DOI:
10.1039/C6RA03078E
(Paper)
RSC Adv., 2016,
6, 57154-57162
One-step approach for the synthesis of functionalized quinoxalines mediated by T3P®–DMSO or T3P® via a tandem oxidation–condensation or condensation reaction†
Received
2nd February 2016
, Accepted 26th February 2016
First published on 26th February 2016
Abstract
An easy and efficient propylphosphonic anhydride (T3P®)–DMSO or T3P® mediated oxidation–condensation or condensation reaction for the synthesis of quinoxalines derived from the interaction of different arrays of condensing partners with ortho-phenylene diamines (o-PDs) under simple and mild reaction conditions in one step has been reported for the first time.
Introduction
New synthetic organic transformations for the synthesis of functionalized N-containing heterocyclic small molecules from simple starting materials are of great importance in the field of synthetic organic chemistry. Quinoxalines, or benzopyrazines, in particular have been studied for their antibacterial, antiviral, anthelmintic, antiinflammatory, kinase inhibitory and anticancer activities1 and they also have a wide array of other applications.2 In addition, the quinoxaline nucleus is contained in some important antibiotics such as olaquindox, carbadox, echinomycin, levomycin and actinoleutin. Owing to their wide ranging biological activity and for technical interest, a number of synthetic strategies have been developed for the synthesis of various substituted quinoxalines. Out of these methods of synthesis, oxidation–condensation and condensation reactions are major synthetic steps used for the synthesis of the bioactive quinoxaline nucleus from readily available precursors like o-PDs and α-hydroxy ketones.3 In addition, several other methods are also available for the synthesis of quinoxalines from the reaction of phenacyl bromides,4 1,2-diketones,5 alkynes,6 epoxides,7 and arylimino oximes8 with o-PDs.
Most of the methods reported above suffer from one or more disadvantages such as poor substrate scope, harsh reaction conditions, laborious and complex work-up procedures, expensive and moisture sensitive reagents, undesirable side products and unsatisfactory yields. During the course of a recent investigation, our research group found that T3P® (propylphosphonic anhydride) can be used in the synthesis of peptides,9 as well as the synthesis of polysubstituted quinolines and naphthyridines,10 β-lactams,11 pyrimidinones using the Biginelli reaction,12 1,2,4-oxadiazoles, 1,3,4-oxadiazoles, 1,3,4-thiadiazoles13 and Fischer indoles.14 Due to T3P®’s significant properties as a water scavenger in oxidation–cyclization reactions, we recently reported several one-pot tandem approaches for the synthesis of benzimidazoles and benzothiazoles,15a as well as 4-thiazolidinones15b and imidazo[1,2-a]pyridines15c from alcohols, using DMSO–propylphosphonic anhydride (T3P®) as an oxidizing medium as well as a cyclodehydrating agent. As a continuation of our research into the synthesis of heterocycles,16 we report, herein, a new strategic one-step method for the synthesis of quinoxalines from the reaction of α-hydroxy ketones, α-halo ketones and 1,2-diketones with o-PDs using DMSO–propylphosphonic anhydride (T3P®) or propylphosphonic anhydride (T3P®) in ethyl acetate (EtOAc) at room temperature. The reaction occurred either via an oxidation followed by condensation or a simple condensation reaction. To optimize the reaction conditions, we initially selected the reaction between o-PD 1a and 2-hydroxy-1,2-diphenylethanone 2a (Scheme 1) in a mixed solvent of EtOAc![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMSO (ratio 2
DMSO (ratio 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) followed by the addition of 1 equivalent of T3P® (50% solution in EtOAc) under stirring at 0 °C, then RT for 10 h, which afforded the desired 2,3-diphenylquinoxaline 3e in 25% yield. For the same reaction we checked if different equivalents of T3P® could affect the course of the reaction and the yield of 3e. Therefore, we carried out the oxidation–condensation using various equivalents of T3P®. The best result was obtained when the reaction between 1,2-diphenylethanone and o-PD was carried out with 2.0 equivalents of T3P® in the presence of DMSO and stirred at RT for 6 h. We also studied the reaction by keeping the equivalent of T3P® (2.0 eq.) constant and varying both the temperature and time. Notably, we were not able to achieve a greater percentage yield compared to the yield obtained at RT conditions. The optimization results are summarized in Table 1. Thus, a clear optimization of the reaction conditions to obtain quinoxalines from a α-hydroxyl ketone with o-PD was revealed from our studies, which involved using 2.0 equivalents of T3P® and stirring the reaction mixture at 0 °C then RT for 6 h. The product was isolated by initially washing the reaction mixture with water followed by brine, then purified by column chromatography using 60–120 silica gel. The oxidation–condensation or cyclodehydrating properties of T3P® helped to generalize the reaction procedure. Next, we carried out reactions with a series of electronically diverse α-hydroxy ketones and o-PDs mediated by T3P® in a solvent mixture of DMSO
1) followed by the addition of 1 equivalent of T3P® (50% solution in EtOAc) under stirring at 0 °C, then RT for 10 h, which afforded the desired 2,3-diphenylquinoxaline 3e in 25% yield. For the same reaction we checked if different equivalents of T3P® could affect the course of the reaction and the yield of 3e. Therefore, we carried out the oxidation–condensation using various equivalents of T3P®. The best result was obtained when the reaction between 1,2-diphenylethanone and o-PD was carried out with 2.0 equivalents of T3P® in the presence of DMSO and stirred at RT for 6 h. We also studied the reaction by keeping the equivalent of T3P® (2.0 eq.) constant and varying both the temperature and time. Notably, we were not able to achieve a greater percentage yield compared to the yield obtained at RT conditions. The optimization results are summarized in Table 1. Thus, a clear optimization of the reaction conditions to obtain quinoxalines from a α-hydroxyl ketone with o-PD was revealed from our studies, which involved using 2.0 equivalents of T3P® and stirring the reaction mixture at 0 °C then RT for 6 h. The product was isolated by initially washing the reaction mixture with water followed by brine, then purified by column chromatography using 60–120 silica gel. The oxidation–condensation or cyclodehydrating properties of T3P® helped to generalize the reaction procedure. Next, we carried out reactions with a series of electronically diverse α-hydroxy ketones and o-PDs mediated by T3P® in a solvent mixture of DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc in order to obtain the desired quinoxalines 3a–k. The o-PD and α-hydroxy ketone without any substitution afforded a good yield of product, while the α-hydroxy ketone containing an electron-donating group favoured the formation of the desired quinoxalines in high yields, when compared to reactions where o-PD contained an electron-withdrawing group. These results are summarized in Scheme 2. The scope of the reaction was successfully extended to other substrates such as electronically demanding α-bromo ketones with o-PD or substituted o-PDs, which also underwent the title reaction under RT conditions to obtain the corresponding quinoxalines (4a–y). Similarly, the reaction of an α-halo ketone and o-PD without any substitution favoured the formation of a quinoxaline in slightly higher yield when compared to reactions using substituted α-halo ketones or o-PDs. These results are summarized in Scheme 3. We then extended our studies to further increase the substrate scope. We carried out the condensation of 1,2-dicarbonyl compounds with o-PD under mild reaction conditions at RT in the presence of T3P® to produce the desired quinoxaline derivatives (5a–p). Interestingly, the reaction between an aliphatic diketone and o-PD underwent the title reaction to afford the desired quinoxaline in lower yield when compared to aromatic 1,2-diketones. In the case where both aromatic starting materials were without substitution, the reaction favoured the formation of the quinoxaline in the highest yield. These results are summarized in Scheme 4.
EtOAc in order to obtain the desired quinoxalines 3a–k. The o-PD and α-hydroxy ketone without any substitution afforded a good yield of product, while the α-hydroxy ketone containing an electron-donating group favoured the formation of the desired quinoxalines in high yields, when compared to reactions where o-PD contained an electron-withdrawing group. These results are summarized in Scheme 2. The scope of the reaction was successfully extended to other substrates such as electronically demanding α-bromo ketones with o-PD or substituted o-PDs, which also underwent the title reaction under RT conditions to obtain the corresponding quinoxalines (4a–y). Similarly, the reaction of an α-halo ketone and o-PD without any substitution favoured the formation of a quinoxaline in slightly higher yield when compared to reactions using substituted α-halo ketones or o-PDs. These results are summarized in Scheme 3. We then extended our studies to further increase the substrate scope. We carried out the condensation of 1,2-dicarbonyl compounds with o-PD under mild reaction conditions at RT in the presence of T3P® to produce the desired quinoxaline derivatives (5a–p). Interestingly, the reaction between an aliphatic diketone and o-PD underwent the title reaction to afford the desired quinoxaline in lower yield when compared to aromatic 1,2-diketones. In the case where both aromatic starting materials were without substitution, the reaction favoured the formation of the quinoxaline in the highest yield. These results are summarized in Scheme 4.
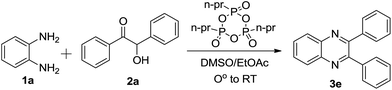 |
| | Scheme 1 The synthesis of a 2,3-diphenyl quinoxaline (3e) from an α-hydroxy ketone. | |
Table 1 The T3P®–DMSO mediated synthesis of 3e under different reaction conditions
| Entry |
Solventa |
T3P®b (equiv.) |
Time (h) |
T [°C] |
Yieldc (%) of 3e |
Reactions were performed with the solvent and DMSO in a volume ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. 1 mmol of 1a, 1.2 mmol of 2a were used in the case of 3e. T3P® (50% solution in EtOAc) was used to carry out the reactions. Isolated yield after purification by column chromatography. 1. 1 mmol of 1a, 1.2 mmol of 2a were used in the case of 3e. T3P® (50% solution in EtOAc) was used to carry out the reactions. Isolated yield after purification by column chromatography. |
| 1 |
EtOAc |
1.0 |
10 |
0–25 |
25 |
| 2 |
EtOAc |
1.5 |
6 |
0–25 |
54 |
| 3 |
EtOAc |
2.0 |
6 |
0–25 |
94 |
| 4 |
EtOAc |
2.5 |
6 |
0–25 |
94 |
| 5 |
EtOAc |
3.0 |
6 |
0–25 |
93 |
| 6 |
EtOAc |
3.5 |
6 |
0–25 |
93 |
| 7 |
CH2Cl2 |
2.5 |
6 |
0–25 |
48 |
| 8 |
CHCl3 |
2.5 |
6 |
0–25 |
44 |
| 9 |
CH3CN |
2.5 |
6 |
0–25 |
55 |
| 10 |
THF |
2.5 |
6 |
0–25 |
49 |
| 11 |
Toluene |
2.5 |
6 |
0–25 |
58 |
| 12 |
Dioxane |
2.5 |
6 |
0–25 |
56 |
| 13 |
Benzene |
2.5 |
6 |
0–25 |
40 |
| 14 |
EtOAc |
2.5 |
5.5 |
50 |
87 |
| 15 |
EtOAc |
2.5 |
5 |
60 |
86 |
| 16 |
EtOAc |
2.5 |
4 |
65 |
88 |
| 17 |
EtOAc |
2.5 |
3 |
75 |
80 |
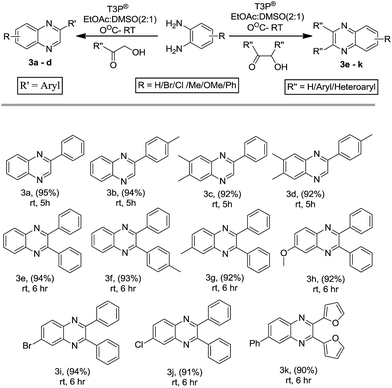 |
| | Scheme 2 The synthesis of quinoxalines from α-hydroxy ketones. T3P® (2.0 mmol), α-hydroxy ketone (1.2 mmol), 1,2 diamine (1.0 mmol). | |
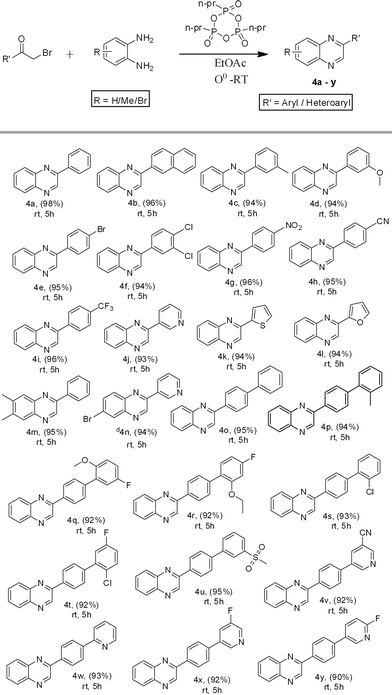 |
| | Scheme 3 The synthesis of quinoxalines from an α-halo ketone. T3P® (2.0 mmol), α-bromo ketone (1.2 mmol), diamine (1.0 mmol). dA mixture of regioisomers. | |
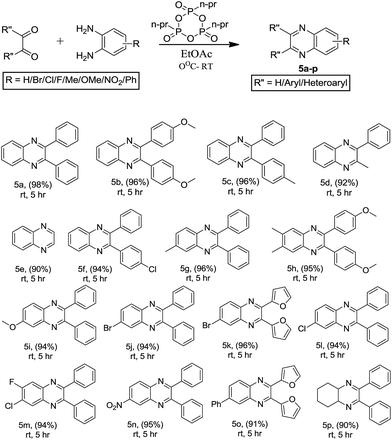 |
| | Scheme 4 The synthesis of quinoxalines from 1,2-diketones. T3P® (2.0 mmol), 1,2-diketone (1.2 mmol), diamine (1.0 mol). | |
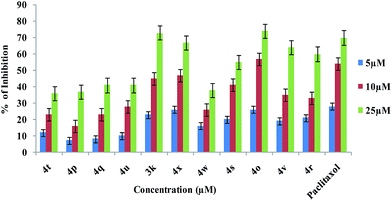
Selected quinoxaline compounds from Schemes 1 and 3, including 3k, 4o, 4p, 4q, 4r, 4s, 4t, 4u, 4v, 4w and 4x, were evaluated as cytotoxic agents against A549 human lung carcinoma cells, which have frequently been chosen by our group,17 as a suitable cell culture to perform cytotoxicity assays. Concentration–response experiments were performed to establish the cytotoxic activity of each selected compound (Fig. 1).
 |
| | Fig. 1 Bar graphs showing cytotoxic activity of different quinoxalines towards the A549 cell line. | |
We observed that quinoxalines 3k, 4o, 4s, 4v and 4u were the most efficient cytotoxic agents when a high concentration (25 μM) was used, resulting in the death of 74% of the A549 cells in case of 4o. The IC50 values, expressed in μM, are summarized in Table 2 and establish the relative order of cytotoxic effectiveness for the quinoxalines as: 3k ≈ 4o > 4s > 4v > 4u > 4q > 4r > 4t > 4p > 4x > 4w. From these results we can conclude that the slight differences in the observed cytotoxicity might be due to differences in the nature of the substituents present in the core moiety and the percentage uptake of the compounds by the cells (Scheme 5).
Table 2 IC50 (μM) inhibitory concentrations with human lung carcinoma cells (A549)
| Quinoxaline |
3k |
4o |
4p |
4q |
4r |
4s |
4t |
4u |
4v |
4w |
4x |
| IC50 (μM) |
2.14 |
2.11 |
27.3 |
17.4 |
19.4 |
2.8 |
20.3 |
10.3 |
5.3 |
33.2 |
30.3 |
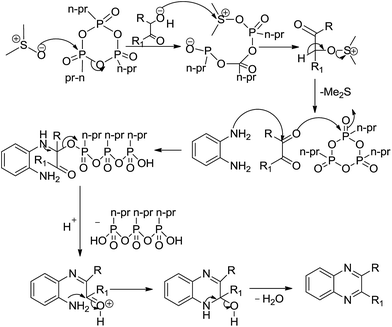 |
| | Scheme 5 Proposed mechanism for the formation of quinoxalines in Scheme 1. | |
Conclusion
This synthetic methodology provides a versatile, one-step protocol for the preparation of libraries of quinoxalines with broad substrate scope and in high yield. Preliminary investigations into the cytotoxic effects of the studied quinoxaline compounds against A-549 cancer cells showed that the lowest IC50 values were observed with phenyl substituted o-PD derived quinoxaline 3k and phenyl substituted α-halo ketone derived quinoxaline 4o. These promising results show that the application of DMSO–T3P® or T3P® mediated reactions can open access to new chemical space in the field of synthetic organic chemistry and also allow the exploration of new synthetic strategies for the synthesis of biologically active heterocyclic small molecules in the field of medicinal chemistry.
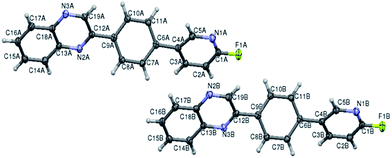 |
| | Fig. 2 Single crystal XRD structure of compound 4y. | |
Experimental section
Materials and instruments
Purification of the reaction products was carried out by normal column chromatography using Sorbent Technologies Standard Grade silica gel (60–120 mesh). Analytical thin layer chromatography was performed on Merck silica gel 60 F254 plates. Visualization was accomplished with UV light and potassium permanganate. Melting points were recorded on a Thomas Hoover capillary melting point apparatus and are uncorrected. Infrared spectra were recorded on an ATI Mattson Genesis Series FT-infrared spectrophotometer. Proton nuclear magnetic resonance spectra (1HNMR) were recorded on an Agilent 400 MHz spectrometer and are reported in ppm using CDCl3 as the internal standard (7.24 ppm). Proton-decoupled carbon nuclear magnetic resonance spectra (13C-NMR) were recorded on a Agilent 100 MHz spectrometer and reported in ppm using CDCl3 as the internal standard (77.0 ppm). Mass spectra were recorded on a Waters instrument, column – BEH C18 1.7 μM 2.1 × 50 mm; solvents – 0.1% formic acid in water (a) and CH3CN (b) with a solvent gradient-varied concentration of solvent (a) and (b) at different time intervals; detector – MS detector. Elemental analyses were recorded using a HERAEUS Vario EL-III instrument (for CHN).
General procedure for the synthesis of quinoxalines from α-hydroxy ketones
To a stirred suspension of α-hydroxy ketone (1.2 mmol) in a solvent mixture of DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc (ratio 2
EtOAc (ratio 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was added ortho-phenylenediamine (1 mmol). This was followed by the addition of T3P® (2.0 mmol) at 0 °C, then the reaction mixture was kept at RT for 6 h. The progress of the reaction was monitored by TLC. The reaction mixture was quenched with ice cold water and the reaction mixture was extracted with EtOAc (×2). The collected organic layers were washed with brine solution and dried over anhydrous sodium sulphate. The organic solvent was removed under reduced pressure to afford the desired quinoxaline, which was purified by column chromatography using hexanes
1) was added ortho-phenylenediamine (1 mmol). This was followed by the addition of T3P® (2.0 mmol) at 0 °C, then the reaction mixture was kept at RT for 6 h. The progress of the reaction was monitored by TLC. The reaction mixture was quenched with ice cold water and the reaction mixture was extracted with EtOAc (×2). The collected organic layers were washed with brine solution and dried over anhydrous sodium sulphate. The organic solvent was removed under reduced pressure to afford the desired quinoxaline, which was purified by column chromatography using hexanes![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc as an eluent.
EtOAc as an eluent.
General procedure for the synthesis of quinoxalines from α-haloketone and 1,2-diketone
To a stirred reaction mixture of an α-bromo ketone or 1,2-diketone (1.2 mmol) in EtOAc was added ortho-phenylenediamine (1 mmol) followed by the addition of T3P® (2 mmol) at 0 °C. The reaction mixture was then kept at RT for 5 h. The progress of the reaction was monitored by TLC. The reaction mixture was quenched with ice cold water and the reaction mixture was extracted with EtOAc (×2). The collected organic layers were washed with brine solution and dried over anhydrous sodium sulphate. The organic solvent was removed under reduced pressure to afford the desired quinoxalines, which were purified by column chromatography using hexanes![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc as an eluent.
EtOAc as an eluent.
2-Phenylquinoxaline (3a or 4a) 18a. Pale yellow solid; yield 95%; (Rf = 0.48 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 75–77 °C (Lit: 75–78);18c IR (KBr): 3055, 1545, 1480, 1445, 1312, 1035 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.33 (s, 1H), 8.21–8.11 (m, 4H), 7.81–7.73 (m, 2H), 7.59–7.52 (m, 3H); LCMS (ESI) [M + H]+ calculated C14H10N2 207.0877 found 207.14.
05 v/v); MP 75–77 °C (Lit: 75–78);18c IR (KBr): 3055, 1545, 1480, 1445, 1312, 1035 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.33 (s, 1H), 8.21–8.11 (m, 4H), 7.81–7.73 (m, 2H), 7.59–7.52 (m, 3H); LCMS (ESI) [M + H]+ calculated C14H10N2 207.0877 found 207.14.
2-(p-Tolyl)quinoxaline (3b). Yellow solid; yield 94%; (Rf = 0.46 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 92–94 °C (Lit: 91–93);18a IR (KBr): 3058, 1615, 1554, 1475, 1445, 1310, 1040 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.40 (s, 1H), 8.55–8.38 (m, 4H), 8.37–8.20 (m, 2H), 7.88 (d, J = 8 Hz, 2H), 2.40 (s, 3H); LCMS (ESI) [M + H]+ calculated C15H12N2 221.10 found 221.16.
05 v/v); MP 92–94 °C (Lit: 91–93);18a IR (KBr): 3058, 1615, 1554, 1475, 1445, 1310, 1040 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.40 (s, 1H), 8.55–8.38 (m, 4H), 8.37–8.20 (m, 2H), 7.88 (d, J = 8 Hz, 2H), 2.40 (s, 3H); LCMS (ESI) [M + H]+ calculated C15H12N2 221.10 found 221.16.
6,7-Dimethyl-2-phenylquinoxaline (3c or 4m). Yellow solid; yield 92%; (Rf = 0.54 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 129–130 °C (Lit: 130–131);18a IR (KBr): 3055, 3040, 2965, 1535, 1480, 1445, 1310, 1210, 1020, 1005 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.22 (s, 1H), 8.20 (d, J = 11.2 Hz, 2H), 7.89 (s, 1H), 7.88 (s, 1H), 7.65–7.36 (m, 3H), 3.41 (s, 6H); LCMS (ESI) [M + H]+ calculated C16H14N2 235.11 found 235.18.
05 v/v); MP 129–130 °C (Lit: 130–131);18a IR (KBr): 3055, 3040, 2965, 1535, 1480, 1445, 1310, 1210, 1020, 1005 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.22 (s, 1H), 8.20 (d, J = 11.2 Hz, 2H), 7.89 (s, 1H), 7.88 (s, 1H), 7.65–7.36 (m, 3H), 3.41 (s, 6H); LCMS (ESI) [M + H]+ calculated C16H14N2 235.11 found 235.18.
6,7-Dimethyl-2-(p-tolyl)quinoxaline (3d). Yellow solid; yield 92%; (Rf = 0.62 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 113–115 °C; (Lit: 114–115);18h IR (KBr): 3025, 2973, 1605, 1530, 1480, 1442, 1315, 1045, 1025, 860, 825 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.36 (s, 1H), 8.13 (d, J = 7.2 Hz, 2H), 8.12 (s, 1H), 7.82 (s, 1H), 7.81 (s, 1H), 7.09 (d, J = 8 Hz, 2H), 2.63 (s, 3H), 2.33 (s, 6H); LCMS (ESI) [M + H]+ calculated C17H16N2 249.13 found 249.19.
05 v/v); MP 113–115 °C; (Lit: 114–115);18h IR (KBr): 3025, 2973, 1605, 1530, 1480, 1442, 1315, 1045, 1025, 860, 825 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.36 (s, 1H), 8.13 (d, J = 7.2 Hz, 2H), 8.12 (s, 1H), 7.82 (s, 1H), 7.81 (s, 1H), 7.09 (d, J = 8 Hz, 2H), 2.63 (s, 3H), 2.33 (s, 6H); LCMS (ESI) [M + H]+ calculated C17H16N2 249.13 found 249.19.
2,3-Diphenylquinoxaline (3e or 5a). Yellow solid; yield 94%; (Rf = 0.58 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 116–118 °C; (Lit: 117–119);18a IR (KBr): 3056, 1635, 1475, 1440, 1345, 1075, 1055, 770, 695 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.18 (dd, J = 6.8 Hz, 3.2 Hz, 2H), 7.77 (dd, J = 3.6 Hz, 3.2 Hz, 2H), 7.53–7.50 (m, 4H), 7.38–7.31 (m, 6H); LCMS (ESI) [M + H]+ calculated C20H14N2 283.12 found 283.18.
05 v/v); MP 116–118 °C; (Lit: 117–119);18a IR (KBr): 3056, 1635, 1475, 1440, 1345, 1075, 1055, 770, 695 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.18 (dd, J = 6.8 Hz, 3.2 Hz, 2H), 7.77 (dd, J = 3.6 Hz, 3.2 Hz, 2H), 7.53–7.50 (m, 4H), 7.38–7.31 (m, 6H); LCMS (ESI) [M + H]+ calculated C20H14N2 283.12 found 283.18.
2-Phenyl-3-p-tolylquinoxaline (3f or 5c). Yellow solid; yield 93%; (Rf = 0.62 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 110–112 °C; (Lit: 112–114);18a IR (KBr): 3055, 2915, 1614, 1553, 1519, 1470, 1438, 1388, 1345, 1056, 1020, 974, 754, 696 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.26–8.14 (m, 2H), 7.99–7.95 (m, 2H), 7.83–7.78 (m, 2H), 7.72 (d, J = 7.6 Hz, 2H), 7.69–7.40 (m, 3H), 7.13 (d, J = 6.8 Hz, 2H), 2.42 (s, 3H); LCMS (ESI) [M + H]+ calculated C21H16N2 297.13 found 297.20.
05 v/v); MP 110–112 °C; (Lit: 112–114);18a IR (KBr): 3055, 2915, 1614, 1553, 1519, 1470, 1438, 1388, 1345, 1056, 1020, 974, 754, 696 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.26–8.14 (m, 2H), 7.99–7.95 (m, 2H), 7.83–7.78 (m, 2H), 7.72 (d, J = 7.6 Hz, 2H), 7.69–7.40 (m, 3H), 7.13 (d, J = 6.8 Hz, 2H), 2.42 (s, 3H); LCMS (ESI) [M + H]+ calculated C21H16N2 297.13 found 297.20.
6-Methyl-2,3-diphenylquinoxaline (3g or 5g). White solid; yield 92%; (Rf = 0.54 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 111–113 °C; (Lit: 110–112);18a IR (KBr): 3054, 2938, 1617, 1485, 1444, 1200, 1056, 1021, 812, 768, 698 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.10 (d, J = 8.4 Hz, 1H), 8.09 (s, 1H), 7.92 (dd, J = 9.6 Hz, 1.6 Hz, 1H), 7.78–7.69 (m, 4H), 7.68–7.31 (m, 6H), 2.77 (s, 3H); LCMS (ESI) [M + H]+ calculated C21H16N2 297.13 found 297.19.
05 v/v); MP 111–113 °C; (Lit: 110–112);18a IR (KBr): 3054, 2938, 1617, 1485, 1444, 1200, 1056, 1021, 812, 768, 698 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.10 (d, J = 8.4 Hz, 1H), 8.09 (s, 1H), 7.92 (dd, J = 9.6 Hz, 1.6 Hz, 1H), 7.78–7.69 (m, 4H), 7.68–7.31 (m, 6H), 2.77 (s, 3H); LCMS (ESI) [M + H]+ calculated C21H16N2 297.13 found 297.19.
6-Methoxy-2,3-diphenylquinoxaline (3h or 5i). White solid; yield 92%; (Rf = 0.52 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 160–162 °C; (Lit: 117–119);18a IR (KBr): 2965, 2936, 1756, 1617, 1482 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.04 (d, J = 8.8 Hz, 1H), 7.95–7.92 (m, 4H), 7.91 (d, J = 2 Hz, 1H), 7.59 (dd, J = 9.2 Hz, 2.8 Hz, 1H), 7.37–7.34 (m, 6H), 3.83 (s, 3H); LCMS (ESI) [M + H]+ calculated C21H16N2O 313.12 found 313.18.
05 v/v); MP 160–162 °C; (Lit: 117–119);18a IR (KBr): 2965, 2936, 1756, 1617, 1482 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.04 (d, J = 8.8 Hz, 1H), 7.95–7.92 (m, 4H), 7.91 (d, J = 2 Hz, 1H), 7.59 (dd, J = 9.2 Hz, 2.8 Hz, 1H), 7.37–7.34 (m, 6H), 3.83 (s, 3H); LCMS (ESI) [M + H]+ calculated C21H16N2O 313.12 found 313.18.
6-Bromo-2,3-diphenylquinoxaline (3i or 5j). Yellow solid; yield 94%; (Rf = 0.48 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 117–119 °C; (Lit: 118–120);18a IR (KBr); 3040, 1588, 1542, 1466, 1442, 1390, 1318, 1182, 1060, 1022, 973, 914, 828, 764, 694 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.35 (s, 1H), 8.03 (d, J = 9.2 Hz, 1H), 7.83 (d, J = 8.4 Hz, 1H), 7.50 (d, J = 6.8 Hz, 4H), 7.39–7.31 (m, 6H); LCMS (ESI) [M + H]+ calculated C20H13BrN2 361.03 found 361.09.
05 v/v); MP 117–119 °C; (Lit: 118–120);18a IR (KBr); 3040, 1588, 1542, 1466, 1442, 1390, 1318, 1182, 1060, 1022, 973, 914, 828, 764, 694 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.35 (s, 1H), 8.03 (d, J = 9.2 Hz, 1H), 7.83 (d, J = 8.4 Hz, 1H), 7.50 (d, J = 6.8 Hz, 4H), 7.39–7.31 (m, 6H); LCMS (ESI) [M + H]+ calculated C20H13BrN2 361.03 found 361.09.
6-Chloro-2,3-diphenylquinoxaline (3j or 5l). White solid; yield 80%; (Rf = 0.46 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 118–120 °C; (Lit: 117–120);18a IR (KBr): 3047, 1602, 1548, 1464, 1442, 1338, 1068, 828, 800, 764, 694 cm−1; 1H NMR (400 MHz, CDCl3): δ = 7.96 (d, J = 8.4 Hz, 1H), 7.90–7.64 (m, 5H), 7.60–7.34 (m. 6H), 2.77 (s, 3H), 2.48 (s, 3H); LCMS (ESI) [M + H]+ calculated C21H16N2 317.08 found 317.14.
05 v/v); MP 118–120 °C; (Lit: 117–120);18a IR (KBr): 3047, 1602, 1548, 1464, 1442, 1338, 1068, 828, 800, 764, 694 cm−1; 1H NMR (400 MHz, CDCl3): δ = 7.96 (d, J = 8.4 Hz, 1H), 7.90–7.64 (m, 5H), 7.60–7.34 (m. 6H), 2.77 (s, 3H), 2.48 (s, 3H); LCMS (ESI) [M + H]+ calculated C21H16N2 317.08 found 317.14.
2,3-Di(furan-2-yl)-6-phenylquinoxaline (3k or 5o). White solid; yield 91%; (Rf = 0.55 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 131–133 °C; IR (KBr): 3356, 1614, 1538, 1438, 1081, 835, 1H NMR (CDCl3, 400 MHz): δ = 8.31 (s, 1H), 8.20 (d, J = 8.4 Hz, 1H), 7.94 (t, J = 4 Hz, 1H), 7.92 (t, J = 3.6 Hz, 1H), 7.65–7.64 (m, 2H), 7.59 (dd, J = 7.2 Hz, 2.8 Hz, 2H), 7.39–7.34 (m, 1H), 7.17 (t, J = 9.6 Hz, 1H), 6.72–6.69 (m, 2H), 6.59–6.58 (m, 2H); 13C NMR (CDCl3, 100 MHz): δ = 144.9, 144.4, 130.6, 130.3, 129.6, 129.3, 128.0, 127.2, 126.2, 113.6, 113.2, 112.1; LCMS (ESI) [M + H]+ calculated C22H14N2O2 339.10 found 339.17; anal. calcd for C21H16N2: C, 78.09; H, 4.17; N, 8.28; found: C, 77.78; H, 3.97; N, 7.96.
05 v/v); MP 131–133 °C; IR (KBr): 3356, 1614, 1538, 1438, 1081, 835, 1H NMR (CDCl3, 400 MHz): δ = 8.31 (s, 1H), 8.20 (d, J = 8.4 Hz, 1H), 7.94 (t, J = 4 Hz, 1H), 7.92 (t, J = 3.6 Hz, 1H), 7.65–7.64 (m, 2H), 7.59 (dd, J = 7.2 Hz, 2.8 Hz, 2H), 7.39–7.34 (m, 1H), 7.17 (t, J = 9.6 Hz, 1H), 6.72–6.69 (m, 2H), 6.59–6.58 (m, 2H); 13C NMR (CDCl3, 100 MHz): δ = 144.9, 144.4, 130.6, 130.3, 129.6, 129.3, 128.0, 127.2, 126.2, 113.6, 113.2, 112.1; LCMS (ESI) [M + H]+ calculated C22H14N2O2 339.10 found 339.17; anal. calcd for C21H16N2: C, 78.09; H, 4.17; N, 8.28; found: C, 77.78; H, 3.97; N, 7.96.
(Naphthalen-2-yl)quinoxaline (4b). Yellow solid; yield 94%; (Rf = 0.60 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 136–138 °C; (Lit: 138);18a IR (KBr): 3038, 1546, 1488, 1306, 1196 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.47 (s, 1H), 8.65 (s, 1H), 8.37–8.34 (dd, J = 10.8 Hz, 2 Hz, 1H), 8.21–8.13 (m, 2H), 8.03–7.99 (m, 2H), 7.90 (t, J = 3.6 Hz, 1H), 7.82–7.73 (m, 2H), 7.54–7.50 (m, 2H); LCMS (ESI) [M + H]+ calculated C18H12N2 257.10 found 257.17.
05 v/v); MP 136–138 °C; (Lit: 138);18a IR (KBr): 3038, 1546, 1488, 1306, 1196 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.47 (s, 1H), 8.65 (s, 1H), 8.37–8.34 (dd, J = 10.8 Hz, 2 Hz, 1H), 8.21–8.13 (m, 2H), 8.03–7.99 (m, 2H), 7.90 (t, J = 3.6 Hz, 1H), 7.82–7.73 (m, 2H), 7.54–7.50 (m, 2H); LCMS (ESI) [M + H]+ calculated C18H12N2 257.10 found 257.17.
2-(m-Tolyl)quinoxaline (4c). Yellow solid; yield 94%; (Rf = 0.52 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 92–94 °C; (Lit: 84–86);18f IR (KBr): 3054, 1612, 1542, 1308 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.33 (s, 1H), 8.19–8.13 (m, 2H), 8.04 (s, 1H), 7.97 (d, J = 8 Hz, 1H), 7.98–7.75 (m, 2H), 7.47 (t, J = 8 Hz, 1H), 7.36 (d, J = 8.4 Hz, 1H), 2.51 (s, 3H); LCMS (ESI) [M + H]+ calculated C15H12N2 221.10 found 221.17.
05 v/v); MP 92–94 °C; (Lit: 84–86);18f IR (KBr): 3054, 1612, 1542, 1308 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.33 (s, 1H), 8.19–8.13 (m, 2H), 8.04 (s, 1H), 7.97 (d, J = 8 Hz, 1H), 7.98–7.75 (m, 2H), 7.47 (t, J = 8 Hz, 1H), 7.36 (d, J = 8.4 Hz, 1H), 2.51 (s, 3H); LCMS (ESI) [M + H]+ calculated C15H12N2 221.10 found 221.17.
2-(3-Methoxyphenyl)quinoxaline (4d). Pale yellow solid; yield 92%; (Rf = 0.46 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 96–98 °C; (Lit: 87–88);18h IR (KBr): 1604, 1539, 1442, 1186, 1031 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.32 (s, 1H), 8.18–8.11 (m, 2H), 7.81–7.73 (m, 4H), 7.47 (t, J = 8 Hz, 1H), 7.07 (dd, J = 10.4 Hz, 2 Hz, 1H), 3.94 (s, 3H); LCMS (ESI) [M + H]+ calculated C15H12N2O 237.09 found 237.15.
05 v/v); MP 96–98 °C; (Lit: 87–88);18h IR (KBr): 1604, 1539, 1442, 1186, 1031 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.32 (s, 1H), 8.18–8.11 (m, 2H), 7.81–7.73 (m, 4H), 7.47 (t, J = 8 Hz, 1H), 7.07 (dd, J = 10.4 Hz, 2 Hz, 1H), 3.94 (s, 3H); LCMS (ESI) [M + H]+ calculated C15H12N2O 237.09 found 237.15.
2-(4-Bromophenyl)quinoxaline (4e). Pale yellow solid; yield 94%; (Rf = 0.56 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 136–138 °C; (Lit: 134–137);18a IR (KBr): 1588, 1536, 1480, 1122, 1068, 1043 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.29 (s, 1H), 8.15–8.07 (m, 4H), 7.81–7.73 (m, 2H), 7.69 (d, J = 8.4 Hz, 2H); LCMS (ESI) [M + H]+ calculated C14H10BrN2O 285.00 found 284.79.
05 v/v); MP 136–138 °C; (Lit: 134–137);18a IR (KBr): 1588, 1536, 1480, 1122, 1068, 1043 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.29 (s, 1H), 8.15–8.07 (m, 4H), 7.81–7.73 (m, 2H), 7.69 (d, J = 8.4 Hz, 2H); LCMS (ESI) [M + H]+ calculated C14H10BrN2O 285.00 found 284.79.
2-(3,4-Dichlorophenyl)quinoxaline (4f). Yellow solid; yield 90%; (Rf = 0.58 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 186–188 °C; (Lit: 187–189);18a IR (KBr): 1540, 1474, 1308, 1146, 1030 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.31 (s, 1H), 8.43 (s, 1H), 8.09 (t, J = 6 Hz, 2H), 7.80 (d, J = 7.2 Hz, 1H), 7.80–7.72 (m, 2H); LCMS (ESI) [M + H]+ calculated C14H9Cl2N2 275.01 found 275.07.
05 v/v); MP 186–188 °C; (Lit: 187–189);18a IR (KBr): 1540, 1474, 1308, 1146, 1030 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.31 (s, 1H), 8.43 (s, 1H), 8.09 (t, J = 6 Hz, 2H), 7.80 (d, J = 7.2 Hz, 1H), 7.80–7.72 (m, 2H); LCMS (ESI) [M + H]+ calculated C14H9Cl2N2 275.01 found 275.07.
2-(4-Nitrophenyl)quinoxaline (4g). Colorless solid; yield 92%; (Rf = 0.64 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 191–193 °C; (Lit: 190–192);18a IR (KBr): 3056, 1529, 1352, 1096 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.11 (s, 1H), 8.40–8.37 (m, 4H), 8.21–7.84 (m, 2H), 7.83 (d, J = 8 Hz, 2H); LCMS (ESI) [M + H]+ calculated C14H9N3O2 252.07 found 252.13.
05 v/v); MP 191–193 °C; (Lit: 190–192);18a IR (KBr): 3056, 1529, 1352, 1096 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.11 (s, 1H), 8.40–8.37 (m, 4H), 8.21–7.84 (m, 2H), 7.83 (d, J = 8 Hz, 2H); LCMS (ESI) [M + H]+ calculated C14H9N3O2 252.07 found 252.13.
4-(Quinoxalin-2-yl)benzonitrile (4h). Pale yellow solid; yield 93%; (Rf = 0.60 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 114–116 °C; (Lit: 193–195); IR (KBr): 3058, 2230, 1548, 1480, 1435, 1322, 1036; 1H NMR (CDCl3, 400 MHz): δ = 9.36 (s, 1H), 8.35 (d, J = 8.8 Hz, 2H), 8.19–8.15 (m, 2H), 7.88–7.80 (m, 4H); 13C NMR (CDCl3, 100 MHz): δ = 143.0, 142.6, 142.3, 141.7, 140.6, 133.0, 131.8, 130.5, 129.6, 129.1, 128.2, 127.7, 118.8, 113.8; LCMS (ESI) [M + H]+ calculated C15H9N3 232.08 found 232.14. Anal. calcd for C15H9N3: C, 77.91; H, 3.92; N, 18.17; found: C, 77.61, H, 3.56, N; 17.98.
05 v/v); MP 114–116 °C; (Lit: 193–195); IR (KBr): 3058, 2230, 1548, 1480, 1435, 1322, 1036; 1H NMR (CDCl3, 400 MHz): δ = 9.36 (s, 1H), 8.35 (d, J = 8.8 Hz, 2H), 8.19–8.15 (m, 2H), 7.88–7.80 (m, 4H); 13C NMR (CDCl3, 100 MHz): δ = 143.0, 142.6, 142.3, 141.7, 140.6, 133.0, 131.8, 130.5, 129.6, 129.1, 128.2, 127.7, 118.8, 113.8; LCMS (ESI) [M + H]+ calculated C15H9N3 232.08 found 232.14. Anal. calcd for C15H9N3: C, 77.91; H, 3.92; N, 18.17; found: C, 77.61, H, 3.56, N; 17.98.
2-(4-(Trifluoromethyl)phenyl)quinoxaline (4i). Pale yellow solid; yield 92%; (Rf = 0.40 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 142–143 °C; (Lit: 142–143);18i IR (KBr): 3036, 1545, 1488, 1315, 1120, 645, 554 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.33 (s, 1H), 8.31 (d, J = 8.4 Hz, 2H), 8.12–8.10 (m, 2H), 7.88–7.76 (m, 4H); LCMS (ESI) [M + H]+ calculated C15H10N2F3 275.07 found 275.14.
05 v/v); MP 142–143 °C; (Lit: 142–143);18i IR (KBr): 3036, 1545, 1488, 1315, 1120, 645, 554 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.33 (s, 1H), 8.31 (d, J = 8.4 Hz, 2H), 8.12–8.10 (m, 2H), 7.88–7.76 (m, 4H); LCMS (ESI) [M + H]+ calculated C15H10N2F3 275.07 found 275.14.
2-(Pyridin-3-yl)quinoxaline (4j). White solid; yield 90%; (Rf = 0.44 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 110–112 °C; (Lit: 114–115);18j IR (KBr): 3052, 1612, 1588, 1542, 1491, 1370, 1025, 1312; 1H NMR (CDCl3, 400 MHz): δ = 9.35 (s, 1H), 9.34 (s, 1H), 8.77 (dd, J = 6 Hz, 1.2 Hz, 1H), 8.54 (dt, J = 12.4 Hz, 2 Hz, 1H), 8.20–8.12 (m, 2H), 7.85–7.77 (m, 2H), 7.53–7.50 (m, 1H); LCMS (ESI) [M + H]+ calculated C13H9N3 208.08 found 208.14.
05 v/v); MP 110–112 °C; (Lit: 114–115);18j IR (KBr): 3052, 1612, 1588, 1542, 1491, 1370, 1025, 1312; 1H NMR (CDCl3, 400 MHz): δ = 9.35 (s, 1H), 9.34 (s, 1H), 8.77 (dd, J = 6 Hz, 1.2 Hz, 1H), 8.54 (dt, J = 12.4 Hz, 2 Hz, 1H), 8.20–8.12 (m, 2H), 7.85–7.77 (m, 2H), 7.53–7.50 (m, 1H); LCMS (ESI) [M + H]+ calculated C13H9N3 208.08 found 208.14.
2-(Thiophen-2-yl)quinoxaline (4k). Pale yellow solid; yield 94%; (Rf = 0.42 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 116–118 °C; (Lit: 116–117);18a IR (KBr): 3120, 3056, 1546, 1424, 1328, 1052 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.16 (s, 1H), 8.08–7.87 (m, 2H), 7.78–7.68 (m, 3H), 7.58 (d, J = 4 Hz, 1H), 7.56 (dd, J = 6.4 Hz, 1.6 Hz, 1H); LCMS (ESI) [M + H]+ calculated C12H9N2S 213.04 found 213.10.
05 v/v); MP 116–118 °C; (Lit: 116–117);18a IR (KBr): 3120, 3056, 1546, 1424, 1328, 1052 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.16 (s, 1H), 8.08–7.87 (m, 2H), 7.78–7.68 (m, 3H), 7.58 (d, J = 4 Hz, 1H), 7.56 (dd, J = 6.4 Hz, 1.6 Hz, 1H); LCMS (ESI) [M + H]+ calculated C12H9N2S 213.04 found 213.10.
2-(Furan-2-yl)quinoxaline (4l). Pale yellow solid; yield 93%; (Rf = 0.46 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 131–133 °C; (Lit: 131–136);18a IR (KBr): 3134, 3118, 1608, 1548, 1444, 1296, 1225, 1126, 1080 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.25 (s, 1H), 8.11–8.06 (m, 2H), 7.78–7.68 (m, 3H), 7.31 (d, J = 4 Hz, 1H), 6.63 (dd, J = 4.8 Hz, 1.2 Hz, 1H); LCMS (ESI) [M + H]+ calculated C12H8N2O 197.06 found 197.12.
05 v/v); MP 131–133 °C; (Lit: 131–136);18a IR (KBr): 3134, 3118, 1608, 1548, 1444, 1296, 1225, 1126, 1080 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.25 (s, 1H), 8.11–8.06 (m, 2H), 7.78–7.68 (m, 3H), 7.31 (d, J = 4 Hz, 1H), 6.63 (dd, J = 4.8 Hz, 1.2 Hz, 1H); LCMS (ESI) [M + H]+ calculated C12H8N2O 197.06 found 197.12.
6-Bromo-2-(pyridin-3-yl)quinoxaline (regioisomers) (4n). A mixture of two regioisomers, which were not separable by column chromatography, was obtained as a white solid. The NMR spectra indicated that 4n was a mixture. The NMR spectra indicated that it is a mixture. White solid; yield 92%; (Rf = 0.42 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); IR (KBr): 3066, 1608, 1548, 1490, 1318, 648 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.46 (s, 1H), 9.38 (s, 1H), 8.83 (s, 1H), 8.57–8.56 (t, J = 3.6 Hz, 1H), 8.36 (t, J = 2 Hz, 1H), 8.08–8.03 (m, 1H), 7.94–7.88 (m, 1H), 7.58–7.54 (m, 1H) LCMS (ESI) [M + H]+ calculated 285.99 found 286.05.
05 v/v); IR (KBr): 3066, 1608, 1548, 1490, 1318, 648 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.46 (s, 1H), 9.38 (s, 1H), 8.83 (s, 1H), 8.57–8.56 (t, J = 3.6 Hz, 1H), 8.36 (t, J = 2 Hz, 1H), 8.08–8.03 (m, 1H), 7.94–7.88 (m, 1H), 7.58–7.54 (m, 1H) LCMS (ESI) [M + H]+ calculated 285.99 found 286.05.
2-([1,1′-Biphenyl]-4-yl)quinoxaline (4o). Colorless solid; yield 95%; (Rf = 0.44 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 132–134 °C; (130–133);18a IR (KBr): 3060, 1534, 1486, 1420, 1317, 1054 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.38 (s, 1H), 8.33 (d, J = 8 Hz, 2H), 8.19–8.13 (m, 2H), 7.95 (t, J = 6.4 Hz, 2H), 7.83–7.75 (m, 2H), 7.69 (t, J = 7.6 Hz, 1H), 7.47–7.40 (m, 2H); 13C NMR (CDCl3, 100 MHz): δ = 150.99, 142.96, 142.36, 141.81, 141.57, 14.50, 140.67, 136.70, 132.19, 130.72, 130.47, 130.01, 129.63, 129.04, 128.24, 127.90, 126.43, 125.83, 121.04, 119.17; LCMS (ESI) [M + H]+ calculated C20H14N2 283.11 found 283.17.
05 v/v); MP 132–134 °C; (130–133);18a IR (KBr): 3060, 1534, 1486, 1420, 1317, 1054 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.38 (s, 1H), 8.33 (d, J = 8 Hz, 2H), 8.19–8.13 (m, 2H), 7.95 (t, J = 6.4 Hz, 2H), 7.83–7.75 (m, 2H), 7.69 (t, J = 7.6 Hz, 1H), 7.47–7.40 (m, 2H); 13C NMR (CDCl3, 100 MHz): δ = 150.99, 142.96, 142.36, 141.81, 141.57, 14.50, 140.67, 136.70, 132.19, 130.72, 130.47, 130.01, 129.63, 129.04, 128.24, 127.90, 126.43, 125.83, 121.04, 119.17; LCMS (ESI) [M + H]+ calculated C20H14N2 283.11 found 283.17.
2-(2′-Methylbiphenyl-4-yl)quinoxaline (4p). White solid; yield 94%; (Rf = 0.46 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 128–130 °C; IR (KBr): 3065, 2910, 1560, 1480, 1415, 1312, 1052 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.39 (s, 1H), 8.26 (dd, J = 2 Hz, 8.4 Hz, 2H), 8.19–8.13 (m, 2H), 7.83–7.75 (m, 2H), 7.55 (d, J = 8 Hz, 2H), 7.32–7.29 (m, 4H), 2.34 (s, 3H); 13C NMR (CDCl3, 100 MHz): δ = 151.7, 144.0, 143.3, 141.5, 141.0, 135.3, 135.2, 130.5, 130.1, 129.6, 129.5, 129.1, 127.6, 127.3, 125.9, 20.5; anal. calcd for C21H16N2: C, 85.11; H, 5.44; N, 9.45. Found: C, 84.90; H, 5.06; N, 9.38.
05 v/v); MP 128–130 °C; IR (KBr): 3065, 2910, 1560, 1480, 1415, 1312, 1052 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.39 (s, 1H), 8.26 (dd, J = 2 Hz, 8.4 Hz, 2H), 8.19–8.13 (m, 2H), 7.83–7.75 (m, 2H), 7.55 (d, J = 8 Hz, 2H), 7.32–7.29 (m, 4H), 2.34 (s, 3H); 13C NMR (CDCl3, 100 MHz): δ = 151.7, 144.0, 143.3, 141.5, 141.0, 135.3, 135.2, 130.5, 130.1, 129.6, 129.5, 129.1, 127.6, 127.3, 125.9, 20.5; anal. calcd for C21H16N2: C, 85.11; H, 5.44; N, 9.45. Found: C, 84.90; H, 5.06; N, 9.38.
2-(5′-Fluoro-2′-methoxybiphenyl-4yl)quinoxaline (4q). White solid; yield 92%; (Rf = 0.42 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 134–136 °C; IR (KBr): 3058, 2858, 1528, 1482, 1418, 1145, 1315, 1052, 686 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.38 (s, 1H), 8.26 (d, J = 8.4 Hz, 2H), 8.19–8.12 (m, 2H), 7.82–7.77 (m, 2H), 7.76–7.72 (m, 2H), 7.13 (dd, J = 2.8 Hz, 8.8 Hz, 1H), 7.07–6.93 (m, 2H), 3.8 (s, 3H); 13C NMR (CDCl3, 100 MHz): δ = 158.3, 155.9, 152.7, 151.5, 143.3, 142.3, 141.5, 139.4, 135.6, 130.3, 130.1, 129.6, 129.5, 129.1, 127.2, 56.2; anal. calcd for C21H16N2: C, 76.35; H, 4.58; N, 8.48; found: C, 75.92, H, 4.66, N, 8.38.
05 v/v); MP 134–136 °C; IR (KBr): 3058, 2858, 1528, 1482, 1418, 1145, 1315, 1052, 686 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.38 (s, 1H), 8.26 (d, J = 8.4 Hz, 2H), 8.19–8.12 (m, 2H), 7.82–7.77 (m, 2H), 7.76–7.72 (m, 2H), 7.13 (dd, J = 2.8 Hz, 8.8 Hz, 1H), 7.07–6.93 (m, 2H), 3.8 (s, 3H); 13C NMR (CDCl3, 100 MHz): δ = 158.3, 155.9, 152.7, 151.5, 143.3, 142.3, 141.5, 139.4, 135.6, 130.3, 130.1, 129.6, 129.5, 129.1, 127.2, 56.2; anal. calcd for C21H16N2: C, 76.35; H, 4.58; N, 8.48; found: C, 75.92, H, 4.66, N, 8.38.
2-(2′-Ethoxy-4′-fluoro-[1,1′-biphenyl]-4-yl)quinoxaline (4r). White solid; yield 92%; (Rf = 0.42 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 136–138 °C; IR (KBr): 3052, 2890, 1532, 1483, 1427, 1319, 1055, 812 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.37 (s, 1H), 8.18 (d, J = 1.6 Hz, 1H), 8.15 (d, J = 6.8 Hz, 2H), 8.14–8.05 (m, 2H), 8.04 (d, J = 8 Hz, 2H), 7.81–7.31 (m, 3H), 7.05 (dd, J = 11.6 Hz, 2.8 Hz, 1H), 3.99 (q, J = 16 Hz, 7.2 Hz, 2H), 1.58 (t, J = 7.2 Hz, 3H); 13C NMR (CDCl3, 100 MHz); 157.47, 152.02, 141.81, 141.57, 140.67, 136.50, 132.29, 130.89, 130.47, 129.04, 128.24, 127.90, 126.53, 125.73, 121.04, 114.27, 110.08, 68.64, 15.47; anal. calcd for C22H17FN2O: C, 76.73; H, 4.98; N, 8.13; found: C, 76.33, H, 4.88, N, 7.98.
05 v/v); MP 136–138 °C; IR (KBr): 3052, 2890, 1532, 1483, 1427, 1319, 1055, 812 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.37 (s, 1H), 8.18 (d, J = 1.6 Hz, 1H), 8.15 (d, J = 6.8 Hz, 2H), 8.14–8.05 (m, 2H), 8.04 (d, J = 8 Hz, 2H), 7.81–7.31 (m, 3H), 7.05 (dd, J = 11.6 Hz, 2.8 Hz, 1H), 3.99 (q, J = 16 Hz, 7.2 Hz, 2H), 1.58 (t, J = 7.2 Hz, 3H); 13C NMR (CDCl3, 100 MHz); 157.47, 152.02, 141.81, 141.57, 140.67, 136.50, 132.29, 130.89, 130.47, 129.04, 128.24, 127.90, 126.53, 125.73, 121.04, 114.27, 110.08, 68.64, 15.47; anal. calcd for C22H17FN2O: C, 76.73; H, 4.98; N, 8.13; found: C, 76.33, H, 4.88, N, 7.98.
2-(2′-Chloro-[1,1′-biphenyl]-4-yl)quinoxaline (4s). White solid; yield 93%; (Rf = 0.50 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 133–135 °C; IR (KBr): 3052, 1528, 1480, 1476, 1416, 1312, 1051, 748 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.37 (s, 1H), 8.27 (d, J = 12 Hz, 2H), 8.17–8.11 (m, 2H), 7.77 (m, 2H), 7.65 (d, J = 8 Hz, 2H), 7.50 (d, J = 7.6 Hz, 1H), 7.40–7.29 (m, 3H); 13C NMR (CDCl3, 100 MHz): δ = 151.4, 143.2, 142.3, 141.6, 141.3, 139.7, 136.0, 132.5, 131.2, 130.2, 130.0, 129.6, 129.5, 129.1, 128.9, 127.2, 126.9; anal. calcd for C20H13ClN2: C, 75.83; H, 4.14; N, 8.84 found: C, 74.82, H, 4.08, N, 8.92.
05 v/v); MP 133–135 °C; IR (KBr): 3052, 1528, 1480, 1476, 1416, 1312, 1051, 748 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.37 (s, 1H), 8.27 (d, J = 12 Hz, 2H), 8.17–8.11 (m, 2H), 7.77 (m, 2H), 7.65 (d, J = 8 Hz, 2H), 7.50 (d, J = 7.6 Hz, 1H), 7.40–7.29 (m, 3H); 13C NMR (CDCl3, 100 MHz): δ = 151.4, 143.2, 142.3, 141.6, 141.3, 139.7, 136.0, 132.5, 131.2, 130.2, 130.0, 129.6, 129.5, 129.1, 128.9, 127.2, 126.9; anal. calcd for C20H13ClN2: C, 75.83; H, 4.14; N, 8.84 found: C, 74.82, H, 4.08, N, 8.92.
2-(2′-Chloro-5′-fluorobiphenyl-4-yl)quinoxaline (4t). White solid; yield 90%; (Rf = 0.42 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 141–143 °C; IR (KBr): 3060, 1532, 1484, 1416, 1316, 1050, 734, 688 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.37 (s, 1H), 8.26 (dd, J = 2 Hz, 8.8 Hz, 2H), 8.18–8.12 (m, 2H), 7.81–7.72 (m, 2H), 7.13 (dd, J = 2.8 Hz, 12 Hz, 1H), 7.06–7.02 (m, 1H), 6.94 (dd, J = 4 Hz, 13.2 Hz, 1H); 13C NMR (CDCl3, 100 MHz): δ = 158.3, 155.9, 152.7, 151.52, 143.3, 141.5, 139.4, 135.6, 130.3, 130.2, 129.6, 129.5, 129.1, 127.2; anal. calcd for C20H12ClFN2: C, 71.75; H, 3.61; N, 8.37; found: C, 71.14, H, 3.48, N, 8.32.
05 v/v); MP 141–143 °C; IR (KBr): 3060, 1532, 1484, 1416, 1316, 1050, 734, 688 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.37 (s, 1H), 8.26 (dd, J = 2 Hz, 8.8 Hz, 2H), 8.18–8.12 (m, 2H), 7.81–7.72 (m, 2H), 7.13 (dd, J = 2.8 Hz, 12 Hz, 1H), 7.06–7.02 (m, 1H), 6.94 (dd, J = 4 Hz, 13.2 Hz, 1H); 13C NMR (CDCl3, 100 MHz): δ = 158.3, 155.9, 152.7, 151.52, 143.3, 141.5, 139.4, 135.6, 130.3, 130.2, 129.6, 129.5, 129.1, 127.2; anal. calcd for C20H12ClFN2: C, 71.75; H, 3.61; N, 8.37; found: C, 71.14, H, 3.48, N, 8.32.
2-(3′-(Methylsulfonyl)biphenyl-4-yl)quinoxaline (4u). Pale yellow solid; yield 95%; (Rf = 0.48 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 137–139 °C; IR (KBr): 3062, 2886, 1530, 1488, 1414, 1317, 1112, 1060 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.39 (s, 1H), 8.34 (d, J = 8.8 Hz, 2H), 8.24 (t, J = 4 Hz, 1H), 8.21–8.14 (m, 2H), 7.99–7.95 (m, 2H), 7.83 (d, J = 8 Hz, 2H), 7.81–7.78 (m, 2H), 7.71 (t, J = 12 Hz, 1H), 7.48–7.40 (m, 2H), 3.14 (s, 3H); 13C NMR (CDCl3, 100 MHz): δ = 156.9, 151.0, 143.0, 142.3, 141.7, 140.6, 136.6, 132.2, 130.7, 129.0, 128.2, 127.9, 126.4, 125.8, 121.1, 44.5; anal. calcd for C21H16N2O2S: C, 69.98; H, 4.47; N, 7.77; found: C, 68.92, H, 4.40, N, 7.28.
05 v/v); MP 137–139 °C; IR (KBr): 3062, 2886, 1530, 1488, 1414, 1317, 1112, 1060 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.39 (s, 1H), 8.34 (d, J = 8.8 Hz, 2H), 8.24 (t, J = 4 Hz, 1H), 8.21–8.14 (m, 2H), 7.99–7.95 (m, 2H), 7.83 (d, J = 8 Hz, 2H), 7.81–7.78 (m, 2H), 7.71 (t, J = 12 Hz, 1H), 7.48–7.40 (m, 2H), 3.14 (s, 3H); 13C NMR (CDCl3, 100 MHz): δ = 156.9, 151.0, 143.0, 142.3, 141.7, 140.6, 136.6, 132.2, 130.7, 129.0, 128.2, 127.9, 126.4, 125.8, 121.1, 44.5; anal. calcd for C21H16N2O2S: C, 69.98; H, 4.47; N, 7.77; found: C, 68.92, H, 4.40, N, 7.28.
4′-(Quinoxalin-2-yl)-[1,1′-biphenyl]-3-carbonitrile (4v). White solid; yield 90%; (Rf = 0.46 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 129–131 °C; IR (KBr): 3056, 2228, 1531, 1482, 1417, 1319, 1052 cm−1; 1H NMR (CDCl3, 400 MHz); δ = 9.57 (s, 1H), 9.37 (s, 1H), 9.21 (s, 1H), 8.50 (s, 1H), 8.32 (d, J = 8.4 Hz, 2H), 8.18–8.12 (m, 2H), 7.79 (d, J = 8.4 Hz, 2H); 13C NMR (CDCl3, 100 MHz): δ = 164.6, 162.2, 150.9, 145.9, 145.9, 145.84, 142.3, 141.6, 139.7, 139.6, 138.3, 136.5, 134.0, 130.4, 129.6, 128.2, 115.6, 109.8, 109.4; anal. calcd for C21H13N2: C, 77.91; H, 3.92; N, 18.17; found: C, 76.88, H, 3.94, N, 18.12.
05 v/v); MP 129–131 °C; IR (KBr): 3056, 2228, 1531, 1482, 1417, 1319, 1052 cm−1; 1H NMR (CDCl3, 400 MHz); δ = 9.57 (s, 1H), 9.37 (s, 1H), 9.21 (s, 1H), 8.50 (s, 1H), 8.32 (d, J = 8.4 Hz, 2H), 8.18–8.12 (m, 2H), 7.79 (d, J = 8.4 Hz, 2H); 13C NMR (CDCl3, 100 MHz): δ = 164.6, 162.2, 150.9, 145.9, 145.9, 145.84, 142.3, 141.6, 139.7, 139.6, 138.3, 136.5, 134.0, 130.4, 129.6, 128.2, 115.6, 109.8, 109.4; anal. calcd for C21H13N2: C, 77.91; H, 3.92; N, 18.17; found: C, 76.88, H, 3.94, N, 18.12.
2-(4-(Pyridin-4-yl)phenyl)quinoxaline (4w). White solid; yield 91%; (Rf = 0.44 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 138–140 °C; IR (KBr): 3056, 1529, 1486, 1418, 1315, 1062 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.39 (s, 1H), 8.7 (d, J = 7.2 Hz, 1H), 8.51 (d, J = 2.4 Hz, 1H), 8.36 (dt, J = 2 Hz, 8.8 Hz, 2H), 8.20–8.14 (m, 2H), 7.83 (dd, J = 1.6 Hz, 8.4 Hz, 1H), 7.82–7.76 (m, 4H), 7.68 (dt, J = 2.8 Hz, 12 Hz, 1H); 13C NMR (CDCl3, 100 MHz): δ = 150.8, 144.0, 143.0, 142.32, 141.7, 138.0, 137.0, 130.4, 129.6, 129.1, 128.3, 127.9, 121.1, 120.9; anal. calcd for C19H13N3: C, 80.54; H, 4.62; N, 14.83; found: C, 80.48, H, 4.06, N, 14.44.
05 v/v); MP 138–140 °C; IR (KBr): 3056, 1529, 1486, 1418, 1315, 1062 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.39 (s, 1H), 8.7 (d, J = 7.2 Hz, 1H), 8.51 (d, J = 2.4 Hz, 1H), 8.36 (dt, J = 2 Hz, 8.8 Hz, 2H), 8.20–8.14 (m, 2H), 7.83 (dd, J = 1.6 Hz, 8.4 Hz, 1H), 7.82–7.76 (m, 4H), 7.68 (dt, J = 2.8 Hz, 12 Hz, 1H); 13C NMR (CDCl3, 100 MHz): δ = 150.8, 144.0, 143.0, 142.32, 141.7, 138.0, 137.0, 130.4, 129.6, 129.1, 128.3, 127.9, 121.1, 120.9; anal. calcd for C19H13N3: C, 80.54; H, 4.62; N, 14.83; found: C, 80.48, H, 4.06, N, 14.44.
2-(4-(5-Fluoropyridin-3-yl)phenyl)quinoxaline (4x). White solid; yield 90%; (Rf = 0.46 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 139–141 °C; IR (KBr): 3056, 1528, 1489, 1413, 1316, 1048, 694 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.38 (s, 1H), 8.52 (d, J = 2 Hz, 1H), 8.34 (d, J = 8.4 Hz, 2H), 8.20–8.13 (m, 2H), 8.09–8.05 (m, 1H), 7.84–7.78 (m, 2H), 7.76 (d, J = 8.4 Hz, 2H), 7.07 (dd, J = 3.2 Hz, 12 Hz, 1H); 13C NMR (CDCl3, 100 MHz); δ = 150.9, 145.9, 145.8, 143.0, 141.6, 139.3, 136.5, 134.0, 130.5, 129.6, 129.1, 128.1, 128.2, 127.7, 109.8, 109.5; anal. calcd for C19H12FN3: C, 75.74; H, 4.01; N, 13.95; found: C, 75.24, H, 3.94, N, 13.68.
05 v/v); MP 139–141 °C; IR (KBr): 3056, 1528, 1489, 1413, 1316, 1048, 694 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.38 (s, 1H), 8.52 (d, J = 2 Hz, 1H), 8.34 (d, J = 8.4 Hz, 2H), 8.20–8.13 (m, 2H), 8.09–8.05 (m, 1H), 7.84–7.78 (m, 2H), 7.76 (d, J = 8.4 Hz, 2H), 7.07 (dd, J = 3.2 Hz, 12 Hz, 1H); 13C NMR (CDCl3, 100 MHz); δ = 150.9, 145.9, 145.8, 143.0, 141.6, 139.3, 136.5, 134.0, 130.5, 129.6, 129.1, 128.1, 128.2, 127.7, 109.8, 109.5; anal. calcd for C19H12FN3: C, 75.74; H, 4.01; N, 13.95; found: C, 75.24, H, 3.94, N, 13.68.
2-(4-(6-Fluoropyridin-3-yl)phenyl)quinoxaline (4y). White solid; yield 90%; (Rf = 0.44 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 143–146 °C; IR (KBr): 3048, 1532, 1482, 1428, 1323, 1052, 702 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.35 (s, 1H), 8.54 (d, J = 8 Hz, 2H), 8.41 (s, 1H), 8.31–8.24 (m, 2H), 8.20–8.12 (m, 1H), 7.88–7.7 (m, 2H), 7.74 (d, J = 8.8 Hz, 2H), 7.16 (dd, J1 = 12.4 Hz, J2 = 3.2 Hz, 1H); 13C NMR (CDCl3, 100 MHz); δ = 164.3, 153.5, 147.1, 145.8, 143.0, 142.8, 141.7, 138.50, 136.5, 134.0, 130.5, 129.8, 129.6, 129.1, 128.2, 127.7, 109.8, 109.3; anal. calcd for C19H12FN3: C, 75.74, H, 4.01, N, 13.95; found: C, 75.44, H, 3.96, N, 13.56.
05 v/v); MP 143–146 °C; IR (KBr): 3048, 1532, 1482, 1428, 1323, 1052, 702 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.35 (s, 1H), 8.54 (d, J = 8 Hz, 2H), 8.41 (s, 1H), 8.31–8.24 (m, 2H), 8.20–8.12 (m, 1H), 7.88–7.7 (m, 2H), 7.74 (d, J = 8.8 Hz, 2H), 7.16 (dd, J1 = 12.4 Hz, J2 = 3.2 Hz, 1H); 13C NMR (CDCl3, 100 MHz); δ = 164.3, 153.5, 147.1, 145.8, 143.0, 142.8, 141.7, 138.50, 136.5, 134.0, 130.5, 129.8, 129.6, 129.1, 128.2, 127.7, 109.8, 109.3; anal. calcd for C19H12FN3: C, 75.74, H, 4.01, N, 13.95; found: C, 75.44, H, 3.96, N, 13.56.
2,3-Bis(4-methoxyphenyl)quinoxaline (5b). White solid; yield 96%; (Rf = 0.42 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 146–148 °C; (Lit: 147–148);18a IR (KBr): 3008, 2964, 2842, 1604, 1508, 1460, 1392, 1346, 1286, 1240, 1172, 1024, 828, 764 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.14–7.91 (m, 2H), 7.6–7.37 (m, 3H), 7.34 (d, J = 8.4 Hz, 4H), 7.33 (d, J = 7.2 Hz, 4H), 3.82 (s, 6H); LCMS (ESI) [M + H]+ calculated C22H18N2O2 343.14 found 343.20.
05 v/v); MP 146–148 °C; (Lit: 147–148);18a IR (KBr): 3008, 2964, 2842, 1604, 1508, 1460, 1392, 1346, 1286, 1240, 1172, 1024, 828, 764 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.14–7.91 (m, 2H), 7.6–7.37 (m, 3H), 7.34 (d, J = 8.4 Hz, 4H), 7.33 (d, J = 7.2 Hz, 4H), 3.82 (s, 6H); LCMS (ESI) [M + H]+ calculated C22H18N2O2 343.14 found 343.20.
2-Methyl-3-phenylquinoxaline (5d). White solid; yield 92%; (Rf = 0.44 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 54–56 °C; (Lit: 54–55);18b IR (KBr): 3122, 1510, 1461, 1318, 1054, 719 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.25–8.11 (m, 2H), 7.73–7.17 (m, 4H), 7.5–7.51 (m, 3H), 2.73 (s, 3H); LCMS (ESI) [M + H]+ calculated C15H12N2 221.10 found 221.17.
05 v/v); MP 54–56 °C; (Lit: 54–55);18b IR (KBr): 3122, 1510, 1461, 1318, 1054, 719 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.25–8.11 (m, 2H), 7.73–7.17 (m, 4H), 7.5–7.51 (m, 3H), 2.73 (s, 3H); LCMS (ESI) [M + H]+ calculated C15H12N2 221.10 found 221.17.
Quinoxaline (5e). White solid; yield 90%; (Rf = 0.42 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 30–32 °C; (Lit: 32–33);18c IR (KBr); 1498, 1364, 1202, 1128, 1028, 956 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.82 (s, 2H), 8.12 (d, J = 8 Hz, 2H), 7.76 (d, J = 8.8 Hz, 2H); LCMS (ESI) [M + H]+ calculated C8H6N2 131.05 found 131.10.
05 v/v); MP 30–32 °C; (Lit: 32–33);18c IR (KBr); 1498, 1364, 1202, 1128, 1028, 956 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.82 (s, 2H), 8.12 (d, J = 8 Hz, 2H), 7.76 (d, J = 8.8 Hz, 2H); LCMS (ESI) [M + H]+ calculated C8H6N2 131.05 found 131.10.
2-(4-Chlorophenyl)-3-phenylquinoxaline (5f). White solid; yield 94%; (Rf = 0.40 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 135–137 °C; (Lit: 136–138);18a IR (KBr): 3058, 2926, 1724, 1588, 1490, 1472, 1440, 1398, 1342, 1087, 1056, 1012, 966, 848, 806, 760, 696 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.20–8.13 (m, 2H), 8.09–8.07 (m, 2H), 8.05–7.82 (m, 4H), 7.80–7.78 (m, 3H), 7.75 (d, J = 8.4 Hz, 2H); LCMS (ESI) [M + H]+ calculated C20H13Cl2N2 317.08 found 317.14.
05 v/v); MP 135–137 °C; (Lit: 136–138);18a IR (KBr): 3058, 2926, 1724, 1588, 1490, 1472, 1440, 1398, 1342, 1087, 1056, 1012, 966, 848, 806, 760, 696 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.20–8.13 (m, 2H), 8.09–8.07 (m, 2H), 8.05–7.82 (m, 4H), 7.80–7.78 (m, 3H), 7.75 (d, J = 8.4 Hz, 2H); LCMS (ESI) [M + H]+ calculated C20H13Cl2N2 317.08 found 317.14.
2,3-Bis(4-methoxyphenyl)-6,7-dimethylquinoxaline (5h). Yellow solid; yield 95%; (Rf = 0.42 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 122–124 °C; (Lit: 123–125);18a IR (KBr): 2930, 2833, 1603, 1508, 1455, 1417, 1340, 1298, 1172, 1023, 965, 828 cm−1; 1H NMR (400 MHz, CDCl3): δ = 7.82 (s, 2H), 7.60 (d, J = 8.4 Hz, 4H), 7.10 (d, J = 7.6 Hz, 4H), 3.92 (s, 6H), 2.42 (s, 6H); LCMS (ESI) [M + H]+ calculated C24H22Cl2N2O2 371.17 found 371.23.
05 v/v); MP 122–124 °C; (Lit: 123–125);18a IR (KBr): 2930, 2833, 1603, 1508, 1455, 1417, 1340, 1298, 1172, 1023, 965, 828 cm−1; 1H NMR (400 MHz, CDCl3): δ = 7.82 (s, 2H), 7.60 (d, J = 8.4 Hz, 4H), 7.10 (d, J = 7.6 Hz, 4H), 3.92 (s, 6H), 2.42 (s, 6H); LCMS (ESI) [M + H]+ calculated C24H22Cl2N2O2 371.17 found 371.23.
6-Bromo-2,3-di(furan-2-yl)quinoxaline (5k). White solid; yield 96%; (Rf = 0.54 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 132–134 °C; (Lit: 131–133);18k IR (KBr): 3359, 1541, 1428, 1071, 835, 756 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.36 (d, J = 1.6 Hz, 1H), 8.03 (d, J = 8.4 Hz, 1H), 7.86 (dd, J = 2.4 Hz, 2.4 Hz, 1H), 7.68 (t, J = 1.6 Hz, 2H), 6.76–6.75 (m, 2H), 6.63–6.61 (m, 2H); LCMS (ESI) [M + H]+ calculated C16H9BrN2O2 341.98 found 341.06.
05 v/v); MP 132–134 °C; (Lit: 131–133);18k IR (KBr): 3359, 1541, 1428, 1071, 835, 756 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.36 (d, J = 1.6 Hz, 1H), 8.03 (d, J = 8.4 Hz, 1H), 7.86 (dd, J = 2.4 Hz, 2.4 Hz, 1H), 7.68 (t, J = 1.6 Hz, 2H), 6.76–6.75 (m, 2H), 6.63–6.61 (m, 2H); LCMS (ESI) [M + H]+ calculated C16H9BrN2O2 341.98 found 341.06.
6-Chloro-7-fluoro-2,3-diphenylquinoxaline (5m). White solid; yield 94%; (Rf = 0.58 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 158–160 °C; (Lit: 150–152);18f IR (KBr): 3418, 1466, 1344, 1216, 702 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.24 (d, J = 8.4 Hz, 1H), 7.83 (d, J = 8.8 Hz, 1H), 7.80–7.28 (m, 4H), 7.14–7.01 (m, 6H); LCMS (ESI) [M + H]+ calculated C20H12Cl2N2FCl 335.07 found 335.14.
05 v/v); MP 158–160 °C; (Lit: 150–152);18f IR (KBr): 3418, 1466, 1344, 1216, 702 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.24 (d, J = 8.4 Hz, 1H), 7.83 (d, J = 8.8 Hz, 1H), 7.80–7.28 (m, 4H), 7.14–7.01 (m, 6H); LCMS (ESI) [M + H]+ calculated C20H12Cl2N2FCl 335.07 found 335.14.
6-Nitro-2,3-diphenylquinoxaline (5n). Yellow solid; yield 95%; (Rf = 0.52 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 183–185 °C; (Lit: 184–186);18a IR (KBr): 3080, 3057, 1612, 1518, 1398, 1340, 1054, 1023, 810, 767, 698 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.08 (d, J = 2.4 Hz, 1H), 8.54 (dd, J = 11.6 Hz, 2.4 Hz, 1H), 8.30 (d, J = 9.2 Hz, 1H), 7.58–7.54 (m, 4H), 7.43–7.36 (m, 6H); LCMS (ESI) [M + H]+ calculated C20H13Cl2N3O2 328.10 found 328.17.
05 v/v); MP 183–185 °C; (Lit: 184–186);18a IR (KBr): 3080, 3057, 1612, 1518, 1398, 1340, 1054, 1023, 810, 767, 698 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.08 (d, J = 2.4 Hz, 1H), 8.54 (dd, J = 11.6 Hz, 2.4 Hz, 1H), 8.30 (d, J = 9.2 Hz, 1H), 7.58–7.54 (m, 4H), 7.43–7.36 (m, 6H); LCMS (ESI) [M + H]+ calculated C20H13Cl2N3O2 328.10 found 328.17.
2,3-Diphenyl-4a,5,6,7,8,8a-hexahydroquinoxaline (5p). White solid; yield 90%; (Rf = 0.48 in hexanes/EtOAc 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 171–176 °C; (Lit: 170–172);18d IR (KBr): 3048, 2936, 1612 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 7.55–7.49 (m, 4H), 7.28–7.20 (m, 6H), 2.84 (t, J = 9.2 Hz, 2H), 2.50 (d, J = 13.6 Hz, 2H), 1.89 (d, J = 9.2 Hz, 2H), 1.64–1.62 (m, 2H), 1.47–1.39 (m, 2H); LCMS (ESI) [M + H]+ calculated C20H20N2 289.16 found 289.22.
05 v/v); MP 171–176 °C; (Lit: 170–172);18d IR (KBr): 3048, 2936, 1612 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 7.55–7.49 (m, 4H), 7.28–7.20 (m, 6H), 2.84 (t, J = 9.2 Hz, 2H), 2.50 (d, J = 13.6 Hz, 2H), 1.89 (d, J = 9.2 Hz, 2H), 1.64–1.62 (m, 2H), 1.47–1.39 (m, 2H); LCMS (ESI) [M + H]+ calculated C20H20N2 289.16 found 289.22.
Note added after first publication
This article replaces the version first published on 26th February 2016, which contained errors in the characterisation data. The original HRMS data has been removed and replaced with LCMS data and elemental analysis for all new compounds reported. Previously known compounds have been referenced to the literature and LCMS data has been added. A new compound 4y has been added and the crystal structure determined as shown in Fig. 2.
Acknowledgements
KSR thanks to DST Indo-Korea (grant no. INT/Korea/dated 13.09.2011 and KBH thanks to UGC for providng UGC-BSR fellowship. We are also thankful to the Institute of Excellence (IOE) for providing NMR and XRD facility at the University of Mysore, Manasagangotri, Mysuru-570006, India, for the spectral data.
References
-
(a) A. Gomtsyan, E. K. Bayburt, R. G. Schmidt, G. Z. Zheng, R. J. Perner, S. Didomenico, J. R. Koenig, S. Turner, T. Jinkerson and I. Drizin, J. Med. Chem., 2005, 48, 744–752 CrossRef CAS PubMed;
(b) W. He, M. R. Myers, B. Hanney, A. P. Spada, G. Bilder, H. Galzcinski, D. Amin, S. Needle, K. Page and Z. Jayyosi, Bioorg. Med. Chem. Lett., 2003, 13, 3097–3100 CrossRef CAS PubMed;
(c) A. Jaso, B. Zarranz, I. Aldana and A. Monge, J. Med. Chem., 2005, 48, 2019–2025 CrossRef CAS PubMed;
(d) R. Sarges, H. R. Howard, R. G. Browne, L. A. Lebel, P. A. Seymour and B. K. Koe, J. Med. Chem., 1990, 33, 2240–2254 CrossRef CAS PubMed;
(e) L. E. Seitz, W. J. Suling and R. C. Reynolds, J. Med. Chem., 2002, 45, 5604–5606 CrossRef CAS PubMed.
-
(a) P. P. Castro, G. Zhao, G. A. Masangkay, C. Hernandez and L. M. Gutierrez-Tunstad, Org. Lett., 2004, 6, 333–336 CrossRef CAS PubMed;
(b) S. Dailey, W. J. Feast, R. J. Peace, I. C. Sage, S. Till and E. L. Wood, J. Mater. Chem., 2001, 11, 2238–2243 RSC;
(c) M. Kulkarni, A. Kumbhar, H. Mohan and B. Rao, Dalton Trans., 2009, 6185–6191 RSC;
(d) L.-C. Rong, X.-Y. Li, C.-S. Yao, H.-Y. Wang and D.-Q. Shi, Acta Crystallogr., Sect. E: Struct. Rep. Online, 2006, 62, o1959–o1960 CrossRef CAS;
(e) S. Saravanakumar, M. K. Kindermann, J. Heinicke and M. Köckerling, Chem. Commun., 2006, 640–664 RSC;
(f) J. L. Sessler, H. Maeda, T. Mizuno, V. M. Lynch and H. Furuta, J. Am. Chem. Soc., 2002, 124, 13474–13479 CrossRef CAS PubMed;
(g) J. L. Sessler, H. Maeda, T. Mizuno, V. M. Lynch and H. Furuta, J. Am. Chem. Soc., 2002, 124, 13474–13479 CrossRef CAS PubMed;
(h) K. J. Thomas, J. T. Lin, Y.-T. Tao and C. Chuen, J. Mater. Chem., 2002, 12, 3516–3522 RSC.
-
(a) C. S. Cho and S. G. Oh, Tetrahedron Lett., 2006, 47, 5633–5636 CrossRef CAS;
(b) V. Jeena and R. S. Robinson, Tetrahedron Lett., 2014, 55, 642–645 CrossRef CAS;
(c) M. Jeganathan, A. Dhakshinamoorthy and K. Pitchumani, Tetrahedron Lett., 2014, 55, 1616–1620 CrossRef CAS;
(d) A. Kamal, K. S. Babu, S. Faazil, S. A. Hussaini and A. B. Shaik, RSC Adv., 2014, 4, 46369–46377 RSC;
(e) A. Kamal, K. S. Babu, S. A. Hussaini, R. Mahesh and A. Alarifi, Tetrahedron Lett., 2015, 56, 2803–2808 CrossRef CAS;
(f) F. Pan, T.-M. Chen, J.-J. Cao, J.-P. Zou and W. Zhang, Tetrahedron Lett., 2012, 53, 2508–2510 CrossRef CAS;
(g) J. Richard, Chem. Commun., 2003, 2286–2287 Search PubMed;
(h) S. Sithambaram, Y. Ding, W. Li, X. Shen, F. Gaenzler and S. L. Suib, Green Chem., 2008, 10, 1029–1032 RSC;
(i) S. YeonáKim, K. HyunáPark and Y. KeunáChung, Chem. Commun., 2005, 1321–1323 Search PubMed.
-
(a) K. Kumar, S. R. Mudshinge, S. Goyal, M. Gangar and V. A. Nair, Tetrahedron Lett., 2015, 56, 1266–1271 CrossRef CAS;
(b) B. Madhav, S. N. Murthy, V. P. Reddy, K. R. Rao and Y. Nageswar, Tetrahedron Lett., 2009, 50, 6025–6028 CrossRef CAS;
(c) H. Meshram, G. S. Kumar, P. Ramesh and B. C. Reddy, Tetrahedron Lett., 2010, 51, 2580–2585 CrossRef CAS;
(d) B. Tanwar, P. Purohit, B. N. Raju, D. Kumar, D. N. Kommi and A. K. Chakraborti, RSC Adv., 2015, 5, 11873–11883 RSC;
(e) J.-P. Wan, S.-F. Gan, J.-M. Wu and Y. Pan, Green Chem., 2009, 11, 1633–1637 RSC.
-
(a) D. Bandyopadhyay, S. Mukherjee, R. R. Rodriguez and B. K. Banik, Molecules, 2010, 15, 4207–4212 CrossRef CAS PubMed;
(b) R. S. Bhosale, S. R. Sarda, S. S. Ardhapure, W. N. Jadhav, S. R. Bhusare and R. P. Pawar, Tetrahedron Lett., 2005, 46, 7183–7186 CrossRef CAS;
(c) J.-J. Cai, J.-P. Zou, X.-Q. Pan and W. Zhang, Tetrahedron Lett., 2008, 49, 7386–7390 CrossRef CAS;
(d) H. R. Darabi, K. Aghapoor, F. Mohsenzadeh, F. Taala, N. Asadollahnejad and A. Badiei, Catal. Lett., 2009, 133, 84–89 CrossRef CAS;
(e) A. Dhakshinamoorthy, K. Kanagaraj and K. Pitchumani, Tetrahedron Lett., 2011, 52, 69–73 CrossRef CAS;
(f) A. Hasaninejad, M. Shekouhy and A. Zare, Catal. Sci. Technol., 2012, 2, 201–214 RSC;
(g) M. M. Heravi, M. H. Tehrani, K. Bakhtiari and H. A. Oskooie, Catal. Commun., 2007, 8, 1341–1344 CrossRef CAS;
(h) H. K. Kadam, S. Khan, R. A. Kunkalkar and S. G. Tilve, Tetrahedron Lett., 2013, 54, 1003–1007 CrossRef CAS;
(i) D. Kumar, K. Seth, D. N. Kommi, S. Bhagat and A. K. Chakraborti, RSC Adv., 2013, 3, 15157–15168 RSC;
(j) A. Kumbhar, S. Kamble, M. Barge, G. Rashinkar and R. Salunkhe, Tetrahedron Lett., 2012, 53, 2756–2760 CrossRef CAS;
(k) S. V. More, M. Sastry, C.-C. Wang and C.-F. Yao, Tetrahedron Lett., 2005, 46, 6345–6348 CrossRef CAS;
(l) S. V. More, M. Sastry and C.-F. Yao, Green Chem., 2006, 8, 91–95 RSC;
(m) S. Paul and B. Basu, Tetrahedron Lett., 2011, 52, 6597–6602 CrossRef CAS;
(n) J. S. Yadav, B. V. Subba Reddy, K. Premalatha and K. Shiva Shankar, Synthesis, 2008, 3787–3792 CrossRef CAS.
-
(a) S. Okumura, Y. Takeda, K. Kiyokawa and S. Minakata, Chem. Commun., 2013, 49, 9266–9268 RSC;
(b) K. S. Vadagaonkar, H. P. Kalmode, K. Murugan and A. C. Chaskar, RSC Adv., 2015, 5, 5580–5590 RSC;
(c) K. D. R. Viswanadham, M. P. Reddy, P. Sathyanarayana, O. Ravi, R. Kant and S. R. Bathula, Chem. Commun., 2014, 50, 13517–13520 RSC;
(d) N. Xu, D.-W. Gu, Y.-S. Dong, F.-P. Yi, L. Cai, X.-Y. Wu and X.-X. Guo, Tetrahedron Lett., 2015, 56, 1517–1519 CrossRef CAS;
(e) Y. Xu and X. Wan, Tetrahedron Lett., 2013, 54, 642–645 CrossRef CAS.
-
(a) S. Antoniotti and E. Duñach, Tetrahedron Lett., 2002, 43, 3971–3973 CrossRef CAS;
(b) M. M. Ibrahim, D. Grau, F. Hampel and S. B. Tsogoeva, Eur. J. Inorg. Chem., 2014, 1401–1405 CrossRef CAS;
(c) E. C. Taylor, C. A. Maryanoff and J. S. Skotnicki, J. Org. Chem., 1980, 45, 2512–2515 CrossRef CAS.
- N. Xekoukoulotakis, C. Hadjiantoniou-Maroulis and A. Maroulis, Tetrahedron Lett., 2000, 41, 10299–10302 CrossRef CAS.
-
(a) J. R. Dunetz, Y. Xiang, A. Baldwin and J. Ringling, Org. Lett., 2011, 13, 5048–5051 CrossRef CAS PubMed;
(b) L. Carpino, Chem. Commun., 1999, 1847–1848 Search PubMed;
(c) L. Carpino, Chem. Commun., 1999, 1847–1848 Search PubMed.
-
(a) J. K. Augustine, A. Bombrun and S. Venkatachaliah, Tetrahedron Lett., 2011, 52, 6814–6818 CrossRef CAS;
(b) M. Jida and B. Deprez, New J. Chem., 2012, 36, 869–873 RSC.
- G. Coulthard, W. P. Unsworth and R. J. Taylor, Tetrahedron Lett., 2015, 56, 3113–3116 CrossRef CAS.
- F. L. Zumpe, M. Flüβ, K. Schmitz and A. Lender, Tetrahedron Lett., 2007, 48, 1421–1423 CrossRef CAS.
- J. K. Augustine, V. Vairaperumal, S. Narasimhan, P. Alagarsamy and A. Radhakrishnan, Tetrahedron, 2009, 65, 9989–9996 CrossRef CAS.
- M. Desroses, K. Wieckowski, M. Stevens and L. R. Odell, Tetrahedron Lett., 2011, 52, 4417–4420 CrossRef CAS.
-
(a) G. M. Raghavendra, A. B. Ramesha, C. N. Revanna, K. N. Nandeesh, K. Mantelingu and K. S. Rangappa, Tetrahedron Lett., 2011, 52, 5571–5574 CrossRef CAS;
(b) K. S. S. Kumar, T. R. Swaroop, K. B. Harsha, K. H. Narasimhamurthy and K. S. Rangappa, Tetrahedron Lett., 2012, 53, 5619–5623 CrossRef;
(c) A. B. Ramesha, G. M. Raghavendra, K. N. Nandeesh, K. S. Rangappa and K. Mantelingu, Tetrahedron Lett., 2013, 54, 95–100 CrossRef CAS.
-
(a) C. S. P. Kumar, K. B. Harsha, K. Mantelingu and K. S. Rangappa, RSC Adv., 2015, 5, 61664–61670 RSC;
(b) C. S. P. Kumar, K. B. Harsha, N. C. Sandhya, A. B. Ramesha, K. Mantelingu and K. S. Rangappa, New J. Chem., 2015, 39, 8397–8404 RSC;
(c) K. H. Narasimhamurthy, S. Chandrappa, K. S. S. Kumar, K. B. Harsha, H. Ananda and K. S. Rangappa, RSC Adv., 2014, 4, 34479–34486 RSC.
-
(a) R. Roopashree, C. D. Mohan, T. R. Swaroop, S. Jagadish, B. Raghava, K. S. Balaji, S. Jayarama and K. S. Rangappa, Bioorg. Med. Chem. Lett., 2015, 25, 2589–2593 CrossRef CAS PubMed;
(b) H. Bharathkumar, C. D. Mohan, S. Rangappa, T. Kang, H. Keerthy, J. E. Fuchs, N. H. Kwon, A. Bender, S. Kim and K. S. Rangappa, Org. Biomol. Chem., 2015, 13, 9381–9387 RSC.
-
(a) M. Lian, Q. Li, Y. Zhu, G. Yin and A. Wu, Tetrahedron, 2012, 68, 9598–9605 CrossRef CAS;
(b) D. Kumar, K. Seth, D. N. Kommi, S. Bhagat and A. K. Chakraborti, RSC Adv., 2013, 3, 15157–15168 RSC;
(c) S. Paul and B. Basu, Tetrahedron Lett., 2011, 52, 6597–6602 CrossRef CAS;
(d) H. K. Kadam, S. Khan, R. A. Kunkalkar and S. G. Tilve, Tetrahedron Lett., 2013, 54, 1003–1007 CrossRef CAS;
(e) A. Kamal, K. S. Babu, S. A. Hussaini, R. Mahesh and A. Alarifi, Tetrahedron Lett., 2015, 56, 2803–2808 CrossRef CAS;
(f) K. S. Vadagaonkar, H. P. Kalmode, K. Murugan and A. C. Chaskar, RSC Adv., 2015, 5, 5580–5590 RSC;
(g) L. J. Martin, A. L. Marzinzik, S. V. Ley and I. R. Baxendale, Org. Lett., 2010, 13, 320–323 CrossRef PubMed;
(h) J.-P. Wan, S.-F. Gan, J.-M. Wu and Y. Pan, Green Chem., 2009, 11, 1633–1637 RSC;
(i) W. Lv, B. Xiong, Z. Tan, H. Jiang and M. Zhang, Asian J. Org. Chem., 2015, 4, 1127–1131 CrossRef CAS;
(j) A. Fürstner, A. Leitner, M. Méndez and H. Krause, J. Am. Chem. Soc., 2002, 124, 13856–13863 CrossRef;
(k) D. Q. Shi, G. L. Dou, S. N. Ni, J. W. Shi and X. Y. Li, J. Heterocycl. Chem., 2008, 45, 1797–1801 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. 1H and 13C NMR data and single crystal X-ray data for compound 4y. CCDC 1480288. For ESI and crystallographic data in CIF or other electronic format. See DOI: 10.1039/c6ra03078e |
|
| This journal is © The Royal Society of Chemistry 2016 |
Click here to see how this site uses Cookies. View our privacy policy here. ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMSO (ratio 2
DMSO (ratio 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) followed by the addition of 1 equivalent of T3P® (50% solution in EtOAc) under stirring at 0 °C, then RT for 10 h, which afforded the desired 2,3-diphenylquinoxaline 3e in 25% yield. For the same reaction we checked if different equivalents of T3P® could affect the course of the reaction and the yield of 3e. Therefore, we carried out the oxidation–condensation using various equivalents of T3P®. The best result was obtained when the reaction between 1,2-diphenylethanone and o-PD was carried out with 2.0 equivalents of T3P® in the presence of DMSO and stirred at RT for 6 h. We also studied the reaction by keeping the equivalent of T3P® (2.0 eq.) constant and varying both the temperature and time. Notably, we were not able to achieve a greater percentage yield compared to the yield obtained at RT conditions. The optimization results are summarized in Table 1. Thus, a clear optimization of the reaction conditions to obtain quinoxalines from a α-hydroxyl ketone with o-PD was revealed from our studies, which involved using 2.0 equivalents of T3P® and stirring the reaction mixture at 0 °C then RT for 6 h. The product was isolated by initially washing the reaction mixture with water followed by brine, then purified by column chromatography using 60–120 silica gel. The oxidation–condensation or cyclodehydrating properties of T3P® helped to generalize the reaction procedure. Next, we carried out reactions with a series of electronically diverse α-hydroxy ketones and o-PDs mediated by T3P® in a solvent mixture of DMSO
1) followed by the addition of 1 equivalent of T3P® (50% solution in EtOAc) under stirring at 0 °C, then RT for 10 h, which afforded the desired 2,3-diphenylquinoxaline 3e in 25% yield. For the same reaction we checked if different equivalents of T3P® could affect the course of the reaction and the yield of 3e. Therefore, we carried out the oxidation–condensation using various equivalents of T3P®. The best result was obtained when the reaction between 1,2-diphenylethanone and o-PD was carried out with 2.0 equivalents of T3P® in the presence of DMSO and stirred at RT for 6 h. We also studied the reaction by keeping the equivalent of T3P® (2.0 eq.) constant and varying both the temperature and time. Notably, we were not able to achieve a greater percentage yield compared to the yield obtained at RT conditions. The optimization results are summarized in Table 1. Thus, a clear optimization of the reaction conditions to obtain quinoxalines from a α-hydroxyl ketone with o-PD was revealed from our studies, which involved using 2.0 equivalents of T3P® and stirring the reaction mixture at 0 °C then RT for 6 h. The product was isolated by initially washing the reaction mixture with water followed by brine, then purified by column chromatography using 60–120 silica gel. The oxidation–condensation or cyclodehydrating properties of T3P® helped to generalize the reaction procedure. Next, we carried out reactions with a series of electronically diverse α-hydroxy ketones and o-PDs mediated by T3P® in a solvent mixture of DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc in order to obtain the desired quinoxalines 3a–k. The o-PD and α-hydroxy ketone without any substitution afforded a good yield of product, while the α-hydroxy ketone containing an electron-donating group favoured the formation of the desired quinoxalines in high yields, when compared to reactions where o-PD contained an electron-withdrawing group. These results are summarized in Scheme 2. The scope of the reaction was successfully extended to other substrates such as electronically demanding α-bromo ketones with o-PD or substituted o-PDs, which also underwent the title reaction under RT conditions to obtain the corresponding quinoxalines (4a–y). Similarly, the reaction of an α-halo ketone and o-PD without any substitution favoured the formation of a quinoxaline in slightly higher yield when compared to reactions using substituted α-halo ketones or o-PDs. These results are summarized in Scheme 3. We then extended our studies to further increase the substrate scope. We carried out the condensation of 1,2-dicarbonyl compounds with o-PD under mild reaction conditions at RT in the presence of T3P® to produce the desired quinoxaline derivatives (5a–p). Interestingly, the reaction between an aliphatic diketone and o-PD underwent the title reaction to afford the desired quinoxaline in lower yield when compared to aromatic 1,2-diketones. In the case where both aromatic starting materials were without substitution, the reaction favoured the formation of the quinoxaline in the highest yield. These results are summarized in Scheme 4.
EtOAc in order to obtain the desired quinoxalines 3a–k. The o-PD and α-hydroxy ketone without any substitution afforded a good yield of product, while the α-hydroxy ketone containing an electron-donating group favoured the formation of the desired quinoxalines in high yields, when compared to reactions where o-PD contained an electron-withdrawing group. These results are summarized in Scheme 2. The scope of the reaction was successfully extended to other substrates such as electronically demanding α-bromo ketones with o-PD or substituted o-PDs, which also underwent the title reaction under RT conditions to obtain the corresponding quinoxalines (4a–y). Similarly, the reaction of an α-halo ketone and o-PD without any substitution favoured the formation of a quinoxaline in slightly higher yield when compared to reactions using substituted α-halo ketones or o-PDs. These results are summarized in Scheme 3. We then extended our studies to further increase the substrate scope. We carried out the condensation of 1,2-dicarbonyl compounds with o-PD under mild reaction conditions at RT in the presence of T3P® to produce the desired quinoxaline derivatives (5a–p). Interestingly, the reaction between an aliphatic diketone and o-PD underwent the title reaction to afford the desired quinoxaline in lower yield when compared to aromatic 1,2-diketones. In the case where both aromatic starting materials were without substitution, the reaction favoured the formation of the quinoxaline in the highest yield. These results are summarized in Scheme 4.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. 1 mmol of 1a, 1.2 mmol of 2a were used in the case of 3e.b T3P® (50% solution in EtOAc) was used to carry out the reactions.c Isolated yield after purification by column chromatography.
1. 1 mmol of 1a, 1.2 mmol of 2a were used in the case of 3e.b T3P® (50% solution in EtOAc) was used to carry out the reactions.c Isolated yield after purification by column chromatography.



![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc (ratio 2
EtOAc (ratio 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was added ortho-phenylenediamine (1 mmol). This was followed by the addition of T3P® (2.0 mmol) at 0 °C, then the reaction mixture was kept at RT for 6 h. The progress of the reaction was monitored by TLC. The reaction mixture was quenched with ice cold water and the reaction mixture was extracted with EtOAc (×2). The collected organic layers were washed with brine solution and dried over anhydrous sodium sulphate. The organic solvent was removed under reduced pressure to afford the desired quinoxaline, which was purified by column chromatography using hexanes
1) was added ortho-phenylenediamine (1 mmol). This was followed by the addition of T3P® (2.0 mmol) at 0 °C, then the reaction mixture was kept at RT for 6 h. The progress of the reaction was monitored by TLC. The reaction mixture was quenched with ice cold water and the reaction mixture was extracted with EtOAc (×2). The collected organic layers were washed with brine solution and dried over anhydrous sodium sulphate. The organic solvent was removed under reduced pressure to afford the desired quinoxaline, which was purified by column chromatography using hexanes![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc as an eluent.
EtOAc as an eluent.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc as an eluent.
EtOAc as an eluent.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 75–77 °C (Lit: 75–78);18c IR (KBr): 3055, 1545, 1480, 1445, 1312, 1035 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.33 (s, 1H), 8.21–8.11 (m, 4H), 7.81–7.73 (m, 2H), 7.59–7.52 (m, 3H); LCMS (ESI) [M + H]+ calculated C14H10N2 207.0877 found 207.14.
05 v/v); MP 75–77 °C (Lit: 75–78);18c IR (KBr): 3055, 1545, 1480, 1445, 1312, 1035 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.33 (s, 1H), 8.21–8.11 (m, 4H), 7.81–7.73 (m, 2H), 7.59–7.52 (m, 3H); LCMS (ESI) [M + H]+ calculated C14H10N2 207.0877 found 207.14.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 92–94 °C (Lit: 91–93);18a IR (KBr): 3058, 1615, 1554, 1475, 1445, 1310, 1040 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.40 (s, 1H), 8.55–8.38 (m, 4H), 8.37–8.20 (m, 2H), 7.88 (d, J = 8 Hz, 2H), 2.40 (s, 3H); LCMS (ESI) [M + H]+ calculated C15H12N2 221.10 found 221.16.
05 v/v); MP 92–94 °C (Lit: 91–93);18a IR (KBr): 3058, 1615, 1554, 1475, 1445, 1310, 1040 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.40 (s, 1H), 8.55–8.38 (m, 4H), 8.37–8.20 (m, 2H), 7.88 (d, J = 8 Hz, 2H), 2.40 (s, 3H); LCMS (ESI) [M + H]+ calculated C15H12N2 221.10 found 221.16.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 129–130 °C (Lit: 130–131);18a IR (KBr): 3055, 3040, 2965, 1535, 1480, 1445, 1310, 1210, 1020, 1005 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.22 (s, 1H), 8.20 (d, J = 11.2 Hz, 2H), 7.89 (s, 1H), 7.88 (s, 1H), 7.65–7.36 (m, 3H), 3.41 (s, 6H); LCMS (ESI) [M + H]+ calculated C16H14N2 235.11 found 235.18.
05 v/v); MP 129–130 °C (Lit: 130–131);18a IR (KBr): 3055, 3040, 2965, 1535, 1480, 1445, 1310, 1210, 1020, 1005 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.22 (s, 1H), 8.20 (d, J = 11.2 Hz, 2H), 7.89 (s, 1H), 7.88 (s, 1H), 7.65–7.36 (m, 3H), 3.41 (s, 6H); LCMS (ESI) [M + H]+ calculated C16H14N2 235.11 found 235.18.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 113–115 °C; (Lit: 114–115);18h IR (KBr): 3025, 2973, 1605, 1530, 1480, 1442, 1315, 1045, 1025, 860, 825 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.36 (s, 1H), 8.13 (d, J = 7.2 Hz, 2H), 8.12 (s, 1H), 7.82 (s, 1H), 7.81 (s, 1H), 7.09 (d, J = 8 Hz, 2H), 2.63 (s, 3H), 2.33 (s, 6H); LCMS (ESI) [M + H]+ calculated C17H16N2 249.13 found 249.19.
05 v/v); MP 113–115 °C; (Lit: 114–115);18h IR (KBr): 3025, 2973, 1605, 1530, 1480, 1442, 1315, 1045, 1025, 860, 825 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.36 (s, 1H), 8.13 (d, J = 7.2 Hz, 2H), 8.12 (s, 1H), 7.82 (s, 1H), 7.81 (s, 1H), 7.09 (d, J = 8 Hz, 2H), 2.63 (s, 3H), 2.33 (s, 6H); LCMS (ESI) [M + H]+ calculated C17H16N2 249.13 found 249.19.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 116–118 °C; (Lit: 117–119);18a IR (KBr): 3056, 1635, 1475, 1440, 1345, 1075, 1055, 770, 695 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.18 (dd, J = 6.8 Hz, 3.2 Hz, 2H), 7.77 (dd, J = 3.6 Hz, 3.2 Hz, 2H), 7.53–7.50 (m, 4H), 7.38–7.31 (m, 6H); LCMS (ESI) [M + H]+ calculated C20H14N2 283.12 found 283.18.
05 v/v); MP 116–118 °C; (Lit: 117–119);18a IR (KBr): 3056, 1635, 1475, 1440, 1345, 1075, 1055, 770, 695 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.18 (dd, J = 6.8 Hz, 3.2 Hz, 2H), 7.77 (dd, J = 3.6 Hz, 3.2 Hz, 2H), 7.53–7.50 (m, 4H), 7.38–7.31 (m, 6H); LCMS (ESI) [M + H]+ calculated C20H14N2 283.12 found 283.18.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 110–112 °C; (Lit: 112–114);18a IR (KBr): 3055, 2915, 1614, 1553, 1519, 1470, 1438, 1388, 1345, 1056, 1020, 974, 754, 696 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.26–8.14 (m, 2H), 7.99–7.95 (m, 2H), 7.83–7.78 (m, 2H), 7.72 (d, J = 7.6 Hz, 2H), 7.69–7.40 (m, 3H), 7.13 (d, J = 6.8 Hz, 2H), 2.42 (s, 3H); LCMS (ESI) [M + H]+ calculated C21H16N2 297.13 found 297.20.
05 v/v); MP 110–112 °C; (Lit: 112–114);18a IR (KBr): 3055, 2915, 1614, 1553, 1519, 1470, 1438, 1388, 1345, 1056, 1020, 974, 754, 696 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.26–8.14 (m, 2H), 7.99–7.95 (m, 2H), 7.83–7.78 (m, 2H), 7.72 (d, J = 7.6 Hz, 2H), 7.69–7.40 (m, 3H), 7.13 (d, J = 6.8 Hz, 2H), 2.42 (s, 3H); LCMS (ESI) [M + H]+ calculated C21H16N2 297.13 found 297.20.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 111–113 °C; (Lit: 110–112);18a IR (KBr): 3054, 2938, 1617, 1485, 1444, 1200, 1056, 1021, 812, 768, 698 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.10 (d, J = 8.4 Hz, 1H), 8.09 (s, 1H), 7.92 (dd, J = 9.6 Hz, 1.6 Hz, 1H), 7.78–7.69 (m, 4H), 7.68–7.31 (m, 6H), 2.77 (s, 3H); LCMS (ESI) [M + H]+ calculated C21H16N2 297.13 found 297.19.
05 v/v); MP 111–113 °C; (Lit: 110–112);18a IR (KBr): 3054, 2938, 1617, 1485, 1444, 1200, 1056, 1021, 812, 768, 698 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.10 (d, J = 8.4 Hz, 1H), 8.09 (s, 1H), 7.92 (dd, J = 9.6 Hz, 1.6 Hz, 1H), 7.78–7.69 (m, 4H), 7.68–7.31 (m, 6H), 2.77 (s, 3H); LCMS (ESI) [M + H]+ calculated C21H16N2 297.13 found 297.19.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 160–162 °C; (Lit: 117–119);18a IR (KBr): 2965, 2936, 1756, 1617, 1482 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.04 (d, J = 8.8 Hz, 1H), 7.95–7.92 (m, 4H), 7.91 (d, J = 2 Hz, 1H), 7.59 (dd, J = 9.2 Hz, 2.8 Hz, 1H), 7.37–7.34 (m, 6H), 3.83 (s, 3H); LCMS (ESI) [M + H]+ calculated C21H16N2O 313.12 found 313.18.
05 v/v); MP 160–162 °C; (Lit: 117–119);18a IR (KBr): 2965, 2936, 1756, 1617, 1482 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.04 (d, J = 8.8 Hz, 1H), 7.95–7.92 (m, 4H), 7.91 (d, J = 2 Hz, 1H), 7.59 (dd, J = 9.2 Hz, 2.8 Hz, 1H), 7.37–7.34 (m, 6H), 3.83 (s, 3H); LCMS (ESI) [M + H]+ calculated C21H16N2O 313.12 found 313.18.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 117–119 °C; (Lit: 118–120);18a IR (KBr); 3040, 1588, 1542, 1466, 1442, 1390, 1318, 1182, 1060, 1022, 973, 914, 828, 764, 694 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.35 (s, 1H), 8.03 (d, J = 9.2 Hz, 1H), 7.83 (d, J = 8.4 Hz, 1H), 7.50 (d, J = 6.8 Hz, 4H), 7.39–7.31 (m, 6H); LCMS (ESI) [M + H]+ calculated C20H13BrN2 361.03 found 361.09.
05 v/v); MP 117–119 °C; (Lit: 118–120);18a IR (KBr); 3040, 1588, 1542, 1466, 1442, 1390, 1318, 1182, 1060, 1022, 973, 914, 828, 764, 694 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.35 (s, 1H), 8.03 (d, J = 9.2 Hz, 1H), 7.83 (d, J = 8.4 Hz, 1H), 7.50 (d, J = 6.8 Hz, 4H), 7.39–7.31 (m, 6H); LCMS (ESI) [M + H]+ calculated C20H13BrN2 361.03 found 361.09.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 118–120 °C; (Lit: 117–120);18a IR (KBr): 3047, 1602, 1548, 1464, 1442, 1338, 1068, 828, 800, 764, 694 cm−1; 1H NMR (400 MHz, CDCl3): δ = 7.96 (d, J = 8.4 Hz, 1H), 7.90–7.64 (m, 5H), 7.60–7.34 (m. 6H), 2.77 (s, 3H), 2.48 (s, 3H); LCMS (ESI) [M + H]+ calculated C21H16N2 317.08 found 317.14.
05 v/v); MP 118–120 °C; (Lit: 117–120);18a IR (KBr): 3047, 1602, 1548, 1464, 1442, 1338, 1068, 828, 800, 764, 694 cm−1; 1H NMR (400 MHz, CDCl3): δ = 7.96 (d, J = 8.4 Hz, 1H), 7.90–7.64 (m, 5H), 7.60–7.34 (m. 6H), 2.77 (s, 3H), 2.48 (s, 3H); LCMS (ESI) [M + H]+ calculated C21H16N2 317.08 found 317.14.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 131–133 °C; IR (KBr): 3356, 1614, 1538, 1438, 1081, 835, 1H NMR (CDCl3, 400 MHz): δ = 8.31 (s, 1H), 8.20 (d, J = 8.4 Hz, 1H), 7.94 (t, J = 4 Hz, 1H), 7.92 (t, J = 3.6 Hz, 1H), 7.65–7.64 (m, 2H), 7.59 (dd, J = 7.2 Hz, 2.8 Hz, 2H), 7.39–7.34 (m, 1H), 7.17 (t, J = 9.6 Hz, 1H), 6.72–6.69 (m, 2H), 6.59–6.58 (m, 2H); 13C NMR (CDCl3, 100 MHz): δ = 144.9, 144.4, 130.6, 130.3, 129.6, 129.3, 128.0, 127.2, 126.2, 113.6, 113.2, 112.1; LCMS (ESI) [M + H]+ calculated C22H14N2O2 339.10 found 339.17; anal. calcd for C21H16N2: C, 78.09; H, 4.17; N, 8.28; found: C, 77.78; H, 3.97; N, 7.96.
05 v/v); MP 131–133 °C; IR (KBr): 3356, 1614, 1538, 1438, 1081, 835, 1H NMR (CDCl3, 400 MHz): δ = 8.31 (s, 1H), 8.20 (d, J = 8.4 Hz, 1H), 7.94 (t, J = 4 Hz, 1H), 7.92 (t, J = 3.6 Hz, 1H), 7.65–7.64 (m, 2H), 7.59 (dd, J = 7.2 Hz, 2.8 Hz, 2H), 7.39–7.34 (m, 1H), 7.17 (t, J = 9.6 Hz, 1H), 6.72–6.69 (m, 2H), 6.59–6.58 (m, 2H); 13C NMR (CDCl3, 100 MHz): δ = 144.9, 144.4, 130.6, 130.3, 129.6, 129.3, 128.0, 127.2, 126.2, 113.6, 113.2, 112.1; LCMS (ESI) [M + H]+ calculated C22H14N2O2 339.10 found 339.17; anal. calcd for C21H16N2: C, 78.09; H, 4.17; N, 8.28; found: C, 77.78; H, 3.97; N, 7.96.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 136–138 °C; (Lit: 138);18a IR (KBr): 3038, 1546, 1488, 1306, 1196 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.47 (s, 1H), 8.65 (s, 1H), 8.37–8.34 (dd, J = 10.8 Hz, 2 Hz, 1H), 8.21–8.13 (m, 2H), 8.03–7.99 (m, 2H), 7.90 (t, J = 3.6 Hz, 1H), 7.82–7.73 (m, 2H), 7.54–7.50 (m, 2H); LCMS (ESI) [M + H]+ calculated C18H12N2 257.10 found 257.17.
05 v/v); MP 136–138 °C; (Lit: 138);18a IR (KBr): 3038, 1546, 1488, 1306, 1196 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.47 (s, 1H), 8.65 (s, 1H), 8.37–8.34 (dd, J = 10.8 Hz, 2 Hz, 1H), 8.21–8.13 (m, 2H), 8.03–7.99 (m, 2H), 7.90 (t, J = 3.6 Hz, 1H), 7.82–7.73 (m, 2H), 7.54–7.50 (m, 2H); LCMS (ESI) [M + H]+ calculated C18H12N2 257.10 found 257.17.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 92–94 °C; (Lit: 84–86);18f IR (KBr): 3054, 1612, 1542, 1308 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.33 (s, 1H), 8.19–8.13 (m, 2H), 8.04 (s, 1H), 7.97 (d, J = 8 Hz, 1H), 7.98–7.75 (m, 2H), 7.47 (t, J = 8 Hz, 1H), 7.36 (d, J = 8.4 Hz, 1H), 2.51 (s, 3H); LCMS (ESI) [M + H]+ calculated C15H12N2 221.10 found 221.17.
05 v/v); MP 92–94 °C; (Lit: 84–86);18f IR (KBr): 3054, 1612, 1542, 1308 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.33 (s, 1H), 8.19–8.13 (m, 2H), 8.04 (s, 1H), 7.97 (d, J = 8 Hz, 1H), 7.98–7.75 (m, 2H), 7.47 (t, J = 8 Hz, 1H), 7.36 (d, J = 8.4 Hz, 1H), 2.51 (s, 3H); LCMS (ESI) [M + H]+ calculated C15H12N2 221.10 found 221.17.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 96–98 °C; (Lit: 87–88);18h IR (KBr): 1604, 1539, 1442, 1186, 1031 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.32 (s, 1H), 8.18–8.11 (m, 2H), 7.81–7.73 (m, 4H), 7.47 (t, J = 8 Hz, 1H), 7.07 (dd, J = 10.4 Hz, 2 Hz, 1H), 3.94 (s, 3H); LCMS (ESI) [M + H]+ calculated C15H12N2O 237.09 found 237.15.
05 v/v); MP 96–98 °C; (Lit: 87–88);18h IR (KBr): 1604, 1539, 1442, 1186, 1031 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.32 (s, 1H), 8.18–8.11 (m, 2H), 7.81–7.73 (m, 4H), 7.47 (t, J = 8 Hz, 1H), 7.07 (dd, J = 10.4 Hz, 2 Hz, 1H), 3.94 (s, 3H); LCMS (ESI) [M + H]+ calculated C15H12N2O 237.09 found 237.15.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 136–138 °C; (Lit: 134–137);18a IR (KBr): 1588, 1536, 1480, 1122, 1068, 1043 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.29 (s, 1H), 8.15–8.07 (m, 4H), 7.81–7.73 (m, 2H), 7.69 (d, J = 8.4 Hz, 2H); LCMS (ESI) [M + H]+ calculated C14H10BrN2O 285.00 found 284.79.
05 v/v); MP 136–138 °C; (Lit: 134–137);18a IR (KBr): 1588, 1536, 1480, 1122, 1068, 1043 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.29 (s, 1H), 8.15–8.07 (m, 4H), 7.81–7.73 (m, 2H), 7.69 (d, J = 8.4 Hz, 2H); LCMS (ESI) [M + H]+ calculated C14H10BrN2O 285.00 found 284.79.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 186–188 °C; (Lit: 187–189);18a IR (KBr): 1540, 1474, 1308, 1146, 1030 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.31 (s, 1H), 8.43 (s, 1H), 8.09 (t, J = 6 Hz, 2H), 7.80 (d, J = 7.2 Hz, 1H), 7.80–7.72 (m, 2H); LCMS (ESI) [M + H]+ calculated C14H9Cl2N2 275.01 found 275.07.
05 v/v); MP 186–188 °C; (Lit: 187–189);18a IR (KBr): 1540, 1474, 1308, 1146, 1030 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.31 (s, 1H), 8.43 (s, 1H), 8.09 (t, J = 6 Hz, 2H), 7.80 (d, J = 7.2 Hz, 1H), 7.80–7.72 (m, 2H); LCMS (ESI) [M + H]+ calculated C14H9Cl2N2 275.01 found 275.07.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 191–193 °C; (Lit: 190–192);18a IR (KBr): 3056, 1529, 1352, 1096 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.11 (s, 1H), 8.40–8.37 (m, 4H), 8.21–7.84 (m, 2H), 7.83 (d, J = 8 Hz, 2H); LCMS (ESI) [M + H]+ calculated C14H9N3O2 252.07 found 252.13.
05 v/v); MP 191–193 °C; (Lit: 190–192);18a IR (KBr): 3056, 1529, 1352, 1096 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.11 (s, 1H), 8.40–8.37 (m, 4H), 8.21–7.84 (m, 2H), 7.83 (d, J = 8 Hz, 2H); LCMS (ESI) [M + H]+ calculated C14H9N3O2 252.07 found 252.13.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 114–116 °C; (Lit: 193–195); IR (KBr): 3058, 2230, 1548, 1480, 1435, 1322, 1036; 1H NMR (CDCl3, 400 MHz): δ = 9.36 (s, 1H), 8.35 (d, J = 8.8 Hz, 2H), 8.19–8.15 (m, 2H), 7.88–7.80 (m, 4H); 13C NMR (CDCl3, 100 MHz): δ = 143.0, 142.6, 142.3, 141.7, 140.6, 133.0, 131.8, 130.5, 129.6, 129.1, 128.2, 127.7, 118.8, 113.8; LCMS (ESI) [M + H]+ calculated C15H9N3 232.08 found 232.14. Anal. calcd for C15H9N3: C, 77.91; H, 3.92; N, 18.17; found: C, 77.61, H, 3.56, N; 17.98.
05 v/v); MP 114–116 °C; (Lit: 193–195); IR (KBr): 3058, 2230, 1548, 1480, 1435, 1322, 1036; 1H NMR (CDCl3, 400 MHz): δ = 9.36 (s, 1H), 8.35 (d, J = 8.8 Hz, 2H), 8.19–8.15 (m, 2H), 7.88–7.80 (m, 4H); 13C NMR (CDCl3, 100 MHz): δ = 143.0, 142.6, 142.3, 141.7, 140.6, 133.0, 131.8, 130.5, 129.6, 129.1, 128.2, 127.7, 118.8, 113.8; LCMS (ESI) [M + H]+ calculated C15H9N3 232.08 found 232.14. Anal. calcd for C15H9N3: C, 77.91; H, 3.92; N, 18.17; found: C, 77.61, H, 3.56, N; 17.98.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 142–143 °C; (Lit: 142–143);18i IR (KBr): 3036, 1545, 1488, 1315, 1120, 645, 554 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.33 (s, 1H), 8.31 (d, J = 8.4 Hz, 2H), 8.12–8.10 (m, 2H), 7.88–7.76 (m, 4H); LCMS (ESI) [M + H]+ calculated C15H10N2F3 275.07 found 275.14.
05 v/v); MP 142–143 °C; (Lit: 142–143);18i IR (KBr): 3036, 1545, 1488, 1315, 1120, 645, 554 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.33 (s, 1H), 8.31 (d, J = 8.4 Hz, 2H), 8.12–8.10 (m, 2H), 7.88–7.76 (m, 4H); LCMS (ESI) [M + H]+ calculated C15H10N2F3 275.07 found 275.14.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 110–112 °C; (Lit: 114–115);18j IR (KBr): 3052, 1612, 1588, 1542, 1491, 1370, 1025, 1312; 1H NMR (CDCl3, 400 MHz): δ = 9.35 (s, 1H), 9.34 (s, 1H), 8.77 (dd, J = 6 Hz, 1.2 Hz, 1H), 8.54 (dt, J = 12.4 Hz, 2 Hz, 1H), 8.20–8.12 (m, 2H), 7.85–7.77 (m, 2H), 7.53–7.50 (m, 1H); LCMS (ESI) [M + H]+ calculated C13H9N3 208.08 found 208.14.
05 v/v); MP 110–112 °C; (Lit: 114–115);18j IR (KBr): 3052, 1612, 1588, 1542, 1491, 1370, 1025, 1312; 1H NMR (CDCl3, 400 MHz): δ = 9.35 (s, 1H), 9.34 (s, 1H), 8.77 (dd, J = 6 Hz, 1.2 Hz, 1H), 8.54 (dt, J = 12.4 Hz, 2 Hz, 1H), 8.20–8.12 (m, 2H), 7.85–7.77 (m, 2H), 7.53–7.50 (m, 1H); LCMS (ESI) [M + H]+ calculated C13H9N3 208.08 found 208.14.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 116–118 °C; (Lit: 116–117);18a IR (KBr): 3120, 3056, 1546, 1424, 1328, 1052 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.16 (s, 1H), 8.08–7.87 (m, 2H), 7.78–7.68 (m, 3H), 7.58 (d, J = 4 Hz, 1H), 7.56 (dd, J = 6.4 Hz, 1.6 Hz, 1H); LCMS (ESI) [M + H]+ calculated C12H9N2S 213.04 found 213.10.
05 v/v); MP 116–118 °C; (Lit: 116–117);18a IR (KBr): 3120, 3056, 1546, 1424, 1328, 1052 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.16 (s, 1H), 8.08–7.87 (m, 2H), 7.78–7.68 (m, 3H), 7.58 (d, J = 4 Hz, 1H), 7.56 (dd, J = 6.4 Hz, 1.6 Hz, 1H); LCMS (ESI) [M + H]+ calculated C12H9N2S 213.04 found 213.10.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 131–133 °C; (Lit: 131–136);18a IR (KBr): 3134, 3118, 1608, 1548, 1444, 1296, 1225, 1126, 1080 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.25 (s, 1H), 8.11–8.06 (m, 2H), 7.78–7.68 (m, 3H), 7.31 (d, J = 4 Hz, 1H), 6.63 (dd, J = 4.8 Hz, 1.2 Hz, 1H); LCMS (ESI) [M + H]+ calculated C12H8N2O 197.06 found 197.12.
05 v/v); MP 131–133 °C; (Lit: 131–136);18a IR (KBr): 3134, 3118, 1608, 1548, 1444, 1296, 1225, 1126, 1080 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.25 (s, 1H), 8.11–8.06 (m, 2H), 7.78–7.68 (m, 3H), 7.31 (d, J = 4 Hz, 1H), 6.63 (dd, J = 4.8 Hz, 1.2 Hz, 1H); LCMS (ESI) [M + H]+ calculated C12H8N2O 197.06 found 197.12.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); IR (KBr): 3066, 1608, 1548, 1490, 1318, 648 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.46 (s, 1H), 9.38 (s, 1H), 8.83 (s, 1H), 8.57–8.56 (t, J = 3.6 Hz, 1H), 8.36 (t, J = 2 Hz, 1H), 8.08–8.03 (m, 1H), 7.94–7.88 (m, 1H), 7.58–7.54 (m, 1H) LCMS (ESI) [M + H]+ calculated 285.99 found 286.05.
05 v/v); IR (KBr): 3066, 1608, 1548, 1490, 1318, 648 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.46 (s, 1H), 9.38 (s, 1H), 8.83 (s, 1H), 8.57–8.56 (t, J = 3.6 Hz, 1H), 8.36 (t, J = 2 Hz, 1H), 8.08–8.03 (m, 1H), 7.94–7.88 (m, 1H), 7.58–7.54 (m, 1H) LCMS (ESI) [M + H]+ calculated 285.99 found 286.05.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 132–134 °C; (130–133);18a IR (KBr): 3060, 1534, 1486, 1420, 1317, 1054 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.38 (s, 1H), 8.33 (d, J = 8 Hz, 2H), 8.19–8.13 (m, 2H), 7.95 (t, J = 6.4 Hz, 2H), 7.83–7.75 (m, 2H), 7.69 (t, J = 7.6 Hz, 1H), 7.47–7.40 (m, 2H); 13C NMR (CDCl3, 100 MHz): δ = 150.99, 142.96, 142.36, 141.81, 141.57, 14.50, 140.67, 136.70, 132.19, 130.72, 130.47, 130.01, 129.63, 129.04, 128.24, 127.90, 126.43, 125.83, 121.04, 119.17; LCMS (ESI) [M + H]+ calculated C20H14N2 283.11 found 283.17.
05 v/v); MP 132–134 °C; (130–133);18a IR (KBr): 3060, 1534, 1486, 1420, 1317, 1054 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.38 (s, 1H), 8.33 (d, J = 8 Hz, 2H), 8.19–8.13 (m, 2H), 7.95 (t, J = 6.4 Hz, 2H), 7.83–7.75 (m, 2H), 7.69 (t, J = 7.6 Hz, 1H), 7.47–7.40 (m, 2H); 13C NMR (CDCl3, 100 MHz): δ = 150.99, 142.96, 142.36, 141.81, 141.57, 14.50, 140.67, 136.70, 132.19, 130.72, 130.47, 130.01, 129.63, 129.04, 128.24, 127.90, 126.43, 125.83, 121.04, 119.17; LCMS (ESI) [M + H]+ calculated C20H14N2 283.11 found 283.17.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 128–130 °C; IR (KBr): 3065, 2910, 1560, 1480, 1415, 1312, 1052 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.39 (s, 1H), 8.26 (dd, J = 2 Hz, 8.4 Hz, 2H), 8.19–8.13 (m, 2H), 7.83–7.75 (m, 2H), 7.55 (d, J = 8 Hz, 2H), 7.32–7.29 (m, 4H), 2.34 (s, 3H); 13C NMR (CDCl3, 100 MHz): δ = 151.7, 144.0, 143.3, 141.5, 141.0, 135.3, 135.2, 130.5, 130.1, 129.6, 129.5, 129.1, 127.6, 127.3, 125.9, 20.5; anal. calcd for C21H16N2: C, 85.11; H, 5.44; N, 9.45. Found: C, 84.90; H, 5.06; N, 9.38.
05 v/v); MP 128–130 °C; IR (KBr): 3065, 2910, 1560, 1480, 1415, 1312, 1052 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.39 (s, 1H), 8.26 (dd, J = 2 Hz, 8.4 Hz, 2H), 8.19–8.13 (m, 2H), 7.83–7.75 (m, 2H), 7.55 (d, J = 8 Hz, 2H), 7.32–7.29 (m, 4H), 2.34 (s, 3H); 13C NMR (CDCl3, 100 MHz): δ = 151.7, 144.0, 143.3, 141.5, 141.0, 135.3, 135.2, 130.5, 130.1, 129.6, 129.5, 129.1, 127.6, 127.3, 125.9, 20.5; anal. calcd for C21H16N2: C, 85.11; H, 5.44; N, 9.45. Found: C, 84.90; H, 5.06; N, 9.38.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 134–136 °C; IR (KBr): 3058, 2858, 1528, 1482, 1418, 1145, 1315, 1052, 686 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.38 (s, 1H), 8.26 (d, J = 8.4 Hz, 2H), 8.19–8.12 (m, 2H), 7.82–7.77 (m, 2H), 7.76–7.72 (m, 2H), 7.13 (dd, J = 2.8 Hz, 8.8 Hz, 1H), 7.07–6.93 (m, 2H), 3.8 (s, 3H); 13C NMR (CDCl3, 100 MHz): δ = 158.3, 155.9, 152.7, 151.5, 143.3, 142.3, 141.5, 139.4, 135.6, 130.3, 130.1, 129.6, 129.5, 129.1, 127.2, 56.2; anal. calcd for C21H16N2: C, 76.35; H, 4.58; N, 8.48; found: C, 75.92, H, 4.66, N, 8.38.
05 v/v); MP 134–136 °C; IR (KBr): 3058, 2858, 1528, 1482, 1418, 1145, 1315, 1052, 686 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.38 (s, 1H), 8.26 (d, J = 8.4 Hz, 2H), 8.19–8.12 (m, 2H), 7.82–7.77 (m, 2H), 7.76–7.72 (m, 2H), 7.13 (dd, J = 2.8 Hz, 8.8 Hz, 1H), 7.07–6.93 (m, 2H), 3.8 (s, 3H); 13C NMR (CDCl3, 100 MHz): δ = 158.3, 155.9, 152.7, 151.5, 143.3, 142.3, 141.5, 139.4, 135.6, 130.3, 130.1, 129.6, 129.5, 129.1, 127.2, 56.2; anal. calcd for C21H16N2: C, 76.35; H, 4.58; N, 8.48; found: C, 75.92, H, 4.66, N, 8.38.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 136–138 °C; IR (KBr): 3052, 2890, 1532, 1483, 1427, 1319, 1055, 812 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.37 (s, 1H), 8.18 (d, J = 1.6 Hz, 1H), 8.15 (d, J = 6.8 Hz, 2H), 8.14–8.05 (m, 2H), 8.04 (d, J = 8 Hz, 2H), 7.81–7.31 (m, 3H), 7.05 (dd, J = 11.6 Hz, 2.8 Hz, 1H), 3.99 (q, J = 16 Hz, 7.2 Hz, 2H), 1.58 (t, J = 7.2 Hz, 3H); 13C NMR (CDCl3, 100 MHz); 157.47, 152.02, 141.81, 141.57, 140.67, 136.50, 132.29, 130.89, 130.47, 129.04, 128.24, 127.90, 126.53, 125.73, 121.04, 114.27, 110.08, 68.64, 15.47; anal. calcd for C22H17FN2O: C, 76.73; H, 4.98; N, 8.13; found: C, 76.33, H, 4.88, N, 7.98.
05 v/v); MP 136–138 °C; IR (KBr): 3052, 2890, 1532, 1483, 1427, 1319, 1055, 812 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.37 (s, 1H), 8.18 (d, J = 1.6 Hz, 1H), 8.15 (d, J = 6.8 Hz, 2H), 8.14–8.05 (m, 2H), 8.04 (d, J = 8 Hz, 2H), 7.81–7.31 (m, 3H), 7.05 (dd, J = 11.6 Hz, 2.8 Hz, 1H), 3.99 (q, J = 16 Hz, 7.2 Hz, 2H), 1.58 (t, J = 7.2 Hz, 3H); 13C NMR (CDCl3, 100 MHz); 157.47, 152.02, 141.81, 141.57, 140.67, 136.50, 132.29, 130.89, 130.47, 129.04, 128.24, 127.90, 126.53, 125.73, 121.04, 114.27, 110.08, 68.64, 15.47; anal. calcd for C22H17FN2O: C, 76.73; H, 4.98; N, 8.13; found: C, 76.33, H, 4.88, N, 7.98.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 133–135 °C; IR (KBr): 3052, 1528, 1480, 1476, 1416, 1312, 1051, 748 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.37 (s, 1H), 8.27 (d, J = 12 Hz, 2H), 8.17–8.11 (m, 2H), 7.77 (m, 2H), 7.65 (d, J = 8 Hz, 2H), 7.50 (d, J = 7.6 Hz, 1H), 7.40–7.29 (m, 3H); 13C NMR (CDCl3, 100 MHz): δ = 151.4, 143.2, 142.3, 141.6, 141.3, 139.7, 136.0, 132.5, 131.2, 130.2, 130.0, 129.6, 129.5, 129.1, 128.9, 127.2, 126.9; anal. calcd for C20H13ClN2: C, 75.83; H, 4.14; N, 8.84 found: C, 74.82, H, 4.08, N, 8.92.
05 v/v); MP 133–135 °C; IR (KBr): 3052, 1528, 1480, 1476, 1416, 1312, 1051, 748 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.37 (s, 1H), 8.27 (d, J = 12 Hz, 2H), 8.17–8.11 (m, 2H), 7.77 (m, 2H), 7.65 (d, J = 8 Hz, 2H), 7.50 (d, J = 7.6 Hz, 1H), 7.40–7.29 (m, 3H); 13C NMR (CDCl3, 100 MHz): δ = 151.4, 143.2, 142.3, 141.6, 141.3, 139.7, 136.0, 132.5, 131.2, 130.2, 130.0, 129.6, 129.5, 129.1, 128.9, 127.2, 126.9; anal. calcd for C20H13ClN2: C, 75.83; H, 4.14; N, 8.84 found: C, 74.82, H, 4.08, N, 8.92.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 141–143 °C; IR (KBr): 3060, 1532, 1484, 1416, 1316, 1050, 734, 688 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.37 (s, 1H), 8.26 (dd, J = 2 Hz, 8.8 Hz, 2H), 8.18–8.12 (m, 2H), 7.81–7.72 (m, 2H), 7.13 (dd, J = 2.8 Hz, 12 Hz, 1H), 7.06–7.02 (m, 1H), 6.94 (dd, J = 4 Hz, 13.2 Hz, 1H); 13C NMR (CDCl3, 100 MHz): δ = 158.3, 155.9, 152.7, 151.52, 143.3, 141.5, 139.4, 135.6, 130.3, 130.2, 129.6, 129.5, 129.1, 127.2; anal. calcd for C20H12ClFN2: C, 71.75; H, 3.61; N, 8.37; found: C, 71.14, H, 3.48, N, 8.32.
05 v/v); MP 141–143 °C; IR (KBr): 3060, 1532, 1484, 1416, 1316, 1050, 734, 688 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.37 (s, 1H), 8.26 (dd, J = 2 Hz, 8.8 Hz, 2H), 8.18–8.12 (m, 2H), 7.81–7.72 (m, 2H), 7.13 (dd, J = 2.8 Hz, 12 Hz, 1H), 7.06–7.02 (m, 1H), 6.94 (dd, J = 4 Hz, 13.2 Hz, 1H); 13C NMR (CDCl3, 100 MHz): δ = 158.3, 155.9, 152.7, 151.52, 143.3, 141.5, 139.4, 135.6, 130.3, 130.2, 129.6, 129.5, 129.1, 127.2; anal. calcd for C20H12ClFN2: C, 71.75; H, 3.61; N, 8.37; found: C, 71.14, H, 3.48, N, 8.32.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 137–139 °C; IR (KBr): 3062, 2886, 1530, 1488, 1414, 1317, 1112, 1060 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.39 (s, 1H), 8.34 (d, J = 8.8 Hz, 2H), 8.24 (t, J = 4 Hz, 1H), 8.21–8.14 (m, 2H), 7.99–7.95 (m, 2H), 7.83 (d, J = 8 Hz, 2H), 7.81–7.78 (m, 2H), 7.71 (t, J = 12 Hz, 1H), 7.48–7.40 (m, 2H), 3.14 (s, 3H); 13C NMR (CDCl3, 100 MHz): δ = 156.9, 151.0, 143.0, 142.3, 141.7, 140.6, 136.6, 132.2, 130.7, 129.0, 128.2, 127.9, 126.4, 125.8, 121.1, 44.5; anal. calcd for C21H16N2O2S: C, 69.98; H, 4.47; N, 7.77; found: C, 68.92, H, 4.40, N, 7.28.
05 v/v); MP 137–139 °C; IR (KBr): 3062, 2886, 1530, 1488, 1414, 1317, 1112, 1060 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.39 (s, 1H), 8.34 (d, J = 8.8 Hz, 2H), 8.24 (t, J = 4 Hz, 1H), 8.21–8.14 (m, 2H), 7.99–7.95 (m, 2H), 7.83 (d, J = 8 Hz, 2H), 7.81–7.78 (m, 2H), 7.71 (t, J = 12 Hz, 1H), 7.48–7.40 (m, 2H), 3.14 (s, 3H); 13C NMR (CDCl3, 100 MHz): δ = 156.9, 151.0, 143.0, 142.3, 141.7, 140.6, 136.6, 132.2, 130.7, 129.0, 128.2, 127.9, 126.4, 125.8, 121.1, 44.5; anal. calcd for C21H16N2O2S: C, 69.98; H, 4.47; N, 7.77; found: C, 68.92, H, 4.40, N, 7.28.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 129–131 °C; IR (KBr): 3056, 2228, 1531, 1482, 1417, 1319, 1052 cm−1; 1H NMR (CDCl3, 400 MHz); δ = 9.57 (s, 1H), 9.37 (s, 1H), 9.21 (s, 1H), 8.50 (s, 1H), 8.32 (d, J = 8.4 Hz, 2H), 8.18–8.12 (m, 2H), 7.79 (d, J = 8.4 Hz, 2H); 13C NMR (CDCl3, 100 MHz): δ = 164.6, 162.2, 150.9, 145.9, 145.9, 145.84, 142.3, 141.6, 139.7, 139.6, 138.3, 136.5, 134.0, 130.4, 129.6, 128.2, 115.6, 109.8, 109.4; anal. calcd for C21H13N2: C, 77.91; H, 3.92; N, 18.17; found: C, 76.88, H, 3.94, N, 18.12.
05 v/v); MP 129–131 °C; IR (KBr): 3056, 2228, 1531, 1482, 1417, 1319, 1052 cm−1; 1H NMR (CDCl3, 400 MHz); δ = 9.57 (s, 1H), 9.37 (s, 1H), 9.21 (s, 1H), 8.50 (s, 1H), 8.32 (d, J = 8.4 Hz, 2H), 8.18–8.12 (m, 2H), 7.79 (d, J = 8.4 Hz, 2H); 13C NMR (CDCl3, 100 MHz): δ = 164.6, 162.2, 150.9, 145.9, 145.9, 145.84, 142.3, 141.6, 139.7, 139.6, 138.3, 136.5, 134.0, 130.4, 129.6, 128.2, 115.6, 109.8, 109.4; anal. calcd for C21H13N2: C, 77.91; H, 3.92; N, 18.17; found: C, 76.88, H, 3.94, N, 18.12.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 138–140 °C; IR (KBr): 3056, 1529, 1486, 1418, 1315, 1062 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.39 (s, 1H), 8.7 (d, J = 7.2 Hz, 1H), 8.51 (d, J = 2.4 Hz, 1H), 8.36 (dt, J = 2 Hz, 8.8 Hz, 2H), 8.20–8.14 (m, 2H), 7.83 (dd, J = 1.6 Hz, 8.4 Hz, 1H), 7.82–7.76 (m, 4H), 7.68 (dt, J = 2.8 Hz, 12 Hz, 1H); 13C NMR (CDCl3, 100 MHz): δ = 150.8, 144.0, 143.0, 142.32, 141.7, 138.0, 137.0, 130.4, 129.6, 129.1, 128.3, 127.9, 121.1, 120.9; anal. calcd for C19H13N3: C, 80.54; H, 4.62; N, 14.83; found: C, 80.48, H, 4.06, N, 14.44.
05 v/v); MP 138–140 °C; IR (KBr): 3056, 1529, 1486, 1418, 1315, 1062 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.39 (s, 1H), 8.7 (d, J = 7.2 Hz, 1H), 8.51 (d, J = 2.4 Hz, 1H), 8.36 (dt, J = 2 Hz, 8.8 Hz, 2H), 8.20–8.14 (m, 2H), 7.83 (dd, J = 1.6 Hz, 8.4 Hz, 1H), 7.82–7.76 (m, 4H), 7.68 (dt, J = 2.8 Hz, 12 Hz, 1H); 13C NMR (CDCl3, 100 MHz): δ = 150.8, 144.0, 143.0, 142.32, 141.7, 138.0, 137.0, 130.4, 129.6, 129.1, 128.3, 127.9, 121.1, 120.9; anal. calcd for C19H13N3: C, 80.54; H, 4.62; N, 14.83; found: C, 80.48, H, 4.06, N, 14.44.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 139–141 °C; IR (KBr): 3056, 1528, 1489, 1413, 1316, 1048, 694 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.38 (s, 1H), 8.52 (d, J = 2 Hz, 1H), 8.34 (d, J = 8.4 Hz, 2H), 8.20–8.13 (m, 2H), 8.09–8.05 (m, 1H), 7.84–7.78 (m, 2H), 7.76 (d, J = 8.4 Hz, 2H), 7.07 (dd, J = 3.2 Hz, 12 Hz, 1H); 13C NMR (CDCl3, 100 MHz); δ = 150.9, 145.9, 145.8, 143.0, 141.6, 139.3, 136.5, 134.0, 130.5, 129.6, 129.1, 128.1, 128.2, 127.7, 109.8, 109.5; anal. calcd for C19H12FN3: C, 75.74; H, 4.01; N, 13.95; found: C, 75.24, H, 3.94, N, 13.68.
05 v/v); MP 139–141 °C; IR (KBr): 3056, 1528, 1489, 1413, 1316, 1048, 694 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.38 (s, 1H), 8.52 (d, J = 2 Hz, 1H), 8.34 (d, J = 8.4 Hz, 2H), 8.20–8.13 (m, 2H), 8.09–8.05 (m, 1H), 7.84–7.78 (m, 2H), 7.76 (d, J = 8.4 Hz, 2H), 7.07 (dd, J = 3.2 Hz, 12 Hz, 1H); 13C NMR (CDCl3, 100 MHz); δ = 150.9, 145.9, 145.8, 143.0, 141.6, 139.3, 136.5, 134.0, 130.5, 129.6, 129.1, 128.1, 128.2, 127.7, 109.8, 109.5; anal. calcd for C19H12FN3: C, 75.74; H, 4.01; N, 13.95; found: C, 75.24, H, 3.94, N, 13.68.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 143–146 °C; IR (KBr): 3048, 1532, 1482, 1428, 1323, 1052, 702 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.35 (s, 1H), 8.54 (d, J = 8 Hz, 2H), 8.41 (s, 1H), 8.31–8.24 (m, 2H), 8.20–8.12 (m, 1H), 7.88–7.7 (m, 2H), 7.74 (d, J = 8.8 Hz, 2H), 7.16 (dd, J1 = 12.4 Hz, J2 = 3.2 Hz, 1H); 13C NMR (CDCl3, 100 MHz); δ = 164.3, 153.5, 147.1, 145.8, 143.0, 142.8, 141.7, 138.50, 136.5, 134.0, 130.5, 129.8, 129.6, 129.1, 128.2, 127.7, 109.8, 109.3; anal. calcd for C19H12FN3: C, 75.74, H, 4.01, N, 13.95; found: C, 75.44, H, 3.96, N, 13.56.
05 v/v); MP 143–146 °C; IR (KBr): 3048, 1532, 1482, 1428, 1323, 1052, 702 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.35 (s, 1H), 8.54 (d, J = 8 Hz, 2H), 8.41 (s, 1H), 8.31–8.24 (m, 2H), 8.20–8.12 (m, 1H), 7.88–7.7 (m, 2H), 7.74 (d, J = 8.8 Hz, 2H), 7.16 (dd, J1 = 12.4 Hz, J2 = 3.2 Hz, 1H); 13C NMR (CDCl3, 100 MHz); δ = 164.3, 153.5, 147.1, 145.8, 143.0, 142.8, 141.7, 138.50, 136.5, 134.0, 130.5, 129.8, 129.6, 129.1, 128.2, 127.7, 109.8, 109.3; anal. calcd for C19H12FN3: C, 75.74, H, 4.01, N, 13.95; found: C, 75.44, H, 3.96, N, 13.56.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 146–148 °C; (Lit: 147–148);18a IR (KBr): 3008, 2964, 2842, 1604, 1508, 1460, 1392, 1346, 1286, 1240, 1172, 1024, 828, 764 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.14–7.91 (m, 2H), 7.6–7.37 (m, 3H), 7.34 (d, J = 8.4 Hz, 4H), 7.33 (d, J = 7.2 Hz, 4H), 3.82 (s, 6H); LCMS (ESI) [M + H]+ calculated C22H18N2O2 343.14 found 343.20.
05 v/v); MP 146–148 °C; (Lit: 147–148);18a IR (KBr): 3008, 2964, 2842, 1604, 1508, 1460, 1392, 1346, 1286, 1240, 1172, 1024, 828, 764 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.14–7.91 (m, 2H), 7.6–7.37 (m, 3H), 7.34 (d, J = 8.4 Hz, 4H), 7.33 (d, J = 7.2 Hz, 4H), 3.82 (s, 6H); LCMS (ESI) [M + H]+ calculated C22H18N2O2 343.14 found 343.20.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 54–56 °C; (Lit: 54–55);18b IR (KBr): 3122, 1510, 1461, 1318, 1054, 719 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.25–8.11 (m, 2H), 7.73–7.17 (m, 4H), 7.5–7.51 (m, 3H), 2.73 (s, 3H); LCMS (ESI) [M + H]+ calculated C15H12N2 221.10 found 221.17.
05 v/v); MP 54–56 °C; (Lit: 54–55);18b IR (KBr): 3122, 1510, 1461, 1318, 1054, 719 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.25–8.11 (m, 2H), 7.73–7.17 (m, 4H), 7.5–7.51 (m, 3H), 2.73 (s, 3H); LCMS (ESI) [M + H]+ calculated C15H12N2 221.10 found 221.17.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 30–32 °C; (Lit: 32–33);18c IR (KBr); 1498, 1364, 1202, 1128, 1028, 956 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.82 (s, 2H), 8.12 (d, J = 8 Hz, 2H), 7.76 (d, J = 8.8 Hz, 2H); LCMS (ESI) [M + H]+ calculated C8H6N2 131.05 found 131.10.
05 v/v); MP 30–32 °C; (Lit: 32–33);18c IR (KBr); 1498, 1364, 1202, 1128, 1028, 956 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.82 (s, 2H), 8.12 (d, J = 8 Hz, 2H), 7.76 (d, J = 8.8 Hz, 2H); LCMS (ESI) [M + H]+ calculated C8H6N2 131.05 found 131.10.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 135–137 °C; (Lit: 136–138);18a IR (KBr): 3058, 2926, 1724, 1588, 1490, 1472, 1440, 1398, 1342, 1087, 1056, 1012, 966, 848, 806, 760, 696 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.20–8.13 (m, 2H), 8.09–8.07 (m, 2H), 8.05–7.82 (m, 4H), 7.80–7.78 (m, 3H), 7.75 (d, J = 8.4 Hz, 2H); LCMS (ESI) [M + H]+ calculated C20H13Cl2N2 317.08 found 317.14.
05 v/v); MP 135–137 °C; (Lit: 136–138);18a IR (KBr): 3058, 2926, 1724, 1588, 1490, 1472, 1440, 1398, 1342, 1087, 1056, 1012, 966, 848, 806, 760, 696 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.20–8.13 (m, 2H), 8.09–8.07 (m, 2H), 8.05–7.82 (m, 4H), 7.80–7.78 (m, 3H), 7.75 (d, J = 8.4 Hz, 2H); LCMS (ESI) [M + H]+ calculated C20H13Cl2N2 317.08 found 317.14.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 122–124 °C; (Lit: 123–125);18a IR (KBr): 2930, 2833, 1603, 1508, 1455, 1417, 1340, 1298, 1172, 1023, 965, 828 cm−1; 1H NMR (400 MHz, CDCl3): δ = 7.82 (s, 2H), 7.60 (d, J = 8.4 Hz, 4H), 7.10 (d, J = 7.6 Hz, 4H), 3.92 (s, 6H), 2.42 (s, 6H); LCMS (ESI) [M + H]+ calculated C24H22Cl2N2O2 371.17 found 371.23.
05 v/v); MP 122–124 °C; (Lit: 123–125);18a IR (KBr): 2930, 2833, 1603, 1508, 1455, 1417, 1340, 1298, 1172, 1023, 965, 828 cm−1; 1H NMR (400 MHz, CDCl3): δ = 7.82 (s, 2H), 7.60 (d, J = 8.4 Hz, 4H), 7.10 (d, J = 7.6 Hz, 4H), 3.92 (s, 6H), 2.42 (s, 6H); LCMS (ESI) [M + H]+ calculated C24H22Cl2N2O2 371.17 found 371.23.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 132–134 °C; (Lit: 131–133);18k IR (KBr): 3359, 1541, 1428, 1071, 835, 756 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.36 (d, J = 1.6 Hz, 1H), 8.03 (d, J = 8.4 Hz, 1H), 7.86 (dd, J = 2.4 Hz, 2.4 Hz, 1H), 7.68 (t, J = 1.6 Hz, 2H), 6.76–6.75 (m, 2H), 6.63–6.61 (m, 2H); LCMS (ESI) [M + H]+ calculated C16H9BrN2O2 341.98 found 341.06.
05 v/v); MP 132–134 °C; (Lit: 131–133);18k IR (KBr): 3359, 1541, 1428, 1071, 835, 756 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.36 (d, J = 1.6 Hz, 1H), 8.03 (d, J = 8.4 Hz, 1H), 7.86 (dd, J = 2.4 Hz, 2.4 Hz, 1H), 7.68 (t, J = 1.6 Hz, 2H), 6.76–6.75 (m, 2H), 6.63–6.61 (m, 2H); LCMS (ESI) [M + H]+ calculated C16H9BrN2O2 341.98 found 341.06.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 158–160 °C; (Lit: 150–152);18f IR (KBr): 3418, 1466, 1344, 1216, 702 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.24 (d, J = 8.4 Hz, 1H), 7.83 (d, J = 8.8 Hz, 1H), 7.80–7.28 (m, 4H), 7.14–7.01 (m, 6H); LCMS (ESI) [M + H]+ calculated C20H12Cl2N2FCl 335.07 found 335.14.
05 v/v); MP 158–160 °C; (Lit: 150–152);18f IR (KBr): 3418, 1466, 1344, 1216, 702 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 8.24 (d, J = 8.4 Hz, 1H), 7.83 (d, J = 8.8 Hz, 1H), 7.80–7.28 (m, 4H), 7.14–7.01 (m, 6H); LCMS (ESI) [M + H]+ calculated C20H12Cl2N2FCl 335.07 found 335.14.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 183–185 °C; (Lit: 184–186);18a IR (KBr): 3080, 3057, 1612, 1518, 1398, 1340, 1054, 1023, 810, 767, 698 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.08 (d, J = 2.4 Hz, 1H), 8.54 (dd, J = 11.6 Hz, 2.4 Hz, 1H), 8.30 (d, J = 9.2 Hz, 1H), 7.58–7.54 (m, 4H), 7.43–7.36 (m, 6H); LCMS (ESI) [M + H]+ calculated C20H13Cl2N3O2 328.10 found 328.17.
05 v/v); MP 183–185 °C; (Lit: 184–186);18a IR (KBr): 3080, 3057, 1612, 1518, 1398, 1340, 1054, 1023, 810, 767, 698 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 9.08 (d, J = 2.4 Hz, 1H), 8.54 (dd, J = 11.6 Hz, 2.4 Hz, 1H), 8.30 (d, J = 9.2 Hz, 1H), 7.58–7.54 (m, 4H), 7.43–7.36 (m, 6H); LCMS (ESI) [M + H]+ calculated C20H13Cl2N3O2 328.10 found 328.17.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05 v/v); MP 171–176 °C; (Lit: 170–172);18d IR (KBr): 3048, 2936, 1612 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 7.55–7.49 (m, 4H), 7.28–7.20 (m, 6H), 2.84 (t, J = 9.2 Hz, 2H), 2.50 (d, J = 13.6 Hz, 2H), 1.89 (d, J = 9.2 Hz, 2H), 1.64–1.62 (m, 2H), 1.47–1.39 (m, 2H); LCMS (ESI) [M + H]+ calculated C20H20N2 289.16 found 289.22.
05 v/v); MP 171–176 °C; (Lit: 170–172);18d IR (KBr): 3048, 2936, 1612 cm−1; 1H NMR (CDCl3, 400 MHz): δ = 7.55–7.49 (m, 4H), 7.28–7.20 (m, 6H), 2.84 (t, J = 9.2 Hz, 2H), 2.50 (d, J = 13.6 Hz, 2H), 1.89 (d, J = 9.2 Hz, 2H), 1.64–1.62 (m, 2H), 1.47–1.39 (m, 2H); LCMS (ESI) [M + H]+ calculated C20H20N2 289.16 found 289.22.


