Synthesis of plasmonic Au–CuS hybrid nanocrystals for photothermal transduction and chemical transformations†
Chao Sun‡
ab,
Mengya Liu‡b,
Yu Zou*b,
Jiandong Weia and
Jiang Jiang*b
aCollege of Environmental and Chemical Engineering, Shanghai University, Shanghai 200444, China
bCAS Key Laboratory of Nano-Bio Interface, i-Lab and Division of Nanobiomedicine, Suzhou Institute of Nano-Tech and Nano-Bionics, Chinese Academy of Sciences, Suzhou 215123, China. E-mail: jjiang2010@sinano.ac.cn; yzou2012@sinano.ac.cn
First published on 4th March 2016
Abstract
Plasmonic Au–copper sulfides hybrid nanocrystals are of great interest for both fundamental research and application in optoelectronics, catalysis, and biomedicine, in which a precise and high-yield synthesis of the hybrid nanocrystals is required. Herein, we investigated colloidal synthesis of Au–CuS hybrid nanocrystals based on a seed-mediated method. Different from previously reported deposition of Cu-rich Cu chalcogenides, we found that decreasing the Cu–S reaction rate is critical for the deposition of relatively S-rich CuS on Au seeds in high yield, which can be achieved by using a chlorobenzene solution containing a small amount of oleylamine as the solvent. The influence of the S/Cu feed molar ratio and reaction temperature on the formation of Au–CuS hybrid nanocrystals has also been investigated. The obtained Au–CuS hybrids showed better photothermal transduction efficiency over CuS nanocrystals prepared under identical conditions. In addition, Au–CuS hybrids could be used as templates to synthesize Au–CuInS2 and Au–Cu2ZnSnS4 hybrid nanocrystals via subsequent reactions with In or Zn/Sn precursors at high temperature. These results provide insight into the critical parameters governing the growth of Au–copper sulfides hybrid nanostructures, which can be useful for the design of other noble metal–semiconductor hybrid nanostructures.
Introduction
Hybrid nanocrystals composed of plasmonic metal and semiconductor components have emerged as a new kind of plasmonic materials in recent years.1–5 By combining the unique properties of both plasmonic metal and semiconductor components in a single particle, these hybrid nanocrystals can exhibit multiple and often synergistically enhanced properties.1,5Among such plasmonic hybrid nanocrystals, Au–copper chalcogenide hybrid nanocrystals have attracted significant attention.1,2 In such hybrids, the metal domains are Au nanocrystals, with size and shape dependent plasmonic properties, useful in surface enhanced Raman scattering, optical imaging and as X-ray computed tomography (CT) imaging contrast agents;6–8 the semiconductor domains are self-doped copper chalcogenides, whose plasmonic absorption is located in the near infrared (NIR) and can be tuned by controlling the doping level of Cu vacancies, useful as photoacoustic contrast agent and photothermal material for cancer therapy.9–13 Thus, Au–Cu chalcogenides hybrid nanocrystals are very promising multifunctional biomaterials for theranostics as well as for efficient solar energy harvesting. Furthermore, these Au–binary copper chalcogenide hybrid nanocrystals can serve as the templates for synthesizing Au–multinary copper chalcogenide hybrids, which are important in optoelectronics and photocatalysis applications.
Although some pioneering work on plasmonic Au–copper chalcogenides hybrid nanocrystals, such as Au–Cu1.8S1 and Au–Cu2−xSe,2 have been reported, the growth mechanism and structural control on these hybrid nanocrystals remain elusive. In addition, the diversity of copper chalcogenides renders the synthesis of specific Au–copper chalcogenides hybrid nanocrystals more challenging. Taking copper sulfides as an example, at least four crystalline phases display plasmonic properties, including Cu1.97S (djurleite), Cu1.8S (digenite), Cu1.75S (anilite), and CuS (covellite).14–18 As a result, the study on nucleation and growth dynamic of copper sulfides especially S-rich CuS on Au seeds has been very limited, in stark contrast to the work conducted on Au–CdS19–21 and Au–PbS,22–24 whose compositions are less sensitive to reaction parameters.
Herein, we present the synthesis of rarely investigated Au–CuS hybrid nanocrystals using a facile seed-mediated method. We optimized the Cu–S reaction rate to obtain Au–CuS hybrid nanocrystals in high yield, and revealed the effects of reaction rate and Ostwald ripening on the formation of Au–CuS hybrid nanocrystals. These Au–CuS hybrid nanocrystals exhibited enhanced photothermal transduction property compared to CuS nanocrystals. Furthermore, more complex Au–CuInS2 and Au–Cu2ZnSnS4 hybrid structures have been realized through simple chemical transformations from Au–CuS.
Experimental section
Materials
Tetrachloroauric(III) acid hydrate (HAuCl4·4H2O), thiourea, and chlorobenzene (99.5%) were purchased from Sinopharm Chemical Reagent Co., Ltd. Copper acetylacetonate (Cu(acac)2), sublimed sulfur (S), oleylamine (OM, 80–90%), trioctylamine (TOA, 90%), 1,2-dichlorobenzene (DCB, 99%) and 1,2,4-trichlorobenzene (99%), were purchased from Aladdin Chemistry Co., Ltd. Zinc acetylacetonate monohydrate (Zn(acac)2·H2O), indium acetylacetonate (In(acac)3, 98%) and tin bis(acetylacetonate) chloride (Sn(acac)2Cl2, 95%) were purchased from Alfa Aesar. Octadecene (ODE, 90%) and poly(maleic anhydride-alt-1-octadecene) (PMAO) were purchased from Sigma-Aldrich.Synthesis of Au nanocrystals
5 mL OM in a 100 mL three-neck flask was first heated up to 160 °C under N2, followed by injection of 0.1 mL 1 M HAuCl4 aqueous solution. Au3+ was immediately reduced by OM to Au0. After holding at 160 °C for 60 minutes, the solution was cooled down to room temperature naturally. Au nanocrystals were isolated by adding ethanol followed by centrifugation, and then dispersed in 5 mL specific solvent (for example, dichlorobenzene).Synthesis of Au–CuS hybrid nanocrystals
Typically, 0.1 mmol Cu(acac)2, 1 mL OM and 5 mL as-prepared dichlorobenzene dispersion of Au nanocrystals were mixed and heated up to 80 °C. At this point, 1 mL 0.1 M dichlorobenzene solution of S was injected swiftly. The temperature of the mixture was then raised to 100 °C and held for 30 min under N2. After the reaction was stopped, Au–CuS hybrid nanocrystals were isolated by adding ethanol and then centrifugation, and purified by a hexane and ethanol washing step. Finally, the nanocrystals were dispersed in hexane or chloroform for characterization, or in ODE for subsequent chemical transformation reactions.Photothermal transduction of Au–CuS hybrid nanocrystals
The purified Au–CuS hybrid nanocrystals dispersed in chloroform were first transferred into water by encapsulation using amphiphilic PMAO.25 Subsequently, 0.6 mL aqueous dispersion of hybrid nanocrystals was loaded into a cuvette and then exposed to a 1064 nm laser at a power density of 1 W cm−2 for 5 min. The temperature of the dispersion was recorded every 20 s using an inserted thermocouple probe with 0.1 °C accuracy. The pure CuS nanocrystals used for comparison were prepared in the absence of Au seeds at 100 °C, with the phase transfer and photothermal measurement procedures kept the same.Chemical transformation of Au–CuS hybrid nanocrystals
The purified Au–CuS hybrid nanocrystals were dispersed in 5 mL ODE and loaded into a flask. Subsequently, 0.1 mmol metal precursors, 0.15 mmol thiourea and 1 mL OM were added, and the mixture was then heated up to 240 °C quickly and kept at this temperature for 30 min under N2. The metal precursors could be In(acac)3 or a combination of 0.05 mmol Zn(acac)2·H2O and 0.05 mmol Sn(acac)2Cl2. The remaining separation and washing steps were the same as those used for the preparation of Au–CuS hybrid nanocrystals.Characterizations
Transmission electron microscopy (TEM) and high-resolution transmission electron microscopy (HRTEM) images were taken by FEI Technai G2 S-Twin at an accelerating voltage of 200 kV. The TEM samples were prepared by dropping a small volume of diluted nanocrystal dispersion onto a carbon-coated copper grid followed by solvent evaporation. X-ray diffraction (XRD) analysis of drop-cast films of nanocrystals was conducted using a Bruker D8 Advance X-ray diffractometer (with Cu Kα radiation at 0.15418 nm). Fourier transform infrared (FTIR) spectra were recorded with a Nicolet 6700 IR spectrometer (KBr wafer technique). The energy dispersive X-ray (EDX) analysis of the nanocrystals was conducted on a Quanta 250 FEG (FEI, USA) scanning electron microscope (SEM). UV-vis-NIR extinction spectra were recorded on a PerkinElmer Lambda-25 spectrometer. The Cu concentrations were measured by an Inductively Coupled Plasma Optical Emission Spectrometer (PerkinElmer ICP-OES 2100 DV).Results and discussion
Chlorobenzenes as solvent for hybrid nanocrystal synthesis
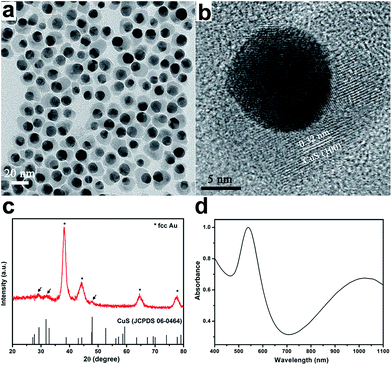
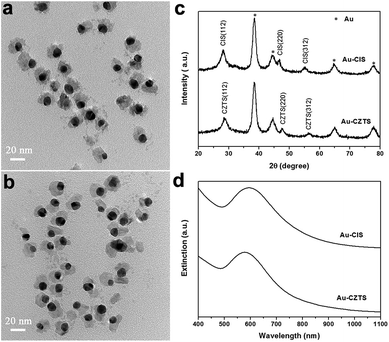
The reported methods for Au–Cu2−xS hybrid nanocrystals synthesis can be divided into two main categories: sulfidation of AuCu alloy nanocrystals26,27 and seeded growth.1,28 As the latter is more convenient for morphology and structure control, we adopted the seeded growth protocol to investigate the key parameters governing the formation of Au–CuS hybrid nanocrystals. First, Au seed nanocrystals were prepared via reduction of Au3+ by OM. Then, the purified Au seeds were mixed with Cu precursors and OM in a chosen solvent, followed by injection of S precursors at elevated temperatures. The reaction between Cu and S precursors induced nucleation and growth of copper sulfides on Au seeds. The formation of copper sulfides in the presence of Au seeds was more rapid than that without Au seeds, judging from the color change of the reaction solution, which is expected due to the lower heterogenous nucleation energy relative to that of homogenous nucleation.Fig. 1a shows a representative TEM image of the Au–CuS hybrid nanocrystals obtained under the conditions described in the experimental section. All CuS nanocrystals were found to grow on Au seeds, which looked darker in the image contrast due to the higher electron density of Au than Cu and S. EDX analysis indicated the ratio of Cu/S in the hybrid nanocrystals was close to 1. The coating of CuS on Au seeds was not complete, namely Au–CuS heterodimers rather than core–shell nanocrystals were formed. The mean size of CuS domains is 6.4 ± 2.1 nm (ESI Fig. S1†). The lattice spacing of the lighter part observed in HRTEM was measured to be 0.32 nm (Fig. 1b), matching well with the (100) planes of covellite CuS. The structure of the hybrids was further characterized by XRD. The XRD pattern (Fig. 1c) revealed that the hybrids were composed of face-centered cubic (fcc) structured Au and hexagonal covellite CuS, although the diffraction intensity of CuS was much weaker compared to that of Au due to heavy atom effect, and similar phenomena have also been observed in other Au–copper sulfide hybrid nanocrystals.1,26–28 The XRD pattern of the CuS nanocrystals prepared under identical reaction conditions in the absence of Au seeds supported the assignment of covellite structure (Fig. S2†). It was worth noting that the single peak at ∼48° is characteristic for CuS, corresponding to (110) planes, which helps to distinguish CuS from other Cu sulfides phases. The surfaces of Au–CuS hybrid nanocrystals were capped with OM molecules, as confirmed by FTIR (Fig. S3†), which ensured high dispersity of these nanocrystals in nonpolar solvents. Similar with those of previously reported Au–Cu1.8S1 and Au–Cu2−xSe2 hybrid nanocrystals, the Au–CuS hybrid nanocrystals exhibited dual surface plasmon resonances: one in the visible region arising from Au component and another one in the NIR region from CuS component (Fig. 1d). Upon the deposition of CuS, the plasmonic peak of Au component shifted from 520 nm to ∼540 nm.
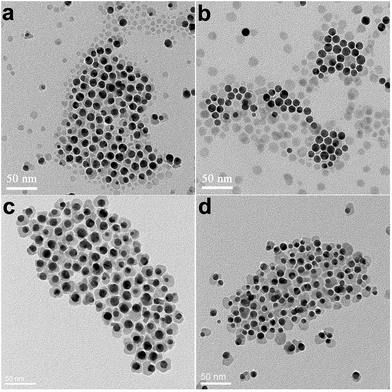
The choice of using DCB as solvent was based on the consideration to lower reactivity of S precursor, so that the homogeneous nucleation of CuS can be suppressed. When using elemental S powder as S source and commonly used amine as solvent, S is first activated by forming ionic ammonium polythioamine salts with amines,29 which will then react with Cu2+ ions to make copper sulfide. Taking previously used TOA and OM in Au–Cu1.8S and Au–Cu2−xSe synthesis as a comparison, a large amount (>40%) of free CuS nanocrystals were formed when pure TOA or OM was used instead of DCB (Fig. 2a and b), indicating the nucleation dynamics of CuS is faster in amine based solvents. Mechanistically, the nucleation rates should be determined by two important factors: precursor concentration and reactivity. Compared to other Cu-rich phases, the formation of CuS requires higher concentration of S precursor (S/Cu molar ratio should be at least 1).30,31 If the concentration of amines (activating agents) in reaction medium was high, then the Cu–S reaction would be too fast, resulting in homogenous nucleation of free CuS nanocrystals. To verify the effect of amines on the formation of Au–CuS hybrids, a series of experiments with varying concentrations of OM in DCB have been carried out (Fig. S4†). The experimental observations confirmed that the proportion of free CuS nanocrystals in the products increased with increasing concentration of OM. Therefore, although amines are essential for the formation of CuS, the concentration of amines in the reaction mixture should be kept at an optimal low concentration to obtain Au–CuS hybrid nanocrystals in high yield. We found that chlorinated benzenes were good solvents for the synthesis of Au–CuS hybrid nanocrystals. Besides dichlorobenzene, Au–CuS hybrid nanocrystals could also be formed effectively in chlorobenzene or trichlorobenzene, as shown in Fig. 2c and d, respectively.
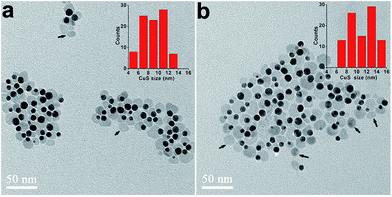
As discussed above, the S/Cu molar ratio should not be smaller than 1 for the formation of CuS phase. Using DCB solution of OM with low concentration as solvent to suppress the homogenous nucleation events, pure Au–CuS hybrid nanocrystals can be obtained when the S/Cu molar ratio was set to 1. When further increasing the amount of S precursor, the CuS domains on Au seeds became bigger due to more precursors participating in the growth of CuS and exhibited the shape of hexagonal nanoplates, which were commonly observed for large CuS nanocrystals (Fig. 3a and b).30–32 However, the appearance of free CuS nanocrystals (indicated by the arrow heads) was also observed, likely due to the accelerated nucleation of CuS at high precursor concentrations.
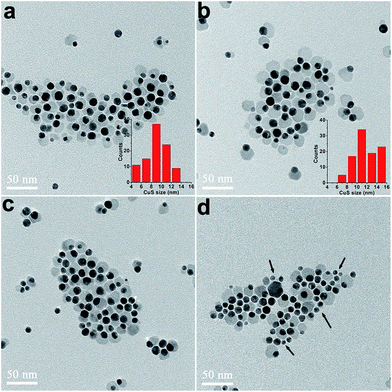
Reaction temperature is another determining factor of reaction dynamics. To suppress the homogenous nucleation, S precursors were injected at low temperature (80 °C). Subsequently, the reaction temperature was raised to 100 °C to complete the growth of CuS. Increasing reaction temperature enabled further growth of CuS domains. However, when the growth temperature was set too high (>140 °C), inter-particle Ostwald ripening was observed due to the exhaustion of precursors in the reaction mixture, leading to growth of large CuS domains at the expense of smaller ones (Fig. 4). As a consequence, a large number (20–50%) of bare Au nanocrystals emerged in the products at higher reaction temperatures (arrow heads in Fig. 4d). Moreover, Au–Cu2−xS (x < 1) rather than Au–CuS hybrid nanocrystals were obtained at the growth temperature of 180 °C (Fig. S5†). An et al. also found that CuS nanocrystals could be converted to Cu2S at 220 °C in the presence of OM.33 Such phase transformations of CuS could be explained by the increased reducing ability of OM at high temperatures.
The above results indicated that S/Cu molar ratio and reaction temperature had a complicated effect on the formation of Au–CuS hybrid nanocrystals. Increasing S/Cu molar ratio or reaction temperature was useful to some extent for growing bigger CuS domains on Au seeds, but too large S/Cu molar ratio resulted in the formation of free CuS nanocrystals, while too high growth temperatures led to the degradation of the hybrids back to bare Au nanocrystals and also the reduction of CuS domains to Cu-rich Cu2−xS phases. To obtain Au–CuS hybrid nanocrystals in high yield, the concentration of amine, S/Cu feed molar ratio and growth temperature should be all balanced to control the nucleation and growth dynamics of CuS on Au seeds.
Au–CuS hybrid nanocrystals as Cu-efficient photothermal transduction agents
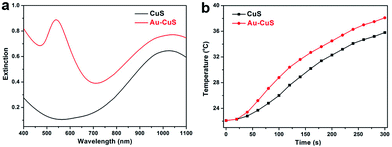
CuS nanocrystals have been widely researched as photothermal transduction agents in photoacoustic imaging and photothermal therapy for cancer owing to their intense NIR absorption.9,11,34–36 However, the concern on the toxicity of Cu ions (the highest safe intake level of Cu is 10 mg daily in adults)37 calls for the amount of CuS used for in vivo applications as little as possible. Therefore, increasing the photothermal transduction capacity per unit of CuS employed is of great importance for practical applications. Growth of CuS on Au to construct hybrid nanostructure provides a feasible solution to this problem, as the local near field on Au surface can enhance the NIR absorption of CuS.1The extinction spectrum of Au–CuS hybrid nanocrystals was not a simple sum of that of Au and CuS component when they are in close contact. In the Au–CuS hybrids, the carrier concentration in CuS domain relative to isolated individual nanocrystals was changed to balance the Fermi level. Moreover, it has been found that the SPR of copper sulfide component could be enhanced by Au due to SPR coupling effect in Au–Cu sulfide hybrids.1 These lead to stronger NIR absorption of Au–CuS hybrids than that of CuS nanocrystals prepared under identical experimental conditions with equal concentration of Cu, as shown in Fig. 5a. In other words, CuS nanocrystals could absorb more NIR photons when they are on Au surface. As a consequence, the aqueous dispersion of Au–CuS hybrids exhibited larger temperature increment relative to CuS dispersion with equal Cu concentration under NIR laser irradiation (Fig. 5b), indicating less Cu was needed to obtain the same imaging or curing effect in photoacoustic imaging and photothermal therapy when Au–CuS hybrids were used instead of plain CuS nanocrystals.
Au–CuS hybrid nanocrystals as template for chemical transformation
The obtained Au–CuS hybrid nanocrystals can also be used as templates to synthesize more complex Au–semiconductor nanostructures such as Au–CuInS2 (Au–CIS) and Au–Cu2ZnSnS4 (Au–CZTS) hybrid nanomaterials. For Au–CIS, purified Au–CuS hybrid nanocrystals were reacted with In(acac)3 and thiourea in the presence of OM at 240 °C. From the TEM image shown in Fig. 6a, it can be seen that the main products obtained were still hybrid nanocrystals. XRD (Fig. 6c) and EDX (Fig. S6a†) analysis indicated these hybrid nanocrystals were composed of Au and tetragonal chalcopyrite CIS. Wu et al. proposed that half of the Cu+ ions would be replaced by In3+ ions and dissolved into the solution to react with the indium and sulfur precursors again in the transformation of CuS to CIS.38 The observed small free CIS nanocrystals in TEM image were probably formed in this process. According to EDX analysis, the molar ratio of Cu/In/S in hybrid nanocrystals was determined to be 1.00/0.90/1.77. In a similar manner, CuS component of the hybrid nanocrystals could be also transformed to CZTS (Fig. 6b), with Cu/Zn/Sn/S ratio of 1.00/0.88/0.43/2.23 (Fig. S6b†). Although the CZTS component was Zn-rich, it still maintained a tetragonal kesterite structure (Fig. 6c). After the chemical transformation to CIS and CZTS with larger refractive index, the SPR band of Au was red-shifted to ∼590 nm, overlapping with the interband absorption of CIS and CZTS, while the NIR extinction peak disappeared due to the low carrier concentrations of CIS and CZTS (Fig. 6d). In these hybrids, the coupling between metal and semiconductor could induce a variety of effects, including SPR enhanced absorption, SPR hot electron transfer and Schottky junction effect, rendering Au/Cu based sulfides hybrid nanocrystals great potential in enhanced solar energy conversion,39 photoelectric response,40 efficient visible-light photocatalyst for hydrogen evolution from water,41,42 as well as for dye degradation.43Conclusions
In summary, we have investigated the critical factors governing the seed-mediated synthesis of Au–CuS hybrid nanocrystals. The effects of the reaction conditions on the nucleation and growth dynamics of CuS on Au seeds were thoroughly studied. High concentrations of amines or large S/Cu molar ratios in the reaction medium would increase the Cu–S reaction rate significantly, resulting in unwanted homogenous nucleation of free CuS nanocrystals. Too high growth temperatures would induce severe inter-particle Ostwald ripening, resulting in bare Au nanocrystals with no CuS deposited, as well as the reduction of CuS domains to Cu-rich Cu2−xS phases. The application potentials of Au–CuS hybrid nanocrystals as Cu-efficient agents for photothermal transduction and as templates for chemical transformation to other Au/Cu-based sulfide nanocrystals have been also investigated. Collectively, we present a systematic study on Au–CuS hybrid nanocrystals, which is expected to foster the extended synthesis of other hybrid nanostructures and study on the applications of these plasmonic materials with interesting optical properties in various fields.Acknowledgements
This work was funded by the “Hundred Talents” program of the Chinese Academy of Sciences, the National Natural Science Foundation of China (Grant No. 21473243 and 51202283).Notes and references
- X. G. Ding, C. H. Liow, M. X. Zhang, R. J. Huang, C. Y. Li, H. Shen, M. Y. Liu, Y. Zou, N. Gao, Z. J. Zhang, Y. G. Li, Q. B. Wang, S. Z. Li and J. Jiang, J. Am. Chem. Soc., 2014, 136, 15684–15693 CrossRef CAS PubMed.
- X. Liu, C. Lee, W. C. Law, D. W. Zhu, M. X. Liu, M. Jeon, J. Kim, P. N. Prasad, C. H. Kim and M. T. Swihart, Nano Lett., 2013, 13, 4333–4339 CrossRef CAS PubMed.
- T. R. Gordon and R. E. Schaak, Chem. Mater., 2014, 26, 5900–5904 CrossRef CAS.
- X. C. Ye, D. R. Hickey, J. Y. Fei, B. T. Diroll, T. J. Paik, J. Chen and C. B. Murray, J. Am. Chem. Soc., 2014, 136, 5106–5115 CrossRef CAS PubMed.
- S. Ghosh, M. Saha, S. Paul and S. K. De, Nanoscale, 2015, 7, 18284–18298 RSC.
- L. A. Lane, X. M. Qian and S. M. Nie, Chem. Rev., 2015, 115, 10489–10529 CrossRef CAS PubMed.
- A. M. Gobin, M. H. Lee, N. J. Halas, W. D. James, R. A. Drezek and J. L. West, Nano Lett., 2007, 7, 1929–1934 CrossRef CAS PubMed.
- D. K. Kim, S. J. Park, J. H. Lee, Y. Y. Jeong and S. Y. Jon, J. Am. Chem. Soc., 2007, 129, 7661–7665 CrossRef CAS PubMed.
- G. Ku, M. Zhou, S. L. Song, Q. Huang, J. Hazle and C. Li, ACS Nano, 2012, 6, 7489–7496 CrossRef CAS PubMed.
- X. Liu, W. C. Law, M. Jeon, X. L. Wang, M. X. Liu, C. H. Kim, P. N. Prasad and M. T. Swihart, Adv. Mater., 2013, 2, 952–957 CAS.
- Y. B. Li, W. Lu, Q. Huang, M. Huang, C. Li and W. Chen, Nanomedicine, 2010, 5, 1161–1171 CrossRef CAS PubMed.
- C. M. Hessel, V. P. Pattani, M. Rasch, M. G. Panthani, B. Koo, J. W. Tunnell and B. A. Korgel, Nano Lett., 2011, 11, 2560–2566 CrossRef CAS PubMed.
- Q. W. Tian, F. R. Jiang, R. J. Zou, Q. Liu, Z. G. Chen, M. F. Zhu, S. P. Yang, J. L. Wang, J. H. Wang and J. Q. Hu, ACS Nano, 2011, 5, 9761–9771 CrossRef CAS PubMed.
- Y. X. Zhao, H. C. Pan, Y. B. Lou, X. F. Qiu, J. J. Zhu and C. Burda, J. Am. Chem. Soc., 2009, 131, 4253–4261 CrossRef CAS PubMed.
- M. Kanehara, H. Arakawa, T. Honda, M. Saruyama and T. Teranishi, Chem.–Eur. J., 2012, 18, 9230–9238 CrossRef CAS PubMed.
- Y. Xie, L. Carbone, C. Nobile, V. Grillo, S. D'Agostino, F. D. Sala, C. Giannini, D. Altamura, C. Oelsner, C. Kryschi and P. D. Cozzoli, ACS Nano, 2013, 7, 7352–7369 CrossRef CAS PubMed.
- S. W. Hsu, C. Ngo and A. R. Tao, Nano Lett., 2014, 14, 2372–2380 CrossRef CAS PubMed.
- D. X. Zhu, A. W. Tang, H. H. Ye, M. Wang, C. H. Yang and F. Teng, J. Mater. Chem. C, 2015, 3, 6686–6691 RSC.
- H. Y. Lin, Y. F. Chen, J. G. Wu, D. I. Wang and C. C. Chen, Appl. Phys. Lett., 2006, 88, 161911 CrossRef.
- J. T. Zhang, Y. Tang, K. Lee and M. Ouyang, Science, 2010, 327, 1634–1638 CrossRef CAS PubMed.
- Q. Zhao, M. W. Ji, H. M. Qian, B. S. Dai, L. Weng, J. Gui, J. T. Zhang, M. Ouyang and H. S. Zhu, Adv. Mater., 2014, 26, 1387–1392 CrossRef CAS PubMed.
- J. Yang, H. I. Elim, Q. B. Zhang, J. Y. Lee and W. Ji, J. Am. Chem. Soc., 2006, 128, 11921–11926 CrossRef CAS PubMed.
- J. S. Lee, E. V. Shevchenko and D. V. Talapin, J. Am. Chem. Soc., 2008, 130, 9673–9675 CrossRef CAS PubMed.
- Y. Zhang, X. Wang, S. Y. Song, D. P. Liu and C. Wang, CrystEngComm, 2012, 14, 7552–7555 RSC.
- R. D. Corato, A. Quarta, P. Piacenza, A. Ragusa, A. Figuerola, R. Buonsanti, R. Cingolani, L. Manna and T. Pellegrino, J. Mater. Chem., 2008, 18, 1991–1996 RSC.
- N. E. Motl, J. F. Bondi and R. E. Schaak, Chem. Mater., 2012, 24, 1552–1554 CrossRef CAS.
- X. G. Ding, Y. Zou and J. Jiang, J. Mater. Chem., 2012, 22, 23169–23174 RSC.
- Y. H. Kim, K. Y. Park, D. M. Jang, Y. M. Song, H. S. Kim, Y. J. Cho, Y. Myung and J. Park, J. Phys. Chem. C, 2010, 114, 22141–22146 CAS.
- J. W. Thomson, K. Nagashima, P. M. Macdonald and G. A. Ozin, J. Am. Chem. Soc., 2011, 133, 5036–5041 CrossRef CAS PubMed.
- H. Zhang, Y. Q. Zhang, J. X. Yu and D. R. Yang, J. Phys. Chem. C, 2008, 112, 13390–13394 CAS.
- M. X. Liu, X. Z. Xue, C. Ghosh, X. Liu, Y. Liu, E. P. Furlani, M. T. Swihart and P. N. Prasad, Chem. Mater., 2015, 27, 2584–2590 CrossRef CAS.
- Y. P. Du, Z. Y. Yin, J. X. Zhu, X. Huang, X. J. Wu, Z. Y. Zeng, Q. Y. Yan and H. Zhang, Nat. Commun., 2012, 3, 1177 CrossRef PubMed.
- L. An, P. P. Zhou, J. Yin, H. Liu, F. J. Chen, H. Y. Liu, Y. P. Du and P. X. Xi, Inorg. Chem., 2015, 54, 3281–3289 CrossRef CAS PubMed.
- Q. W. Tian, M. H. Tang, Y. G. Sun, R. J. Zo, Z. G. Chen, M. F. Zhu, S. P. Yang, J. L. Wang, J. H. Wang and J. Q. Hu, Adv. Mater., 2011, 23, 3542–3547 CrossRef CAS PubMed.
- A. Riedinger, T. Avellini, A. Curcio, M. Asti, Y. Xie, R. Y. Tu, S. Marras, A. Lorenzoni, S. Rubagotti, M. Iori, P. C. Capponi, A. Versari, L. Manna, E. Seregni and T. Pellegrino, J. Am. Chem. Soc., 2015, 137, 15145–15151 CrossRef CAS PubMed.
- M. Zhou, S. L. Song, J. Zhao, M. Tian and C. Li, J. Mater. Chem. B, 2015, 3, 8939–8948 RSC.
- L. R. Guo, I. Panderi, D. D. Yan, K. Szulak, Y. J. Li, Y. T. Chen, H. Ma, D. B. Niesen, N. Seeram, A. Ahmed, B. F. Yan, D. Pantazatos and W. Lu, ACS Nano, 2013, 7, 8780–8793 CrossRef CAS PubMed.
- X. J. Wu, X. Huang, X. Y. Qi, H. Li, B. Li and H. Zhang, Angew. Chem., Int. Ed., 2014, 53, 8929–8933 CrossRef CAS PubMed.
- Y. T. Yen, C. W. Chen, M. Fang, Y. Z. Chen, C. C. Lai, C. H. Hsu, W. C. Wang, H. Lin, C. H. Shen, J. M. Shieh, J. C. Ho and Y. L. Chueh, Nano Energy, 2015, 15, 470–478 CrossRef CAS.
- B. K. Patra, A. Shit, A. K. Guria, S. Sarkar, G. Prusty and N. Pradhan, Chem. Mater., 2015, 27, 650–657 CrossRef CAS.
- E. Ha, L. Y. S. Lee, J. C. Wang, F. H. Li, K. Y. Wong and S. C. E. Tsang, Adv. Mater., 2014, 26, 3496–3500 CrossRef CAS PubMed.
- X. L. Yu, A. Shavel, X. Q. An, Z. S. Luo, M. Ibáñez and A. Cabot, J. Am. Chem. Soc., 2014, 136, 9236–9239 CrossRef CAS PubMed.
- P. S. Dilsaver, M. D. Reichert, B. L. Hallmark, M. J. Thompson and J. Vela, J. Phys. Chem. C, 2014, 118, 21226–21234 CAS.
Footnotes |
| † Electronic supplementary information (ESI) available: TEM images of Au–CuS hybrid nanocrystals synthesized with different amounts of oleylamine, XRD pattern of CuS synthesized at 100 °C, Au–Cu2−xS hybrid synthesized at 180 °C, FTIR and EDX results. See DOI: 10.1039/c6ra02425d |
| ‡ These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2016 |