An NHC silver(I) macrometallocycle: synthesis, structure and selective recognition of iodide anions†
Qing-Xiang Liu*,
Jun-Ren Chen,
Xiao-Feng Sun,
Xiao-Jun Zhao,
Zhi-Xiang Zhao and
Kang-Qing Cai
Key Laboratory of Inorganic-Organic Hybrid Functional Materials Chemistry (Tianjin Normal University), Ministry of Education, Tianjin Key Laboratory of Structure and Performance for Functional Molecules, College of Chemistry, Tianjin Normal University, Tianjin 300387, China. E-mail: tjnulqx@163.com
First published on 15th January 2016
Abstract
A bis-benzimidazolium salt with oligoether-linker LH2·(PF6)2 and its 13-membered N-heterocyclic carbene silver(I) crown ether 1 have been synthesized and characterized. The coordination geometry of the silver(I) ion of 1 is nearly linear with a C(7)–Ag(1)–C(23) bond angle of 179.6(1)°. In the crystal packing of 1, a 2D supramolecular layer is formed by π–π stacking interactions from benzimidazole rings. In particular, the selective recognition of I− using 1 as a receptor was studied on the basis of fluorescence and UV/vis spectroscopic titrations.
Introduction
Molecular recognition is a process where a host binds selectively to a guest to produce a specific function.1 A traditional covalent bond is the main force between hosts and guests. However, non-covalent forces, such as van der Waals forces, hydrogen bonds and electrostatic interactions, also play important roles. The recognition of anionic species is a thriving field of research due to the significance of this in biology, medicine and the environment.2 The detection, isolation and identification of anions have shown broad application prospects in membrane transport carriers, chemical sensing and imitation-enzyme-catalyzed organic synthesis.3 Some anionic receptors that can selectively identify alkaline ions (like fluoride ions and acetate ions) and other anions have been reported.4–6 Iodine is a trace element in human beings, and it has an important influence on brain function, cell growth, nerve activity, metabolism and thyroid function. A deficiency or excess of iodine in the human body can cause goiter, hypothyroidism, hyperthyroidism and some other diseases.7 Additionally, iodine has also been widely used in the synthesis of drugs.8 Thus, the analysis and detection of iodide anions is of great importance in daily life. Common detection methods of iodide anions are GC, capillary electrophoresis and atomic absorption spectroscopy.9 In general, it is difficult to bind iodide anions to receptors due to their spherical structure and weak alkaline characteristics. Up to now, only several receptors that can bind iodide selectively have been reported.10 Therefore, the design and synthesis of iodide receptors with relatively simple structures are still a challenge in host–guest chemistry.During the course of searching for receptors, we became interested in N-heterocyclic carbene (NHC) metal crown ethers. In this paper, we report the synthesis and structure of a new N-heterocyclic carbene silver(I) crown ether silver-{C,C′-1,1′-[1,2-ethanediyl-bis(oxy-1,2-ethanediyl)]-bis(3-npropyl-benzimidazolium-1-yl)} hexafluorophosphate (1). In particular, the selective recognition of I− using 1 as a receptor was studied on the basis of fluorescence and UV/vis spectroscopic titrations.
Results and discussion
Synthesis and general characterization of bis-benzimidazolium salt LH2·(PF6)2 and NHC silver(I) crown ether 1
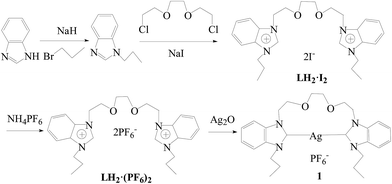
As shown in Scheme 1, benzimidazole as a starting material was alkylated with 1-bromopropane, followed by quaternization with 1,2-bis(2-chloroethoxy)ethane in the presence of NaI to afford bis-benzimidazolium salt, 1,1′-[1,2-ethanediyl-bis(oxy-1,2-ethanediyl)]-bis(3-npropylbenzimidazolium-1-yl) diiodide (LH2·I2). Then anion exchange was carried out with ammonium hexafluorophosphate in methanol to give LH2·(PF6)2. Precursor LH2·(PF6)2 is stable to air and moisture, and soluble in organic solvents such as dichloromethane and acetonitrile, and scarcely soluble in diethyl ether, petroleum ether and water. The structure of LH2·(PF6)2 was confirmed by 1H NMR and 13C NMR spectroscopy and elemental analysis. In the 1H NMR spectra of LH2·(PF6)2, the benzimidazolium proton signal (NCHN) appears at δ = 9.58 ppm, which is consistent with the chemical shifts of the known benzimidazolium salts.11Reaction of precursor LH2·(PF6)2 with Ag2O in CH2Cl2 afforded an N-heterocyclic carbene silver(I) complex silver-{C,C′-1,1′-[1,2-ethanediyl-bis(oxy-1,2-ethanediyl)]-bis(3-npropyl-benzimidazolium-1-yl)} hexafluorophosphate (1). Complex 1 is soluble in DMSO and CH3CN, and almost insoluble in diethyl ether. Complex 1 is slightly light-sensitive in solution, but light-stable as a solid. In the 1H NMR spectra of 1, the resonance for the benzimidazolium proton (NCHN) disappeared, and the chemical shifts of other protons are similar to those of the corresponding precursor LH2·(PF6)2. In the 13C NMR spectra of 1, the signal of the carbene carbon was not observed. The absence of the carbene carbon resonance is not unusual, this phenomenon has been reported for some silver(I)-carbene complexes, and may result from the fluxional behavior of NHC complexes.12
Structure of NHC silver(I) crown ether 1
The formation of silver(I) carbene complex 1 was confirmed by 1H NMR and 13C NMR spectroscopy and X-ray crystallography. Crystals of 1 suitable for X-ray diffraction were obtained by slow evaporation of CH2Cl2 in air. The cation of complex 1 contains a 13-membered NHC silver crown ether formed by a silver(I) ion and a chelate biscarbene ligand with a flexible linkage as shown in Fig. 1. The benzimidazole rings within each molecule form a dihedral angle of 16.5°, and the two n-propyl group chains point in contrary directions. The coordination geometry on the silver(I) ion is nearly linear with a C(7)–Ag(1)–C(23) bond angle of 179.5(8)°. The Ag–Ccarbene bond distance is 2.090(3) Å. The internal ring angle (N–C–N) at the carbene center is 106.3(3)°. These values are similar to the corresponding values of other reported NHC silver complexes.13 The Ag⋯O separations are 3.344(3) Å and 3.533(3) Å, respectively, which shows that Ag⋯O interactions can be neglected (sum of van der Waals radii of silver and oxygen = 3.10 Å).In the crystal packing of 1, a 2D supramolecular layer (Fig. 2) is formed by π–π stacking interactions of benzimidazole rings with a face-to-face distance of 3.479(4) Å (the center-to-center distance being 4.276(7) Å).14
 | ||
| Fig. 2 2D supramolecular layer of complex 1 via π–π stacking interactions. All hydrogen atoms are omitted for clarity. | ||
Recognition of iodide anions by NHC silver(I) crown ether 1
The selective recognition of some anions (F−, Cl−, Br−, I−, H2PO4−, HSO4−, OAc− and NO3−, with their cation being tetrabutyl ammonium (TBA+)) using 1 as a receptor was investigated by fluorescence and UV/vis titrations in acetonitrile at 25 °C. As shown in Fig. 3, receptor 1 displays an emission at 362 nm (λex = 235 nm, excitation and emission slits: 3 nm and 3 nm), which is attributed to the emission of benzimidazole. Upon the addition of 15 equiv. of F−, Cl−, Br−, H2PO4−, HSO4−, OAc− and NO3−, respectively, the fluorescence emission intensity of 1 does not obviously change, whereas the addition of the same amount of I− causes a great decrease of the emission intensity at 362 nm. | ||
| Fig. 3 Fluorescence spectra of 1 (1.0 × 10−5 mol L−1) upon addition of different tetrabutyl ammonium salts (15.0 equiv.) in acetonitrile (λex = 235 nm). | ||
As shown in Fig. 4, upon the titration of I− into a solution of 1, the fluorescence intensities at 362 nm decrease gradually as the concentration of I− increases. As shown in the inset of Fig. 4, when the ratio of CI−/C1 is below 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, the fluorescence intensity decreases sharply with the increasing concentration of I−. When the ratio is in the range of 1
1, the fluorescence intensity decreases sharply with the increasing concentration of I−. When the ratio is in the range of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 20
1 to 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, the inclination of the fluorescence intensity decrease becomes slow. When the ratio exceeded 20
1, the inclination of the fluorescence intensity decrease becomes slow. When the ratio exceeded 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, higher CI− will not lead to a further emission decrease. The Stern–Volmer constant KSV (ref. 10q–s) was calculated to be 2.3 × 104 M−1 (R = 0.994) using the conventional Stern–Volmer relationship in acetonitrile at 25 °C (eqn (1), Fig. S1†).
1, higher CI− will not lead to a further emission decrease. The Stern–Volmer constant KSV (ref. 10q–s) was calculated to be 2.3 × 104 M−1 (R = 0.994) using the conventional Stern–Volmer relationship in acetonitrile at 25 °C (eqn (1), Fig. S1†).
| F0/F = 1 + KSVCI− | (1) |
In UV/vis titration experiments (Fig. 5), the UV/vis absorption spectra of 1 increased gradually with the enhancement of the molar fraction of I−. In addition, a red shift at 201–210 nm with increasing concentration of I− was observed, which indicates that the π–π transition energy of the benzene rings in 1 decreased due to the interactions between 1 and I−.16 It is notable that a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complexation stoichiometry for 1·I− was established by Job’s plot analysis at 205 nm (inset of Fig. 5),17 where the products (χΔA) between molar fractions and the discrepancy of the absorption bands were plotted against the molar fractions (χ) of 1. When the molar fraction of 1 was 0.5, the χΔA values for 1·I− reach a maximum.18 The stability constant K for 1·I− calculated from the plot of A0/ΔA versus 1·I− based on the standard Benesi–Hildebrand method is 1.18 × 105 M−1 (R = 0.999) (eqn (2), Fig. S3†).
1 complexation stoichiometry for 1·I− was established by Job’s plot analysis at 205 nm (inset of Fig. 5),17 where the products (χΔA) between molar fractions and the discrepancy of the absorption bands were plotted against the molar fractions (χ) of 1. When the molar fraction of 1 was 0.5, the χΔA values for 1·I− reach a maximum.18 The stability constant K for 1·I− calculated from the plot of A0/ΔA versus 1·I− based on the standard Benesi–Hildebrand method is 1.18 × 105 M−1 (R = 0.999) (eqn (2), Fig. S3†).
| A0/(A0 − A) = [εr/(εr − εc)](1/KCI− + 1) | (2) |
In order to further explore the special selective ability of I− using 1 as a receptor, competition experiments were conducted. Receptor 1 (1.0 × 10−5 mol L−1) is firstly mixed with 5 equiv. of various anions (F−, Cl−, Br−, H2PO4−, HSO4−, OAc− and NO3−), and then 5 equiv. of I− are added. Fluorescence spectra are exploited to monitor the competition events. As displayed in Fig. S4,† only H2PO4− causes the fluorescence intensity to increase by about 20% compared with that in the presence of I− alone. The fluorescence intensities of other anions are similar to that in the presence of I− alone. The results show that host 1 can discriminate between I− and other anions.
A fluorescence decrease similar to that shown in Fig. 3 was also observed after the addition of other iodide salts (15.0 × 10−5 mol L−1) with different countercations (K+, Na+, NH4+, Cu+ and Hg2+) to 1 (1.0 × 10−5 mol L−1) (Fig. S5†). Thus, the different cations had no obvious influence on the binding between 1 and iodide anions.
Interactions of 1 with I−
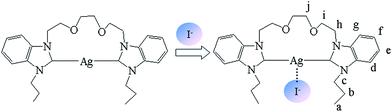
From the structure of 1, we can see that the silver(I) ion is the most likely binding site for I− via Ag⋯I interactions. To obtain detailed information on the binding mode between 1 and I−, 1H NMR titration experiments were carried out in DMSO-d6. The spectral differences are depicted in Fig. 6 (whole 1H NMR spectra are given in Fig. S6 in ESI†). The signals of Hd, Hg, He and Hf of benzimidazole shifted upfield by 0.03–0.04 ppm upon the addition of 1 equiv. of I−, and the signals of Hc and Hh of the CH2 around benzimidazole (Scheme 2) shifted upfield by 0.05 ppm upon the addition of 1 equiv. of I−. These changes should be attributed to the interactions between I− and Ag+. As expected, these observations show that an iodide anion is captured by Ag+ via Ag⋯I interactions. In addition, the signals of the above-mentioned protons do not obviously change upon the addition of further equivalents of I− (Fig. 6(c) and (d)). This indicates that 1 and I− have 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complexation. In high-resolution mass spectrometry (HRMS) analysis of 1·I− (Fig. S7†), an M+ (669.07) of 1·I− is observed, which provides additional evidence for the formation of a 1
1 complexation. In high-resolution mass spectrometry (HRMS) analysis of 1·I− (Fig. S7†), an M+ (669.07) of 1·I− is observed, which provides additional evidence for the formation of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex between 1 and I−. These results are consistent with the results of the UV/vis titration experiments (shown in the inset of Fig. 5).
1 complex between 1 and I−. These results are consistent with the results of the UV/vis titration experiments (shown in the inset of Fig. 5).
 | ||
| Fig. 6 Partial 1H NMR spectra in DMSO-d6. (a) Complex 1; (b) 1 and 1 equiv. of TBAI; (c) 1 and 1.5 equiv. of TBAI; (d) 1 and 2 equiv. of TBAI. | ||
To further understand the complexation behavior of 1 and I−, infrared spectroscopy of 1 and 1·TBAI was performed. In the infrared spectra (Fig. S8†), we observed that the ν(C–N) absorption bands move from 1122 cm−1 in free 1 to 1140 cm−1 in 1·I−, which is attributed to the electron-donating effect of I− as shown in Scheme 2.
By comprehensive analysis of the structure of 1, and the 1H NMR spectra, high-resolution mass spectra and infrared spectra of 1 and 1·TBAI, we can see that the binding force of 1 to I− originates mainly from Ag⋯I interactions due to the strong affinity of silver(I) ions to I−. When I− is bound by silver(I) ions, the remarkable decrease of the fluorescence intensity of 1 may be attributed to a metal (dπ) to ligand (π*) charge transfer (MLCT) process promoted by the electron-donating effect of I−.20
Conclusions
In summary, a new NHC silver(I) crown ether 1 has been synthesized and characterized. Fluorescence and UV/vis spectroscopic titrations indicate that complex 1 has great affinity to I−. 1H NMR , HRMS and IR spectra show that I− is captured through Ag⋯I interactions. The special selective ability of 1 for I− means that it can discriminate between I− and other anions in acetonitrile. Even a detection limit as low as 6.1 × 10−7 mol L−1, 1 is still able to sensitively detect I−. A high selectivity and sensitivity and the great affinity to I− mean this complex has a potential application for the quantitative measurement of I−.Experimental section
General procedures
All manipulations were performed using Schlenk techniques, and solvents were purified by standard procedures. All the reagents for syntheses and analyses were of analytical grade and used without further purification. Melting points were determined with a Boetius Block apparatus. 1H and 13C NMR spectra were recorded on a Bruker Avance III spectrometer at 400 MHz and 100 MHz, respectively. Chemical shifts, δ, are reported in ppm relative to the internal standard TMS for both 1H and 13C NMR. J values are given in Hz. Elemental analyses were measured using a Perkin-Elmer 2400C Elemental Analyzer. The luminescence spectra were conducted on a Cary Eclipse fluorescence spectrophotometer. UV-vis spectra were obtained using a JASCO-V570 spectrometer. EI mass spectra were recorded on a VG ZAB-HS mass spectrometer (VG, U.K.). IR spectra (KBr) were recorded on an Bruker Equinox 55 spectrometer.Preparation of 1,1′-[1,2-ethanediyl-bis(oxy-1,2-ethanediyl)]-bis(3-npropyl-benzimidazolium-1-yl) dihexafluorophosphate (LH2·(PF6)2)
A tetrahydrofuran (THF) (15 mL) solution of benzimidazole (1.000 g, 8.5 mmol) was added to a suspension of oil-free sodium hydride (0.244 g, 10.2 mmol) in THF (25 mL) and stirred for 1 h at 60 °C. Then a THF (20 mL) solution of n-propyl bromide (1.145 g, 9.3 mmol) was dropwise added to the above mixture. The mixture was continually stirred for 48 h at 60 °C. After filtration, a yellow solution was obtained. The solvent was removed with a rotary evaporator and H2O (30 mL) was added to the residue. The solution was extracted with CH2Cl2 (3 × 20 mL), and the extracting solution was dried over anhydrous MgSO4. After removing CH2Cl2, a pale yellow oil 1-npropyl-benzimidazole was obtained. Yield: 1.221 g (90%).A THF (50 mL) suspension of 1-npropyl-benzimidazole (1.429 g, 8.9 mmol) and 1,2-bis(2-chloroethoxy)ethane (0.766 g, 4.1 mmol) in the presence of NaI (1.474 g, 9.8 mmol) was stirred for three days in a reflux, and a white precipitate was formed. The product was filtered and washed with THF. The white powder of 1,1′-[1,2-ethanediyl-bis(oxy-1,2-ethanediyl)]-bis(3-npropyl-benzimidazolium-1-yl) diiodide (LH2·I2) was obtained by recrystallization from methanol/diethyl ether. Yield: 2.207 g (78%). Mp: 170–172 °C. MS (EI): m/z [M − I−]+ = 563.1869, m/z [M − 2I−]+ = 435.2749.
NH4PF6 (1.705 g, 10.5 mmol) was added to a methanol (50 mL) solution of LH2·I2 (3.000 g, 4.4 mmol) whilst stirring and a white precipitate was formed immediately. The product was collected by filtration, washed with small portions of cold methanol, and dried in a vacuum to give 1,1′-[1,2-ethanediyl-bis(oxy-1,2-ethanediyl)]-bis(3-npropyl-benzimidazolium-1-yl) hexafluorophosphate (LH2·(PF6)2). Yield: 2.875 g (90%). Mp: 150–152 °C. Anal. calcd for C26H36F12N4O2P2: C, 42.98; H, 5.00; N, 7.71%. Found: C, 42.72; H, 4.63; N, 7.95%. 1H NMR (400 MHz, DMSO-d6): δ 0.84 (t, J = 7.2, 6H, CH3), 1.89 (q, J = 7.2 Hz, 4H, CH2), 3.46 (s, 4H, CH2), 4.43 (t, J = 7.2 Hz, 4H, CH2), 4.58 (t, J = 4.8, 4H, CH2), 7.63–7.69 (m, 4H, PhH), 7.96–7.98 (q, J = 2.9 Hz, 2H, PhH), 8.02–8.04 (q, J = 3.0 Hz, 2H, PhH), 9.58 (s, 2H, 2-bimiH). 13C NMR (100 MHz, DMSO-d6): δ 10.4 (CH3), 21.9 (CH2), 46.6 (CH2), 48.0 (CH2), 67.4 (CH2), 69.4 (CH2), 113.5 (PhC), 113.7 (PhC), 126.5 (PhC), 126.6 (PhC), 130.8 (PhC), 131.1 (PhC), 142.1 (2-bimiC) (bimi = benzimidazole). MS (EI): m/z [M − (PF6−)]+ = 581.2462, m/z [M − 2(PF6−)]+ = 435.2752.
Preparation of silver-{C,C′-1,1′-[1,2-ethanediyl-bis(oxy-1,2-ethanediyl)]-bis(3-npropyl-benzimidazolium-1-yl)} hexafluorophosphate (1)
Silver oxide (0.070 g, 0.3 mmol) was added to a dichloromethane (30 mL) solution of precursor LH2·(PF6)2 (0.200 g, 0.3 mmol) and the suspension was stirred for 24 h under reflux. The mixture was filtered and concentrated to 5 mL, and diethyl ether (5 mL) was added to precipitate a white powder. Isolation by filtration yielded complex 1. Yield: 0.072 g (38%). Mp: 230–231 °C. Anal. calcd for C26H34F6AgN4O2P: C, 45.43; H, 4.99; N, 8.15%. Found: C, 45.68; H, 4.62; N, 8.53%. 1H NMR (400 MHz, DMSO-d6): δ 0.97 (m, J = 1.0, 6H, CH3), 1.91–1.96 (m, J = 1.6, 4H, CH2), 3.51 (s, 4H, CH2), 3.78 (t, J = 1.0, 4H, CH2), 4.56 (t, J = 2.0, 4H, CH2), 4.74 (t, J = 1.0, 4H, CH2), 7.46–7.49 (m, J = 1.0, 4H, PhH), 7.86–7.89 (m, J = 0.6, 4H, PhH). 13C NMR (100 MHz, DMSO-d6): δ 11.1 (CH3), 23.2 (CH2), 48.3 (CH2), 50.0 (CH2), 67.7 (CH2), 69.4 (CH2), 111.7 (PhC), 112.0 (PhC), 124.0 (PhC), 133.0 (PhC), 133.2 (PhC). The carbene carbon was not observed. MS (EI): m/z [M − (PF6−) − 1]+ = 541.1733.UV titrations
UV titrations were performed on a JASCO-V570 spectrometer using a 1 cm path-length quartz cuvette. The acetonitrile used in the titrations was freshly distilled over calcium hydride. Titrations were carried out by placing the sensor 1 (1 × 10−5 mol L−1) into the 4 mL cuvette and adding increasing amounts of I− (0.0–20.0 × 10−5 mol L−1) using a microsyringe. The absorption spectra were recorded in the range of 200–400 nm. After each addition, an equilibration time of 8–10 min was allowed before the absorption spectra were recorded. Statistical analysis of the data was carried out using Origin 8.0.Fluorescence titrations
Fluorescence titrations were performed on a Cary Eclipse fluorescence spectrophotometer using a 1 cm path-length quartz fluorescence cell. The acetonitrile used in the titrations was freshly distilled over calcium hydride. Titrations were carried out by placing the sensor 1 (1 × 10−5 mol L−1) into the 4 mL cuvette and adding increasing amounts of I− (0.0–24.0 × 10−5 mol L−1) using a microsyringe. The sensor solution was excited at 235 nm, and the emission spectra were recorded in the range of 305–480 nm. After each addition, an equilibration time of 8–10 min was allowed before the fluorescence intensity was recorded. Statistical analysis of the data was carried out using Origin 8.0.X-ray structure determinations
For complex 1 selected single crystals were mounted on a Bruker APEX II CCD diffractometer at 296(2) K with Mo–K radiation (λ = 0.71073 Å) and an ω scan mode. Data collection and reduction were performed using SMART and SAINT software21 with frames of 0.6° oscillation in the range of 1.8° < θ < 25°. An empirical absorption correction was applied using the SADABS program.22 The structures were solved by direct methods and all non-hydrogen atoms were subjected to anisotropic refinement by full-matrix least squares on F2 using the SHELXTL package.23 All hydrogen atoms were generated geometrically (C–H bond lengths fixed at 0.96 Å), assigned appropriated isotropic thermal parameters and included in the final calculations. Crystallographic data are summarized in Table 1 for 1.| a GOF = [Σw(Fo2 − Fc2)2/(n − p)]1/2, where n is the number of reflection and p is the number of parameters refined.b R1 = Σ(||Fo| − |Fc||)/Σ|Fo|; wR2 = [Σ[w(Fo2 − Fc2)2 ]/Σw(Fo2)2]1/2. | |||
|---|---|---|---|
| Chemical formula | C26H34AgF6N4O2P | F(000) | 700 |
| Formula weight | 687.41 | Cryst size, mm | 0.25 × 0.17 × 0.16 |
| Cryst syst | Triclinic | θmin, θmax, deg | 1.82, 25.01 |
| Space group | P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
T, K | 296(2) |
| a, Å | 11.291(2) | No. of data collected | 7363 |
| b, Å | 11.448(2) | No. of unique data | 5021 |
| c, Å | 11.816(2) | No. of refined params | 418 |
| α, deg | 108.288(3) | Goodness-of-fit on F2a | 1.042 |
| β, deg | 90.856(3) | Final R indicesb [I > 2σ(I)] | |
| γ, deg | 96.519(3) | R1 | 0.0382 |
| V, Å3 | 1438.7(4) | wR2 | 0.0978 |
| Z | 2 | R indices (all data) | |
| Dcalcd, mg m−3 | 1.587 | R1 | 0.0423 |
| Abs coeff, mm−1 | 0.826 | wR2 | 0.1016 |
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (No. 21172172 and 21572159), Tianjin Natural Science Foundation (No. 11JCZDJC22000) and The Program for Innovative Research Team in University of Tianjin (TD12-5038).References
- E. T. Kool, Chem. Rev., 1997, 97, 1473 CrossRef CAS PubMed.
- (a) A. Bianchi, K. Bowman-James and E. Garcia-Espana, Supramol. Chem. Anions, 1997 Search PubMed; (b) P. A. Gale, S. E. Garcia-Garrido and J. Garric, Chem. Soc. Rev., 2008, 27, 151 RSC; (c) V. Amendola, M. Bonizzoni, D. Esteban-Gomez and A. P. Davis, Coord. Chem. Rev., 2006, 250, 2939 CrossRef; (d) M. X. Wang, X. H. Zhang and Q. Y. Zheng, Angew. Chem., Int. Ed., 2004, 43, 838 CrossRef CAS PubMed; (e) S. O. Kang, M. A. Hossain and K. Bowman-James, Coord. Chem. Rev., 2006, 250, 3038 CrossRef CAS; (f) S. S. Huang, S. He, Y. Lu, F. F. Wei, X. S. Zeng and L. C. Zhao, Chem. Commun., 2011, 47, 2408 RSC; (g) E. Garcia-Espana, P. Diaz, M. J. Llinares and A. Bianchi, Coord. Chem. Rev., 2006, 250, 2952 CrossRef CAS; (h) L. Fabbrizzi, M. Licchelli, F. Sancenon and A. Taglietti, Coord. Chem. Rev., 2006, 250, 1451 CrossRef; (i) X. Q. Zhang, Y. P. Qiu, B. Rao and M. M. Luo, J. Organomet. Chem., 2009, 28, 3093 CrossRef CAS.
- (a) V. Kral and J. L. Sessler, Tetrahedron, 1995, 51, 539 CrossRef CAS; (b) P. Buhlmann, E. Pretsch and E. Bakker, Chem. Rev., 1998, 98, 1593 CrossRef PubMed; (c) G. M. Hubner, J. Glaser and C. Seel, Angew. Chem., Int. Ed. Engl., 1999, 38, 383 CrossRef CAS; (d) P. A. Gale, Coord. Chem. Rev., 2000, 199, 181 CrossRef CAS; (e) P. D. Beer and P. A. Gale, Angew. Chem., Int. Ed., 2001, 40, 486 CrossRef CAS; (f) F. Han, Y. Bao, Z. Yang, T. M. Fyles, J. Zhao, X. Peng, J. Fan, Y. Wu and S. Sun, Chem.–Eur. J., 2007, 13, 2880 CrossRef CAS PubMed.
- J. Yoo, M. S. Kim, S. J. Hong, J. L. Sessler and C. H. Lee, J. Org. Chem., 2009, 74, 1065 CrossRef CAS PubMed.
- (a) X. D. Yu, H. Lin, Z. S. Cai and H. K. Lin, Tetrahedron Lett., 2007, 48, 8615 CrossRef CAS; (b) J. Shao, H. Lin, M. Yu, Z. Cai and H. Lin, Talanta, 2008, 75, 551 CrossRef CAS PubMed.
- Z. Ekmekci, M. D. Yilmaz and E. U. Akkaya, Org. Lett., 2008, 10, 461 CrossRef CAS PubMed.
- (a) WHO, A Ssessment of Iodine Deficiency Disorders and Monitoring Their Elimination: A Guide for Programm Emanagers, 2nd edn, 2001 Search PubMed; (b) F. Delange, B. Benoist and E. Pretell, Thyroid, 2001, 11, 437 CrossRef CAS PubMed; (c) J. T. Dunn, Thyroid, 2001, 11, 363 CrossRef CAS PubMed.
- A. K. Singh and S. Mehtab, Talanta, 2008, 74, 806 CrossRef CAS PubMed.
- C. Caltagirone and P. A. Gale, Chem. Soc. Rev., 2009, 38, 520 RSC.
- (a) K. Mitachi, S. Siricilla, L. Klaic, W. M. Clemons and M. Kurosu, Tetrahedron Lett., 2015, 56, 3441 CrossRef CAS PubMed; (b) S. Memon, A. A. Bhatti, U. Ocak and M. Ocak, Anal. Methods, 2015, 7, 5114 RSC; (c) S. Rastegarzadeh, N. Pourreza and I. Saeedi, Talanta, 2009, 77, 1032 CrossRef CAS PubMed; (d) N. Singh and D. O. Jang, Org. Lett., 2007, 9, 1991 CrossRef CAS PubMed; (e) V. Balzani, New J. Chem., 1996, 20, 723 CAS; (f) W. G. Yang, S. H. Yanga, Q. R. Guo, T. Zhang, K. Y. Wu and Y. H. Hu, Sens. Actuators, B, 2015, 213, 404 CrossRef CAS; (g) R. Indumathy, P. S. Parameswaran, C. V. Aiswarya and B. U. Nair, Polyhedron, 2014, 75, 22 CrossRef CAS; (h) N. Kumari, M. A. Hasan, B. D. Ward and L. Mishra, Ind. Eng. Chem. Res., 2013, 52, 15007 CrossRef CAS; (i) P. Arora, K. Suyal, N. K. Joshi, H. C. Joshi and S. Pant, Spectrochim. Acta, Part A, 2012, 94, 119 CrossRef CAS PubMed; (j) D. Y. Lee, N. Singh, M. J. Kim and D. O. Jang, Org. Lett., 2011, 13, 3024 CrossRef CAS PubMed; (k) H. A. Ho and M. Leclerc, J. Am. Chem. Soc., 2003, 125, 4412 CrossRef CAS PubMed; (l) V. Suresh, N. Ahmed, I. S. Youn and K. S. Kim, Chem.–Asian J., 2012, 7, 658 CrossRef CAS PubMed; (m) N. Ahmed, B. Shirinfar, I. Geronimo and K. S. Kim, Org. Lett., 2011, 13, 5476 CrossRef CAS PubMed; (n) C. Kar, A. Basu and G. Das, Tetrahedron Lett., 2012, 53, 4754 CrossRef CAS; (o) N. Singh, H. J. Jung and D. O. Jang, Tetrahedron Lett., 2009, 50, 71 CrossRef CAS; (p) H. B. Li, C. P. Han and L. Zhang, J. Mater. Chem., 2008, 18, 4543 RSC; (q) E. Bernal, J. A. Lebron, B. Sarrion, P. Lopez-Cornejo, C. Carmona, F. S. Burgos and M. Lopez-Lopez, RSC Adv., 2015, 5, 46485 RSC; (r) E. Carbonell, E. Delgado-Pinar, J. Pitarch-Jarque, J. Alarcon and E. García-España, J. Phys. Chem. C, 2013, 117, 14325 CrossRef CAS; (s) M. Vetrichelvan, R. Nagarajan and S. Valiyaveettil, Macromolecules, 2006, 39, 8303 CrossRef CAS.
- (a) J. C. C. Chen and I. J. B. Lin, J. Am. Chem. Soc., 2000, 839 Search PubMed; (b) B. Liu, Q. Q. Xia and W. Z. Chen, Angew. Chem., Int. Ed., 2009, 48, 5513 CrossRef CAS PubMed; (c) F. E. Hahn, M. C. Jahnke and T. Pape, Organometallics, 2007, 26, 150 CrossRef CAS; (d) J. C. Garrison and W. J. Youngs, Chem. Rev., 2005, 105, 3978 CrossRef CAS PubMed; (e) D. J. Nielsen, K. J. Cavell, B. W. Skelton and A. H. White, Inorg. Chim. Acta, 2002, 327, 116 CrossRef CAS; (f) M. Alcarazo, S. J. Roseblade, A. R. Cowley, R. Fernandez, J. M. Brown and J. M. Lassaletta, J. Am. Chem. Soc., 2005, 127, 3290 CrossRef CAS PubMed; (g) Q. Li, X. Li, J. Yang, H. B. Song and L. F. Tang, Polyhedron, 2013, 59, 29 CrossRef CAS; (h) J. H. Chen, X. Q. Zhang, Q. Feng and M. M. Luo, J. Organomet. Chem., 2006, 691, 470 CrossRef CAS; (i) F. E. Hahn and M. C. Jahnke, Angew. Chem., Int. Ed., 2008, 47, 3122 CrossRef CAS PubMed.
- (a) A. A. D. Tulloch, A. A. Danopoulos, S. Winston, S. Kleinhenz and G. Eastham, J. Chem. Soc., Dalton Trans., 2000, 4499 RSC; (b) X. Q. Zhang, Y. P. Qiu, B. Rao and M. M. Luo, J. Organomet. Chem., 2009, 28, 3093 CrossRef CAS; (c) J. W. Wang, Q. S. Li, F. B. Xu, H. B. Song and Z. Z. Zhang, Eur. J. Org. Chem., 2006, 1310 CrossRef CAS; (d) Q. Li, Y. F. Xie, B. C. Sun, J. Yang, H. B. Song and L. F. Tang, Organometallics, 2013, 745, 106 CrossRef; (e) B. Liu, C. Y. Chen, Y. J. Zhang, X. L. Liu and W. Z. Chen, Organometallics, 2013, 32, 5451 CrossRef CAS.
- (a) I. J. B. Lin and C. S. Vasam, Coord. Chem. Rev., 2007, 251, 642 CrossRef CAS; (b) D. J. Nielsen, K. J. Cavell, B. W. Sketon and A. H. White, Organometallics, 2006, 25, 4850 CrossRef CAS; (c) Q. X. Liu, Z. Q. Yao, X. J. Zhao, A. H. Chen, X. Q. Yang, S. W. Liu and X. G. Wang, Organometallics, 2011, 30, 3732 CrossRef CAS.
- A. L. Pickering, G. Seeber, D. L. Long and L. Cronin, CrystEngComm, 2005, 7, 504 RSC.
- A. Caballero, R. Martınez, V. Lloveras, I. Ratera, J. Vidal-Gancedo, K. Wurst, A. Tarraga, P. Molina and J. Veciana, J. Am. Chem. Soc., 2005, 127, 15666 CrossRef CAS PubMed.
- D. B. Qin, F. B. Xu, X. J. Wan, Y. J. Zhao and Z. Z. Zhang, Tetrahedron Lett., 2006, 47, 5641 CrossRef CAS.
- (a) J. Polster and H. Lachmann, Spectrometric Titrations, VCH, Weinheim, 1989, p. 256 Search PubMed; (b) J. Wang, S. G. Bodige, W. H. Watson and C. D. Gutsche, J. Org. Chem., 2000, 65, 8260 CrossRef CAS PubMed.
- G. Shyamaprosad, H. Anita, C. Rinku and K. F. Hoong, Org. Lett., 2009, 11, 4350 CrossRef PubMed.
- (a) A. Easwaran, A. Ayyappanpillai and D. Jorg, J. Am. Chem. Soc., 2005, 127, 3156 CrossRef PubMed; (b) G. Shyamaprosad, H. Anita, C. Rinku and K. F. Hoong, Org. Lett., 2009, 11, 4350 CrossRef PubMed.
- (a) A. S. Polo, M. K. Itokazu, K. M. Frin, A. O. T. Patrocínio and N. Y. M. Iha, Coord. Chem. Rev., 2006, 250, 1669 CrossRef CAS; (b) M. R. Gonçalves and K. P. M. Frin, Polyhedron, 2015, 97, 112 CrossRef.
- SMART 5.0 and SAINT 4.0 for Windows NT, Area Detector Control and Integration Software, Bruker Analytical X-Ray Systems, Inc., Madison, WI, USA, 1998 Search PubMed.
- G. M. Sheldrick, SADABS, Program for Empirical Absorption Correction of Area Detector Data, Univ. of Göttingen, Germany, 1996 Search PubMed.
- G. M. Sheldrick, SHELXTL 5.10 for Windows NT, Structure Determination Software, Brucker Analytical X-Ray Systerms, Inc., Madison, WI, USA, 1997 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Text, tables, figures, and CIF files giving crystallographic data and the crystal packings for LH2·(PF6)2 and complex 1, and 1H NMR and 13C NMR spectra for the precursor and complex. CCDC 853182. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c5ra23814e |
| This journal is © The Royal Society of Chemistry 2016 |