DOI:
10.1039/C6RA04633A
(Paper)
RSC Adv., 2016,
6, 46889-46899
Synthesis of stable core–shell structured TiO2@Fe3O4 based on carbon derived from yeast with an enhanced photocatalytic ability
Received
21st February 2016
, Accepted 4th May 2016
First published on 6th May 2016
Abstract
By using yeast as the carbon source, stable core–shell structured TiO2@Fe3O4 based on carbon (TiO2@Fe3O4-carbon) was obtained. The photocatalytic activity of TiO2@Fe3O4-carbon reached 82.6%, which was much higher than that of others; the enhanced photocatalytic activity originated from the good conductivity of carbon. Meanwhile, TiO2@Fe3O4-carbon also exhibited excellent stability during the photocatalytic reaction after 6 repeated cycles. In addition, a plausible mechanism was proposed and discussed on the basis of experimental results.
1. Introduction
Semiconductor nanostructures are of both theoretical and technological importance and exhibit a wide range of electrical and optical properties that can offer widespread applications in photocatalysis, solar energy conversion, energy storage, etc.1–5 Among them, TiO2 is one of the most studied semiconductor nanostructures.6–8 Titanium dioxide, commonly called titania, plays a central role in energy and environmental research, finding applications in dye-sensitized solar cells, in lithium ion batteries, as a photocatalyst for hydrogen production by water splitting, in self-cleaning coatings, and as a photocatalyst for water treatment.9–15 However, the TiO2 photocatalytic efficiency is limited by its wide band-gap energy (∼3.2 eV), which limits the spectrum of photons that can create electron–hole pairs to the ultraviolet (UV) light.16,17 The methods to enhance the photocatalytic properties of TiO2 are mainly based on the inhibition of the photo-generated electrons–holes recombination, on the increase of the exposed surface area, and on the decrease of the TiO2 band-gap energy.18–21
Currently, increasing attention is being focused on carbon based photocatalysts with enhanced photocatalytic activities.22 The utilization of carbon in the composites of photocatalysts not only improves the separation of photogenerated electron–hole pairs due to its high electronic conductivity but also facilitates the concentration of pollutant molecules at the carbon/catalyst interface. The high surface area, large pores (macroporosity), and the presence of surface hydroxyl groups make carbon substance an ideal catalyst support.23–25 Hou et al. reported that the carbon modified Bi12TiO20 showed an excellent photocatalytic activity for degradation of rhodamine-B ascribing to the reduced recombination of photogenerated electron–hole pairs associated with the carbon.26 Li et al. prepared the P25–graphene composite showing a significant enhancement in photodegradation of methylene blue due to enhanced charge separation and transportation.27 Like the titania photocatalysts supported on the carbon matrix by means of several strategies28–30 appear to have various benefits and advantages for providing a cheap and effective wastewater treatment and remediation options.31,32 Liu et al.33 demonstrated the formation of TiO2/carbon fibers (ACFs) composite photocatalyst via sol–gel method and the TiO2/ACFs is especially helpful for the removal of low molecular weight organic pollutants in the contaminated water. The yeast-carbon is a porous and amorphous solid carbon material, which is derived mainly from baker's yeast. For instance, Nacoo and Aquarone34 reported the fabrication of yeast-carbon by carbonizing in a gas-heated muffle. The obtained yeast-carbon possessed higher surface area. The resultant carbon material displayed effective adsorption of organic chemicals in wastewater treatment.
On the other hand, although TiO2 has been proved to be an ideal candidate for treatment of water pollution, the efficient separation and recycling of this finely powdered catalyst is still a scientific problem when applied in practice. Thus a facile and effective approach to recover suspended TiO2 particles is highly desirable. Fortunately, magnetic separation provides a suitable solution to this issue. By immobilized on various magnetic supports (such as carbon35) to form hybrid materials, TiO2 catalyst can not only be collected with an external magnetic field but also inherit from the supports excellent surface chemistry, high mechanical properties, and good thermal stability.36 As a result, successful strategies for preparation of magnetic TiO2 hybrid catalysts are recognized as one of major advances in nanotechnology.
Herein, we carefully designed and synthesized a TiO2@Fe3O4-carbon core–shell structured catalyst which well addressed both the catalytic activity and recovery issues for photocatalysts. We reported a novel TiO2@Fe3O4-carbon photocatalyst by simple-step method for utilization of photodegrading tetracycline (TC) under simulated solar light irradiation. The characteristics of obtained samples were studied by transmission electron microscopy (TEM), high-resolution transmission electron microscopy (HRTEM) images, fast Fourier transformation (FFT), scanning electron microscope (SEM), energy dispersive X-ray (EDX) analysis, X-ray diffraction (XRD), UV-Vis diffuse reflectance spectroscopy (DRS), Fourier-transformed infrared spectra (FT-IR), Raman spectroscopy, X-ray photoelectron spectroscopy (XPS), Brunauer, Emmett and Teller (BET) surface area analysis, vibrating sample magnetometer (VSM), Electron Spin Resonance (ESR) spectroscopy, the photocatalytic properties were obtained by photodegrading TC solution and the absorbance datum and spectra were observed by UV-Vis spectrophotometer. In addition, the stability and mechanism of the composites during photodegrading TC were attentively studied.
2. Materials and method
2.1 Materials and reagents
The powdered yeast was purchased from Angel Yeast Company. Other reagents including tetracycline (TC), ferric chloride hexahydrate (FeCl3·6H2O), ethylene glycol (EG), diethylene glycol (DEG), sodium acrylate (CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHCOONa, Na acrylate), sodium acetate (CH3COONa, NaOAc), benzoquinone (BQ), isopropanol (IPA), triethanolamine (TEOA), 5,5-diamethyl-1-pyrroline N-oxide (DMPO) were obtained from Aladdin. Tetrabutyl titanate (CR), hydrochloric acid (HCl, AR), alcohol (AR) were purchased from Sinopharm Chemical Reagent Co., Ltd. All of the chemicals were analytical grade and used directly without further purification.
CHCOONa, Na acrylate), sodium acetate (CH3COONa, NaOAc), benzoquinone (BQ), isopropanol (IPA), triethanolamine (TEOA), 5,5-diamethyl-1-pyrroline N-oxide (DMPO) were obtained from Aladdin. Tetrabutyl titanate (CR), hydrochloric acid (HCl, AR), alcohol (AR) were purchased from Sinopharm Chemical Reagent Co., Ltd. All of the chemicals were analytical grade and used directly without further purification.
2.2 Preparation of Fe3O4/yeast
Yeast (0.1 g), FeCl3·6H2O (2.4 g, 9 mmol), CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHCOONa (3.4 g), NaOAc (3.4 g) were added into a mixture of EG (22.5 mL) and DEG (22.5 mL) under ultrasonication for about 1 h. The homogeneous solution obtained was transferred to a Teflon-lined stainless-steel autoclave and sealed to heat at 200 °C. After reaction for 10 h, the autoclave was cooled to room temperature. The Fe3O4/yeast obtained was washed several times with ethanol and water, and then dried in vacuum at 65 °C for 10 h.
CHCOONa (3.4 g), NaOAc (3.4 g) were added into a mixture of EG (22.5 mL) and DEG (22.5 mL) under ultrasonication for about 1 h. The homogeneous solution obtained was transferred to a Teflon-lined stainless-steel autoclave and sealed to heat at 200 °C. After reaction for 10 h, the autoclave was cooled to room temperature. The Fe3O4/yeast obtained was washed several times with ethanol and water, and then dried in vacuum at 65 °C for 10 h.
2.3 Preparation of TiO2@Fe3O4/yeast
The TiO2/Fe3O4/yeast photocatalyst was prepared via a mild sol–gel method. 10 mL tetrabutyl titanate was dissolved in 36 mL alcohol with mechanical agitation for 15 min at 313 K. Subsequently, the mixed solution (contained 36 mL alcohol, 3 mL deionized water and 0.2 mL hydrochloric acid) was dropwise added into above mixture. When the sol was formed, 0.5 g Fe3O4/yeast was added into the sol and kept stirring to the gel. After the gel was dried under vacuum at room temperature, the TiO2/Fe3O4/yeast photocatalyst was obtained.
2.4 Preparation of TiO2@Fe3O4-carbon
The TiO2@Fe3O4-carbon photocatalyst was prepared via a simple method. 2.0 g TiO2/Fe3O4/yeast was dissolved in EG (30 mL) with mechanical agitation for 1 h at ambient temperature, the sample was centrifuged and washed with deionized water and absolute ethanol for several times and then dried at 60 °C in an electric oven. Finally, the sample was heated to 200 °C, 300 °C, 400 °C, 500 °C, 600 °C at a rate of 2 °C min−1 in dry synthetic air for 3 h in a tubular furnace and then cooled to ambient temperature normally. These samples were designated as TiO2@Fe3O4-carbon-200, TiO2@Fe3O4-carbon-300, TiO2@Fe3O4-carbon-400, TiO2@Fe3O4-carbon-500, TiO2@Fe3O4-carbon-600, respectively. The preparation procedure of this catalyst is shown in Scheme 1.
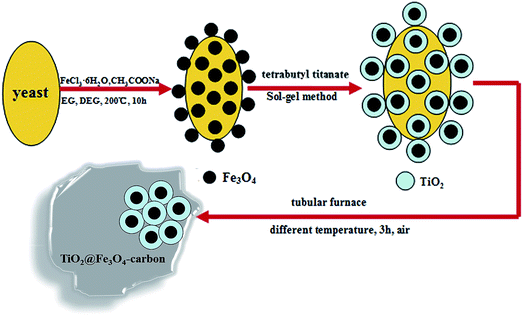 |
| | Scheme 1 Schematic preparation of TiO2@Fe3O4-carbon. | |
2.5 Adsorption experiment
The adsorption capacity was tested by the following steps: 0.1 g photocatalyst was added into 100 mL 20 mg L−1 TC aqueous solution, and then the sampling analysis was conducted in the interval of 10 min at 30 °C. After 60 min in the dark, the photocatalyst was isolated by a magnet, the concentration was measured with the UV-Vis spectrophotometer 2450 (Shimadzu Co., Japan). In order to ensure the authenticity of the experimental data, three repeated experiments were conducted.
2.6 Activity test
The photocatalytic activities of various catalysts were evaluated by the level of photodegradation of TC with an initial concentration of 20 mg L−1 TC solution under simulated solar light irradiation. Briefly, the photochemical reactor contains 100 mL TC solution and 0.1 g photocatalysts with recycling water (30 °C) to keep the reactor temperature constant. The reaction solution achieved adsorption and desorption balance after stirring for 30 min in the dark. The photocatalytic reaction was started by irradiating with a 350 W xenon lamp (without the filter, the wavelength is 200–800 nm). Samples were taken from the suspension in 20 min interval and centrifugally separated to remove catalysts for cyclic utilization in additional runs. The photocatalytic efficiency was calculated according to Dr = [(C0 − C)/C0] × 100%, where Dr is the photocatalytic efficiency, C0 is the adsorption equilibrium concentration of solution, C is the concentration of solution at time t. The concentration of TC was measured by a UV-Vis spectrophotometer at a wavelength of 357 nm. After the experiment, recycled sample was collected by an external magnet to ascertain the reproducibility of TiO2@Fe3O4-carbon photocatalyst.
2.7 Characterization
The SEM images were examined with XL30-ESEM scanning electron microscopy (Philips-FEI Co., Netherlands). The transmission electron microscopy (TEM), high-resolution transmission electron microscopy (HRTEM) images, and fast Fourier transformation (FFT) were examined by transmission electron microscopy (TEM: JEM-2010, Japan). Energy dispersive X-ray spectroscopy (EDS) was recorded on a FEI Tecnai G2F20 instrument and operated at an accelerating voltage of 200 kV. The crystal phase was carried out by powder X-ray diffraction (XRD), which is obtained by X-ray diffractometer (MAC Science, Japan) equipped with Ni-filtrated Cu Kα radiation (40 kV, 30 mA). The 2θ scanning angle range was 5–80° at a scanning rate of 5° min−1. Fourier-transformed infrared (FT-IR) spectra were recorded on a Nicolet Nexus 470 FT-IR (Thermo Nicolet Co., USA) with 2.0 cm−1 resolution in the range 400–4000 cm−1, using KBr pellets. The optical properties of the samples were analyzed by UV-Vis diffuse reflectance spectroscopy (DRS) with a Specord 2450 spectrometer (Shimadzu, Japan) in the wavelength range of 200–800 nm, in which BaSO4 was used as the internal reflectance standard. Raman experiments were performed using a DXR spectrometer, and measurements were made in backscattering geometry. Surface electronic states were analyzed on X-ray photoelectron spectroscopy (XPS, PerkinElmer PHI 5300). The specific surface area (BET) was estimated from the N2 adsorption–desorption isotherms, measured by a Quantachrome NOVA4000 surface area apparatus. Magnetic measurement was carried out using a VSM (7300, Lakeshore) under a magnetic field up to 10 kOe. Active free radicals were identified with electron spin resonance (ESR) on a JEOL FA-200. For the measurement, the samples (100 μL) were collected from the reaction solution after being reacted for 4 min, and immediately mixed with 20 μL DMPO (20 mmol L−1) to form DMPO–radicals adduct. Because of the instability in water solutions, the ESR signal of ˙O2− was detected by dimethyl sulfoxide.
3. Results and discussion
3.1 Structural characterization
Fig. 1a shows the XRD patterns of the as-prepared TiO2@Fe3O4-carbon composites. It shows that all diffraction peaks can be fully indexed to standard peaks of anatase crystalline phase of TiO2 (JCPDS 21-1272). With the temperature of TiO2@Fe3O4-carbon increased, the XRD peak intensities of anatase steadily increase and the widths of peaks become narrower.37 It can also be proved by matching the interplanar spacing d = 0.352 nm and 0.238 nm of TiO2@Fe3O4-carbon with the (101) and (004) crystallographic plane of anatase TiO2. And the diffraction peaks of the composite in each case correspond to the spinel lattice of Fe3O4 (magnetite, JCPDS card no. 19-629). Since maghemite (Fe2O3) also exhibits similar peaks, it is difficult to identify the precise nature of the oxide phase based on XRD alone. The absence of XRD peaks corresponding to any crystalline phases of carbon implies that carbon in the material is amorphous in nature.38
 |
| | Fig. 1 XRD patterns of TiO2@Fe3O4-carbon photocatalyst with different temperatures (a) and Raman spectra of TiO2@Fe3O4-carbon photocatalyst with different temperatures (b). | |
Raman spectroscopy, a powerful and more surface-sensitive technique than XRD for the investigation of crystalline phases, is applied. Typically, there are Raman active fundamental modes observed at 144 cm−1 (Eg), 397 cm−1 (B1g), 518 cm−1 (A1g + B1g) and 640 cm−1 (Eg) for anatase TiO2.39 Fig. 1b shows the resonant Raman scattering spectroscopy of the TiO2@Fe3O4-carbon. The curve of TiO2 exhibits five peaks at 144, 394, 512, 642 and 636 cm−1 which are attributed to ν1 + ν2 vibration of A1g + B1g, ν3 vibration of B1g, and ν4, ν5, ν6 vibration of Eg, respectively. The results indicate the anatase TiO2 referring to the standard anatase spectrum. In addition, it could be seen that the two new weak peaks appear at about 1352 cm−1 and 1586 cm−1 which are called D band and G band compared with TiO2. However, as temperatures rise, peak strength is weak, might be carbon content is less.
3.2 SEM, TEM, HRTEM and EDS images
The morphology and microstructure of TiO2@Fe3O4-carbon-300 is revealed by SEM, TEM, HRTEM and EDS, respectively. The SEM images of TiO2@Fe3O4-carbon-300 with different sizes presented in Fig. 2a illustrate their typical spherical morphology accumulation on the surface carbon materials. From Fig. 2b, the SEM images under higher magnifications reveal interesting details on how the TiO2@Fe3O4-carbon-300 are uniformly and randomly distributed within the granules. Further morphology observations by TEM are presented in Fig. 2c, it can be established that the TiO2@Fe3O4 nanoparticles are dispersed in carbon materials. From Fig. 2d, the spheres have diameters in the range of 200–300 nm. The inset shows a high magnification of an individual sphere, the sphere surface with a layer of 20 nm thickness uniform particles. The high-resolution TEM (HRTEM) images taken from the core and the shell of the TiO2@Fe3O4-carbon-300 microspheres, as shown in Fig. 2f, confirm that the Fe3O4 core and the TiO2 shell are well crystallized. The lattice spacing of 0.296 nm corresponds to the (220) plane of Fe3O4,40 and the d-spacing of 0.351 nm matches to the d101 of anatase TiO2. The fast Fourier transform (FFT) patterns show the corresponding reflections, thus confirming crystallinity of the calcined sample. In order to obtain more detailed contents information of the TiO2@Fe3O4-carbon-300, the EDS spectra from the Fig. 2g gives the signals of C, O, Ti, and Fe elements in the composites. The results of the elemental analysis are shown in Table 1. The content of C was the highest in TiO2@Fe3O4-carbon-300 samples, weight percentage reached 3.57%, and the atomic percentage was 7.98%.
 |
| | Fig. 2 The SEM of TiO2@Fe3O4-carbon-300 (a) and detailed aspects of the surface of the granules showing the uniform distribution of TiO2@Fe3O4-carbon-300 (b), the TEM of TiO2@Fe3O4-carbon-300 (c–e), HRTEM image and the FFT pattern of TiO2@Fe3O4-carbon-300 (f), and the EDS images of TiO2@Fe3O4-carbon-300 (g). | |
Table 1 EDS elemental analysis results of TiO2@Fe3O4-carbon
| Samples |
Element |
C |
O |
Ti |
Fe |
| TiO2@Fe3O4-carbon-200 |
Weight (%) |
3.21 |
32.67 |
54.39 |
3.25 |
| Atomic (%) |
7.48 |
53.91 |
30.29 |
1.57 |
| TiO2@Fe3O4-carbon-300 |
Weight (%) |
3.57 |
34.25 |
58.48 |
3.71 |
| Atomic (%) |
7.98 |
57.46 |
32.77 |
1.78 |
| TiO2@Fe3O4-carbon-400 |
Weight (%) |
3.01 |
34.68 |
58.98 |
3.76 |
| Atomic (%) |
6.48 |
57.86 |
32.97 |
1.85 |
| TiO2@Fe3O4-carbon-500 |
Weight (%) |
2.54 |
35.21 |
59.48 |
3.81 |
| Atomic (%) |
6.01 |
58.46 |
33.67 |
1.88 |
| TiO2@Fe3O4-carbon-600 |
Weight (%) |
2.05 |
35.26 |
59.68 |
3.83 |
| Atomic (%) |
5.38 |
58.31 |
33.98 |
1.89 |
3.3 XPS spectrum of TiO2@Fe3O4-carbon photocatalyst
Fig. 3 shows the surface chemical states of the TiO2@Fe3O4-carbon-300 with further investigation by XPS. The peak positions in all of the XPS spectra were calibrated with C 1s at 284.6 eV. The survey XPS spectrum of the TiO2@Fe3O4-carbon-300 indicates that the product consists of C, O and Ti elements (Fig. 3a). Fig. 3b shows the XPS spectrum of C 1s of the TiO2@Fe3O4-carbon-300. The peak at 284.58 eV of the C–C bond is detected and the other peaks are relatively decreased, which suggests the partly removal of oxygen-containing groups.41 The XPS peak of O 1s is illustrated in Fig. 3c. The peaks centered at 530.85 eV, indicating that calcium exists O. From Fig. 3d, the Ti 2p3/2 of TiO2@Fe3O4-carbon-300 are located at the binding energies of 463.77 and 459.31 eV, respectively, which are consistent with the binding energies TiO2.42
 |
| | Fig. 3 XPS spectrum of the TiO2@Fe3O4-carbon (a), high resolution scanning XPS of C 1s (b), high resolution scanning XPS of O 1s (c), and high resolution scanning XPS spectrum of Ti 2p3/2 of the TiO2@Fe3O4-carbon photocatalyst (d). | |
3.4 FT-IR spectra of photocatalysts
Fig. 4 shows the collected FT-IR spectra of the TiO2@Fe3O4-carbon composites. The peaks at 3411 cm−1 and 1633 cm−1 are attributed to the stretching vibration of H–O–H bond from the molecularly adsorbed H2O and OH groups on the TiO2 surface.43 The peak at 645 cm−1 can be looked at as a combination of Ti–O–Ti vibration and Ti–O–C vibration. Moreover, compared with the as prepared TiO2, another peaks appear at 1435 cm−1 and 1536 cm−1 on the composites' FT-IR spectra, which are corresponding to the vibration of C–C groups.44 Those results indicate that there exist many carbon-contain groups on the surface of TiO2@Fe3O4-carbon composites, which will enhance the absorption of simulated solar light irradiation. In addition, the characteristic band of Fe–O stretching vibration at about 540 cm−1 cannot be observed for the TiO2@Fe3O4-carbon composite, demonstrating the successful synthesis of Fe3O4 coated with TiO2.
 |
| | Fig. 4 FT-IR spectra of TiO2@Fe3O4-carbon photocatalyst with different temperatures. | |
3.5 UV-Vis DRS spectra of photocatalysts
Optical absorption properties and band structure of as-prepared nanomaterials are investigated by UV-Vis diffuse reflectance spectra (DRS). The UV-Vis absorption spectroscopy of TiO2@Fe3O4-carbon photocatalysts is shown in Fig. 5. It is widely acknowledged that the photocatalytic activity is closely associated with the light absorption ability. From Fig. 5, all of the as-prepared samples have photo-responses from simulated solar light irradiation region and show high absorption ranging from 200 nm to 600 nm. The TiO2@Fe3O4-carbon-300 composite can cause an obvious red shift of UV-Vis spectra and the absorption ability of the TiO2@Fe3O4-carbon-300 composite to simulated solar light irradiation region is remarkably enhanced. The greater band gap narrowing occurring on the TiO2@Fe3O4-carbon-300 composite is possibly attributed to the formation of Ti–O–C bond and TiO2@Fe3O4 core–shell structure which build new molecular orbital and narrow the band gap.43,45 The narrowing of band gap means the prepared TiO2@Fe3O4-carbon-300 composite photocatalysts could be excited to produce more hole–electron pairs under simulated solar light irradiation, which could result in higher photocatalytic activities. In addition, the Tauc relation for optical band-gap calculations of photocatalysts is written as
where a is the absorption coefficient, k is the parameter that is related to the effective masses associated with the valence and conduction bands, n is 1/2 for a direct transition, hν is the absorption energy, and Eg is the band gap energy. Fig. 5b shows a Tauc plot for the TiO2@Fe3O4-carbon systems where (ahν)1/2 was plotted against the photon energy (hν). The band gaps estimated from the extrapolated Tauc plot about 2.85 eV for TiO2@Fe3O4-carbon-300, and 3.15 eV for TiO2@Fe3O4-carbon-600, other catalysts band gap energy between the two.
 |
| | Fig. 5 UV-Vis diffuse reflectance spectra of TiO2@Fe3O4-carbon photocatalyst with different temperatures (a) and band gap energies of TiO2@Fe3O4-carbon photocatalyst with different temperatures (b). | |
3.6 VSM of composite photocatalysts
Magnetically separable carbon materials are highly attractive for the adsorption and enrichment of organic pollutants associated with liquid-phase processes. As shown in Fig. 6, the saturation magnetization (Ms), remanence magnetization (Mr), and coercivity (HC) can be obtained from the hysteresis loop. When the naked iron oxide nanoparticles and iron oxide–semiconductor composite nanomaterials exhibit superparamagnetic, the M–H curve should show no hysteresis at a certain temperature.46 Comparing with the pure Fe3O4 particles, the magnetic saturation (Ms) values of the TiO2@Fe3O4-carbon-300 composites was decreased. The observed reduction of Ms values of the TiO2@Fe3O4-carbon-300 composites was mainly due to the non-magnetic titanium dioxide coating on the surface of Fe3O4 particles. It is clearly seen in Fig. 6 (inset) that the magnetic sensitive property would favor the separation and recovery of TiO2@Fe3O4-carbon photocatalyst from the aqueous solution by imparting an external magnetic field simply.
 |
| | Fig. 6 The hysteresis loops of Fe3O4 particles, TiO2@Fe3O4-carbon-300 and the photographs of TiO2@Fe3O4-carbon-300 suspended in water in the absence and presence of an externally placed magnet (inset). | |
3.7 Nitrogen adsorption–desorption isotherm of composite photocatalysts
The hierarchically porous structure of the TiO2@Fe3O4-carbon-300 samples has also been probed by nitrogen adsorption/desorption experiments. Fig. 7 presents the nitrogen adsorption–desorption isotherms and Barrett–Joyner–Halenda (BJH) pore sized is distribution curves (inset) of the TiO2@Fe3O4-carbon-300 samples.
 |
| | Fig. 7 Nitrogen adsorption–desorption isotherm and Barrett–Joyner–Halenda (BJH) pore size distribution plot (inset) of the TiO2@Fe3O4-carbon-300. | |
To investigate the pore structure of TiO2@Fe3O4-carbon-300, the sample is analyzed by N2 adsorption and the BJH results are shown in Fig. 7. The adsorption isotherm exhibits IV-type with a H3-type hysteresis loops indicating that the pore size is close to the mesoporous structure for TiO2@Fe3O4-carbon-300. The BET surface area and total pore volume are calculated to be 309 m2 g−1 and 0.14 cm3 g−1, respectively. In addition, the TiO2@Fe3O4-carbon-300 sample has a wide pore size distribution among the mesopores scale from 3.5 nm to 20 nm, as shown in the inset. Besides, several important structure parameters such as BET surface area and pore volume obtained from N2 adsorption–desorption isotherms are listed in Table 2. Compared with the as-prepared samples, the TiO2@Fe3O4-carbon-300 has the largest BET specific surface area values (309.4217 m2 g−1). Larger surface area may lead to the better photocatalytic activity.
Table 2 Several important structure parameters of TiO2@Fe3O4-carbon
| Sample |
BET surface area (m2 g−1) |
Pore volume (cm3 g−1) |
Pore size (nm) |
| TiO2@Fe3O4-carbon-200 |
201.0213 |
0.073424 |
2.69435 |
| TiO2@Fe3O4-carbon-300 |
309.4217 |
0.140442 |
1.81554 |
| TiO2@Fe3O4-carbon-400 |
60.5952 |
0.052117 |
3.44033 |
| TiO2@Fe3O4-carbon-500 |
26.1874 |
0.030255 |
4.62138 |
| TiO2@Fe3O4-carbon-600 |
10.1744 |
0.019835 |
7.79791 |
3.8 Photocatalytic process
In order to investigate the desired time for adsorption of TC with TiO2@Fe3O4-carbon photocatalyst, the experiment was carried out in accordance with 2.5, and the value of adsorption capacity was shown in Fig. 8a. It could be clearly seen that, before 30 min, the value of adsorption capacity increased significantly. While after 30 min, the value of adsorption capacity fluctuated almost no change, and the reactive process was adsorption–desorption–adsorption. Thus, at the following experiments, 30 min was chosen as the desired time to use.
 |
| | Fig. 8 Adsorption curves of TiO2@Fe3O4-carbon photocatalyst with different temperatures (a) and tetracycline photodegradation efficiency of prepared TiO2@Fe3O4-carbon photocatalyst with different temperatures (b) and tetracycline photodegradation efficiency of prepared TiO2@Fe3O4 photocatalyst with different temperatures (c). | |
The photocatalytic activities of as-prepared samples are evaluated by the degradation of TC under simulated solar light irradiation. From degradation of TC over the TiO2@Fe3O4-carbon samples with the different temperature in Fig. 8b, the TiO2@Fe3O4-carbon-300 exhibits the highest photocatalytic activity (82.6%). We can clearly see from the figure, the catalytic efficiency is in the order of TiO2@Fe3O4-carbon-600 < TiO2@Fe3O4-carbon-500 < TiO2@Fe3O4-carbon-400 < TiO2@Fe3O4-carbon-200 < TiO2@Fe3O4-carbon-300. It is showed that when the temperature exceeds 300 °C, photocatalytic activity and decreased with the increase of temperature. The reason may be that the rise of temperature and the decrease of carbon content, lead to reduce of the electron transfer, reduce the photocatalytic activity. Fig. 8c shows the photocatalytic performance of TiO2@Fe3O4 samples at different temperatures under simulated solar light irradiation. When the temperature is 300 °C, TiO2@Fe3O4-400-300 exhibits the highest photocatalytic activity. The catalytic efficiency is in the order of TiO2@Fe3O4-600 < TiO2@Fe3O4-500 < TiO2@Fe3O4-400 < TiO2@Fe3O4-200 < TiO2@Fe3O4-300. The results show that the activity of TiO2@Fe3O4-carbon catalyst is higher than that of TiO2@Fe3O4 catalyst.
3.9 Stability of composite photocatalysts
Both the photocatalytic efficiency and stability are the important factors to measure the quality of the catalysts. Because of the photocorrosion or photodissolution might occur on the photocatalyst surface in the photocatalytic process. Therefore, we examined the stability of TiO2@Fe3O4-carbon-300 with the optimum value by photodegrading TC for 6 cycles under the same conditions. As shown in Fig. 9a, the catalysts retained a high photocatalytic activity with no decrease during the photodegradation of TC, even after 6 cycles, indicating that TiO2@Fe3O4-carbon-300 was relatively stable. The difference in crystal structure of as obtained samples before and after degradation of TC was compared by examining XRD patterns in Fig. 9b. It is almost the same in the position of diffraction peaks shown from the figure. From the above all, the TiO2@Fe3O4-carbon-300 exhibited relatively good stability during the photocatalytic reaction.
 |
| | Fig. 9 Repeated experiments of photocatalytic degradation of TC by the prepared TiO2@Fe3O4-carbon-300 (a) and XRD patterns of TiO2@Fe3O4-carbon-300 before TC degradation and after TC degradation for the sixth cycle (b). | |
4. Photocatalytic mechanism
It has been widely acknowledged that ˙O2−, h+ and ˙OH are the major factors for the photocatalytic oxidation. Fig. 10 displays the trapping experiment of active species during the photocatalytic reaction. For investigation of ˙O2−, the experiment was started by adding 0.5 g L−1 TiO2@Fe3O4-carbon-300 catalyst and 1 mmol BQ (a quencher of ˙O2−) for photodegradation TC, and the concentration was measured by a UV-Vis spectrophotometer with the maximum absorption wavelength at 357 nm. While for the investigation of h+ and ˙OH, the steps were similar with the procedure of the research of ˙O2−, but adding 1 mmol TEOA (a quencher of h+) and IPA (a quencher of ˙OH) instead of BQ,47 respectively. From the above results, the photocatalytic efficiency of TC was 82.6% without adding quencher. With the introduction of TEOA, the photocatalytic efficiency dropped to 20%. However, when the IPA and BQ were added into reaction solution, the degradation efficiencies of TC were 60% and 38%, respectively. These results further confirm that holes are the primary active species in this system, while superoxide radicals and hydroxyl radicals are also involved.
 |
| | Fig. 10 Photodegradation rates of TC over TiO2@Fe3O4-carbon-300 in the present of different scavengers. | |
To further verify the formation of active species involved in the photocatalytic process, the ESR spin-trap technique (with DMPO) was employed to characterize the photo-generated reactive oxygen species over TiO2@Fe3O4-carbon-300 under simulated solar light irradiation. The characteristic four peaks of DMPO–˙OH with intensity 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 can be observed in the ESR signal (Fig. 11a), and this is similar to spectra reported by other researchers,48–50 elucidating that ˙OH radicals were generated on the electrode under irradiation and no signals were observed in dark condition. Besides, it is well known that oxygen can easily trap an electron from the conduction band of TiO2 forming superoxide. The formation of superoxide radicals were also examined by DMPO spin trapping ESR techniques in methanolic media. Fig. 11b displays the ESR spectrum of DMPO–˙O2− simulated under light irradiation and the characteristics peaks can be observed in the TiO2@Fe3O4-carbon electrode while no signals in the dark, showing that superoxide radicals were efficiently generated over TiO2@Fe3O4-carbon under simulated solar light irradiation.51
1 can be observed in the ESR signal (Fig. 11a), and this is similar to spectra reported by other researchers,48–50 elucidating that ˙OH radicals were generated on the electrode under irradiation and no signals were observed in dark condition. Besides, it is well known that oxygen can easily trap an electron from the conduction band of TiO2 forming superoxide. The formation of superoxide radicals were also examined by DMPO spin trapping ESR techniques in methanolic media. Fig. 11b displays the ESR spectrum of DMPO–˙O2− simulated under light irradiation and the characteristics peaks can be observed in the TiO2@Fe3O4-carbon electrode while no signals in the dark, showing that superoxide radicals were efficiently generated over TiO2@Fe3O4-carbon under simulated solar light irradiation.51
 |
| | Fig. 11 DMPO spin-trapping ESR spectra with TiO2@Fe3O4-carbon-300 samples in the dark and under simulated solar light irradiation for DMPO–˙OH in aqueous dispersion (a), DMPO–˙O2− in methanol dispersion (b). | |
The schematic illustration of the mechanism of the enhancement of photocatalytic activity for TiO2@Fe3O4-carbon is summarized in Fig. 12. Firstly, when TiO2@Fe3O4-carbon is exposed to simulated solar light irradiation, the TiO2 produced electrons and holes (eqn (1)). Then, the electrons on conduction band of the TiO2 rapidly transfer to carbon surface (eqn (2)). Secondly, since O2 may be adsorbed on the surface of carbon, the e− in the carbon reacts with O2 to produce the active species ˙O2− (eqn (3)), then ˙O2− may also react with H+ to further produce ˙OH, which oxidizes the adsorbed TC directly on the surface (eqn (4)). At the same time, the holes from TiO2 react with H2O to further produce ˙OH (eqn (5)). Finally, the h+, ˙O2− and ˙OH all can oxidize TC (eqn (6)). Based on the above analysis, the charge carrier transfers and degradation reactions are proposed as follows:
| | |
TiO2 + hν → TiO2 (e− + h+)
| (1) |
| | |
e− (TiO2) → e− (carbon)
| (2) |
| | |
h+, ˙O2−, ˙OH + TC → CO2 + H2O + other molecules
| (6) |
 |
| | Fig. 12 The schematic illustration of the mechanism of the activation of photocatalytic activity for TiO2@Fe3O4-carbon. | |
5. Conclusions
A stable core–shell structured TiO2@Fe3O4 based on carbon was success-fully prepared using a simple method and using yeast as a source of carbon. The TiO2@Fe3O4-carbon composite catalyst not only retains the recycled magnetic property, but also improves photocatalytic activity and stability for degradation of TC under simulated solar light irradiation, which are of benefit to the application and recovery of the catalysts. When the temperature is 300 °C, the highest photocatalytic activity, can reach 82.6%. The prepared magnetic composite catalyst has repeat-ability and no obvious decline in photocatalytic efficiency was observed after 6 repeated cycles. Moreover, the free radicals were also detected by ESR and the photodegradation of TC were mainly associated with ˙O2− and the photogenerated holes. The results of the study showed the feasible and potential use of the TiO2@Fe3O4-carbon composites in photocatalytic degradation of antibiotic pollutants.
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (No. 21306068, 21407064 and 21546013), the Natural Science Foundation of Jiangsu Province (No. K20130487, BK20140532 and BK20150536), the Postdoctoral Science Foundation of Jiangsu Province (No. 1501102B).
References
- H. Barndõk, D. Hermosilla, C. Han, D. D. Dionysiou, C. Negro and Á. Blanco, Appl. Catal., B, 2016, 180, 44–52 CrossRef.
- T. Bi, J. Wan, S. Yang, X. Yu and F. Ma, Nano, 2015, 10, 1550076 CrossRef CAS.
- X. Cheng, H. Liu, Q. Chen, J. Li and P. Wang, Carbon, 2014, 66, 450–458 CrossRef CAS.
- C. Liu, J. Tang, H. M. Chen, B. Liu and P. Yang, Nano Lett., 2013, 13, 2989–2992 CrossRef CAS PubMed.
- Z. Lu, X. Zhao, Z. Zhu, Y. Yan, W. Shi, H. Dong, Z. Ma, N. Gao, Y. Wang and H. Huang, Chem.–Eur. J., 2015, 21, 18528–18533 CrossRef PubMed.
- X. Chen, S. Shen, L. Guo and S. S. Mao, Chem. Rev., 2010, 110, 6503–6570 CrossRef CAS PubMed.
- Y. Yin and D. Talapin, Chem. Soc. Rev., 2013, 42, 2484–2487 RSC.
- P. A. Soares, T. F. Silva, D. R. Manenti, S. M. Souza, R. A. Boaventura and V. J. Vilar, Environ. Sci. Pollut. Res., 2014, 21, 932–945 CrossRef CAS PubMed.
- Z. Lu, Z. Zhu, D. Wang, Z. Ma, W. Shi, Y. Yan, X. Zhao, H. Dong, L. Yang and Z. Hua, Catal. Sci. Technol., 2016, 6, 1367–1377 CAS.
- Z. Lu, F. Chen, M. He, M. Song, Z. Ma, W. Shi, Y. Yan, J. Lan, F. Li and P. Xiao, Chem. Eng. J., 2014, 249, 15–26 CrossRef CAS.
- A. Fujishima and K. Honda, Nature, 1972, 238, 37–38 CrossRef CAS PubMed.
- K. Yasui, T. Isobe, S. Matsushita and A. Nakajima, J. Mater. Sci., 2013, 48, 2290–2298 CrossRef CAS.
- N. K. Youn, J. E. Heo, O. S. Joo, H. Lee, J. Kim and B. K. Min, J. Hazard. Mater., 2010, 177, 216–221 CrossRef CAS PubMed.
- D. Qi, M. Xing and J. Zhang, J. Phys. Chem. C, 2014, 118, 7329–7336 CAS.
- C. W. Lai and S. Sreekantan, Mater. Sci. Semicond. Process., 2013, 16, 947–954 CrossRef CAS.
- C. W. Lai and S. Sreekantan, Nano, 2012, 7, 1250051 CrossRef.
- H. Li, X. Zhang, X. Cui and Y. Lin, J. Nanosci. Nanotechnol., 2012, 12, 1806–1811 CrossRef CAS PubMed.
- V. Scuderi, G. Impellizzeri, L. Romano, M. Scuderi, M. V. Brundo, K. Bergum, M. Zimbone, R. Sanz, M. A. Buccheri and F. Simone, Nanoscale, 2014, 6, 11189–11195 RSC.
- V. Scuderi, G. Impellizzeri, L. Romano, M. Scuderi, G. Nicotra, K. Bergum, A. Irrera, B. G. Svensson and V. Privitera, Nanoscale Res. Lett., 2014, 9, 1–7 CrossRef CAS PubMed.
- S. Malato, P. Fernández-Ibáñez, M. Maldonado, J. Blanco and W. Gernjak, Catal. Today, 2009, 147, 1–59 CrossRef CAS.
- G. Impellizzeri, V. Scuderi, L. Romano, E. Napolitani, R. Sanz, R. Carles and V. Privitera, J. Appl. Phys., 2015, 117, 105308 CrossRef.
- Y. H. Ng, S. Ikeda, M. Matsumura and R. Amal, Energy Environ. Sci., 2012, 5, 9307–9318 CAS.
- X. Zhang, M. Zhou and L. Lei, Mater. Res. Bull., 2005, 40, 1899–1904 CrossRef CAS.
- A. H. El-Sheikh, A. P. Newman, H. Al-Daffaee, S. Phull, N. Cresswell and S. York, Surf. Coat. Technol., 2004, 187, 284–292 CrossRef CAS.
- Y. Li, X. Li, J. Li and J. Yin, Water Res., 2006, 40, 1119–1126 CrossRef CAS PubMed.
- J. Hou, S. Jiao, H. Zhu and R. Kumar, CrystEngComm, 2011, 13, 4735–4740 RSC.
- H. Zhang, X. Lv, Y. Li, Y. Wang and J. Li, ACS Nano, 2009, 4, 380–386 CrossRef PubMed.
- J. Pouilleau, D. Devilliers, F. Garrido, S. Durand-Vidal and E. Mahé, J. Mater. Sci. Eng. B, 1997, 47, 235–243 CrossRef.
- S. Karuppuchamy, K. Nonomura, T. Yoshida, T. Sugiura and H. Minoura, Solid State Ionics, 2002, 151, 19–27 CrossRef CAS.
- F.-D. Duminica, F. Maury and F. Senocq, Surf. Coat. Technol., 2004, 188, 255–259 CrossRef.
- C.-S. Kuo, Y.-H. Tseng, C.-H. Huang and Y.-Y. Li, J. Mol. Catal. A: Chem., 2007, 270, 93–100 CrossRef CAS.
- B. Y. Jia, L. Y. Duan, C. L. Ma and C. M. Wang, Chin. J. Chem., 2007, 25, 553–557 CrossRef CAS.
- J.-H. Liu, R. Yang and S.-M. Li, J. Environ. Sci., 2006, 18, 979–982 CrossRef CAS.
- R. Nacco and E. Aquarone, Carbon, 1978, 16, 31–34 CrossRef CAS.
- F. Shi, Y. Li, Q. Zhang and H. Wang, Int. J. Photoenergy, 2012, 2012 DOI:10.1155/2012/365401.
- J. Bao, W. Chen, T. Liu, Y. Zhu, P. Jin, L. Wang, J. Liu, Y. Wei and Y. Li, ACS Nano, 2007, 1, 293–298 CrossRef CAS PubMed.
- Y. Shao, C. Cao, S. Chen, M. He, J. Fang, J. Chen, X. Li and D. Li, Appl. Catal., B, 2015, 179, 344–351 CrossRef CAS.
- A. Banerjee, R. Gokhale, S. Bhatnagar, J. Jog, M. Bhardwaj, B. Lefez, B. Hannoyer and S. Ogale, J. Mater. Chem., 2012, 22, 19694–19699 RSC.
- T. Bi, J. Wan, S. Yang, X. Yu and F. Ma, Nano, 2015, 10, 1550076 CrossRef CAS.
- G. Liu, F. He, J. Zhang, L. Li, F. Li, L. Chen and Y. Huang, Appl. Catal., B, 2014, 150–151, 515–522 CrossRef CAS.
- K. Krishnamoorthy, M. Veerapandian, R. Mohan and S.-J. Kim, Appl. Phys. A: Mater. Sci. Process., 2012, 106, 501–506 CrossRef CAS.
- S. Kim, M. Kim, Y. K. Kim, S.-H. Hwang and S. K. Lim, Appl. Catal., B, 2014, 148, 170–176 CrossRef.
- S. Liu, H. Sun, S. Liu and S. Wang, Chem. Eng. J., 2013, 214, 298–303 CrossRef CAS.
- X. Nie, G. Li, P.-K. Wong, H. Zhao and T. An, Catal. Today, 2014, 230, 67–73 CrossRef CAS.
- Q. He, Z. Zhang, J. Xiong, Y. Xiong and H. Xiao, Opt. Mater., 2008, 31, 380–384 CrossRef CAS.
- W. Wu, J. Changzhong and V. A. Roy, Nanoscale, 2015, 7, 38–58 CAS.
- X. Liu, Y. Yan, Z. Da, W. Shi, C. Ma, P. Lv, Y. Tang, G. Yao, Y. Wu and P. Huo, Chem. Eng. J., 2014, 241, 243–250 CrossRef CAS.
- J. Su, P. Geng, X. Li, Q. Zhao, X. Quan and G. Chen, Nanoscale, 2015, 7, 16282–16289 RSC.
- Y. Lu, H. Yu, S. Chen, X. Quan and H. Zhao, Environ. Sci. Technol., 2012, 46, 1724–1730 CrossRef CAS PubMed.
- Y. Hou, X. Li, Q. Zhao, G. Chen and C. L. Raston, Environ. Sci. Technol., 2012, 46, 4042–4050 CrossRef CAS PubMed.
- J. Su, L. Zhu and G. Chen, Appl. Catal., B, 2016, 186, 127–135 CrossRef CAS.
|
| This journal is © The Royal Society of Chemistry 2016 |
Click here to see how this site uses Cookies. View our privacy policy here. ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHCOONa, Na acrylate), sodium acetate (CH3COONa, NaOAc), benzoquinone (BQ), isopropanol (IPA), triethanolamine (TEOA), 5,5-diamethyl-1-pyrroline N-oxide (DMPO) were obtained from Aladdin. Tetrabutyl titanate (CR), hydrochloric acid (HCl, AR), alcohol (AR) were purchased from Sinopharm Chemical Reagent Co., Ltd. All of the chemicals were analytical grade and used directly without further purification.
CHCOONa, Na acrylate), sodium acetate (CH3COONa, NaOAc), benzoquinone (BQ), isopropanol (IPA), triethanolamine (TEOA), 5,5-diamethyl-1-pyrroline N-oxide (DMPO) were obtained from Aladdin. Tetrabutyl titanate (CR), hydrochloric acid (HCl, AR), alcohol (AR) were purchased from Sinopharm Chemical Reagent Co., Ltd. All of the chemicals were analytical grade and used directly without further purification.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHCOONa (3.4 g), NaOAc (3.4 g) were added into a mixture of EG (22.5 mL) and DEG (22.5 mL) under ultrasonication for about 1 h. The homogeneous solution obtained was transferred to a Teflon-lined stainless-steel autoclave and sealed to heat at 200 °C. After reaction for 10 h, the autoclave was cooled to room temperature. The Fe3O4/yeast obtained was washed several times with ethanol and water, and then dried in vacuum at 65 °C for 10 h.
CHCOONa (3.4 g), NaOAc (3.4 g) were added into a mixture of EG (22.5 mL) and DEG (22.5 mL) under ultrasonication for about 1 h. The homogeneous solution obtained was transferred to a Teflon-lined stainless-steel autoclave and sealed to heat at 200 °C. After reaction for 10 h, the autoclave was cooled to room temperature. The Fe3O4/yeast obtained was washed several times with ethanol and water, and then dried in vacuum at 65 °C for 10 h.




![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 can be observed in the ESR signal (Fig. 11a), and this is similar to spectra reported by other researchers,48–50 elucidating that ˙OH radicals were generated on the electrode under irradiation and no signals were observed in dark condition. Besides, it is well known that oxygen can easily trap an electron from the conduction band of TiO2 forming superoxide. The formation of superoxide radicals were also examined by DMPO spin trapping ESR techniques in methanolic media. Fig. 11b displays the ESR spectrum of DMPO–˙O2− simulated under light irradiation and the characteristics peaks can be observed in the TiO2@Fe3O4-carbon electrode while no signals in the dark, showing that superoxide radicals were efficiently generated over TiO2@Fe3O4-carbon under simulated solar light irradiation.51
1 can be observed in the ESR signal (Fig. 11a), and this is similar to spectra reported by other researchers,48–50 elucidating that ˙OH radicals were generated on the electrode under irradiation and no signals were observed in dark condition. Besides, it is well known that oxygen can easily trap an electron from the conduction band of TiO2 forming superoxide. The formation of superoxide radicals were also examined by DMPO spin trapping ESR techniques in methanolic media. Fig. 11b displays the ESR spectrum of DMPO–˙O2− simulated under light irradiation and the characteristics peaks can be observed in the TiO2@Fe3O4-carbon electrode while no signals in the dark, showing that superoxide radicals were efficiently generated over TiO2@Fe3O4-carbon under simulated solar light irradiation.51








