Tunable photoluminescence emissions and large dielectric constant of the electroactive poly(vinylidene fluoride–hexafluoropropylene) thin films modified with SnO2 nanoparticles
Nur Amin Hoquea,
Pradip Thakurab,
Niranjan Balaac,
Arpan Koola,
Sukhen Das†
*a and
Partha Pratim Ray*a
aDepartment of Physics, Jadavpur University, Kolkata-700032, India. E-mail: sdasphysics@gmail.com; Tel: +91-9433091337
bDepartment of Physics, Netaji Nagar College for Women, Kolkata-700092, India
cDepartment of Botany, Sree Gopal Banerjee College, Hooghly-712503, India
First published on 10th March 2016
Abstract
In the present study, SnO2 nanoparticles (NPs) have been synthesized hydrothermally at 100 °C and 150 °C. The NPs have been characterized by X-ray diffraction (XRD), UV-visible spectroscopy and field emission scanning electron microscopy (FESEM). Thereafter, poly(vinylidene fluoride–hexafluoropropylene) (PVDF–HFP) with different contents (1–15 wt%) of SnO2 NPs has been synthesized by a simple solution-casting method. An electroactive β-phase formation mechanism in the NP-incorporated PVDF–HFP thin films is confirmed by XRD, Fourier transform infrared spectroscopy and differential scanning calorimetry. FESEM images show interfacial interaction in the thin films between the polymer matrix and the NPs. Maximum β-phase nucleation of ∼74% has been reached by 10 wt% addition of SnO2 NPs synthesized hydrothermally at 100 °C. The photoluminescence emission spectra show two UV and visible emissions at two excitation wavelengths. Uniform distribution of the NPs in the polymer matrix leads to large dielectric constant ∼2578.44 at 20 Hz due to addition of 10 wt% SnO2 NPs (100SO) in PVDF–HFP matrix. The large increase in dielectric constant has been successfully explained in terms of MWS polarization effect and percolation theory.
1. Introduction
Over the last decade, electroactive polymer nanocomposites (NCs), with high dielectric constant, excellent piezoelectric, pyroelectric, ferroelectric and electro-optic properties, have gained interest due to their variety of potential applications. These include high energy density arrays of capacitors, acoustic transducers, actuators, infrared detectors, piezoelectric sensors and nanogenerator, rail runs, pulsed lasers, information storage memory devices, developed flexible and stretchable energy harvesting systems in both biomedical and electronic fields.1–10 Among these, energy harvesting technologies, which are produced with piezoelectric materials have attracted great attention because these allow the conversion of ambient energy, such as sunlight, wind, vibration, rain drops and sea waves, to electric energy.11Among the most important electroactive semicrystalline thermoplastic polymers, the poly(vinylidene fluoride) (PVDF) homopolymer and its copolymers (such as poly(vinylidene fluoride-co-hexafluoropropylene) (PVDF–HFP), poly(vinylidene fluoride-co-trifluoroethylene) (P(VDF–TrFE)), and poly(vinylidene fluoride–trifluoroethylene–chlorofluoroethylene) (P(VDF–TrFE–CFE))), with good peizo-electric, pyro-electric, ferro-electric coefficients, excellent thermal stability and chemical resistance, have been considered as prospective materials for the abovementioned applications.12–18 Nowadays, there has been a great interest to enhance the piezoelectricity and electrochemical coupling factor of PVDF, which is sweeping real applications. The semicrystalline polymer of PVDF occurs in at least five forms, i.e. α, β, γ, δ and ε, but the most common crystalline phases of PVDF are α, β and γ; moreover, the β form assumes paramount importance among all other forms due to its excellent piezoelectricity and good elastic strength and therefore study on improvement of the β-phase nucleation in PVDF has currently received a lot of attention.19–22 The thermodynamically stable polymorph of PVDF and its co-polymers is the nonpolar α-phase with TGTG′ (T-trans, G-gauche+, G′-gauche) dihedral conformation.23 However, the all trans (TTTT) conformation, i.e. the electroactive β-phase, leads to highest spontaneous polarization per unit cell.24 The polar γ-phase with TTTGTTG′ conformational sequence shows average piezoelectricity.26
Thus, there is a great challenge to develop cost-effective and reliable methods to improve the fraction of electroactive β-phase in PVDF and its co-polymers. The polar β-phase nucleation in PVDF and its co-polymers can be obtained from the nonpolar α-phase by, for example, controlled annealing or melting crystallization under high pressure27 of α PVDF and its co-polymer, the electro-spinning method,28 low-temperature polar solvent crystallization,29 addition of nucleation agents or surface-charged nucleation agents,30 applying a strong electric field31 and, most recently, using supercritical carbon dioxide (SC CO2).32 Recent studies are suggesting that the addition of nanoparticle (NPs),33 some fillers, such as metal oxide NPs, metal NPs, carbon black, carbon nanotubes, graphene, carbon nanofibers, inorganic salts, clays, ceramic nanofillers, transition metal oxide NPs, nickel hydroxide, and some other polymer, into the polymer matrix not only effectively enhances the polar β-phase in PVDF and its co-polymers but also improves the dielectric properties of the polymer NCs thin films.33–40 Several efforts have been made41 to improve the nucleation of β crystallites in PVDF, dielectric properties and optical properties, but rarely the study deal with use of the cost-effective electroactive PVDF–HFP to develop optically active and high dielectric electroactive PVDF–HFP NCs. Some studies on PVDF–HFP/SnO2 composite films used as anode materials in lithium ion batteries have been reported earlier.42 However, studies on the effect of SnO2 NPs on the electroactive β-phase and its dielectric properties are very rare. Tin oxide (SnO2) is an important metal oxide and n-type wide band gap semiconductor and has outstanding optical, electrical and electrochemical properties.43
Our present study deals with fabrication of low cost, high dielectric, electroactive PVDF–HFP NC thin films by incorporating the optically active SnO2 NPs. First, SnO2 NPs have been prepared via a hydrothermal process, treating for 12 hours at two different temperatures, 100 °C and 150 °C. Therefore, PVDF–HFP thin films with different percentages of SnO2 NPs have been synthesized by a solution-casting method. The effect of the NPs on electroactive β-phase crystallization, optical and dielectric properties in PVDF films has been investigated thoroughly in terms of surface charge, size and the interfacial polarization, which occurs between the interfaces of the NPs and the polymer chains near the percolation threshold.
2. Experimental
2.1. Materials
The materials used in our present studies are stannic chloride (SnCl4·5H2O) (Merck, India), lithium hydroxide (Merck, India), poly(vinylidene fluoride-co-hexafluoropropylene) (PVDF–HFP) pellets (Aldrich, Germany. Mw: 180![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 GPC, Mn: 71
000 GPC, Mn: 71![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000) and dimethyl sulfoxide (DMSO) (Merck, India).
000) and dimethyl sulfoxide (DMSO) (Merck, India).
2.2. Preparation of SnO2 NPs and PVDF–HFP composites
0.5 M stannic chloride (SnCl4·5H2O) was dissolved in 50 mL de-ionized water with vigorous stirring. Then, 1.0 M LiOH was added after dissolving SnCl4·5H2O. The mixture was vigorously stirred for 1 hour and then transferred into two 25 mL Teflon-lined stainless-steel autoclaves. The two autoclaves were heated, one at 100 °C the other at 150 °C, for 12 hours and subsequently cooled to room temperature. The products were collected by centrifugation and rinsed with deionized water several times to adjust the pH to 7. Finally, the obtained samples were dried at 80 °C overnight and collected in vacuum for further characterizations.PVDF–HFP/SnO2 NPs NC thin films were prepared via simple solution-casting method. PVDF–HFP (0.2 g) was dissolved in 5 mL DMSO and the desired percentage of SnO2 NPs added to this solution. The mixture was continuously stirred at 60 °C overnight. The mixture was then cast onto clean Petri dishes and dried at 80 °C in an oven until complete evaporation of the solvent. The films were collected and stored in a vacuum. Pure PVDF–HFP film was also prepared by the same procedure for comparison. The thickness of the synthesized films was in the range of 50–75 μm. Table 1 presents details of the different amounts of NPs added to the PVDF–HFP matrix.
| Sample name | Amount of PVDF–HFP (g) | Hydrothermal temperature of SnO2 NPs (°C) | Percentage SnO2 NPs (mass%) | Amount of SnO2 NPs (g) |
|---|---|---|---|---|
| Pure PVDF–HFP | 0.2 | NA | 0 | 0 |
| 100SO1 | 0.2 | 100 | 1 | 0.002 |
| 100SO2.5 | 0.2 | 100 | 2.5 | 0.005 |
| 100SO5 | 0.2 | 100 | 5 | 0.01 |
| 100SO7.5 | 0.2 | 100 | 7.5 | 0.015 |
| 100SO10 | 0.2 | 100 | 10 | 0.02 |
| 100SO15 | 0.2 | 100 | 15 | 0.03 |
| 150SO1 | 0.2 | 150 | 1 | 0.002 |
| 150SO2.5 | 0.2 | 150 | 2.5 | 0.005 |
| 150SO5 | 0.2 | 150 | 5 | 0.01 |
| 150SO7.5 | 0.2 | 150 | 7.5 | 0.015 |
| 150SO10 | 0.2 | 150 | 10 | 0.02 |
| 150SO15 | 0.2 | 150 | 15 | 0.03 |
2.3. Characterization
The fraction of β-phase (F(β)) in the films was evaluated using the Lambert–Beer law:
 | (1) |
The heat of melting or enthalpies of fusion (ΔHm) of pure PVDF–HFP and SnO2 NP-doped PVDF–HFP thin films (100SO and 150SO) were calculated from DSC curves. The degree of crystallinity (Xc) of the samples was evaluated using the equation as follows:
| Xc = ΔHm/ΔH100% | (2) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) were collected in the frequency range of 20 Hz to 2 MHz at room temperature using a digital LCR meter (Agilent, E4980A). 1 V AC voltage was applied across the two opposite surfaces of the samples. The dielectric constant (ε) and the AC conductivity (σac) of the samples were evaluated using eqn (3) and (4), respectively, as follows:
δ) were collected in the frequency range of 20 Hz to 2 MHz at room temperature using a digital LCR meter (Agilent, E4980A). 1 V AC voltage was applied across the two opposite surfaces of the samples. The dielectric constant (ε) and the AC conductivity (σac) of the samples were evaluated using eqn (3) and (4), respectively, as follows:| ε = Cd/ε0A | (3) |
σac = 2πfε0ε![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) tan tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ δ
| (4) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ are the capacitance, thickness, area and tangent loss of the samples, respectively, f is the applied frequency in Hz and ε0 is the permittivity of free space (8.854 × 10−12 F m−1).44
δ are the capacitance, thickness, area and tangent loss of the samples, respectively, f is the applied frequency in Hz and ε0 is the permittivity of free space (8.854 × 10−12 F m−1).443. Results and discussion
3.1. Characterization of SnO2 particles
We prepared SnO2 NPs for two different hydrothermal temperatures, i.e. 100 °C and 150 °C. The XRD patterns of the as-synthesized SnO2 NPs (synthesized at 100 and 150 °C) are show in Fig. 1a. All the diffraction peaks from X-ray reflections confirm the formation of tetragonal phase SnO2 NPs (JCPDS no. 41-1445); there are no other peaks, suggesting high purity of the synthesized SnO2 NPs. The UV-Vis spectroscopy of both sample 100SO and 150SO also have been shown in Fig. 1b. Absorption peaks for both samples arise at ∼255 nm.45 Fig. 1c and d show the FESEM images of SnO2 NPs synthesized hydrothermally at 100 °C and 150 °C, respectively. Formation of comparatively large particles with diameter ∼800 nm has been observed at 100 °C and well-defined, uniformly-distributed SnO2 NPs with diameter ∼30–50 nm are formed at 150 °C. | ||
| Fig. 1 (a) XRD spectra of SnO2 NPs synthesized at 100 °C and 150 °C; (b) UV-visible absorption spectra in DMSO medium and FESEM images of SnO2 NPs: (c) 100SO and (d) 150SO. | ||
3.2. Characterization of PVDF–HFP/SnO2 NPs NCs
The standard XRD peaks of pure PVDF at 2θ = 17.7° (100), 18.4° (020), 19.9° (110), and 26.5° (021) all are assigned to the α-PVDF, whereas the 2θ = 20.8° peak, due to the superposition of the (110) and (200) plane, corresponds to β-PVDF and that at 38.9° (211) is due to the γ-phase. However, all the peaks for the pure PVDF–HFP are well matched with the standard peaks of α-PVDF.46 However, for the PVDF–HFP/SnO2 NPs films the intensities of all the characteristic diffraction peaks corresponding to α- and γ-phases become less significant but the diffraction peak at 20.4° ((110), (200)) appeared more strongly, indicating a conversion of the crystalline structure from the nonpolar α-phase to electroactive β-phase in the SnO2 NPs-modified PVDF–HFP thin films. It may be suggested from the diffraction patterns that the highest β-phase conversion has occurred at 10 wt% addition of SnO2 NPs prepared at 100 °C, whereas only 5 wt% loading of the SnO2 NPs prepared at 150 °C in the polymer matrix obtains the maximum β-phase crystallization in NC thin films. Smaller sizes of the SnO2 NPs prepared at 150 °C play a crucial role in developing and stabilizing more β polymorphs or crystallites in the NPs-modified PVDF–HFP thin films at low concentration. Further addition of NPs shows again the conversion of nonpolar α-crystallites.
More quantitatively, the α-phase and β-phase nucleation may be assessed through the ratio of the intensity of the peaks at 20.4° (200) and 18.2° (020) (I20.4/I18.2).45 For pure PVDF–HFP thin film, this ratio is found to be 0.94. Results suggest that this ratio depends on wt% of NPs. 10 wt% loading of SnO2 NPs prepared at 100 °C in the polymer matrix (Fig. 2c) shows the highest value ∼5.35798 and 4.61594 has been found for 5 wt% loading of SnO2 NPs prepared at 150 °C (Fig. 2d).
Fig. 3 illustrates the IR spectra of the pure PVDF–HFP and NPs-loaded PVDF–HFP thin films. The absorbance bands due to nonpolar α-phase (TGTG′) centered at 488 cm−1 (CF2 wagging), 532 cm−1 (CF2 bending), 615 and 764 cm−1 (CF2 bending and skeletal bending, respectively), 796 and 976 cm−1 (CH2 rocking) are clearly observed in the spectrum of pure PVDF–HFP thin film. Two tiny bands also observed at 840 cm−1 (CH2 rocking, CF2 stretching and skeletal C–C stretching) and 813 cm−1 (CF2 asymmetric stretching), which correspond to electroactive or polar β-phase and γ-phase, respectively.
All characteristic absorbance bands corresponding to nonpolar α-phase are present in the pure PVDF–HFP spectrum. For the 100SO/PVDF–HFP NC thin films, the IR spectra confirm the nucleation of polar β-phase by showing the strong appearance of the peaks at 444 cm−1 (CF2 rocking), 510 cm−1 (CF2 stretching), and 840 cm−1 (CH2 rocking, CF2 stretching and skeletal C–C stretching). For the sample 100SO10, maximum enhancement of the characteristic peaks related to the electroactive β-phase (TTTT conformation) has been observed (Fig. 3a). However, for 5 wt% the doping of 150SO in PVDF–HFP matrix (Fig. 3c) obtains the maximum intensity of the main β-phase peak at 840 cm−1 (CH2 rocking, CF2 stretching and skeletal C–C stretching).13,14,19,25 The characteristic absorption peak related to the polar γ-phase at 812 cm−1 (CF2 asymmetric stretching)46 is totally absent in the NC thin films.
The relative fraction of electroactive β-phase content (F(β)) has been calculated using eqn (1). The calculated value of (F(β)) for pure PVDF–HFP film has been found to be 32% and for the 100SO/PVDF–HFP thin films, a maximum ∼74% β-phase nucleation is achieved by loading 10 wt% of SnO2 NPs synthesized at 100 °C (Fig. 3b). The relative fraction of β-phase (F(β)) attained a maximum value ∼70.9% for 5 wt% loading of SnO2 NPs synthesized at 150 °C. The FTIR data are well consistent with the XRD results.
 | ||
| Fig. 4 DSC thermographs of (a) pure PVDF–HFP 100SO-loaded PVDF–HFP thin films and (b) pure PVDF–HFP 150SO-loaded PVDF–HFP thin films. | ||
The appearance of a broader endothermic peak at 147.86 °C in the thermograph of pure PVDF–HFP thin film suggests crystallization of the nonpolar α polymorph.49 From Fig. 4a it can be found that the melting temperature increases to higher temperature up to 10 wt% loading of 100SO in PVDF–HFP matrix and decreases for higher loading of the NPs. A similar type of shifting of melting temperature to higher temperature in 150SO/PVDF–HFP NCs has also been observed (Fig. 4b). Maximum shifting in melting temperature to higher value is observed up to 5 wt% loading of SnO2 NPs prepared at 150 °C in PVDF–HFP matrix. This increase in melting temperatures up to a certain concentration of NPs (10 wt% for 100SO and 5 wt% for 1500SO) may be due to the formation of the more crystalline polar β-phase in polymer thin films by catalytic action of the NPs.48 These results are also consistent with XRD and FTIR data.
The melting enthalpies (ΔHm) can be calculated from the DSC thermographs and then the degree of crystallinity (Xc) of the samples may also be obtained using eqn (2). Fig. 5 represents the change of ΔHm and Xc with the NPs content. Melting enthalpy (ΔHm) and the degree of crystallinity (Xc) increase up to 10 wt% for 100SO-loaded PVDF–HFP thin films and then the values decrease. Herein, also maximum melting enthalpy (ΔHm) and the degree of crystallinity (Xc) value have been found for 5 wt% loading of 150SO-loaded PVDF–HFP thin films.
 | ||
| Fig. 6 FESEM micrograph of (a) pure PVDF–HFP, (b) 100SO2.5, (c) 100SO5, (d) 100SO7.5, (e) 100SO10 and (f) 100SO15 samples. | ||
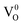 ,
, 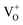 and
and  states. The oxygen vacancy is an intrinsic donor in SnO2.51 Because
states. The oxygen vacancy is an intrinsic donor in SnO2.51 Because 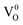 is a very shallow donor, it is expected that most oxygen vacancies will be in their paramagnetic
is a very shallow donor, it is expected that most oxygen vacancies will be in their paramagnetic  states under the flat band condition and thus the emission of SnO2NPs/PVDF–HFP thin films in the UV region is directly attributed to the formation of oxygen vacancies in paramagnetic
states under the flat band condition and thus the emission of SnO2NPs/PVDF–HFP thin films in the UV region is directly attributed to the formation of oxygen vacancies in paramagnetic 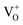 states. The visible broad emission in our samples around 400–500 nm may be assigned to the formation of a
states. The visible broad emission in our samples around 400–500 nm may be assigned to the formation of a  luminescent center. The recombination of a conduction band electron with the
luminescent center. The recombination of a conduction band electron with the  center results in visible emission in the samples when the surface-trapped hole recombines with an electron in a deeply trapped
center results in visible emission in the samples when the surface-trapped hole recombines with an electron in a deeply trapped  to form a
to form a  center.52
center.52
 | ||
| Fig. 9 Photo luminescence spectra of pure PVDF–HFP and SnO2 NPs-PVDF–HFP thin films: (a) and (c) λex = 256 nm and (b) and (d) λex = 303 nm at ambient conditions. | ||
3.2.7.1. SnO2 NPs content dependence. The dependence of dielectric constant and tangent loss of SnO2 NPs-loaded PVDF–HFP thin films on NPs content at two different frequencies, 20 Hz and 1 MHz, have been shown in Fig. 10. Remarkable improvement in dielectric constant has been observed due to incorporation of both NPs in the PVDF–HFP matrix. The dielectric constant has been observed to increase up to 10 wt% loading of the SnO2 NPs synthesized at 100 °C in PVDF–HFP in both frequency and dielectric value but has decreased for further loading of the NPs (Fig. 10a and b). The maximum values of the dielectric constant, ∼2578.45 at 20 Hz and 400.6 at 1 MHz, have been achieved by embedding 10 wt% loading of SnO2 NPs synthesized at 100 °C. Only 5 wt% incorporation of SnO2 NPs prepared at 150 °C into the polymer matrix shows a fast improvement in dielectric constant, with values ∼865.55 at 20 Hz and 200.4 at 1 MHz, and then a decrease in dielectric constant has been noted for further loading of 150SO. Tangent losses for both the NPs-loaded samples (100SO and 150SO) have increased non-linearly due to the occurrence of different Debye-like relaxation.16,25
The improvement of dielectric constant is very slow up to 7.5 wt% for 100SO-doped polymer thin films and 2.5 wt% for 150SO-loaded samples, but the dielectric value increase very fast at 10 and 5 wt% addition of 100SO and 150SO in PVDF–HFP matrix, respectively, which may be due to reaching the percolation threshold value.16,25 The remarkable improvement of dielectric constants may be explained according to percolation theory as follows:
| ε = ε0(1 − f/fc)−q | (5) |
Fig. 11a and b display variation of the AC conductivity of the samples with different concentration of NPs at 20 Hz, 10 kHz, 100 kHz and 1 MHz. Almost sharp increase in the AC conductivity of the NPs-doped samples has been noticed upon the addition of 100SO up to 10 wt% and 150SO up to 5 wt% at the high frequency of 1 MHz, but for lower frequencies (100 kHz, 10 kHz and 20 Hz), the variation of AC conductivity of the NC thin films with NPs content shows a slow increase without altering the nature of variation at 1 MHz. With increase of the frequency the AC conductivity also increase on the basis of an apparent power law: σ(ω) = ωn (0.6 ≤ n ≤ 1).16
 | ||
| Fig. 11 SnO2 NPs content dependence of AC conductivities at 20 Hz, 10 kHz, 100 kHz and 1 MHz (a) 100SO-loaded samples and (b) 150SO-loaded samples. | ||
3.2.7.2. Frequency dependence of dielectric properties. The frequency dependence of dielectric constant, tangent loss (tan
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) and AC conductivity of pure PVDF–HFP and PVDF–HFP/SnO2 NPs NC thin films at ambient conditions have been shown in Fig. 12. The dielectric constant of the samples decreases with frequency increasing when the NPs concentration increases up to 15 wt% for 100SO- and 150SO-doped polymer nanocomposite films. At low frequency, space charge, i.e., dipoles, may follow the direction of the applied electric field, but for higher frequencies the dipoles are unable to follow the electric field and lag behind the electric field resulting in the decrease of dielectric constant.12,16 At the maximum value of frequency, ∼1/t, the dielectric constant shows a relaxation (t is the relaxation time).46 This reduction of dielectric constant can also be explained by the MWS polarization effect. Upon the application of the electric field, the charges move freely and accumulate at the interfaces between the polymer matrix and the NPs due to differences in conductivity and dielectric properties of insulating PVDF–HFP and semiconducting SnO2 NPs.25 At low frequencies, the movement and transfer of charge carriers occurs effortlessly due to there being enough time to move to the electrode, but at higher frequencies, the interfacial movement of the dipoles is restricted resulting in reduction of the dielectric constant. Similarly, tan
δ) and AC conductivity of pure PVDF–HFP and PVDF–HFP/SnO2 NPs NC thin films at ambient conditions have been shown in Fig. 12. The dielectric constant of the samples decreases with frequency increasing when the NPs concentration increases up to 15 wt% for 100SO- and 150SO-doped polymer nanocomposite films. At low frequency, space charge, i.e., dipoles, may follow the direction of the applied electric field, but for higher frequencies the dipoles are unable to follow the electric field and lag behind the electric field resulting in the decrease of dielectric constant.12,16 At the maximum value of frequency, ∼1/t, the dielectric constant shows a relaxation (t is the relaxation time).46 This reduction of dielectric constant can also be explained by the MWS polarization effect. Upon the application of the electric field, the charges move freely and accumulate at the interfaces between the polymer matrix and the NPs due to differences in conductivity and dielectric properties of insulating PVDF–HFP and semiconducting SnO2 NPs.25 At low frequencies, the movement and transfer of charge carriers occurs effortlessly due to there being enough time to move to the electrode, but at higher frequencies, the interfacial movement of the dipoles is restricted resulting in reduction of the dielectric constant. Similarly, tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ for all samples decreases with increasing frequency in the same manner. Only the tan
δ for all samples decreases with increasing frequency in the same manner. Only the tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ of the 150SO5 sample shows a Debye-like dipolar relaxation peak (Fig. 12e). The highest dielectric constants observed have been ε ∼ 2578.45 for 100SO10 and ε ∼ 865.55 for 150SO5 at 20 Hz.
δ of the 150SO5 sample shows a Debye-like dipolar relaxation peak (Fig. 12e). The highest dielectric constants observed have been ε ∼ 2578.45 for 100SO10 and ε ∼ 865.55 for 150SO5 at 20 Hz.
Fig. 12c and f represent the variation of the AC conductivity of pure PVDF–HFP and NC thin films with increasing frequency. The AC conductivities of the samples increase almost linearly with increasing frequency. The conductivity of the sample can be divided into two regions, one is for the lower frequency range dominated by DC conductivity, and the other region is a higher frequency region characterized by AC conductivity, which mainly depends on frequency. In our samples, a lower frequency plateau is totally absent. Thus, our samples pose only AC conductivities. Therefore, the increase in AC conductivity of the NC samples with increasing frequency and up to a certain percentage of the fillers (10 wt% for 100SO and 5 wt% for 150SO), is mainly due to the MWS interfacial contribution and dipolar relaxation modes.46
4. Conclusions
The effect of SnO2 NPs (of two different sizes) on the electroactive β-phase crystallization, photoluminescence emissions and dielectric properties of PVDF–HFP thin films has been studied in detail in the present study. Incorporation of SnO2 NPs promotes ∼74% electroactive β-phase nucleation for 10 wt% loading of NPs synthesized at 100 °C and ∼70.9% for 5 wt% addition of NPs prepared at 150 °C. Strong ion-dipole or electrostatic interaction between the –CH2 dipoles and negatively-charged NPs' surfaces transform the polymer chains from nonpolar TGTG′ conformation to polar all trans (TTTT) conformation or electroactive β polymorphs. Tunable photoluminescence emissions, both in the UV and visible regions, have been observed for both NPs-loaded samples. Remarkable improvement in dielectric constant due to incorporation of the SnO2 NPs in the PVDF–HFP matrix has also been noticed. The very large increase of dielectric constant at certain doping concentrations is due to reaching the percolation threshold value and the large accumulation of charge due to the MWS effect. The highest dielectric constants achieved were ∼2578.45 for 10 wt% loading of 100SO in PVDF–HFP matrix and ∼865.55 for 5 wt% loading of 150SO, at 20 Hz. Therefore, these successfully fabricated electroactive, optically active and high dielectric polymer NC thin films may be used suitably to develop the energy harvesters, sensors, actuators, energy storage devices, thin film capacitors, solar cells, optical detectors, optoelectronic devices and piezoelectric nanogenerators.Acknowledgements
Authors acknowledge University Grand Commission (UGC), Government of India for the financial support to this study.References
- P. Martins, C. Caparros, R. Gonçalves, P. M. Martins, M. Benelmekki, G. Botelho and S. Lanceros-Mendez, J. Phys. Chem. C, 2012, 116(29), 15790–15794 CAS.
- L. Xie, X. Huang, K. Yang, S. Li and P. Jiang, J. Mater. Chem. A, 2014, 2, 5244 CAS.
- J. H. Kim, J. H. Kim, E. S. Choi, J. H. Kim and S. Y. Lee, RSC Adv., 2014, 4, 54312 RSC.
- S. H. Lim, A. C. Rastogi and S. B. Desu, J. Appl. Phys., 2004, 96, 5673 CrossRef CAS.
- N. S. Choi, Z. Chen, S. A. Freunberger, X. Ki, Y. K. Sun, K. Amine, G. Yushin, L. F. Nazar, J. Cho and P. G. Bruce, Angew. Chem., Int. Ed., 2012, 51, 9994 CrossRef CAS PubMed.
- V. Etacheri, R. Marom, R. Elazari, G. Salitra and D. Aurbach, Energy Environ. Sci., 2011, 4, 3243 CAS.
- G. Jeong, Y. U. Kim, H. Kim, Y. J. Kim and H. J. Sohn, Energy Environ. Sci., 2011, 4, 1986 CAS.
- R. C. G. Naber, C. Tanase, P. W. M. Blom, G. H. Gelinck, A. W. Marsman, F. J. Touwslager, S. Setayesh and D. M. De Leeuw, Nat. Mater., 2005, 4, 243–248 CrossRef CAS.
- E. Y. Tsymbal and H. Kohlstedt, Science, 2006, 313, 181–183 CrossRef CAS PubMed.
- T. T. Wang, J. M. Herbert and A. M. Glass, The Applications of Ferroelectric Polymers, Chapman and Hall, New York, 1988 Search PubMed.
- A. J. Lovinger, Science, 1983, 220, 1115–1121 CAS.
- P. Thakur, A. Kool, B. Bagchi, S. Das and P. Nandy, Phys. Chem. Chem. Phys., 2015, 17, 1368–1378 RSC.
- P. Martins, A. C. Lopes and S. Lanceros-Mendez, Prog. Polym. Sci., 2013, 39(4), 683–706 CrossRef.
- P. Thakur, A. Kool, B. Bagchi, S. Das and P. Nandy, Appl. Clay Sci., 2014, 99, 149–159 CrossRef CAS.
- G. H. Kim, S. M. Hong and Y. Seo, Phys. Chem. Chem. Phys., 2009, 11, 10506–10512 RSC.
- Y. Feng, W. L. Li, Y. F. Hou, Y. Yu, W. P. Cao, T. D. Zhang and W. D. Fei, J. Mater. Chem. C, 2015, 3, 1250 RSC.
- D. Yuan, Z. Li, W. Thitsartarn, X. Fan, J. Sun, H. Lia and C. He, J. Mater. Chem. C, 2015, 3, 3708 RSC.
- B. S. I. Gunduz, R. Alpern, D. Amare, J. Crawford, B. Dolan, S. Jones, R. Kobylarz, M. Reveley and P. Cebe, Polymer, 2010, 51, 1485–1493 CrossRef.
- P. Thakur, A. Kool, B. Bagchi, N. A. Hoque, S. Das and P. Nandy, RSC Adv., 2015, 5, 28487–28496 RSC.
- D. Mandal, K. J. Kim and J. S. Lee, Langmuir, 2012, 28, 10310–10317 CrossRef CAS PubMed.
- S. Yu, W. Zheng, W. Yu, Y. Zhang, Q. Jiang and Z. Zhao, Macromolecules, 2009, 42, 8870–8874 CrossRef CAS.
- J.-W. Park, Y.-A. Seo, I. Kim and C.-S. Ha, Macromolecules, 2004, 37, 429–436 CrossRef CAS.
- L. Huang, C. Lu, F. Wangd and L. Wang, RSC Adv., 2014, 4, 45220–45229 RSC.
- M. Sharma, G. Madrasa and S. Bose, Phys. Chem. Chem. Phys., 2014, 16, 14792 RSC.
- P. Thakur, A. Kool, B. Bagchi, N. A. Hoque, S. Das and P. Nandy, Phys. Chem. Chem. Phys., 2015, 17, 13082–13091 RSC.
- G. H. Kim, S. M. Hong and Y. Seo, Phys. Chem. Chem. Phys., 2009, 11, 10506–10512 RSC.
- T. Hattori, M. Hikosaka and H. Ohigashi, Polymer, 1996, 37, 85–91 CrossRef CAS.
- P. Sajkiewicz, A. Wasiak and Z. Goclowski, Eur. Polym. J., 1999, 35, 423–427 CrossRef CAS.
- J. G. Bergman, J. H. McFee and G. R. Crane, Appl. Phys. Lett., 1971, 18, 203–209 CrossRef CAS.
- M. Imran-ul-haq, B. Tiersch and S. Beuermann, Macromolecules, 2008, 41, 7453–7458 CrossRef CAS.
- J. S. Andrew and D. R. Clarke, Langmuir, 2008, 24, 670–674 CrossRef CAS PubMed.
- P. Mishra and P. Kumar, Compos. Sci. Technol., 2013, 88, 26–32 CrossRef CAS.
- H. P. Xu and Z. M. Dang, Chem. Phys. Lett., 2007, 438(4–6), 196–202 CrossRef CAS.
- P. Martins, C. M. Costa, M. Benelmekki, G. Botelho and S. L. Mendez, CrystEngComm, 2012, 14(8), 2807–2811 RSC.
- V. Sencadas, P. Martins, A. Pităes, M. Benelmekki, J. L. G. Ribelles and S. Lanceros-Mendez, Langmuir, 2011, 27(11), 7241–7249 CrossRef CAS PubMed.
- R. Song, G. Xia, X. Xing, L. He, Q. Zhao and Z. Mad, J. Colloid Interface Sci., 2013, 401, 50–57 CrossRef CAS PubMed.
- S. L. Jiang, U. Yu, J. J. Xie, L. P. Wang, Y. K. Zeng and M. Fu, J. Appl. Polym. Sci., 2010, 116(2), 838–842 CAS.
- J. K. Yuan, S. H. Yao, Z. M. Dang, A. Sylvestre, M. Genestoux and J. Bai, J. Phys. Chem. C, 2011, 115, 5515–5521 CAS.
- Y. J. Li, M. Xu, J. Q. Feng and Z. M. Dang, Appl. Phys. Lett., 2006, 89(7), 072902 CrossRef.
- W. Wu, X. Huang, S. Li, P. Jiang and T. Toshikatsu, J. Phys. Chem. C, 2012, 116(47), 24887–24895 CAS.
- M. Yu, Y. Z. Long, B. Sunb and Z. Fan, Nanoscale, 2012, 4, 2783–2796 RSC.
- Y. Zhang, Q. Xiao, G. Lei, Z. Li and X. Li, Electrochim. Acta, 2015, 178, 336–343 CrossRef CAS.
- K. Bouras, J.-L. Rehspringer, G. Schmerber, H. Rinnert, S. Colis, G. Ferblantier, M. Balestrieri, D. Ihiawakrim, A. Dinia and A. Slaoui, J. Mater. Chem. C, 2014, 2, 8235–8243 RSC.
- A. Kool, P. Thakur, B. Bagchi, N. A. Hoque and S. Das, Appl. Clay Sci., 2015, 114, 349–358 CrossRef.
- E. A. Floriano, L. V. A. Scalvi, J. R. Sambranoc and V. Geraldo, Mater. Res., 2010, 13(4), 437–443 CrossRef CAS.
- P. Thakur, A. Kool, B. Bagchi, N. A. Hoque, S. Das and P. Nandy, RSC Adv., 2015, 5, 62819–62827 RSC.
- V. Tomer, E. Manias and C. A. Randall, J. Appl. Phys., 2011, 110, 044107 CrossRef.
- L. Wu, W. Yuan, N. Hu, Z. Wang, C. Chen, J. Qiu, J. Ying and Y. Li, J. Phys. D: Appl. Phys., 2014, 47, 135302 CrossRef.
- O. Padmaraj, M. Venkateswarlu and N. Satyanarayana, Ionics, 2013, 19, 1835–1842 CrossRef CAS.
- T. W. Kim, D. U. Lee and Y. S. Yoon, J. Appl. Phys., 2000, 88, 3759 CrossRef CAS.
- F. Gu, S. F. Wang, C. F. Song, M. K. Lu, Y. X. Qi, G. J. Zhou, D. Xu and D. R. Yuan, Chem. Phys. Lett., 2003, 372, 351–354 CrossRef.
- S. Das, S. Kar and S. Chaudhuri, J. Appl. Phys., 2006, 99, 114303 CrossRef.
- C. C. Wang, J. F. Song, H. M. Bao, Q. D. Shen and C. Z. Yang, Adv. Funct. Mater., 2008, 18(8), 1299–1306 CrossRef CAS.
- J. W. Zha, X. Meng, D. Wang, Z. M. Dang and R. K. Y. Li, Appl. Phys. Lett., 2014, 104, 072906–072914 CrossRef.
Footnote |
| † Present address: Department of Physics, IIEST, Howrah, West Bengal – 711103, India. |
| This journal is © The Royal Society of Chemistry 2016 |







