Carbon-based dots co-doped with nitrogen and sulfur for Cr(VI) sensing and bioimaging†
Yang Liu,
Xiaojuan Gong,
Yifang Gao,
Shengmei Song,
Xin Wu,
Shaomin Shuang and
Chuan Dong*
Institute of Environmental Science, and School of Chemistry and Chemical Engineering, Shanxi University, Taiyuan 030006, China. E-mail: dc@sxu.edu.cn; Fax: +86-351-7018613; Tel: +86-351-7018613
First published on 11th March 2016
Abstract
Less toxic elements nitrogen and sulfur co-doped carbon-based dots (NSCDs) have been prepared by microwave-assisted pyrolysis of citric acid and N-acetyl-L-cysteine as the carbon source and N,S-dopant, respectively. The structure and optical properties of NSCDs are characterized by transmission electron microscopy, X-ray photoelectron spectroscopy, elemental analysis, Fourier transform infrared spectroscopy, UV-vis absorption, and photoluminescence spectroscopy. The mechanism for the formation of NSCDs is also discussed. The as-prepared NSCDs show small size distribution and excellent dispersibility. Their strong blue fluorescence is observed when the excitation wavelength is between 260 nm and 380 nm. Moreover, they exhibit high tolerance to various external conditions including external cations, pH values, and continuous UV excitation. More strikingly, as the emission of NSCDs is efficiently quenched by Cr(VI), the as-prepared NSCDs are employed as a highly sensitive and selective probe for Cr(VI) detection. The linear range is 0.5–125 μM Cr(VI) with the detection limit 20 nM. The as-synthesized NSCDs have been successfully applied for Cr(VI) sensing and cell imaging.
Introduction
Quantum dots (QDs) are nanoparticles with size-dependent optical and electronic properties that have been proposed for various applications, such as energy-efficient displays and lighting, photovoltaic devices, and biological markers. Compared with other fluorescent materials, QDs offer many promising advantages, such as bright fluorescence, high photostability, and resistance to metabolic degradation in bioapplications.1 However, the toxicity of most high-performance QDs is of concern due to their compositions of metal elements, such as lead, cadmium, gold, silver, and silicon. In this regard, carbon nanomaterials have fascinating optical properties and already show encouraging performance in bioimaging and biosensing. Fluorescent carbon nanomaterials appear in different forms such as fullerene,2 carbon nanotubes,3 nanodiamonds,4 fluorescent graphene,5 and a rising star: carbon dots (CDs)6,7 which were accidently discovered as a by-product during the electrophoretic purification of single-wall carbon nanotubes fabricated by arc-discharge methods.8 Among all these materials, CDs containing heteroatoms have generated unique excitement and been actively pursued as they are considered the most promising candidate to complement carbon in materials applications because of their intrinsic properties such as electronic properties, surface and local chemical reactivity.9 Most CDs reported so far are hydrophilic in nature and appear to be a promising alternative to semiconductor QDs in the fields of biological labeling,10,11 chemosensing and biosensing,12 and biomedicine.10Up to now, extensive efforts have been devoted to synthesize CDs, and the synthesis methods can be generally classified into top-down and bottom-up approaches.13 “Top-down” methods involve breaking down larger mass carbon materials into individual nanoparticles (NPs) through arc-discharge single-walled carbon nanotubes,8 laser ablation of graphite,14 electrochemical oxidation of graphite and multi-walled carbon nanotubes,15 commercially activated carbon and lampblack,16 carbonizing polymerized resols on silica spheres,17 and chemical oxidation of oxide graphene.18 Complicated synthesis conditions, time and energy consumption, expensive starting materials and/or instruments, and difficulty in preparation of large quantities and high quality of CDs are often problematic for the “top-down” approaches.19 By contrast, “bottom-up” approaches include thermal decomposition, combustion and dehydration of suitable molecular precursors. Generally, these methods involve intricate processes and severe synthetic conditions, and the photoluminescence (PL) quantum yield (Φs) of the obtained CDs is very low with only a few exceptions. Undoubtedly, the preparation of CDs should be carried out toward facile and environmentally benign strategies. Recently, microwave pyrolysis of carbohydrates solution is a rising technique due to its simple experimental setup, easy control of the reaction, and low consumption.7,20 However, to achieve highly luminescent CDs, surface-passivation reagents are usually required.21 Microwave pyrolysis of different carbohydrates either in the absence or presence of any surface passivating agent and dehydration of carbohydrates with subsequent surface passivation have been reported to produce CDs with sufficient Φs. In addition, many efforts have been focused on synthesis of N and/or S doped CDs, owing to their good performances in fluorescence.22 Specifically, heteroatoms doped CDs remain almost all the advantages of blank CDs, and efficiently avoid their self-quenching and relatively low PL Φs.22 Gong et al. synthesized N-CDs for label-free detection of Fe3+ and cell imaging.23 Kwon et al. prepared S-incorporated CDs, which exhibited enhanced absorbance and PL intensity in the long-wavelength regime.24 Guo et al. fabricated N,S-doped graphene, which demonstrated remarkably improved electrocatalytic activity and electrochemical sensing performances.25 Other N,S-co doped CDs have been reported,26–28 but they existed some defects, such as high reaction temperature, long reaction time, and low Φs. Our group constructed NSCDs by microwave-assisted pyrolysis of citric acid (CA) and N-acetyl-L-cysteine (NAC) for Cr(VI) sensing and cellular imaging.
In the present work, we report a facile, economical, and effective approach to synthesize strongly fluorescent nitrogen and sulfur co-doped carbon dots (NSCDs) on a large scale by one-step microwave-assisted pyrolysis of CA and NAC as the precursors. CA rich in carbon can serve as an excellent carbon source. NAC comprising N and S atoms can function as dopants for NSCDs. The resulting NSCD possesses good water-solubility, and is non-toxic and cost-effective. The synthesis method is cost-effective and fast and can be completed within 8 min using a domestic microwave oven. Doping N and S into CDs greatly increases the Φs of CDs. The obtained NSCDs solution exhibits homogeneous phase without any noticeable precipitation at ambient conditions for six months, indicating their long-term colloidal stability. The as-prepared NSCDs display excellent stability under various external conditions including pH, ionic strength, and UV light. These NSCDs have been demonstrated as an effective fluorescent probe for sensitive and selective detection of Cr(VI) with a detection limit as low as 20 nM, which is much lower than other previous reported values was measured by GQDs,29,30 AgNPs,31 and CDs32 and is lower than the maximum allowable level (50 μg L−1) for Cr(VI) in drinking water permitted by the World Health Organization (WHO). Its potential applications in bioimaging and intracellular Cr(VI) monitoring have been explored. The results demonstrate that NSCDs can be applied for detecting Cr(VI) in biosystem and shows almost no interference from other metal ions. Our as-synthesized NSCDs could open up more analytical applications in bioimaging, biosensoring, and biomedicine.
Experimental
Materials
Citric acid (CA), N-acetyl-L-cysteine (NAC) and quinine sulfate were obtained from Aldrich (Milwaukee, WI, USA). Dimethyl sulfoxide (DMSO), Dulbecco's modified Eagle's medium (DMEM), fetal bovine serum (FBS), trypsin, ethylenediaminetetraacetic acid (EDTA), and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) were purchased from Solarbio (Beijing, China). Other reagents were from Beijing Chemical Reagents Company (Beijing, China). All reagents of analytical grade or above were used as received without further purification. All aqueous solutions were prepared with ultrapure water (≥18.25 MΩ cm) from a Milli-Q Plus System (Millipore, Bedford, MA, USA).Synthesis of NSCDs
The NSCDs was synthesized by a facile green route of microwave-assisted pyrolysis method. Briefly, 0.09 g CA and 0.15 g NAC were dissolved with 5 mL ultrapure water in a 50 mL beaker under ultrasonic stirring for 5 min to form a homogeneous solution. Then the reaction mixture was placed at the center of the rotation plate of a domestic microwave oven (600 W) and heated for 8 min. The mixture was cooled down to room temperature and 20 mL of ultrapure water was added. The solution was purified by a dialysis membrane tube with MWCO of 500–1000 Da (Spectrum Laboratories, Rancho Dominguez, CA, USA) in a 2 L ultrapure water every 24 h over a course of 3 days. Finally, a clear and yellow aqueous solution containing NSCDs was obtained and lyophilized to yield the dry NSCDs product.Characterisation
The transmission electron microscopic (TEM) images were acquired on a JEOL JEM-2100 transmission electron microscopy (Tokyo, Japan) with an accelerating voltage of 300 kV. The elemental analysis was conducted on an Elementar Analysen systemevario EL cube elemental analyser (Hanau, Germany). Analyses were performed in triplicate and the average values were obtained. The X-ray photoelectron spectra (XPS) were acquired on an AXIS ULTRA DLD X-ray photoelectron spectrometer (Kratos, Tokyo, Japan) with AlKα radiation operating at 1486.6 eV. Spectra were processed by Case XPS v.2.3.12 software using a peak-fitting routine with symmetrical Gaussian–Lorentzian functions. The Fourier transform infrared spectra (FTIR) were recorded on a Bruker Tensor II FTIR spectrometer (Bremen, German). Absorption and photoluminescence spectra were recorded using a UV-2450-PC spectrophotometer (Shimadzu, Japan) and Varian Cary Eclipse spectrofluorometer (Palo Alto, CA, USA), respectively.Nanosecond fluorescence lifetime experiments were performed using a FLS 920 time-correlated single-photon counting (TCSPC) system under right-angle sample geometry. An Edinburgh EPL 405 nm picosecond diode laser with a repetition rate of 2 MHz was used to excite the samples. The fluorescence was collected by a photomultiplier tube (Hamamatsu H5783p) connected to a Becker & Hickl SPC-130TCSPC board (Berlin, Germany). The time constant of the instrument response function was 50 ns.
Fluorescent sensors of Cr(VI) detection
The freeze-dried NSCDs powder was dissolved in a phosphate buffer saline (PBS comprising 134 mM NaCl, 2.7 mM KCl, 8.0 mM Na2HPO4, and 2.0 mM KH2PO4) solution (pH 7.4) with a concentration of 0.20 mg mL−1. Then, aqueous solutions containing twenty kinds of metal ions, K+, Na+, Ag+, Zn2+, Fe2+, Cd2+, Hg2+, Ba2+, Ca2+, Cu2+, Mg2+, Ni2+, Pb2+, Mn2+, Co2+, Fe3+, Al3+, Cr3+, Cr(VI), and Mn(VII), were prepared with a concentration of 10 mM. To evaluate the selectivity of NSCDs, 0.20 mL of the NSCDs of the above metal ion solution was mixed with 1.8 mL of the NSCDs, and then the PL spectra were measured for recording the fluorescence intensity. To evaluate the detection range of Cr(VI), 0.20 mL of the solutions containing different concentration of Cr(VI) was mixed with 1.8 mL of the NSCDs solution. The control sample was prepared by mixing 0.20 mL of ultrapure water with 1.80 mL of the NSCDs solution. All the samples were excited (λex) at 362 nm, and the intensities of fluorescence emission (λem) at 443 nm for each sample were recorded for comparison.MTT assay
For the cell cytotoxicity test, HepG2 cells were first plated on a Costar® 96-well cell culture cluster and cultured at 37 °C with 5.0% CO2 in air for 3 h to adhere cells onto the surface. The well without cells and treatment with NSCDs were taken as the zero sets. The medium was then changed with 200 μL of fresh DMEM supplemented with 10% FBS containing NSCDs and then the cells were allowed to grow for another 24 h and 48 h, respectively. At least six parallel samples were performed in each group. Cells not treated with NSCDs were taken as the controls. After adding 20 μL of 5.0 mg mL−1 MTT reagent into every well, the cells were further incubated for 4 h. The culture medium with MTT was removed and 150 μL of DMSO was added. The resulting mixture was shaken for ca. 10 min at room temperature. The optical density (OD) of the mixture was measured at 490 nm with a SunRise microplate reader (Tecan Austria GmbH, Grödig, Austria). The cell viability was estimated as: cell viability (%) = (ODTreated/ODControl) × 100%, where ODControl and ODTreated were obtained in the absence and presence of NSCDs, respectively.Multicolor cellular imaging
The human cervical carcinoma SiHa cells were cultured in DMEM supplemented with 10% FBS and the cells were seeded in the culture dish and cultured with 0.50 mg mL−1 NSCDs and 0.5 mg mL−1 NSCDs/1.0 mM Cr(VI), respectively. After incubation at 37 °C for 4 h, the SiHa cells were harvest using 0.25% trypsin/0.020% EDTA, washed three times (1.0 mL each) with pH 7.4 phosphate buffered saline (PBS comprising 137 mM NaCl, 2.7 mM KCl, 8.0 mM Na2HPO4, and 2.0 mM KH2PO4) and kept in PBS for optical imaging by a Olympus FV1000 confocal microscope (Tokyo, Japan) with 20× and 40× objective.Results and discussion
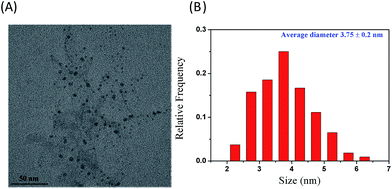
Four types of water-soluble NSCDs were synthesized using various CA/NAC: 0.10, 0.20, 0.40, and 0.60. They all possess similar spectral characteristics but different emission intensities. Among them, the NSCDs synthesized with CA/NAC of 0.60 produces the strongest fluorescence. As such, it was chosen for further characterisation and studies. Higher CA/NAC (>0.60) does not improve the fluorescence of the NSCDs and is not discussed in this work. Reaction time is another important parameter for NSCDs synthesis. As the reaction time proceeds from short to long, the polymer-like NSCDs are converted into carbogenic NSCDs. In a modest reaction time, polymer-like NSCDs are formed and the PL arises from the surface/molecule state (perhaps owing to amide-containing fluorophores). In a short or long reaction time, owing to further carbonization, partial carbogenic NSCDs are formed and the PL is derived from the synergistic effect of the carbogenic core and the surface/molecule state. The carbogenic core plays a greater role in NSCDs no matter the synthesis time decreases or increases. The optimal reaction time is found to be 8.0 min as it produces the highest Φs. The NSCDs were prepared by the microwave-assisted method, which the reaction was conducted by first condensing CA and NAC, whereupon they formed polymer-like NSCDs, which were then carbonized to form the NSCDs.TEM has been used extensively as a powerful tool in the study of nanoparticles from which the morphology and size can be identified. Fig. 1A shows the representative TEM image, which shows the synthesized NSCDs are mostly of spherical morphology and disperse rather evenly on the TEM grid surface. The corresponding histogram obtained by statistical analysis of approximately 150 particles using the ImageJ software are displayed in Fig. 1B. The Gaussian fitting curves reveal that the corresponding particle size distribution of NSCDs is narrowly distributed of 2.25–6.25 nm with average diameter in the range of 3.75 ± 0.2 nm.
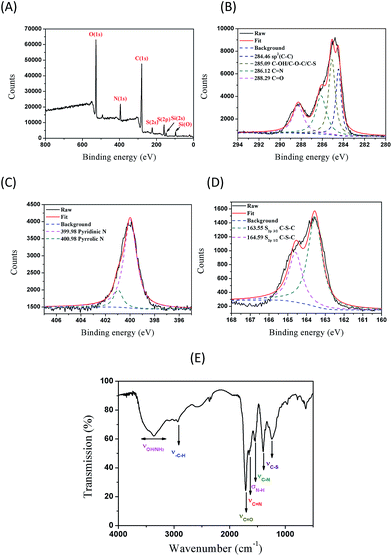
To probe the chemical composition and content of the as-synthesized NSCDs, the elemental analysis and XPS measurements were obtained. The doping of heteroatoms into NSCDs was initially probed by elemental analysis in Table S1A (ESI†) which is consistent with the results of XPS (vide infra), revealing that the NSCDs contain carbon (C), hydrogen (H), oxygen (O), nitrogen (N), and sulfur (S). Higher N and S contents and less O content are found for the NSCDs. The elemental contents are expressed in terms of relative number of atom as depicted in Table S1B.† The empirical formula for NSCDs is approximately C19H28O11N4S2. As shown in Fig. 2A, the survey XPS of NSCDs sample reveals the presence of C, N, S and O as well as limited H without any other impurities. The binding energy peaks at 280.4, 527.0, 159.0 and 527.0 eV correspond to C1s, N1s, S2p and O1s, respectively. In detail, the C1s spectrum (Fig. 2B) are deconvoluted into four peaks at 284.46, 285.09, 286.12 and 288.29 eV, which are attributed to sp2(C–C), C–OH/C–O–C/C–S, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N, and C
N, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, respectively.23–25 Fig. 2C displays the high-resolved N1s spectrum which can be deconvoluted into two peaks at 399.98 and 400.98 eV, representing N1s states in pyridinic N and pyrrolic N, respectively.23 The high-resolution spectrum of S2p (Fig. 2D) reveal the presence of C–S–C units, which could be fitted into two peaks at 163.55 and 164.59 eV corresponding to S2p3/2 and S2p1/2 C–S–C, respectively.25 The O1s of NSCDs spectrum (Fig. S1†) shows four peaks at 531.29, 531.69, 532.19 and 532.59 eV attributing to the O*
O, respectively.23–25 Fig. 2C displays the high-resolved N1s spectrum which can be deconvoluted into two peaks at 399.98 and 400.98 eV, representing N1s states in pyridinic N and pyrrolic N, respectively.23 The high-resolution spectrum of S2p (Fig. 2D) reveal the presence of C–S–C units, which could be fitted into two peaks at 163.55 and 164.59 eV corresponding to S2p3/2 and S2p1/2 C–S–C, respectively.25 The O1s of NSCDs spectrum (Fig. S1†) shows four peaks at 531.29, 531.69, 532.19 and 532.59 eV attributing to the O*![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–O, O
C–O, O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–O*, C–OH/C–O–C, and C
C–O*, C–OH/C–O–C, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups, respectively.23–25 Again, these confirm the doping of N and S onto the surface of the as-synthesized NSCDs. In summary, the XPS data show the presence of C
O groups, respectively.23–25 Again, these confirm the doping of N and S onto the surface of the as-synthesized NSCDs. In summary, the XPS data show the presence of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, C–O, C
C, C–O, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, C–S–C, C–N, and O–C
O, C–S–C, C–N, and O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O surface-functionalities are on the NSCDs. Fig. 2E displays the FTIR of NSCDs. The existence of a broad peak centred at 3367 cm−1 represents the stretching vibrations of O–H or N–H bond.6 Several sharp peaks at 1238, 1397, 1541, 1653, and 1712 cm−1 correspond to C–S, amido CO–N, CON–H, C
O surface-functionalities are on the NSCDs. Fig. 2E displays the FTIR of NSCDs. The existence of a broad peak centred at 3367 cm−1 represents the stretching vibrations of O–H or N–H bond.6 Several sharp peaks at 1238, 1397, 1541, 1653, and 1712 cm−1 correspond to C–S, amido CO–N, CON–H, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N, and C
N, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, respectively, which improve the hydrophilicity and stability of the NSCDs in aqueous system.6 Both XPS and IR confirm the surface of NSCDs is co-doped with N and S atoms. In summary, the pyrolysis of CA and doping of N and S heteroatoms into NSCDs should have taken place at high temperature under microwave irradiation, resulting in the formation of NSCDs covered with carboxylic acid, amido and alkyl sulfide moieties. These functional groups are potential linkers for attachment of therapeutic moieties for targeted drug delivery.
O, respectively, which improve the hydrophilicity and stability of the NSCDs in aqueous system.6 Both XPS and IR confirm the surface of NSCDs is co-doped with N and S atoms. In summary, the pyrolysis of CA and doping of N and S heteroatoms into NSCDs should have taken place at high temperature under microwave irradiation, resulting in the formation of NSCDs covered with carboxylic acid, amido and alkyl sulfide moieties. These functional groups are potential linkers for attachment of therapeutic moieties for targeted drug delivery.
 | ||
| Fig. 2 (A) XPS survey scan, (B) C1s XPS, (C) N1s XPS and (D) S2p XPS of NSCDs. (E) FTIR spectrum of NSCDs. | ||
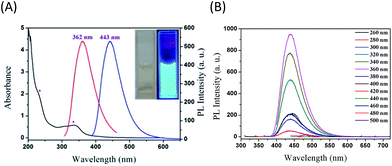
In quest of exploring the optical properties of the as-prepared NSCDs, the UV-vis absorption and PL spectra were acquired and depicted in Fig. 3. As shown in the UV-vis absorption spectrum (blank line in Fig. 3A), we propose that the strong absorption peak of NSCDs at 335 nm probably attributing to the formation of excited defect surface states induced by the N and S heteroatoms and the weak absorption peak at 232 nm is attributed to the π → π* transition of the aromatic sp2 bond.22 The PL spectra of NSCDs are shown in Fig. 3A (red and blue lines). It can be seen that the optimal excitation (λex) and emission (λem) wavelengths are located at 362 and 443 nm, respectively. The photograph of the NSCDs dispersion under UV light (365 nm) exhibits strong blue emission (the inset of Fig. 3A). To further explore the optical properties of the as-prepared NSCDs, the PL spectra of NSCDs under various λex was investigated and depicted in Fig. 3B. The λem peak of NSCDs remains constant ca. 439–446 nm when λex is 260–380 nm; however, it is red-shift from 480 to 621 nm when λex moves from 400 to 500 nm. The λex-independent PL spectra are probably attributed to the π* → π transitions of the graphitic structure of the carbon cores whereas the λex-dependent PL spectra are derived from the π* → n transitions (surface states) of the surface-attached functionalities (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O/C–NH2/C–S–C). The λex-dependent PL spectra are bathochromically shifted with the increase in λex, indicating that the PL band can be tuned by adjusting λex. The λex-dependent PL behavior is common with most CDs. This means that λem can be tuned by just controlling λex without changing CDs. The λex-dependent PL behavior is useful for multicolor imaging applications (vide infra). Furthermore, using quinine sulfate (Fig. S2A†) as a reference, the fluorescence Φs of the as-synthesized NSCDs (Fig. S2B†) was measured to be 14.4%. The as-prepared NSCDs exhibits excellent stability which is essential for practical applications.
O/C–NH2/C–S–C). The λex-dependent PL spectra are bathochromically shifted with the increase in λex, indicating that the PL band can be tuned by adjusting λex. The λex-dependent PL behavior is common with most CDs. This means that λem can be tuned by just controlling λex without changing CDs. The λex-dependent PL behavior is useful for multicolor imaging applications (vide infra). Furthermore, using quinine sulfate (Fig. S2A†) as a reference, the fluorescence Φs of the as-synthesized NSCDs (Fig. S2B†) was measured to be 14.4%. The as-prepared NSCDs exhibits excellent stability which is essential for practical applications.
The effects of ionic strength (in terms of the concentration of KCl), pHs, and UV irradiation on the PL stability of NSCDs were investigated. The PL intensity and spectral feature of NSCDs do not change much under different concentrations of KCl (Fig. S3A†), which is beneficial since it is necessary for NSCDs to be used in the presence of various salt concentrations in practical applications. Fig. S3B† displays the effect of pH on the PL intensity of NSCDs. The PL intensity increases with the increase in pH and reaches the highest at pH 7.0–10.0; however, the PL intensity drops significantly when the pH is higher than 11.0. Since the PL intensity is highest and maintains fairly constant at the physiological pH 7.0–7.5, it has potential for application in cellular imaging (vide infra). No obvious photobleaching was observed (>79%) after 3 h of continuous UV irradiation as shown in Fig. S3C,† suggesting that the photostability of NSCDs is good. The dry NSCDs powder sample could be repeatedly re-dispersed in water without any aggregation which is advantageous for preservation and transportation. The obtained NSCDs solution exhibits homogeneous phase without any noticeable precipitation at ambient conditions for six months, indicating their long-term colloidal stability.
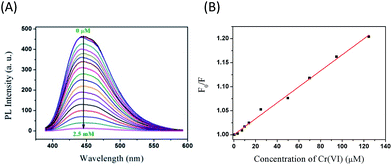
Of particular interest and significance is the finding that the as-prepared NSCDs can be utilized as a highly efficient nanoprobe for Cr(VI) detection. Most reported Cr(VI) detection probes are commonly suffered from the drawbacks of narrow range of detection concentration,30 low accuracy,30,32 or low selectivity.31 Herein, Cr(VI) can be detected with NSCDs via luminescence measurements. Fig. 4A shows the fluorescent quenching of NSCDs at various concentrations of Cr(VI). Fig. 4B displays the Stern–Volmer plot of NSCDs with increasing concentration of Cr(VI) (F0/F against concentration of Cr(VI)), where F0 and F are the PL intensities of NSCDs at λex/λem of 362/443 nm in the absence and presence of Cr(VI), respectively. The quenching efficiency is fitted by the Stern–Volmer equation, F0/F = 1 + Ksv[Q], where Ksv is the Stern–Volmer quenching constant and [Q] is the Cr(VI) concentration. The Ksv is calculated to be 1.66 × 103 L mol−1 with a correlation coefficient r2 of 0.994. The F0/F curve is linearly related to the concentration of Cr(VI) in the range 0.5–125 μM (i.e., 26–6500 μg L−1 of Cr(VI)), indicating their excellent sensing properties in the detection of trace Cr(VI). The limit of detection (LOD) and limit of quantification (LOQ) were calculated by taking the PL intensity equal to 3 times the standard deviation of the intensity at the blank (n = 10) divided by the slope of the calibration graph, and three times the LOD, respectively. The LOD and LOQ of the proposed sensor were determined as 20 (1.04 μg L−1) and 60 nM at a signal-to-noise ratio of 3,23,32 respectively. The World Health Organization (WHO) stipulates that Cr(VI) concentrations lower than 50 μg L−1 are acceptable in drinking water,33 inferring that our NSCDs-based fluorescent method is suitable for monitoring Cr(VI) concentration in drinking water.
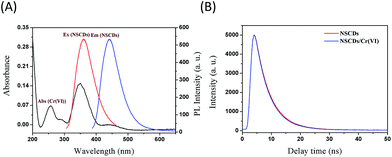
IFE phenomenon of fluorescence usually occurs between absorber and fluorophore resulting from the absorption of the excitation and/or emission light by absorbers in the detection system. The effective IFE requires the complementary overlap of the absorber's absorption band with the excitation and/or emission bands of the fluorophore. Therefore, it is important to choose a suitable absorber and fluorophore pair for the IFE-based fluorescent chemosensor. As shown in Fig. 5A, the excitation spectrum of NSCDs has a band at 362 nm (red line in Fig. 5A), and the emission band of NSCDs under the excitation of 362 nm is centered at 443 nm (blue line in Fig. 5A); however, Cr(VI) exhibits broad absorption at 257, 350, and 450 nm (black line in Fig. 5A), respectively, showing quite precise overlapping with the excitation and emission bands of NSCDs. Consequently, Cr(VI) can not only shield the excitation light for NSCDs but also absorb the emission light from NSCDs. Naturally, the absorbance enhancement of Cr(VI) could be successfully converted to fluorescence quenching of NSCDs, which ensures that the IFE occurs in a highly efficient way. To obtain further insight into the PL quenching mechanism between NSCDs fluorophore and Cr(VI) absorber, time-correlated single-photon counting (TCSPC) was used to study the lifetime of NSCDs in the absence and presence of Cr(VI) as depicted in Fig. 5B. The lifetime data are summarized in Table S2.† The PL decay curves can be well-fitted by a double-exponential function: I(t) = A1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−t/τ1) + A2
exp(−t/τ1) + A2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−t/τ2), where τ1 and τ2 are the time constants of the two radiative decay channels; A1 and A2 are the corresponding amplitudes. From the best fit of the data, τ1(A1) and τ2(A2) of NSCDs and NSCDs/Cr(VI) are derived to be 10.19 ns (83.24%) and 1.486 ns (16.76%), and 9.56 ns (90.72%) and 0.4619 ns (9.275%), respectively. In the absence (the red line in Fig. 5B) and presence (the blue line in Fig. 5B) of Cr(VI), the average lifetime values of NSCDs and NSCDs/Cr(VI) were 8.732 ns and 8.717 ns, respectively. In terms of statistical significance, the fluorescence lifetime of NSCDs was almost no change in the absence and presence of Cr(VI), which indicated that there is no significant excited-state interaction between NSCDs and Cr(VI), demonstrating that the fluorescence quenching of NSCDs by Cr(VI) resulted from the simple absorption of the excitation and emission light by the absorber. Also, Cr(VI) is positively charged, and no electrostatic interaction took place between NSCDs and Cr(VI). This can facilitate our demonstration of the present concept that the fluorescence sensing would be clearly based on the IFE rather than other possible approaches. In addition, sensitivity and selectivity are important parameters to evaluate the performance of the sensing system. The effect of representative metal ions (K+, Na+, Ag+, Zn2+, Fe2+, Cd2+, Hg2+, Ba2+, Ca2+, Cu2+, Mg2+, Ni2+, Pb2+, Mn2+, Co2+, Fe3+, Al3+, Cr3+, Cr(VI), Mn(VII)) on NSCDs fluorescence quenching under the same conditions are investigated and displayed in Fig. S4.† Except Cr2O72−, most of these metal ions of ultrahigh concentrations (10 mM) do not induce significant decrease in PL intensity of PNHCDs; thus do not cause any interference in detection of Cr(VI). This PNHCDs-based “turn-off” fluorescent probe provides obvious advantages of simplicity, convenience, and rapid implementation and thus has potential application for the detection of Cr(VI) in environment and industry.
exp(−t/τ2), where τ1 and τ2 are the time constants of the two radiative decay channels; A1 and A2 are the corresponding amplitudes. From the best fit of the data, τ1(A1) and τ2(A2) of NSCDs and NSCDs/Cr(VI) are derived to be 10.19 ns (83.24%) and 1.486 ns (16.76%), and 9.56 ns (90.72%) and 0.4619 ns (9.275%), respectively. In the absence (the red line in Fig. 5B) and presence (the blue line in Fig. 5B) of Cr(VI), the average lifetime values of NSCDs and NSCDs/Cr(VI) were 8.732 ns and 8.717 ns, respectively. In terms of statistical significance, the fluorescence lifetime of NSCDs was almost no change in the absence and presence of Cr(VI), which indicated that there is no significant excited-state interaction between NSCDs and Cr(VI), demonstrating that the fluorescence quenching of NSCDs by Cr(VI) resulted from the simple absorption of the excitation and emission light by the absorber. Also, Cr(VI) is positively charged, and no electrostatic interaction took place between NSCDs and Cr(VI). This can facilitate our demonstration of the present concept that the fluorescence sensing would be clearly based on the IFE rather than other possible approaches. In addition, sensitivity and selectivity are important parameters to evaluate the performance of the sensing system. The effect of representative metal ions (K+, Na+, Ag+, Zn2+, Fe2+, Cd2+, Hg2+, Ba2+, Ca2+, Cu2+, Mg2+, Ni2+, Pb2+, Mn2+, Co2+, Fe3+, Al3+, Cr3+, Cr(VI), Mn(VII)) on NSCDs fluorescence quenching under the same conditions are investigated and displayed in Fig. S4.† Except Cr2O72−, most of these metal ions of ultrahigh concentrations (10 mM) do not induce significant decrease in PL intensity of PNHCDs; thus do not cause any interference in detection of Cr(VI). This PNHCDs-based “turn-off” fluorescent probe provides obvious advantages of simplicity, convenience, and rapid implementation and thus has potential application for the detection of Cr(VI) in environment and industry.
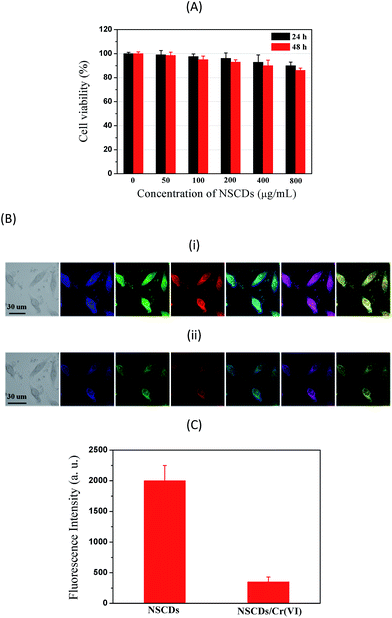
The cytotoxicity of NSCDs is a natural concern because of their potential for bioimaging and nanoscale dimensions. To evaluate the cytotoxicity of the NSCDs, the viability of human cervical carcinoma SiHa cells treated with NSCDs was measured using the MTT method. In the MTT assay, MTT could be reduced by the active cellular enzymes in the cells to insoluble blue-violet formazan crystals. The quantitative information about the cytotoxicity of NSCDs can be obtained by measuring the absorption on the cells. As shown in Fig. 6A, SiHa cells were incubated with concentration ranging from 50 μg mL−1 to 800 μg mL−1 of NSCDs for 24 and 48 h, respectively. The viability of the cells remained greater than 86% even incubated with ultrahigh concentration (800 μg mL−1) of NSCDs for 48 h, demonstrating instinctively low toxicity of NSCDs (without any further functionalization).
In order to explore the potential application of PNHCDs in vitro imaging of living cells, SiHa cells were exposed to NSCDs aqueous solutions for 4 h. The cellular uptake of NSCDs was then observed by laser scanning confocal microscopic (LSCM) as depicted in Fig. 6B(i). NSCDs were well-dispersed in the cytoplasm region between the nucleus and the cell membrane, and the cells stained with NSCDs display strongest blue emissions. When Cr2O72− was added into SiHa cells culture medium, the blue emissions were hardly found as shown in Fig. 6B(ii). The bright-field images of SiHa cells incubated with (i) NSCDs and (ii) NSCDs/Cr(VI) (first panels in Fig. 6B) indicate clearly the normal morphology of the cells, verifying that NSCDs and NSCDs/Cr(VI) are biocompatible and possess minimum toxicity to the cells. The cells display blue (second panels), green (third panels), and red (fourth panels in Fig. 6B) emissions when they were excited with 405, 488, and 543 nm lasers, respectively. The merged images (the fifth panels in Fig. 6B) of the second and third panels, merged images (the sixth panels in Fig. 6B) of the second and fourth panels, and merged images (the seventh panels in Fig. 6B) of the third and fourth panels demonstrate the ability of NSCDs to penetrate into the cell membrane without any further surface passivation. This observation demonstrates NSCDs potential as a bioimaging agent for living cells. The cell incubated with NSCDs emit stronger than that of NSCDs/Cr(VI), attributing to the fact that Cr(VI) could quench NSCDs in aqueous solution (vide supra). Fig. 6C displays the intracellular fluorescence intensities of NSCDs in the absence and presence of Cr2O72−, respectively. It is obvious that the cells show the emission intensity without Cr2O72− > with Cr2O72−. These results further demonstrate that NSCDs are efficient fluorescent probes for “on–off” monitoring the Cr(VI) in live cells.
Conclusions
For the first time, less toxic elements nitrogen and sulfur co-doped CDs (NSCDs) have been synthesized from a fast, simple and “green” route with CA and NAC as the precursors with a Φs as high as 14.4%. The morphology and chemical structures have been investigated extensively by TEM, XPS, FTIR, UV-vis absorption, and PL spectroscopy. The as-prepared NSCDs show good fluorescence without further surface treatment. In addition, NSCDs display excellent stability under various conditions, including external metal ionic, pH, and continuous excitation. Moreover, NSCDs show good sensitivity and selectivity to Cr(VI) in complex environments, and has a remarkable selectivity over other representative metal ions, it can be serve as an effective probe for PL detection of Cr(VI) with a detection limit as low as 20 nM. Furthermore, NSCDs could be utilized as a reagent capable of detecting Cr(VI) in biosystem. Combining its simple and fast synthetic method, it is anticipated that NSCDs could have potential applications in biological labeling, disease diagnosis, biosensing, biomedical and biological fields.Acknowledgements
This work was supported by the National Science Foundation of China (21575084 and 21475080), the Hundred Talent Programme of Shanxi Province, and Shanxi Scholarship Council of China.Notes and references
- X. Michalet, F. F. Pinaud, L. A. Bentolila, J. M. Tsay, S. Doose, J. Li, G. A. Sundaresan, M. Wu, S. S. Gambhir and S. Weiss, Science, 2005, 307, 538 CrossRef CAS PubMed.
- J. Jeong, M. Cho, Y. T. Lim, N. W. Song and B. H. Chung, Angew. Chem., Int. Ed., 2009, 48, 5296 CrossRef CAS PubMed.
- K. Welsher, Z. Liu, S. P. Scherlock, J. T. Robinson, Z. Chen, D. Daranciang and H. Dai, Nat. Nanotechnol., 2009, 4, 773 CrossRef CAS PubMed.
- A. Krueger, Adv. Mater., 2008, 20, 2445 CrossRef CAS.
- S. Zhuo, M. Shao and S.-T. Lee, ACS Nano, 2012, 6, 1059 CrossRef CAS PubMed.
- X. Gong, Q. Hu, M. C. Paau, Y. Zhang, S. Shuang, C. Dong and M. M. F. Choi, Nanoscale, 2014, 6, 8162 RSC.
- Y. Zhai, Z. Zhu, C. Zhu, J. Ren, E. Wang and S. Dong, J. Mater. Chem. B, 2014, 2, 6995 RSC.
- X. Xu, R. Ray, Y. Gu, H. J. Ploehn, L. Gearheart, K. Raker and W. A. Scrivens, J. Am. Chem. Soc., 2004, 126, 12736 CrossRef CAS PubMed.
- A. B. Bourlinos, A. Bakandritsos, A. Kouloumpis, D. Gournis, M. Krysmann, E. P. Giannelis, K. Polakova, K. Safarova, K. Hola and R. Zboril, J. Mater. Chem., 2012, 22, 23327 RSC.
- S. C. Ray, A. Saha, N. R. Jana and R. Sarkar, J. Phys. Chem. C, 2009, 113, 18546 CAS.
- L. Shen, L. Zhang, M. Chen, X. Chen and J. Wang, Carbon, 2013, 55, 343 CrossRef CAS.
- X. Ran, H. Sun, F. Pu, J. Ren and X. Qu, Chem. Commun., 2013, 49, 1079 RSC.
- S. N. Baker and G. A. Baker, Angew. Chem., Int. Ed., 2010, 49, 6726 CrossRef CAS PubMed.
- Y. Sun, B. Zhou, Y. Lin, W. Wang, K. A. S. Fernando, P. Pathak, M. J. Meziani, B. A. Harruff, X. Wang, H. Wang, P. G. Luo, H. Yang, M. E. Kose, B. Chen, L. M. Veca and S.-Y. Xie, J. Am. Chem. Soc., 2006, 128, 7756 CrossRef CAS PubMed.
- Q.-L. Zhao, Z.-L. Zhang, B.-H. Huang, J. Peng, M. Zhang and D.-W. Pang, Chem. Commun., 2008, 5116 RSC.
- V. N. Mochalin and Y. Gogotsi, J. Am. Chem. Soc., 2009, 131, 4594 CrossRef CAS PubMed.
- A. B. Bourlinos, A. Stassinopoulos, D. Anglos, R. Zboril, V. Georgakilas and E. P. Giannelis, Chem. Mater., 2008, 20, 4539 CrossRef CAS.
- X. Sun, Z. Liu, K. Welsher, J. T. Robinson, A. Goodwin, S. Zaric and H. Dai, Nano Res., 2008, 1, 203 CrossRef CAS PubMed.
- C.-I. Wang, W.-C. Wu, A. P. Periasamy and H.-T. Chang, Green Chem., 2014, 16, 2509 RSC.
- S. Zhu, Q. Meng, L. Wang, J. Zhang, Y. Song, H. Jin, K. Zhang, H. Sun, H. Wang and B. Yang, Angew. Chem., Int. Ed., 2013, 52, 3953 CrossRef CAS PubMed.
- Z. Zhang, J. Hao, J. Zhang, B. Zhang and J. Tang, RSC Adv., 2012, 2, 8599 RSC.
- X. Wang, L. Cao, S. T. Yang, F. Lu, M. J. Meziani, L. Tian, K. W. Sun, M. A. Bloodgood and Y. Sun, Angew. Chem., Int. Ed., 2010, 49, 5310 CrossRef CAS PubMed.
- X. Gong, W. Lu, M. C. Paau, Q. Hu, X. Wu, S. Shuang, C. Dong and M. M. F. Choi, Anal. Chim. Acta, 2015, 861, 74 CrossRef CAS PubMed.
- W. Kwon, J. Lim, J. Lee, T. Park and S.-W. Rhee, J. Mater. Chem. C, 2013, 1, 2002 RSC.
- P. Guo, F. Xiao, Q. Liu, H. Liu, Y. Guo, J. R. Gong, S. Wang and Y. Liu, Sci. Rep., 2013, 3, 3499 Search PubMed.
- W. Wang, Y.-C. Lu, H. Huang, A.-J. Wang, J.-R. Chen and J.-J. Feng, Biosens. Bioelectron., 2015, 64, 517–522 CrossRef CAS PubMed.
- W. Wang, Y.-C. Lu, H. Huang, A.-J. Wang, J.-R. Chen and J.-J. Feng, Sens. Actuators, B, 2014, 202, 741–747 CrossRef CAS.
- H. Ding, J.-S. Wei and H.-M. Xiong, Nanoscale, 2014, 6, 13817–13823 RSC.
- F. Cai, X. Liu, S. Liu, H. Liu and Y. Huang, RSC Adv., 2014, 4, 52016 RSC.
- E. Punrat, C. Maksuk, S. Chuanuwatanakul, W. Wonsawat and O. Chailapakul, Talanta, 2016, 150, 198 CrossRef CAS PubMed.
- C. K. Balavigneswaran, T. S. J. Kumar, R. M. Packiaraj and S. Prakash, Appl. Nanosci., 2014, 4, 367 CrossRef CAS.
- Y. Liu, J. Hu, Y. Li, H.-P. Wei, X.-S. Li, X.-H. Zhang, S.-M. Chen and X.-Q. Chen, Talanta, 2015, 134, 16 CrossRef CAS PubMed.
- World Health Organization, Guidelines for Drinking Water Quality, World Health Organization, Geneva, Switzerland, 1997 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Elemental analysis, O1s XPS of NSCDs, plots of integrated PL intensity against absorbance of quinine sulfate and NSCDs, effect of ionic strength, pH, and UV excitation time on PL intensity of NSCDs, double-exponential fitting of NSCDs and NSCDs/Cr(VI) decay curves, comparison of fluorescence of NSCDs after addition of different metal ions. See DOI: 10.1039/c6ra02653b |
| This journal is © The Royal Society of Chemistry 2016 |