Effect of a static magnetic field on the preparation of MnOOH and Mn3O4 by a hydrothermal process
L. C. Dong,
Y. B. Zhong*,
S. Zhe,
T. Y. Zheng and
H. Wang
Shanghai Key Laboratory of Advanced Special Steels, Shanghai University, Shanghai 200072, China. E-mail: yunboz@staff.shu.edu.cn
First published on 3rd February 2016
Abstract
In this paper, manganese oxides/oxyhydroxides were prepared by a low-temperature hydrothermal process in a static magnetic field. The products were analyzed with XRD, TEM, HRTEM and VSM. Single-crystalline nanowires (20 nm in diameter and 1 μm in length) of MnOOH were obtained under zero magnetic fields. However, some cubic particles of Mn3O4 were formed when a static magnetic field was applied. Furthermore, the quantity of Mn3O4 increased with the increase of the magnetic flux density. Quantitative thermodynamic calculation results showed that the magnetic Gibbs energy would change the direction of the chemical reaction to obtain Mn3O4.
Introduction
Over the past few years, manganese compounds, such as various manganese oxides/oxyhydroxides have attracted considerable research interest due to their promising application potentials in several fields, e.g., catalysis,1–6 chemical sensing,7 ion exchange,8 molecular adsorption,9 magnetic applications,10,11 batteries,12,13 and supercapacitors.14–16 For instance, MnOOH, is used as an effective precursor to synthesize intercalation compounds and lithium manganese oxides for rechargeable lithium-ion batteries.17 Moreover, Mn3O4 is known to be an active catalyst in several oxidations and reductions. All of the above reports reveal the great interest in synthesizing manganese oxides with various forms/structures for a very wide variety of applications. On the basis of the above application potentials, various methods have been developed to synthesis manganese oxides with desired shape in order to obtain ideal functions for their unique applications.The high static magnetic field could be easily obtained with the development of superconducting technology, which has opened a promising way to modulate the morphology and magnetic property of nanomaterials. So the influence of magnetic field on the preparation of nanomaterials has been the subject of interest to researchers in recent years. On one hand, the magnetic field can significantly influence the growth behavior of nanoparticles. Wang18 found that single-crystalline nanowires of Fe3O4 were formed under 0.35 T magnetic field, while square and hexagonal particles were formed under zero magnetic fields. On the other hand, the magnetic field can affect the direction of the chemical reaction. Wang19 obtained γ-Fe2O3 nanotubes under 12 T magnetic field, while the as-synthesized samples were a single phase of α-Fe2O3. Furthermore, due to the effect of static magnetic field, manganese oxides/oxyhydroxides were prepared under high magnetic field.
Hydrothermal synthesis is a common method widely used to prepare nano-materials in industry. The advantage of the hydrothermal method compared with others is that the crystalline precipitates at relatively low temperature with high deposition rates. In this communication, we report the preparation of manganese oxides/oxyhydroxides by a hydrothermal process in a static magnetic field, and the effects of the static magnetic field on the morphology and magnetic property of nanoparticles were studied.
Results and discussion
Fig. 1 shows the XRD patterns of the samples synthesized by hydrothermal process in various magnetic flux densities (MFD). It could be known that the samples directly extracted were monoclinic MnOOH with lattice constants a = 5.3 Å, b = 5.278 Å, and c = 5.307 Å (JCPDS 41-1379) in zero magnetic fields. When a static magnetic filed was introduced, some diffraction peaks appeared which could be indexed to the tetragonal Mn3O4 with lattice constants a = b = 5.762 Å and c = 9.470 Å (JCPDS 24-0734). In addition, the diffraction peaks of Mn3O4 increased while the diffraction peaks of MnOOH decreased with the increase of MFD. When the MFD reached 6 T, all diffraction peaks can be indexed to Mn3O4, accompanied by the disappearance of MnOOH.Fig. 2 displays TEM images of the samples prepared under static magnetic fields, which clearly reveal the effect of magnetic field on the nucleation and growth process of manganese oxides/oxyhydroxides. It can be found that the products consisted of a large quantity of nanowires (diameter 20 nm and length about 1 μm) without magnetic field. However, considerable changes in the morphology of the particles could be found when a static magnetic field was applied. In addition to nanowires, some cubic particles of size 20–50 nm were observed in the samples when a magnetic field was applied. Moreover, with the increase of MFD, more and more cubic particles were obtained. Finally, a large number of cubic particles formed, accompanied by the disappearance of the nanowires when MFD reached 6 T. The proportion of cubic particles to the samples obtained in various magnetic field intensities was summarized in Table 1. The proportion of cubic particles to the samples was increased with the increase of MFD. The results clearly showed that the static magnetic field could be responsible for the formation of cubic particles.
 | ||
| Fig. 2 SEM images of the samples obtained in various magnetic field intensities. (a) 0 T, (b) 0.6 T, (c) 1 T, (d) 2 T, (e) 4 T, (f) 6 T. | ||
| Magnetic field | 0 T | 0.6 T | 1 T | 2 T | 4 T | 6 T |
| Cubic particles | 5.5% | 10.7% | 51.2% | 73.6% | 87.7% | 98.3% |
When a 2 T magnetic field was applied, there were not only nanowires products but also cubic nanoparticles. The representative HRTEM images of manganese oxides/oxyhydroxides products are shown in Fig. 3. The HRTEM and electron diffraction pattern (Fig. 3(a)) demonstrated that the single-crystalline nature of nanowires corresponded to the plane of MnOOH, compared with the XRD results (shown in Fig. 1). The direction of the zone of electron beam, [101], could be clearly defined from the direction of (020) and (202) diffraction dots. The HRTEM image showed that MnOOH nanowires grew along [011], with the distinct lattice spacing of 3.5 Å. What's more, the HRTEM and electron diffraction pattern (Fig. 3(b)) demonstrated that the single-crystalline nature of cubic particles corresponded to the plane of Mn3O4, compared with the XRD results. Due to the results of Fig. 2 and 3, the shape of Mn3O4 as-prepared was cubic, while MnOOH preferred to form nanowires. In addition, the samples directly extracted were MnOOH nanowires in zero magnetic fields, while Mn3O4 nanoparticles increased with the increase of MFD. The results of TEM and HRTEM were consistent with the results of XRD.
 | ||
| Fig. 3 HRTEM images of the samples obtained in 2 T magnetic field. (a) Nanowires, (b) nanoparticles. | ||
In order to examine the growth evolution process of manganese oxides/oxyhydroxides, the SEM images of products prepared at different times were shown in Fig. 4. Fig. 4(a)–(c) revealed the growth of nanowires prepared without magnetic field, while Fig. 4(d)–(f) indicated the growth of cubic particles obtained with magnetic field. The results showed that a lot of nanowires (diameter 5–10 nm and length about 200 nm) were formed in 1 h without magnetic field. In addition, the nanowires grew larger when the reaction time was extended. However, when the magnetic field was introduced, numbers of spherical and cubic particles were obtained in 1 h. What's more, the size of cubic particles increased over time.
 | ||
| Fig. 4 SEM images of the samples prepared at different times. (a) 0 T-1 h, (b) 0 T-2 h, (c) 0 T-4 h, (d) 6 T-1 h, (e) 6 T-2 h, (f) 6 T-4 h. | ||
Schematic illustration of the formation of different manganese oxides/oxyhydroxides under magnetic field was shown Fig. 5. MnOOH was formed without magnetic field. The growth of MnOOH nuclei would be modified into small nanowires due to the anisotropic growth, and then these smaller nanowires might grow larger.20 However, when a magnetic field was applied, Mn3O4 was obtained. Crystal growth should occur rapidly on high-free-energy facets as suggested by the Wuff facet theory. Mn3O4 preferred to form cubic particles.21
 | ||
| Fig. 5 Schematic illustration of the formation of different manganese oxides/oxyhydroxides under magnetic field. | ||
The TGA cures of the samples prepared with and without magnetic field were shown in Fig. 6. The samples were heated at a rate of 10 °C min−1 from 50 °C to 700 °C in flowing N2 air. From Fig. 6(a), there were two significant weight loss presenting at the temperature range 250–310 °C and 520–550 °C. The weight loss at 250–300 °C should correspond to the deoxylation,22 accompanied by the nucleation of β-MnO2. The sharp weight loss at 520–550 °C was due to the oxygen release for the reduction of β-MnO2 to α-Mn2O3.23 However, there was only one significant weight loss presenting at the temperature range 250–310 °C in Fig. 6(b), which may be resulted from the loss of crystal water.
The former results indicate that the magnetic field affected the phase of samples intensively, because the only difference in preparation method between the samples is the MFD. From a thermodynamic perspective, magnetic field, like temperature, is an independent variable that changes the free energy, and makes the chemical reaction turn to generate Mn3O4, which may be a major reason. The formation reactions can be expressed by following equations (eqn (1) is for formation of MnOOH and eqn (2) is for formation of Mn3O4). The free energy changed by magnetic field can be expressed as eqn (3).18
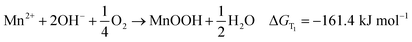 | (1) |
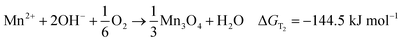 | (2) |
When a static magnetic field is applied, the resulting free-energy changes are classified into two terms:16 the thermal Gibbs free energy, ΔGT, and the magnetic Gibbs free energy, ΔGM. Therefore, if a static magnetic field is imposed during the preparation of manganese oxides/oxyhydroxides, a new equilibrium, (ΔGT + ΔGM), will be reached.19
ΔGT1 is the thermal Gibbs free energy for formation of MnOOH, and ΔGT2 is the thermal Gibbs free energy for formation of Mn3O4. ΔGT1 is lower than ΔGT2 at 120 °C, which indicates that the product of the decomposition for MnCl2 was MnOOH without a static magnetic field. However, when a static magnetic field is applied, a new equilibrium, (ΔGT + ΔGM), will be reached. The magnetic Gibbs free energy, ΔGM, can be expressed as
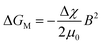 | (3) |
The magnetic property of the as-prepared samples under a static magnetic field was also investigated. Fig. 7 shows magnetic hysteresis curves for the samples measured at room temperature. It showed that all of the samples were superparamagnetic. When the particle sizes are below a critical size (DC), which typically lies below 100 nm, normal microscopic multidomain ferromagnetic (FM) structure is energetically unfavorable, and the particles were in the single domain state.25 In this state, the mechanism of magnetization reversal can only occur via the rotation of the magnetization vector from one easy magnetic direction to another over the magnetic anisotropy barrier, which would result in the remanence and coercivity being zero. In addition, the hysteresis curves indicated a trend that the magnetism of the samples was increased with the enhancement of the magnetic field intensity. On one hand, a large number of Mn3O4 were formed, accompanied by the disappearance of MnOOH with increase of magnetic field intensity, and the magnetism of Mn3O4 is stronger than MnOOH, which might be a major reason. On the other hand, the high shape anisotropy of the nanowires may prevent them from magnetizing in directions other than along their easy magnetic axes, which might result that the magnetism of nanowires was lower than nanoparticles. For these two reasons, the magnetism of the samples was increased with the enhancement of MFD. Further work should be done to clarify the physical origin completely.
 | ||
| Fig. 7 M–H curves of the samples by hydrothermal synthesis in various magnetic field intensities at room temperature. | ||
Experimental section
The chemical reagents used in the work were manganese(II) chloride tetrahydrate (MnCl2·4H2O, CAS no. 13446-34-9) and sodium hydroxide (NaOH, CAS no. 1310-73-2). All chemicals were analytical grade and purchased from Sinopharm Chemical Reagent Co. Ltd. In a typical process, 0.004 mol of MnCl2·4H2O was put into distilled water at room temperature to form an aqueous solution. 20 ml alkaline solution of 3 M NaOH was slowly dripped into the above solution to precipitate Mn2+ ions during constant magnetic stirring, and then a brown suspension solution was formed. The suspension solution was transferred to a stainless steel Teflon-lined autoclave (15 ml effective volume) with a filling 80% volume of the autoclave for hydrothermal treatment. The autoclave was sealed and maintained at 120 °C for 4 h with and without magnetic field. Finally, they were cooled down to room temperature. The products were filtered and washed with distilled water and absolute ethanol for few times, and then dried in an oven at 60 °C for 8 h. A schematic diagram of the apparatus used in this experiment is shown in Fig. 8. The autoclave was put in the center of the magnetic field. The magnetic field (up to 6 T) was applied parallel to the longitudinal direction and the temperature in the furnace chamber could be adjusted automatically by the temperature controlling apparatus.The morphologies of as-prepared samples were observed by a transmission electron microscopy (TEM) and high-resolution transmission electron microscopy (HRTEM) (JEOL JEM-2010F, Japan). The crystal structures were characterized by X-ray diffraction (XRD; Rigaku D/MAX-2200, Tokyo, Japan) with Cu Kα radiation (60 kV and 40 mA). The thermogravimetric analysis was carried out on DSC/DTA-TG simultaneous thermal analyzer (STA 449 F3 Jupiter, NETZSCH, Germany). The magnetic properties were also measured using a vibrating sample magnetometer (VSM, LakeShore 7407, America) at room temperature.
Conclusions
In summary, a static magnetic field can significantly influence the phase and magnetic property of the samples synthesized by low-temperature hydrothermal method. Single-crystalline nanowires (diameter 20 nm and length about 1 μm) of MnOOH were obtained under zero magnetic fields. However, some cubic particles of Mn3O4 were formed when a 1 T magnetic field was applied. What's more, the yield of Mn3O4 increased with the increase of MFD. VSM results confirmed that all of the samples were superparamagnetic, and the magnetism of the samples was increased with the enhancement of the magnetic field intensity. All results showed that a static magnetic field could destroy the stability of the chemical reaction, and make the chemical reaction turn to generate the products with higher magnetism. It is excited that the process could be a promising technique to synthesize other materials, and the related work is still under investigation.Acknowledgements
The authors would like to thank National Science Foundation of China (No. 50974085, No. 51034010), Key Project supported by Science and Technology Commission of Shanghai Municipality (No. 13JC1402500), Development Foundation for Talents in Shanghai, China (No. 2009046), National High-tech R&D Program of China (No. 2009AA03Z109). Financial Supported by Natural Science Foundation of Jiangsu Province, China (No. BK2011501), Financial Supported by Innovation Fund for Technology Based Firms, China (12C26213202452).Notes and references
- T. Yamashita and A. Vannice, J. Catal., 1996, 161, 254 CrossRef CAS.
- V. D. Makwana, Y. C. Son, A. R. Howell and S. L. Suib, J. Catal., 2002, 210, 46 CrossRef CAS.
- C. S. Liu, L. J. Zhang, F. B. Li, Y. Wang, Y. Gao, X. Z. Li, W. D. Cao, C. H. Feng, J. Dong and L. N. Sun, Ind. Eng. Chem. Res., 2009, 48, 10408 CrossRef CAS.
- H. Huang, S. Sithambaram, C. H. Chen, C. K. Kithongo, L. P. Xu, A. Iyer, H. F. Garces and S. L. Suib, Chem. Mater., 2010, 22, 3664 CrossRef CAS.
- M. Huynh, D. Kwabena Bediako and D. G. Nocera, J. Am. Chem. Soc., 2014, 136, 6002 CrossRef CAS PubMed.
- T.-D. Dang, A. N. Banerjee, S. W. Joo and B.-K. Min, Ind. Eng. Chem. Res., 2014, 53, 9743 CrossRef CAS.
- M. Nogami, T. Maeda and T. Uma, Sens. Actuators, B, 2009, 137, 603 CrossRef CAS.
- O. Giraldo, S. L. Brock, W. S. Willis, M. Marquez, S. L. Suib and S. J. Ching, J. Am. Chem. Soc., 2000, 122, 9330 CrossRef CAS.
- Y. F. Shen, R. P. Zerger, R. N. Deguzman, S. L. Suib, L. Mccurdy, D. I. Potter and C. L. Oyoung, Science, 1993, 260, 511 CAS.
- W. S. Seo, H. H. Jo, K. Lee, B. Kim, S. J. Oh and J. T. Park, Angew. Chem., Int. Ed., 2004, 43, 1115 CrossRef CAS PubMed.
- L. Luo, H. T. Zhu, J. K. Liang, G. H. Rao, J. B. Li and Z. M. Du, J. Phys. Chem. C, 2010, 114, 8782 Search PubMed.
- K. S. Park, M. H. Cho, S. H. Park, K. S. Nahm, Y. K. Sun, Y. S. Lee and M. Yoshio, Electrochim. Acta, 2002, 47, 2937–2942 CrossRef CAS.
- J. Kim and A. Manthiram, Nature, 1997, 390, 265 CrossRef CAS.
- C. C. Hu and T. W. Tsou, Electrochem. Commun., 2002, 4, 105 CrossRef CAS.
- M. Q. Wu, G. A. Snook, G. C. Chen and D. J. Fray, Electrochem. Commun., 2004, 6, 499 CrossRef CAS.
- S. W. Lee, J. Kim, S. Chen, P. T. Hammond and Y. Shao-Horn, ACS Nano, 2010, 4, 3889 CrossRef CAS PubMed.
- M. M. Thackery, A. Dekock, M. H. Rossouw, D. Liles, R. Bittihn and D. J. Hoge, Electrochem. Soc., 1992, 139, 363 CrossRef.
- J. Wang, Q. W. Chen, C. Zeng and B. Y. Hou, Adv. Mater., 2004, 16, 137 CrossRef CAS.
- J. Wang, Y. Ma and K. Watanabe, Chem. Mater., 2008, 20, 20 CrossRef CAS.
- L. L. Lan, Q. J. Li and G. R. Gu, et al., J. Alloys Compd., 2015, 644, 430–437 CrossRef CAS.
- N. N. Zhao, W. Nei and X. B. Liu, et al., Small, 2008, 4, 77–81 CrossRef CAS PubMed.
- R. Giovanoli and V. Leuenberger, Helv. Chim. Acta, 1969, 52, 2333 CrossRef CAS.
- R. Giovanoli, Thermochim. Acta, 1994, 234, 303 CrossRef CAS.
- D. R. Lide, Handbook of Chemistry and Physics, CRC Press, 2009, pp. 4–132 Search PubMed.
- N. Paunovic, Z. V. Popovic and Z. D. Dohcevic-Mitrovic, J. Phys.: Condens. Matter, 2012, 18, 177 Search PubMed.
| This journal is © The Royal Society of Chemistry 2016 |



