by powder X-ray diffraction (XRD) using a Bruck D2 X-ray diffractometer with Cu Kα radiation (k = 1.5418 Å) at a scanning rate of 0.1° min−1 in the 2θ range of 20–85°. The chemical compositions, the sizes and morphologies, and the microstructures of the samples were characterized by field-emission scanning electron microscopy (FE-SEM, JSM-6330F), transmission electron microscopy (TEM, JEM2010-HR), and X-ray photoelectron spectroscopy (XPS, ESCALab 250). Nitrogen sorption isotherms were measured using a Micromeritics ASAP 2020 analyzer at −196 °C. The weight of the sample used in the measurement was 100–200 mg. The Brunauer–Emmett–Teller (BET) method was utilized to calculate the specific surface area (SBET) using the adsorption data at p/p0 = 0.06–0.30.Electrochemical measurements
The electrochemical tests were carried out in a conventional three electrode electrochemical cell by using a CHI660E. A Hg/HgO (1 M NaOH) and Pt wire were used as the reference electrode and the counter electrode, respectively. The as prepared samples on Ni foams were directly used as the working electrodes. All the electrochemical measurements of catalysts were measured in 1 M NaOH solution (pH = 14) after purging the electrolyte with N2 gas for 30 min at 25 °C. The potential, measured against a Hg/HgO (1 M NaOH) electrode, was converted to the potential versus the reversible hydrogen electrode (RHE) according to Evs. RHE = Evs. Hg/HgO + EHg/HgOθ + 0.059pH − iR, and all the polarization curves were iR corrected using the data of the electrochemical impedance spectroscopy (EIS) measurements. The EIS was measured between the frequency of 0.01 Hz to 105 Hz with an amplitude of 0.005 V at different potentials. Linear sweep voltammetry was carried out at 1 mV s−1 for the polarization curves. Chronoamperometry was measured under overpotentials of 60 and 115 mV, respectively.Results and discussion
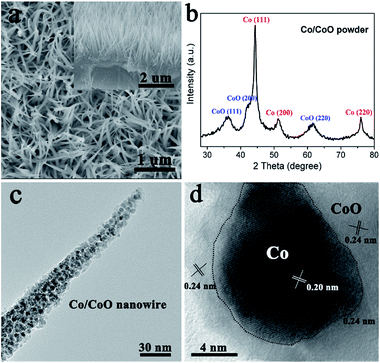
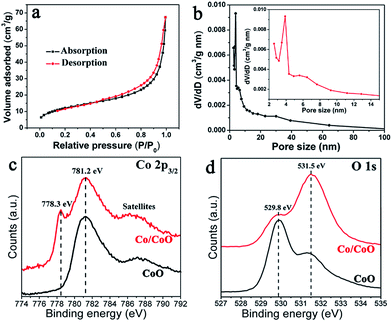
Oriented Co2(OH)2CO3·nH2O nanowire arrays on nickel foams were first prepared by a hydrothermal method,29 confirmed by XRD and SEM results (Fig. S1†). Then they were treated by a non-contact Al-reduction process in a vacuum two-zone tube furnace,31 in order to obtain the desired nanowire arrays of nanoscale Co/CoO. Fig. 1a shows the scanning electron microscopy (SEM) image and the cross-section analysis of the Co/CoO prepared using Al powder at 300 °C. These results indicate the Co/CoO on Ni foam remains the aligned array structures, and each Co/CoO unit is vertically contacted on the substrate. The XRD result of the corresponding Co/CoO powder reveals the co-existing of the metallic cobalt and the cobalt oxide (Fig. 1b). TEM image shows the Co/CoO nanowire consists of opaque nanodots uniformly interconnected by the light fine plates, with an uniform size of ∼10 nm (Fig. 1c). The opaque nanodots are identified to be metallic Co, seen from the interplanar spacings of 0.20 nm for Co (111) planes in their lattice-resolved HRTEM image (Fig. 1d). The light plates surrounding the metals are identified to be CoO species, confirmed by the larger interplanar spacings, about 0.24 nm for CoO (111) planes (Fig. 1d). The Co and CoO are well matched at their interface, where their lattice fringes are overlapped (Fig. 1d). The homogeneous Co/CoO structure affords large interface area for electrocatalysis. Moreover, numerous inter-particle mesopores with a size ranged from 2 to 5 nm in these Co/CoO units can be clearly seen (Fig. 1c). These pores are originated from the release of water and oxygen molecules during the reduction process. It is well known that the nanoporous structures are important to facilitate the mass transport of electrolytes and gas bubbles within the electrodes, and will also provide large electroactive area for the electrochemical reaction.32The pore structures of mesoporous Co/CoO were further examined by N2 adsorption–desorption measurements. The isotherms with H3 hysteresis loops were observed for our Co/CoO (Fig. 2a), which are commonly observed for the mesoporous transitional metal oxides.33–35 The pore size distributions calculated from the desorption isotherms by the BJH method are shown as a plot of the relative pore filling versus pore size in Fig. 2b. It indicates that mesopores with size of 2–50 nm are in the Co/CoO nanowires, with the main peak at 3.9 nm. This is in accordance with the TEM results. The nanoporous structures are important to facilitate the mass transport of electrolytes and gas bubbles, and will also provide large electroactive area for the electrochemical reaction.28
X-ray photoelectron spectroscopy (XPS) study was performed in order to further study the chemical compositions of Co/CoO nanocomposites, shown in Fig. 2. Both the CoO and Co/CoO nanocomposite show the main CoII 2p3/2 peaks at 781.2 eV,36 accompanied with the broad satellites. Contrastively, the spectrum of Co/CoO nanocomposite shows an additional metallic Co peak at 778.3 eV, seen from Fig. 2c. This result demonstrates the co-existing of CoO and metallic Co in our hybrid Co/CoO catalysts. The O 1s XPS spectra are shown in Fig. 2d. The CoO sample has two peaks, one at 529.85 eV attributed to the lattice O2− and one small shoulder at 531.1 eV from the hydroxyl groups on the surface. For the Co/CoO nanocomposite, the intensity of the hydroxyl groups at 531.1 eV increased dramatically, indicating that many more hydroxyl groups were created on the CoO layers. This phenomenon is perhaps related to the oxygen-defective nature in the Co/CoO sample resulting from the Al-reduction process,37 consistent with the observations for reduced metal oxides according to previous studies.27,28,38,39 The oxygen-defect can afford active centers to efficiently cleave water molecules.28,40 And these surface hydroxyl groups could make more hydrophilic electrode surface, facilitating the electrolyte to infiltrate the nanopores in the electrode materials. Furthermore, surface hydroxyl groups could directly participate in the catalytic reactions of water cleavation.28,41
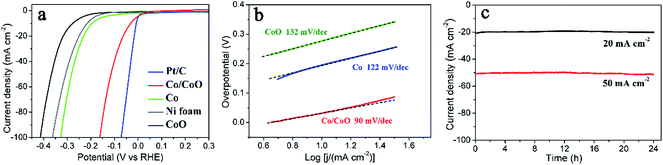
The HER catalytic property of the hybrid Co/CoO nanowire arrays were measured in a three-electrode configuration in 1 M NaOH with a scan rate of 1 mV s−1. Metallic Co electrode, CoO electrode and Pt/C electrode were also investigated under the same condition for comparison. The pure Co electrode and CoO electrode were obtained from the same precursor Co2(OH)2CO3·nH2O/Ni foam annealed in H2/Ar and Ar atmosphere, respectively. The corresponding XRD of powder samples are shown in Fig. S2.† Fig. 3a shows the electro-polarization curves. The Co/CoO array exhibits excellent HER activity with near-zero onset overpotential as same as the Pt/C, largely better than the Co electrode (onset overpotential of 150 mV) and CoO electrode (onset overpotential of 240 mV). And Co/CoO array requires overpotentials of 60, 115 and 167 mV to afford current densities of 20, 50 and 100 mA cm2, which are just 43, 77 and 95 mV larger than the Pt/C electrode, respectively. And these overpotentials are also smaller than that of other reported HER electrodes at the same current density measured in alkaline solutions, including RANEY®,19 Ni–Mo alloy,17 Ni(OH)2 modified Ni metal,23 and comparable to the CNT-Ni/NiO.22 Our Co/CoO array electrode even exhibited smaller HER overpotentials than those star catalysts measured in acid electrolytes, such as NiP,10,42 CoP,43 CuP,12 CoSe2,8 MoS2,44 WS2,45 Mo2N13 and Mo2C.14 Tafel slopes of the samples were calculated from their corresponding polarization curves. Co/CoO array exhibits a Tafel slope of 90 mV dec−1 in the overpotential region of 0–100 mV, smaller than that of Co electrode (122 mV dec−1) and CoO electrodes (132 mV dec−1), shown in Fig. 3b. The small Tafel slope means the fast HER kinetics and the consequent high current density at a lower overpotential. Generally, the alkaline HER follows a two-step mechanism: the first Volmer step where water is cleaved to generate the reactive intermediate H* (2H2O + M + 2e− ⇆ 2M–H* + 2OH−), followed by either the Heyrovsky step (H2O + M–H* + e− ⇆ H2 + M + OH−) or the Tafel step (2M–H* ⇆ H2 + 2M). When Volmer reaction is the rate-determining step (r.d.s.), the Tafel slope should be above 100 mV dec−1, and when the r.d.s. is Heyrovsky reaction or Tafel reaction, the Tafel slope should be about 40 mV dec−1 or 30 mV dec−1, respectively.46 The Tafel slopes of the present Co-based electrodes lie in the range of 100 mV dec−1 to 140 mV dec−1, evidencing the r.d.s. is the first Volmer step. Water dissociation on the pure Co surface and H* absorption on CoO surface are all kinetically difficult, which accounts for the larger Tafel slopes and higher HER onset overpotentials of the Co and CoO electrodes. For the Co/CoO electrode, the HO–H bonds can be efficiently cleaved at the interface of Co/CoO, resulting in the formation of OH* on CoO and H* on the neighboured Co site. The as-derived H* are then proficiently converted to H2 by the Co nanoparticles.21 Thus a faster HER kinetics can be expected on the Co/CoO hybrids. Additionally, the porous array structures and the highly conductive Co nanoparticles can further benefit the charge transfer and mass transport (inwards diffusion of electrolyte and outwards diffusion of gas bubbles). Furthermore, the specific surface areas of Co powder, CoO powder and the Co/CoO powder were calculated to be as 39.5, 90.9 and 43.4 m2 g−1. That is to say the surface area of Co/CoO nanocomposite is comparable to that of the metal Co nanoparticles, and even smaller than that of CoO sample. So the enhanced HER activity of Co/CoO has little relationship with increased surface area.
The durabilities of the hybrid Co/CoO nanowire array was also examined using chronoamperometry method. Fig. 3c shows the time-dependent current density curves under static overpotentials of 60 and 115 mV, respectively. After a long period (24 h), the current densities (20 and 50 mA cm−2) show barely degradations with the retention above 90%. Additionally, the nanowire-array structure of our Co/CoO is well maintained after the long-period HER measurement, seen from the corresponding SEM image (Fig. S3†). Thus, this Co/CoO electrode has superior long-term stability, which is crucial for real alkali-electrolyzer operations.
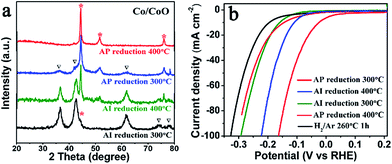
To study the affect of different components in Co/CoO catalyst on its HER activity, Co/CoO nanowire arrays with different ratio of Co/CoO were prepared by controlling reduction degree of the cobalt hydroxy-carbonate precursors, and they were studied by linear sweep voltammetry. The annealing temperature and the surface area of the Al reductant have significant influences on the reduction degree. Al powder (AP) with a higher surface area and Al ingot (AI) with a lower one were respectively employed in our experiments. Four Co/CoO samples were prepared by AI-reduction at 300 °C (sample I), AI-reduction at 400 °C (sample II), AP-reduction at 300 °C (sample III) and AP-reduction at 400 °C (sample IV), respectively. Fig. 4a shows the XRD results of the four Co/CoO powders. Only a weak cobalt XRD peak is observed for sample I, indicating a slight component of metallic cobalt. With improved reduction degree, increased ratio of Co/CoO is obtained for sample II and sample III, and only cobalt XRD peaks are observed in sample IV. From sample I to sample II to sample III, the HER performances are more and more improved, seen from the decreasing onset overpotentials and the increasing current densities at large overpotentials, as shown in Fig. 4b. The sample III exhibits the best HER performance, indicating a higher percentage of metallic cobalt is better for HER catalysis. However, the sample IV, with a further greater reduction degree, shows a degraded HER performance. Although it displays a relatively low onset overpotential (close to sample II), its current densities at high overpotentials are obviously lower than the sample I–III. It is worth noting sample IV still outperforms the H2-reduced sample (sample V). And the corresponding TEM images (Fig. S4†) reveal that a ultrathin layer of CoO exists in sample IV, but no obvious CoO species are observed for the sample V. These results indicate that a suitable balance between Co and CoO in the catalyst is crucial for high catalytic activity, resulted from the synergistic effect as stated above. The CoO species could lower the water-dissociation energy barrier and the metallic Co acts as a relay to further convert H* to H2 molecular. The sample III might have the optimal balanced Co/CoO configuration that can take full advantage of this synergistic effect to achieve the best HER activity.
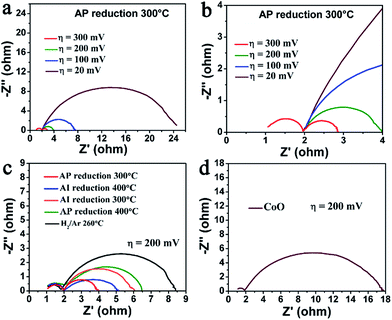
To further reveal the roles of Co and CoO in HER kinetics of the hybrid catalysts, the impedance spectroscopy (EIS) analyses were performed. Fig. 5a and Fig. 5b show the Nyquist plots of sample III at different overpotentials. Two semicircles are observed in these plots: a small one in the high frequency domain that is independent of the potential, which is attributed to the electrode porosity; and a bigger one in the middle and low frequency domain that was varied with the potential, which is related to the HER kinetics.47 The charge-transfer resistance (RCT) of HER can be calculated from the diameter of the second semicircle.47 Fig. 5c shows the Nyquist plots of the samples with different component ratio of Co/CoO at the same overpotential 200 mV. All the samples display similar semicircles in the high frequency domain but significantly different ones in the middle and low frequency domain, indicating the substantial correlation between the HER-RCT and the Co/CoO components. The value of HER-RCT of these samples are summarized in Table 1. The sample I–IV have obviously lower HER-RCT compared to the sample V, which accounts for their lower onset overpotentials and confirms the important role of CoO in lowering the H* adsorption energy barrier. While the HER-RCT of pure CoO catalyst (sample VI) is very high to 15.8 Ω from its corresponding Nyquist plot (Fig. 5d), which accounts for its bad HER activity. That means fast HER kinetics also need proper metallic Co species to further produce H2 products. Sample III has the smallest HER-RCT among the Co/CoO hybrid catalysts, consistent with previous HER performances. In a word, the Co/CoO hybrid catalyst can enormously decrease the HER charge transfer resistance compared to the single component Co or CoO catalyst, and the lowest charge transfer resistance can be achieved by well balancing the ratio of Co/CoO components.
| HER-RCT (Ω) at η = 200 mV | |
|---|---|
| Sample I: AI reduction 300 °C | 3.9 |
| Sample II: AI reduction 400 °C | 3.0 |
| Sample III: AP reduction 300 °C | 1.8 |
| Sample IV: AP reduction 400 °C | 4.5 |
| Sample V: H2/Ar reduction 260 °C | 6.6 |
| Sample VI: Ar annealing 350 °C | 15.8 |
Conclusions
In summary, we prepared Co nanoparticles embedded CoO nanowire arrays via a template-free method, including cobalt hydroxy-carbonate nanowire arrays growth on Ni foam and the following non-contact Al-reduction process. They were endowed with improved properties of charge transfer, mass transport and faster HER kinetics. A suitable balance between Co and CoO in the catalyst is crucial for high catalytic activity. And the optimal Co/CoO array exhibited outstanding HER activity in alkaline media, approaching to the commercial Pt/C catalyst, and they also showed good long-term stabilities. This hybrid Co/CoO nanowire array may pave ways for designing new nanomaterials for efficient hydrogen production from water-splitting.Acknowledgements
This work was financially supported from NSF of China (Grant no. 61376056, 51125006, 91122034, 51121064), and Science and Technology Commission of Shanghai (Grant no. 13JC1405700, 14520722000).Notes and references
- K. Zeng and D. Zhang, Prog. Energy Combust. Sci., 2010, 36, 307–326 CrossRef CAS.
- Y. Jiao, Y. Zheng, M. Jaroniec and S. Z. Qiao, Chem. Soc. Rev., 2015, 44, 2060–2086 RSC.
- M. S. Faber and S. Jin, Energy Environ. Sci., 2014, 7, 3519–3542 CAS.
- Y. Zheng, Y. Jiao, M. Jaroniec and S. Z. Qiao, Angew. Chem., Int. Ed., 2015, 54, 52–65 CrossRef CAS PubMed.
- S. Peng, L. Li, X. Han, W. Sun, M. Srinivasan, S. G. Mhaisalkar, F. Cheng, Q. Yan, J. Chen and S. Ramakrishna, Angew. Chem., Int. Ed., 2014, 53, 12594–12599 CAS.
- J. Kibsgaard, T. F. Jaramillo and F. Besenbacher, Nat. Chem., 2014, 6, 248–253 CrossRef CAS PubMed.
- Y. H. Chang, C. T. Lin, T. Y. Chen, C. L. Hsu, Y. H. Lee, W. Zhang, K. H. Wei and L. J. Li, Adv. Mater., 2013, 25, 756–760 CrossRef CAS PubMed.
- D. Kong, H. Wang, Z. Lu and Y. Cui, J. Am. Chem. Soc., 2014, 136, 4897–4900 CrossRef CAS PubMed.
- E. J. Popczun, C. G. Read, C. W. Roske, N. S. Lewis and R. E. Schaak, Angew. Chem., 2014, 126, 5531–5534 CrossRef.
- E. J. Popczun, J. R. McKone, C. G. Read, A. J. Biacchi, A. M. Wiltrout, N. S. Lewis and R. E. Schaak, J. Am. Chem. Soc., 2013, 135, 9267–9270 CrossRef CAS PubMed.
- P. Jiang, Q. Liu, Y. Liang, J. Tian, A. M. Asiri and X. Sun, Angew. Chem., Int. Ed., 2014, 53, 12855–12859 CrossRef CAS PubMed.
- J. Tian, Q. Liu, N. Cheng, A. M. Asiri and X. Sun, Angew. Chem., Int. Ed., 2014, 53, 9577–9581 CrossRef CAS PubMed.
- D. H. Youn, S. Han, J. Y. Kim, J. Y. Kim, H. Park, S. H. Choi and J. S. Lee, ACS Nano, 2014, 8, 5164–5173 CrossRef CAS PubMed.
- W.-F. Chen, C.-H. Wang, K. Sasaki, N. Marinkovic, W. Xu, J. Muckerman, Y. Zhu and R. Adzic, Energy Environ. Sci., 2013, 6, 943–951 CAS.
- W.-F. Chen, J. T. Muckerman and E. Fujita, Chem. Commun., 2013, 49, 8896–8909 RSC.
- W. F. Chen, K. Sasaki, C. Ma, A. I. Frenkel, N. Marinkovic, J. T. Muckerman, Y. Zhu and R. R. Adzic, Angew. Chem., Int. Ed., 2012, 51, 6131–6135 CrossRef CAS PubMed.
- J. R. McKone, B. F. Sadtler, C. A. Werlang, N. S. Lewis and H. B. Gray, ACS Catal., 2013, 3, 166–169 CrossRef CAS.
- N. Krstajić, V. Jović, L. Gajić-Krstajić, B. Jović, A. Antozzi and G. Martelli, Int. J. Hydrogen Energy, 2008, 33, 3676–3687 CrossRef.
- E. Endoh, H. Otouma, T. Morimoto and Y. Oda, Int. J. Hydrogen Energy, 1987, 12, 473–479 CrossRef CAS.
- B. Tilak, A. Ramamurthy and B. Conway, High performance electrode materials for the hydrogen evolution reaction from alkaline media, 1986 Search PubMed.
- R. Subbaraman, D. Tripkovic, D. Strmcnik, K.-C. Chang, M. Uchimura, A. P. Paulikas, V. Stamenkovic and N. M. Markovic, Science, 2011, 334, 1256–1260 CrossRef CAS PubMed.
- M. Gong, W. Zhou, M. C. Tsai, J. Zhou, M. Guan, M. C. Lin, B. Zhang, Y. Hu, D. Y. Wang, J. Yang, S. J. Pennycook, B. J. Hwang and H. Dai, Nat. Commun., 2014, 5, 4695 CrossRef CAS PubMed.
- N. Danilovic, R. Subbaraman, D. Strmcnik, K. C. Chang, A. Paulikas, V. Stamenkovic and N. M. Markovic, Angew. Chem., 2012, 124, 12663–12666 CrossRef.
- H. Jin, J. Wang, D. Su, Z. Wei, Z. Pang and Y. Wang, J. Am. Chem. Soc., 2015, 137, 2688–2694 CrossRef CAS PubMed.
- X. Liu, W. Liu, M. Ko, M. Park, M. G. Kim, P. Oh, S. Chae, S. Park, A. Casimir, G. Wu and J. Cho, Adv. Funct. Mater., 2015, 25, 5799–5808 CrossRef CAS.
- X. Yan, L. Tian and X. Chen, J. Power Sources, 2015, 300, 336–343 CrossRef CAS.
- X. Yan, L. Tian, M. He and X. Chen, Nano Lett., 2015, 15, 6015–6021 CrossRef CAS PubMed.
- X. Liu, X. Wang, X. Yuan, W. Dong and F. Huang, J. Mater. Chem. A, 2016, 4, 167–172 CAS.
- J. Jiang, J. Liu, X. Huang, Y. Li, R. Ding, X. Ji, Y. Hu, Q. Chi and Z. Zhu, Cryst. Growth Des., 2009, 10, 70–75 Search PubMed.
- Z. Wang, C. Yang, T. Lin, H. Yin, P. Chen, D. Wan, F. Xu, F. Huang, J. Lin, X. Xie and M. Jiang, Energy Environ. Sci., 2013, 6, 3007 CAS.
- Z. Wang, C. Yang, T. Lin, H. Yin, P. Chen, D. Wan, F. Xu, F. Huang, J. Lin and X. Xie, Energy Environ. Sci., 2013, 6, 3007–3014 CAS.
- X. Lu and C. Zhao, Nat. Commun., 2015, 6(6616), 1–7 Search PubMed.
- C. Yu, X. Dong, L. Guo, J. Li, F. Qin, L. Zhang, J. Shi and D. Yan, J. Phys. Chem. C, 2008, 112, 13378–13382 CAS.
- W. Luo, X. Hu, Y. Sun and Y. Huang, ACS Appl. Mater. Interfaces, 2013, 5, 1997–2003 CAS.
- R. B. Rakhi, W. Chen, M. N. Hedhili, D. Cha and H. N. Alshareef, ACS Appl. Mater. Interfaces, 2014, 6, 4196–4206 CAS.
- C.-T. Ho, T.-H. Weng, C.-Y. Wang, S.-J. Yen and T.-R. Yew, RSC Adv., 2014, 4, 20053–20057 RSC.
- J. L. Mackay and V. E. Henrich, Phys. Rev. B: Condens. Matter Mater. Phys., 1989, 39, 6156 CrossRef CAS.
- X. Chen, L. Liu, Y. Y. Peter and S. S. Mao, Science, 2011, 331, 746–750 CrossRef CAS PubMed.
- T. Xia, C. Zhang, N. A. Oyler and X. Chen, Adv. Mater., 2013, 25, 6905–6910 CrossRef CAS PubMed.
- Y. Wang, T. Zhou, K. Jiang, P. Da, Z. Peng, J. Tang, B. Kong, W. B. Cai, Z. Yang and G. Zheng, Adv. Energy Mater., 2014, 4(16), 1–7 Search PubMed.
- Y.-F. Li and A. Selloni, ACS Catal., 2014, 4, 1148–1153 CrossRef CAS.
- Z. Pu, Q. Liu, C. Tang, A. M. Asiri and X. Sun, Nanoscale, 2014, 6, 11031–11034 RSC.
- J. Tian, Q. Liu, A. M. Asiri and X. Sun, J. Am. Chem. Soc., 2014, 136, 7587–7590 CrossRef CAS PubMed.
- M. A. Lukowski, A. S. Daniel, F. Meng, A. Forticaux, L. Li and S. Jin, J. Am. Chem. Soc., 2013, 135, 10274–10277 CrossRef CAS PubMed.
- D. Voiry, H. Yamaguchi, J. Li, R. Silva, D. C. Alves, T. Fujita, M. Chen, T. Asefa, V. B. Shenoy and G. Eda, Nat. Mater., 2013, 12, 850–855 CrossRef CAS PubMed.
- B. Conway and B. Tilak, Electrochim. Acta, 2002, 47, 3571–3594 CrossRef CAS.
- B. Łosiewicz, A. Budniok, E. Rówiński, E. Łągiewka and A. Lasia, Int. J. Hydrogen Energy, 2004, 29, 145–157 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available: XRD pattern and SEM image of Co2(OH)2CO3·nH2O nanowire arrays, XRD pattern of CoO powder and Co powder, TEM images of sample IV and sample V. See DOI: 10.1039/c6ra07538j |
| This journal is © The Royal Society of Chemistry 2016 |