Green synthesis of highly fluorescent AuNCs with red emission and their special sensing behavior for Al3+†
Dan Li,
Zhenhua Chen*,
Tiezhu Yang,
Hao Wang,
Nan Lu and
Xifan Mei*
Liaoning Medical University, Jinzhou, 121001, People's Republic of China. E-mail: zhchen561@yahoo.com; meixifan1971@163.com
First published on 12th February 2016
Abstract
Facile methods were fabricated for preparation of highly fluorescent AuNCs with red emission using L-cysteine. Water soluble and aggregation induced AuNCs have been prepared by the employment of the same ligand. The emission wavelength of the as-prepared water soluble AuNCs can be tuned by simply adjusting the amounts of the reactants. Interestingly, it is further found that the fluorescence of AuNCs which has been quenched by Pb2+ can be recovered by Al3+. This provides a new approach for ‘turn-on’ detection of Al3+ in aqueous solution. A mechanism for sensing that is related to the residual reducing reagent (THPC) has been proposed base on the investigation of various related nanoclusters-based systems.
Introduction
Gold nanoclusters (AuNCs) have been developed as promising materials for various applications in recent years, due to their unique optical, electronic, and low toxic properties.1–4 They exhibit great performance in the area of catalysis, sensing and medicine.5–8 Though great attention has been directed to the application of AuNCs, the synthesis of AuNCs with bright fluorescence using cost effective protection and green ligands is still a challenge.9 AuNCs with red emission have been obtained by using large molecules such as bovine serum albumin (BSA) protein, glutathione (GSH) peptide, and DNA.10–12 However, each of them describes disadvantages. For instance, besides the expensive price and low temperature storage requirement, BSA protein and GSH peptide require elevated temperature for the synthesis.10,13 Additionally, protein protected AuNCs are very difficult to be separated; DNA protected AuNCs require complicate procedures for the preparation (therefore the procedures are difficult to be repeatable).12 Meanwhile, small molecules such as (dihydrolipoic acid) DHLA have been employed for the preparation of AuNCs, which require simple procedures at room temperatures, but the quantum yield is quite low.14,15 As well as this, 11-MUA (11-mercaptoundecanoic acid, 98%) (500 mg, ca. 400 EUR, Sigma-Aldrich) has been successfully applied for synthesis of AuNCs with red emission,16 but the ligand is very expensive. Moreover, the excitation wavelength (ca. 300 nm, not visible) is quite short, which is not environment-friendly. Compared to these ligands, the amino acids of native protein that exists in people will provide more advantages. They are not only cost effective, but with low toxic, biocompatible, and environment-friendly properties. Until now, common amino acids of native protein such as L-histidine, L-cysteine, methionine, and L-proline have been designed as templates for synthesis of fluorescent AuNCs with blue/green emission.17–20 However, when these amino acids are used as templates, no fluorescent AuNCs with red emission (more environmentally friendly) have been obtained yet. For example, L-cysteine is a market available amino acid (>99%, 1 kg, ca. 14 EUR, alibaba), which can perform as a green ligand if AuNCs with red emission can be successfully obtained. AuNCs with green emission have been obtained by using L-cysteine, but red emitted AuNCs hasn't been fabricated yet.18It has been reported that the composition of L-cysteine may play an important role for the culture of BSA protected AuNCs with red emission.21,22 In that case, tryptophan or some other amino acid may act as a weak reduction reagent. It is worth simulating a similar culture environment like BSA or other ligands where AuNCs with red emission can be found.10 However, the reduction composition such as tryptophan of BSA is not well applied in water as a reductant by itself. When tryptophan was separated from protein, it didn't exhibit the same performance. Thus, other weak but independent reducing reagent should be found.23 Then, AuNCs with red emission will possibly be obtained using L-cysteine as a protection ligand.
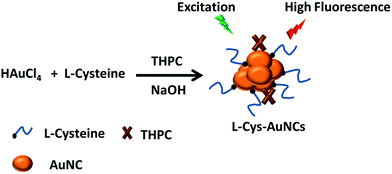
In the case of sensing, the detection of Al3+ is of great importance. If high concentration of Al3+ is present in the environment, it will cause serious disease.24 Fluorescent sensors have attracted interest for the detection of Al3+.25–27 Some organic sensors have been designed for detecting Al3+, but these systems may bring new toxic media for aqueous solution.28–31 The green sensing materials such as noble metal nanoclusters are expected. Recently, two methods for determination of Al3+ using nanoclusters have been reported. The PEGylated MSA-AgAuNCs can be used for sensing of Al3+ in aqueous solution.32 However, only 3-fold fluorescence enhancement was observed upon the addition of 2 mM of Al3+. The other report found that the tyrosine-stabilized fluorescent AuNCs could be used for sensing of Al3+ though less than one fold enhancement was obtained.33 In this work, we develop a facile method for synthesis of water soluble L-cysteine capped AuNCs (L-Cys-AuNCs) with bright red emission. The emission wavelength can be tuned by simply adjusting the amounts of the reactants. The protocol for synthesis is described in Scheme 1, which is similar like the culture of BSA protected AuNCs with red emission. However, our protocols are much more cost effective and the product is with high yield. It is proposed that tetrakis(hydroxymethyl)phosphonium chloride (THPC) may play a similar role like tryptophan or other amino acid with reducing ability of BSA.
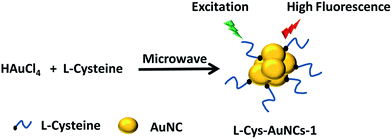
At the same time, a microwave assisted method was employed for the fabrication of L-cysteine protected AuNCs with red emission (L-Cys-AuNCs-1) in the absence of additional reducing reagent. However, the as prepared AuNCs are not water soluble. Therefore, the red emission is relied on ‘aggregation induced fluorescence’.34 The protocol for the formation of L-Cys-AuNCs-1 is demonstrated in Scheme 2.
Furthermore, the water soluble L-Cys-AuNCs were fabricated for turn-on sensing of Al3+ by a special process based on the recovery of the fluorescence, which has been quenched by Pb2+. To the best of our knowledge, when a common amino acid of native protein is used as a capping ligand, this is the first work that obtains AuNCs with red emission. Also, we report a first strategy for sensing of Al3+ based on the recovery of the fluorescence of noble metal nanoclusters that has been quenched by Pb2+.
Results and discussion
To confirm the formation of the water soluble L-Cys-AuNCs, we use UV-vis, fluorescence spectra, the comparison of photograph under dark and UV light, as well as XPS to investigate the properties (see Fig. 1). It can be seen from Fig. 1a that the synthesized L-Cys-AuNCs display a broad absorption band with a weak peak at ca. 450 nm. Just like previous AuNCs, ultra-small sizes of L-Cys-AuNCs demonstrate molecular-like properties, which are different from relatively large size gold nanoparticles. Thus, it is not surprising there is no obvious surface plasma resonance peak at 520 nm in the visible absorption region for L-Cys-AuNCs. The inset picture in Fig. 1a describes the as prepared sample under dark and UV light (365 nm UV lamp, 0.7 mW). Bright red fluorescence can be observed. On the other hand, no obvious red fluorescence can be seen by eyes using our UV lamp for some other small molecular protected AuNCs such as DHLA-AuNCs, which was reported by previous papers, though there was no big difference for their maxim excitation wavelength.14,35 Meanwhile, the as-prepared L-Cys-AuNCs exhibit strong red fluorescence with a maximum emission peak at ca. 700 nm under 493 nm excitation wavelength, see Fig. 1b. These results reveal the formation of L-Cys-AuNCs with bright red fluorescence. The fluorescence quantum yield was calculated at 4.3% using acridine red (Qf = 0.51) as a reference. Meanwhile, the emission wavelength can be tuned by adjusting the amounts of the reagents while fixing other conditions. The fluorescence emission spectra for the samples with various wavelengths are described in Fig. S1–S3.† It can be seen from Fig. S1 and S2† that the emission wavelength demonstrates red shift as a function of L-cysteine or HAuCl4. With higher concentration of either L-cysteine or HAuCl4, the forward reaction for the formation of AuNCs can proceed much faster, which enable the formation of larger sizes of AuNCs. Therefore the presence of larger size of AuNCs for the reaction product will induce the red shift of the emission wavelength. On the contrary, in the presence of more amounts of THPC, the emission wavelength demonstrates blue shift, see Fig. S3.† Notwithstanding, there is no big difference for the emission wavelength when 3–10 μL of THPC is used. Higher concentrations of the reactant were supposed to increase the reaction rate, which should cause similar effect like higher concentration of HAuCl4 and L-cysteine, but the blue shifts of the emission wavelength indicated the formation of smaller size of AuNCs. It is concluded that THPC not only acts as a reducing reagent but also it is capable of playing a role as a complexant to inhibit the reaction rate, which will be further discussed for the mechanism of sensing.A further inspection of the composition of L-Cys-AuNCs was performed using XPS (see Fig. 1c and d). The valence states of the Au atoms in L-Cys-AuNCs were studied. The XPS analysis in Fig. 1c reveals the Au 4f7/2 binding energy for L-Cys-AuNCs of 84.1 eV, locates between those of 84.0 eV for Au(0) and 86.0 eV for Au(I). This finding suggests that Au(0) and Au(I) species coexist in L-Cys-AuNCs. However, the binding energy describes blue shift compared to AuNCs in most papers.36 Since the binding energy locates quite near Au(0), it indicates Au(0) plays a more important role. According to the papers, if the ratio of Au(I) was reduced to Au(0) by the introduction of NaBH4, the fluorescence intensity would be significantly decreased while Au(0) played a major ratio.37 Notwithstanding, the fluorescence is still strong for our L-Cys-AuNCs though the ratio Au(0) plays a more important role. As a result, it can be concluded that AuNCs can still exhibit bright fluorescence even though Au(I) hasn't played the major role in our case. As well as this, it can be further observed from the XPS survey (Fig. 1d) that the presence of S, N and C belongs to the L-cysteine capping ligand. It also indicates the as prepared AuNCs are protected by L-cysteine.
As well as THPC, weather water soluble AuNCs with red emission could be obtained by using other reducing reagents was investigated, which had been often applied for the preparation of noble metal nanoclusters. For the formation of water soluble L-Cys-AuNCs with our conditions, NaOH is used to dissolve the aggregates that has been formed by the complex of L-cysteine and gold precursor. Then, various reducing reagents have been investigated, but no fluorescent AuNCs can be found. The typical fluorescent emission spectra for the products are described in Fig. S4.† It can be seen that no fluorescence but the noise signal can be detected by the instrument. At the same time, no fluorescent AuNCs can be obtained in the absence of reducing reagent either. If NaOH is not used to dissolve the aggregates between L-cysteine and gold precursor, there will be no fluorescence when no further treatment is performed, see Fig. S5a and b.† According to the paper, L-cysteine is capable of acting as a reducing reagent itself to reduce Au3+ as Au0,18 but no fluorescent AuNCs are found at the current condition. It can be concluded that the ambient condition is not suitable for the formation of Au0 to occur spontaneously. Furthermore, microwave assistance was found as a successful strategy for the formation of L-Cys-AuNCs-1 with aggregation induced red emission. The characterization for L-Cys-AuNCs-1 including photograph under visible light and UV light, UV-vis, fluorescence spectra, as well XPS are described in Fig. S5–S7.† Fig. S5† describes the photograph of the samples that obtained without (Fig. S5a and b†) and with (Fig. S5c and d†) the assistance of microwave heating. Without the assistance of microwave heating, it can be seen that the as formed colloid describe pale white colour under visible light, but no fluorescence can be seen under UV light (365 nm UV lamp, 0.7 mW). On the other hand, the as-prepared sample after microwave heating was pale white under visible light. Meanwhile, red emission under UV light can be observed, which indicates that highly fluorescent AuNCs protected by L-cysteine (L-Cys-AuNCs-1) are formed. The maximum fluorescence intensity of L-Cys-AuNCs-1 can be obtained at 360 nm excitation wavelength, which is in consistent with the UV-vis absorption spectrum (373 nm) (Fig. S6a†). As demonstrated in Fig. S6b,† the maxim emission wavelength is located at 605 nm with a Stokes shift of 302 nm. The fluorescence quantum yield is calculated at 3.1% using acridine red (Qf = 0.51) as a reference. To further confirm the formation of L-Cys-AuNCs-1, XPS was performed. It can be seen in Fig. S7† that the Au 4f7/2 peak being at 84.5 eV indicates the valence state for Au is located between 0 and 1, which is similar to other AuNCs in most papers.37–39 As well as this, the XPS survey indicates the presence of S, C and N, which reveals the presence of the L-cysteine ligand. As summary, it can be concluded that the as selected reducing reagent (THPC) is of key importance for the formation of water soluble L-Cys-AuNCs at the current condition, but no additional reducing reagent is required for the formation of L-Cys-AuNCs-1 with aggregation induced emission.
Next, our samples were studied for the application as a sensor for cations. First, we performed a study for the selectivity of our L-Cys-AuNCs based sensor (S0) toward various cations, examining the properties of the designed system, see Fig. 2. As indicated in Fig. 2a, the presence of Ag+ and Pb2+ resulted in quenching of the fluorescence of S0 sensor. Meanwhile, an enhancement in fluorescence was found for the sensor in the presence of Al3+. In contrast, no obvious signal change occurred in the presence of other cations including Ag+, Ba2+, Ca2+, Co2+, Cu2+, Fe2+, Na+, Ni2+, NH4+ and Cd2+.
Additionally, the sensing behaviours for Fe3+ and Cr3+ of S0 sensor are compared, which are described in Fig. S8.† No enhancement is observed like Al3+ though they are with the same valence states. It can be concluded that the as prepared sensor might be applied for sensing of Al3+. However, according to Fig. 2b, the enhancement didn't describe great advantages over the Al3+ sensors in previous papers.32 The fluorescence intensity only described 1–2 times enhancement and the selectivity was not excellent enough. Therefore, further fabrication was required for realizing better performance of the sensor.
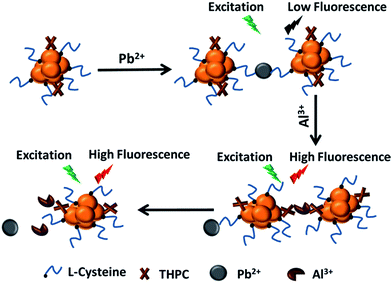
We try to investigate whether Al3+ can recover the fluorescence that has been quenched by Ag+ or Pb2+. It is found that the fluorescence will no longer be enhanced in the presence of Ag+. Based on this phenomenon, it is concluded that some chemical reaction may happen so that the decrease of the fluorescence is irreversible. For instance, Ag+ can be reduced to Ag0 on the surface of Au0 by the residual adsorbed THPC. After the formation of Au–Ag, the fluorescence will be quenched due to the formation of bigger particles. In the presence of Al3+, the particle cannot be recovered as fluorescent AuNCs.
On the other hand, it was found the presence of Al3+ can not only describe enhancement for the fluorescence of L-Cys-AuNCs based sensor, but also it can be used to recover the fluorescence that has been quenched by Pb2+. Then, it was expected that much greater enhancement factor would be demonstrated if the fluorescence for the mixture of Pb2+ and L-Cys-AuNCs was considered as a sensing system. Then, the new sensor would be produced in the presence of Pb2+ and L-Cys-AuNCs (S1). As expected, we observed a much more significant enhancement factor of fluorescence in the presence of Al3+, see Fig. 3. Fig. 3a describes the fluorescence emission spectra for the sensor (S0) (L-Cys-AuNCs), L-Cys-AuNCs in the presence of Pb2+ (S1), S1 in the presence of Al3+ (S1-Al), as well as S0 in the presence of Al3+ (S0-Al). Meanwhile, the comparison of the enhancement was demonstrated in Fig. 3b. For S0 sensor, the enhancement was ca. 1–2 times for Al3+ (S0-Al). On the other hand, if we consider S1 as the sensor, the fluorescence shows more than 14 times enhancement for sensing of Al3+ (S1-Al). This indicates that a highly enhancement strategy for sensing of Al3+ will be presented by S1 compared to S0 sensor. The enhancement is also much higher than previous papers using other nanoclusters as sensors.32,33 Furthermore, the corresponding pictures for the mixture of S0, S1, S1-Al, as well as S0-Al under dark and UV light are described in Fig. 3c and d respectively. It can be observed from Fig. 3d that there is no big difference for the comparison between S0 and S0-Al. On the other hand, obvious contrast can be seen for the comparison between S1 and S1-Al. As a result, advantages can be demonstrated by the new proposed sensing system in consideration of the enhancement and the contrast in the picture.
Fig. 4 displays TEM images of the as-prepared L-Cys-AuNCs (S0) as well as S1, S1-Al, and S0-Al. It can be seen from Fig. 4a that the average diameter of L-Cys-AuNCs (S0) is smaller than 1 nm, as judged from image analysis of the particles. In the presence of Pb2+ (S1), the sizes are larger than 1 nm (Fig. 4b). This indicated the partly aggregation (soft) of the small nanoclusters, which could be caused by the complex between L-cysteine ligand and Pb2+. Notwithstanding, the aggregation is not so significant that they cannot be reversible based on the TEM image. These phenomena inspire us to develop a sensor based on the mixture of Pb2+ and L-Cys-AuNCs (S1). The fluorescence for the mixture of Pb2+ and L-Cys-AuNCs may possibly be recovered since their binding is not very strong just by complexing. In the presence of additional Al3+ for S1 (S1-Al), the sizes are smaller than 1 nm. In the presence of Al3+ for S0 (S0-Al), the sizes of the particles are also smaller than 1 nm according to the evaluation from the TEM image. It can be clearly observed from TEM that the sizes for the particles of S0 (Fig. 4a), S1-Al (Fig. 4c) and S0-Al (Fig. 4d) are small enough to describe quantum effect as well as fluorescence.
According to the TEM images, the aggregation induced enhancement is not the reason for sensing since no significant aggregation has been observed. Then, we further investigated various related systems for studying the phenomena of sensing. Since THPC was used as a reducing reagent for the preparation of water soluble L-Cys-AuNCs, it is worth investigating whether the residual THPC plays an important role for the fluorescence enhancement.
Initially, we study the sensing behaviour of L-Cys-AuNCs-1 where no THPC is present. The sensing behaviours including the fluorescence emission spectra in the presence of various cations and the selectivity were described in Fig. S9.† It can be seen that the fluorescence is not only quenched in the presence of Ag+, but also it is quenched by some other heavy cations such as Fe3+ and Cu2+. On the other hand, there is no big difference for the intensity in the presence of Pb2+. If we consider L-Cys-AuNCs-1 in the presence of Pb2+ as S1, the fluorescence cannot be enhanced by further addition of Al3+ (S1-Al). It can be concluded that the sensing behaviour for L-Cys-AuNCs-1 is quite different from that for L-Cys-AuNCs. The difference for the formation of L-Cys-AuNCs-1 is that no NaOH and THPC were used. Thus, it is worth investigating some other similar systems where both NaOH and THPC are employed. Initially, we perform similar sensing processes for AuNCs protected by L-cysteine with using less amounts of THPC (3 μL) as reducing reagent (L-Cys-AuNCs-2). Meanwhile, several types of other metal nanoclusters that use both NaOH and THPC are also studied. The sensing behaviours for Pb2+, Al3+, the mixture of Pb2+ and Al3+ were studied before and after the separation of the residual THPC. The results are described in Fig. S10–S14.† Based on Fig. S3† and 1b, it is found that the emission wavelength for L-cysteine protected AuNCs doesn't describe significant difference when 3–10 μL of THPC is used as the reducing reagent. Thus, it can be concluded there is no big difference for the as formed AuNCs except the amounts of the adsorbed THPC. The selectivity study for L-Cys-AuNCs-2 is described in Fig. S10.† It can be seen that the fluorescence can still be quenched in the presence of Ag+ (Fig. S10a†). However, in the presence of Pb2+, the fluorescence intensity hasn't significantly decreased. Meanwhile, if we consider S0 in the presence of Pb2+ as another sensor (S1), the fluorescence cannot be enhanced by the further addition of Al3+. For the L-Cys-AuNCs-2 sensor, only 3 μL of THPC was used. Then, less amounts of residual THPC would be adsorbed on the surface of AuNCs. As a result, it can been revealed that the residual adsorbed THPC would influence the fluorescence behaviour of the L-Cys-AuNCs in the presence of Al3+ since different sensing behaviors were demonstrated for L-Cys-AuNCs (10 μL), L-Cys-AuNCs-1 (0 μL) and L-Cys-AuNCs-2 (3 μL) when various amounts of THPC had been used.
After that, several kinds of AuNCs that were prepared using THPC as reducing reagent were investigated. The synthesis procedures are described by ESI.† The effect of the application of THPC was studied before and after the separation. First, Fig. S11† describes the fluorescence emission spectra of L-Cys-AuNCs in the presence of cations before and after the separation of THPC. After separation, it can be observed from Fig. S11b† that the presence of Al3+ will partly quench the fluorescence of the purified L-Cys-AuNCs. The presence of Pb2+ will completely quench the fluorescence of the purified L-Cys-AuNCs. After further addition of Al3+ to the purified L-Cys-AuNCs-Pb2+ system, the fluorescence cannot be recovered. It can be seen the sensing behaviour for Al3+ can't be described when THPC is separated. In order to further confirm the importance of the residual THPC for sensing of Al3+. Other AuNCs protected by DPA and 11-MUA were synthesized using THPC as reducing reagent. The effects of the residual THPC were also studied before and after the separation, see Fig. S12 and S13.† It can be seen from Fig. S12a† that the fluorescence of DPA-AuNCs is enhanced in the presence of Al3+. In the presence of Pb2+, the fluorescence was also quenched. Furthermore, the fluorescence of the DPA-AuNCs-Pb2+ system was recovered by the addition of Al3+. The phenomena are quite similar to our L-Cys-AuNCs-Pb2+ (S1) sensing system without the purification of the residual THPC (Fig. S11a†). Also, similarly, after the separation of THPC, no fluorescence enhancement can be achieved by Al3+ (Fig. S12b†). Additionally, the 11-MUA-AuNCs system was studied (Fig. S13†). It can be seen that similar phenomena happens as DPA-AuNCs and L-Cys-AuNCs based system. All the AuNCs prepared using THPC as reduction reagent have exhibited similar sensing behaviours for Al3+ when the residual THPC is not separated. It can be concluded that the residual THPC plays a vital role for our sensing strategy. On the other hand, we synthesized two types of AuNCs using BSA and GSH as the protection ligand without the introduction of THPC (Fig. S14†). When we used the AuNCs-Pb2+ system as the sensor, no fluorescence enhancement would be observed in the presence of Al3+. Herein, it was also confirmed that the sensing strategy couldn't be employed without using THPC as a reducing reagent. According to the results, it can be concluded that the application of THPC as a reducing reagent plays an important role in sensing of Al3+. Based on the above phenomena, the sensing process was described in Scheme 3. In the presence of Pb2+, the complex between Pb2+ and the capping ligand (–COO) induce the quenching of the fluorescence due to the formation of ‘soft’ aggregation. However, since Al3+ has a higher binding ability with –COO in this case, after further addition of Al3+, Pb2+ will less significantly bind with the capping ligand (–COO). Meanwhile, the complex between Al3+ and the adsorbed THPC can enhance the fluorescence of the L-Cys-AuNCs-Pb2+ (S1), which is similar to the enhancement of S0. As a result, the further addition of Al3+ enables the enhancement of the fluorescence that has been quenched by Pb2+. This phenomenon can be employed for detection of Al3+.
To investigate the selectivity for S1 sensing system, other cations including Ag+, Ba2+, Ca2+, Co2+, Cu2+, Fe3+, Na+, Ni2+, NH4+, Pb2+ and Cd2+ on the influence of the fluorescence at the same sensing conditions are described, see Fig. 5. The comparison for sensing of Fe3+ and Cr3+ are described in Fig. S15.† The results in Fig. 5b and S15b† indicate the selectivity of the S1 sensor exhibits more excellent advantages over the S0 system and previous papers.32,33 The corresponding pictures for the sensing systems (Fig. 5) under dark and UV light are described in Fig. S16.† It can be seen that fluorescence can only be observed for S1 sensor in the presence of Al3+. On the other hand, for S1 sensor in the presence of other cations, no fluorescence can be seen under the UV lamp. The high efficiency and excellent selectivity of the S1 system toward Al3+ suggests that our strategy is promising for the detection of selected Al3+ in water samples.
 | ||
| Fig. 5 Fluorescence spectra (a) of L-Cys-AuNCs-Pb2+ sensor (S1) in the presence of various metal ions and the corresponding selectivity (b) of the S1 sensor (Fe indicates Fe2+). | ||
Furthermore, the selectivity behaviour for S1 sensor to Al3+ over other cations with different pH values is investigated. The results are described in Fig. S17.† It can be seen the sensing behaviours are quite similar from pH 4 to pH 8. However, at pH 10, the enhancement for Al3+ was relative low. This is possibly because the binding between Al3+ and THPC tend to be destroyed at high pH values.
Additionally, the titration behaviour of the as prepared sensor was studied, see Fig. 6. The fluorescence turn-on spectra in Fig. 6a reveal that the fluorescence intensity of S1 sensor increased upon increasing the concentration of Al3+. Fig. 6b presents the fluorescence enhancements (F/F0) plotted with respect to the concentrations of the Al3+. It is observed polynomial relationships in the ranges from 25 to 400 μM for Al3+ (R2 = 0.998; Y = −0.0000739X2 + 0.0706X + 0.872).
 | ||
| Fig. 6 Fluorescence spectra (a) of S1 probe in the presence of different concentrations of Al3+; fluorescence intensity enhancement (b) as a function of the concentration. | ||
As well as Al3+, we found some rare earth ions could be used for recovering the fluorescence that has been quenched by Pb2+ too. Fig. 7 describes the fluorescence of S1 in the presence of Al3+ as well as S1 in the presence of various rare earth cations. It can be seen that the presence of most rare earth ions with trivalent ions like Al3+ enable the partly recovery of the fluorescence for S1 sensing system. This is different from Fe3+ and Cr3+ since heavy metal ions tend to be quenching rather than enhancing the fluorescence of nanoclusters. Therefore, it indicated the presence of rare earth ions with comparable amounts might interfere with the detection of Al3+. However, the concentration of rare earth ions in natural water is too little to compare to Al3+. Still, the proposed method can be applied in the environment. On the other hand, it can be concluded that the system might be further applied in other area based on fluorescence enhancement behaviours by the combination of rare earth.
Experimental
Reagents and instruments
All the materials for the experiments were commercially available and used as received without further purification. Tetrachloroauric-III-acid hydrate (HAuCl4·3H2O, ≥99.9%), L-cysteine (≥97%), sodium hydroxide (NaOH, ≥98%) and tetrakis(hydroxymethyl)phosphonium chloride solution (THPC, 80% in H2O) and metal salts that will be used for investigating the sensing behaviours of cations were purchased from Sigma Aldrich. Metal salts are including silver nitrate (AgNO3, >99%), barium nitrate (Ba(NO3)2, ≥99%), calcium nitrate tetrahydrate (Ca(NO3)2, ≥99.0%), cobalt(II) nitrate hexahydrate (Co(NO3)2·6H2O, ≥98%), copper(II) nitrate trihydrate (Cu(NO3)2, 98–103%), iron(II) sulfate heptahydrate (FeSO4, 99%), sodium sulfate anhydrous (Na2SO4, ≥99%), nickel(II) nitrate hexahydrate (Ni(NO3)2·6H2O, ≥98.5%), ammonium chloride (NH4Cl, 99%), lead(II) nitrate (Pb(NO3)2, ≥99%), cadmium nitrate tetrahydrate (Cd(NO3)2·4H2O, ≥99.0%), aluminum nitrate nonahydrate (Al(NO3)3·9H2O, >99%), chromium(III) nitrate nonahydrate (Cr(NO3)3·9H2O, >99%), iron(III) nitrate nonahydrate (Fe(NO3)3·9H2O, 99.85%), yttrium(III) nitrate hexahydrate (Y(NO3)3·6H2O, 99.8%), lanthanum(III) nitrate hexahydrate (La(NO3)3·6H2O, >99%), ytterbium(III) nitrate pentahydrate (Yb(NO3)3·5H2O, 99.9%), erbium(III) nitrate pentahydrate (Er(NO3)3·5H2O, 99.9%), gadolinium(III) nitrate hexahydrate (Gd(NO3)3·6H2O, 99.9%), thulium(III) nitrate pentahydrate (Tm(NO3)3·6H2O, 99.9%), cerium(III) nitrate hexahydrate (Ce(NO3)3·6H2O, >99.9%), were purchased from Sigma Aldrich. All the solvents were of analytical grades. Deionized water was used through the experiment. Fluorescence spectra were recorded on a FS-2 spectrophotometer (SCINCO). Transmission electron microscope (TEM) images were obtained on a Hitachi transmission electron microscope operated at 120 kV. UV-visible (UV-vis) absorption spectra were performed on a UV-1600 spectrometer (Shangdong). X-ray photoelectron spectroscopy (XPS) spectra were obtained on Escalab 250 X-ray photoelectron spectroscope.Preparation of L-Cys-AuNCs
In a typical procedure, 250 μL of HAuCl4·3H2O (1%) and 5 mL of 0.1 M L-cysteine were combined in a 10 mL vial with stirring. Then, white colour aggregates were formed, but no fluorescence could be detected yet. After that, 300 μL of 1 M NaOH was combined to dissolve the aggregates. Subsequently, 10 μL of THPC (80%) was added. The solution was stirred overnight. The aqueous phase turned from colourless to bright yellow. Then, the water soluble AuNCs with red emission that are protected by L-cysteine (L-Cys-AuNCs) are formed. For further application as sensor, the as obtained colloid will be directly used. For separation of the unreacted reactants, 10 mL of ethanol was added to 5 mL of the mixture. The yellow powder with high yield can be obtained after centrifugation at 4000 rpm. After washing the powder by ethanol–water mixture (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) for several times, it can be re-dispersed in water for further applications. For long term storage, the powder can be stored in refrigerator (freezing chamber), which will be stable for more than two years.
1) for several times, it can be re-dispersed in water for further applications. For long term storage, the powder can be stored in refrigerator (freezing chamber), which will be stable for more than two years.
For preparation of L-cysteine protected AuNCs with other wavelength, the conditions are kept same as the typical procedure except the mentioned parameter.
Preparation of L-Cys-AuNCs-1
In a typical procedure, freshly prepared aqueous L-cysteine (5 mL, 0.1 M) were mixed with HAuCl4 (250 μL, 1%) solution in a 250 mL beaker. Under gentle stirring for 5 min, the white colour suspension was transferred to a domestic microwave oven. The irradiation time for heating was set for 30 s. After cooling down, the solution was irradiated for another 30 s. Then, the L-cysteine protected AuNCs (L-Cys-AuNCs-1) that are not soluble in water with bright red emission can be obtained. The L-Cys-AuNCs-1 can be easily precipitated and separated by centrifugation (4000 rpm).In a typical experiment process, a 1.0 cm quartz cell was used for the fluorescence measurement. The sample was excited at 493 nm, and the emission was collected from 550 to 800 nm. The intensity was read at ca. 700 nm. 500 μL of L-Cys-AuNCs solution and 1500 μL of 1 mM of phosphate buffers buffer (pH = 7) containing various concentrations of Al3+ were added into the quartz cell. For the selectivity experiment, other cations (400 μM) were studied in the place of Al3+. For other sensing study, other AuNCs are investigated in the place of L-Cys-AuNCs with the same conditions.
Conclusions
We found facile and green approaches for the synthesis of water-soluble L-Cys-AuNCs with red emission using a common amino acid of native protein as a capping ligand. Meanwhile, a microwave assisted method for fabrication of L-Cys-AuNCs-1 with aggregation induced red emission was proposed. The water soluble L-Cys-AuNCs can be used as probes for detection of Al3+. Furthermore, by the combination of Pb2+, the enhancement describes advantages over the previous Al3+ sensor in the papers. The system can be further applied in other area based on fluorescence enhancement behaviours by the combination of rare earth ions.Acknowledgements
This work is supported by Excellent Talents program of Liaoning Provincial Universities (No. LJQ2013089), National Natural Science Foundation of China (No. 51202199 and No. 81471854), Natural Science Foundation of Liaoning Province (No. 2014022038), Liaoning Medical University Principal Fund (No. XZJJ20130104-01), Liaoning Medical University Principal Fund-Aohong Boze Students Researching Training Program (No. 2014D22) and Liaoning Provincial University Students Researching Training Programs (No. 201210160012).Notes and references
- C. J. Zeng, Y. X. Chen, A. Das and R. C. Jin, J. Phys. Chem. Lett., 2015, 6, 2976–2986 CrossRef CAS PubMed.
- H. Li, L. Li, A. Pedersen, Y. Gao, N. Khetrapal, H. Jonsson and X. C. Zeng, Nano Lett., 2015, 15, 682–688 CrossRef CAS PubMed.
- Y. X. Chen, C. J. Zeng, D. R. Kauffman and R. C. Jin, Nano Lett., 2015, 15, 3603–3609 CrossRef CAS PubMed.
- K. Pyo, V. D. Thanthirige, K. Kwak, P. Pandurangan, G. Ramakrishna and D. Lee, J. Am. Chem. Soc., 2015, 137, 8244–8250 CrossRef CAS PubMed.
- H. B. Chong and M. Z. Zhu, ChemCatChem, 2015, 7, 2296–2304 CrossRef CAS.
- F. Molaabasi, S. Hosseinkhani, A. A. Moosavi-Movahedi and M. Shamsipur, RSC Adv., 2015, 5, 33123–33135 RSC.
- L. L. Wang, J. Qiao, L. Qi, X. Z. Xu and D. Li, Sci. China: Chem., 2015, 58, 1508–1514 CrossRef CAS.
- W. Ge, Y. Y. Zhang, J. Ye, D. H. Chen, F. U. Rehman, Q. W. Li, Y. Chen, H. Jiang and X. M. Wang, J. Nanobiotechnol., 2015, 13, 1–8 CrossRef PubMed.
- S. X. Wang, X. M. Meng, A. Das, T. Li, Y. B. Song, T. T. Cao, X. Y. Zhu, M. Z. Zhu and R. C. Jin, Angew. Chem., Int. Ed., 2014, 53, 2376–2380 CrossRef CAS PubMed.
- J. P. Xie, Y. G. Zheng and J. Y. Ying, J. Am. Chem. Soc., 2009, 131, 888–889 CrossRef CAS PubMed.
- H. Y. Zhang, Q. Liu, T. Wang, Z. J. Yun, G. L. Li, J. Y. Liu and G. B. Jiang, Anal. Chim. Acta, 2013, 770, 140–146 CrossRef CAS PubMed.
- G. Y. Liu, Y. Shao, K. Ma, Q. H. Cui, F. Wu and S. J. Xu, Gold Bull., 2012, 45, 69–74 CrossRef CAS.
- J. Zhang, Y. Yuan, G. L. Liang, M. N. Arshad, H. A. Albar, T. R. Sobahi and S. H. Yu, Chem. Commun., 2015, 51, 10539–10542 RSC.
- L. Shang, L. X. Yang, F. Stockmar, R. Popescu, V. Trouillet, M. Bruns, D. Gerthsen and G. U. Nienhaus, Nanoscale, 2012, 4, 4155–4160 RSC.
- S. H. Xu, P. P. Liu, Q. W. Song, L. Wang and X. L. Luo, RSC Adv., 2015, 5, 3152–3156 RSC.
- H. C. Chang, Y. F. Chang, N. C. Fan and J. A. A. Ho, ACS Appl. Mater. Interfaces, 2014, 6, 18824–18831 CAS.
- Y. Zhang, Q. Hu, M. C. Paau, S. P. Xie, P. F. Gao, W. Chan and M. M. F. Choi, J. Phys. Chem. C, 2013, 117, 18697–18708 CAS.
- Y. Chen, W. Y. Li, Y. Wang, X. D. Yang, J. Chen, Y. N. Jiang, C. Yu and Q. Lin, J. Mater. Chem. C, 2014, 2, 4080–4085 RSC.
- H. H. Deng, L. N. Zhang, S. B. He, A. L. Liu, G. W. Li, X. H. Lin, X. H. Xia and W. Chen, Biosens. Bioelectron., 2015, 65, 397–403 CrossRef CAS PubMed.
- X. Y. Mu, L. Qi, P. Dong, J. Qiao, J. Hou, Z. X. Nie and H. M. Ma, Biosens. Bioelectron., 2013, 49, 249–255 CrossRef CAS PubMed.
- Y. L. Xu, J. Sherwood, Y. Qin, D. Crowley, M. Bonizzoni and Y. P. Bao, Nanoscale, 2014, 6, 1515–1524 RSC.
- B. A. Russell, K. Kubiak-Ossowska, P. A. Mulheran, D. J. S. Birch and Y. Chen, Phys. Chem. Chem. Phys., 2015, 17, 21935–21941 RSC.
- J. Sun, Y. Yue, P. Wang, H. L. He and Y. D. Jin, J. Mater. Chem. C, 2013, 1, 908–913 RSC.
- A. J. Thenmozhi, T. R. W. Raja, U. Janakiraman and T. Manivasagam, Neurochem. Res., 2015, 40, 2006 CrossRef CAS PubMed.
- A. Sahana, A. Banerjee, S. Lohar, B. Sarkar, S. K. Mukhopadhyay and D. Das, Inorg. Chem., 2013, 52, 3627–3633 CrossRef CAS PubMed.
- S. Goswami, K. Aich, S. Das, A. K. Das, D. Sarkar, S. Panja, T. K. Mondal and S. Mukhopadhyay, Chem. Commun., 2013, 49, 10739–10741 RSC.
- S. G. Sangita Das, K. Aich, K. Ghoshal, C. Kheng Quah, M. Bhattacharyya and H.-K. Fu, New J. Chem., 2015, 39, 8582–8587 RSC.
- Y. T. Wu, Y. J. Liu, X. Gao, K. C. Gao, H. Xia, M. F. Luo, X. J. Wang, L. Ye, Y. Shi and B. Lu, Chemosphere, 2015, 119, 515–523 CrossRef CAS PubMed.
- W. T. Xu, Y. F. Zhou, D. C. Huang, M. Y. Su, K. Wang and M. C. Hong, Inorg. Chem., 2014, 53, 6497–6499 CrossRef CAS PubMed.
- C. H. Chen, D. J. Liao, C. F. Wan and A. T. Wu, Analyst, 2013, 138, 2527–2530 RSC.
- O. Alici and S. Erdemir, Sens. Actuators, B, 2015, 208, 159–163 CrossRef CAS.
- T. Y. Zhou, L. P. Lin, M. C. Rong, Y. Q. Jiang and X. Chen, Anal. Chem., 2013, 85, 9839–9844 CrossRef CAS PubMed.
- X. Y. Mu, L. Qi, J. Qiao and H. M. Ma, Anal. Methods, 2014, 6, 6445–6451 RSC.
- Z. T. Luo, X. Yuan, Y. Yu, Q. B. Zhang, D. T. Leong, J. Y. Lee and J. P. Xie, J. Am. Chem. Soc., 2012, 134, 16662–16670 CrossRef CAS PubMed.
- L. Shang, N. Azadfar, F. Stockmar, W. Send, V. Trouillet, M. Bruns, D. Gerthsen and G. U. Nienhaus, Small, 2011, 7, 2614–2620 CrossRef CAS PubMed.
- D. P. Anderson, J. F. Alvino, A. Gentleman, H. Al Qahtani, L. Thomsen, M. I. J. Polson, G. F. Metha, V. B. Golovko and G. G. Andersson, Phys. Chem. Chem. Phys., 2013, 15, 3917–3929 RSC.
- C. Zhou, C. Sun, M. X. Yu, Y. P. Qin, J. G. Wang, M. Kim and J. Zheng, J. Phys. Chem. C, 2010, 114, 7727–7732 CAS.
- X. J. Tu, W. B. Chen and X. Q. Guo, Nanotechnology, 2011, 22, 095701 CrossRef PubMed.
- X. Le Guevel, B. Hotzer, G. Jung, K. Hollemeyer, V. Trouillet and M. Schneider, J. Phys. Chem. C, 2011, 115, 10955–10963 CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra00912c |
| This journal is © The Royal Society of Chemistry 2016 |