Osteoblastogenic activity of ark shell protein hydrolysates with low molecular weight in mouse mesenchymal stem cells
Jun-Ho Hyung†
a,
Chang-Bum Ahn†b and
Jae-Young Je *a
*a
aDepartment of Marine-Bio Convergence Science, Pukyong National University, Busan 48547, Republic of Korea. E-mail: jjy1915@pknu.ac.kr; Fax: +82-51-629-6865; Tel: +82-51-629-6871
bDivision of Food and Nutrition, Chonnam National University, Gwangju 61186, Republic of Korea
First published on 16th March 2016
Abstract
Aging of human bone is characterized by decreased bone formation and bone mass. In this study, ark shell protein hydrolysates (ASPHs) were prepared by peptic hydrolysis with optimal conditions and were fractionated into 3–10 kDa, 1–3 kDa and <1 kDa fractions. Modulating the effects of ASPH with low molecular weight (ASPH < 1 kDa), which exhibited the highest stimulation effect on alkaline phosphatase (ALP) activity in mouse mesenchymal stem cells (MSCs), was investigated by measuring osteogenic biomarkers including bone morphogenetic protein-2 (BMP-2), p-Smad1/5, Runx2, Dlx5, osterix, and MAPKs as well as ALP activity, type I collagen, mineralization and osteocalcin. Treatment with ASPH < 1 kDa significantly increased the expressions of osteogenic biomarkers and also up-regulated ALP activity, mineralization, type I collagen and osteocalcin in MSCs. This study may provide new insights in the osteoblastic differentiation and ASPH < 1 kDa may be useful for health-promoting functional food ingredients against osteoporosis.
1 Introduction
Bone is an ossified tissue, which is delicately regulated by the balance between bone resorption by osteoclasts and bone formation by osteoblasts. This remodeling process is tightly regulated by bone cells themselves and also by a number of growth factors, cytokines, and environmental factors.1,2 Imbalance between bone formation and resorption results in development of a variety of bone-decreasing disorders including osteoporosis, hypercalcemia, rheumatoid arthritis, tumor metastasis into bone, periodontitis and Paget's disease.3 Currently, a number of therapeutic drugs for osteoporosis are available, however, unfortunately the long-term use of anti-osteoporotic medications is associated with serious side effects.4 Since osteoblasts play a crucial role in bone formation, activation of mesenchymal stem cells (MSCs) to an osteoblast lineage is an important step in the process of new bone formation. Therefore, the development of osteogenic agents without side effects is urgently needed.A number of natural substances including flavonoid, sulphated polysaccharide, and collagen-derived dipeptide have been developed to treat bone-related diseases.5–7 These natural products have shown beneficial effects on osteoblast differentiation and bone regeneration through activation of bone morphogenetic proteins (BMPs) and transcriptional factors including runt-related gene 2 (Runx2) and osterix, which regulate the expression of the osteoblast-related genes encoding alkaline phosphatase (ALP), type I collagen and osteocalcin. However, few studies have examined marine protein-derived food factors that have a beneficial effect on bone health. Ark shell (Scapharca subcrenata) belongs to the phylum Mollusca, class Bivalvia, suborder Taxodonta and family Arcidae and is important shellfish which is widely cultured in Korea. In 2013, the total output of ark shell was approximately 5000 M/T. Previous works reported that shellfish protein-derived peptides have shown versatile bioactivities such as antioxidant, antihypertensive, and anticancer.8,9 Therefore, the aim of this study was to produce ark shell protein-derived bioactive peptides with beneficial effect on bone health and to determine the effect on the osteoblast differentiation of mesenchymal stem cells (MSCs).
2 Materials and methods
2.1. Materials
Ark shell was purchased from a local fish market (Suncheon, Korea) and protein content of ark shell was 68.24% (dry basis) by Kjeldahl method. Cell culture media and materials were purchased from Gibco-BRL (Gaithersburg, MD, USA). Phospho-Smad1/5, Smad1/5/8, Dlx5, Runx2, osterix, p-ERK, p-JNK, p-p38, and β-actin was purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). All other chemicals were purchased from Sigma-Aldrich (St. Louis, MO, USA).2.2. Preparation and membrane fractionation of ark shell protein hydrolysates (ASPH)
Before digestion, ark shell was washed using tap water and boiled to inactivate internal enzymes for 10 min. Then ark shell was freeze-dried and pulverized. ASPH were prepared by gastointestinal digestion with pepsin at enzyme/substrate (E/S) ratios of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100, 1
100, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500, and 1
500, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000 for 120 min, and then the reaction was terminated by boiling for 10 min to inactivate pepsin. After measuring ALP activity, new digestions using the selected E/S ratio were conducted to determine the optimal digestion time for 30, 60, 120, 240, and 360 min followed by measuring ALP activity.
1000 for 120 min, and then the reaction was terminated by boiling for 10 min to inactivate pepsin. After measuring ALP activity, new digestions using the selected E/S ratio were conducted to determine the optimal digestion time for 30, 60, 120, 240, and 360 min followed by measuring ALP activity.
ASPH by enzymatic reaction of selecting E/S ratio and hydrolysis time were further fractionated based on its molecular weight using molecular weight cut-off (MWCO) membranes of 10, 3, and 1 kDa using Quixstand benchtop system (GE Healthcare, Buckinghamshire, UK). The resulted permeates were designated as 3–10 kDa, 1–3 kDa and <1 kDa peptide fractions. All fractions were freeze-dried and stored at −20 °C until needed for further analysis.
2.3. Cell culture
Murine mesenchymal stem cells (D1 cell, CRL-12424) were purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA). The cells were maintained in Dulbecco's low glucose modified Eagle's medium (DMEM) containing 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin in a 5% CO2 incubator at 37 °C. All experiments were started when the cells reached 80% confluence and used for the assays at passage 3 to 6. For osteogenic differentiation, culture media were changed at 2 days to ODM (DMEM supplemented with 50 μg mL−1 ascorbic acid, 10 mM β-glycerolphosphate, and 10−7 M dexamethasone).2.4. MTT assay
Cells were seeded into a 96 well-plate at a density of 5 × 103 cells per well. After cells were grown to confluence, they were cultured in osteogenic differentiation conditions with or without ASPHs for 24 h. The MTT solution (1 mg mL−1) was added and further incubated for 4 h. Formazan crystals were dissolved in DMSO and absorbance was measured at 540 nm.2.5. ALP activity assay and ALP staining
Cells were seeded into a 96 well-plate at a density of 5 × 103 cells per well and treated with various concentrations of ASPH and its membrane fractions for 7 days. After treatments, the cells were washed two times with PBS and lysed with 25 mM sodium carbonate buffer (pH 10) containing 0.1% triton X-100. After centrifugation at 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 15 min at 4 °C, 100 μL of supernatant was incubated with an enzyme assay buffer (25 mM carbonate buffer containing 1.5 mM MgCl2, 3.8 mM p-nitrophenyl phosphate) at 37 °C for 90 min. The absorbance was measured at 405 nm using a microplate reader and calculated using the following equation in which A and A0 were relative absorbance with and without the sample, respectively.
000 rpm for 15 min at 4 °C, 100 μL of supernatant was incubated with an enzyme assay buffer (25 mM carbonate buffer containing 1.5 mM MgCl2, 3.8 mM p-nitrophenyl phosphate) at 37 °C for 90 min. The absorbance was measured at 405 nm using a microplate reader and calculated using the following equation in which A and A0 were relative absorbance with and without the sample, respectively.| ALP activity (%) = (A − A0)/A0 × 100 |
For ALP staining, cells were seeded into a 12 well-plate and treated with ASPH < 1 kDa (400 μg mL−1) for 7 days. Cells were washed two times with PBS and fixed with 10% formalin for 5 min, and then washed with PBS. BCIP/NBT substrate solution was added into a 12 well-plate and incubated at 37 °C for 15 min. Cell images were observed on a light microscope.
2.6. Alizarin red S staining
The level of mineralization was determined by Alizarin red S staining using a 12 well-plate after 21 days treatment. Briefly, the cells were rinsed with PBS, fixed with 70% ethanol for 1 h at 4 °C, and then stained with 2% Alizarin red S solution (pH 4.2) for 15 min at room temperature. The cells were washed four times with distilled water, and allowed to dry completely. Images of calcium deposits were collected with a microscope. For quantification of mineralization, the cells were destained for 15 min with 10% of cetylpyridinium chloride in 10 mM sodium phosphate buffer (pH 7.0). Then the extracted stain was measured at 562 nm using a microplate reader. The level of mineralization was calculated using the following equation in which A and A0 were relative absorbance with and without the sample, respectively.| Mineralization level (%) = (A − A0)/A0 × 100 |
2.7. BMP-2, type I collagen, and osteocalcin assay
The levels of BMP-2 (BMP-2 Quantikine ELISA Kit, R&D systems Inc., Minneapolis, MN, USA), type I collagen (SIRCOL Collagen Assay Kit, Biocolor, UK), and osteocalcin (Osteocalin EIA kit, Biomedical Technologies Inc., Stoughton, MA, USA) were determined using the culture media according to the manufacture's instructions.2.8. Western blot analysis
Cell lysates were prepared using RIPA lysis buffer (Sigma Chemical Co.) containing protease and phosphatase inhibitor (Roche Applied Science, IN, USA). Protein concentration was quantified using the BCA protein assay kit (Thermo Scientific, MA, USA). Proteins were separated using 10% SDS-PAGE and transferred onto PVDF membrane. After 1 h blocking using 5% skim milk or BSA in TBS-T (20 mM Tris–HCl, 150 mM NaCl, 0.02% Tween-20) at room temperature, the membrane was incubated with indicated primary antibodies (overnight 4 °C). After washing with TBS-T, the membrane was incubated with horseradish peroxidase-conjugated secondary antibody at room temperature for 1 h. The blot was visualized by the enhanced chemiluminescence western blotting detection kit (Pierce Biotechnology, IL, USA).2.9. Determination of amino acid profile of ASPH < 1 kDa
Amino acid compositional analysis was performed using amino acid analyzer (S433-H, Sykam GmbH, Germany). Briefly, acid hydrolysis of ASPH < 1 kDa using 6 N HCl at 110 °C for 24 h was conducted in a sealed ampoule, and then made up to 10 mL in a volumetric flask using 0.2 M sodium citrate buffer (pH 2.2) after removal of HCl. A cation separation column (LCA K06/Na, 4.6 × 150 mm) was used at a flow rate of 0.45 mL min−1 (buffer) and 0.25 mL min−1 (reagent) at 440 nm and 570 nm.2.10. Statistics
All results are expressed as the mean ± standard deviation of three determinations. Differences between means of each group were assessed by one-way analysis of variance followed by Duncan's test using PASW Statistics 19.0 software (SPSS, Chicago, IL, USA). A P-value <0.05 was considered statistically significant.3 Results
3.1. Production of ASPHs by enzymatic hydrolysis for osteoblastic cell differentiation
Bioactive peptides, which can induce osteoblastic cell differentiation in MSCs, were produced by peptic hydrolysis and also optimal hydrolysis conditions were determined by measuring ALP activity in MSCs. Firstly, three E/S ratios of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100, 1
100, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500, and 1
500, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000 with hydrolysis time of 120 min were employed and ALP activities of the resulted bioactive peptides were displayed in Fig. 1A. ASPHs differently affected on ALP activities in MSCs and ASPH produced by E/S ratio of 1
1000 with hydrolysis time of 120 min were employed and ALP activities of the resulted bioactive peptides were displayed in Fig. 1A. ASPHs differently affected on ALP activities in MSCs and ASPH produced by E/S ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 exhibited the most ALP activity with 202% at 400 μg mL−1 compared to the blank group (without treatment). Thus, in order to determine optimal hydrolysis time at E/S ratio of 1
500 exhibited the most ALP activity with 202% at 400 μg mL−1 compared to the blank group (without treatment). Thus, in order to determine optimal hydrolysis time at E/S ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500, enzymatic hydrolysis was performed at different hydrolysis time (30, 60, 120, 240, and 360 min). Cytotoxicities of the resulted ASPHs on MSCs were determined using the MTT assay and all ASPHs showed no cytotoxicity against MSCs (Fig. 1B). As shown in Fig. 1C, ALP activities were increased up to 120 min thereafter these increments were decreased. Thus, we selected the E/S ratio of 1
500, enzymatic hydrolysis was performed at different hydrolysis time (30, 60, 120, 240, and 360 min). Cytotoxicities of the resulted ASPHs on MSCs were determined using the MTT assay and all ASPHs showed no cytotoxicity against MSCs (Fig. 1B). As shown in Fig. 1C, ALP activities were increased up to 120 min thereafter these increments were decreased. Thus, we selected the E/S ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 with hydrolysis time of 120 min and the resulted ASPH was further separated into 3–10 kDa, 1–3 kDa, and <1 kDa peptide fractions using a MWCO membrane. The peptide fractions were further evaluated their stimulation effects on ALP in MSCs. As depicted in Fig. 1D, all peptide fractions stimulated ALP activities, and moreover the low molecular weight peptide fraction (ASPH < 1 kDa) showed the most stimulation activity compared to other peptide fractions in a dose-dependent manner.
500 with hydrolysis time of 120 min and the resulted ASPH was further separated into 3–10 kDa, 1–3 kDa, and <1 kDa peptide fractions using a MWCO membrane. The peptide fractions were further evaluated their stimulation effects on ALP in MSCs. As depicted in Fig. 1D, all peptide fractions stimulated ALP activities, and moreover the low molecular weight peptide fraction (ASPH < 1 kDa) showed the most stimulation activity compared to other peptide fractions in a dose-dependent manner.
3.2. ALP staining and mineralization assays
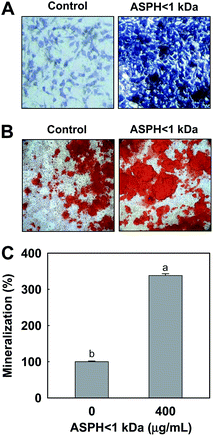
To confirm the effect of ASPH < 1 kDa on stimulation of ALP activity, we further examined staining for ALP after treatment with ASPH < 1 kDa (400 μg mL−1) for 7 days. The results revealed that ASPH < 1 kDa increased the intensity of ALP staining, indicating ASPH < 1 kDa upregulated ALP activity in MSCs (Fig. 2A). To evaluate osteoblastic mineralization, MSCs were incubated with/without ASPH < 1 kDa (400 μg mL−1) in the presence of ODM for 21 days. Fig. 2B shows the representative image of the mineralization in MSCs after treatment with ASPH < 1 kDa and ASPH < 1 kDa significantly increased the mineralization in MSCs visualized by Alizarin red S staining. Mineralization by treatment with ASPH < 1 kDa in MSCs was increased 338% compared with the untreated ODM-only control cells (Fig. 2C).3.3. ASPH < 1 kDa stimulates type I collagen and osteocalcin expressions
Type I collagen and osteocalcin as an early stage and later stage osteoblastic markers are measured using ELISA kits. Type I collagen and osteocalcin in cultured media were significantly (p < 0.05) elevated after treatment with ODM in the presence of ASPH < 1 kDa (0–400 μg mL−1) for 7 days (Fig. 3), indicating that ASPH < 1 kDa enhanced type I collagen and osteocalcin protein productions.3.4. ASPH < 1 kDa promotes BMP-2 expression
BMP-2 is an important component of osteoblastic differentiation, therefore we measured BMP-2 concentration in cultured media using an ELISA kit. As shown in Fig. 4, BMP-2 concentration was significantly (p < 0.05) increased after treatment with ODM in the presence of ASPH < 1 kDa (0–400 μg mL−1) for 7 days, suggesting that osteoblastic differentiation of MSCs may attribute to stimulation of BMP-2 expression by ASPH < 1 kDa.3.5. Effects of ASPH < 1 kDa on BMP-2 signaling pathway
To verify underlying mechanisms of ASPH < 1 kDa on osteoblastic differentiation, MSCs were cultured with ODM in the presence of ASPH < 1 kDa (400 μg mL−1) for 7 days, and then the cell lysates were prepared for western blot analysis. As described in Fig. 4, western blot analysis also revealed that ASPH < 1 kDa stimulated BMP-2/4 expression (Fig. 5A). We also analyzed downstream BMP signaling pathway and the results showed that the expressions of p-Smad1/5, Dlx-5, Runx2, and osterix proteins were up-regulated by treatment with ASPH < 1 kDa. These expressions are attributed to upregulation of BMP-2/4 expression by ASPH < 1 kDa treatment.3.6. Effects of ASPH < 1 kDa on MAPK signaling pathway
Phosphorylation of MAPKs such as ERK, JNK, and p38 is known to be involved in cell differentiation. The MSCs were cultured with ODM in the presence of ASPH < 1 kDa (400 μg mL−1) for 7 days, and the cell lysates were prepared for western blot analysis. As shown in Fig. 5B, the phosphorylation levels of ERK, JNK, and p38 in MSCs were significantly increased by treatment with ASPH < 1 kDa, but not changed the expressions of total forms of three MAPKs, indicating that ASPH < 1 kDa is capable of participating in osteoblastic differentiation through activation of MAPK signaling pathway.3.7. Amino acid composition
Amino acid compositions of fish and shellfish protein hydrolysates are important because of their nutritional value and biological activities. Amino acid composition of ark shell (AS) and ASPH < 1 kDa was determined and summarized in Table 1. ASP was most rich in aspartic acid, glutamic acid, leucine, lysine, and arginine. There is no particular trend in amino acid composition of ASPH < 1 kDa and aspartic acid, glutamic acid, leucine, arginine, and glycine are rich in ASPH < 1 kDa.| AS | ASPH < 1 kDa | |
|---|---|---|
| Aspartic acid | 0.89 ± 0.02 | 3.25 ± 0.15 |
| Threonine | 0.52 ± 0.01 | 1.04 ± 0.11 |
| Serine | 0.46 ± 0.01 | 1.01 ± 0.08 |
| Glutamic acid | 1.74 ± 0.12 | 3.69 ± 0.23 |
| Proline | 0.40 ± 0.02 | 0.77 ± 0.10 |
| Glycine | 0.65 ± 0.03 | 1.73 ± 0.21 |
| Alanine | 0.63 ± 0.02 | 1.59 ± 0.22 |
| Cysteine | 0.17 ± 0.01 | 0.24 ± 0.17 |
| Valine | 0.50 ± 0.02 | 1.15 ± 0.19 |
| Methionine | 0.27 ± 0.02 | 0.70 ± 0.08 |
| Isoleucine | 0.47 ± 0.03 | 0.85 ± 0.10 |
| Leucine | 0.81 ± 0.05 | 1.71 ± 0.16 |
| Tyrosine | 0.46 ± 0.02 | 0.86 ± 0.12 |
| Phenylalanine | 0.47 ± 0.03 | 0.96 ± 0.12 |
| Histidine | 0.22 ± 0.01 | 0.58 ± 0.09 |
| Lysine | 0.80 ± 0.06 | 1.10 ± 0.12 |
| Arginine | 0.81 ± 0.05 | 1.85 ± 0.21 |
4 Discussion
Utilization of food-derived bioactive peptides is of great interest in health promotion because bioactive peptides have shown various biological activities. These bioactive peptides can be generated by enzymatic hydrolysis with specific proteases. In the present study, for the first time, we reported osteogenic effect of ark shell protein hydrolysate with low molecular weight (ASPH < 1 kDa) in MSCs. As shown in Fig. 1, we determined the E/S ratio and hydrolysis time for the preparation of ASPH < 1 kDa, which exhibited the most stimulation effect on ALP activity in MSCs, and demonstrated that ASPH < 1 kDa increased the ALP activity and the mineralization visualized by ALP and Alizarin red S staining assays (Fig. 2). Further, the effects of ASPH < 1 kDa on osteoblast differentiation of MSCs were measured by determining osteoblastic biomarkers, including type I collagen and osteocalcin as an early stage and later stage osteoblastic markers. ASPH < 1 kDa promoted the production of type I collagen and osteocalcin in a dose-dependent manner (Fig. 3).BMPs belong to transforming growth factor-β superfamily and modulate osteoblast differentiation.10 BMPs bind to BMP receptors (BMPR-I and BMPR-2), thereafter BMPR-I and BMPR-2 form a heterodimeric receptor complex, which can phosphorylate receptor-regulated Smads (R-Smads, Smad1/5).10 This phosphorylation initiates signal transduction. Phosphorylated Smad1/5 then combined with Smad4, thereafter the complex formed translocates to the nucleus to stimulate other transcriptional factors.11 Many researches reported that BMP-2 induced osteoblast differentiation in various cell types through phosphorylation of downstream molecules such as R-Smads.7,12–14 In the present study, we demonstrated that ASPH < 1 kDa stimulated the protein expression of BMP-2/4 in MSCs (Fig. 4 and 5A). Western blot analysis also evidenced that ASPH < 1 kDa up-regulated the phosphorylation of Smad1/5 (R-Smads), indicating the phosphorylation of Smad1/5 are attributed to BMP-2/4 induction by ASPH < 1 kDa treatment.
In addition, we examined the effects of ASPH < 1 kDa on the expression of transcriptional factors such as distal-less homebox 5 (Dlx5), Runx2, and osterix by western blot analysis. Dlx5 is an essential transcriptional factor that is induced by BMP-2 and is expressed in the latter stage of osteoblast differentiation.15 Dlx5 expression stimulates osteocalcin expression and matrix mineralization, and several lines of evidence demonstrated that Dlx5 may be considered to be the upstream regulator of Runx2 and oxterix.16,17 Runx2 and osterix are also essential transcriptional factors that regulate osteoblast differentiation and bone formation through up-regulating the expression of type I collagen and osteocalcin.18,19 In this study, we found that treatment with ASPH < 1 kDa activated the expression of Dlx5, Runx2, and osterix in MSCs. Kimira et al., demonstrated that treatment with collagen-derived dipeptide up-regulated gene expression of Runx2 and osterix in MC3T3-E1 osteoblastic cells.5 Kim et al., also reported that fucoidan promotes osteoblast differentiation via BMP-2-induced Runx2 expression.12 Our results were also similar to the findings from Kimira et al. and Kim et al.5,12
MAPKs play an important role in osteoblast differentiation and are activated along with various osteoblastic biomarkers. However, the roles of p38, ERK and JNK in osteoblast differentiation are disputable. It has been reported that the activation of p38 MAPK is required for the expression of ALP in primary calvarial osteoblasts but inhibition of ERK had no significant effect on cell differentiation.20 In contrast, Kim et al., demonstrated that osteoblast differentiation induced by fucoidan in human mesenchymal stem cells is required for activation of ERK and JNK activation but not p38.12 It has been also demonstrated that the activation of ERK, which is modulated by Smads, up-regulates fibronectin and osteopontin expression.21 These discrepant roles of MAPKs in osteoblast differentiation may be attributed to the different cell types. In the present study, we examined the phosphorylation of ERK, p38 and JNK in MSCs after treatment with ASPH < 1 kDa and found that ASPH < 1 kDa activated three MAPKs (Fig. 5B). Activations of Smad proteins and MAPKs are required for osteoblast differentiation in many cell types. It has been reported that BMPs also initiate non-Smad intracellular signaling pathway and activate ERK, p38 and JNK in different cell types.21,22 In the present study, therefore, the activation of MAPKs by treatment with ASPH < 1 kDa may be attributed to the up-regulation of BMP-2 in MSCs.
5 Conclusion
In conclusion, it was shown that ASPH < 1 kDa promoted osteoblast differentiation in mouse MSCs (Fig. 6). ASPH < 1 kDa up-regulated ALP activity, mineralization, osteocalcin and type I collagen expression through activation of BMP-2 signaling pathway along with R-Smads and transcriptional factors (Dlx5, Runx2, and osterix). Moreover, ASPH < 1 kDa up-regulated the phosphorylation of MAPKs, which is required for osteoblast differentiation. Collectively, these findings suggest that ASPH < 1 kDa may be useful as an ingredient in functional foods, which can promote bone health.Conflict of interest
The authors declare that there is no conflict of interest.Acknowledgements
This study was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2013R1A1A2057530).References
- K. Matsuo and N. Irie, Arch. Biochem. Biophys., 2008, 473, 201–209 CrossRef CAS PubMed.
- N. J. Panetta, D. M. Gupta and M. T. Longaker, Curr. Stem Cell Res. Ther., 2010, 5, 122–128 CrossRef CAS PubMed.
- G. A. Rodan and T. J. Martin, Science, 2000, 289, 1508–1514 CrossRef CAS PubMed.
- S. F. Yu, T. S. Yang, W. C. Chiu, C. Y. Hsu, C. L. Chou, Y. J. Su, H. M. Lai, Y. C. Chen, C. J. Chen and T. T. Cheng, J. Bone Miner. Metab., 2013, 31, 351–359 CrossRef PubMed.
- Y. Kimira, K. Ogura, Y. Taniuchi, A. Kataoka, N. Inoue, F. Sugihara, S. Nakatani, J. Shimizu, M. Wada and H. Mano, Biochem. Biophys. Res. Commun., 2014, 453, 498–501 CrossRef CAS PubMed.
- H. Y. Yoon, S. I. Yun, B. Y. Kim, Q. Jin, E. R. Woo, S. Y. Jeong and Y. S. Chung, Eur. J. Pharmacol., 2011, 664, 54–59 CrossRef CAS PubMed.
- Y. S. Cho, W. K. Jung, J. A. Kim, I. W. Choi and S. K. Kim, Food Chem., 2009, 116, 990–994 CrossRef CAS.
- S. Y. Park, C. B. Ahn and J. Y. Je, J. Food Biochem., 2014, 38, 460–468 CrossRef CAS.
- P. A. Harnedy and R. J. FitzGerald, J. Funct. Foods, 2012, 4, 6–24 CrossRef CAS.
- C. J. Wu and H. K. Lu, Journal of Dental Science, 2008, 3, 13–21 Search PubMed.
- S. M. Abdelmagid, M. F. Barbe, I. Arango-Hisijara, T. A. Owen, S. N. Popoff and F. F. Safadi, J. Cell. Physiol., 2008, 210, 26–37 CrossRef PubMed.
- B. S. Kim, H. J. Kang, J. Y. Park and J. Lee, Exp. Mol. Med., 2015, 47, e128 CrossRef CAS PubMed.
- H. Li, H. Guo and H. Li, Steroids, 2013, 78, 426–433 CrossRef CAS PubMed.
- T. Yonezawa, J. W. Lee, A. Hibino, M. Asai, H. Hojo, B. Y. Cha, T. Teruya, K. Nagai, U. I. Chung, K. Yagasaki and J. T. Woo, Biochem. Biophys. Res. Commun., 2011, 409, 260–265 CrossRef CAS PubMed.
- H. M. Ryoo, H. M. Hoffmann, T. Beumer, B. Frenkel, D. A. Towler, G. S. Stein, J. L. Stein, A. J. van Wijnen and J. B. Lian, Mol. Endocrinol., 1997, 11, 1681–1694 CrossRef CAS PubMed.
- H. M. Ryoo, M. H. Lee and Y. J. Kim, Gene, 2006, 366, 51–57 CrossRef CAS PubMed.
- T. Tadic, M. Dodig, I. Erceg, I. Marijanovic, M. Mina, Z. Kalajzic, D. Velonis, M. S. Kronenberg, R. A. Kosher, D. Ferrari and A. C. Lichtler, J. Bone Miner. Res., 2002, 17, 1008–1014 CrossRef CAS PubMed.
- T. Komori, Cell Tissue Res., 2010, 339, 189–195 CrossRef CAS PubMed.
- P. J. Marie, Arch. Biochem. Biophys., 2008, 473, 98–105 CrossRef CAS PubMed.
- Y. Hu, E. Chan, S. X. Wang and B. Li, Endocrinology, 2003, 144, 2068–2074 CrossRef CAS PubMed.
- A. Nohe, E. Keating, P. Knaus and N. O. Petersen, Cell. Signalling, 2004, 16, 291–299 CrossRef CAS PubMed.
- M. Wan and X. Cao, Biochem. Biophys. Res. Commun., 2005, 328, 651–657 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2016 |