Adsorption of phosphate on surface of magnetic reed: characteristics, kinetic, isotherm, desorption, competitive and mechanistic studies†
Tiantian Wangab,
Xing Xu*a,
Zhongfei Rena,
Baoyu Gaoa and
Hantao Wanga
aShandong Key Laboratory of Water Pollution Control and Resource Reuse, School of Environmental Science and Engineering, Shandong University, Jinan 250100, PR China. E-mail: xuxing@sdu.edu.cn; Fax: +86-531-88364513; Tel: +86-531-88361912
bSchool of resource and environment, Huazhong Agricultural University, Wuhan, PR China
First published on 6th January 2016
Abstract
A magnetic biocomposite was prepared by Fe3O4 in situ co-precipitation and amine functionalization processes by using virgin reed as starting material. The characteristics of amine cross-linked magnetic reed (ACMR) as well as phosphate-loaded/regenerated ACMR were evaluated by using FTIR, TEM, SEM, VSM and XPS. Adsorption properties of ACMR for phosphate were also determined. Results indicated that the average particle diameter of present Fe3O4 in ACMR was approximately 10.4 nm. Analysis of XPS indicated that uptake of phosphate by ACMR was based on N+ in quaternary nitrogen N 1s. The atomic ratio of Fe and N in regenerated ACMR was about 0.41% and 10.75%, which was a bit lower as compared with that in clean sample (0.46% and 11.02%); this indicated a small loss of Fe3O4 and amine groups on surface of ACMR during adsorption–desorption cycles. The calculated Qmax from Langmuir model were about 31.4–37.0 mg g−1 at 20–45 °C, which has shown comparable phosphate uptake capacity by contrast with most reported work. In addition, competitive adsorption revealed that the inhibition effect was more significant for anions with greater tendency to undergo ion exchange reaction (e.g. sulfate, nitrate and chloride) with quaternary ammonium groups on ACMR.
1. Introduction
The presence of phosphate in wastewaters provides an additional nutrient in the near static water bodies. As a result, an excessive growth of photosynthetic aquatic micro- and macro-organisms is encouraged in such water bodies which ultimately becomes a major cause for the eutrophication of such receiving waters.1–3 Therefore wastes containing phosphate must meet the discharge limits for phosphates as 0.5–1.0 mg L−1 P. In order to meet effluent quality standards, the removal of phosphate from wastewaters prior to discharge into natural waters is required.In wastewater-treatment technology, various techniques such as chemical precipitation, reverse osmosis, electrodialysis, contact filtration, adsorption and advanced biological methods have been successfully applied for phosphate removal.4,5 Adsorption is one of the techniques, which is comparatively more useful and economical for phosphate removal. Adsorption has been using as an alternative method for phosphate removal in wastewater treatment since 1960s. Due to intensified ‘‘green thinking’’ in process industry as well as environmental protection, the applications of low cost and easily available natural products in wastewater treatment have emerged as a viable option.6,7 These available materials included agricultural products and by-products, industrial by-products/wastes, hydrotalcites, soils and constituents, oxides and clay minerals, etc.2,3,5,7
Using the low-cost agricultural by-products as the effective adsorbents seems to be an acceptable access to uptake the phosphate from aqueous media. These agricultural by-products based bio-sorbents were prepared by impregnating with metal ions or amine groups which can catch phosphate by ligand/ion exchange mechanisms or activating with porous structures for adsorption of phosphate through pore adsorption.7–9 However, conventional cellulose-based adsorbents are difficult to be recovered from wastewater except by filter or high speed centrifugation. The preparation of magnetic bio-sorbents would be a good alternative to solve this problem.9–12 Compared to the reported bio-sorbents, a combination between bio-sorbents and iron oxide particles can pose an efficient biocomposite material, which could possibly show high adsorption capacity, intensified stability, and easy recovery from treated effluents by applying an external magnetic field. At present, there is only a few researches on the applications of these magnetic biomaterials, which mainly focus on the removal of dyes, Cr(VI) and heavy metal cations from liquids as well as enzyme immobilization.10–14 Therefore, to explore a magnetic bio-sorbent with a large phosphate adsorption capacity has an important realistic meaning.
In this study, virgin reed was used as the starting material to prepare the magnetic biosorbent. Fe3O4 was first introduced onto the surface of virgin reed by the in situ co-precipitation method.15,16 It was then modified with amine groups through grafting of epichlorohydrin, and ethanediamine followed by reaction with trimethylamine, forming the amine crosslinked magnetic reed (ACMR).1–3,17–19 The resulting biocomposite materials well as phosphate laden sample was characterized by Fourier transform infrared spectroscopy (FTIR), transmission electron microscopy (TEM), Scanning Electron Microscope (SEM), vibrating sample magnetometer (VSM), X-ray photoelectron spectroscopy (XPS) and X-ray diffraction (XRD). Subsequently, the biocomposite material was used for the removal of phosphate from liquid by evaluating its adsorption capacities as a function of pH, concentration, contact time, temperature and reusability.
2. Methods and materials
2.1. Materials and reagents
The virgin reed was obtained from Weishan Lake in Shandong Province, China. It was first dried and then sieved to 1–4 mm before use. All reagents used in tests including FeSO4, FeCl3, NaH2PO4, epichlorohydrin, N,N-dimethylformamide, ethanediamine and trimethylamine were of analytically pure and were bought from Sinopharm Group Co. Ltd.2.2. Preparation of ACMR
The preparation of amine crosslinked magnetic sample was conducted in a two-step reaction composed of (i) Fe3O4 in situ co-precipitation, and (ii) amine functionalization process (Fig. 1).(i) The Fe3O4 was first introduced onto the surface of virgin reed by the in situ co-precipitation method based on hydrogen bond. In the co-precipitation process, virgin reed (4.0 g) was suspended in a 240 mL mixed solution with FeSO4 of 0.125 mol L−1 and FeCl3 of 0.25 mol L−1. The reaction system was kept in an oxygen-free condition by purging with N2. After adding 25 mL of NH3·H2O (25%), the reaction temperature was raised to 70 °C and held for about 4 h. Then the Fe3O4 loaded reed was washed with deionized water and dried in a vacuum oven at 85 °C for 4 h.
(ii) Thereafter, 15 mL of epichlorohydrin and 10 mL of N,N-dimethylformamide were reacted with 6.0 g of magnetic reed and the mixture was stirred for 1 h at 85 °C. Ethanediamine (5 mL) was then added dropwise into the system and stirred for 30 min, followed by adding 10 mL of trimethylamine (stirred for 1 h at 85 °C). The primary products were washed with deionized water and dried for 12 h at 80 °C. The final samples (ACMR) were then collected and stored for use (ACMR with 19.6 g was obtained).
2.3. Characteristics of all magnetic samples
The magnetic properties of the biocomposite material (ACMR), phosphate laden ACMR and regenerated ACMR were measured as VSM by magnetometer (LDJ9500) at room temperature. Scanning electron microscopy (SEM) images and transmission electron microscopy (TEM) of ACMR were obtained with NoVa™ Nano SEM 250 and FEI Tecnai G20, respectively.The functional groups in ACMR and phosphate laden ACMR as well as phosphate solid were evaluated by using the FTIR technique (Perkin-Elmer “Spectrum BX” spectrometer) with spectrums canned from 400 to 4000 cm−1.
The textural structure including BET surface area, pore volume and pore size was determined by nitrogen adsorption/desorption isotherms at 196 C according to the Brunauer–Emmett–Teller (BET) principle (JW-BK122W, Beijing JWGB Sci. & Tech. Co., Ltd., China).
The surface binding state and elemental speciation of ACMR, phosphate laden ACMR and regenerated ACMR were analyzed by XPS. The measurements were performed by a spectrometer (ESCALAB 250) with MgKα irradiation (1486.71 eV of photons) as X-ray source. The phosphate laden ACMR was prepared by mixing 0.1 g of ACMR with 50 mL of phosphate solution (1000 mg L−1) for 12 h.
XRD patterns of powdered ACMR samples were carried out using a Rigaku D/MAX-YA diffractometer with Cu-Kα radiation at a voltage of 40 kV and a current of 40 mA, employing a scanning speed of 0.5 s per step in the range of diffracting angles 2θ from 10° to 70°.
2.4. Adsorption tests
In the pH effect experiment, the initial pH of phosphate solution (40 mg P per L) was first adjusted to pH range from 3.4 to 10.8 by 1 mol L−1 of HCl/NaOH solutions. Thereafter, 0.1 g ACMR was added to 25 mL of respective solutions in 100 mL Erlenmeyer flasks. Mixing was carried out at room temperature (20 ± 1 °C) on a rotary mixer and then the residual phosphate was determined at the wavelengths of UV-maximum (λmax) at 700 nm by ammonium molybdate spectrophotometric method through a UV-visible spectrophotometer (model UV754GD, Shanghai).Isotherm experiments was carried out by maintaining the water bath at 20, 35 and 45 °C. ACMR (0.1 g) were added to a set of 100 mL Erlenmeyer flasks containing 25 mL of phosphate solutions (pH: 6.0–7.0) with concentrations range of 25–500 mg P per L. After mixing for 6 h, the residual phosphate was then detected.
Adsorption kinetics experiments were carried out by mixing a series of ACMR (0.1 g) with 25 mL of phosphate solutions with concentrations of 10, 20 and 40 mg P per L (20 ± 1 °C). Each samples were extracted at required time intervals and then were filtered to analyze the residual phosphate concentrations in solutions.
The spent ACMR was mixed with NaCl, NaOH or HCl solution (0.1 mol L−1) for 6 h. After washed with distilled water for three times, the regenerated ACMR were used again in the subsequent experiments. The adsorption–desorption tests were conducted for 4 cycles and each regeneration efficiency was evaluated.
The competitive adsorption tests were evaluated by mixing the individual anions (e.g. nitrate, chloride, sulfate, and carbonate) with phosphate. The concentrations of individual anions were in the range of 0.5–5 mmol L−1. AMCR (0.1 g) were added to a set of 100 mL Erlenmeyer flasks containing 25 mL of solutions with co-existed anions. After stirring for 60 min, the residual nitrate was determined.
3. Results and discussions
3.1. Characteristics of all magnetic samples
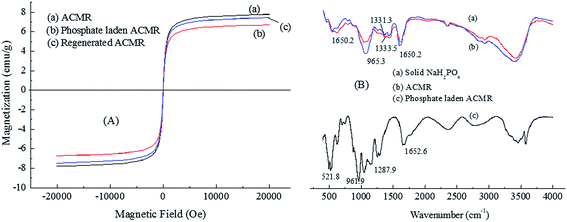
The magnetic property of the spent ACMR after the adsorption of phosphate was also determined and its saturated magnetization value was measured to be 6.77 emu g−1. Although the magnetic property of ACMR was weakened after the adsorption process, the spent ACMR still presented a high saturated magnetization, which could be separated easily from treated solution by an external magnetic field.
The magnetic property of the regenerated ACMR was also recovered after the 4 cycles of brine regeneration process. The saturated magnetization of the regenerated sample was measured to be 7.49 emu g−1; this was very close to the clean ACMR but higher than that of spent sample. This result indicated that effect of brine desorption process on magnetic property of ACMR was negligible. As a result, ACMR could be reused without significant loss in magnetization.
The powder XRD patterns of ACMR and Fe3O4 are shown in Fig. S1A.† For XRD pattern of Fe3O4 five characteristic peaks at 30.1°, 35.4°, 43.0°, 56.9° and 62.5° were corresponding to the (220), (311), (400), (511) and (440) crystal planes of a pure Fe3O4 with a spinal structure.15The chemical reaction of Fe3O4 formation can be written as:
| Fe2+ + 2Fe3+ + 8OH− → Fe3O4 + 4H2O |
Compared with the XRD pattern of Fe3O4, we can see that the Fe3O4 crystal has been successfully grown onto the virgin reed template. In addition, it was observed that diffraction peaks at 2θ = 15.5° and 22.6° assigned to crystalline cellulose can be found for ACMR.15
The specific peaks of solid Na2HPO4 were observed at 521.8, 961.9, 1287.9 and 1652.6 cm−1. It was obvious that these peaks were also overlapped with the bands of phosphate-loaded ACMR and the specific bands (1100–1250 cm−1 and 1650 cm−1) in phosphate-loaded ACMR were greatly enhanced. In addition, C–N bond (1331.3 cm−1) in ACMR was shifted to 1333.5 cm−1 after the adsorption of phosphate. These results indicated that phosphate was adsorbed onto the surface of ACMR by interactions between amine groups and phosphate.
 | ||
| Fig. 3 SEM images of ACMR at different magnification (a) ×200, (b) ×2000; TEM of ACMR (c) 200 nm, (d) 20 nm. | ||
The typical TEM images of ACMR was shown in Fig. 3c and d. It was evident that TEM image of ACMR was of irregular shape with Fe3O4 nanoparticles embedded in virgin reed. The average particle diameter of present Fe3O4 was approximately 10.4 nm; this corresponded well to the previous study.15,16,24,25 As a result, a large number of Fe3O4 had been introduced onto the skeleton of reed, which made the magnetic reed easily be separated from aqueous solution by a magnetic process.
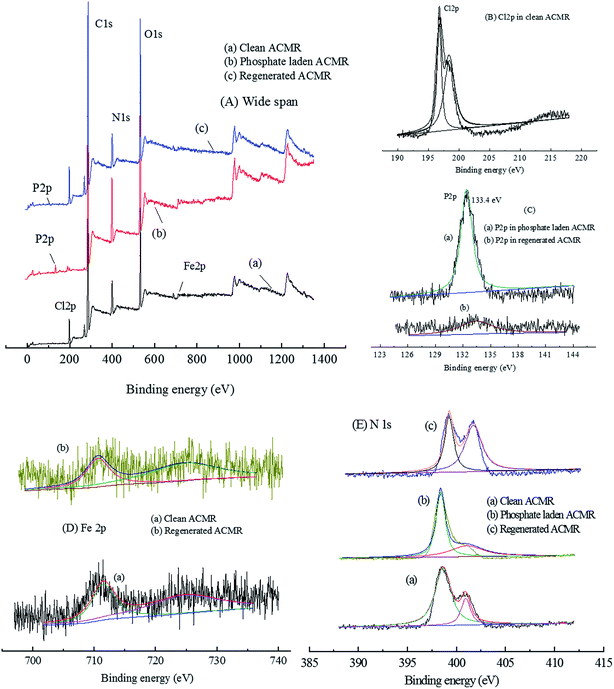 | ||
| Fig. 4 XPS of Clean ACMR and phosphate laden ACMR (A) wide scan (0–1400 eV); (B) Cl 2p (190–217 eV); (C) P 2p (125–145 eV); (D) Fe 2p (695–740 eV); (E) N 1s (387–412 eV). | ||
The Cl 2p (196.6 eV) in clean ACMR was derived from epichlorohydrin, which corresponded to the adsorption sites for phosphate. This binding energy almost disappeared after the adsorption of phosphate; it was replaced by the new binding energy at 133.4 eV, which was assigned to the adsorbed phosphate on surface of ACMR. After the brine regeneration, the atomic ratio of P significantly decreased from 1.61% to 0.27% with the increase of Cl atomic ratio from 0.45 to 4.04%. This indicated that brine regeneration process was effective to desorb the laden phosphate from ACMR.
The typical peaks for Fe in XPS analysis were at 711, and 729 eV, representing binding energies of Fe 2p. The peaks at 710.1 eV related to Fe(II) and the shoulder peak at 724.3 eV assigned to Fe(III) suggested the co-existence of Fe(III) and Fe(II) of Fe3O4 in all ACMR samples. After four cycles of adsorption–desorption processes, the atomic ratio of Fe in regenerated ACMR was about 0.41%, which was a bit lower as compared with that of clean sample (0.46%); this indicated a small loss of Fe3O4 on surface of ACMR during adsorption–desorption cycles.
The narrow scan of N 1s at 397–415 eV was shown in Fig. 4E. The one with lower binding energy (∼399.4 eV) could be interpreted as N in amide.26 The binding energy value at higher N state (401.4 eV) was assigned to the N+ in quaternary nitrogen N 1s.26 After phosphate adsorption, N 1s of N+ in quaternary nitrogen was weakened and shifted to 401.8 eV. This peak was recovered after the brine desorption process with binding energy at 401.3 eV. In contrast, the N 1s at ∼399.4 eV was almost constant. These results indicated that the adsorption of phosphate was mainly based on the N+ in quaternary nitrogen. The atomic ratio of N in regenerated ACMR was about 10.75%, which was a slight lower than that in clean ACMR (11.05%); this indicated an extent of destruction of amine groups during several cycles of adsorption–desorption processes.
3.2. Adsorption tests
 | ||
| Fig. 5 Adsorption of phosphate by ACMR as a function of pH (phosphate concentration: 40 mg P per L, contact time: 60 min, ACMR dosage: 4 g L−1, temperatures: 20 °C). | ||
Both Fe3O4 particles and virgin reed were used for uptake of phosphate, but their uptake capacities were lower than 4% at all pH conditions (Fig. 5). Results shown in Fig. 5 also indicated that uptake of phosphate by ACMR was increased from 78.7% to 97.1% with an increased pH from 3.1 to 7.5; thereafter the uptake was slightly decreased to 94.8% with the pH increased from 7.5 to 10.8. Based on the adsorption data, it is apparent that the phosphate adsorption capacities are reduced with pHs at strong acid and strong base conditions.
When the pH was lower than 4.0, the species of phosphate ions in the system mainly exist in the form of H3PO4 and H2PO4−. The lowering of pH caused phosphate to be protonated and the increased phosphoric acid will interfere with the adsorption of phosphate by ACMR. When the pH increased beyond 10.0, the OH− will increase significantly, and the excess OH− in the system would compete with phosphate ions for adsorption sites on surface of ACMR. In addition, the surface of ACMR will be negatively charged at higher pHs.27 Both these factors weakened the interactions between the negatively charged phosphate ions and positively charged ACMR, etc. resulting in a decreased uptake of phosphate at higher pH conditions.
All equilibrium results were fit with the Langmuir, Freundlich, and Temkin models followed as:28–31
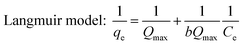 | (1) |
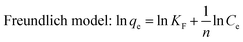 | (2) |
Temkin equation: qe = B![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(ACe) ln(ACe)
| (3) |
The fit of the three isotherms (Langmuir, Freundlich, and Temkin models) was shown in Fig. 6b–d and their parameters were summarized in Table 1. The experimental data were fit well by the Langmuir with correlation coefficient R2 between 0.994–0.998. Since the Langmuir equation assumes that the surface is homogenous, as a result, the adsorption of phosphate by ACMR was based on the homogenous distribution of adsorption sites on surface of the magnetic biocomposite. The calculated Qmax from Langmuir model were about 37.0, 33.0 and 31.4 mg g−1 at 20, 35 and 45 °C; this was very close to the experimental data. The phosphate capacities of different magnetic composites was evaluated by Yan et al.,32 and they found that uptake of phosphate by reported Fe3O4 composites were in range of 2.0–37.0 mg g−1. As a result, the magnetic biocomposite prepared in this work has shown comparable phosphate uptake capacity by contrast with most reported work.
| Temperature (°C) | Langmuir | Freundlich | Temkin | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Qmax (mg g−1) | b | R2 | KF (mg g−1) (mg L−1)−1 | n | R2 | A | B | R2 | |
| 20 | 37.0 | 0.0878 | 0.994 | 5.62 | 2.74 | 0.938 | 3.65 | 5.01 | 0.987 |
| 35 | 33.0 | 0.0925 | 0.998 | 4.93 | 2.69 | 0.906 | 2.72 | 4.82 | 0.990 |
| 45 | 31.4 | 0.0720 | 0.995 | 4.43 | 2.68 | 0.930 | 2.28 | 4.55 | 0.991 |
Temkin isotherm contains a factor that explicitly takes into account adsorbing species–adsorbate interactions.31,33 This model assumes the following: (i) the adsorption is characterized by a uniform distribution of binding energies, up to some maximum binding energy, and that (ii) the heat of adsorption of all molecules in the layer decreases linearly with coverage due to adsorbent–adsorbate interactions. The correlation coefficients R2 obtained from Temkin model (0.987–0.991) were relatively higher than those of Freundlich model. As a result, the adsorption of phosphate onto ACMR could also be characterized by a uniform distribution of binding energies and potential interactions between the phosphate and homogenous surface of ACMR.
Results shown in Fig. 7 indicated that the amount of adsorbed phosphate increased with time t and the adsorption reached an equilibrium state within 10–15 min. There was two adsorption stages before the equilibrium. It was obvious that about 70% of phosphate was removed within 1–5 min in stage 1; this represented a rapid adsorption rate partially due to the instantaneous monolayer adsorption on surface of ACMR. The rapid adsorption process was then followed by a gradually reduced adsorption rate prior to reaching equilibrium (stage 2).
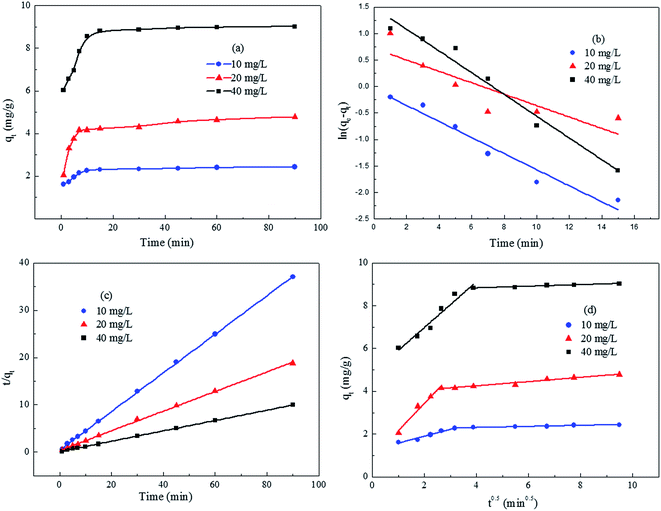 | ||
| Fig. 7 (a) Adsorption kinetic of phosphate onto ACMR and (b) its fit with pseudo first-order model; (c) pseudo second-order model; (d) intra-particle diffusion model. | ||
To analyze the adsorption rate of phosphate onto MWS, the pseudo first-order equation, pseudo second-order equation and intra-particle diffusion equation were evaluated based on the experimental data.14,34,35
(1) Pseudo first-order model. A kinetic model for adsorption analysis is the pseudo first-order rate expressed in the form:
ln(qe − qt) = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe − k1t qe − k1t
| (4) |
(2) Pseudo second-order model. The pseudo second-order kinetic rate equation is given as follows:
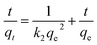 | (5) |
(3) Intra-particle diffusion model. The intra-particle diffusion model was proposed to identify the diffusion mechanism. If the plot of uptake, qt, versus square root of time, t1/2 passes through the origin, the intra-particle diffusion will be the sole rate-limiting process. The initial rate of intra-particle equation is as follows:
| qt = kpt0.5 | (6) |
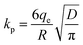 | (7) |
Kinetic parameters at different phosphate concentrations were evaluated from the Fig. 7b–d and all results were given in Table 2. The correlation coefficients (R2) obtained from the pseudo first-order kinetic model were only 0.699–0.947, which were significantly lower than those of the correlation coefficients (R2) from pseudo second-order kinetic model (>0.999). And also, the calculated qe2 values (2.45–9.12 mg g−1) obtained at different phosphate concentrations (10–40 mg g−1) agree well with experimental qe,exp values (2.42–9.05 mg g−1).
| C0 (mg L−1) | qe,expa (mg g−1) | Pseudo first-order | Pseudo second-order | Intra-particle diffusion | |||||
|---|---|---|---|---|---|---|---|---|---|
| k1 (min−1) | qe1b (mg g−1) | R2 | k2 (g (mg−1 min−1)) | qe2b (mg g−1) | R2 | kp (mg (g−1 min−1)) | R2 | ||
| a qe,exp is experimental values.b qe1, qe2 are calculated values. | |||||||||
| 10 | 2.42 | 0.155 | 0.95 | 0.943 | 0.391 | 2.45 | 1 | 0.327 | 0.951 |
| 20 | 4.75 | 0.108 | 2.04 | 0.699 | 0.118 | 4.81 | 0.999 | 1.27 | 0.962 |
| 40 | 9.05 | 0.201 | 4.44 | 0.947 | 0.119 | 9.12 | 1 | 1.07 | 0.941 |
Based on the results from Fig. 7d, it was obvious that the plots of qt versus t1/2 exhibited an initial linear portion followed by a plateau after 10–15 min. The initial curved portion of the plots corresponded to the boundary layer adsorption in the first adsorption stage and the linear portion to intra-particle diffusion, with the plateau corresponding to equilibrium. In addition, the plots did not pass through the origin. This indicated that intra-particle diffusion was not the only rate-limiting step; other kinetic processes were also simultaneously involved in the adsorption, which both contributed to the adsorption mechanisms for phosphate onto ACMR.
A decrease in adsorbed amount of phosphate may be, in part, due to loss of quaternary ammonium functional groups from the surface of ACMR during the brine desorption processes; this was validated by the XPS results of N 1s. Another possibility of reduced adsorption was that the laden phosphate ions were continuously trapped within the inner surface of the channels or pores in ACMR, resulting in the small decrease in adsorption sites that were available throughout the adsorption–desorption cycles.
The atomic ratio of Fe in all regenerated ACMR samples (regenerated by HCl solution) was evaluated based on the XPS analysis (Fig. 8c). A gradual decrease in regenerated ACMR samples was observed as the adsorption–desorption cycles was carried out. The atomic ratio of Fe was almost reduced by 20% after 4 cycles of adsorption–desorption. However, the saturated magnetization value of regenerated ACMR was decreased only lower than 5% (Fig. 2); this indicated that the loss of Fe during desorption process was mainly assigned to the Fe2O3 or other Fe formations, not to the Fe3O4 nano-particles in ACMR.
As discussed in this work, the adsorption of phosphate by ACMR was based on ions exchange on quaternary ammonium groups. As a result, the inhibition effect was more significant for anions with greater tendency to undergo ion exchange reaction (e.g. sulfate, nitrate and chloride) with quaternary ammonium groups on ACMR.
4. Conclusions
ACMR was prepared by Fe3O4 in situ co-precipitation and amine functionalization methods by using virgin reed as starting material. The characteristics of virgin, spent and regenerated samples were measured by FTIR, TEM, SEM, VSM and XPS. Results from FTIR and XPS analysis indicated that phosphate was adsorbed onto the surface of ACMR by interactions between amine groups and phosphate. The adsorption of phosphate onto ACMR could also be characterized by a uniform distribution of binding energies and potential interactions between the phosphate and homogenous surface of ACMR. Effect of competitive ions was the greatest for NO3−, followed by SO42− and Cl− while there was no effect of carbonate. In addition, no significant loss in adsorption capacity during four adsorption–desorption cycles (HCl and NaCl as eluents) demonstrated that the magnetic biocomposite was very suitable for the design of a continuous adsorption process.Acknowledgements
The research was supported by the National Natural Science Foundation of China (51178252, 51508307), China Postdoctoral Science Foundation funded project (2014M560556, 2015T80721). This work was also supported by grants from Tai Shan Scholar Foundation.References
- Z. Ren, X. Xu, B. Gao, Q. Yue and W. Song, Integration of adsorption and direct bio-reduction of perchlorate on surface of cotton stalk based resin, J. Colloid Interface Sci., 2015, 459, 127–135 CrossRef CAS PubMed.
- X. Xu, B. Gao, W. Wang, Q. Yue, Y. Wang and S. Ni, Adsorption of phosphate from aqueous solutions onto modified wheat residue: characteristics, kinetic and column studies, Colloids Surf., B, 2009, 70, 46–52 CrossRef CAS PubMed.
- X. Xu, B.-Y. Gao, X. Tan, Q.-Y. Yue, Q.-Q. Zhong and Q. Li, Characteristics of amine-crosslinked wheat straw and its adsorption mechanisms for phosphate and chromium(VI) removal from aqueous solution, Carbohydr. Polym., 2011, 84, 1054–1060 CrossRef CAS.
- J. Lalley, C. Han, X. Li, D. D. Dionysiou and M. N. Nadagouda, Phosphate adsorption using modified iron oxide-based sorbents in lake water: kinetics, equilibrium, and column tests, Chem. Eng. J., 2016, 284, 1386–1396 CrossRef CAS.
- Y. Yan, X. Sun, F. Ma, J. Li, J. Shen, W. Han, X. Liu and L. Wang, Removal of phosphate from wastewater using alkaline residue, J. Environ. Sci., 2014, 26, 970–980 CrossRef CAS.
- T. A. H. Nguyen, H. H. Ngo, W. S. Guo, T. Q. Pham, F. M. Li, T. V. Nguyen and X. T. Bui, Adsorption of phosphate from aqueous solutions and sewage using zirconium loaded okara (ZLO): fixed-bed column study, Sci. Total Environ., 2015, 523, 40–49 CrossRef CAS PubMed.
- T. A. H. Nguyen, H. H. Ngo, W. S. Guo, J. Zhang, S. Liang, D. J. Lee, P. D. Nguyen and X. T. Bui, Modification of agricultural waste/by-products for enhanced phosphate removal and recovery: potential and obstacles, Bioresour. Technol., 2014, 169, 750–762 CrossRef CAS PubMed.
- M. R. Awual, M. A. Shenashen, A. Jyo, H. Shiwaku and T. Yaita, Preparing of novel fibrous ligand exchange adsorbent for rapid column-mode trace phosphate removal from water, J. Ind. Eng. Chem., 2014, 20, 2840–2847 CrossRef CAS.
- W. Wang, J. Zhou, D. Wei, H. Wan, S. Zheng, Z. Xu and D. Zhu, ZrO2-functionalized magnetic mesoporous SiO2 as effective phosphate adsorbent, J. Colloid Interface Sci., 2013, 407, 442–449 CrossRef CAS PubMed.
- S. Lata and S. R. Samadder, Removal of arsenic from water using nano adsorbents and challenges: a review, J. Environ. Manage., 2016, 166, 387–406 CrossRef CAS PubMed.
- D. Mehta, S. Mazumdar and S. K. Singh, Magnetic adsorbents for the treatment of water/wastewater—a review, Journal of Water Process Engineering, 2015, 7, 244–265 CrossRef.
- V. K. Sharma, T. J. McDonald, H. Kim and V. K. Garg, Magnetic graphene–carbon nanotube iron nanocomposites as adsorbents and antibacterial agents for water purification, Adv. Colloid Interface Sci., 2015, 225, 229–240 CrossRef CAS PubMed.
- A. E. Chávez-Guajardo, J. C. Medina-Llamas, L. Maqueira, C. A. S. Andrade, K. G. B. Alves and C. P. de Melo, Efficient removal of Cr(VI) and Cu(II) ions from aqueous media by use of polypyrrole/maghemite and polyaniline/maghemite magnetic nanocomposites, Chem. Eng. J., 2015, 281, 826–836 CrossRef.
- X. Sun, L. Yang, Q. Li, J. Zhao, X. Li, X. Wang and H. Liu, Amino-functionalized magnetic cellulose nanocomposite as adsorbent for removal of Cr(VI): synthesis and adsorption studies, Chem. Eng. J., 2014, 241, 175–183 CrossRef CAS.
- W. Song, B. Gao, X. Xu, F. Wang, N. Xue, S. Sun, W. Song and R. Jia, Adsorption of nitrate from aqueous solution by magnetic amine-crosslinked biopolymer based corn stalk and its chemical regeneration property, J. Hazard. Mater., 2016, 304, 280–290 CrossRef CAS PubMed.
- W. Song, B. Gao, T. Zhang, X. Xu, X. Huang, H. Yu and Q. Yue, High-capacity adsorption of dissolved hexavalent chromium using amine-functionalized magnetic corn stalk composites, Bioresour. Technol., 2015, 190, 550–557 CrossRef CAS PubMed.
- X. Xu, B. Gao, X. Huang, J. Ling, W. Song and Q. Yue, Physicochemical characteristics of epichlorohydrin, pyridine and trimethylamine functionalized cotton stalk and its adsorption/desorption properties for perchlorate, J. Colloid Interface Sci., 2015, 440, 219–228 CrossRef CAS PubMed.
- X. Xu, B. Gao, W. Wang, Q. Yue, Y. Wang and S. Ni, Effect of modifying agents on the preparation and properties of the new adsorbents from wheat straw, Bioresour. Technol., 2010, 101, 1477–1481 CrossRef CAS PubMed.
- X. Xu, B. Gao, Q. Yue and Q. Zhong, Sorption of phosphate onto giant reed based adsorbent: FTIR, Raman spectrum analysis and dynamic sorption/desorption properties in filter bed, Bioresour. Technol., 2011, 102, 5278–5282 CrossRef CAS PubMed.
- H. Ma, J.-B. Li, W.-W. Liu, M. Miao, B.-J. Cheng and S.-W. Zhu, Novel synthesis of a versatile magnetic adsorbent derived from corncob for dye removal, Bioresour. Technol., 2015, 190, 13–20 CrossRef CAS PubMed.
- X.-F. Sun, B. Liu, Z. Jing and H. Wang, Preparation and adsorption property of xylan/poly(acrylic acid) magnetic nanocomposite hydrogel adsorbent, Carbohydr. Polym., 2015, 118, 16–23 CrossRef CAS PubMed.
- N. M. Mahmoodi, Synthesis of core–shell magnetic adsorbent nanoparticle and selectivity analysis for binary system dye removal, J. Ind. Eng. Chem., 2014, 20, 2050–2058 CrossRef CAS.
- F.-L. Mi, S.-J. Wu and Y.-C. Chen, Combination of carboxymethyl chitosan-coated magnetic nanoparticles and chitosan–citrate complex gel beads as a novel magnetic adsorbent, Carbohydr. Polym., 2015, 131, 255–263 CrossRef CAS PubMed.
- N. Farzin Nejad, E. Shams and M. K. Amini, Synthesis of magnetic ordered mesoporous carbon (Fe-OMC) adsorbent and its evaluation for fuel desulfurization, J. Magn. Magn. Mater., 2015, 390, 1–7 CrossRef CAS.
- C. Zhang, L. Chen, T.-J. Wang, C.-L. Su and Y. Jin, Synthesis and properties of a magnetic core–shell composite nano-adsorbent for fluoride removal from drinking water, Appl. Surf. Sci., 2014, 317, 552–559 CrossRef CAS.
- R. Pietrzak, XPS study and physico-chemical properties of nitrogen-enriched microporous activated carbon from high volatile bituminous coal, Fuel, 2009, 88, 1871–1877 CrossRef CAS.
- X. Xu, B.-Y. Gao, Q.-Y. Yue and Q.-Q. Zhong, Preparation and utilization of wheat straw bearing amine groups for the sorption of acid and reactive dyes from aqueous solutions, J. Hazard. Mater., 2010, 182, 1–9 CrossRef CAS PubMed.
- L. Cui, L. Hu, X. Guo, Y. Zhang, Y. Wang, Q. Wei and B. Du, Kinetic, isotherm and thermodynamic investigations of Cu2+ adsorption onto magnesium hydroxyapatite/ferroferric oxide nano-composites with easy magnetic separation assistance, J. Mol. Liq., 2014, 198, 157–163 CrossRef CAS.
- A. A. El-Bindary, M. A. Hussien, M. A. Diab and A. M. Eessa, Adsorption of Acid Yellow 99 by polyacrylonitrile/activated carbon composite: kinetics, thermodynamics and isotherm studies, J. Mol. Liq., 2014, 197, 236–242 CrossRef CAS.
- T. A. Khan, S. A. Chaudhry and I. Ali, Equilibrium uptake, isotherm and kinetic studies of Cd(II) adsorption onto iron oxide activated red mud from aqueous solution, J. Mol. Liq., 2015, 202, 165–175 CrossRef CAS.
- A. Mittal, D. Jhare and J. Mittal, Adsorption of hazardous dye eosin yellow from aqueous solution onto waste material de-oiled Soya: isotherm, kinetics and bulk removal, J. Mol. Liq., 2013, 179, 133–140 CrossRef CAS.
- L.-G. Yan, K. Yang, R.-R. Shan, T. Yan, J. Wei, S.-J. Yu, H.-Q. Yu and B. Du, Kinetic, isotherm and thermodynamic investigations of phosphate adsorption onto core–shell Fe3O4@LDHs composites with easy magnetic separation assistance, J. Colloid Interface Sci., 2015, 448, 508–516 CrossRef CAS PubMed.
- K.-Y. A. Lin, Y.-T. Liu and S.-Y. Chen, Adsorption of fluoride to UiO-66-NH2 in water: stability, kinetic, isotherm and thermodynamic studies, J. Colloid Interface Sci., 2016, 461, 79–87 CrossRef PubMed.
- J. Liu, R. Zhu, T. Xu, Y. Xu, F. Ge, Y. Xi, J. Zhu and H. He, Co-adsorption of phosphate and zinc(II) on the surface of ferrihydrite, Chemosphere, 2016, 144, 1148–1155 CrossRef CAS PubMed.
- L. Song, J. Huo, X. Wang, F. Yang, J. He and C. Li, Phosphate adsorption by a Cu(II)-loaded polyethersulfone-type metal affinity membrane with the presence of coexistent ions, Chem. Eng. J., 2016, 284, 182–193 CrossRef CAS.
- D.-W. Cho, C.-M. Chon, Y. Kim, B.-H. Jeon, F. W. Schwartz, E.-S. Lee and H. Song, Adsorption of nitrate and Cr(VI) by cationic polymer-modified granular activated carbon, Chem. Eng. J., 2011, 175, 298–305 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra25280f |
| This journal is © The Royal Society of Chemistry 2016 |