Enhanced hydrogen production by methanol decomposition using a novel rotating gliding arc discharge plasma
Hao Zhanga,
Fengsen Zhua,
Xiaodong Li*a,
Kefa Cena,
Changming Dub and
Xin Tu*c
aState Key Laboratory of Clean Energy Utilization, Zhejiang University, Hangzhou 310027, China. E-mail: lixd@zju.edu.cn
bSchool of Environmental Science and Engineering, Sun Yat-Sen University, Guangdong 510275, China
cDepartment of Electrical Engineering and Electronics, University of Liverpool, Liverpool L69 3GJ, UK. E-mail: Xin.tu@liv.ac.uk
First published on 19th January 2016
Abstract
Hydrogen production from methanol decomposition was performed in a novel direct current (DC) rotating gliding arc (RGA) plasma reactor. The effects of various important parameters (feed flow rate, applied voltage, CH3OH concentration, operating current, preheating temperature, and water addition) on the reaction performance of the plasma methanol decomposition were investigated. The results showed that increasing the applied voltage and the operating current remarkably enhanced the CH3OH conversion in contrast to the effects of increasing feed flow rate, CH3OH concentration, and water addition. The selectivities of gas products (primarily H2 and CO) appeared to be positively correlated with the specific energy input. A comparison of the methanol decomposition processes using different non-thermal plasmas (e.g., dielectric barrier discharge and corona discharge) clearly showed that the RGA plasma provided a significantly higher CH3OH conversion (28.6–95.6%) and a relatively high energy yield of H2 (8.5–32.0 g kW−1 h−1) while maintaining a processing capacity that was several orders of magnitude higher than the other plasmas. A mathematical model was established to predict the CH3OH conversion and energy yield of H2. The model sensitivity analysis indicated that the CH3OH concentration was the most influential parameter, whereas the water addition was the least important parameter for the reaction performance.
1. Introduction
Clean electricity generation from H2-fueled fuel cells has been considered as one of the possible near to mid-term solutions for electronic devices and distributed power, as well as for various auxiliary power units in homes, businesses, and transportation.1–3 However, technical problems with the handling, storage, and transport of hydrogen severely limit the widespread use of portable fuel cells.4–6 Therefore, increasing attention has been focused on the development of new and efficient mobile and portable hydrogen production technologies that can overcome these problems by providing a source of clean hydrogen for fuel cells.7,8Methanol has been considered as an excellent H2-containing source for portable hydrogen production due to its high hydrogen to carbon ratio, simple transportation, low boiling point, biodegradability, and absence of sulfur content.6,9–11 Conventional hydrogen production strategies based on methanol rely on the use of catalysts including transition or noble metals.12–14 Although significant efforts have been devoted to find active, stable, and inexpensive catalysts to produce hydrogen from methanol, the problems of rapid loss of catalyst activity associated with high capital costs, the requirement of high temperatures, and large equipment size still limit its further applications, particularly for on-board hydrogen production systems.14
Non-thermal plasma technology provides an attractive alternative to the conventional catalytic route for methanol reforming at low temperatures. In non-thermal plasmas, the overall gas temperature can be as low as room temperature, which eliminates the quenching requirements and electrode erosion problems, and the electrons are highly energetic, with a typical electron temperature of 1–10 eV, which is sufficient to break down most of the chemical bonds in molecules and produce highly reactive species for the initiation and propagation of chemical reactions.15,16 In addition to the highly energetic electrons, the reactive radicals, excited species and ions formed in the plasma system can significantly enhance the chemical reaction rates, thus avoiding expensive catalysts and those vulnerable to impurities.17 A high reaction rate and fast attainment of the steady state in plasma processes allow a rapid start-up and shutdown of the process, providing high flexibility for integration into portable hydrogen production systems.15
Different non-thermal plasma systems have been investigated for methanol conversion into hydrogen to maximize hydrogen production and to reduce energy consumption, such as microhollow cathode discharge (MHCD),7 microwave discharge,2,12,18,19 and dielectric barrier discharge (DBD).20 However, these plasmas cannot simultaneously provide a high level of nonequilibrium, high electron temperature, and high electron density, making it difficult to achieve a highly efficient conversion of methanol at a high gas flow rate, which restricts the potential scale-up of this process.21 For example, in a DBD-assisted methanol decomposition process, Futamura et al. reported a methanol conversion of only 8–26% at a carrier gas (N2) flow rate of 100 ml min−1 and a CH3OH concentration input of 1%.22
In the past decade, gliding arc discharge (GAD) has attracted increasing interest because it is considered a powerful transitional plasma that can simultaneously provide a relatively high energy density, high electron temperature, good chemical selectivity, and low energy consumption,15,21,23–25 providing high flexibility to work in a wide range of reactant flow rates and plasma power levels (up to several kW).24 In a traditional flat GAD reactor consisting of two divergent knife-shaped electrodes, a high flow rate (e.g., 10–20 l min−1) is generally required to push the arc as it moves along the electrodes and to sustain a relatively long arc, generating a discharge zone for chemical reactions. Therefore, the fast gas speed and limited two-dimensional plasma reaction area confined by the flat electrodes lead to a low retention time of reactants, restricting the conversions of reactants and the energy efficiency of the processes.21,26
To overcome these problems, a direct current (DC) three-dimensional rotating gliding arc (RGA) reactor has been developed for fuel reforming in our lab.26,27 Compared with the traditional flat GAD reactor, the RGA reactor can generate a synergetic effect resulting from the combination of swirling flow and Lorentz force, which can make the arc rotate rapidly and steadily around the cone-shaped inner electrode without extinction (with a rotational speed of up to 100 rotations per second) over a wide flow rate range (e.g., 0.05–40 l min−1). Thus, a three-dimensional stable plasma area can be created in which the retention time of the reactants in the plasma is significantly increased, and the reactor efficiency is largely enhanced. Our previous study has shown that compared with other typical non-thermal plasmas (e.g., DBD, microwave, radio-frequency, and flat GAD), this RGA system showed significant advantages in the methane decomposition process in terms of methane conversion (maximum, 91.8%), hydrogen selectivity (maximum, 80.7%), and energy consumption for hydrogen production (minimum, 14.3 kJ l−1) while maintaining a reactant flow rate, or processing capacity, several orders of magnitude higher than the other plasmas.26
In this study, the RGA plasma reactor was developed for the decomposition of methanol into hydrogen in N2 carrier gas. The effects of different operating parameters (flow rate, applied voltage, CH3OH concentration, operating current, preheating temperature, and water addition) on the performance of the plasma chemical reaction were investigated. A mathematical model was established to predict the CH3OH conversion and H2 energy yield of the process based on the obtained experimental data. In addition, the model sensitivity analysis was performed to obtain new insights into the effect and relative importance of different parameters on the performance of the methanol decomposition process.
2. Experimental section
2.1. Experimental setup
As shown in Fig. 1, the experimental setup primarily consists of an RGA reactor, a mass flow controller (MFC), a syringe pump, temperature controllers, a DC power supply, a two-stage condenser system, and a gas chromatography (GC) system. A cone-shaped inner electrode (anode) is placed inside of a grounded circular cylinder (cathode). N2 was injected into the bottom of the reactor by three tangential inlets to form a swirling flow field in the reactor. An upward magnetic field formed by a magnet was placed around the entire reactor for stabilization and acceleration of the arc. With the combined effect of swirling flow and Lorentz force, the arc rotated rapidly around the inner electrode with a frequency of up to 100 rotations per second, forming a stable plasma “disc” area (see Fig. 1). A more detailed description of the RGA reactor is available in our previous work.26The plasma reactor was powered by a DC power supply (380 V/10 kV WWL-SS), and a 40 kΩ resistance was connected in series in the circuit to limit and stabilize the current. To investigate the effect of applied voltage, the power supply was set to the constant-voltage mode, and the output voltage was adjusted from 6 to 10 kV. When the effect of operating current was studied, the power supply was then switched to the constant-current mode, and the output current was gradually reduced until a critical limit of current was reached where an instability of the arc was encountered (the arc reignited at the narrowest gap repeatedly without rotating steadily on a plane).
High-purity nitrogen (≥99.99%) was used as the carrier gas, and the flow rate was controlled by an MFC (Sevenstar D07-series). A silica gel desiccant was placed after the MFC for moisture removal in the gas line. Anhydrous methanol (≥99.5%) or a mixture of methanol/water was controlled and injected into the gas line by a high-resolution syringe pump (Harvard, 11 plus). Thus, the total feed flow rate (0.3–0.7 mol min−1) of the mixed stream and the CH3OH concentration input (5–30 mol%) could be controlled. The mixed stream was then preheated to 353–393 K in a stainless steel pipe (4 mm in inner diameter and 40 cm in length) equipped with a temperature controller system to form a steady state vapor flow before flowing into the reactor. The quartz cover of the reactor was also heated to 373 K to prevent vapor condensation on the inner wall. A two-stage condenser was placed at the exit of the reactor to condense and collect the condensable vapors in the effluent stream, consisting of a first-stage condenser pipe equipped with an ice water circulation system and a second-stage liquid trap placed inside of an ice water container.
The gaseous products were measured by a GC (GC9790A, Fuli Analytical Instrument) equipped with a thermal conductivity detector (TCD) for detecting H2, O2, and N2 and a flame ionization detector (FID) for detecting CO, CO2, CH4, C2H6, C2H4, and C2H2. The preliminary analysis of the condensed liquid using a gas chromatography – mass spectrometry (JEOL, JMS-Q1050GC) showed that the condensed liquid was mainly methanol with trace amounts of ethanol, propanol and ethylene glycol. Thus, the volume of the condensed liquid can be roughly regarded as the volume of the unreacted methanol after the plasma reaction.2,28–31 Each experiment was repeated three times with similar results and the average results were given in this paper.
The experimental conditions are listed in Table 1.
| Parameters | Total flow rate (mol min−1) | CH3OH molar concentration (%) | Applied voltage (kV) | Operating current (mA) | Preheating temperature (K) | Water additiona (vol%) |
|---|---|---|---|---|---|---|
| a The volume concentration of water in the feed methanol/water liquid mixture. | ||||||
| Symbols | Q | C(CH3OH) | U | I | TP | A(H2O) |
| Values | 0.3–0.7 | 5–30 | 6, 8 and 10 | 50–210 | 353–393 | 0–5 |
2.2. Reaction performance assessment
For the methanol decomposition process, the conversion of CH3OH (X(CH3OH)) was defined as follows:
 | (1) |
The selectivities (S) of the products can be calculated as follows:
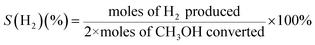 | (2) |
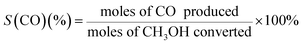 | (3) |
 | (4) |
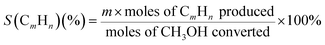 | (5) |
The energy yield of H2 (EY) was calculated by the following equation:
 | (6) |
The energy conversion efficiency of the process (ECE) was calculated based on the change of the lower heating values (LHV) of the fuels before and after the reaction, as follows:
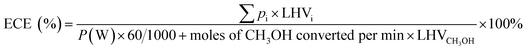 | (7) |
The specific energy input (SEI) was defined as follows to represent the energy density applied to the reaction area.
 | (8) |
3. Results and discussion
3.1. Effect of feed flow rate
The feed flow rate exerts a significant effect on the retention time of reactants within the plasma area, thus strongly affecting the performance of the plasma chemical reactions. The effect of flow rate on the CH3OH conversion, specific energy input (SEI), the selectivities of H2 (S(H2)) and CO (S(CO)), H2/CO ratio, and the selectivities of byproducts (S(CO2), S(CH4), S(C2H2), S(C2H4)) is presented in Fig. 2, whereas the CH3OH concentration input, applied voltage, and preheating temperature were kept constant. In agreement with other works,7,9,28,30 increasing the flow rate from 0.3 to 0.7 mol min−1 leads to a continuous drop of CH3OH conversion from 91.6 to 58.7%, as shown in Fig. 2(a). The decrease in the retention time of reactant in the plasma and SEI (Fig. 2(a)) with an increase in flow rate contributes to the decline in CH3OH conversion. In plasma, the production of reactant species, such as radicals, ions, and excited molecules, is vital for the initiation and propagation of chemical reactions. The SEI has a direct relationship on the formation of these species; thus, decreasing the SEI always shows a continuously negative effect on reactant conversions in plasma-assisted fuel conversion processes.16,32–35 | ||
| Fig. 2 Effect of total flow rate on (a) SEI, CH3OH conversion, (b) selectivities of H2 and CO, H2/CO ratio, and (c) selectivities of byproducts (C(CH3OH) = 15%, U = 10 kV, TP = 373 K, A(H2O) = 0%). | ||
Fig. 2(b) and (c) show that H2 and CO are the major gaseous products in the RGA-assisted methanol decomposition process, and CO2, CH4, C2H2, and C2H4 are also formed with an extremely low total selectivity of 1.4–5.4% under all of the tested conditions. No detectable amounts of C2H6, C3, or C4 gas products were generated in this study. However, C2H6 was commonly detected in the methanol decomposition processes using other types of non-thermal plasmas, such as MHCD7 and DBD.22 The relatively high electron density of the gliding arc discharge (1014 to 1015 cm−3),15,36 which significantly increases the collision probability between heavy particles and highly energetic electrons, could account for the limited formation of large molecules.37 It should also be noted that plasma-assisted partial oxidation or steam reforming of methanol often showed a significant formation of undesired gaseous products, such as HCHO (45–60% in selectivity)38 and CO2 (11.8–95% in selectivity).20,39 Therefore, the RGA-assisted methanol decomposition process could produce more clean gas products, primarily producing syngas, which is beneficial to the application to mobile or portable systems (e.g., fuel cells).
As seen from Fig. 2(b), the selectivities of H2 and CO show a nearly linear decrease as the flow rate increased, most likely from the decrease of SEI, which reduced the probability of heavy particles being broken into small molecules. The maximum selectivities towards H2 and CO are 56.1% and 69.1%, respectively, at a flow rate of 0.3 mol min−1. From the stoichiometry of the methanol decomposition reaction, a H2/CO molar ratio of 2.0 would be expected, whereas in this case, the H2/CO ratio slightly escalates from 1.63 to 1.78 as the flow rate increases from 0.3 to 0.7 mol min−1, which should arise from the formation of H-containing byproducts in this process. It is proposed that several N-containing products (e.g., NH3 and HCN) are formed in the methanol decomposition process in N2 due to the abundance of excited N atoms and/or excited N2 molecules in the plasma bulk, as evidenced from the occurrence of the NH and CN bands in the CH3OH/N2 emission spectra.40 In addition, the formation of trace amounts of liquid byproducts, such as ethanol, propanol, and ethylene glycol, which was also reported in a corona plasma-assisted methanol decomposition process,39 probably contribute to the decrease in the H2/CO ratio as well.
Fig. 2(c) shows that the selectivities towards CH4, C2H4, and CO2 tend to decrease with the increase of flow rate. In contrast, the C2H2 selectivity initially declines with an increase in flow rate and then levels off or slightly increases at a higher flow rate.
3.2. Effects of applied voltage and CH3OH concentration input
The applied voltage could significantly affect the characteristics of plasmas, as well as the energy input in reaction systems; thus it is an important parameter in plasma chemical reactions.22,28,41,42 Our previous works have shown that an increase in applied voltage would lead to a remarkable increase in electron temperature in the RGA plasmas.43 In addition, the performance of plasma-assisted fuel reforming processes has been found to strongly depend on the reactant composition.7,9,42In the RGA-assisted methanol decomposition process with N2 as the carrier gas, the initial dissociation of CH3OH molecules could be initiated by three factors, thermal activation, direct electron impact, and collisions with the nitrogen active species (e.g., nitrogen atoms N(2D), electronically excited N2(A3Σ+u), and vibrationally excited N2(X1Σ+g,ν)44–46), resulting in the formation of CH3, CH2, CH2OH, CH3O, CH2O, OH, H2O, H, and H2 via eqn (9)–(13).39,47–49 Our optical emission spectroscopy (OES) study showed that the addition of 5% CH3OH into N2 led to a nearly complete disappearance of N2 spectral lines, indicating efficient energy transfers from excited nitrogen species to reactant molecules.
| CH3OH → CH3 + OH | (9) |
| CH3OH → CH2 + H2O | (10) |
| CH3OH → CH2OH + H | (11) |
| CH3OH → CH3O + H | (12) |
| CH3OH → CH2O + H2 | (13) |
The produced CH3 and CH2 radicals will then result in the formation of CH4, C2H2, and C2H4 (in trace amounts) via the coupling, hydrogenation, and/or dehydrogenation reactions between the CmHn species,31 during which H2 may also be produced in small amounts.
CH2O is the key intermediate product of the methanol decomposition process. CH2O is unstable in non-thermal plasmas and can be readily further decomposed to form H2 and CO through the following reactions:28
| CH2O → CHO + H | (14) |
| CHO → CO + H | (15) |
| H + H → H2 | (16) |
Eqn (13) and (14)–(16) are clearly dominant in the plasma methanol decomposition process, considering that H2 and CO are the primary products and the H2/CO ratio is close to 2.0 under all of the tested conditions.
CO2 is possibly primarily produced from the reactions of CO with OH via eqn (17).
| CO + OH → H + CO2 | (17) |
The effects of applied voltage and CH3OH concentration input on the performance of the methanol decomposition process are shown in Fig. 3. As expected, the SEI and CH3OH conversion increase with an increase in applied voltage at the same CH3OH concentration (Fig. 3(a) and (b)). Previous studies have shown a facilitating effect of the N2 carrier gas on the decomposition of fuels in non-thermal plasmas,7,26,29,50 which should be attributed to the formation of a variety of nitrogen active species (as previously mentioned), that could substantially contribute to the decomposition of reactants.44–46 Therefore, for a fixed applied voltage, the increase in the CH3OH concentration (i.e., decrease in N2 concentration) results in a pronounced drop of CH3OH conversion (Fig. 3(b)). For example, at an applied voltage of 8 kV, the CH3OH conversion declines drastically from 95.6 to 35.2% as the CH3OH concentration increases from 5 to 30%.
 | ||
| Fig. 3 Effect of applied voltage and CH3OH concentration input on (a) SEI, (b) CH3OH conversion, (c) H2 selectivity, H2/CO ratio, and (d) CO selectivity (Q = 0.6 mol min−1, TP = 373 K, A(H2O) = 0%). | ||
As shown in Fig. 3(c) and (d), for most of the conditions (CH3OH concentration ≥ 15%), increasing the applied voltage enhances the selectivities of H2 and CO due to the increased SEI. As the CH3OH concentration increases (at the same applied voltage), the selectivities of H2 and CO follow a similar evolution to that of SEI (Fig. 3(a)). Initially, there is a remarkable increase as the CH3OH concentration increases to 15%, which then tends to level off with a further increase in the CH3OH concentration to 30%. It is worth noting that a plateau of H2 and CO selectivities with the variation of CH3OH concentration in non-thermal plasma-assisted methanol decomposition processes has also been reported.7,28 In contrast, Burlica et al. reported a “dropping” curve of H2 selectivity versus CH3OH concentration in a pulsed gliding arc plasma.9
Fig. 3(c) shows that the H2/CO ratio is weakly dependent on the CH3OH concentration, irrespective of applied voltage, and varies slightly in the range from 1.80 to 2.50.
3.3. Effect of operating current
The highly energetic electrons in plasmas are responsible for the first step of the decomposition process and the formation of excited species (as previously mentioned) via different types of collisions with the gas atoms and molecules.18 The operating current of plasma is linearly correlated with the electron number density, thus significantly influencing the performance of the methanol decomposition process.51The effect of current on the CH3OH conversion, SEI, and the selectivities towards H2 and CO is presented in Fig. 4. In agreement with other studies,52 a linear increase of SEI from 11.4 to 40.0 kJ mol−1 can be observed with an increase of current from 50 to 210 mA, which leads to a linear increase of CH3OH conversion from 38.7 to 72.4%. Similar to the CH3OH conversion and SEI, the selectivities of H2 and CO also drastically increase with the increase of current.
 | ||
| Fig. 4 Effect of operating current on (a) SEI, CH3OH conversion, (b) selectivities of H2 and CO, and H2/CO ratio. (Q = 0.5 mol min−1, C(CH3OH) = 15%, TP = 373 K, A(H2O) = 0%). | ||
Interestingly, a comparison of Fig. 2(a)–(c), 3(a), (c) and (d), and 4(a) and (b) shows that the product selectivities always follow a similar evolution to the SEI as each parameter varies, revealing that the SEI appears to directly affect the product selectivities.
We can see from Fig. 4(b) that an increasing discharge current leads to a slight drop in the H2/CO ratio, which might be attributed to the formation of different byproducts. The maximum H2/CO ratio reaches 2.2 at a current of 50 mA.
3.4. Effect of preheating temperature
The influence of preheating temperature on the performance of this process is shown in Fig. 5. An enhancement in CH3OH conversion would be expected with the increase of preheating temperature due to the increased energy input into the plasma system. However, in this study, the CH3OH conversion initially increases to a maximum of 76.3% at a preheating temperature of 363 K but then decreases to a plateau of between 73.4 and 73.7% with a further increase in the preheating temperature. A similar phenomenon also occurred in a flat GAD-assisted methanol decomposition process, and the authors attributed these results to the increased feed flow speed (resulting in a decreased retention time of reactants in plasma) because of the increase in preheating temperature.28 | ||
| Fig. 5 Effect of preheating temperature on (a) SEI, CH3OH conversion, (b) selectivities of H2 and CO, and H2/CO ratio. (Q = 0.5 mol min−1, C(CH3OH) = 15%, U = 10 kV, A(H2O) = 0%). | ||
The selectivities of H2 and CO initially decrease but then tend to level off with the increase in preheating temperature. In addition, an increase in preheating temperature from 353 to 393 K results in a slight enhancement of the H2/CO ratio from 1.96 to 2.02.
3.5. Effect of water addition
Steam reforming of methanol has also been extensively investigated in both traditional catalytic and plasma-assisted processes,20,30,53 and the effect of the steam reforming reaction on the plasma methanol decomposition process was studied in this work by introducing a small amount of water (1–5 vol% in the methanol/water liquid mixture) into the system. The results are presented in Fig. 6.We can see from Fig. 6(a) that the SEI initially slightly increases with the addition of 1% water into methanol and then plateaus to a constant value of 41.6 kJ mol−1 with a further increase in the amount of water addition. The increase of the water addition amount from 0 to 5% leads to a remarkable drop in the CH3OH conversion from 73.7 to 67.0%, revealing that the addition of water significantly inhibits the decomposition reaction of methanol. This is probably ascribed to the decrease in the gas temperature in the plasma bulk with an increasing amount of water addition due to the cooling effect of water molecules on the plasma system. It is considered that in plasma chemical reactions, the reactants can be dissociated via thermal activation and via plasma chemistry.49 The methanol decomposition reaction (eqn (18)) is strongly endothermic; thus, it is significantly thermally dependent. Consequently, the decrease in gas temperature with the increase in the water addition amount could substantially contribute to the decline of the CH3OH conversion.
| CH3OH → 2H2 + CO, ΔH298 K = 128.1 kJ mol−1 | (18) |
Due to the steam reforming reaction of methanol (eqn (19)), an enhancement in the selectivities of H2 and CO2 (Fig. 6(b) and (c)), as well as a slight increase in the H2/CO ratio (Fig. 6(b)) are observed with an increasing amount of water addition, agreeing well with other works.22,41 It should be noted that the methanol decomposition reaction (eqn (18)) is followed by the so-called water gas shift reaction (eqn (20)) when water is introduced into the process.
| CH3OH + H2O → 3H2 + CO2, ΔH298 K = 49 kJ mol−1 | (19) |
| CO + H2O → H2 + CO2, ΔH298 K = −41 kJ mol−1 | (20) |
Although CO undergoes the water gas shift reaction in the presence of water, its selectivity increases slightly with an increasing amount of water introduced (Fig. 6(b)). This probably arises from the enhanced CO formation through the Boudouard reaction (eqn (21)), considering the increasing production of CO2 in the system.
| C + CO2 → 2CO, ΔH298 K = 172 kJ mol−1 | (21) |
Fig. 6(c) shows that the selectivity towards CH4 is weakly dependent on the amount of water addition. However, the C2H2 selectivity drops nearly linearly and the C2H4 decreases slightly with an increasing amount of water addition. The possible dominant pathways for the formation of C2H2 and C2H4 are as follows:44
| CH2 → CH → C2H2 | (22) |
| CH2 → C2H4 | (23) |
The CH2 is primarily produced via eqn (10). The addition of water probably inhibits eqn (10), thus inhibiting eqn (22) and (23), leading to a decrease in the selectivities of C2H2 and C2H4.
3.6. Energy efficiency of the RGA plasma process
As shown in Fig. 7, the effects of different parameters on the efficiency of the plasma-assisted methanol decomposition reaction were examined in terms of the energy yield of H2 (EY) and energy conversion efficiency (ECE), as defined in eqn (6) and (7), respectively. We can see that an increase in flow rate from 0.3 to 0.7 mol min−1 leads to a remarkable increase in EY from 16.7 to 20.7 g kW−1 h−1, but a decrease in ECE from 44.2 to 39.5% (Fig. 7(a)). Fig. 7(b) indicates that a maximum applied voltage of 10 kV is beneficial for improving the EY and ECE, especially for a higher CH3OH concentration of ≥15%. The efficiency of the methanol decomposition process is shown to be strongly dependent on the CH3OH concentration input. At an applied voltage of 10 kV, a pronounced increase of EY from 8.5 to 32.0 g kW−1 h−1 and a drastic increase of ECE from 18.2 to 52.5% appear with an increase in CH3OH concentration from 5 to 30%. Interestingly, Burlica et al. also reported that the EY was approximately linearly dependent on the CH3OH concentration in a pulsed GAD system.9 Fig. 7(c) shows that the operating current plays an important role that is comparable to that of CH3OH concentration in this process. An increase in current from 50 to 210 mA leads to a remarkable decrease of EY from 25.0 to 21.3 g kW−1 h−1 but significantly enhances the ECE from 32.1 to 44.4%. The effects of preheating temperature and water addition on the methanol conversion efficiency are relatively weak. A slight enhancement of EY can be observed when increasing the preheating temperature from 353 to 373 K.To achieve a better performance, a compromise needs to be made between the CH3OH conversion and the EY, since the EY shows a negative correlation with the CH3OH conversion. According to the experimental results (Fig. 2–7), the “best results” appear to be obtained at a flow rate of 0.5–0.6 mol min−1, a CH3OH concentration input of 15–20%, an applied voltage of 10 kV (corresponding operating current of around 210 mA), a preheating temperature of 363 K, together with zero water addition, yielding a CH3OH conversion of 60–70%, a H2 selectivity of 50–52%, an EY of H2 of 22–26 g kW−1 h−1, and an ECE of 41–47%.
To compare our results with the current state-of-the-art from literature, Fig. 8 illustrates the various experimental data for EY vs. processing capacity (feed CH3OH flow rate) in plasma-assisted methanol decomposition process using different non-thermal plasmas. The experimental conditions from literature, and their corresponding detailed results are summarized in Table 2. We can see from Table 2 that the DBD reactor shows a low energy efficiency, as evidenced by the low values of both the energy yield of H2 (0.05–9.00 g kW−1 h−1) and the energy conversion efficiency (0.4–4.9%).22,54,55 The corona discharge produced a relatively high CH3OH conversion of 10–80% but with an energy yield of H2 of lower than 4.5 g kW−1 h−1.29 In the MHCD7 and traditional GAD28 plasmas, a moderate CH3OH conversion, a moderate energy yield of H2, as well as a moderate energy conversion efficiency can be achieved, whereas the processing capacity of the system was at a low level (feed methanol flow rate of only 0.02–1.66 mmol min−1).
 | ||
| Fig. 8 Performance comparisons of the RGA plasmas with other non-thermal plasmas for hydrogen production from methanol decomposition. | ||
| Method | Power | Carrier gas (ml min−1) | CH3OH concentration/molar flow rate | CH3OH conversion (%) | Selectivity (%) | H2/CO | EY (H2) (g kW−1 h−1) | ECE (%) | |
|---|---|---|---|---|---|---|---|---|---|
| S(H2) | S(CO) | ||||||||
| Corona29 | AC, 2 kHz, 0.8 kV, 12 W | Ar, 40 | 20–75%, 0.45–5.36 mmol min−1 | 10–80 | — | — | — | <4.5 | — |
| DBD22 | AC, 50 Hz, 0.21–1.99 W | N2, 100 | 1%, 0.04 mmol min−1 | 8–26 | 12–21.7 | 11.7–20.0 | 1.96–2.70 | ∼0.05–0.63 | 0.4–4.9 |
| DBD54 | 0.75–4.5 kHz | — | 0.5–1.0 mmol min−1 | ∼55–95 | — | — | — | 0.5–9 | — |
| DBD55 | 20 W | N2/Ar, 100 | 1%, 0.04 mmol min−1 | 76 | 77.6 | 53.2 | 2.9 | 0.3 | 1.9 |
| MHCD7 | DC, ∼0.8–1.3 W | N2, 10.5–23.7 | 4.7–56.7%, 0.02–0.60 mmol min−1 | 7.4–47.0 | 55–80 | 50–80 | 1.5–2.8 | 1.2–10.8 | 7.7–37.0 |
| GAD28 | AC, <300 W | Ar, 43.3–86.6 | 17.4–37.8%, 0.83–1.66 mmol min−1 | 51.8–81.7 | 43.4–49.5 | 81.4–81.9 | 1.14–1.20 | <6.9 | — |
| RGA (this work) | DC, 10 kV, 192.4–336.0 W | N2, 5700–13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
5–30%, 30.1–212.3 mmol min−1 | 28.6–95.6 | 24.4–56.4 | 19.4–69.1 | 1.63–2.51 | 8.5–32.0 | 18.2–52.5 |
| Thermodynamic limits9 CH3OH → 2H2 + CO | 290 | 100 | |||||||
As shown in Fig. 8, the EY of H2 appears to be positively correlated with the feed CH3OH flow rate, irrespective of the plasma source types. It is clear from Fig. 8 and Table 2 that compared with other plasma systems, the RGA plasma provides a significantly higher CH3OH conversion (28.6–95.6%), a highest EY of H2 (8.5–32.0 g kW−1 h−1), as well as a highest ECE (18.2–52.5%). It is also interesting to note that the RGA plasma can simultaneously provide a processing capacity of several orders of magnitude higher (30.1–212.3 mmol min−1 methanol) than that of other plasma systems (e.g., 0.02–0.60 mmol min−1 in MHCD plasma7) while allowing for a wider range of reactant concentration (5–30%). Therefore, the RGA plasmas appear to be optimal for future industrial applications compared with different non-thermal plasmas. The higher energy efficiency of the RGA plasma chemical process should be attributed to its unique characteristics with a high electron density (1014 to 1015 cm−3), moderate electron temperature (≈1 eV), which is associated with a relatively high vibrational temperature (4200–4400 K) and rotational temperature (1400–2200 K).26,36 The rotating plasma area should also substantially contribute to the improvement of energy efficiency by enhancing the retention time of reactant in the plasma.
To evaluate the commercial viability of the RGA plasma process, the ratio (η) of the molar flow rate of hydrogen produced in this process (QH2, produced) to that needed by a fuel cell (QH2, needed) to power the RGA plasma reformer at that rate of hydrogen production was calculated (eqn (24)).7
 | (24) |
Notice that the energy spent in the other units that needed in a practical methanol-based fuel processor for fuel cell system (e.g., the gas clean-up stage downstream the reactor) was not taken into consideration.
Obviously, the value η = 1.0 represents the energy breakeven point where the plasma reactor produces just enough hydrogen for fuel cell to sustain its own operation. We consider that the entire CO produced is then converted into H2 by the water gas shift reaction (eqn (20)), which is commonly used in on-board hydrogen production system for fuel cell.56–58 Therefore, the CO produced was taken into account. The calculation assumes a generic fuel cell with an efficiency of 50%.57
Based on this method, the calculated η varied in the range of between 0.5 and 1.1 under the studied conditions, indicating that the present process performance is even insufficient for a self-sustaining system. In order to be competitive with current technologies, the energy conversion efficiency of the plasma process should be enhanced to >85%, which is comparable with the traditional catalytic route.59
Further improvement in the energy efficiency of the RGA plasma chemical reaction can be expected from the optimization of the plasma power and reactor geometry. For example, pulsed DC power has been found to remarkably reduce the thermal loading and enhance the reactant conversion and energy efficiency.60 Burlica et al. have reported that an energy yield for H2 of up to 175 g kW−1 h−1 can be achieved when using a pulsed power (250 Hz) in a GAD-assisted methanol reforming process.9
Reactor geometry is another fruitful direction to achieve gains in energy efficiency. By insulating the reactor, the endothermic methanol decomposition reaction could potentially use the energy dissipated as heat, leading to an enhancement in efficiency.7 A supersonic nozzle configuration can also be used for reactant injection to allow for a lower feed flow rate with an increased retention time of reactant in plasma.61 In addition, a modelling study by Lindner et al. suggested that reducing the volume within the reactor or creating a microplasma device with an enclosed volume would lead to a significant increase in energy efficiency.7
The combination of plasma with catalysts (e.g., Cu/Al2O3 and Cu/ZuO) was also found to enhance the CH3OH conversion in DBD plasma-assisted methanol steam reforming31 or partial oxidation38 processes. However, the conditions of the reaction must be carefully controlled to reduce the formation of undesired byproducts, such as CO2 and HCHO.31,38
3.7. Mathematical model and sensitivity analysis
To obtain new insights into the relationships between the various operating parameters and the performance of the methanol decomposition process, a mathematical model was established using the method reported by Hsieh et al.62 In this model, the feed flow rate (Q, mmol min−1), CH3OH concentration input (C(CH3OH), %), discharge power (P, W), preheating temperature (Tp, K), and water addition (A(H2O), %) were selected to estimate the CH3OH conversion (X(CH3OH), %) and the energy yield of H2 (EY, g kW−1 h−1), which are the two most important indicators of the performance of the methanol decomposition process. It should be noted that the discharge power, but not the applied voltage or current, was selected to describe the energy input because the voltage and current are mutually influenced and cannot be considered as two independent variables.Based on the multiple regression methods presented by Hsieh et al.,62 we can obtain the functional relationships between the dependent variables (X(CH3OH) and EY) and the independent variables (Q, C(CH3OH), P, Tp, and A(H2O)) as follows:
 | (25) |
 | (26) |
It is important to note that these models are obtained based on the experimental data under the following conditions: Q = 300–700 mmol min−1, C(CH3OH) = 5–30%, P = 96–356 W, Tp = 353–393 K, and A(H2O) = 0–5%. There is relatively good agreement between the experimental results and the predicted data using the models with an error of within 10.24% (i.e., 9.27% and 10.24% for X(CH3OH) and EY, respectively).
The model sensitivity analysis was also performed with the aim of gaining better insight into the relative importance of the various operating parameters for the CH3OH conversion and the energy yield of H2 of the plasma-assisted methanol decomposition reaction. The results are shown in Fig. 9. For each sensitivity analysis, only one parameter was changed from +10% increase or −10% reduction, and all of the other parameters were fixed at the initial values (Q = 500 mmol min−1, C(CH3OH) = 20%, P = 230 W, Tp = 373 K, and A(H2O) = 3%). The definitions of Δλ/λ and ΔS/S in Fig. 9 are noted as follows:
Δλ/λ: the increase or reduction of each parameter divided by the initial value.
ΔS/S: the response of X(CH3OH) or EY divided by the corresponding value at initial conditions.
As shown in Fig. 9(a), for the effect of CH3OH concentration on the CH3OH conversion, a decrease of Δλ/λ from 0 to −70% leads to a sharp enhancement of ΔS/S from 0 to 117.9%, but an increase of Δλ/λ from 0 to 60% results in a moderate drop of ΔS/S from 0 to −26.2%. It is clear that the CH3OH concentration could strongly affect the CH3OH conversion, particularly at a lower reactant concentration (<20%), which can also be confirmed by Fig. 3(b). The flow rate also has a significantly negative effect on the CH3OH conversion. The ΔS/S decreases pronouncedly from 36.0 to −16.4% as Δλ/λ increases from −50 to 50%. The CH3OH conversion is positively correlated with the discharge power and preheating temperature. For the discharge power, an increase of Δλ/λ from −60 to 60% results in a nearly linear improvement of ΔS/S from −41.7 to 31.9%. The water addition is shown to weakly affect the CH3OH conversion. In general, Fig. 9(a) indicates that the relative significances of the various parameters for the CH3OH conversion decrease in the following order: C(CH3OH) > P > Q > Tp > A(H2O).
Fig. 9(b) clearly shows that the CH3OH concentration has a much stronger influence on the energy yield of H2 compared with the other parameters, which is also evidenced by Fig. 7. For the change in CH3OH concentration, the increase of Δλ/λ from −70 to 60% drastically increases the ΔS/S from −52.5 to 33.7%. The energy yield of H2 is weakly positively correlated with the flow rate and preheating temperature but weakly negatively correlated with discharge power. The effect of water addition on the energy yield of H2 is negligible. Briefly, the importance of sensitivity for the energy yield of H2 is C(CH3OH) > Tp > P > Q > A(H2O).
4. Conclusions
A DC rotating gliding arc discharge plasma was investigated for enhanced hydrogen production from methanol decomposition. The effects of feed flow rate, applied voltage, CH3OH concentration, operating current, preheating temperature, and water addition on the reaction performance were studied. The feed flow rate and CH3OH concentration were found to have a strong negative effect on the CH3OH conversion, whereas increasing the applied voltage and operating current remarkably enhanced the CH3OH conversion. The addition of water into the system was found to inhibit the decomposition of CH3OH but improved the selectivity towards H2.Thermal activation, direct electron impact, and collisions with nitrogen active species (e.g., nitrogen atoms N(2D), electronically excited N2(A3Σ+u), and vibrationally excited N2(X1Σ+g,ν)) can contribute to the dissociation of CH3OH molecules. CH2O was considered the key intermediate product of the process, leading to the formation of H2 and CO.
The RGA plasma showed significant advantages in CH3OH conversion and the energy yield of H2 compared with other non-thermal plasmas. It is also important to note that the RGA plasmas can provide a processing capacity of several orders of magnitude higher (30.1–212.3 mmol min−1 methanol) than that of other plasma systems (e.g., 0.02–0.60 mmol min−1 in MHCD plasma) while allowing for a wider range of reactant concentration (5–30%). Therefore, the RGA plasmas appeared to be optimal for future industrial applications.
The established mathematical model could accurately predict the CH3OH conversion and energy yield of H2 with an error of within 10.24%. The model sensitivity analysis showed that the relative significances of the various parameters for the CH3OH conversion decrease in the following order: CH3OH concentration > discharge power > feed flow rate > preheating temperature > water addition.
Acknowledgements
This work is supported by the National Natural Science Foundation of China (51576174), the Specialized Research Fund for the Doctoral Program of Higher Education of China (20120101110099), and the Fundamental Research Funds for the Central Universities (2015FZA4011).References
- L. Qin, G. Si, X. Li and S. Kang, RSC Adv., 2015, 5, 102593–102598 RSC
.
- T. Durka, G. D. Stefanidis, T. Van Gerven and A. I. Stankiewicz, Int. J. Hydrogen Energy, 2011, 36, 12843–12852 CrossRef CAS
.
- S. Nishioka and K. Maeda, RSC Adv., 2015, 5, 100123–100128 RSC
.
- L. Roses, G. Manzolini, S. Campanari, E. De Wit and M. Walter, Energy Fuels, 2013, 27, 4423–4431 CrossRef CAS
.
- S. H. Ma, D. H. Choi, S. M. Chun, S. S. Yang and Y. C. Hong, Energy Fuels, 2014, 28, 7721–7725 CrossRef CAS
.
- P. Hu, Y. Diskin-Posner, Y. Ben-David and D. Milstein, ACS Catal., 2014, 4, 2649–2652 CrossRef CAS
.
- P. J. Lindner and R. S. Besser, Int. J. Hydrogen Energy, 2012, 37, 13338–13349 CrossRef CAS
.
- E. Tatarova, N. Bundaleska, J. P. Sarrette and C. M. Ferreira, Plasma Sources Sci. Technol., 2014, 23, 63002 CrossRef CAS
.
- R. Burlica, K. Shih, B. Hnatiuc and B. R. Locke, Ind. Eng. Chem. Res., 2011, 50, 9466–9470 CrossRef CAS
.
- H. M. AbdelDayem, S. S. Al-Shihry and S. A. Hassan, Ind. Eng. Chem. Res., 2014, 53, 19884–19894 CrossRef CAS
.
- B. Zhang, D. Zhao, Y. Wu, H. Liu, T. Wang and J. Qiu, Ind. Eng. Chem. Res., 2015, 54, 623–632 CrossRef CAS
.
- Y. Wang, Y. You, C. Tsai and L. Wang, Int. J. Hydrogen Energy, 2010, 35, 9637–9640 CrossRef CAS
.
- A. G. Gribovskiy, L. L. Makarshin, D. V. Andreev, O. P. Klenov and V. N. Parmon, Chem. Eng. J., 2015, 273, 130–137 CrossRef CAS
.
- S. Sá, H. Silva, L. Brandão, J. M. Sousa and A. Mendes, Appl. Catal., B, 2010, 99, 43–57 CrossRef
.
- X. Tu and J. C. Whitehead, Int. J. Hydrogen Energy, 2014, 39, 9658–9669 CrossRef CAS
.
- S. Y. Liu, D. H. Mei, Z. Shen and X. Tu, J. Phys. Chem. C, 2014, 118, 10686–10693 CAS
.
- B. Hrycak, D. Czylkowski, R. Miotk, M. Dors, M. Jasinski and J. Mizeraczyk, Int. J. Hydrogen Energy, 2014, 39, 14184–14190 CrossRef CAS
.
- J. Henriques, N. Bundaleska, E. Tatarova, F. M. Dias and C. M. Ferreira, Int. J. Hydrogen Energy, 2011, 36, 345–354 CrossRef CAS
.
- N. Bundaleska, D. Tsyganov, R. Saavedra, E. Tatarova, F. M. Dias and C. M. Ferreira, Int. J. Hydrogen Energy, 2013, 38, 9145–9157 CrossRef CAS
.
- W. Ge, X. Duan, Y. Li and B. Wang, Plasma Chem. Plasma Process., 2015, 35, 187–199 CrossRef CAS
.
- C. S. Kalra, Y. I. Cho, A. Gutsol, A. Fridman and T. S. Rufael, Rev. Sci. Instrum., 2005, 76, 25110 CrossRef
.
- S. Futamura and H. Kabashima, IEEE Trans. Ind. Appl., 2004, 40, 1459–1466 CrossRef CAS
.
- C. Du, H. Li, L. Zhang, J. Wang, D. Huang, M. Xiao, J. Cai, Y. Chen, H. Yan and Y. Xiong, Int. J. Hydrogen Energy, 2012, 37, 8318–8329 CrossRef CAS
.
- A. Fridman, S. Nester, L. A. Kennedy, A. Saveliev and O. Mutaf-Yardimci, Prog. Energy Combust. Sci., 1998, 25, 211–231 CrossRef
.
- A. Indarto, D. R. Yang, C. H. Azhari, W. H. W. Mohtar, J. Choi, H. Lee and H. K. Song, Chem. Eng. J., 2007, 131, 337–341 CrossRef CAS
.
- H. Zhang, C. Du, A. Wu, Z. Bo, J. Yan and X. Li, Int. J. Hydrogen Energy, 2014, 39, 12620–12635 CrossRef CAS
.
- H. Zhang, X. D. Li, Y. Q. Zhang, T. Chen, J. H. Yan and C. M. Du, IEEE Trans. Plasma Sci., 2012, 40, 3493–3498 CrossRef
.
- Y. LÜ, W. Yan, S. Hu and B. Wang, J. Fuel Chem. Technol., 2012, 40, 698–706 CrossRef
.
- H. Li, J. Zou, Y. Zhang and C. Liu, Chem. Lett., 2004, 33, 744–745 CrossRef CAS
.
- D. H. Lee and T. Kim, Int. J. Hydrogen Energy, 2013, 38, 6039–6043 CrossRef CAS
.
- T. Kim, S. Jo, Y. Song and D. H. Lee, Appl. Energy, 2014, 113, 1692–1699 CrossRef CAS
.
- X. Tu and J. C. Whitehead, Appl. Catal., B, 2012, 125, 439–448 CrossRef CAS
.
- D. H. Lee, K. Kim, M. S. Cha and Y. Song, Proc. Combust. Inst., 2007, 31, 3343–3351 CrossRef
.
- D. H. Lee, K. Kim, M. S. Cha and Y. Song, Int. J. Hydrogen Energy, 2010, 35, 10967–10976 CrossRef CAS
.
- I. Rusu and J. Cormier, Chem. Eng. J., 2003, 91, 23–31 CrossRef CAS
.
- S. P. Gangoli, PhD Thesis, Drexel University, 2007
.
- M. Jimenez, R. Rincon, A. Marinas and M. D. Calzada, Int. J. Hydrogen Energy, 2013, 38, 8708–8719 CrossRef CAS
.
- S. Yao, X. Zhang and B. Lu, AIChE J., 2005, 51, 1558–1564 CrossRef CAS
.
- Y. Han, J. Wang, D. Cheng and C. Liu, Ind. Eng. Chem. Res., 2006, 45, 3460–3467 CrossRef CAS
.
- H. Zhang, X. Li, F. Zhu, Z. Bo, K. Cen and X. Tu, Int. J. Hydrogen Energy, 2015, 40, 15901–15912 CrossRef CAS
.
- S. Tanabe, H. Matsuguma, K. Okitsu and H. Matsumoto, Chem. Lett., 2000, 1116–1117 CrossRef CAS
.
- X. D. Li, H. Zhang, S. X. Yan, J. H. Yan and C. M. Du, IEEE Trans. Plasma Sci., 2013, 41, 126–132 CrossRef
.
- A. J. Wu, H. Zhang, X. D. Li, S. Y. Lu, C. M. Du and J. H. Yan, IEEE Trans. Plasma Sci., 2014, 42, 3560–3568 CrossRef CAS
.
- A. Oumghar, J. C. Legrand, A. M. Diamy, N. Turillon and R. I. Ben-Aim, Plasma Chem. Plasma Process., 1994, 14, 229–249 CrossRef CAS
.
- C. D. Pintassilgo and J. Loureiro, Adv. Space Res., 2010, 46, 657–671 CrossRef CAS
.
- C. D. Pintassilgo and J. Loureiro, Planet. Space Sci., 2009, 57, 1621–1630 CrossRef CAS
.
- A. Chang and S. H. Lin, Chem. Phys. Lett., 2002, 363, 175–181 CrossRef CAS
.
- L. N. Krasnoperov and J. V. Michael, J. Phys. Chem. C, 2004, 108, 8317–8323 CrossRef CAS
.
- S. Jo, D. H. Lee and Y. Song, Int. J. Hydrogen Energy, 2013, 38, 13643–13648 CrossRef CAS
.
- A. Indarto, J. Choi, H. Lee and H. K. Song, Energy, 2006, 31, 2986–2995 CrossRef CAS
.
- R. Valdivia-Barrientos, J. Pacheco-Sotelo, M. Pacheco-Pacheco, J. S. Benitez-Read and R. López-Callejas, Plasma Sources Sci. Technol., 2006, 15, 237 CrossRef
.
- X. Zhu, T. Hoang, L. L. Lobban and R. G. Mallinson, J. Phys. D: Appl. Phys., 2011, 44, 274002 CrossRef
.
- I. Uriz, G. Arzamendi, P. M. Diéguez, F. J. Echave, O. Sanz, M. Montes and L. M. Gandía, Chem. Eng. J., 2014, 238, 37–44 CrossRef CAS
.
- V. J. Rico, J. L. Hueso, J. Cotrino and A. R. González-Elipe, J. Phys. Chem. A, 2010, 114, 4009–4016 CrossRef CAS PubMed
.
- H. Kabashima, H. Einaga and S. Futamura, IEEE Trans. Ind. Appl., 2003, 39, 340–345 CrossRef CAS
.
- A. P. Simpson and A. E. Lutz, Int. J. Hydrogen Energy, 2007, 32, 4811–4820 CrossRef CAS
.
- J. R. Lattner and M. P. Harold, Appl. Catal., B, 2005, 56, 149–169 CrossRef CAS
.
- G. Petitpas, J. Rollier, A. Darmon, J. Gonzalez-Aguilar, R. Metkemeijer and L. Fulcheri, Int. J. Hydrogen Energy, 2007, 32, 2848–2867 CrossRef CAS
.
- J. Han, I. Kim and K. Choi, Int. J. Hydrogen Energy, 2002, 27, 1043–1047 CrossRef CAS
.
- K. H. Schoenbach, A. El-Habachi, M. M. Moselhy, W. Shi and R. H. Stark, Phys. Plasmas, 2000, 7, 2186–2191 CrossRef CAS
.
- C. Du, D. Huang, J. Mo, D. Ma, Q. Wang, Z. Mo and S. Ma, Int. J. Hydrogen Energy, 2014, 39, 9057–9069 CrossRef CAS
.
- L. Hsieh, W. Lee, C. Chen, M. Chang and H. Chang, Plasma Chem. Plasma Process., 1998, 18, 215–239 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2016 |




