Energy levels of metal porphyrins upon molecular alignment during layer-by-layer electrostatic assembly: scanning tunneling spectroscopy vis-à-vis optical spectroscopy
Biswajit Kundua,
Sudipto Chakrabartia,
Michio M. Matsushitab and
Amlan J. Pal*a
aDepartment of Solid State Physics, Indian Association for the Cultivation of Science, Jadavpur, Kolkata 700032, India. E-mail: sspajp@iacs.res.in
bDepartment of Chemistry & Research Center for Materials Science, Nagoya University, Furo-cho, Chikusa-ku, Nagoya 464-8602, Japan
First published on 9th May 2016
Abstract
We report ultrathin-film formation of metal-porphyrins with their molecular plane aligned parallel to the substrate. Such an alignment has been achieved through application of an external magnetic field to a monolayer of the porphyrin derivatives followed by immobilization of the molecules with a layer of a polyion. The orientation of metal-porphyrin molecules in a monolayer responded to the magnetic field due to their anisotropic magnetic moment arising out of unpaired d-electrons of the central atom. In this work, we compared characteristics of different metal-porphyrin molecules in their aligned and unaligned forms. Scanning tunneling spectroscopy (STS) of a monolayer of the porphyrins were recorded that in turn yielded density of states (DOS) from which highest occupied molecular orbitals (HOMO) and lowest unoccupied molecular orbitals (LUMO) could be located. We observed that the energies responded upon alignment of the molecules; transport gap of metal porphyrins representing both Soret and Q-bands decreased in the aligned films due to an interaction of the molecular planes with the electrode. We compared the transport gap from STS and optical gap representing Soret and Q-bands in films of aligned and unaligned molecules.
1 Introduction
Layer-by-layer (LbL) electrostatic assembly of polyions opened up possibilities to form ultrathin-films for a range of electronic and opto-electronic applications.1,2 The film deposition technique, which is based on sequential adsorption of moieties having an opposite nature of ions, has been applied to a range of materials, such as polyions, conjugated organics, quantum dots of metal and compound semiconductors encapsulated with suitable stabilizers, and so forth.1–4 Since the technique depended on surface charge reversal during the adsorption of anionic and cationic layers in sequence, the morphology of the films depended strongly on the pH of the ionic species in solutions and the functional groups attached to the molecules/polymers/nanostructures.4The morphology of LbL films could also be controlled through chemisorption on a functioned surface via a molecule–surface interaction5,6 and also with the aid of external forces.7–11 Electric field expectedly affected the moieties due to their ionic nature.7,8 For molecules having an anisotropic magnetic moment, a magnetic field can be applied during the film formation process to align the molecules.9–11 Alignment of molecular planes could be achieved since magnetization in such molecules is associated with their molecular structure. Alignment of the molecules had to be immobilized thorough electrostatic binding with a layer of oppositely charged polyion. Depending on the direction of external magnetic field applied to the (magnetic) molecular layer, molecular-alignment to a particular direction could be accomplished. That is, the LbL deposition technique allowed one to achieve a control over the alignment of molecular planes in thin-films.
Thin-films deposited through LbL technique are being used in different (opto)electronic devices ranging from light-emitting diodes12 to photovoltaic devices13 primarily due to the uniformity of film-thickness achieved in this method. The nature of electronic and opto-electronic devices that can be formed with the ultrathin-films primarily depended on the energy levels of the active materials and work-function of metal electrodes that are often depicted in the form of an “energy level diagram”; the other physical parameters, such as extinction coefficient, electrical conductivity, carrier mobility, and so forth contributed in determining efficiency of such devices. Determination of energy of highest occupied molecular orbitals (HOMO) and lowest unoccupied molecular orbitals (LUMO) has hence remained an absolutely important task. Apart from cyclic voltammetry,14,15 scanning tunneling spectroscopy (STS) has been an effective tool to locate energy levels of the active materials. While scanning tunneling microscopy (STM) provides morphology of molecules in assemblies,5,6,16–19 the STS allows one to determine differential conductance (dI/dV) of organic semiconductors and inorganic nanostructures alike that has correspondence to the material's density of states (DOS).20–25 In this report, we present our work on locating HOMO and LUMO energies of a range of porphyrins having a metal from the first (3d) transition series as the central atom. Since the metal porphyrins could be aligned during the adsorption process, we aimed to bring out the role of molecules' alignment on their HOMO and LUMO energies when the molecules formed a monolayer on an electrode.
2 Experimental
A Materials
While tetraphenylporphyrin tetrasulfonic acid hydrate (TPPS hydrate) was purchased from Tokyo Chemical Industry, manganese(II) acetate tetrahydrate, nickel(II) acetate tetrahydrate, and cobalt(II) chloride hexahydrate were purchased from Merck India Private Limited. Dimethyl formamide (DMF) and poly(allylamine hydrochloride) (PAH) were procured from Aldrich Chemical Co. The PAH had an average molecular weight of 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1. All the materials were used without further purification.
000 g mol−1. All the materials were used without further purification.
B Synthesis of tetrasodium salt of metal porphyrins
To synthesize tetrasodium salt of a metal porphyrin, we have followed reported routes with certain modifications.26 In a particular synthesis, the starting compound was TPPS hydrate. Solutions of 0.05 mM of TPPS hydrate and 0.5 mM of the corresponding metal salt were mixed along with 1 mL of DMF. The mixture was taken in a 20 mL round-bottom reaction flask and heated to 80 °C for 36 h. After completion of the reaction, pH of the solution was adjusted to 1 by adding HCl that led to precipitation of the compound. The precipitate was then filtered out and dissolved in 1 M Na2CO3 solution to form tetrasodium salt of the metal porphyrin. The solution was then evaporated in a rotary evaporator to obtain a dry precipitate of the metal porphyrin derivative.C Layer-by-layer (LbL) film deposition
To form LbL films of TPPS hydrate and the metal porphyrin derivatives (metal: Mn, Co, Ni), all of which were anionic in nature, we have used PAH as the polycation. Concentration of PAH bath was 5 × 10−3 M based on its repeat units; concentration of the porphyrins was also the same. LbL films were deposited through alternate adsorption of an anion and the polycation on precleaned quartz substrates for 15 min each followed by a couple of washes in-between in deionized water. A sequence of the processes completed formation of one bilayer of a porphyrin. The dipping sequence was repeated in cycles to obtain a desired number of bilayers of porphyrin LbL films. It may be stated here that the LbL deposition process is based on surface charge reversal during adsorption of a monolayer.D LbL assembly with alignment of molecular orientation
In addition to conventional LbL films of the porphyrins, we formed their LbL films with molecules aligned parallel to the substrate. This particular orientation has been achieved by applying an external magnetic field after absorption of the molecules or formation of a monolayer. It may be stated here that anisotropic magnetic moment appears in metal porphyrins due to unpaired d-electrons of the central atom;27,28 due to axial interactions in the functional molecules, the moment is expected to be perpendicular to the molecular plane.29 In practice, to align the molecules in LbL films, we first adsorbed a monolayer of metal-porphyrin molecules followed by the usual rinsing process. The film was then placed in a magnetic field of 500 mT with the direction of the magnetic field being perpendicular to the substrate (and away from it), which was kept in a suspended position in an empty beaker. This allowed alignment of the molecules with their plane parallel to the substrate. After 2 min, a 5 mM aqueous solution of PAH was poured slowly in the beaker so that the film became completely immersed in PAH solution. Electrostatic adsorption of PAH was then allowed to continue for 15 min in presence of the magnetic field. The polycation immobilized alignment of the molecules through an electrostatic binding process. The field was then turned off and the film was removed from the polycation solution followed by the rinsing protocol as described earlier. This completed adsorption of a bilayer of porphyrin molecules with the “molecules aligned” parallel to the substrate. The dipping process was then repeated in sequence to obtain a desired number of LbL layers. Schematic representation of LBL film deposition process with “molecules aligned” parallel to the substrate is shown in Fig. 1. | ||
| Fig. 1 Schematic representation of LbL deposition process to deposit metal-porphyrin molecules with their plane parallel to the substrate (“molecules aligned”). | ||
E Characterization of LbL films
The LbL films were characterized by optical absorption spectroscopy. Growth process of conventional (unaligned) and aligned films was monitored by recording optical absorption spectrum after adsorption of each layer. Monolayer of the materials in both unaligned and aligned configurations was characterized in a scanning tunneling microscope (Nanosurf Easyscan2). For such measurements, the monolayer was formed on an highly arsenic-doped n-silicon substrate which had a resistivity of 3–10 mΩ cm. Tip-approaching condition was 0.4 nA at 2.0 V for all the systems so that the influence of tip remained invariant in the measurements. Tunneling current was measured with a Pt/Ir (80%/20%) tip. Here, the current was recorded after disabling the scanning and feedback controls. From tunneling current, its numerical derivative (dI/dV) was evaluated that has a correspondence to the density of states (DOS) of the materials. With bias being applied to the tip, peaks in positive voltages of DOS spectrum represented HOMO levels from which electrons could be withdrawn. Similarly, LUMO energies were represented in the form of peaks at negative voltages, at which electrons could be injected to the LUMO levels of the molecules. Tunneling current was measured at many points on each monolayer, so that a histogram of HOMO and LUMO energies of a monolayer could be plotted from DOS spectra of the measurements.3 Result and discussion
A Optical absorption spectra of (metal) porphyrins
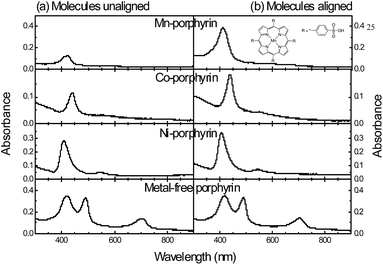
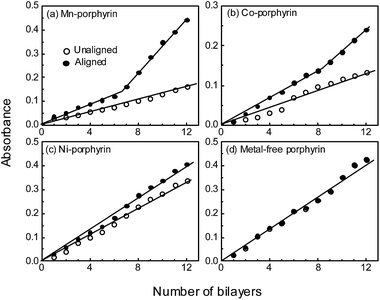
In Fig. 2(a), we show optical absorption spectra of the porphyrins in LbL films. The spectra are similar to those of a number of other porphyrins.30 The films in general exhibited two bands: the strong absorption band at the high energy region corresponded to the characteristic Soret band of porphyrins. The band at the lower energy, which is known as the Q band, was prominent in the metal-free porphyrin. The band shrunk in metal porphyrins especially if the metal had a large number of unpaired d-electrons (hypsoporphyrins). Compared to the π–π* absorption band in aqueous solution, the spectra of LbL films showed a red-shift which has been attributed to strong interactions between individual molecules in densely packed films.31,32Growth of LbL films was monitored by recording optical absorption spectrum after adsorption of each monolayer. We did the measurements for conventional LbL films and also when the metal porphyrin molecules were aligned with the aid of an external magnetic field. Here, the easy-magnetization axis of the metal-porphyrins having an anisotropic moment is perpendicular to the molecular plane. In Fig. 2(b), we show optical absorption spectra of the porphyrins in LbL films, which was formed by applying a magnetic field to align the porphyrin molecules in the films. The absorbance was higher for the metal porphyrins implying that the aligned molecules, when immobilized through a layer of polycation (PAH), augmented the proceeding adsorption process. Since we recorded spectrum after adsorption of each bilayer for both the cases, we plotted absorbance of Soret band of the two types of LbL films as a function of number of bilayers (Fig. 3). Absorbance of the Soret band (and also of the Q band) increased linearly with the bilayer number indicative of a stepwise and regular deposition of porphyrin in each cycle. The figures moreover show that the adsorption process was accelerated when the molecules were aligned. That is, in magnetization vector assisted LbL deposition, the assembly process relied on both electrostatic and magnetic forces of attraction.
The plot clearly brings out the enhancement in optical absorption due to an increase in the adsorption process during LbL film formation. Since the augmentation in LbL deposition process is due to anisotropy in magnetization vector associated to the unpaired d-electrons of the central atom, the enhancement in the adsorption process (and optical absorption) is expected to depend on the metal (number of unpaired d-electrons or degree of anisotropy). The percent of increase in absorbance for any particular number of LbL layers depended on the central atom. The anisotropy of magnetic moment of the molecule has determined the percent of increase in absorbance upon alignment of the moment. That is, the level of increase in absorbance was the highest for Mn-porphyrin and least for the nickel one; the increase was intermediate for Co-porphyrin. For the metal-free porphyrin, the magnetic field could not affect the adsorption phenomenon since the molecules do not have any anisotropy in magnetic moment to align with the external magnetic field. The data points in Fig. 3(d) therefore overlapped. For any metal-porphyrin, the level of increase in absorbance was augmented further in multilayered films, since the magnetization vector available for further adsorption became compounded in such films that in turn accelerated the LbL assembly process. The results hence inferred that the molecular plane of metal-porphyrins could be aligned in mono- and multilayered LbL films through application of an external magnetic field followed by immobilization of the molecules with a polyion in the form of electrostatic binding.
From optical absorption spectra of the porphyrins in their LbL films, we aimed to determine the optical gap of the molecules. In Fig. 4, we show (αhν)2 versus energy characteristics (Tauc plot) to obtain the gap. From the slope of the plot (Kubelka–Munk theory), optical gap of the molecules could be estimated. Presence of two gaps in porphyrins could be visible in the Tauc plots. A gap due to transition to the Soret band, appearing in the higher energy, ranged between 2.7 and 2.8 eV, which matches well with the reported values for porphyrin-like molecules.33 The transition between ground and S1 states (Q band) appeared at lower energies. The intensity of the band is known to depend on the number of unpaired d-electrons in the central atom. In Mn-porphyrin, the Q-band was weak. In part (b) of the figure, similar plots for aligned LbL films are shown. The plots show that the gaps representing Soret and Q-bands did not respond to alignment of (metal) porphyrins in LbL films, since the energy levels are intrinsic parameters of a molecule.
B Scanning tunneling spectroscopy (STS) and density of states (DOS)
We then proceeded to record STS of the monolayers with the molecules remaining unaligned or aligned with their planes parallel to the substrate (electrode). We intended to know if the location of ground state (HOMO) and the two excited states (LUMO and LUMO+1) that yield Q and Soret bands respectively, appears in the DOS spectrum. We also aimed to study if the alignment of the molecules on an electrode that would introduce an interaction between the molecule's plane and the metal would affect their HOMO and LUMO energies. Such studies are important since a monolayer of organic semiconductors is often formed on an electrode to aid carrier transport or to block carriers to suit performance of a device.We accordingly recorded tunneling current through a monolayer of the molecules in unaligned and aligned forms. For each case, we recorded the spectroscopy on many points on a monolayer since STS is a localized measurement and can record characteristics of a particular molecule or of a defect phase. Such a large number of measurements allowed us to form a histogram of HOMO and LUMO energies of the molecules in both the forms (unaligned and aligned). The difference between the energies moreover provided the band gap in terms of transport gap.
Typical DOS spectrum, histogram of the energies, and transport gap of Mn-porphyrin molecules in the two forms are shown in Fig. 5. A typical STM topography of the (aligned) molecules, as presented in Fig. 5(a), shows uniformity of monolayer films. The image remained indistinguishable even after record of STS implying that the molecules did not get detached during the record of the spectra. The functionalized molecules are expected to remain bound to the electrode surface due to a strong electrostatic force involved in the LbL deposition process. DOS spectra of other metal porphyrins have been clubbed in Fig. 6. For metal-free porphyrin, a typical DOS spectrum, histogram of HOMO and LUMO energies and the transport gap are shown in Fig. 7. Here we recorded STS of unaligned molecules only, since an external magnetic field is not expected to affect the orientation of such molecules that have no preferred easy-magnetization axis. The inferences that can be derived from the figure containing a large volume of data are as follows:
− HOMO could be observed in the form of peaks in positive voltages of the DOS spectrum.
− LUMO and LUMO+1 energies could be observed in the form of peaks in negative voltages of the DOS spectrum.
− Gap between HOMO and LUMO energies could be estimated from STS yielding the Q-band. Similarly the difference between HOMO and LUMO+1 energies yields the Soret band.
− In contrast to optical absorption spectra, presence of Q-band is evident in the STS of metal-porphyrins, as LUMO level appeared distinctly in the DOS spectra of such molecules. For metal-free porphyrins, while the optical absorption spectra yielded clear Q- and Soret bands, both LUMO and LUMO+1 energies were also well-resolved in the DOS spectra that take part in resulting the two bands following transitions from the HOMO level.
− Upon alignment of metal-porphyrin molecules parallel to the electrode, LUMO and LUMO+1 energies decreased in magnitude leading to a decrease in the transport gap. The decrease in the latter level was more than the former one. The HOMO level seemed to have experienced a little change upon alignment.
− The transport gap decreased upon alignment in metal-porphyrins. While the quantum of change in transport gap varied in metal porphyrins, the percent of change in the gap remained invariant.
− The gap decreased in the aligned monolayer since the plane of the molecules interacted with the electrode in the aligned films. Such an interaction is known to decrease the gap of metal porphyrins.34,35
− For metal-free porphyrins, neither the energy-levels nor the transport gap responded upon an attempt to align the molecules. This is expected since such molecules could not be aligned due to absence of anisotropic magnetic moment in them.
− The dispersion in energies of HOMO and LUMO levels and also the transport gap narrowed down in the aligned films. The defect states in unaligned films might have arisen out of non-uniform interactions amongst molecules and also between molecules and the substrate electrode.
C Comparison of optical and transport gaps
The results hence infer that alignment of organic molecules on an electrode affects their energy levels. The results moreover allowed us to compare the optical gap and the transport gap involving Soret and Q band due to alignment of metal-porphyrin molecules (Tables 1 & 2, respectively). Here, for both the bands, while the optical band gap was evaluated from Tauc plots (Fig. 4), transport gap was determined from the STS measurements. The tables inferred that the optical gap was in general higher than the transport gap.| Molecule | Optical gap from Tauc plots (eV) | Transport gap from DOS (eV) | ||
|---|---|---|---|---|
| Unaligned | Aligned | Unaligned | Aligned | |
| Mn-porphyrin | 2.80 | 2.79 | 2.02 | 1.90 |
| Co-porphyrin | 2.68 | 2.69 | 2.40 | 2.25 |
| Ni-porphyrin | 2.84 | 2.82 | 2.58 | 2.44 |
| Metal-free porphyrin | 2.72 | — | 1.78 | — |
| Molecule | Optical gap from Tauc plots (eV) | Transport gap from DOS (eV) | ||
|---|---|---|---|---|
| Unaligned | Aligned | Unaligned | Aligned | |
| Mn-porphyrin | 1.61 | 1.66 | 1.26 | 1.22 |
| Co-porphyrin | 1.87 | 1.98 | 2.02 | 2.01 |
| Ni-porphyrin | 2.06 | 2.05 | 2.06 | 1.96 |
| Metal-free porphyrin | 1.68 | — | 1.42 | — |
As such, the transport gap when evaluated from other experimental/simulation methods is typically larger than the optical gap measured by optical spectroscopy,34 since the transport gap involves energies of uncorrelated electrons and holes while the optical gap represents the energy to form a bound electron–hole pair. The difference is expected to match the binding energy of the excitons that forms in the material. We may add here that in STS of small organic molecules, the energies may not provide the exact location HOMO and LUMO due to several reasons.36,37 Apart from the influence of tip on molecules, defects play a major role in STS. When defects are not too few, the STM tip can very often continue to inject or withdraw carriers from the defect sites. With increase in bias in either direction, the tunneling process integrates higher energy levels; the energy levels of defect states still remained to appear as the first peak from 0 V in STS. The difference between HOMO and LUMO energies obtained from STS therefore turned out to be less than the transport gap of defect-free molecules when derived from other experimental or simulation methods.
Upon alignment of metal-porphyrin molecules, while there was a little change in the optical gap, the transport gap decreased. This is expected since the decrease in the transport gap is due to interaction of the molecular plane with the electrode in the aligned films; while recording optical absorption spectra, the molecules did not interact with an electrode since the molecules were deposited and aligned on quartz substrates. The change in the transport gap was predominant in case of Soret band (transition from HOMO to LUMO+1) as compared to that for the Q band (transition from HOMO to LUMO). The percent of change appeared to have remained invariant in the metal-porphyrins.
4 Conclusions
In conclusion, we have shown that metal porphyrins having an anisotropic magnetic moment could be aligned during LbL film formation. With the easy-magnetization axis of metal-porphyrins being perpendicular to the molecular plane and arising out of unpaired d-electrons of the central metal atom and axial interactions, we achieved LbL films with aligned metal porphyrin molecules having their molecular plane aligned parallel to the substrate. STS of a monolayer of the metal-porphyrins inferred location of HOMO, LUMO, LUMO+1 energies that are involved in the appearance of Q and Soret bands. Upon alignment of the molecules on an electrode, the LUMO and LUMO+1 energies responded leading to a decrease in the transport gap as compared to those of unaligned molecules. The percent of decrease remained invariant in different metal porphyrins. The optical gap of the molecules on quartz substrate in contrast did not decrease upon alignment of molecular planes in LbL films. We have explained the results by considering an interaction of the molecular plane with the electrode that was absent while measuring the optical gap.Acknowledgements
The authors acknowledge financial support through India-Japan Cooperative Science Programme (DST/INT/JSPS/P-172/2014). The authors also acknowledge financial support through Nano Mission projects.References
- K. Araki, M. J. Wagner and M. S. Wrighton, Langmuir, 1996, 12, 5393–5398 CrossRef CAS.
- G. Decher, Science, 1997, 277, 1232–1237 CrossRef CAS.
- S. L. Clark, E. S. Handy, M. F. Rubner and P. T. Hammond, Adv. Mater., 1999, 11, 1031–1035 CrossRef CAS.
- S. S. Shiratori and M. F. Rubner, Macromolecules, 2000, 33, 4213–4219 CrossRef CAS.
- V. Arima, E. Fabiano, R. I. R. Blyth, F. Delia Sala, F. Matino, J. Thompson, R. Cingolani and R. Rinaldi, J. Am. Chem. Soc., 2004, 126, 16951–16958 CrossRef CAS PubMed.
- M. Li, H.-J. Xu, K. Deng, Z. Shen, X.-Z. You, Q.-D. Zeng and C. Wang, J. Phys. Chem. C, 2010, 114, 1881–1884 CAS.
- S. W. Wang, L. S. Zhao, X. L. Zhang, Z. S. Shi, Z. C. Cu and Y. Q. Yang, J. Colloid Interface Sci., 2009, 336, 470–476 CrossRef CAS PubMed.
- Y. Wang, X. J. Wang, Y. Guo, Z. C. Cui, Q. Lin, W. Z. Yu, L. Y. Liu, L. Xu, D. M. Zhang and B. Yang, Langmuir, 2004, 20, 8952–8954 CrossRef CAS PubMed.
- Y. Tian, J. G. Park, Q. F. Cheng, Z. Y. Liang, C. Zhang and B. Wang, Nanotechnology, 2009, 20, 335601 CrossRef PubMed.
- S. Dey and A. J. Pal, Langmuir, 2010, 26, 17139–17142 CrossRef CAS PubMed.
- C. M. Miyazaki, A. Riul, D. S. Dos Santos, M. Ferreira, C. J. L. Constantino, M. A. Pereira-da-Silva, R. Paupitz, D. S. Galvao and O. N. Oliveira, Int. J. Mol. Sci., 2013, 14, 12953–12969 CrossRef PubMed.
- A. C. Fou, O. Onitsuka, M. Ferreira, M. F. Rubner and B. R. Hsieh, J. Appl. Phys., 1996, 79, 7501–7509 CrossRef CAS.
- J. A. He, R. Mosurkal, L. A. Samuelson, L. Li and J. Kumar, Langmuir, 2003, 19, 2169–2174 CrossRef CAS.
- F. Armijo, M. C. Goya, Y. Gimeno, M. C. Arevalo, M. J. Aguirre and A. H. Creus, Electrochem. Commun., 2006, 8, 779–784 CrossRef CAS.
- G. S. Wilson and B. P. Neri, Ann. N. Y. Acad. Sci., 1973, 206, 568–578 CrossRef CAS PubMed.
- K. C. Aus Călărasi, in Department Chemie und Pharmazie, Friedrich-Alexander-Universität Erlangen-Nürnberg, Erlangen, 2007 Search PubMed.
- T. Niu and A. Li, J. Phys. Chem. Lett., 2013, 4, 4095–4102 CrossRef CAS.
- F. Schulz, M. Ijaes, R. Drost, S. K. Haemaelaeinen, A. Harju, A. P. Seitsonen and P. Liljeroth, Nat. Phys., 2015, 11, 229–234 CrossRef CAS.
- D. Lensen and J. Elemans, Soft Matter, 2012, 8, 9053–9063 RSC.
- S. A. Krasnikov, C. J. Hanson, D. F. Brougham and A. A. Cafolla, J. Phys.: Condens. Matter, 2007, 19, 446005 CrossRef.
- M. Takada and H. Tada, Mol. Cryst. Liq. Cryst., 2006, 455, 93–97 CrossRef CAS.
- A. Bera, S. Dey and A. J. Pal, Nano Lett., 2014, 14, 2000–2005 CrossRef CAS PubMed.
- S. U. Nanayakkara, J. van de Lagemaat and J. M. Luther, Chem. Rev., 2015, 115, 8157–8181 CrossRef CAS PubMed.
- S. P. Jarvis, S. Taylor, J. D. Baran, D. Thompson, A. Saywell, B. Mangham, N. R. Champness, J. A. Larsson and P. Moriarty, J. Phys. Chem. C, 2015, 119, 27982–27994 CAS.
- J. Mielke, F. Hanke, M. V. Peters, S. Hecht, M. Persson and L. Grill, J. Am. Chem. Soc., 2015, 137, 1844–1849 CrossRef CAS PubMed.
- W. J. Kruper, T. A. Chamberlin and M. Kochanny, J. Org. Chem., 1989, 54, 2753–2756 CrossRef CAS.
- K. Awaga and Y. Maruyama, Phys. Rev. B: Condens. Matter Mater. Phys., 1991, 44, 2589–2594 CrossRef CAS.
- H. Yamada, T. Shimada and A. Koma, J. Chem. Phys., 1998, 108, 10256–10261 CrossRef CAS.
- A. R. Harutyunyan and A. A. Kuznetsov, Chem. Phys. Lett., 1995, 241, 168–172 CrossRef CAS.
- H. Donker, R. B. M. Koehorst and T. J. Schaafsma, J. Phys. Chem. B, 2005, 109, 17031–17037 CrossRef CAS PubMed.
- M. Gouterman, Optical Spectra and Electronic Structure of Porphyrins and Related Rings, Series Title: The Porphyrins: Physical Chemistry, Part A. Series, ed. David Dolphin, 1978 Search PubMed.
- M. J. Prushan, http://www.lasalle.edu/%7Eprushan/Abs%20and%20Fluor%20of%20TPPH2.pdf 2005.
- M. E. Sanchez-Vergara, J. C. Alonso-Huitron, A. Rodriguez-Gomez and J. N. Reider-Burstin, Molecules, 2012, 17, 10000–10013 CrossRef CAS PubMed.
- D. Cahen, A. Kahn and E. Umbach, Mater. Today, 2005, 8, 32–41 CrossRef CAS.
- V. C. Zoldan, R. Faccio and A. A. Pasa, Sci. Rep., 2015, 5, 8350 CrossRef PubMed.
- K. Iketaki, K. Kanai, A. Shimizu, T. Kubo, Z. H. Wang, Y. Ouchi, Y. Morita, K. Nakasuji and K. Seki, J. Phys. Chem. C, 2009, 113, 1515–1519 CAS.
- T. Yamada, M. Isobe, M. Shibuta, H. S. Kato and T. Munakata, J. Phys. Chem. C, 2014, 118, 1035–1041 CAS.
| This journal is © The Royal Society of Chemistry 2016 |