On splitting of the NICS(1) magnetic aromaticity index†
Jan Cz. Dobrowolski*ab and
Piotr F. J. Lipińskic
aInstitute of Chemistry and Nuclear Technology, 16 Dorodna Street, 03-195 Warsaw, Poland
bNational Medicines Institute, 30/34 Chełmska Street, 00-725 Warsaw, Poland. E-mail: j.dobrowolski@nil.gov.pl
cDepartment of Neuropeptides, Mossakowski Medical Research Centre Polish Academy of Sciences, 02-106 Warszawa, Poland
First published on 24th February 2016
Abstract
The NICS(1) magnetic aromaticity index is split into NICS(1) and NICS(−1) indices when the points 1 Å above and below the ring center are inequivalent by symmetry. The two indices characterize the aromaticity of the two ring faces rather than the ring itself. We propose new NICS(1) co-indices to complete aromaticity analysis.
Aromaticity is a complex molecular property described by an enumerative definition rather than a unique formula.1,2 An aromatic system must satisfy most of the following four criteria: (1) energetic, (2) geometric, (3) magnetic, and (4) chemical reactivity. Several indices evaluate these aspects of aromaticity,1,2 but the magnetic NICS3 and structural HOMA4–6 indices are referred to most often. The NICS (nucleus independent chemical shift) index is a reverse chemical shift value calculated either in the middle of the ring or 1 Å above it. A negative NICS indicates aromatic ring, while a positive value indicates an antiaromatic one. There are several versions of the NICS(1) index enabling a better filtering of the valence σ-orbitals' contribution to the NICS index, whose aim is to determine only the effects originating from the π-electron system.7 However, the objective of this paper is to qualitatively demonstrate the NICS(1) splitting phenomenon rather than search for very accurate values. This is also why we use the B3LYP/6-31G** level of theory reproducible in any lab.‡
Although most polycyclic aromatic hydrocarbons are planar, numerous others, e.g., helicenes,8 bowl-shaped,9,10 twisted,11 saddle-shaped,12 or strained PAHs,13 are not. Many more PAHs are predicted to be non-planar.14,15 For planar aromatics, a requirement to determine the nucleus independent chemical shift 1 Å above the ring center is unequivocal. Simply, the NICS(1) values on both sides of the ring are the same, unless the ring substituent or its surrounding is quite asymmetric. Yet, for non-planar systems, for which the point above the ring center is not equivalent by symmetry to the point below the ring, the degeneration of the two NICS(1) values is released.
So far, the NICS values have been almost always determined for the planar rings. However, for the non-planar rings they can also be determined: the coordinates of the ring center can be found as the arithmetic average of the coordinates of the ring heavy atoms. Next, the reference plane can be found by the least square fitting applied to the coordinates of the ring heavy atoms and the ring center. If the ring center is placed in the origin of the system of coordinates, then in the Ax + By + Cz + D = 0 plane, the intercept D is equal to 0, and the vector [A, B, C] is normal to the plane. This allows us to find the points 1 Å above and below the ring center.
Let us agree that the symbols −1 and 1 denote the points at the concave and the convex side of the molecule, respectively. For corannulene, −1 is inside and 1 is outside the bowl. When the convex and concave sides of a molecule cannot be clearly defined, we denote the point of the greater value with 1.
In the vast majority of aromatic rings, NICS(1) and NICS(−1) < NICS(0), while NICS(1) and NICS(−1) > NICS(0) occurs rarely, e.g., in C6(CN)6 or C6F6, where CN or F withdraw the σ-electron charge from the ring. NICS(1) > NICS(0) > NICS(−1) is common for non-planar systems. This has recently been demonstrated thoroughly for the five- and six-membered rings in corannulene and sumanene molecules, for which continuous changes of the NICS index are within a distance from the checkpoint of 2 Å below to 2 Å above a given ring.16
Here, we consider several non-planar and planar systems with and without the NICS splitting (Schemes 1 and 2). But first, let us introduce intrinsic NICS(1) co-indices: the NICS(1) average, the NICS(1) splitting, i.e., the difference between NICS(1) and NICS(−1), the asymmetry of NICS(1), and the NICS(0) bias:
| NICS(1)av = [NICS(−1) + NICS(1)]/2 | (1) |
| NICS(1)diff = NICS(1) − NICS(−1) | (2) |
| NICS(1)as = [NICS(1)/NICS(−1)] − 1 | (3) |
| NICS(0)bia = NICS(0) − NICS(1)av | (4) |
 | ||
| Scheme 1 Non-planar rings, but 4, considered in Table 1. | ||
Notice that co-indices (1)–(4) are well-defined regardless of whether the splitting occurs or not. Each of them introduces new information about the magnetic aromaticity. NICS(1)av informs about the mean magnetic aromaticity 1 Å outside the ring. As NICS(1) index contains less contributions from σ-electrons than NICS(0), the NICS(1)av co-index is unequivocal and probably better describes the magnetic aromaticity of the ring itself than NICS(0). Obviously, if the splitting does not occur, NICS(1)av = NICS(1). Also, it facilities a comparison between the splitted and unsplitted indices. NICS(1)diff is the splitting magnitude showing the difference between the magnetic aromaticities of the two ring faces. NICS(1)as tells us about the asymmetry of NICS(1) and NICS(−1) with respect to the ring center. NICS(0)bia displays a bias in the magnetic aromaticity evaluation by use of the NICS(0) or NICS(1)av value. Its sign shows whether the center of the ring indicates a greater magnetic aromaticity than the points 1 Å outside the ring. It depends on contribution of the σ-valence orbitals of the ring heavy atoms to NICS(0). The new indices are not mutually correlated, which means that they are not linearly dependent. However, the plot of NICS(1)av against NICS(1)as displays some clustering of points which requires further inspection.
Let us measure the ring non-planarity by means of T: the root mean square deviation of all ring CCCC dihedral angles:
 | (5) |
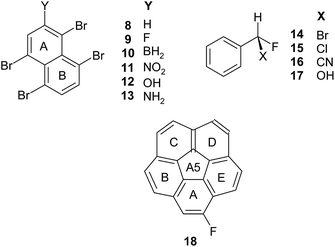
Now consider two series of compounds: (i) the non-planar systems and the reference benzene (Scheme 1), and (ii) the chiral aromatics, which may (but not necessarily) exhibit a significant non-planarity of the ring (Scheme 2).
In the group (i) of the non-planar molecules, the non-planarities of the (1)A, (2)A5, and (2)B rings, in the bowl-shaped corannulene and sumanene, are significant (T = 7.72, 8.89, and 5.05 deg, respectively, Table 1SI in ESI†) and the splittings are significant as well (NICS(1)diff = 6.3, 6.0 and 3.2 ppm, respectively). However, the (1)A5 and (2)A rings in the same systems are planar (T = 0.00) but the splittings can be even greater than before: NICS(1)diff = ca. 5 and 4.5 ppm, respectively (Table 1).
| Ring | NICS (ppm) | ||||||
|---|---|---|---|---|---|---|---|
| (−1) | (0) | (1) | (1)diff | (1)av | (0)bia | (1)as | |
| (1)A5 | −2.37 | 9.39 | 2.56 | 4.94 | 0.10 | 9.30 | −2.08 |
| (1)A | −12.77 | −7.17 | −6.49 | 6.28 | −9.63 | 2.46 | −0.49 |
| (2)A5 | −4.93 | 2.78 | 1.04 | 5.98 | −1.95 | 4.73 | −1.21 |
| (2)A | −6.15 | −3.17 | −1.66 | 4.50 | −3.91 | 0.74 | −0.73 |
| (2)B | −10.92 | −10.29 | −7.74 | 3.18 | −9.33 | −0.96 | −0.29 |
| (3)A | −9.28 | −8.24 | −9.28 | 0.00 | −9.28 | 1.04 | 0.00 |
| (3)B | −12.47 | −11.72 | −12.47 | 0.00 | −12.47 | 0.75 | 0.00 |
| (3)C | −11.11 | −9.46 | −11.11 | 0.00 | −11.11 | 1.65 | 0.00 |
| (4) | −11.31 | −9.85 | −11.31 | 0.00 | −11.31 | 1.46 | 0.00 |
| (5) | −1.48 | −1.22 | −1.48 | 0.00 | −1.48 | 0.26 | 0.00 |
| (6) | 2.09 | 5.48 | 2.09 | 0.00 | 2.09 | 3.39 | 0.00 |
| (7)A5 | −2.58 | 8.83 | 2.25 | 4.84 | −0.17 | 9.00 | −1.87 |
| (7)A | −12.98 | −9.99 | −6.85 | 6.14 | −9.92 | −0.07 | −0.47 |
| (7)B | −12.78 | −7.53 | −6.68 | 6.10 | −9.73 | 2.20 | −0.48 |
| (7)C | −12.76 | −7.22 | −6.46 | 6.30 | −9.61 | 2.39 | −0.49 |
| (8)A | −10.86 | −10.37 | −10.86 | 0.00 | −10.86 | 0.49 | 0.00 |
| (9)A | −10.83 | −11.15 | −10.77 | 0.06 | −10.80 | −0.35 | −0.01 |
| (9)B | −10.78 | −10.20 | −10.73 | 0.06 | −10.75 | 0.55 | −0.01 |
| (10)A | −10.23 | −8.73 | −9.94 | 0.28 | −10.08 | 1.36 | −0.03 |
| (10)B | −10.51 | −9.51 | −10.27 | 0.24 | −10.39 | 0.88 | −0.02 |
| (11)A | −10.37 | −10.40 | −10.23 | 0.14 | −10.30 | −0.10 | −0.01 |
| (11)B | −10.77 | −9.76 | −10.49 | 0.28 | −10.63 | 0.87 | −0.03 |
| (12)A | −10.38 | −10.41 | −10.25 | 0.14 | −10.32 | −0.09 | −0.01 |
| (12)B | −10.64 | −10.21 | −10.57 | 0.08 | −10.60 | 0.39 | −0.01 |
| (13)A | −9.83 | −9.70 | −9.79 | 0.04 | −9.81 | 0.11 | 0.00 |
| (13)B | −10.42 | −9.88 | −10.19 | 0.22 | −10.31 | 0.43 | −0.02 |
| (14) | −11.25 | −9.71 | −10.89 | 0.36 | −11.07 | 1.36 | −0.03 |
| (15) | −11.36 | −9.87 | −11.07 | 0.30 | −11.22 | 1.34 | −0.03 |
| (16) | −11.32 | −9.73 | −11.07 | 0.26 | −11.20 | 1.47 | −0.02 |
| (17) | −11.41 | −9.98 | −11.38 | 0.04 | −11.39 | 1.42 | 0.00 |
| (18)A5 | −2.46 | 9.11 | 2.42 | 4.88 | −0.02 | 9.13 | −1.98 |
| (18)A | −12.62 | −8.40 | −6.41 | 6.20 | −9.51 | 1.11 | −0.49 |
| (18)B | −12.5 | −7.03 | −6.29 | 6.20 | −9.40 | 2.37 | −0.50 |
| (18)C | −12.88 | −7.22 | −6.55 | 6.34 | −9.71 | 2.50 | −0.49 |
| (18)D | −12.67 | −7.13 | −6.40 | 6.28 | −9.53 | 2.40 | −0.50 |
| (18)E | −12.99 | −7.60 | −6.76 | 6.22 | −9.88 | 2.28 | −0.48 |
On the other hand, for the A, B and C rings in (3), NICS(1)diff = 0 despite the fact that these rings are significantly distorted due to steric hindrances of four Br substituents (T = 13.30, 12.18, and 1.97 deg, respectively). Also, for strongly non-planar cyclohexane (5) and cyclooctatetraene (6) (T = 54.63 and 78.14 deg, respectively, Table 1SI†) the splittings are null.
For a chiral molecule not superimposable on its mirror image, only translations and rotations are allowed as symmetry elements, and for finite non-periodic systems, only the latter remain.17 In the group (ii) of the chiral molecules, the unsubstituted 1,4,5,8-tetrabromonaphthalene (8), an analogue of (3), is quite non-planar (T = 8.97 deg) but it has C2 symmetry, and NICS(1)diff = 0.0. For the monosubstituted derivatives of (8), T varies from 8.79 to 11.19 deg, yet, these molecules have no symmetry elements and their NICS(1)diff values range between 0.04 and 0.28 ppm (9–13, Scheme 2, Table 1). Thus, a perturbation of symmetry by a substituent attached to the analysed ring may introduce a small but visible NICS(1) splitting. On the other hand, the non-planarity of the phenyl ring in benzene monosubstituted by chiral substituents considered in the same (most stable) conformation is small, T = 0.02 ÷ 0.33 deg. Nevertheless, the splitting varies from 0.04 to 0.36 ppm (14–17, Scheme 2, Table 1). The splitting of the A ring in highly strained, chiral, monofluoro corannulene (18), is equal to 6.20 ppm and is only slightly different from that in the A ring of achiral parent (1) and disubstituted (7).
Thus, ring non-planarity is not a necessary condition for NICS(1) splitting. Also, ring planarity does not guarantee that NICS(1)diff would not be significant. The necessary condition for the NICS(1) splitting to appear is the non-equivalence of the points 1 Å above and below the center of the ring plane by any symmetry operation of the ring. Thus, the points are magnetically inequivalent, too. Also, the molecular chirality induces some NICS(1) splitting if, and only if, the symmetry non-equivalence condition is conserved.
Now consider the new co-indices (1)–(4). For most rings considered here, NICS(0)bia is positive which means that NICS(0) is greater than NICS(1)av (Table 1). This is because in NICS(1)av, the local contributions from σ-valence orbitals are diminished,18 and aromaticity indicated by NICS(0) is smaller than that given by NICS(1)av. However, for the (2)B, (7)A, (9)A, (11)A, and (12)A rings, NICS(0)bia is negative (Table 1). For all these rings except for (2)B, this is a result of the withdrawing of σ-electrons by the –F, –NO2, and –OH substituents.19 However, for the (2)B six-membered ring in non-planar pericondensed summanene, NICS(1)bia < 0 is surprising.
Let us now consider rings with small NICS(1)av values accompanied by relatively large NICS(1)diff ones. A small NICS(1)av of an unsaturated ring may be due to a cancelation of NICS(1) and NICS(−1) values of different sign. This is the case for the five-membered rings in (1), (2), (7), and (18) (Table 1), for which NICS(1)diff exceed 4.8 ppm while the moduli of NICS(1)av are smaller than 2 ppm. Thus, from the concave side of the molecule, the ring is seen as magnetically moderately aromatic, from the convex side, it is perceived as magnetically moderately antiaromatic, and NICS(1)av close to 0 suggests that the ring itself is non-aromatic. According to the classical Hückel's rule, the number of electrons on the convex side of the ring should approach 2n, whereas, at the same time, the number of electrons on the concave side should approach 2n + 2. More generally, for all systems for which the splitting NICS(1)diff is large, the magnetic aromaticities of the two ring faces are strongly different, and the electron densities at the two ring faces are strongly different, too.
Thus, for systems with a strong non-equivalence of the points 1 Å above and below the center of the ring plane, the two NICS(1) and NICS(−1) indices characterize the magnetic aromaticities of the two ring faces rather than the aromaticity of the ring itself. It is likely that the differing aromaticities of the two molecular faces can effect in their different chemical reactions and different physical magnetic phenomena.
Observe that the NICS(0)bia and NICS(1)as indices of the (1)A5, (2)A5, (7)A5, and (18)A5 rings, are much larger than for the other rings (Table 1). However, there is a possibility that the NICS(0)bia index would be large at NICS(1)diff = 0, the NICS(1)as index vanishes at NICS(1)diff = 0. Notice also, that although both NICS(1)diff and NICS(1)as are null without splitting, they change differently in different systems. This may help in a more detailed analysis of systems with differing aromaticities between the two faces.
It is also interesting how the substituent effect influences the splitting. A comparison of the NICS(1) co-indices for 1,2-fluoro substituted and unsubstituted corannulene, (7) and (1), reveals that the NICS(1)as change is the most visible at the (7)A5 ring which is not directly substituted. On the other hand, NICS(0)bia changes as expected, the most for the (7)A ring: from 2.46 ppm for (1)A to −0.07 ppm for (7)A. This is connected with two σ-electron withdrawing –F groups17 decreasing the σ-electron contribution to NICS(0). Interestingly, the substituent effect is practically not reflected in variations of the (1)A, (7)A, (7)B and (7)C ring deformations, for which T is equal to 7.72, 7.63, 7.63, and 7.75 deg, respectively.
Five substituents of different electron donor–acceptor activity towards σ- and π-valence electrons of the core system19 were used to substitute (8) (Scheme 2). The –F group is strongly σ-electron withdrawing but weakly π-electron donating, –BH2 is σ-electron donating and strongly π-electron withdrawing, –NO2 is both σ- and π-electron withdrawing, while –OH and –NH2 are σ-electron withdrawing but they are strong π-electron donors.19 Although the deformations T, NICS(0), NICS(1)av, and NICS(0)bia of the A and B rings correlate quadratically with the pEDA(I) descriptor, the NICS(1)diff indices of the two rings do not change regularly (Fig. 1SI–5SI, ESI†). Nevertheless, for the B ring, one can observe a tendency of NICS(1)diff to increase along with the increase of both the π-electron withdrawing and π-electron donating properties of the substituent, while for the A ring the tendency is perturbed. Clearly, the irregularity is due to intramolecular interaction between the substituent and the neighboring, bulky Br atom. Notice, that in the case of monosubstituted derivatives of tetrabromonaphthalene (8), the ring deformation T correlates linearly with the Sinister-Rectus Mass Chirality Measure, SRCMMr,20–22 and thus it is a (local) measure of chirality for these compounds (Fig. 6SI†).
Let us stress at the end that not only aromaticity is a multidimensional phenomenon1,2 but also the sole magnetic aspect of aromaticity is complex. The magnetic aromaticity is connected to the ring current and its characteristics taken in selected single points, such as the ring center or 1 Å outside, probably cannot fully illustrate its composite nature. Indeed, for example a paratropic ring current, denoting antiaromaticity, has been shown to sustain in the (1)A5 ring.23 This is in agreement with the NICS(0) value reported here (Table 1). However, the prototropic ring current is more intense inside the bowl than outside it.23 The former is in contradiction to the NICS(−1) value, yet the latter is again in accord with the NICS(1) value (Table 1). Moreover, the NICS(1)av co-index suggests non-aromatic character of (1)A5. Furthermore, the NICS(−1)zz, NICS(0)zz and NICS(1)zz values determined for (1)A5 in ref. 16 are all positive suggesting antiaromatic character of the ring. Notice however, that the (prototropic) ring current is the most intense between the ring centre and the ring skeleton. Notice also that although the NICS evolution in the direction perpendicular to the ring plane is remarkable,16 in the direction parallel to the plane it is also significant.24 In fact, between the ring centre and its rim there are other local NICS extrema. This is why further studies enabling a deeper insight into the criteria of the magnetic aromaticity of the rings and finding more connections between the ring currents and the NICS values are desirable.
Finally, before taking the NICS(1) difference as a solid criterion of the two faces' different aromaticity remark that the NICS value for a single ring in a polycyclic system can be composed of several factors. One of them is connected to the fact that the NICS value is affected by ring currents in all circuits.25–27 Thus, the difference between the NICS(−1) and NICS(1) may have contributions from both σ- and π-orbitals of the substituents as well as the currents present in the ring nonplanar surrounding of the (poly)condensed rings. This aspect of the NICS(1) splitting need to be addressed and clarified in future studies.
Conclusions
In conclusion, the NICS(1) magnetic aromaticity index of a ring, measured 1 Å above or below the ring center, can be split into NICS(1) and NICS(−1) indices when the two checkpoints are non-equivalent by symmetry, and thus are magnetically inequivalent. The presence of two different NICS(1) indices suggests that NICS(1) is the ring face magnetic aromaticity index rather than the aromaticity index of the ring itself. Indeed, according to NICS(1), the outer face of some rings is (moderately) antiaromatic and the inner is (moderately) aromatic, while an average NICS(1)av close to 0 suggests the ring to be non-aromatic. We suppose that the different aromaticities of the two molecular faces can induce different chemical reactions and different physical magnetic phenomena. For the systems with the NICS(1) splitting, the NICS(1)as co-index shows an asymmetry of the NICS(1) and NICS(−1) indices with respect to the ring center. It is 0 without splitting, but when it approached −2 for the studied systems, the two ring faces had the opposite magnetic aromaticity characters.Acknowledgements
This work was supported by the National Science Centre in Poland Grant No. 2013/09/B/ST5/03664. Computational Grants G19-4 from the Interdisciplinary Centre of Mathematical and Computer Modelling (ICM) at University of Warsaw and “substeff” Grant from PL-Grid Infrastructure are gratefully acknowledged. Critical comments of anonymous referees to this paper which helped us to clarify several problems are gratefully acknowledged.Notes and references
- P. von Ragué Schleyer and H. Jiao, What is aromaticity?, Pure Appl. Chem., 1996, 68, 209–218 Search PubMed
.
- T. M. Krygowski and H. Szatylowicz, Aromaticity: what does it mean?, ChemTexts, 2015, 1–12 Search PubMed
.
- Z. Chen, C. S. Wannere, C. Corminboeuf, R. Puchta and P. von Ragué Schleyer, Nucleus-independent chemical shifts (NICS) as an aromaticity criterion, Chem. Rev., 2005, 105, 3842–3888 CrossRef CAS PubMed
.
- T. M. Krygowski and M. K. Cyrański, Structural aspects of aromaticity, Chem. Rev., 2001, 101, 1385–1420 CrossRef CAS PubMed
.
- S. Ostrowski and J. C. Dobrowolski, What does the HOMA index really measure?, RSC Adv., 2014, 4, 44158–44161 RSC
.
- J. C. Dobrowolski and S. Ostrowski, On the HOMA index of some acyclic and conducting systems, RSC Adv., 2015, 5, 9467–9471 RSC
.
- H. Fallah-Bagher-Shaidaei, C. S. Wannere, C. Corminboeuf, R. Puchta and P. von Ragué Schleyer, Org. Lett., 2006, 8, 863–866 CrossRef CAS PubMed
.
- Y. Shen and C. F. Chen, Helicenes: synthesis and applications, Chem. Rev., 2012, 112, 1463–1535 CrossRef CAS PubMed
.
- Y.-T. Wu and J. S. Siegel, Aromatic Molecular-Bowl Hydrocarbons:
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Synthetic Derivatives, Their Structures, and Physical Properties, Chem. Rev., 2006, 106, 4843–4867 CrossRef CAS PubMed
Synthetic Derivatives, Their Structures, and Physical Properties, Chem. Rev., 2006, 106, 4843–4867 CrossRef CAS PubMed .
- D. Myśliwiec and M. Stępień, Angew. Chem., Int. Ed., 2013, 52, 1713–1717 CrossRef PubMed
.
- R. Herges, Organic chemistry: aromatics with a twist, Nature, 2007, 450, 36–37 CrossRef CAS PubMed
.
- K. Y. Cheung, X. Xu and Q. Miao, Aromatic Saddles Containing Two Heptagons, J. Am. Chem. Soc., 2015, 137, 3910–3914 CrossRef CAS PubMed
.
- H. Dodziuk, Strained Hydrocarbons, Wiley-VCH, 2009 Search PubMed
.
- J. Cz. Dobrowolski, On the Belt and Moebius Isomers of the Coronene Molecule, J. Chem. Inf. Comput. Sci., 2002, 42, 490–499 CrossRef CAS PubMed
.
- D. J. Klein and A. T. Balaban, Clarology for Conjugated Carbon Nano-Structures: Molecules, Polymers, Graphene, Defected Graphene, Fractal Benzenoids, Fullerenes, Nano-Tubes, Nano-Cones, Nano-Tori, etc., Open Org. Chem. J., 2011, 5(1–M3), 27–61 CrossRef CAS PubMed
.
- A. Reisi-Vanania and A. A. Rezaei, Evaluation of the aromaticity of non-planar and bowl-shaped molecules by NICS criterion, J. Mol. Graphics Modell., 2015, 61, 85–88 CrossRef PubMed
.
- G. P. Moss, International, basic terminology of stereochemistry, Pure Appl. Chem., 1996, 68, 2193–2222 CrossRef CAS
.
- P. von Ragué Schleyer, M. Manoharan, Z.-X. Wang, B. Kiran, H. Jiao, R. Puchta and N. J. R. van Eikema Hommes, Dissected Nucleus-Independent Chemical Shift Analysis of π-Aromaticity and Antiaromaticity, Org. Lett., 2001, 3, 2465–2468 CrossRef
.
- W. P. Ozimiński and J. Cz. Dobrowolski, On the Substituent Effect. The Natural Population Analysis Approach, J. Phys. Org. Chem., 2009, 22, 769–778 CrossRef
.
- S. Ostrowski, M. H. Jamróz, J. E. Rode and J. Cz. Dobrowolski, On Stability, Chirality Measures, and Theoretical VCD Spectra of the Chiral C58X2 Fullerenes (X = N, B), J. Phys. Chem. A, 2012, 116, 631–643 CrossRef CAS PubMed
.
- M. H. Jamróz, J. E. Rode, S. Ostrowski, P. F. J. Lipiński and J. Cz. Dobrowolski, Chirality Measures of α-Amino Acids, J. Chem. Inf. Model., 2012, 52, 1462–1479 CrossRef PubMed
.
- P. F. J. Lipiński and J. Cz. Dobrowolski, Local chirality measures in QSPR: IR and VCD spectroscopy, RSC Adv., 2014, 4, 47047–47055 RSC
.
- E. Steiner, P. W. Fowler and L. W. Jenneskens, Counter-Rotating Ring Currents in Coronene and Corannulene, Angew. Chem., Int. Ed., 2001, 40, 362–366 CrossRef CAS
.
- K. E. Horner and P. B. Karadakov, Shielding in
and around Oxazole, Imidazole, and Thiazole: How Does the Second Heteroatom Affect Aromaticity and Bonding?, J. Org. Chem., 2015, 80, 7150–7157 CrossRef CAS PubMed
.
- S. Fias, S. van Damme and P. Bultinck, Multidimensionality of delocalization indices and nucleus-independent chemical shifts in polycyclic aromatic hydrocarbons II: proof of further nonlocality, J. Comput. Chem., 2010, 31, 2286–2293 CAS
.
- S. Fias, S. van Damme and P. Bultinck, Multidimensionality of delocalization indices and nucleus independent chemical shifts in polycyclic aromatic hydrocarbons, J. Comput. Chem., 2008, 29, 358–366 CrossRef CAS PubMed
.
- P. Bultinck, S. Fias and R. Ponec, Local Aromaticity in Polycyclic Aromatic Hydrocarbons: Electron Delocalization versus Magnetic Indices, Chem.–Eur. J., 2006, 12, 8813–8818 CrossRef CAS PubMed
.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr, J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, T. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, and D. J. Fox, Gaussian 09, Revision B.01, Gaussian, Inc., Wallingford CT, 2010 Search PubMed
.
- SigmaPlot for Windows Version 12.0, Systat Software, Inc., Copyright© 2011 Search PubMed
.
Footnotes |
| † Electronic supplementary information (ESI) available: The deformation parameters T and correlations between T and NICS(1) co-indices and pEDA substituent effect descriptor for (8) are given in ESI file. See DOI: 10.1039/c6ra03246j |
| ‡ The calculations were performed using Gaussian 09 suite of programs.28 The molecules were optimized (to true minima, ascertained by no imaginary frequencies) at the B3LYP/6-31G** level. The NICS values were calculated using GIAO approach. The reference ring plane in non-planar systems was determined by the least square routine using the SigmaPlot 12 program.29 |
| This journal is © The Royal Society of Chemistry 2016 |