Design of three dimensional hybrid Co3O4@NiMoO4 core/shell arrays grown on carbon cloth as high-performance supercapacitors
Yongfeng Li*,
Hui Wang,
Jianming Jian,
Yun Fan,
Lin Yu,
Gao Cheng,
Junli Zhou and
Ming Sun
Key Laboratory of Clean Chemistry Technology of Guangdong Higher Education Institutions, School of Chemical Engineering and Light Industry, Guangdong University of Technology, Guangzhou, 510006, P. R. China. E-mail: gdliyf@163.com
First published on 26th January 2016
Abstract
Electrodes with rationally designed hybrid nanostructure composites can have superior electrochemical performance for supercapacitors to single structured materials. In this work, the uniform three dimensional (3D) hybrid Co3O4@NiMoO4 nanowire/nanosheet arrays directly grown on carbon cloth were designed and synthesized via a two-step hydrothermal method. A series of characterizations of SEM, XRD, XPS, TEM and N2 adsorption/desorption isotherms were used to verify the hierarchical core/shell hybrid nanostructure of as-prepared products, which combines the advantages of the good rate capability of Co3O4 nanowires and the high surface area of NiMoO4 nanosheets. As a binder-free electrode, the fabricated 3D hybrid nanocomposite achieves a high areal capacitance of 3.61 F cm−2 at a current density of 3 mA cm−2 and a capacitance retention of 82% with the increase of current density from 3 to 15 mA cm−2. Besides, the hybrid nanostructured product also exhibits lower bulk resistance and lower charge transfer resistance than single structured Co3O4 material. The outstanding electrochemical behaviour and the facile fabrication process suggest that this hybrid nanoarchitecture material has potential application in high-performance supercapacitors.
1 Introduction
The ever-increasing demand for energy and power has driven the development of electrochemical energy storage devices. Supercapacitors (SCs), as a class of electrical energy storage device, are outstanding for their high power density, long cycle life and low maintenance cost.1,2 Taking energy storage mechanisms into consideration, SCs are classified into two types: electrical double layer capacitors3 and pseudocapacitors.4 Compared to electrical double layer capacitors dominated by ion adsorption on the surface of active materials, pseudocapacitors can provide a much higher specific capacitance and energy density due to their fast and reversible redox reaction.5 In this regard, transition metal compounds, including oxides,6,7 hydroxides,8,9 and sulfides10,11 are investigated as potential electrode materials for pseudocapacitors. Among these materials, binary metal oxides exhibit a higher performance than single-component metal oxides because of their feasible oxidation state, high electrical conductivity, potential low-cost and behaviour as environmental-friendly energy storage materials.12,13 For instance, the metal molybdates, including NiMoO4,14,15 CoMoO4 (ref. 16) and MnMoO4,17 are promising potential electrode materials owing to their low-cost, abundant resources and environmental compatibility. In particular, NiMoO4 has received increasing interest in the past several years for the high electrochemical activity of Ni ion.18 However, the reported electrode materials based on transition metal oxides are usually made by a traditional slurry-coating method, where the involved polymer binders and conducting additives can generate series resistance and deterioration of capacitance during redox reactions.19,20 So the design and development of the new framework of metal oxide electrode materials are in progress.Currently, the three dimensional hybrid core/shell nanostructures directly grown on conducting substrates as binder-free electrode have attracted more attention because they have higher surface/body ratios, larger surface areas, better permeabilities and more electroactive sites.21–25 The synergistic effect caused by such hybrid core/shell nanostructures can facilitate the diffusion of active species and the transport of electrons.26 For instance, Lou et al. synthesized hierarchical NiCo2O4@MnO2 core/shell heterostructured nanowire arrays on Ni foam, which delivered a capacitance of 2.05 F cm−2 at a current density of 20 mA cm−2 with a cycling stability of 85% after 1000 cycles.27 Wang et al. reported a facile two-step hydrothermal method to integrate two ternary metal oxides (NiCo2O4 and CoMoO4) into core/shell nanowire arrays on Ni foam, and the resulting NiCo2O4@CoMoO4 hybrid electrode exhibited areal capacitance of 14.67 F cm−2 at a current density of 10 mA cm−2, which are several times larger than the pristine NiCo2O4 electrode (1.42 F cm−2).28 Zou et al. described the fabrication of 3D NiCo2S4@NiO2 dendritic heterojunction arrays on Ni foam with a high capacitance of 4.28 F cm−2 even at a current density of 50 mA cm−2 and a high capacitance retention of 96.36% after 3000 cycles.29 In this work, we present a facile strategy to prepare Co3O4 nanowire@NiMoO4 nanosheet core/shell hybrid structures on carbon cloth for supercapacitor electrode application. The crystalline Co3O4 nanowires uniformly grown on carbon cloth current collector are used as the backbone to support and provide reliable electrical connection to the NiMoO4 nanosheets coatings with mesoporous structure, enabling the full utilization of NiMoO4 by offering more electroactive sites and faster electronic/ionic conductivity. Meanwhile, in contrast to Ni foam, carbon cloth as a flexible substrate also has many advantages of low-cost, good electrical conductivity, chemical stability, light weight, flexibility and high porosity. So such three dimensional hybrid Co3O4@NiMoO4 core/shell nanocomposites are expected to exhibit good electrochemical performance as supercapacitor electrode materials.
2 Experimental
2.1 Synthesis of Co3O4 nanowires
The Co3O4 nanowires array was grown on carbon cloth by a facile hydrothermal method.30 Prior to the experiment, the carbon cloth substrates were carefully cleaned in 4 M HCl solution, ethanol and deionized water successively with the assistance of ultrasonication. In a typical synthesis, 1.5 mmol of Co(NO3)2·6H2O, 6.0 mmol of NH4F and 7.5 mmol of urea were dissolved in 15 mL deionized water, then the obtained homogeneous clear pink solution was transferred into a Teflon-lined stainless steel autoclave with a piece of pretreated carbon cloth (1 × 2 cm2) at 120 °C for 9 h. After that, the sample was collected and rinsed several times with deionized water and ethanol, followed by drying at 60 °C for 12 h. Finally, the carbon cloth with the as-prepared hydrate precursors was annealed at 350 °C for 2 h to obtain Co3O4 nanowires.2.2 Synthesis of the hybrid Co3O4@NiMoO4 core/shell nanocomposite
To obtain hybrid nanostructures of Co3O4@NiMoO4, a secondary hydrothermal reaction was adopted to grow the NiMoO4 nanosheets on the backbone of Co3O4 nanowires. For this, the obtained Co3O4 nanowires supported on carbon cloth substrate was directly immersed in an solution containing 1 mmol of Ni(CH3COO)2·4H2O, 0.2 g of (NH4)2Mo7O24·4H2O, 0.24 g of urea and 10 mL of deionized water in an autoclave at 160 °C for 10 h. Afterwards, the sample was collected and rinsed with deionized water and ethanol several times, followed by annealing at 400 °C in air for 2 h.2.3 Material characterization
X-ray diffraction (XRD) patterns were recorded on a Bruker D8 advance diffractometer, using Cu-Kα radiation at 40 kV and 40 mA. The nanostructured samples were observed by using a field emission scanning electron microscope (FESEM, JEOL JSM-7001F) and a field emission transmission electron microscope (FETEM, FEI Tecnai G20). X-ray photoelectron spectroscopy (XPS) was carried out by using Thermo Scientific ESCALAB 250XI spectrometer. Nitrogen adsorption/desorption isotherms were measured on ASAP 2020 analyzer.2.4 Electrochemical measurements
Electrochemical measurements were performed on the Autolab PGSTAT302N electrochemical workstation using a three-electrode test system in 2 M KOH aqueous solution. The Co3O4@NiMoO4 core/shell arrays or pristine Co3O4 arrays on carbon cloth (≈1 × 1 cm2; hybrid Co3O4@NiMoO4 mass: ≈2.0 mg, pristine Co3O4 mass: ≈1.2 mg) were directly used as the working electrode. A platinum electrode and a saturated calomel electrode (SCE) served as the counter electrode and the reference electrode respectively. The areal capacitance of the electrode was calculated according to the following equations:
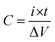 | (1) |
3 Results and discussion
As shown in Fig. 1, the fabrication of hybrid Co3O4@NiMoO4 nanostructure as a binder-free electrode involves two key steps of hydrothermal reactions. First, the aligned pristine Co3O4 nanowires were grown on the fibers of carbon cloth substrate by a hydrothermal and post-annealing process. Second, interconnected NiMoO4 nanosheets with different lateral sizes were controllably grown on the backbone of Co3O4 nanowires through a secondary hydrothermal process. | ||
| Fig. 1 Schematic illustration of the fabrication process for 3D hybrid Co3O4@NiMoO4 core/shell nanowires/nanosheets array on carbon cloth. | ||
The SEM images of the as-synthesized pristine Co3O4 nanowires and hybrid Co3O4@NiMoO4 core/shell nanostructures grown on carbon cloth are shown in Fig. 2. From Fig. 2a–c, it can be observed that the free standing Co3O4 nanowires are densely and uniformly distributed on the fibers of the carbon cloth substrate with an average diameter of ca. 50–80 nm and length up to ca. 5 μm. And the high magnification SEM image of the sample reveals the rather smooth surface and sharp tip of the Co3O4 nanowires with diameter gradually decreasing from the bottom to the tip in Fig. 2c. Moreover, the slim Co3O4 nanowires are also observed to be homogenously aligned and separated apart adequately, which can serve as the ideal conductive scaffolds for the subsequent growth of the NiMoO4 nanosheets shell. From the SEM images of the hybrid sample shown in Fig. 2d–f, it is clearly observed that the Co3O4 “branches” are tightly bonded and totally covered with “leaves” of NiMoO4 nanosheets after the secondary hydrothermal reaction. In addition, the NiMoO4 nanosheets are typically interconnected with each other to construct a highly porous and rough morphology on the surface of Co3O4 nanowires, resulting in the creation of three dimensional hybrid core/shell nanostructures on a large scale on the carbon cloth. These NiMoO4 nanosheets not only have a robust adhesion and electrical contact to the Co3O4 nanowires, but also connect to each other and finally form a network like structure. Moreover, the interconnected nanosheets with a large lateral size produce abundant space and electroactive sites, making them fully accessible to electrolyte.31,32
 | ||
| Fig. 2 SEM images of (a–c) the Co3O4 nanowires array on the carbon cloth, (d–f) the hybrid Co3O4@NiMoO4 core/shell nanostructure array on carbon cloth. | ||
The phase structure of the as-synthesized pristine Co3O4 nanowires and hybrid Co3O4@NiMoO4 core/shell nanostructures on carbon cloth are displayed in Fig. 3a. Apparently, the main reflection peaks at a 2θ value of 31.9°, 36.9°, 44.8°, 59.4° and 65.2° can be well indexed to the (220), (311), (400), (511) and (440) planes of Co3O4 (JCPDS no. 42-1467).33,34 Unfortunately, no new peaks are observed in the XRD pattern of the Co3O4@NiMoO4 hybrid nanocomposite, indicating the low content and poor crystallinity of the NiMoO4 nanosheets compared with Co3O4 nanowires.35 However, the surface chemical composition of the hybrid sample can be verified by XPS, as shown in Fig. 3b–d. The Co 2p core level spectrum shows two peaks at 781 and 796 eV, corresponding to the Co 2p3/2 and Co 2p1/2 for Co3O4 respectively.36 The Ni 2p core level spectrum reveals two major peaks at 857.2 and 874.9 eV, coupling with two satellite peaks at 862.9 and 881.1 eV, which correspond to Ni 2p3/2 and Ni 2p1/2 respectively. The main binding energy peaks of Ni 2p3/2 and Ni 2p1/2 are separated by 17.7 eV, which is a signature of the Ni2+ oxidation state.34,37 Two binding energy peaks located at 232.6 and 235.7 eV with splitting width 3.1 eV can be observed in the region of Mo 3d, which are characteristics of the Mo6+ oxidation state.38 The XPS results confirm that the valence of Ni and Mo elements are +2 and +6 respectively, which is in accordance with the phase structure of NiMoO4.
 | ||
| Fig. 3 (a) XRD patterns of the as-synthesized Co3O4 nanowires and hybrid Co3O4@NiMoO4 core/shell array on carbon cloth. (b–d) XPS spectra of Co 2p, Ni 2p and Mo 3d for hybrid Co3O4@NiMoO4. | ||
More information about the morphological and structural features of the hybrid Co3O4@NiMoO4 nanocomposite was obtained by TEM and high-resolution TEM (HRTEM). As illustrated in Fig. 4a and b, the pristine Co3O4 “nanocore” is tightly bonded with ultrathin NiMoO4 nanosheets, forming a typical core/shell structure, which is in good accordance with the SEM observation. Moreover, the TEM results clearly indicate that the interconnected NiMoO4 nanosheets around Co3O4 nanowires are highly porous and ultrathin with a few nanometers in thickness. Interestingly, the ultrathin NiMoO4 nanosheets exhibit graphene-like morphology with transparent feature, further suggesting the ultrathin nature (Fig. 4b). HRTEM measurements of the shell materials (Fig. 4c) typically display a set of clear lattice fringe with an interplanar space of ca. 0.214 nm, corresponding to the (121) planes of NiMoO4 nanosheets in the hybrid structure.24 The corresponding selected area electron diffraction (SAED) pattern (as shown in the inset of Fig. 4c) also indicates the polycrystalline nature of the ultrathin nanosheets, which is consistent with the XPS results.
As shown in Fig. 5, the N2 adsorption/desorption isotherms of pristine Co3O4 nanowires and Co3O4@NiMoO4 nanocomposite are similar in shape. Both can be observed a type-IV isotherms with a type-H3 hysteresis loop in the relative pressure range of 0.5–1.0 P/P0, which is ascribed to the presence of mesoporous structure in the samples. The mesoporous feature can be further confirmed by the pore size distribution analysis in the inset of Fig. 5. The sharp peaks in the pore size range of 2–4 nm and 7–15 nm are all attributed to mesoporous channels in the sample architectures, which is in good accordance with the TEM observation. Besides, it can be found that there are also many micropores in the samples, indicating the meso–micro dual porous structure in the as-prepared products, especially the hybrid Co3O4@NiMoO4 nanocomposite. Moreover, in contrast to the Co3O4 nanowires, the hybrid Co3O4@NiMoO4 nanocomposite show a more distinct hysteresis loop and more meso–micro porous volume, which can be attributed to the presence of ultrathin NiMoO4 nanosheets grown on Co3O4 nanowires. These ultrathin NiMoO4 nanosheets create voids between adjacent particles that increase the surface area.7 So the specific surface area of Co3O4@NiMoO4 nanocomposite was calculated to be 251.2 m2 g−1, which was much higher than that of pristine Co3O4 nanowires (23.5 m2 g−1). This meso–micro dual porous structure with high surface area of composites can play an important role in providing more electroactive sites and shorter diffusion paths for electrochemical reactions on the electrode surface.39,40
 | ||
| Fig. 5 Nitrogen adsorption/desorption isotherms plots and BJH pore size distribution of (a) Co3O3 nanowires and (b) Co3O4@NiMoO4 hybrid nanocomposite. | ||
The electrochemical properties of the as-prepared pristine Co3O4 nanowires and hybrid Co3O4@NiMoO4 nanocomposite as electrodes investigated in detail are as follows. The typical cyclic voltammetry (CV) curves of Co3O4 nanowires and Co3O4@NiMoO4 nanocomposite electrodes within the potential window of −0.2 to 0.8 V (vs. SCE) at various scan rates are shown in Fig. 6a and c. Both the CV curves are consisted of obvious redox peaks, indicating that the specific capacitance characteristics are mainly attributed to the faradaic redox reactions of Co3+/Co4+ and/or Ni2+/Ni3+.15,41,42 Besides, compared with the CV curves of Co3O4 nanowires, the Co3O4@NiMoO4 nanocomposite has more distinct redox peaks and larger enclosed CV curve area at the same scan rate (as seen in Fig. 7a), reflecting that the hybrid Co3O4@NiMoO4 electrode has higher specific capacitance. This is in good accordance with the results obtained by the galvanostatic charge–discharge measurement in Fig. 6b and d. The areal capacitances of the electrodes can be calculated from the discharge curves and the corresponding results are plotted in Fig. 7b. Encouragingly, the areal capacitances of hybrid Co3O4@NiMoO4 nanocomposite electrode are 3.61 F cm−2 (1805 F g−1) at low current density of 3 mA cm−2 (1.5 A g−1) and 2.96 F cm−2 (1480 F g−1) at high current density of 15 mA cm−2 (7.5 A g−1), and at least 82% level can be maintained, whereas the maximum areal capacitance of the pristine Co3O4 nanowires is only 0.40 F cm−2 (333 F g−1). Impressively, these results are remarkable as compared to many of the previously reported Co3O4 and NiMoO4 based materials. For example, the ZnO@Co3O4 core/shell hetero-structures on Ni foam is 1.52 F cm−2 at 5 mA cm−2;19 the Co3O4@NiMoO4 nanosheets array on nickel foam is 2.67 F cm−2 at 3 mA cm−2;34 the NiMoO4 nanowires on carbon cloth is 1.27 F cm−2 at 5 mA cm−2.43 Such enhanced pseudocapacitive performance further proves the great advantages of the present two-step hydrothermal method, which would make the Co3O4 nanowires more suitable as backbone for the subsequent construction of 3D hybrid Co3O4@NiMoO4 nanocomposite electrode for supercapacitor applications.
Electrochemical impedance spectroscopy (EIS) was carried out to further explore the electrochemical behaviors of the Co3O4@NiMoO4 hybrid electrode. The Nyquist plots of the pristine Co3O4 nanowires and hybrid Co3O4@NiMoO4 nanocomposite electrodes in the frequency range of 0.01 Hz to 100 kHz are shown in Fig. 8. The two plots all exhibit a semicircle loop followed by a rising along imaginary impedance axis. In the low frequency region, the slope of the curves shows the Warburg resistance, which represents the ion diffusion of the electrolyte into the inner surface of electrode materials.44,45 As compared with the pristine Co3O4 electrode, the hybrid Co3O4@NiMoO4 electrode shows a slightly less vertical straight line along the imaginary axis (as shown in Fig. 8), indicating a bit higher ion diffusion resistance. This can be attributed to the meso–micro dual porous structure of hybrid Co3O4@NiMoO4 nanocomposite, in which the microporous channels are less favorable for the electrolyte penetration and OH− ion diffusion into the inner of electrode materials than mesoporous channels. In the high frequency region, the intersection of the curves at the real impedance axis indicates the bulk resistance of the electrochemical system, including a series resistance of the electrolyte inside the pore and the interfacial resistance at the electroactive material/current collector and at the electrode/electrolyte interface.46,47 In addition, the diameter of the depressed semicircle at high frequency region corresponds to the charge transfer resistance on the electrode surface.48,49 As shown in the inset of Fig. 8, the hybrid electrodes are observed to exhibit much lower bulk resistance and lower charge transfer resistance than the pristine Co3O4 electrode, since the interconnected neighboring NiMoO4 nanosheets can contact with each other and provide more electroactive sites and additional convenient pathways for electron transport in the electrochemical system.
Finally, as one of the key issue for supercapacitor electrode materials, the cycling stability has been tested for the pristine Co3O4 nanowires and hybrid Co3O4@NiMoO4 composite, as shown in Fig. 9. For the pristine Co3O4 electrode, the areal capacitance is about 0.32 F cm−2 after 2500 cycles at the discharge current density of 10 mA cm−2 (about 95.6% retention). Although, the hybrid Co3O4@NiMoO4 electrode displays a declining behaviour during the long-term cycling tests (about 77.4% retention after 3000 cycles), the final capacitance value is still much better than that of pristine Co3O4 even at a higher discharge current density of 15 mA cm−2. In addition, for Co3O4@NiMoO4 electrode, the capacitance loss (16.6%) was mainly during the first 600 cycles, which can be generally explained by the irreversible of the Faraday reactions or the dissolution of active materials from the hybrid nanostructures at the beginning of the cycle test.34,50 However, it still retains 92.7% after the rest of 2400 cycles, exhibiting a relatively good stability.
 | ||
| Fig. 9 Cycling performance of the pristine Co3O4 nanowires and hybrid Co3O4@NiMoO4 electrodes at different current densities. | ||
4 Conclusions
In summary, the uniform three dimensional hybrid Co3O4@NiMoO4 core/shell nanocomposite directly grown on carbon cloth are successfully synthesized by a facile two-step hydrothermal method. As a binder-free electrode, the constructed 3D hybrid nanocomposite delivers impressive pseudocapacitive performances (a high areal capacitance of 3.61 F cm−2 at current density of 3 mA cm−2 and a capacitance retention of 82%), in contrast to the pristine Co3O4 nanowires. Such intriguing capacitive behavior is attributed to the 3D nanocomposite configuration and the synergistic effect of the combined pseudocapacitive contributions from the Co3O4 nanowires and the ultrathin NiMoO4 nanosheets. This work suggests that the aforementioned hybrid core/shell nanoarchitecture is very suitable and promising as advanced electrode materials for high-performance supercapacitors.Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (Grant No. 21576054), Pearl River Nova Program of Guangzhou (Grant No. 2011J2200041), Guangdong Province Science and Technology Project (Grant No. 2012A030600006), Science and Technology Achievements Transformation Projects of Guangdong Higher Education Institutes (Grant No. cgzhzd1104).References
- M. Winter and R. J. Brodd, Chem. Rev., 2004, 104, 4245–4270 CrossRef CAS PubMed.
- C. C. Hu, K. H. Chang, M. C. Lin and Y. T. Wu, Nano Lett., 2006, 6, 2690–2695 CrossRef CAS PubMed.
- G. Wang, J. Zhang, S. Kuang, J. Zhou, W. Xing and S. Zhuo, Electrochim. Acta, 2015, 153, 273–279 CrossRef CAS.
- S. Xiong, C. Yuan, M. Zhang, B. Xi and Y. Qian, Chem.–Eur. J., 2009, 15, 5320–5326 CrossRef CAS PubMed.
- C. Yuan, L. Yang, L. Hou, L. Shen, X. Zhang and X. W. Lou, Energy Environ. Sci., 2012, 5, 7883–7887 CAS.
- Z. Yu, B. Duong, D. Abbitt and J. Thomas, Adv. Mater., 2013, 25, 3302–3306 CrossRef CAS PubMed.
- D. Cai, B. Liu, D. Wang, Y. Liu, L. Wang, H. Li, Y. Wang, C. Wang, Q. Li and T. Wang, Electrochim. Acta, 2014, 115, 358–363 CrossRef CAS.
- L. Su, C. Ma, T. Hou and W. Han, RSC Adv., 2013, 3, 19807 RSC.
- J. Zhao, J. Chen, S. Xu, M. Shao, Q. Zhang, F. Wei, J. Ma, M. Wei, D. G. Evans and X. Duan, Adv. Funct. Mater., 2014, 24, 2938–2946 CrossRef CAS.
- M. Sun, J. Tie, G. Cheng, T. Lin, S. Peng, F. Deng, F. Ye and L. Yu, J. Mater. Chem. A, 2015, 3, 1730–1736 CAS.
- L. Mei, T. Yang, C. Xu, M. Zhang, L. Chen, Q. Li and T. Wang, Nano Energy, 2014, 3, 36–45 CrossRef CAS.
- C. Yuan, J. Li, L. Hou, X. Zhang, L. Shen and X. W. Lou, Adv. Funct. Mater., 2012, 22, 4592–4597 CrossRef CAS.
- D. Guo, P. Zhang, H. Zhang, X. Yu, J. Zhu, Q. Li and T. Wang, J. Mater. Chem. A, 2013, 1, 9024 CAS.
- B. Senthilkumar, K. V. Sankar, R. K. Selvan, M. Danielle and M. Manickam, RSC Adv., 2013, 3, 352–357 RSC.
- S. E. Moosavifard, J. Shamsi, S. Fani and S. Kadkhodazade, RSC Adv., 2014, 4, 52555–52561 RSC.
- X. Xia, W. Lei, Q. Hao, W. Wang and X. Wang, Electrochim. Acta, 2013, 99, 253–261 CrossRef CAS.
- K. K. Purushothaman, M. Cuba and G. Muralidharan, Mater. Res. Bull., 2012, 47, 3348–3351 CrossRef CAS.
- S. Peng, L. Li, H. B. Wu, S. Madhavi and X. W. D. Lou, Adv. Energy Mater., 2015, 5, 1401172–1401178 Search PubMed.
- D. Cai, H. Huang, D. Wang, B. Liu, L. Wang, Y. Liu, Q. Li and T. Wang, ACS Appl. Mater. Interfaces, 2014, 6, 15905–15912 CAS.
- Z. Yin, S. Zhang, Y. Chen, P. Gao, C. Zhu, P. Yang and L. Qi, J. Mater. Chem. A, 2015, 3, 739–745 CAS.
- J. Liu, J. Jiang, C. Cheng, H. Li, J. Zhang, H. Gong and H. J. Fan, Adv. Mater., 2011, 23, 2076–2081 CrossRef CAS PubMed.
- X. Sun, Q. Li, Y. Lu and Y. Mao, Chem. Commun., 2013, 49, 4456–4458 RSC.
- D. Guo, W. Ren, Z. Chen, M. Mao, Q. Li and T. Wang, RSC Adv., 2015, 5, 10681–10687 RSC.
- D. Cheng, Y. Yang, J. Xie, C. Fang, G. Zhang and J. Xiong, J. Mater. Chem. A, 2015, 3, 14348–14357 CAS.
- G. Jiang, M. Zhang, X. Li and H. Gao, RSC Adv., 2015, 5, 69365–69370 RSC.
- W. Ren, D. Guo, M. Zhuo, B. Guan, D. Zhang and Q. Li, RSC Adv., 2015, 5, 21881–21887 RSC.
- L. Yu, G. Zhang, C. Yuan and X. W. Lou, Chem. Commun., 2013, 49, 137–139 RSC.
- D. Cai, B. Liu, D. Wang, L. Wang, Y. Liu, H. Li, Y. Wang, Q. Li and T. Wang, J. Mater. Chem. A, 2014, 2, 4954–4960 CAS.
- R. Zou, Z. Zhang, M. F. Yuen, J. Hu, C.-S. Lee and W. Zhang, Sci. Rep., 2015, 5, 7862 CrossRef PubMed.
- J. Jiang, J. P. Liu, X. T. Huang, Y. Y. Li, R. M. Ding, X. X. Ji, Y. Y. Hu, Q. B. Chi and Z. H. Zhu, Cryst. Growth Des., 2010, 10, 70–75 CAS.
- B. Wang, S. Li, X. Wu, W. Tian, J. Liu and M. Yu, J. Mater. Chem. A, 2015, 3, 13691–13698 CAS.
- F. Deng, L. Yu, G. Cheng, T. Lin, M. Sun, F. Ye and Y. Li, J. Power Sources, 2014, 251, 202–207 CrossRef CAS.
- D. Cai, D. Wang, B. Liu, L. Wang, Y. Liu, H. Li, Y. Wang, Q. Li and T. Wang, ACS Appl. Mater. Interfaces, 2014, 6, 5050–5055 CAS.
- W. Hong, J. Wang, P. Gong, J. Sun, L. Niu, Z. Yang, Z. Wang and S. Yang, J. Power Sources, 2014, 270, 516–525 CrossRef CAS.
- D. Cai, D. Wang, B. Liu, Y. Wang, Y. Liu, L. Wang, H. Li, H. Huang, Q. Li and T. Wang, ACS Appl. Mater. Interfaces, 2013, 5, 12905–12910 CAS.
- Y. Q. Zhang, L. Li, S. J. Shi, Q. Q. Xiong, X. Y. Zhao, X. L. Wang, C. D. Gu and J. P. Tu, J. Power Sources, 2014, 256, 200–205 CrossRef CAS.
- K. Xiao, L. Xia, G. Liu, S. Wang, L.-X. Ding and H. Wang, J. Mater. Chem. A, 2015, 3, 6128–6135 CAS.
- Z. Xu, Z. Li, X. Tan, C. M. B. Holt, L. Zhang, B. S. Amirkhiz and D. Mitlin, RSC Adv., 2012, 2, 2753 RSC.
- A. Ajay, A. Paravannoor, J. Joseph, A. V, A. Ss, S. V. Nair and A. Balakrishnan, Appl . Surf. Sci., 2015, 326, 39–47 CrossRef.
- D. Ghosh, S. Giri and C. K. Das, Nanoscale, 2013, 5, 10428–10437 RSC.
- X.-J. Ma, W.-B. Zhang, L.-B. Kong, Y.-C. Luo and L. Kang, New J. Chem., 2015, 39, 6207–6215 RSC.
- T. Liu, H. Chai, D. Jia, Y. Su, T. Wang and W. Zhou, Electrochim. Acta, 2015, 180, 998–1006 CrossRef CAS.
- D. Guo, Y. Luo, X. Yu, Q. Li and T. Wang, Nano Energy, 2014, 8, 174–182 CrossRef CAS.
- D. Guo, Y. Z. Luo, X. Z. Yu, Q. H. Li and T. H. Wang, Nano Energy, 2014, 8, 174–182 CrossRef CAS.
- P. Liu, Y. Deng, Q. Zhang, Z. Hu, Z. Xu, Y. Liu, M. Yao and Z. Ai, Ionics, 2015, 21, 2797–2804 CrossRef CAS.
- P. H. Yang and W. J. Mai, Nano Energy, 2014, 8, 274–290 CrossRef CAS.
- H. Liu, J. Zhang, D. Xu, B. Zhang, L. Shi, L. Huang and S. Tan, Appl. Surf. Sci., 2014, 317, 370–377 CrossRef CAS.
- X. Li, J. Shen, N. Li and M. Ye, Mater. Lett., 2015, 139, 81–85 CrossRef CAS.
- M. C. Liu, L. Kang, L. B. Kong, C. Lu, X. J. Ma, X. M. Li and Y. C. Luo, RSC Adv., 2013, 3, 6472–6478 RSC.
- H. Zhang, Y. Chen, W. Wang, G. Zhang, M. Zhuo, H. Zhang, T. Yang, Q. Li and T. Wang, J. Mater. Chem. A, 2013, 1, 8593–8600 CAS.
| This journal is © The Royal Society of Chemistry 2016 |




