Disinfection of water by pulsed power technique: a mechanistic perspective
Raj Kamal Singh†a,
Ligy Philip†*a and
Sarathi Ramanujamb
aDepartment of Civil Engineering, Indian Institute of Technology Madras, Chennai, India – 600036. E-mail: ligy@iitm.ac.in; Fax: +91-44-22574252; Tel: +91-44-22574274
bDepartment of Electrical Engineering, Indian Institute of Technology Madras, Chennai, India – 600036
First published on 21st January 2016
Abstract
The mechanism of disinfection by pulsed plasma technology was investigated in detail, with Escherichia coli as the model bacteria. Studies were carried out to investigate the behavior of bacterial surface potential as a function of applied energy, based on molecular level analysis of DNA and proteins. The cell survival and morphological changes during plasma treatment were correlated with membrane damage using SEM analysis, along with DNA degradation and protein leakage. In addition, cell size (Z-average) and poly-dispersive index (PI) of E. coli cells were measured to understand the colloidal behavior of disinfected bacterial cells in plasma reactors. Significant membrane deformities and morphological changes were observed under SEM analysis, and lactate dehydrogenase (LDH) assay confirmed a drastic increase in membrane permeability upon plasma treatment. An energy dependent DNA fragmentation and protein leakage confirmed the killing of bacterial cells. The E. coli cell membrane zeta potential (−14.81 ± 2.08 mV) was neutralized to −1.08 ± 0.83 mV in 6 min of treatment time due to the propensity of H+ ions on bacterial membranes. The FTIR confirmed the changes occurring in the functional groups of bacterial cell membranes, which further emphasized the possible interaction with ROS/RNS and membrane damage.
1. Introduction
Access to clean and safe drinking water is the foremost demand of the current century. It is estimated that about 783 million people in the world do not have access to clean water and about 2.5 billion people do not have access to proper sanitation.1 Over 80% of used water is not collected or treated,2 which further stresses the available potable water sources. Pathogenic contamination of the potable water sources leads to detrimental effects on humans and aquatic species. In humans, the enteric bacteria such as E. coli inhabit the intestine and cause diarrhoea, hemorrhagic colitis and haemolytic uremic syndrome.3 WHO estimated that the 84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 diarrhoeal deaths per year are due to inadequate drinking water, improper sanitation and hygiene.4 Thus, the elimination of pathogens has become a major consideration in any treatment system.
200 diarrhoeal deaths per year are due to inadequate drinking water, improper sanitation and hygiene.4 Thus, the elimination of pathogens has become a major consideration in any treatment system.
Most commonly used disinfection methods for the removal of pathogens from drinking water include chlorination, UV radiation and ozonation. The principle mechanism of disinfection using chlorine, ozone and UV radiation have been well studied and those mechanism include (i) cell wall damage, (ii) alternation of the cell permeability, (iii) alternation of the colloidal nature of the cytoplasm, (iv) inhibition of enzyme activity and (v) alteration of the organism DNA.5 Although chlorination is extensively used, the formation of carcinogenic organochlorine compounds is a major problem of the process.6 Similarly, an inadequate dose of ozone in water leads to various disinfection by-products.7 The harmful consequences of chemical decontamination methods have led to the development of advanced technologies like electrochemical techniques, nanotechnology and advanced oxidation processes (AOPs). Various AOPs such as photo-Fenton,8 O3/UV,9 UV/H2O2,10 electrochemical process11 and photocatalysis12–14 have been extensively studied for the bacterial disinfection. Recent studies have provided the evidence for bacteriological cytotoxicity of engineered nanoparticles (NPs).15 The cytotoxicity effects of NPs are attributed to the involvement of the free radicals, oxidative damage of cell membrane, leakage of extracellular components and lipid peroxidation. Ultimately DNA getting damaged is the predominant cell death mechanism in bacteria.16–18 Although there has been a significant advancement in the bacterial disinfection processes using the advanced techniques mentioned earlier, still there are gaps in the understanding of the underlying mechanisms that govern the complete bacterial kill. So far, all the mechanisms proposed for the AOPs and electrochemical disinfection methods mostly describe the role of electron generated free chlorine and hydroxyl radical in causing the bacterial death. However, the study of bacterial deactivation at the sub-cellular level, involving the mechanisms such as membrane damage, protein and DNA damage, enzyme inhibition etc. is extremely limited.
In recent years, plasma technology for water treatment has been gaining importance due to its rapid disinfection potential.19,20 The generation of reactive oxygen species (ROS) such as OH˙, O3, H2O2, HO2˙, O2˙− and reactive nitrogen species (RNS) such as NO˙ and NO2˙etc., along with UV light and shock waves21–23 in plasma processes, makes the application of plasma technology in water and wastewater treatment feasible. Plasma, either the corona discharge or streamer, has been reported to cause effective bacterial decontamination in gas24 and water.25–29 Although the antimicrobial effect of plasma on different bacterial strains is well established, to the best of authors' knowledge, the disinfection mechanism is not fully understood. Reactive species, UV light and shock waves produced during corona discharge can alter the membrane, proteins and DNA of the bacterial cells.26,29–31 Also, stress caused by corona discharge may enhance the endogenous inactivation mechanism by increasing intracellular ROS level.30 Rapid ionization of air during corona discharge and dissolution of ionized gases in water can alter the pH, conductivity32 and electrophoretic mobility of the ions present in the aqueous phase. These conditions then alter the potential across the cell membrane and thus lead to the leakage of cell materials (proteins, DNA and salts, etc.) in aqueous solution. Therefore, understanding the fate and behaviour of bacteria during plasma treatment is a prerequisite for the assessment of the role of reactive species on the physical and molecular damage of bacterial cells. Also, bacterial cells act as colloidal particles in aqueous solution.33 The behaviour of these colloids may alter during plasma treatment process. The neutralization of negatively charged bacterial cells in the suspension medium can cause flocculation of cells. Understanding the colloidal behaviour during plasma treatment might help to separate untreated, partially treated and/or treated dead cells by electrostatic trapping from large volumes of water.34
This paper reports the studies performed on the effect of pulsed corona discharge on E. coli decontamination in water. In order to understand the disinfection mechanism, the membrane deformities of E. coli upon plasma treatment were first determined using scanning electron microscope (SEM) and lactate dehydrogenase (LDH) release. Bacterial membrane functional group perturbation upon plasma treatment was analysed using FTIR analysis. To understand the cellular biomolecular changes under corona discharge, the DNA degradation study and protein assay have been conducted. Finally, the cell size, polydispersive index (PDI) and zeta potential were measured to determine the effect of aqueous environment on cellular mobility and its colloidal behaviour.
2. Materials and methodology
2.1. Experimental set up
The schematic diagram of corona discharge reactor and set up for the experimental studies is shown in Fig. 1. Corona discharge was obtained using tungsten electrode of needle plane configuration. Needles with a sharp tip radius of 50 μm were used to ensure continuous discharge. High voltage and the ground electrode were fixed in a glass container of a capacity of 50 mL. A rotating spark gap (RSG) was used to generate square pulses, and the duration of pulses was controlled by varying the speed of RSG. The applied voltage magnitude is fixed in such a way that the visual streamer propagation is observed during the test. Iced water was continuously circulated around the reactor to avoid temperature rise. In top electrode, a port was provided for the sampling and analysis of different physiochemical parameters. More details are available elsewhere.35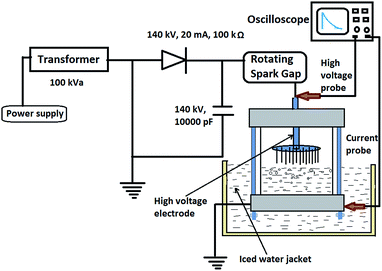 | ||
| Fig. 1 A Schematic diagram of experimental set up and multi-pin plane geometry corona discharge reactor. | ||
2.2. Growth media, bacterial cultures and disinfection study
Escherichia coli BL21 was used as model bacteria for the study of disinfection mechanism. A single colony of E. coli was picked from tryptone bile glucoronic agar (Himedia, India) plate and inoculated into 200 mL Luria broth (Himedia, India). Subsequently, the culture was incubated overnight in an orbital shaker (150 rpm) at 37 °C. In order to separate media and bacterial cells from the culture media, 10 mL of culture was taken, centrifuged at 7000 × g for 5 min and washed twice with physiological saline (0.85% NaCl, Rankem, India) prior to use. The initial cell count of approximately 107 cfu mL−1, was used in all the experiments. To quantify the amount of proteins released from bacterial cells, and also to visualize the DNA bands clearly, a high concentration of initial bacterial cell count was used for the study.Disinfection study was carried out at input voltage of 23 kV and 25 Hz frequency. Samples were withdrawn at different discharge time of 2, 4, 6, 8 and 10 min and the disinfection efficiency was evaluated by plating the samples on tryptone bile glucoronic agar media.
2.3. Molecular analysis
2.4. Bacterial morphology assessment
2.5. E. Coli interfacial potential measurement and size analysis
E. coli cells were incubated overnight for the measurement of surface potential in Zeta analyser (Horiba Scientific nanopartica, SZ 100), E. coli cells were then taken and centrifuged at 7000 × g for 5 min to remove broth. The pellet was isolated and washed two times with physiological saline and re-suspended in the same saline solution. Cell concentration of about 107 cfu mL−1 was used for the plasma treatment. Subsequently, 3 mL of treated and untreated cells at were withdrawn from the reactor at different time intervals. Prior to zeta potential analysis, the cells were sonicated for 5 min for proper dispersion in the medium. The zeta potential and electrophoretic mobility for each sample were calculated using the Smoluchowski equation.38 At least triplicate of samples were used, and each was scanned thrice to reduce error. Similarly, mean particle size diameter (Z-average) and poly-dispersive index (PI) of each sample were measured using the same instrument.2.6. FTIR analysis
The changes in functional groups of bacterial cell membrane upon plasma treatment were confirmed by Fourier Transformed Infrared Spectroscopy (FTIR). 10 mL of treated and untreated cell suspensions were centrifuged for 10 min at 7000 × g. The pellet was washed twice with physiological saline and lyophilized to remove moisture. The dried cells were then subjected to FTIR analysis (Perkin Elmer, USA).2.7. Estimation of nitrate concentration and conductivity
Nitrate ion concentration in liquid samples was measured using AS 11 (4 × 250 mm) column in ion chromatography (DX-100, Dionex, USA), equipped with electron conductivity detector. 3 mM methane sulfonic acid and 12 mM NaOH was used as eluent.Conductivity and pH of liquid samples were measured using multi-parameter meters (Eutech, CyberScan PC 510).
3. Results
3.1. Disinfection effect of pulse power technology (PPT)
A rapid disinfection capability of PPT was observed at applied pulsed voltage of 23 kV and 25 Hz frequency.Fig. 2, shows the variation of log reduction of E. coli cells as a function of time. The complete disinfection (7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log reduction) was obtained in 6 min of treatment time. Streamer discharge leads to enormous ROS/RNS generation in aqueous solution,22,35 and also increases the UV doses. ROS/RNS being strong oxidant may cause atom etching on cell membrane of bacteria.19 Also, UV irradiation and ROS/RNS may damage the DNA of bacterial cells. Individual and combined effect of system parameters such as voltage, frequency, pH, alkalinity, turbidity and presence of organic materials on the disinfection efficiency were extensively studied earlier,35 and it was noted that the disinfection efficiency is significantly affected by the above mentioned system parameters in PPT process.
log reduction) was obtained in 6 min of treatment time. Streamer discharge leads to enormous ROS/RNS generation in aqueous solution,22,35 and also increases the UV doses. ROS/RNS being strong oxidant may cause atom etching on cell membrane of bacteria.19 Also, UV irradiation and ROS/RNS may damage the DNA of bacterial cells. Individual and combined effect of system parameters such as voltage, frequency, pH, alkalinity, turbidity and presence of organic materials on the disinfection efficiency were extensively studied earlier,35 and it was noted that the disinfection efficiency is significantly affected by the above mentioned system parameters in PPT process.
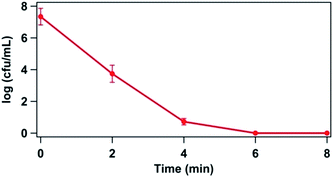 | ||
| Fig. 2 Log reduction of E. coli in physiological saline as a function of time for power delivery of 85.2 kW; applied voltage = 23 kV and frequency = 25 Hz. | ||
However, the mechanistic aspect of bacterial disinfection in PPT process was not explored. In the present study, morphological and molecular analysis was carried out to understand the mechanism of bacterial disinfection in the PPT process.
3.2. E. coli morphology upon plasma treatment
To observe the membrane deformities and physical changes due to plasma treatment, SEM analysis was employed.Interesting morphological changes were observed in the damaged cells after plasma treatment. The SEM images show the micropores, cytoplasm exudates, clumping, wrinkled appearance and complete wreckage of the cells (Fig. 3). The first image (Fig. 3a) shows the intact cells of E. coli with the rod shape, smooth surface and its characteristics size (around 2 μm). Upon application of high electric field (23 kV input voltage) during corona discharge, micropores appeared in the cells first (Fig. 3b) after 2 min of treatment. A similar explanation for bacterial cell death during plasma treatment is reported by Neumann et al.39 After the formation of micropores, the cytoplasm started oozing out (Fig. 3c) and this resulted in cell clumping (Fig. 3d). Moreover, upon plasma treatment, the aqueous solution becomes acidic.32 Due to high concentration of H+ ions, the bacterial cell potential neutralized, resulting in cell clumping. As the duration and intensity of plasma treatment increased, further damage occurred in the cells. As a result, more cytoplasm was oozed out, and cells were wrinkled further (Fig. 3e). Extended plasma treatment led to complete cell devastation and a large amount of cell debris was observed after 8 minutes of treatment (Fig. 3f). The ruptured cells were no longer intact and were often found in clumps or aggregates. Similar cell clumping behavior was observed during application of zinc oxide nanoparticles on E. coli cells.40
Membrane permeability assessment was performed by LDH release. Bacterial cells exposed to ROS and RNS demonstrated the time (energy supply) dependent LDH release (Fig. 4). A higher level of LDH was released after the treatment as compared to that in control. LDH release increased with time of treatment, which confirms membrane damage with treatment time. Dalai et al.16 also reported that the LDH leakage is an indication of membrane damage in bacterial cells.
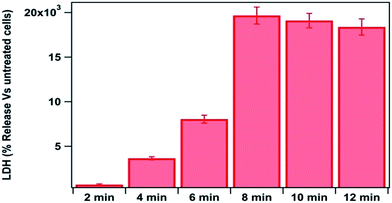 | ||
| Fig. 4 Assessment of membrane damage after different time of plasma treatment by LDH assay; power delivery was 85.2 W during the experimentation. | ||
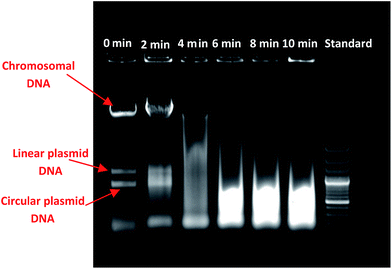
3.3. Fragmentation of chromosomal DNA
The DNA fragmentation study was conducted to prove that the changes were occurring in the bacterial cell at the molecular level, upon the application of plasma. This result is shown in Fig. 5. The characteristic band of E. coli chromosomal DNA was observed in an untreated sample. However, the DNA extracted from plasma treated cells shows important differences compared to DNA extracted from untreated cells. The DNA pattern in Lane 2 shows that the fragmentation in DNA started after 2 min of plasma treatment. A high degree of fragmentation in the form of smear was observed in Lane 3, after 4 min of treatment. Further increase in applied energy (after 6 min), led to increased DNA damage as seen in the Fig. 5 – Lanes 4, 5, and 6 as smears. Comparison with DNA size standard (Lane 7), confirmed that the bacterial chromosomal DNA got damaged leading to the formation of fragments of size lesser than 1000 bp.3.4. Leakage of proteins
Proteins are important biomolecules which have an important role in balancing the physiological activities in a bacterial cell. As shown in Fig. 6, plasma treatment resulted in the leakage of proteins through the membrane of E. coli cells. It was found that the concentration of proteins in the cell-free supernatant increased with treatment time while the concentration of residual proteins in intact cells decreased with time. A high concentration (37.52 ± 3.68 μg mL−1) of proteins in the cell-free supernatant was found to be present after 12 min of treatment. On the other hand, total initial proteins (51.09 ± 1.63 μg mL−1) of untreated E. coli cells decreased to 3.45 ± 2.71 μg mL−1 after 12 min of treatment. Thus, it is evident that the plasma treatment resulted in protein exudation from the bacterial cells, indicating the occurrence of damage inside the cells.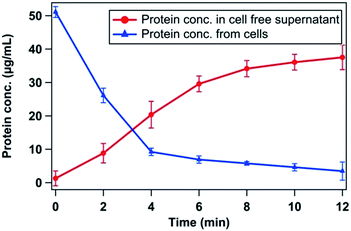 | ||
| Fig. 6 Leakage of protein from E. coli cells after different time of plasma treatment; power deliver was 85.2 W for the study. | ||
3.5. Zeta potential and cell size measurements
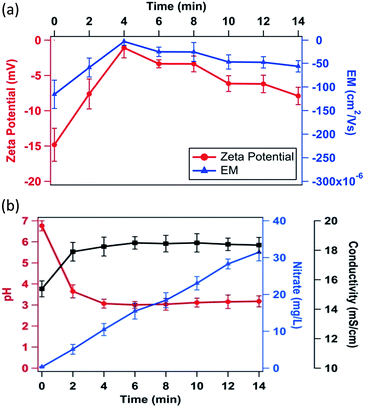
Zeta potential measurements were carried out to examine the effect of plasma-generated reactive species and aqueous environment on the membrane surface potential.As shown in Fig. 7a, untreated E. coli cells displayed zeta potential of −14.81 ± 2.08 mV in physiological saline. However, the potential neutralized to −1.08 ± 0.83 mV upon 4 min of corona discharge with an energy dissipation of 20.4 kJ. On the other hand, a further increase in treatment time showed a significant reversal of charge towards negative potential. After 14 min of corona discharge (85.9 kJ energy), the zeta potential was −7.9 ± 1.26 mV. Corresponding to zeta potential, electrophoretic mobility of cells also displayed similar behaviour (Fig. 7a). The aqueous environmental parameters such as pH, conductivity and nitrate concentrations (Fig. 7b) showed changes during treatment. The pH of the aqueous solution was significantly affected by corona discharge, and it changed from 6.8 ± 0.2 to 3.1 ± 0.2 after 4 min discharge. Further increase in discharge had insignificant effect on pH. Similarly, the conductivity of aqueous solution first increased and then stabilized. On the other hand, the nitrate concentration continuously increased upon corona discharge. Similar surface charge neutralization behaviour was recently observed by Arakha et al.40
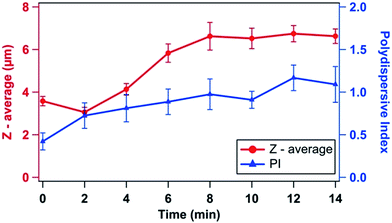
Cell size (Z-average) measurement and PI of individual and clumped E. coli cells were also performed to understand the changes in the colloidal behaviour of cells upon plasma treatment. Results of Z-average size (Fig. 8) indicate that bacterial cells were greatly influenced by plasma treatment. During the initial 2 min of treatment, Z-average size of E. coli cells decreased. However, with the increase in the exposure time to 4 min, the Z-average of clumped E. coli cells was found to increase and it increased steadily with further increase in the treatment time. As the bacterial cells were treated, a significant difference in their sizes was observed, and this was confirmed with the SEM images obtained for the same (Fig. 3). The heterogeneity in cell sizes resulted in increased PDI values for the treated cells, which was found to increase with the treatment time (Fig. 8). A similar trend of PI for dead cells has been reported in the literature.3,34
3.6. FTIR analysis
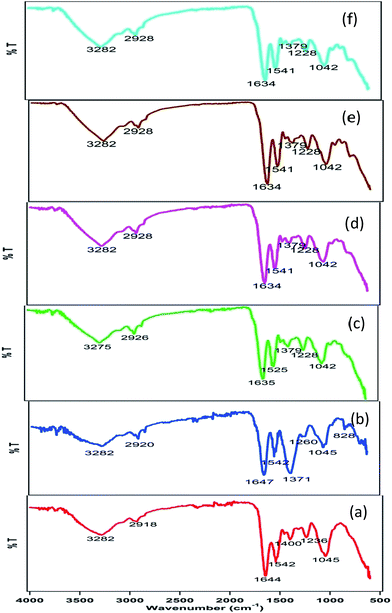
The FTIR spectra obtained for untreated, and plasma-treated E. coli cells are compared in Fig. 9. The spectrum of untreated cells showed the presence of broad band at 3282 cm−1 and peak at 2918 cm−1, corresponds to vibration of OH− due to enhanced hydration of bacterial cells and fatty acids region.41,42 The absorption peak at 1644 and 1542 cm−1 corresponds to amide I and amide II of proteins and peptides.41 The peaks at 1400 cm−1 showed the presence of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O symmetric stretching of COO– groups in amino acids and fatty acids.43 The characteristic peaks at 1236 might be the P
O symmetric stretching of COO– groups in amino acids and fatty acids.43 The characteristic peaks at 1236 might be the P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O asymmetric stretching of phosphodiesters in phospholipids. The broad peak corresponding to C–O–C and C–O bonds in rings of various polysaccharides in the cell wall was noted in the fingerprint region of 1045 cm−1.43 After 2 min of plasma treatment, the peak at 1400 cm−1 disappeared, which suggested that the C
O asymmetric stretching of phosphodiesters in phospholipids. The broad peak corresponding to C–O–C and C–O bonds in rings of various polysaccharides in the cell wall was noted in the fingerprint region of 1045 cm−1.43 After 2 min of plasma treatment, the peak at 1400 cm−1 disappeared, which suggested that the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O symmetric stretching of COO– groups in amino acids and fatty acids was largely affected.
O symmetric stretching of COO– groups in amino acids and fatty acids was largely affected.
A considerable increase in peak intensity at 1371 cm−1 suggested the involvement of C–N stretching vibration in amino acids.44 The appearance of peaks at 1260 cm−1 suggested several coordinate displacements in amide III band components of proteins.41 After 4 min of treatment, the peaks at 3282, 2920, 1647, 1542, 1371 and 1260 cm−1 shifted to 3275, 2926, 1635, 1525, 1379 and 1228, respectively. After 6 min of treatment, no considerable change in the FTIR spectra was observed. This indicated that perturbation process of all the membrane functional groups finished in 4–6 min of treatment time.
4. Discussion
Although several disinfection studies have been carried out earlier to demonstrate the antimicrobial activity of plasma treatment processes, the underlying mechanisms are only vaguely understood and explained. Here, we have used corona discharge as a plasma source due to its strong antimicrobial activity.26,29 In our previous study, we obtained 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log reduction of E. coli in 4 min of treatment at power delivery of 85.2 kW.35 The corona discharge in air above the liquid produced different ROS and RNS, which increase the acidity of water by releasing hydrogen ions.22,45 Plasma dissociates the atmospheric O2 and N2 and leads to the formation of NO2− and NO3− and the dissolution of NOx in water produces H+ in water, which can be understood by eqn (1)–(6).46,47
log reduction of E. coli in 4 min of treatment at power delivery of 85.2 kW.35 The corona discharge in air above the liquid produced different ROS and RNS, which increase the acidity of water by releasing hydrogen ions.22,45 Plasma dissociates the atmospheric O2 and N2 and leads to the formation of NO2− and NO3− and the dissolution of NOx in water produces H+ in water, which can be understood by eqn (1)–(6).46,47| N2 + e → 2N + e, | (1) |
| O2 + e → 2O + e, | (2) |
| N + O → NO, | (3) |
| NO + O → NO2 | (4) |
| 2NO2(aq) + H2O(l) → NO2− + NO3− + 2H+ | (5) |
| NO(aq) + NO2(aq) + H2O(l) → 2NO2− + 2H+ | (6) |
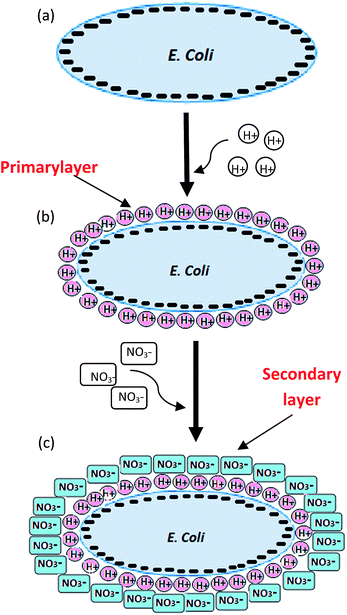
A sharp decrease in pH was observed after 4 min of corona discharge, which stabilized further. However, an increasing trend in NO3− concentration was observed with increasing discharge time (Fig. 7b). On the basis of experimental evidence and reported studies, it is hypothesized that the following sequence of events occur at the membrane of E. coli: (I) attachment of H+ ions and formation of primary layer due to negative potential of E. coli (II) neutralization of the bacteria surface potential, resulting in simultaneous disturbance in bacterial physiological system and molecular changes, and (III) attachment of NO3− on the primary layer and formation of a secondary layer (Fig. 10). These events could be responsible for the non-viability of cells and variation of zeta potential at different treatment times.
The presence of negatively charged lipopolysaccharide, proteins and lipoproteins on the outer membrane of E. coli renders the negative charge to it.48 It was confirmed from the zeta potential measurement studies also (Fig. 7a). Due to acidification of aqueous solution during corona discharge, the negatively charged E. coli was sharply neutralized by lighter H+ ions. Therefore, a reduction in zeta potential was observed during first 4 min of treatment (Fig. 7a and b). In parallel, the concentration of nitrate was also found to be increasing with the treatment time (Fig. 7b), which could slow down the charge neutralization process. However, being lighter in mass, H+ ions could move faster compared to heavier NO3− ions in water. Therefore, the charge neutralization by H+ ions in first 4 min was the predominant mechanism. After 4 min of treatment, the reduction in pH was insignificant. However, the nitrate concentration was found to be increasing. Similar trend of pH change during different electrical discharge treatment methods with liquid water has been reported by other researchers.32,49 However, the reason for such a trend was not very clear. As a result of low pH (high H+ concentration), NO3− ions could be attracted towards the primary layer of H+ ions and attached as a secondary layer due to electrostatic force of attraction. Therefore, negative zeta potential was observed after 4 min of treatment. Once the NO3− ions got saturated with oppositely charged H+ ions, equilibrium was achieved and constant zeta potential was observed after 10 min of treatment.
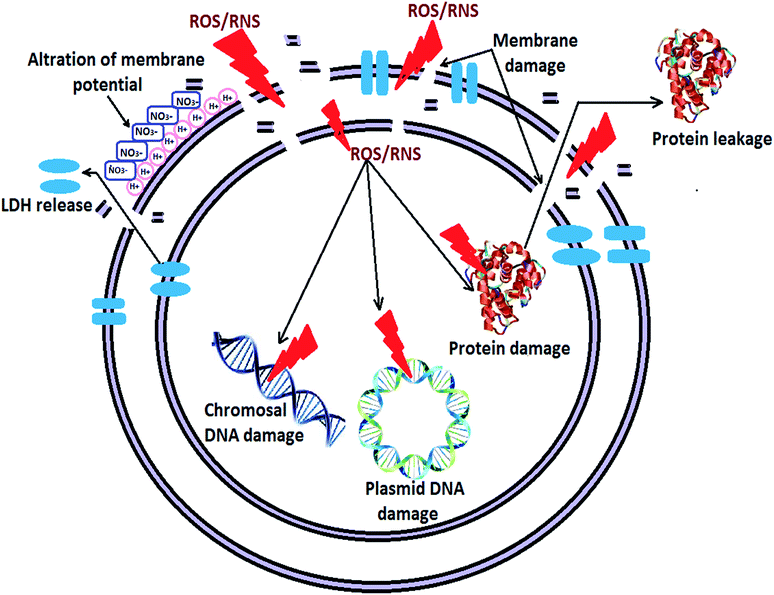
This observation indicates that possibly the interaction between reactive species and bacterial membrane surface results in the perturbation in membrane potential, which could further trigger the reaction inside the bacterial cell, leading to the cellular damage. SEM analysis and molecular analysis were conducted to confirm the bacterial non-viability. Increasing concentrations of ROS, RNS, aqueous electrons, and acidification of aqueous solution neutralize the interfacial potential of bacteria, suggesting the neutralization of functional group present on the lipopolysaccharides and proteins of the bacterial membrane. As a result of neutralization, the energy released during charge neutralization process is possibly utilized either in the change in membrane tension or the production of ROS inside the cell.50 The inherent production of ROS by plasma channel or ROS generated during bacterial stress condition might enhance the antimicrobial activity.19 Also, due to very high electric field, the membrane potential of E. coli cells may get disturbed, which in turn add up to the antimicrobial activity. To understand the change in membrane integrity, SEM analysis was performed, and it confirmed the formation of micropores, indicating the disintegration of the cellular membrane upon plasma treatment (Fig. 3). As a result of micropore formation, the cytoplasm started oozing out. The cytoplasm exudates and neutralized membrane lead to the cell clumping, which can be seen in the Fig. 3d. The wrinkles on the cells appeared due to excessive cytoplasm loss from cells (Fig. 3e), and further complete devastation of cells could be observed (Fig. 3f). Similar abnormal textures of bacteria were observed during bacterial inactivation study using photoactivated ZnO nano particles.40,51 The abnormal morphology of bacterial cell after corona discharge treatment is also reported by Korachi et al.29 Present study provides additional information about the detailed morphological changes such as micropore formation, wrinkle formation and appearance of cell debris, leading to a better understanding of the bacterial membrane deformities.
Assessment of the membrane permeability through LDH release confirmed that the membrane damage increased with treatment time (Fig. 4). The concept of assessment of membrane permeability through LDH release has been used in many earlier studies. It has been shown that increase in membrane permeability is an indication of cell membrane deformities and cell death.52,53 The changes occurred to surface chemistry groups in the bacterial membrane was investigated through FTIR analysis. Considerable shifting of the peak at 1400 cm−1 and a significant increase of peak intensity at 1371 cm−1 – indicated that COO groups of amino acids and fatty acids are greatly affected by the treatment. Considerable changes in the signature spectrum of live bacterial cells explain the fact that the bacterial membrane chemistry also got affected by plasma treatment. Similar FTIR signature at 4, 6, 8 and 10 min treatment time, explains that all the changes occurred within 4 min of treatment time, at an energy delivery of 20.5 kJ.
The effect of the plasma treatment on bacteria cell viability is further explored using molecular level DNA and protein degradation study. The oxidative damage of bacterial DNA and protein was observed (Fig. 5 and 6). ROS and RNS damaged the DNA, causing single or double strand break and directly affecting the sugar and base moiety, which form the backbone of the DNA.54 The oxidation of proteins by reactive species has also been well documented. It has been reported that oxidation of sulfhydryl group, reduction of disulphides, oxidative adduction of amino acid group closed to metal binding site via metal catalyzed oxidation, modification in prosthetic group, protein–protein interaction and peptide fragmentation are main mechanisms for the protein damage in cells.55,56 From Fig. 5, it is clear that the E. coli genomic DNA was completely broken to smaller fragments after 6 min of plasma treatment. Also, as shown in Fig. 6, plasma treatment could enhance the protein leakage through bacterial membrane. Interestingly, more leakage of proteins occurred in first 8 min of treatment, which accounted for 70% of total proteins. Protein content in cell-free supernatant was analysed during the plasma treatment of E. coli. Higher proteins content in the supernatant as compared to that in control indicated the membrane permeabilization caused by plasma treatment. Therefore, it may be concluded that the plasma treatment has a remarkable effect on bacterial proteins loss, resulting in their death. This is in agreement with the results of disinfection of E. coli and Staphylococcus aureus by electrolyzed oxidizing (EO) water.57 They have also reported the leakage of significant amount of proteins from bacterial cells after EO treatment.
The Z-average is the indication of average size of colloidal particles (bacterial cells) in the aqueous phase. The decrease in Z-average in first 2 min of treatment could be due to the leakage of cytoplasm, following the formation of the micropores. Scattered individual E. coli cells were also observed without clumps in SEM image taken after 2 min of treatment (Fig. 3b). Due to the rapid neutralization of negative membrane potential by H+ ions (in low pH conditions), and extracellular cytoplasm release, bacteria had undergone clumping. Therefore, an increase in Z-average of clumped cells was observed after 2 min of treatment time. On the other hand, PI is a function of heterogeneity of particle size. Heterogeneity of bacterial cells solely depends on physiological states of cells (e.g. rich, starved and dead).3 Significant morphological and physiological changes of bacterial cells were observed after treatment, which was the reason for increasing PI value with increasing time. Increasing PI trend is the indication of heterogeneity of bacterial cells due to their abnormal morphology after plasma treatment. Thus, PI provided the direct information about the abnormality of bacterial cells during disinfection process.
Based on the above results, it can be suggested that the PPT exhibited ROS/RNS dependent disinfection of bacterial cells through the following multiple steps: (a) destabilization of bacterial membrane potential; (b) damage of bacterial membrane and leakage of membrane enzyme (LDH) and proteins; (c) ROS/RNS induced fragmentation of DNA and (d) ultimately the cell death, which increases the Z-average and PI of the aqueous solution. The complete picture of sub-cellular disinfection mechanism in PPT is shown in Fig. 11.
5. Conclusions
Physical and molecular level disinfection mechanism of bacterial cells has been studied in detail. Based on the SEM, FTIR and LDH assay, insights into the occurrence of biophysical events on the bacterial cell membrane were gained. Firstly, the change in the bacterial surface potential was explored using zeta potential measurement, and this was used as a guide for understanding the mechanism underlying the interaction of ROS, RNS, and aqueous environment with bacterial membrane. Secondly, molecular level DNA fragmentation and protein release in aqueous environment confirmed the bacterial killing. The total protein loss confirmed the oxidative degradation of protein molecules into smaller amino acids molecules or mineralized products (CO2, H2O, NO3−, etc.). Thus, these studies provided an insight into the disinfection mechanism of PPT.Acknowledgements
The authors place on record their gratitude to the Department of Science and Technology (DST), Government of India for the financial support for this study.References
- UNESCO, 2013, http://www.unwater.org/water-cooperation-2013/water-cooperation/facts-and-figures/en/, Retrieved on Aug 7, 2015.
- E. Corcoran, C. Nellemann, B. Elaine, R. Bos, O. David and H. Savelli, Sick water? The central role of wastewater management in sustainable development, 2010 Search PubMed.
- C. Wang, T. Chang, H. Yang and M. Cui, Food Control, 2015, 47, 231–236 CrossRef CAS.
- WHO, 2014, http://www.who.int/water_sanitation_health/diseases/en/, Retrieved on Aug 7, 2015.
- M. Eddy, G. Tchobanoglous, F. L. Burton and H. D. Stensel, Wastewater engineering: Treatment and reuse, McGraw-Hill, Boston, 4th edn, 2003 Search PubMed.
- C. Tian, R. Liu, H. Liu and J. Qu, Water Res., 2013, 47, 5901–5910 CrossRef CAS PubMed.
- X. Zhang, S. Echigo, H. Lei, M. E. Smith, R. A. Minear and J. W. Talley, Water Res., 2005, 39, 423–435 CrossRef CAS PubMed.
- E. Ortega-Gómez, P. Fernández-Ibáñez, M. M. Ballesteros Martín, M. I. Polo-López, B. Esteban García and J. A. Sánchez Pérez, Water Res., 2012, 46, 6154–6162 CrossRef PubMed.
- S. T. Summerfelt, M. J. Sharrer, S. M. Tsukuda and M. Gearheart, Aquacultural Engineering, 2009, 40, 17–27 CrossRef.
- Y. Penru, A. R. Guastalli, S. Esplugas and S. Baig, J. Photochem. Photobiol., A, 2012, 233, 40–45 CrossRef CAS.
- J. Jeong, C. Kim and J. Yoon, Water Res., 2009, 43, 895–901 CrossRef CAS PubMed.
- J. Marugán, R. van Grieken, C. Sordo and C. Cruz, Appl. Catal., B, 2008, 82, 27–36 CrossRef.
- S. Dong, J. Feng, M. Fan, Y. Pi, L. Hu, X. Han, M. Liu, J. Sun and J. Sun, RSC Adv., 2015, 5, 14610–14630 RSC.
- S. Yu, T. Lin and W. Chen, RSC Adv., 2014, 4, 31370–31377 RSC.
- B. I. Kharisov, H. V. R. Dias, O. V. Kharissova, V. M. Jimenez-Perez, B. O. Perez and B. M. Flores, RSC Adv., 2012, 2, 9325–9358 RSC.
- S. Dalai, S. Pakrashi, R. S. S. Kumar, N. Chandrasekaran and A. Mukherjee, Toxicol. Res., 2012, 1, 116 RSC.
- T. Parandhaman, A. Das, B. Ramalingam, D. Samanta, T. P. Sastry, A. B. Mandal and S. K. Das, J. Hazard. Mater., 2015, 290, 117–126 CrossRef CAS PubMed.
- Y. Long, J. Ni and Z. Wang, Water Res., 2015, 84, 198–206 CrossRef CAS PubMed.
- M. Moreau, N. Orange and M. G. J. Feuilloley, Biotechnol. Adv., 2008, 26, 610–617 CrossRef CAS PubMed.
- S. Maheux, D. Dubey, T. Belmonte, C. Penney, H. M. Cauchie, F. Clement and P. Choquet, RSC Adv., 2015, 5, 42135–42140 RSC.
- B. R. Locke, M. Sato, P. Sunka, M. R. Hoffmann and J.-S. Chang, Ind. Eng. Chem. Res., 2006, 45, 882–905 CrossRef CAS.
- P. Lukes, E. Dolezalova, I. Sisrova and M. Clupek, Plasma Sources Sci. Technol., 2014, 23, 015019 CrossRef.
- P. Attri, B. Arora and E. H. Choi, RSC Adv., 2013, 3, 12540–12567 RSC.
- R. Hackam and H. Akiyama, IEEE Trans. Dielectr. Electr. Insul., 2000, 7, 654–683 CrossRef CAS.
- C. Wang, Y. Wu and G. Li, J. Electrost., 2008, 66, 71–78 CrossRef CAS.
- V. Joubert, C. Cheype, J. Bonnet, D. Packan, J.-P. Garnier, J. Teissié and V. Blanckaert, Water Res., 2013, 47, 1381–1389 CrossRef CAS PubMed.
- J. Julák, V. Scholtz, S. Kotúčová and O. Janoušková, Phys. Med. Biol., 2012, 28, 230–239 Search PubMed.
- A. H. Sari and F. Fadaee, Surf. Coat. Technol., 2010, 205, S385–S390 CrossRef CAS.
- M. Korachi, Z. Turan, K. Şentürk, F. Şahin and N. Aslan, J. Electrost., 2009, 67, 678–685 CrossRef CAS.
- J. Hoon Park, N. Kumar, D. Hoon Park, M. Yusupov, E. C. Neyts, C. C. W. Verlackt, A. Bogaerts, M. Ho Kang, H. Sup Uhm, E. Ha Choi and P. Attri, Sci. Rep., 2015, 5, 13849 CrossRef PubMed.
- T. Ohshima, M. Sato and M. Saito, J. Electrost., 1995, 35, 103–112 CrossRef CAS.
- R. Burlica, M. J. Kirkpatrick and B. R. Locke, J. Electrost., 2006, 64, 35–43 CrossRef CAS.
- D. A. Lytle, E. W. Rice, C. H. Johnson and K. R. Fox, Appl. Environ. Microbiol., 1999, 65, 3222–3225 CAS.
- K. A. Soni, A. K. Balasubramanian, A. Beskok and S. D. Pillai, Curr. Microbiol., 2008, 56, 93–97 CrossRef CAS PubMed.
- R. K. Singh, V. Babu, L. Philip and S. Ramanujam, Chem. Eng. J., 2016, 284, 1184–1195 CrossRef CAS.
- M. M. Bradford, Anal. Biochem., 1976, 72, 248–254 CrossRef CAS PubMed.
- D. M. Brown, M. R. Wilson, W. MacNee, V. Stone and K. Donaldson, Toxicol. Appl. Pharmacol., 2001, 175, 191–199 CrossRef CAS PubMed.
- A. V. Delgado, F. Gonzalez-Caballero, R. J. Hunter, L. K. Koopal and J. Lyklema, Measurement and interpretation of electrokinatic phenomena, Pure Appl. Chem., 2005, 77, 1753–1805 CAS.
- E. Neumann and I. Sugar, J. Electroanal. Chem. Interfacial Electrochem., 1983, 156, 479 CrossRef.
- M. Arakha, M. Saleem, B. C. Mallick and S. Jha, Sci. Rep., 2015, 5, 9578 CrossRef CAS PubMed.
- D. Naumann, D. Helm and H. Labischinski, Nature, 1991, 351, 81–82 CrossRef CAS PubMed.
- A. Kamnev, Spectrosc. Int. J., 2008, 22, 83–95 CrossRef CAS.
- R. Davis and L. Mauer, Current Research, Technology and Education Topics, 2010, 1582–1594 Search PubMed.
- R. Vinodh, L. K. Elumalai, D. Sangeetha and R. Rengasamy, J. Biosci. Technol., 2009, 1, 45–51 Search PubMed.
- J.-L. Brisset, B. Benstaali, D. Moussa, J. Fanmoe and E. Njoyim-Tamungang, Plasma Sources Sci. Technol., 2011, 20, 034021 CrossRef.
- G. Sathiamoorthy, S. Kalyana, W. C. Finney, R. J. Clark and B. R. Locke, Ind. Eng. Chem. Res., 1999, 1844–1855 CrossRef CAS.
- J. H. Seinfeld and S. N. Pandis, Atmospheric chemistry and physics, Wiley, Newyork, 1998, p. 1326 Search PubMed.
- C. S. Alves, M. N. Melo, H. G. Franquelim, R. Ferre, M. Planas, L. Feliu, E. Bardaji, W. Kowalczyk, D. Andreu, N. C. Santos, M. X. Fernandes and M. A. R. B. Castanho, J. Biol. Chem., 2010, 285, 27536–27544 CrossRef CAS PubMed.
- W. Bian, X. Song, J. Shi and X. Yin, J. Electrost., 2012, 70, 317–326 CrossRef CAS.
- P. J. P. Espitia, N. F. F. Soares, J. S. dos Reis Coimbra, N. J. de Andrade, R. S. Cruz and E. A. Alves Medeiros, Food Bioprocess Technol., 2012, 5, 1447–1464 CrossRef CAS.
- K. Kairyte, A. Kadys and Z. Luksiene, J. Photochem. Photobiol., B, 2013, 128, 78–84 CrossRef CAS PubMed.
- T. Y. Suman, S. R. Radhika Rajasree and R. Kirubagaran, Ecotoxicol. Environ. Saf., 2015, 113, 23–30 CrossRef CAS PubMed.
- S. Dalai, S. Pakrashi, M. Bhuvaneshwari, V. Iswarya, N. Chandrasekaran and A. Mukherjee, Aquat. Toxicol., 2014, 146, 28–37 CrossRef CAS PubMed.
- H. Sies and C. F. Menck, Mutat. Res., 1992, 275, 367–375 CAS.
- L. Fucci, C. N. Oliver, M. Coon and E. R. Stadtman, Proc. Natl. Acad. Sci. U. S. A., 1983, 80, 1521–1525 CrossRef CAS.
- E. R. Stadtman, Free Radical Biol. Med., 1990, 9, 315–325 CrossRef CAS PubMed.
- X. Zeng, W. Tang, G. Ye, T. Ouyang, L. Tian, Y. Ni and P. Li, J. Food Sci., 2010, 75, 253–260 CrossRef PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2016 |