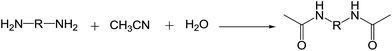Amide exchange reaction: a simple and efficient CuO catalyst for diacetamide synthesis†
Qinghe Liab,
Peixue Wanga and
Youquan Deng*a
aState Key Laboratory for Oxo Synthesis and Selective Oxidation, State Key Laboratory of Solid Lubrication, Lanzhou Institute of Chemical Physics, Chinese Academy of Sciences, Lanzhou 730000, China. E-mail: ydeng@licp.cas.cn; Fax: +86 931 4968116
bGraduate University of Chinese Academy of Sciences, Beijing 100049, China
First published on 12th April 2016
Abstract
A highly copper-catalysed amide exchange reaction of hexamethylenediamine (HDA) with CH3CN and H2O for the synthesis of hexamethylenebisacetamide (HMBA) without an organic solvent or gas protection was developed. 100% HDA conversion and >99% HMBA selectivity was obtained. X-ray diffraction, scanning emission microscopy, and temperature-programmed reduction of hydrogen were used to characterize the structural properties of the catalyst. The reaction mechanism was also investigated.
Diacetamide products (DAPs), which are among the simplest imides, model compounds of biochemical relevance, and good co-crystallising agents, have a variety of applications in numerous industrially important processes, as well as in medicine, as pesticides, and in organic synthesis.1–5 The molecular conformation and basic applications have attracted considerable attention. In recent years, the amide exchange reaction (AER) has been used increasingly to synthesize DAPs by pharmaceutical companies and in research laboratories.6,7 Among the many different DAPs, hexamethylenebisacetamide (HMBA)8–10 is a major anti-carcinogen and medical–chemical intermediate used in the synthesis of different organic chemicals,11–25 including hexamethylenetertamine (HMTA), diethyl hexamethylenedicarbamate (EHDC), and diphenyl hexamethylenedicarbamate (PHDC), (Fig. 1).
Given the potential applications of HMBA, considerable work has gone into its production.26–28 The traditional preparation method of HMBA, however, still utilises a noble metal complex catalyst and an organic solvent under an inert atmosphere and has a low yield. As shown in Scheme 1, RuH2(PPh3)4 was employed in the reaction of HDA, CH3CN, and H2O to prepare HMBA using dimethoxyethane (DME) as the solvent under an argon atmosphere, in only 89% yield.29,30 However, the development of a method using mild conditions and a non-noble metal catalyst for the conversion of diamine to diacetamide is still required. Much effort has gone into identifying alternative non-noble metal catalysts and green ways to produce HMBA. Recently, there has been a lot of focus on different copper-catalysed reaction systems. Copper catalysts have been used as simple and efficient promoters in various reaction systems such as photo-catalytic reactions.31,32
Herein, a simple and efficient copper catalyst was used to replace noble metal catalysts in the reaction of HDA, CH3CN, and H2O without an organic solvent or gas protection with an isolated yield of 96%.
The catalytic performance of different metal oxide catalysts was studied (see ESI Table 1†). From the results, it was identified that the CuO catalyst shows better catalytic performance than the others in this reaction, with 100% HDA conversion and >99% HMBA selectivity. However, the use of other copper catalysts, including Cu, Cu2O, CuCl, CuBr, and CuSO4, was less successful, leading to poor HMBA selectivity compared to CuO. The CuO catalyst was deemed to be more suitable for the reaction of HDA, CH3CN, and H2O (Table 1).
| Entry | Catalyst | Conv. (%) | Sel.b (%) |
|---|---|---|---|
| a Reaction conditions: 0.01 mol HDA, 0.03 mol CH3CN, 0.06 mol H2O, 0.00625 mol catalyst, 180 °C, 2 h. Conversion and selectivity were determined by gas chromatography.b Selectivity of 1b. | |||
| 1 | CuO | 100 | >99 |
| 2 | Cu | 98 | 61 |
| 3 | Cu2O | 98 | 72 |
| 4 | CuCl | 93 | 70 |
| 5 | CuBr | 90 | 84 |
| 6 | CuSO4 | 89 | 60 |
Then, we optimised the reaction conditions of the model reaction of HDA, CH3CN, and H2O (see ESI, Fig. S1†). Primarily, the influence of the ratio of reagents was investigated. In the presence of 0.00625 moles of catalyst at 180 °C for 2 h, the optimal molar ratio of HDA, CH3CN, and H2O was found to be 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3
3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6.
6.
The recyclability of the CuO catalyst was also investigated (Fig. 2). The results show a slight decrease in activity after each run, corresponding to a decrease in HDA conversion from 100% to 98%. This is because CuO recovered only 95% of the charged amount after each run by simply centrifuging, washing, and drying, and it was slightly influenced by water, suggesting that the CuO catalyst could be reused without significant loss of activity. As shown in Fig. 2, HMBA selectivity was also influenced by the slight decrease in activity of the CuO catalyst.
 | ||
| Fig. 2 Cycling tests of CuO. Reaction conditions: 0.01 mol HDA, 0.03 mol CH3CN, 0.06 mol H2O, recovered catalyst, 180 °C, 2 h. The selectivity in this figure was assigned to HMBA. | ||
Subsequently, to demonstrate the utility and generality of this approach to the formation of diacetamides, reactions of different diamines, including aliphatic diamines (linear chain and cyclic diamines) and aromatic diamines, with CH3CN and H2O were carried out under the optimised conditions, the results are shown in Table 2. Excellent yields of the corresponding diacetamides were obtained with the linear chain and cyclic aliphatic diamines (entries 1–5). On the other hand, lower activities were obtained with aromatic diamines towards the production of the corresponding diacetamides (entry 6–10), which might be due to the weak nucleophilicity of the nitrogen atom and the conjugative effect derived from the benzene ring. 2-Methyl-1H-benzo[d]imidazole was easily formed, detected using an Agilent GC-MS 7890B (see ESI Fig. S4†), from the reaction of o-phenylenediamine and CH3CN, which proceeded with only 3% selectivity for the diacetylation product. However, high activity was achieved with 1,3-bisaminomethylbenzene (entry 11) with 100% conversion and 83% diacetamide selectivity due to the benzyl group of this aromatic diamine. The CuO catalytic system was applicable to a wide range of aliphatic and aromatic diamines.
| Entry | Diamine | Major product | T (h) | Conv. (%) | Sel.b (%) |
|---|---|---|---|---|---|
| a Reaction conditions: 0.01 mol diamine, 0.03 mol CH3CN, 0.06 mol H2O, 0.00625 mol catalyst, 180 °C, 2–8 h. Conversion and selectivity were determined by gas chromatography.b Selectivity of diacetamide. | |||||
| 1 |  |
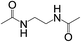 |
2 | 100 | >99 |
| 2 |  |
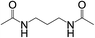 |
2 | 100 | >99 |
| 3 | 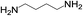 |
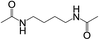 |
2 | 100 | >99 |
| 4 | 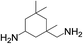 |
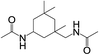 |
8 | 100 | 95 |
| 5 | 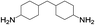 |
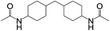 |
8 | 100 | 93 |
| 6 | 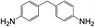 |
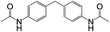 |
8 | 56 | 20 |
| 7 | 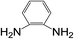 |
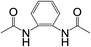 |
8 | 45 | 3 |
| 8 | 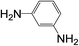 |
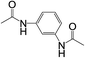 |
8 | 63 | 15 |
| 9 |  |
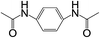 |
8 | 75 | 25 |
| 10 | 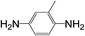 |
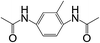 |
8 | 35 | 10 |
| 11 | 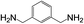 |
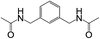 |
2 | 100 | 83 |
X-ray diffraction (XRD), scanning electron microscopy (SEM), and temperature-programmed reduction of hydrogen (TPR-H2) were carried out to examine the relationship between the structure of the CuO catalyst and performance of the reaction system. The XRD results of the prepared and used CuO are shown in Fig. 3. The diffraction peaks at 32°, 35°, 38°, 48°, 53°, 58°, 61°, 66°, 68°, 72°, and 75° of the prepared CuO are in good agreement with pure CuO (JCPDS card no. 801916). In addition, the sharp and narrow peaks indicate that the prepared CuO is well crystallised. The peaks at 18°, 33°, 43°, and 50° 2θ in Fig. 3b were assigned to Cu, which was formed by the reaction of by-product NH3 with CuO, causing a slight decrease in activity of the CuO catalyst.
The CuO catalyst prepared by the hydrothermal method has a nanorod-like morphology with an average diameter of 200 nm, as shown in Fig. 4. For comparison, different morphologies of CuO were also investigated and found to exhibit similar catalytic activities, indicating that the morphology of the CuO catalyst has little influence in this reaction.
The TPR results of the prepared CuO, commercial CuO, and Cu2O catalysts are shown in Fig. 5. A broad peak at 305 °C can be observed for the prepared pure CuO catalyst (Fig. 5a). There are two broad peaks at 367 and 534 °C, which are assigned to Cu2O and CuO, respectively, because Cu2O is more easily reduced than CuO under the same conditions. In addition, the shift in CuO reduction temperature from 305 to 534 °C indicated some type of interaction between Cu2O and CuO, which may inhibit the reduction of CuO (Fig. 5b). On the contrary, the introduction of CuO may favour the reduction of Cu2O, with much lower peaks at 254 and 280 °C observed in Fig. 5c compared to Fig. 5b.
In order to study the mechanism of the reaction of HDA, CH3CN, and H2O catalysed by CuO, reactions of different substrates, including HDA, CH3CN, H2O, and CH3CONH2, were carried out and traced by gas chromatography-mass spectrometry (GC-MS), (Table 3).
| Entry | HDA (mol) | CH3CN (mol) | H2O (mol) | CH3CONH2 (mol) | Conv. (%) | Sel.b (%) |
|---|---|---|---|---|---|---|
| a Reaction conditions: 180 °C, 2 h, magnetic stirring speed of 960 rpm, 100 mL high pressure reactor. Conversion and selectivity were determined by gas chromatography.b Selectivity of HMBA.c Conversion of HDA.d Conversion of CH3CN.e Without the CuO catalyst. | ||||||
| 1 | 0.01 | 0.03 | 0.06 | — | 100c | >99 |
| 2 | 0.01 | 0.03 | — | — | — | — |
| 3 | 0.01 | — | — | 0.03 | 82c | 52 |
| 4 | 0.01 | — | 0.06 | — | — | — |
| 5 | — | — | 0.06 | 0.01 | — | — |
| 6 | — | 0.03 | — | 0.01 | — | — |
| 7e | — | 0.03 | 0.06 | — | 6.5d | — |
| 8 | — | 0.03 | 0.06 | — | 8d | — |
Initially, HDA, CH3CN, and H2O (molar ratio, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3
3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6) were added to the reactor, and 100% HDA conversion and >99% HMBA selectivity were obtained (entry 1). The reactions of HDA with CH3CN, H2O, and CH3CONH2 were investigated. As shown in Table 3 entry 2, no HDA consumption or HMBA was detected in the reaction solution. These results and those of previous reports show that the polymerization reaction of CH3CN was promoted by a catalytic amount of HDA to form 1,3,5-trimethyl-triazine, which was detected by GC-MS in this experiment. Only 82% HDA conversion and 52% HMBA selectivity were obtained when HDA reacted with CH3CONH2 directly (entry 3). There was no reaction between HDA and H2O, CH3CONH2, and H2O, and CH3CONH2 and CH3CN in the presence of the CuO catalyst under the reaction conditions (entries 4–6). In the end, compared to entry 7, which has 6.5% CH3CN conversion under no CuO catalyst, the reaction of CH3CN with H2O catalyzed by CuO showed 8% CH3CN conversion to CH3CONH2 (entry 8), indicating that CuO has little effect on the reaction of CH3CN with H2O.
6) were added to the reactor, and 100% HDA conversion and >99% HMBA selectivity were obtained (entry 1). The reactions of HDA with CH3CN, H2O, and CH3CONH2 were investigated. As shown in Table 3 entry 2, no HDA consumption or HMBA was detected in the reaction solution. These results and those of previous reports show that the polymerization reaction of CH3CN was promoted by a catalytic amount of HDA to form 1,3,5-trimethyl-triazine, which was detected by GC-MS in this experiment. Only 82% HDA conversion and 52% HMBA selectivity were obtained when HDA reacted with CH3CONH2 directly (entry 3). There was no reaction between HDA and H2O, CH3CONH2, and H2O, and CH3CONH2 and CH3CN in the presence of the CuO catalyst under the reaction conditions (entries 4–6). In the end, compared to entry 7, which has 6.5% CH3CN conversion under no CuO catalyst, the reaction of CH3CN with H2O catalyzed by CuO showed 8% CH3CN conversion to CH3CONH2 (entry 8), indicating that CuO has little effect on the reaction of CH3CN with H2O.
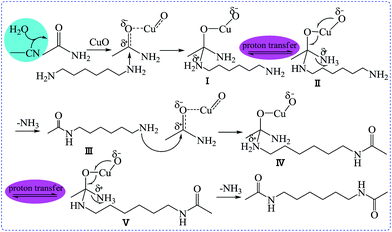
Based on the reactions discussed above, a feasible reaction pathway for the AER of HDA, CH3CN, and H2O catalysed by CuO is proposed, as shown in Scheme 2. The results indicate that HMBA was generated in two consecutive steps. Initially, CH3CONH2 was formed by a reaction of CH3CN and H2O, followed by the formation of HMBA in the second step via the AER of HDA with CH3CONH2, the former reaction was promoted by the latter. It is worth noting that in the second step, CuO plays a significant role in the nucleophilic addition reaction of HDA and CH3CONH2. The electrophilicity of the carbonyl carbon atom in CH3CONH2 was promoted by the CuO catalyst. The carbonyl was then attacked by nucleophilic HDA.
As shown above, the nucleophilic addition of CH3CONH2 activated by CuO with HDA occurred to form I, followed by proton transfer to generate II. The elimination of ammonia from II affords intermediate III, which goes on to react with activated CH3CONH2 to afford IV. V then forms HMBA via the elimination of ammonia.
Conclusions
We proposed a highly copper-catalysed AER of HDA with CH3CN and H2O for the synthesis of HMBA. Under the optimised reaction conditions, good yields of various diacetamides were obtained with different diamines. This optimised catalytic system for the AER of diamines, CH3CN, and H2O is preferable for the synthesis of diacetamides via an environmentally benign route. Further development and application of this reaction are currently underway.Acknowledgements
This study was financially supported by the National Natural Science Foundation of China (No. 21173240).Notes and references
- W. Wang, Y. Huang and N. Tang, Acta Crystallogr., Sect. E: Struct. Rep. Online, 2008, 64, o2237 CAS.
- X. F. Li, Y. An, Q. H. Huang and Y. H. Wen, Acta Crystallogr., Sect. E: Struct. Rep. Online, 2011, 67, o3291 CAS.
- P. Li, C. Zhang and W. Xu, Acta Crystallogr., Sect. E: Struct. Rep. Online, 2011, 67, o3041 CAS.
- H. X. Cai and W. N. Wu, Acta Crystallogr., Sect. E: Struct. Rep. Online, 2012, 68, o933 CAS.
- M. Yusuf, I. Solanki and P. Jain, J. Chem. Sci., 2012, 124, 703–715 CrossRef CAS.
- L. F. Beste and R. C. Houtz, J. Polym. Sci., 1952, 8, 395–407 CrossRef CAS.
- K. Miller, J. Polym. Sci., 1976, 14, 1403–1417 Search PubMed.
- M. Bailey, Acta Crystallogr., 1955, 8, 575–578 CrossRef CAS.
- J. C. Williams and A. E. McDermott, J. Phys. Chem. B, 1998, 102, 6248–6259 CrossRef CAS.
- I. Sandeman, J. Math. Phys. Sci., 1955, 232, 105–113 CrossRef CAS.
- B. A. Conley, M. J. Egorin, V. Sinibaldi, G. Sewack, C. KIoc and L. Roberts, Cancer Chemother. Pharmacol., 1991, 28, 33–38 CrossRef CAS PubMed.
- B. A. Conley, M. J. Egorin, V. Sinibaldi, G. Sewack, C. KIoc and L. Roberts, Cancer Chemother. Pharmacol., 1992, 31, 37–45 CrossRef CAS PubMed.
- C. A. Burris, S. D. Silva, W. C. Narrow, A. E. Casey, L. T. Lotta, H. J. Federoff and W. J. Bowers, J. Gene Med., 2008, 10, 152–164 CrossRef CAS PubMed.
- L. N. Dong, M. Liu, A. J. Chen, D. Z. Sun, X. L. Wei and Y. Y. Di, J. Chem. Eng. Data, 2011, 56, 4031–4039 CrossRef CAS.
- L. N. Dong, M. Liu, A. J. Chen and D. Z. Sun, J. Chem. Eng. Data, 2012, 57, 2456–2464 CrossRef CAS.
- E. Meilhoc, M. J. Moutin and H. B. Osborne, Biochem. J., 1986, 238, 701–707 CrossRef CAS PubMed.
- A. Haces, T. R. Breitman and J. S. Driscol, J. Med. Chem., 1987, 30, 405–409 CrossRef CAS PubMed.
- R. Kitagawa, Y. Takahashi, M. Takahashi, H. Imazu, M. Yasuda, H. Sadanari and J. Tanaka, Virology, 2009, 383, 195–206 CrossRef CAS PubMed.
- G. Q. Li, M. Liu, L. N. Dong, L. L. Wang, D. Z. Sun, X. L. Wei and Y. Y. Di, J. Chem. Thermodyn., 2012, 48, 160–174 CrossRef CAS.
- G. Q. Li, M. Liu, L. L. Wang, L. N. Dong, D. Z. Sun, X. L. Wei and Y. Y. Di, J. Solution Chem., 2012, 41, 849–863 CrossRef CAS.
- G. Q. Li, M. Liu, L. L. Wang, L. Y. Zhu, D. Z. Sun and Y. Di, J. Chem. Eng. Data, 2010, 55, 4239–4243 CrossRef CAS.
- H. Li, C. J. Chi, M. Liu, D. Z. Sun and J. F. Liu, J. Chem. Thermodyn., 2010, 42, 1187–1191 CrossRef CAS.
- P. Papazafiri and H. B. Osborne, Eur. J. Biochem., 1989, 178, 789–793 CrossRef CAS PubMed.
- D. M. L. Goodgame, D. A. Grachvogel, I. Hussain, A. J. P. White and D. J. Williams, Inorg. Chem., 1999, 38, 2057–2063 CrossRef CAS PubMed.
- N. P. Chatterton, D. M. L. Goodgame, D. A. Grachvogel, I. Hussain, A. J. P. White, D. J. Williams, A. J. P. White and D. J. Williams, Inorg. Chem., 2001, 40, 312–317 CrossRef CAS PubMed.
- G. E. Munn and W. Del, US Pat., 2745842, 1956.
- F. E. Gould, G. S. Johnson and A. F. Ferris, J. Org. Chem., 1960, 25, 1658–1660 CrossRef CAS.
- S. Kakaei and J. Xu, Org. Biomol. Chem., 2013, 11, 5481–5490 CAS.
- S. M. Ikeda and T. Naota, US Pat., 4801748, 1989.
- S. M. Ikeda, T. Naota and E. Saito, J. Am. Chem. Soc., 1986, 108, 7846–7847 CrossRef PubMed.
- J. P. Wang, H. Xu, X. F. Qian, Y. Y. Dong, J. K. Gao, G. D. Qian and J. M. Yao, Chem.–Asian J., 2015, 10, 1276–1280 CrossRef CAS PubMed.
- J. K. Gao, J. P. Wang, X. F. Qian, Y. Y. Dong, H. Xu, R. J. Song, C. F. Yan, H. C. Zhu, Q. W. Zhong, G. D. Qian and J. M. Yao, J. Solid State Chem., 2015, 228, 60–64 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Details of experimental procedures of preparation and characterization of catalysts and catalytic data, and characterization of the product. See DOI: 10.1039/c6ra05563j |
| This journal is © The Royal Society of Chemistry 2016 |