The preparation of 3D network pore structure activated carbon as an electrode material for supercapacitors with long-term cycle stability
Changyu Leng and
Kang Sun*
Institute of Chemical Industry of Forest Products, CAF, National Engineering Lab for Biomass Chemical Utilization, Key and Open Lab on Forest Chemical Engineering, SFA, Nanjing 210042, China. E-mail: sunkang0226@163.com
First published on 31st May 2016
Abstract
The high performance bio-based activated carbon (BAC) was prepared via a novel combination of chemical and physical activation processes using coconut shells as a precursor. The as-obtained BAC exhibited a three-dimensional (3D) network pore structure, high specific surface area (3242.05 m2 g−1), unobstructed and interconnected pores, and large pore volume (1.919 cm3 g−1). The BAC exhibited a high specific capacitance of 337 F g−1 (202 F cm−3) and retained a specific capacitance of 331 F g−1 (190 F cm−3) after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles (98% capacitance reservation) at a current density of 0.5 A g−1 in a 6 M KOH electrolyte. AC-W had a gravimetric capacitance value of 240 F g−1 and a volumetric capacitance value of 120 F cm−3 in 1 M TEA BF4/AN. The BAC shows ideal properties as electrode material and also exhibits long-term cycle stability, low capacitance loss rate and high capacitance as an electrode material in aqueous and organic electrolytes. These results are expected to develop efficient, environmentally friendly and low-cost supercapacitors, as well as to promote their application in future electric vehicles, mobile phones and power electronics.
000 cycles (98% capacitance reservation) at a current density of 0.5 A g−1 in a 6 M KOH electrolyte. AC-W had a gravimetric capacitance value of 240 F g−1 and a volumetric capacitance value of 120 F cm−3 in 1 M TEA BF4/AN. The BAC shows ideal properties as electrode material and also exhibits long-term cycle stability, low capacitance loss rate and high capacitance as an electrode material in aqueous and organic electrolytes. These results are expected to develop efficient, environmentally friendly and low-cost supercapacitors, as well as to promote their application in future electric vehicles, mobile phones and power electronics.
1. Introduction
The technology of the electric vehicle has been developed over the previous decades, but there are still only a small proportion of electric vehicles running on the street, which is due to the battery technology of electric vehicles being not qualified. In actual use, a large volume of battery packs are employed to support the electric vehicle for long-time driving. The combined price of all the battery packs is about half of the vehicle price due to the abovementioned problems, such that the frequent replacement of the battery packs is the bottleneck of the promotion of the electric vehicle application. It can be observed that the capacitance of the lithium ion battery (LIB) is large, but its charge rate is very slow and the LIB does not have the capacity of instant high-power discharge. The cost of lead–acid batteries is low, but it is heavy in weight and causes serious environmental pollution.1–7Supercapacitors (SC) have the characteristics of high specific capacitance and rapid charge–discharge. They are an ideal energy storage device of future electric vehicles, mobile phones and power electronics. Obviously, the electrode material is one of the most important factors of a SC. The electrode materials at present include activated carbon, metal oxides, and conducting polymers.8–11 Metal oxides have a high specific capacitance but also have high costs and massive pollution production during the preparation process. Conducting polymers have a high working voltage, but its resistance is high and the rigidity is inferior. It can be clearly observed that they cannot meet the demands of an excellent performing SC. Activated carbon (AC) has environmental friendliness, a high specific surface area, rapid charge–discharge, superior rigidity, a lightweight quality, a high specific capacitance and low cost. Those advantages determine that it is an ideal electrode material of the future SCs. Now, the specific surface area of commercial activated carbon can reach 3000 cm2 g−1, but the AC-based supercapacitor still has fast capacitance loss after multiple charge–discharge processes and a short cycling life.9–14 During the charge–discharge process, the poor connection of pores makes ionic motion difficult such that some electrolyte ions are trapped and deposited in pores, which leads to high ion-transport resistance, insufficient ionic diffusion, and distortion and blockage of pores.33,34 Because of the disconnected, blocked and isolated pores, electrolyte ions can enter the electrode under the charging voltage, but they cannot be thoroughly removed from the electrode during the discharge process. Therefore, electrolyte ions deposit in the pores and channels more and more, which results in a fast capacitance loss and short-term cycle stability.15–21
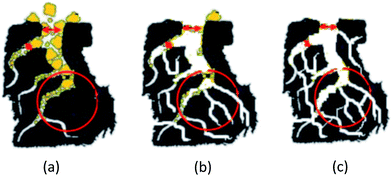
Coconut shells are a type of the renewable biomass resource. They have a dense structure and well-developed cell cavity, which is beneficial to the penetration of the activation reagent. In this study, we designed a novel physical–chemical activation process to prepare the AC from coconut shells with an ideal pore structure. According to the activation mechanism,5,22 in the first step, ZnCl2 was used to prepare AC with mesopores as a foundation framework. (Fig. 1(a)) Then, we employed KOH, which has a stronger activation capability than ZnCl2, to etch a large number of micropores within the mesopores and remove organic impurities and non-carbonated particles inside the porous channels to interconnect meso–mesopores. (Fig. 1(b)) In the final step, high-temperature water vapor was passed though the AC porous channels, further sweeping the remaining rubbish and interconnecting the micro–micropores sufficiently, making the porous structure tidier and cleaner. (Fig. 1(c)) We utilized ZnCl2, KOH and water vapor as activation reagents in succession to obtain the expected activated carbon that has a three-dimensional (3D) interconnected network structure and better electrochemical properties for use as a carbon-based electrode.
2. Experimental
2.1 Preparation of the AC samples
Coconut shells were first ground and then sieved to get 0.6 mm and 1 mm sized particles. Then, 20 g of the abovementioned prepared particles were mixed with zinc chloride23 for activation at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 wt% and maintained at room temperature for 24 h. Subsequently, the mixture was emptied into a muffle furnace. The temperature of the furnace was raised at 10 °C min−1 up to 650 °C, and maintained at 650 °C for 1 h during the activation process. The sample was then washed with 0.1 M HCl and distilled water and then dried at 120 °C for 6 h. The as-prepared sample was named AC-Zn.
3 wt% and maintained at room temperature for 24 h. Subsequently, the mixture was emptied into a muffle furnace. The temperature of the furnace was raised at 10 °C min−1 up to 650 °C, and maintained at 650 °C for 1 h during the activation process. The sample was then washed with 0.1 M HCl and distilled water and then dried at 120 °C for 6 h. The as-prepared sample was named AC-Zn.
The AC-Zn was impregnated with KOH in a covered stainless steel vessel at 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 wt%. 15 g of the AC-Zn was first pretreated at 350 °C for 3 h under air atmosphere and then the mixture was activated at a rate of 10 °C min−1 up to 800 °C and maintained this temperature for 1 h. The sample was also washed in the same manner as the AC-Zn sample to remove excess reagent. The resulting sample was named AC-K.
1 wt%. 15 g of the AC-Zn was first pretreated at 350 °C for 3 h under air atmosphere and then the mixture was activated at a rate of 10 °C min−1 up to 800 °C and maintained this temperature for 1 h. The sample was also washed in the same manner as the AC-Zn sample to remove excess reagent. The resulting sample was named AC-K.
AC-W was prepared from AC-K by water vapor activation as follow steps. 10 g of AC-K was heated at a rate of 10 °C min−1 to 800 °C under nitrogen flow in a horizontal furnace. The sample was then activated in steam (0.2 mL min−1) at 800 °C for 30 min. The sample was subsequently washed with 0.1 M HCl and then distilled water and dried at 120 °C for 6 h.
2.2 Characterizations
Scanning electron microscopy (SEM) was used to observe the morphology of the samples. The properties of interconnected pores and channels were observed with transmission electron microscopy (TEM). Nitrogen (77 K) adsorption was carried out using a Micrometritics ASAP 2020 analyzer. The surface area values, the pore size distribution and total pore volume of the activated carbon samples were calculated by the density functional theory (DFT) and according to the adsorbed nitrogen amount. The elemental analysis was performed according to GB/T12496 with an elemental analyzer. The properties of the activated carbon structure were investigated by Raman spectra and X-ray diffraction (XRD).2.3 Electrochemical measurements
The capacitive measurements of the AC samples were carried out in a two-electrode system. The supercapacitor tests were performed on a CHI 660D electrochemical workstation at room temperature. The double electrodes were composed of 85% activated carbon, 5% acetylene black and 10% PTFE. The mixture was then sliced to make electrodes with a diameter of 10 mm and a thickness of 0.10 mm. The geometric surface area of a slice is 0.785 cm2, herein, the mass of the electrodes range from 13 to 15 mg. The two circular sheet-type carbon-based electrodes were directly placed inside the simulated supercapacitor and separated by polyvinylidene fluoride (PVDF) paper in 6 M KOH. Cyclic voltammetry (CV) was carried out at different scan rates, galvanostatic charge–discharge (GC) curve was obtained at different current densities from 0.01 to 0.9 V, and electrochemical impedance spectroscopy (EIS) was measured at frequencies from 100 kHz to 10 mHz.3. Result and discussion
3.1 Morphology and structure
The surface morphologies of AC-Zn, AC-K and AC-W were analyzed by SEM in Fig. 2(a)–(d). There are some obvious changes of morphologies on the surface of the AC samples. The SEM image of AC-Zn (Fig. 2(a)) exhibited few pores and massive non-carbonized substances on the surface. Fig. 2(b) showed that AC-K still had some non-carbonized substances and organic impurities on the surface, but it had more micropores that were etched by KOH. Furthermore, the surface of AC-W was very clean and smooth, as shown in Fig. 2(c), which confirmed that the role of steam had further swept the non-carbonized substances and organic residues. The SEM of AC-W shown in Fig. 2(d) also confirmed that the surface of the AC-W was clean and smooth. | ||
| Fig. 2 (a) SEM image of AC-Zn; (b) SEM image of AC-K; (c) SEM image of AC-W; and (d) SEM image of AC-W. | ||
From the Fig. 3(a), we can observe that the AC-W has some graphite microcrystals and mesopores (2–50 nm). The TEM of AC-W shown in Fig. 3(b) was typical for the activated carbon with a large number of micropores (the standard of Fig. 3(b) and (d) is 5 nm). Signally, there were elliptic channels of mesopores etched on the carbon walls visible through the TEM observation (Fig. 3(c)). In the TEM of AC-W shown in Fig. 3(d), an unobstructed and interconnected root-type network structure with mesopores and some micropores is observed, which can make ionic motion easy and reduce the ion-transport resistance. Furthermore, the large amount of micropores and mesopores provide sufficient contact inter-area between the carbon-based electrode and electrolyte, which was good for rapid ion transport from the surface to the pores and channels of electrode. The balance of the micro–mesopores can enrich the active sites and allow the micropores to be accessed property, so that the capacitance of the SCs will be improved and the electronic resistance of the electrodes sharply decreased. The well-controlled distribution of pores sizes provides channels for fast ions motion and shortens the diffusion distance, which is better for long-term cycle stability. This structure makes AC-W an ideal electrode material for supercapacitors with low capacitance loss rate and a long cycling life.
3.2 Raman and X-ray diffraction analysis
The structure of the AC samples was investigated by Raman spectroscopy and X-ray diffraction (XRD). There were no sharp and high peaks in the XRD due to the amorphous state of the AC samples, as shown in Fig. 4(a).24 The Raman spectrum of the AC samples was shown in Fig. 4(b), which exhibits a D-band peak at 1330 cm−1 and G-band at 1590 cm−1. The vibrations of the dangling bonds of the carbon atoms were usually related to the peak at 1330 cm−1 (D-band) for the in-plane terminations of disordered graphite and the vibration in all sp2 bonded carbon atoms were closely associated to the peak at 1590 cm−1 (G-band).25 Although the peak shape of AC-W was higher and narrower than AC-Zn and AC-K, the strong D-band peak demonstrated that AC-W still contained an obvious quantity of disordered defect sections and had a low degree of crystallinity.26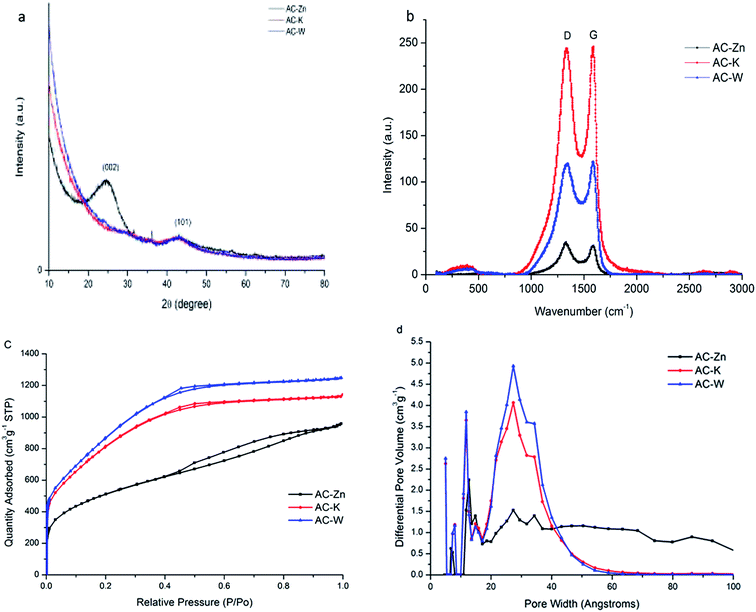 | ||
| Fig. 4 (a) XRD pattern and (b) Raman spectrum of AC samples. (c) N2 adsorption–desorption isotherms and (d) the corresponding pore size distributions of the as-prepared samples. | ||
3.3 Porous texture of AC
As shown in Fig. 4, the SSA of the samples was calculated using the BET method with a p/p0 value between 0.10 and 0.25 for the N2 adsorption data. The SSA and PSD were also calculated by the DFT method using the N2 adsorption data. The total pore volume was determined by the amount of nitrogen absorbed at p/p0 = 0.9. The properties of sample textures were summarized in Table 1. According to IUPAC classification, the presented isotherms of AC-Zn were type IV, as shown in Fig. 4(c). AC-Zn exhibited the lowest isotherm and a hysteresis loop at high relative pressure. This indicated that there were some mesopores formed on the surface of the activated carbon, which were the channels for rapid ions transportation and offered a place for the next step of micropores formation. The AC-K and AC-W showed type I isotherms and AC-W displayed a significantly higher isotherm than AC-K. At a relative pressure of 0.4, the N2 adsorption volumes both showed a gradual increase, so there were more various sizes of micropores in AC-K and AC-W. At a relative pressure of 0.4, it can be clearly observed that the isotherms also showed a hysteresis loop in the desorption branch, which adequately proved that some mesopores still existed in the AC-K and AC-W samples. The total DFT specific surface areas were 3022.70 and 3242.05 m2 g−1 with total pore volumes of 1.742, and 1.919 cm3 g−1 for AC-K and AC-W, respectively. The micropore volumes of AC-K and AC-W were 1.512 and 1.669 cm3 g−1, corresponding to the micropore volume fractions of 86.8, and 87%.| Vmicro (cm3 g−1) | Vmeso (cm3 g−1) | Total pore volume (cm3 g−1) | DFT SSA (m2 g−1) | Average pore width (nm) | |
|---|---|---|---|---|---|
| AC-Zn | 0.836 | 0.36373 | 1.482 | 1250.61 | 3.20350 |
| AC-K | 1.512 | 0.02836 | 1.742 | 3022.70 | 2.30557 |
| AC-W | 1.669 | 0.01276 | 1.919 | 3242.05 | 2.36822 |
The high-mesopores-ratio AC-based supercapacitors showed higher power output, high-frequency performance, and superior capacitive behavior.27 It was demonstrated that high mesopore ratios can enhance the performance of ion diffusion in the pores/channels of the AC. Moreover, a high mesopore ratio was suitable for quick charge and discharge because the high mesopore rate was suitable for electrolyte ions to transport inside the porous structure of the AC. More inner-surfaces of pores inside the AC can be wetted by the electrolyte. Therefore, zinc chloride was used to prepare AC with mesopores as foundation framework and then we used KOH to etch a large number of micropores in the mesopores, which can be observed in Fig. 4(c). Due to the role of steam during reactivation, the micropores widened a bit, which made the small mesopores form. However, reactivation obviously changed the pore size distribution of the AC-K. In addition to the small micropores reserved from the AC-K, a lot of micropores and small mesopores were generated in AC-W with pore sizes between 2 and 4 nm. The change resulted from carbon atoms burned off and carbon atoms reacting with the steam during steam reactivation. The micropore volumes of the AC-K and AC-W were 1.512 and 1.669 cm3 g−1, corresponding to the micropore volume fractions of 86.8, and 87%. The substantial difference between the pore volumes and pore size distributions (Fig. 4(d)) of the AC samples were due to the different roles of the chemical or physical activation methods, which can also be confirmed by SEM and TEM images.29
3.4 Electrochemical properties of the activated carbon
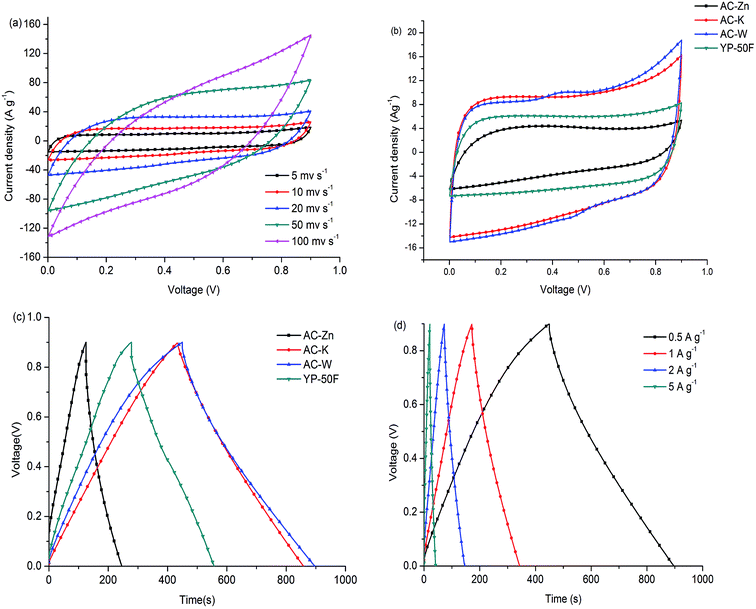
The CV method can exhibit the influence of porous structure on ion transport behavior, which is due to the capacitive performance of supercapacitors. In general, a rectangular shaped voltammogram is required for the desired capacitive performance. Changing the voltage scan rates can estimate the applicability for rapid charge–discharge process. The CV data of AC-Zn, AC-K and AC-W at a scan rate of 5 mV s−1 were shown in Fig. 5. All the CV curves for the AC material electrodes exhibited a rectangular shape, which was a property of electrochemical double-layer capacitance. In addition, the AC-W electrode CV curve exhibited a larger rectangular area and a bigger current response than that of the AC-K and AC-Zn electrodes, an obvious increase in specific capacitance demonstrated during the activation process. Fig. 5(b) showed the CV curves of the AC-W electrode at different scan rates of 5–100 mV s−1. The CV curve gradually became tilted as the scan rate increased, but even at 100 mV s−1 it still maintained a rectangular-like shape, which indicated a good capacitive behavior and a small resistance in the accessible pores of the AC-W electrode.28 Therefore, the electrochemical performance of AC-W is more excellent than the AC-Zn and the AC-K.As shown in Fig. 5(b), AC-W showed rectangular shaped cyclic voltammetry curves from 0 to 1.0 V. The EDLCs had little limitation of electrolyte diffusion, and even at scan rate of 50 mV s−1, the rectangular shape was still maintained due to the interconnected pores structure and the fast ionic motion. There are two formulas that are included in the calculation of capacitance per electrode independently of whether it is CV or GC. The specific capacitance values of a single electrode Cs (F g−1) can be calculated according to eqn (1), with the formula
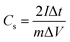 | (1) |
Galvanostatic charge–discharge measurements are used to study the relationship of specific capacitance and different loading current densities and to calculate the specific capacitance. The GC of the AC samples was shown in Fig. 5 at various current densities, which was typically triangular on the charge–discharge curves. The specific capacitance of the AC samples can also calculate based on eqn (2):
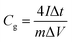 | (2) |
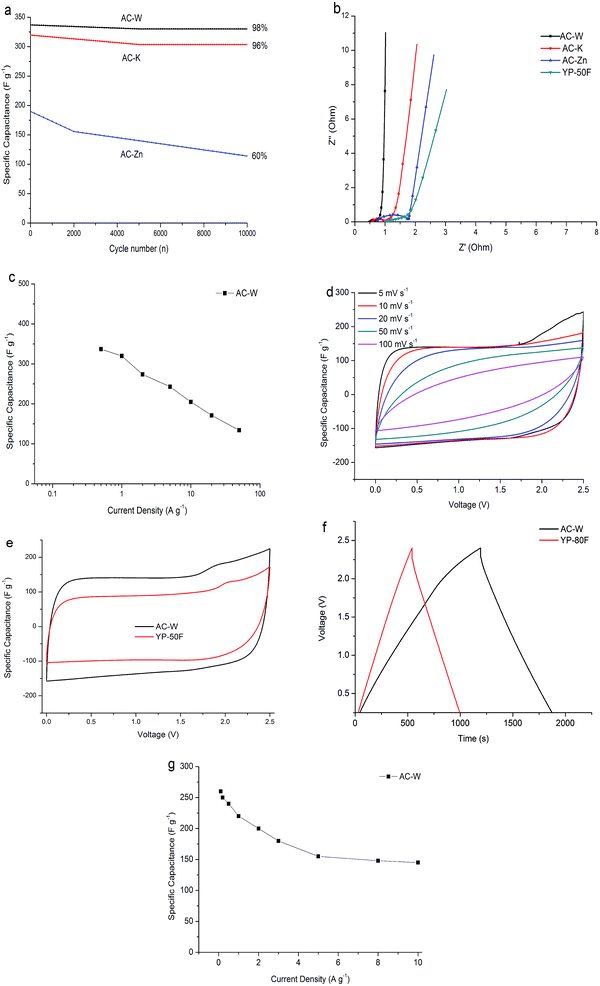
For actual application, durability is the most important property of a capacitor. The high values of capacitance and stable capacitance with cycling are both important for practical use.30 To further understand the capacitive behavior of the AC samples, Fig. 6(a) showed the cyclic stability of the AC-Zn, AC-K and AC-W electrodes measured by galvanostatic charge/discharge for 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles. The specific capacitance of AC-Zn first exhibited a sharp decrease from 190 to 156 F g−1 after 1000 cycles and 114 F g−1 was maintained (60% capacitance retention) after 10
000 cycles. The specific capacitance of AC-Zn first exhibited a sharp decrease from 190 to 156 F g−1 after 1000 cycles and 114 F g−1 was maintained (60% capacitance retention) after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles. The specific capacitance of AC-K was relatively stable (96% capacitance retention) after 10
000 cycles. The specific capacitance of AC-K was relatively stable (96% capacitance retention) after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles. The specific capacitance for AC-W was 337 F g−1 (202 F cm−3) in the first cycle, and after 10
000 cycles. The specific capacitance for AC-W was 337 F g−1 (202 F cm−3) in the first cycle, and after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles, still maintained a coulombic efficiency of 98% (331 F g−1). The AC-W shows higher specific capacitance than other samples. The AC-W exhibits an extraordinary high stability at a high current density of 1 A g−1 after 10
000 cycles, still maintained a coulombic efficiency of 98% (331 F g−1). The AC-W shows higher specific capacitance than other samples. The AC-W exhibits an extraordinary high stability at a high current density of 1 A g−1 after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles, merging the excellent reversibility and stability of the AC-W as electrode material for SCs.
000 cycles, merging the excellent reversibility and stability of the AC-W as electrode material for SCs.
Fig. 6(b) showed the Nyquist plots for the AC-Zn, AC-K and AC-W electrodes. A semicircle in the high-frequency region and a straight line in the low-frequency region were displayed in the three plots. The non-vertical slope line of the low frequency impedance can be considered a deviation from the ideal capacitive behavior. Based on the simulation results of an equivalent circuit, it is believed that the better electrochemical behavior of the AC-W mainly originates from the suitable pore structure, with the concurrence of micropores and small mesopores, and better conductivity. The ideal porous structure can promote quick electrolyte ion diffusion into or out the channels and pores of the carbon-based materials. Moreover, in the low-frequency region, AC-W showed a more vertical line leaning to the imaginary axis, which signified more excellent electrochemical performance than AC-K and AC-Zn. The AC-W electrode had excellent pore accessibility, low internal (Rs) and charge transfer (Rct) resistance for the electrolyte.31 Furthermore, from the high frequency region, the AC-W has a shorter Warburg domain in the plot and a smaller semicircle, which evidences better ion transfer efficiency and smaller contact resistance between the electrolyte solution and electrode material than the other two samples, respectively. Above all, the AC-W has the lowest equivalent series resistance (ESR), which is associated with good intrinsic electronic properties for the samples, low ions transfer resistance in the materials and low contact resistance between electrode and current collector.
Fig. 6(c) is the rate performance of AC-W in 6 M KOH electrolyte. The gravimetric capacitance of AC-W electrodes shows slow fading as the rate increase from 0.5 A g−1 to 1 A g−1. It is due to large number of micropores, which play a role in storing charge. Furthermore, the AC-W shows slow fading under high current density, because it not only has many micropores, which provide high specific capacitance, but also an appropriate numbers of mesopores as channels for fast ions motion and short diffusion distance. It proves that the AC-W has well-balanced micro/mesopores and it is beneficial for a high-rate performance as an electrodes for SCs. A lot of micropores would be bad for capacitive performance under high current density while a lot of mesopores cannot provide high specific capacitance values. Therefore, we should find a balance between the amount of micropores and mesopores and prepare a high-rate performance carbon material as an electrode for SCs.
In addition to the aqueous electrolyte (6 M KOH), we also explored the capacitive performance of the AC-W based supercapacitor in an organic electrolyte (TEA BF4/AN). The voltage window is 2.5 times larger than the aqueous electrolyte and even at 100 mV s−1, the CV curves from 0 to 2.5 V still have the typical rectangular shape, indicating rapid ion transfer and convenient and smooth ion channels. The AC-W exhibits excellent performance with a high specific capacitance at 0.1 A g−1 and still retains a specific capacitance of 240 F g−1 and 210 F g−1 even at high current density of 0.5 A g−1 and 1 A g−1, respectively. The electrochemical properties of AC-W are better than that of carbon derived from rice husk, coconut shell, coffee beans, seaweed (150 F g−1 at 0.2 A g−1, 250 F g−1 in 1 M H2SO4, 286 F g−1 in 1 M H2SO4, 74.5 F g−1 at 0.5 A g−1), and even superior to some advanced carbon materials, such as carbon nanotubes, nanofilms and graphene. In Fig. 6(e), the specific capacitance of AC-W is higher than the commercial activated carbon YP-50F, indicating that the three-step preparation brings high additional values to AC-W. It is really superior than most physically or chemically activated carbon and some novel carbon materials. It can be observed in Fig. 6(g) that the AC-W also has excellent rate performance in the organic electrolyte. Similarly, the gravimetric capacitance of AC-W electrodes also show slow fading as the current density increases, and it still has a specific capacitance of 145 F g−1 at the high current density of 10 A g−1. It proves that the AC-W not only has many micropores, but also has appropriate numbers of mesopores and channels for rapid ion transfer.
The AC-W is very promising as an electrode for SCs with low capacitance loss rate and long-term cycle stability. The clean and interconnected pore structure network, high specific surface area and effective pore volumes contribute to the superior energy storage performance of AC-W.32 The abovementioned factors can be summarized to explain the obvious aspects of low capacitance loss rate and long-term cycle stability achieved in bio-based activated carbon based supercapacitors. First, a sufficient electrode–electrolyte interface as a result of a high specific surface area results in a double-layer capacitance, and provides interconnected electrolyte ion transport channels. Second, shortening the diffusion pathways and connecting porous channels can result in fast ion diffusion. Finally, the AC-W has intrinsic electronic conductivity characteristics due to the intrinsic natural network of pores structure and interconnected channels in the framework. Therefore, the abovementioned porous structure can be used as an electrode material for SCs, which have low capacitance loss rate and long-term cycle stability.
4. Conclusions
A novel biomass based AC with interconnected pore structures has been prepared via a new physical–chemical activation processes using coconut shells as a precursor. First, ZnCl2 was used to prepare AC with mesopores as a foundation framework. Then, KOH as a stronger activation agent was used to etch more micropores and connect the micro–meso pores. In the final step, water vapor was passed though the AC porous channels, further sweeping the remaining impurities and connecting the micro–micropores sufficiently. In addition, the novel activation processes can be developed into an applicative route to realize industrialization production and the high performance AC can be used as an electrode material for SCs with low capacitance loss rates and long-term cycle stability.Acknowledgements
This study is financially supported by the projects in forestry public benefit research sector (201404610).References
- M. Hou, K. Sun and X. Deng, et al., RSC Adv., 2015, 5, 74664–74670 RSC.
- Z. Dong, S. J. Kennedy and Y. Wu, J. Power Sources, 2011, 196, 4886–4904 CrossRef CAS.
- A. Lewandowski, M. Zajder and E. Frąckowiak, et al., Electrochim. Acta, 2001, 46, 2777–2780 CrossRef CAS.
- Y. Zhu, S. Murali, M. D. Stoller and K. J. Ganesh, Science, 2011, 332, 1537–1541 CrossRef CAS PubMed.
- Z. Jin, W. Mu and C. Zhang, et al., Electrochim. Acta, 2012, 59, 100–104 CrossRef CAS.
- Z. Yu, L. Tetard and L. Zhai, et al., Energy Environ. Sci., 2015, 8, 702–730 CAS.
- P. J. Hall, M. Mirzaeian and S. I. Fletcher, et al., Energy Environ. Sci., 2010, 3, 1238–1251 CAS.
- X. He, Y. Geng and J. Qiu, et al., Carbon, 2010, 48, 1662–1669 CrossRef CAS.
- T. Brousse, P. L. Taberna and O. Crosnier, et al., J. Power Sources, 2007, 173, 633–641 CrossRef CAS.
- Z. Fan, Y. Liu and J. Yan, et al., Adv. Energy Mater., 2012, 2, 419–424 CrossRef CAS.
- C. Xiehong, S. Yumeng and S. Wenhui, et al., Small, 2011, 7, 3163–3168 CrossRef PubMed.
- E. Frackowiak and F. Beguin, Carbon, 2001, 39, 937–950 CrossRef CAS.
- H. Jiang, P. S. Lee and C. Li, Energy Environ. Sci., 2012, 6, 41–53 Search PubMed.
- K. J. Chun, H. K. Park and D. Qu, et al., J. Power Sources, 1998, 74, 99–107 CrossRef.
- X. Y. Chen, C. Chen and Z. J. Zhang, et al., J. Power Sources, 2013, 230, 50–58 CrossRef CAS.
- S. R. Sivakkumar and A. G. Pandolfo, Electrochim. Acta, 2012, 65, 280–287 CrossRef CAS.
- M. D. Stoller and R. S. Ruoff, Energy Environ. Sci., 2010, 3, 1294–1301 CAS.
- T. Brousse, P. L. Taberna and O. Crosnier, et al., J. Power Sources, 2007, 173, 633–641 CrossRef CAS.
- S. R. C. Vivekchand, C. S. Rout and K. S. Subrahmanyam, et al., J. Chem. Sci., 2008, 120, 9–13 CrossRef CAS.
- M. Kim, Y. Hwang and K. Min, et al., Electrochim. Acta, 2013, 113, 322–331 CrossRef CAS.
- H. Pin, Z. Zhenhuan and T. Jian, et al., Nanoscale, 2014, 6, 12120–12129 RSC.
- M. M. Thackeray, C. Wolverton and E. D. Isaacs, Energy Environ. Sci., 2012, 5, 7854–7863 CAS.
- Z. Ming, S. Yan and Y. Li, et al., J. Power Sources, 2012, 217, 6–12 CrossRef.
- L. Sun, C. Tian and M. Li, et al., J. Mater. Chem. A, 2013, 1, 6462–6470 CAS.
- E. Redondo, J. Carretero-González and E. Goikolea, et al., Electrochim. Acta, 2015, 160, 178–184 CrossRef CAS.
- E. Frackowiak, G. Lota and G. Gryglewicz, et al., Electrochim. Acta, 2004, 49, 515–523 CrossRef.
- M. Jian, B. Liu and R. Liu, et al., RSC Adv., 2015, 5 Search PubMed.
- A. Perrin, A. Celzard and A. Albiniak, et al., Microporous Mesoporous Mater., 2005, 81, 31–40 CrossRef CAS.
- D. C. S. Azevedo, J. C. S. Araújo and M. Bastos-Neto, et al., Microporous Mesoporous Mater., 2007, 100, 361–364 CrossRef CAS.
- A. Davies and A. Yu, Can. J. Chem. Eng., 2011, 89, 1342–1357 CrossRef CAS.
- J. Yan, Z. Fan and T. Wei, et al., Carbon, 2010, 48, 3825–3833 CrossRef CAS.
- T. Tsubota, Y. Miyauchi and N. Murakami, et al., J. Power Sources, 2011, 196, 5769–5773 CrossRef CAS.
- T. Han, Chem. Sci., 2016, 7, 1791–1796 RSC.
- M. S. Park, J. Kim and K. J. Kim, et al., Phys. Chem. Chem. Phys., 2015, 17 Search PubMed.
| This journal is © The Royal Society of Chemistry 2016 |