One-step in situ calcination synthesis of g-C3N4/N-TiO2 hybrids with enhanced photoactivity
Shanfu Sun,
Mingxuan Sun*,
Yalin Fang,
Ying Wang and
Huiping Wang
School of Materials Engineering, Shanghai University of Engineering Science, Shanghai 201620, China. E-mail: mingxuansun@sues.edu.cn; smxalan@163.com; Fax: +86 21 67791201; Tel: +86 21 67791474
First published on 26th January 2016
Abstract
A series of composites, based on graphitic carbon nitride and nitrogen-doped titanium dioxide (g-C3N4/N-TiO2), were prepared by a solid-phase calcination process, in which a homogenous mixture containing melamine and TiN was calcined at 550 °C for 3 h in air. Various techniques such as X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FT-IR), scanning electron microscopy (SEM), transmission electron microscopy (TEM), X-ray photoelectron spectroscopy (XPS), and UV-vis diffuse reflectance spectroscopy (UV-vis DRS) characterized the as-prepared composites. The results suggested that nitrogen was incorporated into the TiO2 lattice that formed the N–Ti bonds, which was followed by the successful combination of g-C3N4 with N-TiO2. A red-shift of absorption edge in the light region and more absorption in the visible light region were observed for the prepared composites. The synthesized composites exhibited excellent photocatalytic activities, which were evaluated by the degradation of methylene blue (MB) after irradiation with visible light. Especially, g-C3N4/N-TiO2 composites, prepared using melamine and TiN with a mass ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, showed the highest photocatalytic activity, and the corresponding rate of MB degradation was 0.028 min−1, which was 1.9 times greater than that of N-TiO2 (0.015 min−1). Furthermore, the photoelectrochemical performances of the samples on illumination with UV-visible light were evaluated. Remarkable enhancement of photoelectrochemical response of g-C3N4/N-TiO2 composites in UV-visible light, compared to pristine N-TiO2, was noticed. The co-modifications of TiO2 with N doping and g-C3N4 coupling resulted in an enhancement in the capacity of light absorption and an improvement in the transfer of charge carriers, which predominantly contributed to the improvement of photoactivity. This study develops a new and facile fabrication procedure for g-C3N4/N-TiO2 hybrids and represents an important step in the improvement of solar energy conversion using cost-effective materials.
1, showed the highest photocatalytic activity, and the corresponding rate of MB degradation was 0.028 min−1, which was 1.9 times greater than that of N-TiO2 (0.015 min−1). Furthermore, the photoelectrochemical performances of the samples on illumination with UV-visible light were evaluated. Remarkable enhancement of photoelectrochemical response of g-C3N4/N-TiO2 composites in UV-visible light, compared to pristine N-TiO2, was noticed. The co-modifications of TiO2 with N doping and g-C3N4 coupling resulted in an enhancement in the capacity of light absorption and an improvement in the transfer of charge carriers, which predominantly contributed to the improvement of photoactivity. This study develops a new and facile fabrication procedure for g-C3N4/N-TiO2 hybrids and represents an important step in the improvement of solar energy conversion using cost-effective materials.
1. Introduction
Solar energy is an alternative and sustainable source of energy for addressing the global challenges of energy crisis and environmental pollution.1 Semiconductor-based materials, which are used for harvesting visible light, are significantly efficient, stable, and inexpensive and hence are considered to be the key factors governing the utilization of solar energy. Therefore, the development of an efficient, visible-light-driven semiconductor-based material has been extensively investigated. Amongst various semiconductor materials studied, titanium dioxide (TiO2) is by far the most preferred material in a number of sunlight-driven renewable energy and environmental technologies, owing to its low cost, availability, excellent stability and nontoxicity.2 Unfortunately, TiO2 fails to utilize visible light and can absorb only ultraviolet radiations, which account for approximately only 4% of solar energy, due to its wide band gap energy of 3.0–3.2 eV.3 Moreover, fast recombination of photoinduced electron–hole pairs is another factor that hampers its full range of practical applications. As a result, a large number of efforts have been devoted to modify TiO2 by doping metallic4–6 and nonmetallic elements,7 surface sensitization,8 and deposition of noble metals,9,10 which are aimed at utilizing both the merits of visible light response and efficient separation of electron–hole pairs.The doping of TiO2 with nitrogen dopants for visible light-response has now been confirmed in a variety of studies and attempts have been made to enhance its performance by further modifications. Wu et al.11 successfully prepared N-TiO2 particles from a water-based TiO2 sol by hydrothermally treating it with triethylamine at a low temperature. This simple and effective method provided N-TiO2 nanoparticles, which degraded methyl orange excellently under visible light. Amongst these modifying TiO2 photocatalysts, N-doped TiO2 is one of the most studied visible-light photocatalysts, since it is inexpensive, stable, and showed high activity, as reported by Asahi et al.12 It was believed that the nitrogen atoms substituted the oxygen sites in the lattice and were responsible for the visible-light activity. A number of publications have reported the preparation of N-doped TiO2 by physical or chemical methods, including sol–gel,13–15 sputtering,12,16 ion implantation,17 mechanochemical,18 and plasma-enhanced chemical vapor deposition methods.19
Coupling of TiO2 with other visible-light-responsive semiconductors to prepare composite materials seems particularly meaningful and efficient. Very recently, graphitic carbon nitride (g-C3N4), which has a band gap of ca. 2.7 eV and a more negative conduction band as compared to that of TiO2, has been a hot-topic of research. Wang et al.20 were the first to report an organic, metal-free polymeric semiconductor, g-C3N4, as an efficient photocatalyst for the production of hydrogen by splitting up of the water molecules in the presence of visible light, which inspired the design of the photocatalytic system, in the search for energy production. g-C3N4 is an outstanding photocatalyst due to its high reducing power and visible-light adsorption. In addition to this, g-C3N4 is also inexpensive, since this metal-free semiconductor can be synthesized by simple heating of urea or melamine at 500–600 °C. Therefore, sunlight harvesting, photocatalytic stability, and effective charge transfer capability of g-C3N4 make it an excellent candidate for photocatalysis.21 Up to date, a large number of g-C3N4-based composites, such as g-C3N4/MoS2,22 Ag2O/g-C3N4,23 and TiO2/g-C3N4,24 have been reported.
In order to make further use of the advantages offered by g-C3N4 and N-TiO2, composites of N-TiO2 and g-C3N4 were proposed (g-C3N4/N-TiO2). In this regard, Zhou et al.25 synthesized a series of composites based on g-C3N4 and N-TiO2 by a pyrolysis process of urea and Ti(OH)4, and the photocatalytic activities of the as-prepared photocatalysts were investigated for photoreduction of CO2 under simulated light irradiation. Wang et al.26 synthesized a series of porous N-doped TiO2/g-C3N4 heterostructures through a microware-assisted route. The photodegradation behaviors of rhodamine B (RhB) and methylene blue (MB) in the presence of prepared photocatalysts were studied using visible light (λ > 400 nm) irradiation. Therefore, combining N-TiO2 with g-C3N4 to synthesize composite photocatalysts seems to be a facile route to a heterojunction structure, which not only enhances the photoactivity, but also possesses the charge transfer capability and widens the scope of the photocatalytic system. However, the procedures for the preparation of most of the reported g-C3N4/N-TiO2 composites, are complex and hence their applications are restricted.
In this study, the synthesis of a series of composites based on g-C3N4 and N-TiO2 (g-C3N4/N-TiO2) by a facile calcination process, with available TiN and melamine precursors in air atmosphere, is reported. The synthetic route employed not only ensures the successful growth of N-TiO2 nanostructures on lamellar g-C3N4, but also a high dispersion of N-TiO2 nanostructures on g-C3N4 without aggregation. Moreover, by this way, the g-C3N4/N-TiO2 composite catalysts acquire porous structures and large surface areas, thereby increasing the contact area for the objective pollutants. The photodegradation behavior of methylene blue (MB) on irradiation with visible light (λ > 420 nm), in the presence of the prepared photocatalysts, was studied. Based on the relative band positions of the two materials and the experimental results, the photocatalytic mechanism of g-C3N4/N-TiO2 heterojunctions was postulated. A simple synthetic method for g-C3N4/N-TiO2 hybrids will pave a new path for the development of highly efficient catalysts.
2. Experimental section
2.1 Catalysts preparation
The chemicals melamine (C3H6N6, >99%) and TiN were obtained from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China) and used as received without further treatments.A series of g-C3N4/N-TiO2 nanocomposites were prepared via a facile calcination of a mixture of TiN and melamine. The preparation process is illustrated in Scheme 1. Briefly, a certain amount of melamine and TiN were vigorously grinded to obtain a homogeneous mixture and then transferred to porcelain crucible with cover. The calculation process was conducted at 550 °C for 3 h in muffle furnace at atmospheric pressure in air. The investigated mass ratios of melamine and TiN were 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, 2
3, 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, 3
3, 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, and 4
3, and 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, and the relevant products were denoted as CT-1, CT-2, CT-3 and CT-4, respectively. In addition, blank N-TiO2 and g-C3N4 were synthesized via the same procedure as that of g-C3N4/N-TiO2 nanocomposites except the addition of melamine or TiN, respectively.
3, and the relevant products were denoted as CT-1, CT-2, CT-3 and CT-4, respectively. In addition, blank N-TiO2 and g-C3N4 were synthesized via the same procedure as that of g-C3N4/N-TiO2 nanocomposites except the addition of melamine or TiN, respectively.
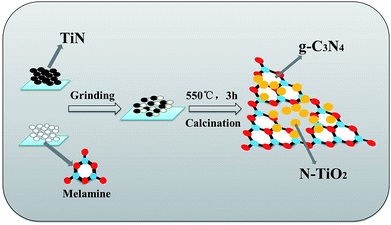 | ||
| Scheme 1 Scheme for the preparation of g-C3N4/N-TiO2 nanocomposites by one-step in situ calcination. | ||
2.2 Characterizations
X-ray diffraction (XRD) patterns were recorded on a Panalytical X'Pert X-ray diffractometer (Holland) using Cu-Kα radiation operated at 40 kV and 40 mA. Fourier transform infrared (FT-IR) spectra were obtained using a Shimadzu IR Prestige 21 spectrometer. The morphologies and microstructures of the samples were characterized by scanning electron microscope (SEM: JEOL JSM-7000F, Japan) and transmission electron microscope (TEM: FEI Tecnai F20, USA). The UV-vis diffuse reflectance spectra (DRS) were recorded between 200 and 800 nm using a Shimadzu UV 3600 (Japan) spectrophotometer. The X-ray photoelectron spectroscopy (XPS) analysis was performed on a RBD upgraded PHI-5000 CESCA system (Perkin-Elmer) with Al/Mg–K radiation.2.3 Photocatalytic measurements
The photocatalytic activities of the samples were tested by the degradation of methyl blue (MB) under visible light irradiation. The visible light source for photocatalysis was a 500 W xenon lamp (CHF-XM35, Trusttech Co., Ltd., Beijing) equipped with a cut filters (λ > 420 nm). The light intensity of the lamp was 120 mW cm−2, which was measured by a light-intensity meter (FZ-A, Photoelectric Instrument Factory of Beijing Normal University). In a typical process, 15 ml of MB solution with an initial concentration of 5 mg l−1 and 7.5 mg of photocatalyst were added to a 50 ml quartz reactor. Prior to irradiation, the suspension was agitated in darkness for 2 hours to ensure adsorption/desorption equilibrium. Once light illumination began, 5 ml of the suspension was sampled at given time intervals, centrifuged, and filtered to remove the photocatalysts. The remaining concentration of MB was analyzed using a UV-vis spectrophotometer (Shimadzu UV 1601-PC, Japan) in the wavelength range of 400–800 nm. The decoloration rate was reported as Ct/C0 and −ln(Ct/C0), respectively, where Ct was the pollutants concentration after adsorption or photocatalysis and C0 was initial concentration.2.4 Photoelectrochemical measurements
The photoelectrochemical measurements were carried out on a CHI-660E electrochemical workstation (Shanghai Chenhua Instrument Co., Ltd, China) in a three-electrode cell with a quartz window. The prepared samples with an active area of 0.72 cm2, a platinum wire, and saturated calomel electrode (SCE) were used as working electrodes, counter electrode, and reference electrode, respectively. The light source used here was the same as that of photocatalytic measurements with an UV-visible light intensity of 165 mW cm−2. Electrochemical impedance spectra (EIS) were recorded in the frequency range from 0.01 Hz to 100 kHz. All measurements were carried out in 0.5 M Na2SO4 aqueous electrolyte under ambient conditions at room temperature.3. Results and discussion
3.1 Characterization of the as-prepared samples
The crystal structures of the as-prepared samples were studied by XRD (Fig. 1). Fig. 1A shows the XRD patterns of pure TiO2 (P25), g-C3N4, N-TiO2, TiN, and CT-4 samples. As shown in Fig. 1A, P25 was composed of anatase and rutile phase, with the anatase content of 88%. The XRD pattern of pure g-C3N4 exhibited two diffraction peaks at 13.1° and 27.4°, which corresponded to (100) and (002) diffraction planes.27 These were attributed to the characteristic in-planar structural packing and inter-planar stacking peaks of the aromatic systems in graphite-like carbon nitride, respectively.20 The results indicated that g-C3N4 could be obtained by a calcination process. Pure TiN shows two distinct peaks at 36.7° and 42.6°, which can be attributed to (111) and (200) crystal planes (PDF 03-065-0565). After calcination at 550 °C for 3 h, peaks corresponding to TiN disappeared, whereas only the peaks corresponding to TiO2 appeared, suggesting the complete transformation of TiN into TiO2. The diffraction peaks at 2θ values of 25.4°, 27.5°, 37.8°, 48.0°, 55.0°, and 56.6° corresponded to the (101), (110), (004), (200), (211), and (220) (PDF 03-065-5714) (PDF 03-065-0191) crystal planes of N-TiO2. Hence, the as-prepared N-TiO2 was composed of both, the anatase and rutile phases. Based on the integration of the intensities of anatase (101) and rutile (110) peaks, the rutile phase was found to be 15%. Scherrer's equation was applied to the anatase (101) planes. The crystallite sized was calculated and found to be around 30 nm for N-TiO2. In addition to this, Fig. 1B showed that the diffraction peaks at 25.4° for the (101) planes of N-TiO2 were slightly higher shift compared to that of P25 (25.3°), which indicated that nitrogen was doped into TiO2 and this resulted in lattice distortion. The diffraction peaks for the (110) plane of CT-4 were slightly shifted higher, became broader and stronger than that of N-TiO2, which was probably due to the overlapping of (110) plane of N-TiO2 and (002) plane of g-C3N4. These results were further confirmed by the XRD patterns of g-C3N4/N-TiO2 with different amounts of g-C3N4 (Fig. 1C). Fig. 1D provided the magnification of (110) and (101) planes in the range of 24–30° of Fig. 1C, for further investigation. The intensities of peak around 27.5° increased gradually with an increase in the amount of g-C3N4 in the composites. The results indicated that CT was composed of g-C3N4 and N-TiO2.The FTIR spectra of g-C3N4, N-TiO2, and CT-4 samples are shown in Fig. 2. The FTIR peaks of the as-prepared g-C3N4 were very similar to the reports published earlier.28 The peaks that were centered at about 1639, 1569, 1460, 1410, 1329, and 1250 cm−1 were ascribed to the typical stretching modes of CN heterocycles.29 Additionally, a broad peak around 3150 cm−1 corresponded to the stretching modes of terminal NH2 of NH groups at the defect sites of the aromatic ring, while the absorption band at 808 cm−1 was attributed to the characteristic breathing mode of tri-s-triazine units.28 For CT-4 and N-TiO2, the main peaks appearing at 400–700 cm−1 were attributed to Ti–O stretching and Ti–O–Ti bridge stretching modes in TiO2.30 Also, the inserted figure showed slightly broadened absorption bands at around 1380–1500 cm−1 in the CT-4 sample, which could be attributed to the characteristic stretching modes of g-C3N4, suggesting the presence of g-C3N4 in the g-C3N4/N-TiO2 composites.
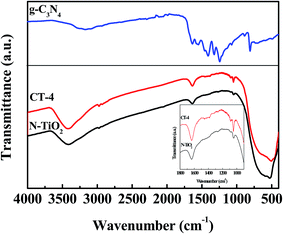 | ||
| Fig. 2 FTIR spectra of g-C3N4, N-TiO2, and CT-4 samples. The inset is the wavenumber range from 1800 to 800 cm−1 for CT-4 and N-TiO2 samples. | ||
The morphologies of the as-prepared samples were examined by FE-SEM (Fig. 3A and B), TEM (Fig. 3C), and HR-TEM (Fig. 3D). As shown in Fig. 3A and B, sphere nanoparticles with grain sizes around 50 nm could be seen. It was difficult to differentiate between the morphologies of pure N-TiO2 and g-C3N4/N-TiO2 composites. The TEM images provided a clearer picture of the two components. As seen in Fig. 3C, g-C3N4 displayed a long and thin lamellar shape and had an amorphous structure, whereas N-TiO2 showed uniform particles with sizes almost consistent with SEM results. N-TiO2 loading on the g-C3N4 surface displayed a stable and intimate interfacial contact, although it was ultrasonically treated, prior to TEM study, which contributed to the improved transfer of charge carriers. Fig. 3D showed the presence of two different interplanar spacings (d) at 0.352 and 0.233 nm that corresponded to the (101) and (112) planes of anatase form of TiO2,31 respectively. The lattice fringe at 0.310 nm was attributed to the (110) planes of rutile form.32 This observation confirmed that titania matrix contained both the phases (anatase and rutile) and these results were consistent with the XRD results. In addition to this, it was evident that g-C3N4 had a stacked layer structure. This proved the successful fabrication of g-C3N4/N-TiO2.
Fig. 4 shows the XPS spectra of g-C3N4/N-TiO2 composites and high-resolution XPS of C, N, O, and Ti elements in CT-3 samples. The spectra exhibited distinctive peaks corresponding to C, N, O, and Ti elements, which confirmed the formation of g-C3N4/N-TiO2 composites (Fig. 4A). The high-resolution XPS of C 1s provided in Fig. 4B was divided into two peaks, which could be ascribed to the adventitious carbon that appeared at 284.6 eV and the carbon atom in N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N group, appearing at 288.1 eV.33,34 Fig. 4C presents the N 1s spectra of g-C3N4, N-TiO2, and CT-3 samples. For pure g-C3N4, the characteristic N 1s peak of g-C3N4 was observed at 398.4 eV, which can be assigned to sp2-hybridizednitrogen (C
N group, appearing at 288.1 eV.33,34 Fig. 4C presents the N 1s spectra of g-C3N4, N-TiO2, and CT-3 samples. For pure g-C3N4, the characteristic N 1s peak of g-C3N4 was observed at 398.4 eV, which can be assigned to sp2-hybridizednitrogen (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–C) involved in the thiazine rings of g-C3N4.35 For N-TiO2, the N 1s peak centered at 399.7 eV was attribute to the N atoms located at the interstitial sites of TiO2 lattice and the O–Ti–N sites incorporated into the TiO2 lattice.36 Gaussian fitting of the N 1s spectra of CT-3 revealed an asymmetrical feature with two main peaks located at the binding energy of 398.4 and 399.7 eV, respectively, suggesting the successful fabrication of g-C3N4/N-TiO2 composites. The O 1s spectra in Fig. 5D showed two peaks for the binding energy at 529.8 and 531.2 eV, which were associated with the O2− in TiO2 and the –OH group on the surface of samples, respectively.37 Fig. 4E shows the Ti 2p high-resolution XPS peak. Two characteristic signals at the binding energies of 458.8 (Ti 2p3/2) and 464.4 eV (Ti 2p1/2) were observed for P25, which was consistent with the ref. 38. With regard to Ti 2p of N-TiO2 and CT-3 sample, the Ti 2p3/2 and Ti 2p1/2 core levels of the composites appear around 458.5 eV and 464.1 eV, respectively. Compared with that of P25, a little shift of 0.3 eV in N-TiO2 and CT-3 sample can be observed. The decrease in the binding energy of Ti 2p after nitrogen doping suggested that the electronic interactions of Ti with anions were considerably different from that of TiO2. Since nitrogen is less electronegative than oxygen, partial transfer of electron from the N to the Ti occurred. Hence, the electron density around the anion decreased, as a result of which the electron density around the cation increased. This further proved that nitrogen was incorporated into the lattice and substituted oxygen. Based on the XPS as well as XRD, FTIR and TEM analysis, it could demonstrate that the incorporation of nitrogen species into TiO2 and the formation of N-doped TiO2 during the calcining process, meanwhile, the g-C3N4 was successfully composited with N-TiO2.
N–C) involved in the thiazine rings of g-C3N4.35 For N-TiO2, the N 1s peak centered at 399.7 eV was attribute to the N atoms located at the interstitial sites of TiO2 lattice and the O–Ti–N sites incorporated into the TiO2 lattice.36 Gaussian fitting of the N 1s spectra of CT-3 revealed an asymmetrical feature with two main peaks located at the binding energy of 398.4 and 399.7 eV, respectively, suggesting the successful fabrication of g-C3N4/N-TiO2 composites. The O 1s spectra in Fig. 5D showed two peaks for the binding energy at 529.8 and 531.2 eV, which were associated with the O2− in TiO2 and the –OH group on the surface of samples, respectively.37 Fig. 4E shows the Ti 2p high-resolution XPS peak. Two characteristic signals at the binding energies of 458.8 (Ti 2p3/2) and 464.4 eV (Ti 2p1/2) were observed for P25, which was consistent with the ref. 38. With regard to Ti 2p of N-TiO2 and CT-3 sample, the Ti 2p3/2 and Ti 2p1/2 core levels of the composites appear around 458.5 eV and 464.1 eV, respectively. Compared with that of P25, a little shift of 0.3 eV in N-TiO2 and CT-3 sample can be observed. The decrease in the binding energy of Ti 2p after nitrogen doping suggested that the electronic interactions of Ti with anions were considerably different from that of TiO2. Since nitrogen is less electronegative than oxygen, partial transfer of electron from the N to the Ti occurred. Hence, the electron density around the anion decreased, as a result of which the electron density around the cation increased. This further proved that nitrogen was incorporated into the lattice and substituted oxygen. Based on the XPS as well as XRD, FTIR and TEM analysis, it could demonstrate that the incorporation of nitrogen species into TiO2 and the formation of N-doped TiO2 during the calcining process, meanwhile, the g-C3N4 was successfully composited with N-TiO2.
 | ||
| Fig. 4 XPS survey spectrum for the CT-3 composite (A) and the high resolution XPS spectra of C 1s (B), N 1s (C), O 1s (D), and Ti 2p (E). | ||
3.2 Evaluation of photocatalytic activity
In order to evaluate the photocatalytic performance of the as-prepared samples, the photoactivities of the samples towards aqueous phase degradation of methylene blue, a well-known organic-based dye pollutant in waste water produced from textile and other industrial processes, was studied. For comparison, the activities of g-C3N4 TiO2 (P25) and N-TiO2 were also determined under the same conditions. The results are shown in Fig. 5.Fig. 5A shows the UV-vis absorption spectra of MB solutions at all irradiated time intervals in the presence of CT-3 photocatalysts, when irradiated with visible light. Weakening in the intensity of absorption indicated that MB degraded on irradiation with visible light for prolonged time periods. In addition, the maximum absorption of wavelength showed a blue-shift, which suggested that intermediate products were formed during the degradation of MB.
Fig. 5B shows the concentrations of residual MB fractions in solution after absorption/desorption equilibrium in the dark for 120 min. As shown in Fig. 5B, the absorption percentages of MB in pure g-C3N4, N-TiO2, and CT-3 samples were 27%, 18%, and 23%, respectively. The results indicated the coupling of g-C3N4 into N-TiO2 was beneficial to enhance the adsorption ability of N-TiO2 to dye molecules.
Fig. 5C shows the comparison of photocatalytic degradation of MB over bare TiO2, g-C3N4, N-TiO2, and g-C3N4/N-TiO2 hybrids with different g-C3N4 contents under visible light irradiation. C0 and Ct were the concentrations of initial MB solution and MB solution remaining after each time interval of irradiation, respectively. As shown in Fig. 5C, the self-degradation of MB was nearly negligible without photocatalysts in the blank experiment. P25 exhibited poor visible photocatalytic activity in MB degradation. It is known that P25 cannot absorb visible light (λ > 420 nm), the observed activity can be attributed to the dye-sensitization effect of MB on TiO2. Both pure g-C3N4 and N-TiO2 has a higher photocatalytic activity than P25 under visible light irradiation. It can be seen that N doping into TiO2 greatly enhances the visible photocatalytic activity of TiO2. With respect to the g-C3N4/N-TiO2 hybrids, all of them showed higher photocatalytic activities than either N-TiO2 or g-C3N4. In addition, the photocatalytic activity increased gradually with the increasing amounts of g-C3N4 from 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 to 1
3 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. However, further increase in g-C3N4 content in the composites up to 4
1. However, further increase in g-C3N4 content in the composites up to 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, led to a decrease in the rate of degradation. Amongst them, CT-3 showed the best photocatalytic activity, which is consistent with the UV-vis DRS results. Notably, the addition of an appropriate proportion of g-C3N4 in the composites was necessary for photoactivity. The suitable concentration of g-C3N4 causes its good dispersion in the composites, which favors better interface contact between the g-C3N4 and N-TiO2 nanoparticles. As a result, high separation of the charge carriers and photoactivity can be achieved on the CT-3 sample.39,40
3, led to a decrease in the rate of degradation. Amongst them, CT-3 showed the best photocatalytic activity, which is consistent with the UV-vis DRS results. Notably, the addition of an appropriate proportion of g-C3N4 in the composites was necessary for photoactivity. The suitable concentration of g-C3N4 causes its good dispersion in the composites, which favors better interface contact between the g-C3N4 and N-TiO2 nanoparticles. As a result, high separation of the charge carriers and photoactivity can be achieved on the CT-3 sample.39,40
Fig. 5D represents the plots of −ln(Ct/C0) versus irradiation time based on above data. The degradation of MB, obviously, followed the pseudo-first-order reaction kinetics. The pseudo-first-order reaction rate constant k was calculated from the kinetics equation: ln(C0/C) = kt. As shown in Fig. 5D, pure g-C3N4 and pristine N-TiO2 exhibited certain photoactivities with reaction rate constants of 0.011 and 0.015 min−1, respectively. Compared to N-TiO2 sample, the g-C3N4/N-TiO2 hybrids showed higher reaction rate constants. The composite CT-3 had the highest reaction rate constant (0.028 min−1), which was increased two-fold in the photodegradation of MB compared to N-TiO2. Fig. 5E showed the degradation ratio of MB in the presence of g-C3N4, N-TiO2, and CT-3 photocatalysts on irradiation with UV-vis light. As shown in Fig. 5E, an increase in the ratio of photodegradation in case of CT-3 composite (90%) was observed and compared with that of g-C3N4 (46%) and N-TiO2 (79%) after 15 min UV-vis light irradiation, which further demonstrated the synergetic effect of N-doping and g-C3N4 coupling on the photoactivity of TiO2.
Stability of photocatalysts is also very important for its practical applications. Hence, in order to assess the stability, degradation of MB was tested by recycling it successively thrice and investigated after irradiation with visible light, as shown in Fig. 5F. The proportions of MB after photolysis (40 min) were found to be 73%, 70%, and 68%, for the CT-3 sample after the first, second, and third cycles, respectively. After three cycles, only a slight loss of activity was observed. However, a larger decrease was observed in the case of pure g-C3N4 and N-TiO2. Thus, g-C3N4/N-TiO2 hybrids can be used as stable visible-light photocatalysts.
3.3 Photoelectrochemical performance
Photoelectrochemical performance of N-TiO2 and the g-C3N4/N-TiO2 composite electrodes were studied, the results of which are shown in Fig. 6. As shown in Fig. 6A, both electrodes showed good reproducibility and stability of transient anodic photocurrent as the UV-visible light illumination was switched-on and switched-off. It was interesting to note that g-C3N4/N-TiO2 composite electrode showed a photocurrent density of 50 μA cm−2, which was about 3.8 times more than that of N-TiO2 electrode, with a photocurrent density of 13 μA cm−2. This remarkably increased photocurrent may be the result of faster electron transfer and a more efficient separation of charge carriers due to the integrating of N-TiO2 with g-C3N4. | ||
| Fig. 6 Transient photocurrent curves (A) and open-circuit potential curves (B) of N-TiO2 (a) and CT-2 nanocomposite electrodes (b) in 0.5 M Na2SO4 under UV-vis illumination. | ||
The transient open-circuit potential of the N-TiO2 electrode and the g-C3N4/N-TiO2 composite electrode on illumination using UV-visible light are shown in Fig. 6B. The measured potential reflected the kinetic behavior of charge transfer at the electrode. A photopotential value of 0.50 V was observed for g-C3N4/N-TiO2 composite electrodes, which was larger than that of pristine N-TiO2 electrode (0.41 V). Enhancement in open-circuit potential implied an increase in the number of carriers in g-C3N4/N-TiO2 composite electrode, which suggested faster rates of separation and transportation of electrons and holes.
3.4 Mechanism discussion
The absorption range of light plays an important role in the application of semiconductors in photocatalysis. The UV-vis diffused reflectance spectra were used to study the optical properties of the samples. It can be seen from Fig. 7A that N-TiO2 exhibits a longer light absorption edge compared with bare TiO2, indicating N doping was an efficient alternative for extending the light absorption of TiO2. Furthermore, there was an obvious red-shift of ca. 20–40 nm in the absorption edges of CT-1, CT-2, CT-3, and CT-4 samples, compared to blank N-TiO2. In addition to this, enhanced absorbance in the visible light region of 400–800 nm was also noticed for all g-C3N4/N-TiO2 composites compared to N-TiO2. This improvement could be attributed to the introduction of g-C3N4, which exhibited an optical absorption edge at 475 nm in the visible light region. Fig. 7B presents the plots obtained from UV-vis absorption spectra by transformations based on the Kubelka–Munk function versus the energy of light. The transformation was carried out according to the formula: (αhν)2 = A(hν − Eg), where α, ν, A, and Eg are the absorption coefficient, light frequency, proportionality constant, and band gap, respectively.41 The intercept of the tangent to the X axis gave a good approximation of the band gap energy of the samples. Notably, the band gap of g-C3N4 was evaluated and found to be 2.55 eV, and the band gaps in case of CT-1, CT-2, CT-3, and CT-4 composites were evaluated and found to be 2.90, 2.88, 2.84, and 2.87 eV, respectively, which were smaller than that of N-TiO2 (2.93 eV) and P25 (3.10 eV). Interestingly, the absorption edge at the longest wavelength and narrowest band gap energy was observed for CT-3 composite, which suggested that an optimum content of g-C3N4 in g-C3N4/N-TiO2 composite could contribute to the best photoresponsive property and the narrowest band gap amongst the as-prepared photocatalysts.To determine the further advantages of g-C3N4/N-TiO2 composites over N-TiO2 for improving the transfer of charge carriers, electrochemical impedance spectra (EIS) were recorded. Typical EIS Nyquist plots are presented in Fig. 8. A large arc (almost a line) suggested that the value of the electron-transfer resistance is very large. The introduction of g-C3N4 resulted in a decrease in the arc for g-C3N4/N-TiO2 composite electrode compared to N-TiO2 electrode, suggesting g-C3N4/N-TiO2 composite to be a more efficient charge carrier than N-TiO2.
 | ||
| Fig. 8 EIS Nyquist plots of N-TiO2 (a) and g-C3N4/N-TiO2 nanocomposite (CT-3 nanocomposite electrode) (b) in 0.5 M Na2SO4 under dark. | ||
On the basis of the above experimental results, a detailed schematic mechanism of charge transfer and separation for g-C3N4/N-TiO2 composites under visible light irradiation is proposed in Scheme 2. It is obvious from the schematic diagram that the conduction band (CB) and valence band (VB) of g-C3N4 are located above the CB and VB of N-TiO2, respectively. Under the irradiation of visible light, both N-TiO2 and g-C3N4 can be photoexcited to generate electron–hole pairs. Because of the matched energy band structure of g-C3N4 and N-TiO2, the photogenerated electrons of g-C3N4 can easily inject into the CB of N-TiO2 while the photogenerated holes of N-TiO2 can transfer to the VB of g-C3N4. Thus, efficient charge separation can be achieved, leaving more electrons in the CB of N-TiO2 for reduction process and more holes in the VB of N-TiO2 for oxidation reaction. In addition, the CB edge potential of N-TiO2 and the CB of g-C3N4 level are more negative than EO2/˙O2− (0.28 V vs. SHE),42 thus, O2 can be captured to generate ˙O2− via the reduction process. These ˙O2− radicals are the most important oxidizing species responsible for the degradation of MB. Meanwhile, holes can react with water moles or hydroxide ions (OH−) to produce hydroxyl radicals (˙OH), which has strong oxidation ability to decompose MB to CO2 and H2O.
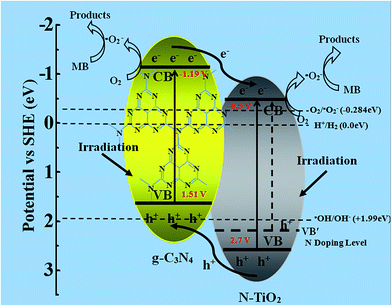 | ||
| Scheme 2 Schematic diagram of the separation and transfer of photogenerated charges in g-C3N4/N-TiO2 hybrids as well as the possible reaction mechanism. | ||
4. Conclusion
Composites, based on g-C3N4 and N-TiO2, were synthesized in situ by a facile heating method. The adopted synthetic route ensured not only the successful growth of lamellar g-C3N4, but also a high dispersion of N-TiO2 nanostructures on g-C3N4, without aggregation. The optimal mass ratio of melamine and TiN composite was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The heterostructure showed a better performance in MB photocatalytic degradation, compared to pure g-C3N4 or N-TiO2. The efficient separation of electrons and holes that originated from the formation of g-C3N4/N-TiO2 heterostructures was found to be beneficial to the photocatalytic activity. This work provides a facile and efficient way to design composite heterostructured photocatalysts, by an in situ growth strategy.
1. The heterostructure showed a better performance in MB photocatalytic degradation, compared to pure g-C3N4 or N-TiO2. The efficient separation of electrons and holes that originated from the formation of g-C3N4/N-TiO2 heterostructures was found to be beneficial to the photocatalytic activity. This work provides a facile and efficient way to design composite heterostructured photocatalysts, by an in situ growth strategy.
Acknowledgements
This work was financially supported by Innovation Program of Shanghai Municipal Education Commission (15ZZ092), Training Program for Young Teachers in Shanghai Colleges and Universities (ZZgcd14010), Startup Foundation of Shanghai University of Engineering Science (No Xiaoqi2014-22) and Graduate Innovation Program of Shanghai University of Engineering Science (15KY0516). The authors also appreciate the referee's very valuable comments, which have greatly improved the quality of the manuscript.References
- J. L. Zhang, Y. M. Wu, M. Y. Xing, S. A. K. Leghari and S. Sajjad, Energy Environ. Sci., 2010, 3, 715–726 CAS.
- E. Bizani, K. Fytianos, I. Poulios and V. Tsiridis, J. Hazard. Mater., 2006, 136, 85–94 CrossRef CAS PubMed.
- R. Daghrir, P. Drogui and D. Robert, Ind. Eng. Chem. Res., 2013, 52, 3581–3599 CAS.
- N. T. Nguyen, J. E. Yoo, M. Altomare and P. Schmuki, Chem. Commun., 2014, 50, 9653–9656 RSC.
- B. Cheng, Y. Le and J. G. Yu, J. Hazard. Mater., 2010, 177, 971–977 CrossRef CAS PubMed.
- X. Gao, Y. Jiang, Y. Zhong, Z. Y. Luo and K. F. Cen, J. Hazard. Mater., 2009, 174, 734–739 CrossRef PubMed.
- M. X. Sun, X. Y. Zhang, J. Li, X. L. Cui, D. L. Sun and Y. H. Lin, Electrochem. Commun., 2012, 16, 26–29 CrossRef CAS.
- A. Di Paola, E. Garcia-Lopez, G. Marci and L. Palmisano, J. Hazard. Mater., 2011, 211, 3–29 Search PubMed.
- W. J. Tseng, S. M. Kao and J. H. Hsieh, Ceram. Int., 2015, 9, 10494–10500 CrossRef.
- F. Q. Gao, Y. Yang and T. H. Wang, Chem.–Eng. J., 2015, 270, 418–427 CrossRef CAS.
- D. Y. Wu, M. C. Long, W. M. Cai, C. Chen and Y. H. Wu, J. Alloys Compd., 2010, 502, 289–294 CrossRef CAS.
- R. Asahi, T. Morikawa, T. Ohwaki, K. Aoki and Y. Taga, Science, 2001, 293, 269–271 CrossRef CAS PubMed.
- H. Irie, Y. Watanabe and K. Hashimoto, J. Phys. Chem. B, 2003, 107, 5483–5486 CrossRef CAS.
- C. Burda, Y. Lou, X. Chen, A. C. S. Samia, J. Stout and J. M. Gole, Nano Lett., 2003, 3, 1049–1051 CrossRef CAS.
- S. Sakthivel, M. Janczarek and H. Kisch, J. Phys. Chem. B, 2004, 108, 19384–19387 CrossRef CAS.
- T. Lindgren, J. M. Mwabora, E. Avendano, J. Jonsson, A. Hoel, C. G. Granqvist and S. E. Lindquist, J. Phys. Chem. B, 2003, 107, 5709–5716 CrossRef CAS.
- M. Kitano, K. Funatsu, M. Matsuoka, M. Ueshima and M. Anpo, J. Phys. Chem. B, 2006, 110, 25266–25272 CrossRef CAS PubMed.
- S. Yin, H. Yamaki, M. Komatsu, Q. W. Zhang, J. S. Wang, Q. Tang, F. Saito and T. Sato, J. Mater. Chem., 2003, 13, 2996–3001 RSC.
- M. Maeda and T. Watanabe, J. Electrochem. Soc., 2006, 153, C186–C189 CrossRef CAS.
- X. C. Wang, K. Maeda, A. Thomas, K. Takanabe, G. Xin, J. M. Carlsson, K. Domen and M. Antonietti, Nat. Mater., 2009, 8, 76–82 CrossRef CAS PubMed.
- P. Wu, J. R. Wang, J. Zhao, L. J. Guo and F. E. Osterloh, Chem. Commun., 2014, 50, 15521–15524 RSC.
- H. Zhao, Y. M. Dong, P. P. Jiang, H. Y. Miao, G. L. Wang and J. Q. Zhang, J. Mater. Chem. A, 2015, 3, 7375–7381 CAS.
- M. Wu, J. M. Yan, X. W. Zhang, M. Zhao and Q. Qiang, J. Mater. Chem. A, 2015, 3, 15710–15714 CAS.
- F. Chang, J. Zhang, Y. C. Xie, J. Chen, C. L. Li, J. Wang, J. R. Luo, B. Q. Deng and X. F. Hu, Appl. Surf. Sci., 2014, 311, 574–581 CrossRef CAS.
- S. Zhou, Y. Liu, J. M. Li, Y. J. Wang, G. Y. Jing, Z. Zhao, D. X. Wang, A. J. Duan, J. Liu and Y. C. Wei, Appl. Catal., B, 2014, 158–159, 20–29 CrossRef CAS.
- X. J. Wang, W. Y. Yang, F. T. Li, Y. B. Xue, R. H. Liu and Y. J. Hao, Ind. Eng. Chem. Res., 2013, 52, 17140–17150 CrossRef CAS.
- Y. M. He, Y. Wang, L. H. Zhang, B. T. Teng and M. H. Fan, Appl. Catal., B, 2015, 168, 1–8 Search PubMed.
- J. Fu, Y. L. Tian, B. B. Chang, F. N. Xi and X. P. Dong, J. Mater. Chem., 2012, 22, 21159–21166 RSC.
- J. G. Yu, S. H. Wang, J. X. Low and W. Xiao, Phys. Chem. Chem. Phys., 2013, 15, 16883–16890 RSC.
- G. S. Pozan and A. Kanbur, Appl. Catal., B, 2013, 129, 409–415 CrossRef CAS.
- Y. Y. Liang, H. L. Wang, H. S. Casalongue, Z. Chen and H. J. Dai, Nano Res., 2010, 3, 701–705 CrossRef CAS.
- A. Ramchiary and S. K. Samdarshi, Sol. Energy Mater. Sol. Cells, 2015, 134, 381–388 CrossRef CAS.
- S. P. Wang, C. J. Li, T. W. Wang, P. Zhang, A. Li and J. L. Gong, J. Mater. Chem. A, 2014, 2, 2885–2890 CAS.
- Y. M. He, L. H. Zhang, X. X. Wang, Y. Wu, H. J. Lin, L. H. Zhao, W. Z. Weng, H. L. Wan and M. H. Fan, RSC Adv., 2014, 4, 13610–13619 RSC.
- X. X. Wang, S. S. Wang, W. D. Hu, J. Cai, L. H. Zhang, L. Z. Dong, L. H. Zhao and Y. M. He, Mater. Lett., 2014, 115, 53–56 CrossRef CAS.
- M. X. Sun, P. Song, J. Li and X. L. Cui, Mater. Res. Bull., 2013, 48, 4271–4276 CrossRef CAS.
- G. M. An, W. H. Ma, Z. Y. Sun, Z. M. Liu, B. X. Han, S. D. Miao, Z. J. Miao and K. L. Ding, Carbon, 2007, 45, 1795–1801 CrossRef CAS.
- H. Q. Wang, J. P. Yan, W. F. Chang and Z. M. Zhang, Catal. Commun., 2009, 10, 989–994 CrossRef CAS.
- T. T. Li, L. H. Zhao, Y. M. He, J. Cai, M. F. Luo and J. J. Lin, Appl. Catal., B, 2013, 129, 255–263 CrossRef CAS.
- J. Cai, Y. M. He, X. X. Wang, L. H. Zhang, L. Z. Dong, H. J. Lin, L. H. Zhao, X. D. Yi, W. Z. Weng and H. L. Wan, RSC Adv., 2013, 3, 20862–20868 RSC.
- M. X. Sun, Y. L. Fang, Y. Wang, S. F. Sun, J. He and Z. Yan, J. Alloys Compd., 2015, 650, 520–527 CrossRef CAS.
- Y. M. He, J. Cai, T. T. Li, Y. Wu, Y. M. Yi, M. F. Luo and L. H. Zhao, Ind. Eng. Chem. Res., 2012, 51, 14729–14737 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2016 |




