Basicity and stability of urea deep eutectic mixtures†
S. P. Simeonov*a and
Carlos A. M. Afonso*b
aInstitute of Organic Chemistry with Centre of Phytochemistry Bulgarian Academy of Sciences, Acad. G. Bonchev str., bl. 9, 1113, Sofia, Bulgaria. E-mail: svilen@orgchm.bas.bg
bThe Research Institute for Medicines (iMed.ULisboa), Faculty of Pharmacy, University of Lisbon, Av. Prof. Gama Pinto, 1649-003, Lisboa, Portugal. E-mail: carlosafonso@ff.ulisboa.pt
First published on 5th January 2016
Abstract
The stability and basicity origin of some common urea based deep eutectic mixtures/solvents (DES) were studied. We have observed an unexpected Hantzsch dihydropyridine reaction in sorbitol/urea DES, where the source of ammonia was found not to be originated from the individual ingredients of the DES. Our results showed that decomposition of urea occurs in DES at lower than expected temperatures, namely below 100 °C and is enhanced in diol DES by the formation of carbonates causing their unexplained basicity. Carbohydrates and choline chloride (ChCl) DES exhibit lower rates of decomposition, while no decomposition was observed from neat urea or MeOH and EtOH urea solutions.
Introduction
During the last decades considerable scientific interest on more green and sustainable approaches in organic synthesis has emerged.1,2 Ionic liquids (ILs) were intensively studied as promising green solvents and catalysts for diverse organic transformations.3–5 They are considered “green” mainly due to their low vapour pressure, high thermal stability and recyclability but despite that there are some serious doubts about their real greenness when their life cycle and toxic and environmental effects during the synthesis, application and disposal are considered.6,7 More recently, after the pioneering and systematic research of Abbott et al.,8–10 a new class of ionic fluids has emerged, namely deep eutectic mixtures/solvents (DES), which exhibit similar properties to the traditional ILs but are much more eco-friendly and cheap, which is of growing interest.11,12DES are typically formed by two or three components which interact each other mainly via hydrogen bond interactions to form an eutectic mixture, which has melting point lower than each of the individual ingredients. Most of them are liquid at temperatures below 70 °C and even at RT, and can be used as safe and inexpensive solvents for diverse applications. Usually DES are obtained by mixing quaternary ammonium salts with metal salts or hydrogen bond donors (HBD).13–15 Among all the HBD, urea is one of the most widely used because it is cheap, readily available and non-toxic. Urea based DES are already applied as solvents for various classical organic reactions13,16–20 and valorisation of biorenewable resources.21,22 Many of these transformations require reaction temperatures up to 100 °C and although some of them could proceed at lower temperatures the initial formation of the DES is usually performed by heating the ingredients till homogeneous mixture is formed. Herein are provided some insides on the stability of choline chloride (ChCl), polyalcohols and carbohydrates DES with urea under heating and the origin of their already observed basicity23 and catalytic activity in base catalysed reactions.16,17,19,20
Results and discussion
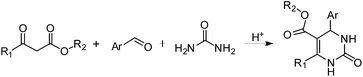
In our research we were interested on the possible application of chiral DES in asymmetric synthesis and in particular asymmetric Biginelli reactions. Such an alternative would be highly attractive because DES can be easily accessed from cheap and environmental friendly natural compounds like carbohydrates or derived polyols.The Biginelli reaction is an acid-catalysed, three-component reaction between an aldehyde, β-ketoester and urea providing dihydropyrimidones,24 compounds with interesting pharmacological properties associated with their heterocyclic scaffold25 (Scheme 1).
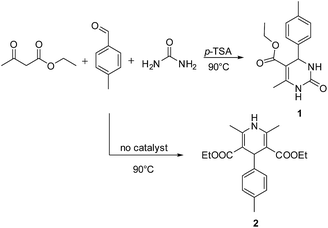
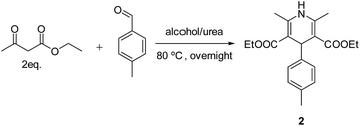
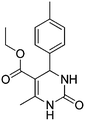
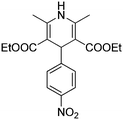
Initially we carried out classical Biginelli reaction between ethyl acetoacetate and p-tolyl aldehyde catalysed by p-toluene sulfonic acid (p-TSA) at 90 °C in sorbitol/urea (47 mol%/53 mol%) DES. The resulting dihydropyrimidone 1 was obtained in 95% yield after 12 h, with no enantiomeric excess. Surprisingly when the same reaction was repeated in absence of p-TSA instead of 1 we observed a new product in 41% yield, which was determined to be diethyl 2,4,6-trimethyl-1,4-dihydropyridine-3,5-dicarboxylate 2 (Scheme 2).
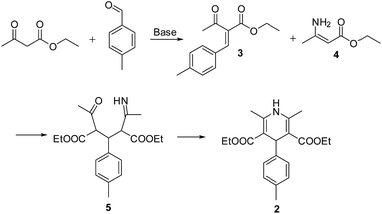
Obviously the formation of 2 was a result of Hantzsch dihydropyridine synthesis26 that proceed via initial Knoevenagel condensation between 1 equiv. ethyl acetoacetate and p-tolyl aldehyde to form intermediate 3, which further reacts with ester enamine 4, produced by the condensation of a second equivalent of ethyl acetoacetate with ammonia. The formed intermediate 5 undergo cyclization to give 2 (Scheme 3).
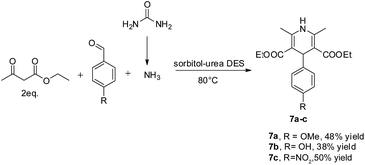
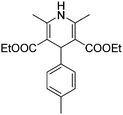
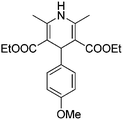
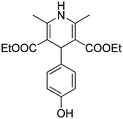
Different aryl aldehydes were screened under the same conditions and 1,4-dihydropyridine derivatives 7a–c were obtained in moderate yields of up to 50% (Scheme 4).
The unexpected formation of 1,4-dihydropyridine derivatives raises the question about the origin of ammonia in the reaction mixture. We used standard Nessler reagent test, which gives yellow colour in presence of ammonia (Fig. 1). The blank tests of pure urea and sorbitol water solutions were negative (Table 1), thereby proving that ammonia is not originated as an impurity in the individual ingredients of the DES. Positive result was observed only for aqueous solution of sorbitol/urea DES (Table 1, Fig. 1), which is somehow unexpected since the significant thermal urea decomposition requires higher temperatures, above 150 °C.27 However, this observation leads to the idea that partial decomposition of urea to ammonia occurs during the heating and formation of urea based DES and is responsible for their basicity.23 Ammonia could also be the catalyst in the reported base catalysed reactions, such as Perkin17 and Knoevenagel condensations.19
The synthesis of 2 was later used as a clock reaction for the decomposition of urea in different alcohols. The reactions were monitored by TLC (Table 2).
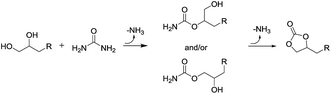
The formation of 2 was found to be enhanced in presence of poly alcohols (Table 2, entries 4 and 5), while no product was detected in presence of mono alcohols (Table 2, entries 1 and 2), thereby bringing to the idea that the formation of ammonia is not a result of a simple urea thermal decomposition. According to the results, the most rationalized explanation of the ammonia origin is the possible formation of cyclic carbonates and their related intermediates from the reaction of urea with poly alcohols (Scheme 5) that can proceed at moderate temperatures, even below 100 °C.
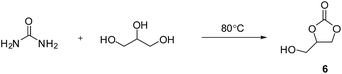
The formation of cyclic carbonates has been confirmed in a test reaction where a mixture of glycerol and urea (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 molar ratio) was heated at 80 °C with steering for 3 weeks to form glycerol carbonate 6 (Scheme 6).
2 molar ratio) was heated at 80 °C with steering for 3 weeks to form glycerol carbonate 6 (Scheme 6).
The reaction mixture was analysed by MS (Fig. 2) and 13C NMR (Fig. 3). The formation of 6 together with other minor side products, which were not assigned, was confirmed.
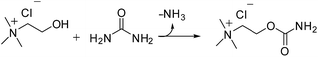
Our results could also explain the basicity of one of the most commonly used ChCl/urea (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), in which some urea decomposition occurred and traces of product 2 were observed by TLC (Table 2, entry 3). We speculate that the lower amount of formed ammonia is due to the fact that ChCl, being mono alcohol, cannot form cyclic carbonates via favourable intermolecular cyclization (Scheme 7).
2), in which some urea decomposition occurred and traces of product 2 were observed by TLC (Table 2, entry 3). We speculate that the lower amount of formed ammonia is due to the fact that ChCl, being mono alcohol, cannot form cyclic carbonates via favourable intermolecular cyclization (Scheme 7).
Having these results in hands, we explored the stability of some already reported DES in a systematic way. The liberated ammonia during the formation of DES was trapped in an aqueous acid solution and after titration with a base, in presence of an indicator, the amount of ammonia was determined and could be directly related to the urea decomposition rate. The temperature of 80 °C was chosen for our study because it can mimic commonly used by many researchers conditions for the formation of DES. A scale of 2.4 g of urea or dimethylurea (DMU) was used for the synthesis of DES and the other ingredients were recalculated as they are reported in the literature. In a standard experiment DES was heated at 80 °C for 7 h and continuously flashed with N2, which was then bubbled trough 10 mL 0.05 M water solution of H2SO4. At the end, the solution was titrated with 0.05 M solution of KOH using phenolphthalein as indicator, the results are summarized in Table 3.
| Entry | DES/molar ratio | 0.05 M KOH mLa | NH3/CH3NH2 mmol | NH3/CH3NH2 yield%c,d |
|---|---|---|---|---|
| a Theoretical volume of 0.05 M KOH aqueous solution needed for complete neutralization of H2SO4 solution (10 mL, 0.05 M) is 20 mL.b Urea/sorbitol DES was previously prepared in this molar ratio in our laboratory.c Calculated as a percent from the theoretical yield of ammonia considering full urea decomposition.d The ammonia/CH3NH2 which may have remained dissolved in the DES is not taken into account and the actual yields are expected to be higher. | ||||
| 1 (ref. 8) | ChCl/urea 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
18.5 | 0.075 | 0.09 |
| 2b | Sorbitol/urea 5.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 4 |
9.3 | 0.535 | 0.66 |
| 3 | Urea | 20 | 0.000 | 0 |
| 4 (ref. 18) | Glucose/urea/CaCl2 1.6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 : 0.5 4 : 0.5 |
19 | 0.005 | 0.006 |
| 5 | Glycerol/urea 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 4 |
4.9 | 0.755 | 0.94 |
| 6 (ref. 8) | ChCl/DMU 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
19.5 | 0.025 | 0.03 |
| 7 | Glycerol/DMU 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 4 |
19.5 | 0.025 | 0.03 |
| 8 | Sorbitol/DMU 5.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 4 |
19.3 | 0.035 | 0.04 |
| 9 (ref. 18) | Fructose/urea 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
18 | 0.100 | 0.12 |
The obtained results were in agreement with our previous observation that the urea decomposition is enhanced in presence of poly alcohols. As it was expected, pure urea was stable at 80 °C and no ammonia was detected (Table 3, entry 3). The highest amount of ammonia was observed in case of glycerol (Table 3, entry 5) and sorbitol (Table 3, entry 2), while only small amount was observed for ChCl (Table 2, entry 1). When carbohydrates such as glucose and fructose were used only minor amounts of ammonia were detected (Table 3, entries 4 and 9). However, after 7 hours at 80 °C the colour of these DES turns brown, probably due to carbohydrates decomposition and formation of humins, which also should be taken into account when they are used as a reaction media at high temperatures. Switching from urea to dimethylurea DMU resulted in more stable DES and only traces of methylamine were observed, even with glycerol and sorbitol (Table 3, entries 7 and 8).
Experimental
General
All the reagents were purchased from Sigma-Aldrich or Merck and were used without further purification. The reaction evolution was followed by TLC using silica Merck Kieselgel 60 F254 plates, and revealed by ultraviolet light at 254 nm and 325 nm. NMR spectra were recorded at room temperature in a Bruker AMX 300 or Bruker AMX 400 using CDCl3 or DMSO-d6 as solvents. The MS spectra of the glycerol urea mixture (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 molar ratio) was recorded using Thermo Scientific High Resolution Magnetic Sector MS DFS by chemical ionization.
2 molar ratio) was recorded using Thermo Scientific High Resolution Magnetic Sector MS DFS by chemical ionization.
Preparation of sorbitol–urea DES
13.96 g of urea and 49.8 g of sorbitol were mixed and stirred at 80 °C for 3 h till homogeneous liquid mixture was formed.5 g of sorbitol/urea DES were placed in a round bottom flask then p-tolyl aldehyde (1 mmol, 120 mg), ethyl acetoacetate (1 mmol, 130 mg) and p-TsOH (0.2 mmol, 34 mg) were added and stirred overnight at 90 °C. The reaction was dissolved in 10 mL of water. After complete dissolution, the product was extracted with ethyl acetate 2 × 25 mL. The organic phase was dried over Na2SO4, evaporated and the crude was purified with automatic flash chromatography machine-CombiFlash using gradient mixing of hexane and ethylacetate to give 260.4 mg (95%).
Reported mp 170–172 °C; found: 171–172 °C.
The spectral data (1H and 13C NMR) is identical with the reported.29
1H NMR (300 MHz, DMSO-d6) δ (ppm) 9.16 (s, 1H, NH), 7.68 (br s, 1H, NH), 7.12 (s, 4H), 5.10 (d, 1H, J = 3.3 Hz), 3.36 (q, 2H, J = 7.1 Hz), 2.49 (s, 3H), 2.24 (s, 3H), d 1.10 (t, 3H, J = 7.1 Hz).
13C NMR (100 MHz, DMSO-d6) δ (ppm) 166.3, 152.9, 148.9, 142.8, 137.3, 129.7, 126.8, 100.2, 59.9, 54.5, 21.4, 18.5, 14.8.
Preparation of Nessler reagent
A saturated solution of HgCl2 (∼2.2 g in 35 mL of distilled water) was added to a solution of 5 g of KI in 5 mL of distilled water until the excess is indicated by the formation of a precipitate. Then 20 mL of 5 N NaOH were added and the mixture diluted to 100 mL with distilled water. The solution was left to settle and the clear liquid was draw off.Nessler test
To a solution of 0.1 g sorbitol/urea DES, urea or sorbitol in 4 mL of distilled water were added several drops of the Nessler reagent. The appearance of yellow colour was followed as a sign for the presence of ammonia.General procedure for the synthesis of 1,4 dihydropyridine derivatives
5 g of sorbitol/urea DES were placed in a round bottom flask then 1 mmol of the corresponding aromatic aldehyde and 2 mmol of ethyl acetoacetate were added and stirred overnight at 90 °C. The reaction was dissolved in 10 mL of water. After complete dissolution, the product was extracted with ethyl acetate 2 × 25 mL. The organic phase was dried over Na2SO4, evaporated and the crude was purified with automatic flash chromatography machine (CombiFlash) using gradient mixing of hexane and ethyl acetate.Light yellow solid, yield: 140 mg, 41%. Reported mp: 136–137 °C;30 found: 140–142 °C.
1H NMR (300 MHz, CDCl3) δ (ppm) 7.17 (d, J = 7.6 Hz, 2H), 7.01 (d, J = 7.6 Hz, 2H), 5.74 (s, 1H), 4.95 (s, 1H), 4.08 (q, J = 7.0 Hz, 4H), 2.31 (s, 6H), 2.27 (s, 3H), 1.22 (t, J = 7.1 Hz, 6H).
13C NMR (75 MHz, CDCl3) δ (ppm) δ 167.70, 144.89, 143.81, 135.50, 128.56, 127.83, 104.23, 59.70, 39.10, 21.05, 19.56, 14.26.
ESI-MS: calculated for [C20H25NO4 + H]+/z: 344.24; found: (M + H)/z: 344.15.
IR: (KBr, cm−1): 3342.64 (NH), 2982.11 (Ar–H), 2936.77 (CH), 1693.65 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O).
O).
Light yellow solid, yield: 160 mg, 48%. Reported mp: 148–153 °C;31 found: 156–157 °C.
1H NMR (400 MHz, CDCl3) δ (ppm) 7.19 (d, J = 8.7 Hz, 2H), 6.74 (d, J = 8.7 Hz, 2H), 5.62 (s, 1H), 4.92 (s, 1H), 4.21–3.99 (m, 4H), 3.75 (s, 3H), 2.32 (s, 6H), 1.22 (t, J = 7.1 Hz, 6H).
13C NMR (101 MHz, CDCl3) δ (ppm) 167.83, 158.00, 143.66, 140.46, 129.10, 113.31, 104.54, 59.84, 55.27, 38.86, 19.74, 14.41.
ESI-MS: calculated for [C20H25NO5 + H]+/z: 360.42; found: (M + H)/z: 360.08.
IR: (KBr, cm−1): 3342.64 (NH), 2983.88 (Ar–H), 2956.87 (CH), 1689.64 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O).
O).
Yellow solid, yield: 131 mg, 38%. Reported mp: 230–231 °C;32 found: 219–220 °C.
1H NMR (400 MHz, DMSO) δ (ppm) 9.07 (s, 1H), 8.70 (s, 1H), 6.92 (d, J = 8.5 Hz, 2H), 6.57 (d, J = 8.5 Hz, 2H), 4.74 (s, 1H), 4.00–3.96 (m, 4H), 2.23 (s, 6H), 1.13 (t, J = 7.1 Hz, 6H).
13C NMR (101 MHz, DMSO) δ (ppm) 167.11, 155.44, 144.77, 138.92, 128.29, 114.52, 102.29, 58.89, 37.87, 18.21, 14.21.
ESI-MS: calculated for [C19H23NO5 + H]+/z: 346.40; found: (M + H)/z: 346.02.
IR: (KBr, cm−1): 3346.50 (N–H), 2985.81 (Ar–H), 2937.59 (CH), 1662.64 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1442.75 (C–OH).
O), 1442.75 (C–OH).
Yellow solid, yield: 189 mg, 50%. Reported mp: 118–127 °C;31 found: 129–130 °C.
1H NMR (400 MHz, CDCl3) δ (ppm) 8.07 (d, J = 8.8 Hz, 2H), 7.44 (d, J = 8.8 Hz, 2H), 5.84 (s, 1H), 5.08 (s, 1H), 4.11–4.05 (m, J = 7.1, 3.1 Hz), 2.34 (s, 6H), 1.21 (t, J = 7.1 Hz, 6H).
13C NMR (101 MHz, CDCl3) δ (ppm) 167.10, 155.14, 146.34, 144.70, 128.92, 123.30, 103.18, 60.02, 40.14, 19.65, 14.27.
ESI-MS: calculated for [C19H22N2O6 + H]+/z: 375.40, found: (M + H)/z: 375.06.
IR: (KBr, cm−1): 3319.49 (NH), 2926.01 (Ar–H), 2852.72 (CH), 1701.22 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O).
O).
Conclusions
The reported unusual basicity of urea based DES17,19,23 can be explained by the urea decomposition and liberation of ammonia, even at lower than the expected temperatures. The presence of ammonia was detected during unexpected formation of dihydropyridines via Hantzsch synthesis in sorbitol/urea DES and further confirmed with Nessler reagent tests. The formation of ammonia was not caused by simple thermal urea decomposition and was enhanced in presence of poly alcohols via the formation of cyclic carbonates. Neat or mono alcohols solutions of urea were observed to be completely stable under heating up to 80 °C for at least 7 h. ChCl was found to be an exception and although being mono alcohol, low rate of urea decomposition was also detected at 80 °C for 12 h. DMU based DES were more stable since DMU is less prompt to react with alcohols forming carbonates and insignificant amounts of methylamine were detected. Besides the extremely useful and high potential of DES clearly demonstrated for different applications after the pioneer discovery of Abbott et al.,8–10 this study provides some guidelines about their use for long time under high temperature in which for some specific DES combination, namely urea/polyalcohols occurs some ammonia liberation. These observations could be of importance for applications using urea based DES in different areas such as organic synthesis since, as we have shown, ammonia could be a catalyst or a reactive nucleophile in organic reactions performed in this reaction media.Acknowledgements
The authors acknowledge Fundação para a Ciência e a Tecnologia (FCT) (ref. SFRH/BD/67025/2009, PTDC/QUI–QUI/119823/2010 and UID/DTP/04138/2013) and European Research Area Network; ERANet LAC (ref. ELAC2014/BEE-0341) for financial support and REDE/1518/REM/2005 (FF-UL) for the mass service.Notes and references
- P. T. Anastas, Chem. Rev., 2007, 107, 2167 CrossRef.
- I. T. Horváth and P. T. Anastas, Chem. Rev., 2007, 107, 2169 CrossRef PubMed.
- J. P. Hallett and T. Welton, Chem. Rev., 2011, 111, 3508 CrossRef CAS PubMed.
- S. Park and R. J. Kazlauskas, Curr. Opin. Biotechnol., 2003, 14, 432 CrossRef CAS PubMed.
- F. van Rantwijk and R. A. Sheldon, Chem. Rev., 2007, 107, 2757 CrossRef CAS PubMed.
- R. Renner, Environ. Sci. Technol., 2001, 35, 410A CrossRef CAS PubMed.
- T. P. Thuy Pham, C.-W. Cho and Y.-S. Yun, Water Res., 2010, 44, 352 CrossRef PubMed.
- A. P. Abbott, G. Capper, D. L. Davies, R. K. Rasheed and V. Tambyrajah, Chem. Commun., 2003, 70 RSC.
- A. P. Abbott, D. Boothby, G. Capper, D. L. Davies and R. K. Rasheed, J. Am. Chem. Soc., 2004, 126, 9142 CrossRef CAS PubMed.
- E. L. Smith, A. P. Abbott and K. S. Ryder, Chem. Rev., 2014, 114, 11060 CrossRef CAS PubMed.
- Q. Zhang, K. de Oliveira Vigier, S. Royer and F. Jerome, Chem. Soc. Rev., 2012, 41, 7108 RSC.
- Y. Dai, J. van Spronsen, G.-J. Witkamp, R. Verpoorte and Y. H. Choi, J. Nat. Prod., 2013, 76, 2162 CrossRef CAS PubMed.
- G. Imperato, S. Hoger, D. Lenoir and B. Konig, Green Chem., 2006, 8, 1051 RSC.
- Z. Maugeri and P. Dominguez de Maria, RSC Adv., 2012, 2, 421 RSC.
- A. P. Abbott, J. C. Barron, K. S. Ryder and D. Wilson, Chem.–Eur. J., 2007, 13, 6495 CrossRef CAS PubMed.
- S. B. Phadtare and G. S. Shankarling, Green Chem., 2010, 12, 458 RSC.
- P. M. Pawar, K. J. Jarag and G. S. Shankarling, Green Chem., 2011, 13, 2130 RSC.
- G. Imperato, E. Eibler, J. Niedermaier and B. Konig, Chem. Commun., 2005, 1170 RSC.
- Y. A. Sonawane, S. B. Phadtare, B. N. Borse, A. R. Jagtap and G. S. Shankarling, Org. Lett., 2010, 12, 1456 CrossRef CAS PubMed.
- B. Singh, H. Lobo and G. Shankarling, Catal. Lett., 2011, 141, 178 CrossRef CAS.
- K. D. O. Vigier, A. Benguerba, J. Barrault and F. Jerome, Green Chem., 2012, 14, 285 RSC.
- F. Ilgen, D. Ott, D. Kralisch, C. Reil, A. Palmberger and B. Konig, Green Chem., 2009, 11, 1948 RSC.
- W. Li, Z. Zhang, B. Han, S. Hu, J. Song, Y. Xie and X. Zhou, Green Chem., 2008, 10, 1142 RSC.
- Suresh and J. S. Sandhu, Arkivoc, 2012, 66 CAS.
- C. O. Kappe, Eur. J. Med. Chem., 2000, 35, 1043 CrossRef CAS PubMed.
- A. K. Saini, S. Sandhu and S. Jagir, J. Sci. Ind. Res., 2008, 67, 95 Search PubMed.
- P. M. Schaber, J. Colson, S. Higgins, D. Thielen, B. Anspach and J. Brauer, Thermochim. Acta, 2004, 424, 131 CrossRef.
- X. Jing, Z. Li, X. Pan, Y. Shi and C. Yan, J. Iran. Chem. Soc., 2009, 6, 514 CrossRef CAS.
- N. A. Liberto, S. de Paiva Silva, Â. de Fátima and S. A. Fernandes, Tetrahedron, 2013, 69, 8245 CrossRef.
- D. Bandyopadhyay, S. Maldonado and B. K. Banik, Molecules, 2012, 17, 2643 CrossRef PubMed.
- R. F. Affeldt, E. V. Benvenutti and D. Russowsky, New J. Chem., 2012, 36, 1502 RSC.
- N. Koukabi, E. Kolvari, A. Khazaei, M. A. Zolfigol, B. Shirmardi-Shaghasemi and H. R. Khavasi, Chem. Commun., 2011, 47, 9230 RSC.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra24558c |
| This journal is © The Royal Society of Chemistry 2016 |