Surface co-modification of TiO2 with N doping and Ag loading for enhanced visible-light photoactivity
Mingxuan Sun†
*,
Yalin Fang†,
Shanfu Sun and
Ying Wang
School of Materials Engineering, Shanghai University of Engineering Science, Shanghai 201620, China. E-mail: mingxuansun@sues.edu.cn; smxalan@163.com; Fax: +86 21 67791201; Tel: +86 21 67791474
First published on 14th January 2016
Abstract
A composite of N-doped TiO2 with Ag loading (Ag/N-TiO2) was successfully synthesized by a facile in situ calcination process using titanium nitride (TiN) and silver nitrate (AgNO3) as the starting materials. The effects of Ag loading and N doping on the structure and properties of TiO2 were characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM), X-ray photoelectron spectroscopy (XPS), and diffuse reflectance spectroscopy (UV-visible DRS). XPS results indicated that the loading content of Ag in Ag/N-TiO2 nanomaterials can be adjusted by varying the amount of AgNO3 in the starting materials. There is an enhanced light absorption and a red shift of the optical edge for the resulting Ag/N-TiO2 compared with bare TiO2 and N-doped TiO2. The photocatalytic activities of the samples were evaluated for the degradation of methylene blue in aqueous solutions under visible light (λ > 420 nm). The superior visible light photodegradation of Ag/N-TiO2 composites was confirmed compared to N-doped TiO2 nanoparticles. In addition, an enhanced visible light photoelectrochemical performance was demonstrated. The photocurrent and photovoltage of Ag/N-TiO2 were about 1.5 and 1.4 times larger than those of N-TiO2, respectively. The technique proposed in this paper can also be applied to fabricate other N-TiO2 based composites.
1. Introduction
Titanium dioxide (TiO2), as one of the most promising semiconductor materials, has attracted tremendous research interest as regards clean energy and environmental remediation due to its low cost, high efficiency, photostability, and no toxicity.1–4 Unfortunately, the wide band gap (3.0–3.2 eV) and the rapid recombination of photogenerated electron–hole pairs of TiO2, as the main bottle neck, restrict its utilization in solar energy applications. Thus, a lot of strategies have been explored to overcome these limitations, such as doping of TiO2 with metals and nonmetals or coupling with other semiconductors with narrow band gap.5–11Among the nonmetal elements, nitrogen, as an effective doping element, has been extensively studied. Numerous researches have demonstrated the high visible light activity of TiO2 doped with nitrogen.12,13 Generally, the doping of nitrogen atoms results in a decrease of the band gap energy of TiO2 by mixing the N 2p with O 2p states and higher separation rate of photogenerated hole–electron pairs.14,15 Li et al. prepared N-doped TiO2 photocatalysts by a sol–gel method using ethylenediamine as the nitrogen source, and demonstrated the improved hydrogen production rate and methyl orange degradation performance over N-doped TiO2 compared with TiO2.16 Also, several other investigations have also indicated that N doping can efficiently yield a narrow band gap and improve the photocatalytic activity of TiO2.17–19 Recently, further modifications of N-doped TiO2 with metal ions, metal oxides and other nonmetal elements have been reported to be an efficient strategy for the enhancement of visible light absorption and photocatalytic activity compared to N-doped TiO2.20–23
Decorating TiO2 with a noble metal, such as Au, Ag, Pt, and Pd, is also a good alternative to enhance the photoactivity of TiO2.24–27 Among them, Ag is the most suitable candidate for industrial applications due to its low cost and easy preparation. The deposition of Ag nanoparticles onto the surface of TiO2 can effectively increase the photocatalytic activity by accelerating charge separation and extending the absorption edge to the visible light region.28–30 Tseng et al. prepared mesoporous TiO2–Ag composite particles by a two-step chemical process and found that the composite had excellent photocatalytic activity.31 Bensouici et al. prepared Ag-doped TiO2 nanostructure thin films by a simple sol–gel route. The as-prepared sample exhibited remarkable visible light-driven photocatalytic activity in the photodegradation of Rhodamine B.32 Jaafar et al. successfully synthesized Ag–TiO2 by an in situ electrochemical method. The as-prepared sample of 5 wt% Ag–TiO2 exhibited the highest photodegradation activity of 2-chlorophenol solution which was related to the greatest number of surface oxygen vacancies.25
Based on the above analysis, the combination of these two effects (N doping and Ag loading) is expected to further improve the photoactivity of TiO2 by multiple synergistic effects. To date, some researchers have reported that the co-modification of TiO2 with N doping and Ag loading could further improve the utilization ratio of visible light.33,34 Zhou et al. synthesized N-doped TiO2/Ag composites by a facile modified sol–gel approach and demonstrated their excellent photocatalytic activity and stability under visible light. It is observed that about 97% of a methylene blue (MB) solution can be degraded using N-doped TiO2/Ag, while only about 80%, 79% and 57% of the solution can be degraded using Ag–TiO2, N-TiO2 and TiO2, respectively, after visible light irradiation for 3 h.35 Dhabbe et al. prepared Ag-loaded N-doped TiO2 nanocomposite by a microwave-assisted and impregnation-reduction method. The photocatalytic activity of the catalyst was tested under both UV and sunlight. A 40% enhancement in the photodegradation efficiency was achieved by Ag loading compared with N-doped TiO2 under sunlight.36 Gao et al. introduced Ag into N-doped TiO2 via a hydrothermal procedure. The dependence of photocatalytic activity on Ag content was investigated through degradation of Rhodamine B. It is reported that a photocatalyst of N-TiO2 modified with Ag is widely utilized to prevent the recombination of photogenerated electron–hole pairs and improve the charge transfer efficiency.37 However, these methods also involved special equipment or complex preparation processes. It is still necessary to develop more efficient approaches for the synthesis of Ag-modified N-doped TiO2.
In this study, Ag/N-TiO2 nanoparticles were successfully fabricated via an in situ calcining procedure. TiN powder and AgNO3 were used as the only reactants involved in the preparation process. Nitrogen atoms originating from the decomposition of TiN were doped into the lattice of TiO2. Simultaneously, AgNO3 was transformed to Ag and deposited on the surface of N-TiO2. The as-prepared samples exhibit excellent photoelectrochemical performance and photodegradation activity under visible light irradiation. The improvement of the photoactivity of Ag/N-TiO2 composites could be attributed to the enhanced light absorption capacity and accelerated separation rate of photogenerated electron–hole pairs due to the synergistic effect of N element doping and Ag nanoparticle loading.
2. Experimental: materials and methods
2.1 Preparation of Ag/N-TiO2
Silver nitrate (AgNO3, 99.8%) was purchased from Sinopharm Chemical Reagent Company. Titanium nitride (TiN, 99.9%, ∼40 nm) was obtained from Shanghai ST-NANO Company. Deionized water was used in all of the experiments.Ag/N-TiO2 composites were obtained by a simple in situ calcination process. Briefly, 0.1 g of TiN was dispersed in 10 mL of silver nitrate solution (0.1 M) and ultrasonically vibrated for 2 h. Then, the precipitates were washed with deionized water three times and dried at 60 °C for 12 h, followed by annealing at 500 °C for 2 h in air atmosphere. Thus, Ag/N-TiO2 nanocomposites were obtained with a light gray color. N-TiO2 nanoparticles were synthesized by annealing TiN at 500 °C for 2 h in air atmosphere for comparison. The loading amount of Ag was adjusted by controlling the concentration of AgNO3 solution (0.05–0.15 M) in the process of preparing the Ag/N-TiO2 nanocomposites. The as-prepared samples were denoted as Ag/N-TiO2 (x), where x represents the concentration of AgNO3 solution.
2.2 Characterizations
The phase composition and crystal structure of as-prepared samples were characterized by X-ray diffraction (XRD) (Panalytical X'Pert powder, Holland) using Cu Kα radiation (λ = 0.1542 nm) at 40 kV and 40 mA. The morphologies and microstructures of the samples were investigated by field emission scanning electron microscopy (FE-SEM; Philips XL30) and transmission electron microscopy (TEM; JEOL JEM-2011, Japan). X-ray photoelectron spectroscopy (XPS) was carried out using a RBD upgraded PHI-5000 C ESCA system (PerkinElmer) with Al/Mg Kα as the radiation source. The binding energies were calibrated based on the C 1s peak at 284.6 eV as a standard. The UV-visible diffuse reflectance spectra (DRS) were recorded using a (Shimadzu UV-3600, Japan) UV-visible spectrophotometer in the region of 200 to 800 nm.2.3 Photoelectrochemical and photocatalytic measurements
Photoelectrochemical performance of the as-prepared samples was measured using a three-electrode system with a CHI660E electrochemical workstation (Shanghai Chenhua instruments, China). The FTO electrode deposited with the as-prepared sample as working electrode, saturated calomel electrode (SCE) as the reference electrode and Pt foil as the counter electrode were positioned in a supporting electrolyte of 0.5 M Na2SO4 aqueous solution for measurements. A 500 W xenon lamp (CHF-XM35, Beijing) combined with an optical filter was used to provide visible light (wavelengths below 420 nm, light intensity of 120 mW cm−2). The electrochemical impedance spectra (EIS) of as-prepared samples were obtained in the range 100 kHz to 100 mHz without light illumination.The visible light photocatalytic activity of as-prepared samples was investigated using the degradation of MB solution under visible light irradiation. Typically, the photocatalyst (7.5 mg) was added into MB solution (15 mL, 5 mg L−1) and stirred for 2 h inside a dark box to reach the adsorption/desorption equilibrium of MB. Then, the photodegradation test was performed under visible light with an intensity of 120.0 mW cm−2. The UV-visible adsorption spectrum of the remaining MB solution was recorded at 10 min intervals for 50 min. In addition, the photoelectrochemical and photodegradation measurements of bare TiO2 (P25, Degussa) were performed under the same conditions for comparison.
3. Results and discussion
The preparation procedures of Ag/N-TiO2 nanocomposites are presented schematically in Scheme 1. Ag nanoparticles are dispersed on the surfaces of N-TiO2 via a calcining process. The three inset photographs on the right-hand side present the color change of TiN, N-TiO2 and Ag/N-TiO2 composites, which is visual evidence for the successful synthesis of Ag/N-TiO2 composites. The raw material TiN exhibits a black color, whereas the obtained N-TiO2 shows a light yellow color, which could be ascribed to the presence of nitrogen element in TiO2. For Ag/N-TiO2, a light grey color is observed, indicating Ag nanoparticles are successfully loaded on the surface of N-TiO2 nanoparticles.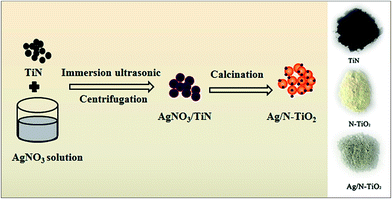 | ||
| Scheme 1 Schematic illustration of the preparation of Ag/N-TiO2. The three inset photographs on the right-hand side present the color evolution of samples TiN, N-TiO2 and Ag/N-TiO2. | ||
The XRD patterns of TiN, N-TiO2 and Ag/N-TiO2 are shown in Fig. 1. A series of characteristic peaks were observed for the raw material of TiN powder at 36.70°, 42.72°, 61.91°, 74.08°, and 78.13°, which were attributed to (111), (200), (220), (311) and (222) crystal planes of cubic phase TiN (JCPDS card no. 03-065-0715), respectively. The XRD patterns of N-TiO2 and Ag/N-TiO2 clearly exhibit the presence of anatase and rutile phases of TiO2 polymorphs. The diffraction peaks located at 25.31°, 37.72°, 47.08°, 55.11°, 62.76°, and 75.16° corresponding to (101), (004), (200), (211), (204), and (215) planes of anatase structure of TiO2 are in agreement with JCPDS card no. 00-004-0477. The rutile peaks at 27.45°, 36.09°, 41.27°, 53.81°, 56.66°, and 70.41° can be indexed to the (110), (101), (111), (211), (220), and (112) planes of rutile TiO2 (JCPDS card no. 01-076-1979), respectively. Notably, the diffraction peak for the (101) crystal planes of N-TiO2 nanoparticles (2θ = 25.31°) is at slightly lower angle compared to that of pristine TiO2 (P25) (2θ = 25.35°), which indicates that nitrogen was doped into the TiO2 lattice and caused lattice distortion.38,39 The results suggest that the N-doped TiO2 was successfully prepared by annealing TiN at 500 °C for 2 h. However, no obvious peak for Ag was detected for the Ag/N-TiO2 composites. This is possibly because of the small amount of Ag loading on the N-TiO2 surface. Another reason is that it might be masked by the strong diffraction peaks of TiO2. Compared with the XRD pattern of N-TiO2, the peak positions of Ag/N-TiO2 composites are only slightly changed to some extent, suggesting that Ag ions do not incorporate into the TiO2 lattice. The results indicate Ag nanoparticles are loaded on the surface of TiO2. The inset exhibits the deconvolution of detailed XRD pattern of Ag/N-TiO2 at 35–40°, which has four fitting curves centered at 36.09°, 36.91°, 37.72°, and 38.10° corresponding to rutile TiO2 (101), anatase TiO2 (103) and (004), and Ag (111), respectively. The results imply the co-existence of rutile TiO2, anatase TiO2 and Ag in the Ag/N-TiO2 composites. The existence of Ag in the composites could be further confirmed by XPS and TEM results, as described later.
 | ||
| Fig. 1 XRD patterns of TiN, N-TiO2 and Ag/N-TiO2; inset is the deconvolution of detailed XRD pattern of Ag/N-TiO2 at 35–40° (A: anatase TiO2; R: rutile TiO2). | ||
Fig. 2 presents the SEM images of the resulting N-TiO2 (Fig. 2A) and Ag/N-TiO2 composites (Fig. 2B). Spherical nanoparticles with grain sizes of about 41.0 nm and 63.5 nm are observed for N-TiO2 and Ag/N-TiO2, respectively. The morphology of N-TiO2 and Ag/N-TiO2 shows slight changes, which is attributed to the small particle size of Ag nanoparticles. Compared with that of N-TiO2, the increased particle size of Ag/N-TiO2 composites indicates that the coupling of Ag could bring about the agglomeration of composite particles. The morphology of the Ag/N-TiO2 composite was also characterized by TEM (Fig. 2C and D). As shown in Fig. 2C, a ternary composites with particle structure is clearly observed, which is consistent with the SEM results. Fig. 2D presents a typical HR-TEM image of an Ag/N-TiO2 sample. The HR-TEM image shows clear lattice fringes of TiO2 and Ag nanoparticles. The lattice fringe of 0.324 nm is assigned to (110) crystal plane of rutile TiO2, whereas that of 0.35 nm corresponds to (101) crystal plane of anatase TiO2. The fringes with a spacing of 0.24 nm match well with (111) crystal plane of Ag. The results further confirm that Ag/N-TiO2 composites were successfully fabricated.
The surface composition and chemical states of Ag/N-TiO2 were further determined by XPS analysis, and the corresponding results are shown in Fig. 3A–D. Fig. 3A illustrates the XPS spectrum of Ag/N-TiO2 composites, which indicates the presence of C, Ti, O, N, and Ag elements. No trace of any impurity is observed, except for C 1s. The appearance of the C peak mainly originates from air pollution. High-resolution spectra of Ti, N and Ag species are shown in Fig. 3B–D, respectively. Two peaks centered at 458.3 eV and 464.0 eV are associated with Ti 2p3/2 and Ti 2p1/2 of Ti(IV), respectively, which confirmed the formation of TiO2. Moreover, both of these binding energy values reduce by 0.5 eV in comparison with the bare TiO2.40 The decrease of binding energy can be ascribed to the lower electronegativity of nitrogen compared to oxygen, which can cause different interactions between Ti and other anions.41,42 To some extent, the phenomenon suggests that oxygen in the TiO2 lattice is partly substituted by nitrogen. The peak for N 1s at 399.6 eV is observed in the N 1s XPS spectrum (Fig. 3C), which is related to the formation of O–Ti–N linkages.43,44 Thus, the substitution of O by N in the TiO2 lattice is achieved, which is responsible for the visible light absorption.45 Fig. 3D displays two characteristic peaks with binding energies located at 368.4 eV and 374.4 eV, which can be identified with Ag 3d5/2 and 3d3/2, respectively. The results indicate that Ag loading on the TiO2 surface mainly exists in the form of metallic nature.46,47 Together, the XRD, TEM, and XPS results confirmed that the composite of Ag/N-TiO2 was successfully formed via the simple in situ calcination process. Furthermore, energy dispersive spectroscopy (EDS) was also applied for elemental analysis. The atomic percentages of Ag/Ti in the samples increased from 0.35 at% to 1.9 at% as the concentration of AgNO3 in the solution ranged from 0.05 M to 0.15 M. Obviously, the content of Ag in the Ag/N-TiO2 nanocomposites is proportional to the amount of AgNO3 in the starting materials.
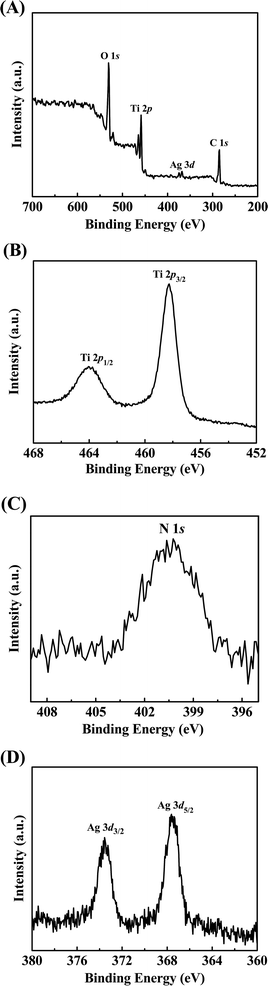 | ||
| Fig. 3 XPS spectrum of Ag/N-TiO2 (A) and the high-resolution spectra of Ti 2p (B), N 1s (C) and Ag 3d (D). | ||
The DRS of as-prepared N-TiO2 and Ag/N-TiO2 (0.10) samples along with the spectrum of bare TiO2 (P25) for comparison are shown in Fig. 4. As shown in Fig. 4A, it can be observed that the absorption edges of N-TiO2 and Ag/N-TiO2 are located at 409 nm and 440 nm, respectively. The N-TiO2 and Ag/N-TiO2 samples exhibited a wider range of absorption of visible light than bare TiO2 (380 nm). Meanwhile, it is clearly seen that the absorption intensity ranging from 400 nm to 600 nm for Ag/N-TiO2 is higher than that for TiO2 and N-TiO2. The optical band-gap energy (Eg) of the samples can be roughly estimated from hν vs. (αhν)1/2 plots as shown in Fig. 4B.48 The band gaps of bare TiO2, N-TiO2, and Ag/N-TiO2 are found to be 3.1 eV, 2.85 eV, and 2.65 eV, respectively. Obviously, Ag/N-TiO2 exhibited the longest absorption edge and minimum band-gap energy, which is beneficial to the improvement of the photocatalytic and photoelectrochemical efficiency under visible light.
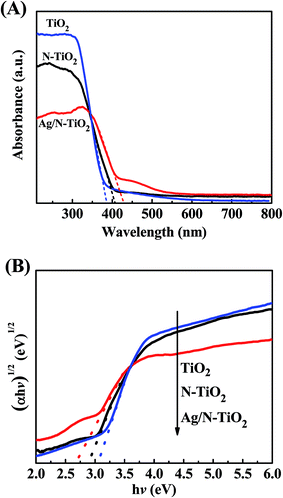 | ||
| Fig. 4 (A) The UV-visible diffuse reflectance absorption spectra of bare TiO2, N-TiO2, Ag/N-TiO2 (0.10); (B) the corresponding hν vs. (αhν)1/2 curves of as-prepared samples. | ||
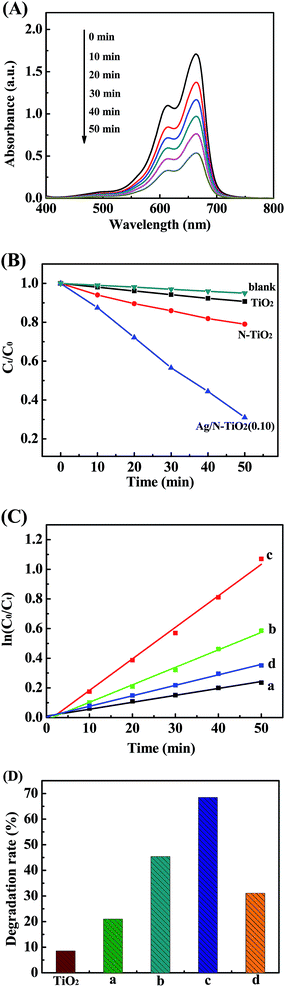
The photocatalytic degradation of MB dye molecules under visible light was used to evaluate the photocatalytic performance of as-prepared samples (Fig. 5). Fig. 5A shows representative variations in the characteristic absorption of MB (λmax = 664 nm) under visible light irradiation over Ag/N-TiO2 (0.10) photocatalyst. The characteristic absorption intensity of MB weakens as the irradiation time increases, indicating that MB gradually decomposed. Fig. 5B shows illustrative photodegradation profiles of MB solution at each irradiated time interval over bare TiO2 (P25), N-TiO2, and Ag/N-TiO2 under visible light irradiation. C0 and Ct are the initial concentration of MB and the remaining concentration of MB after each irradiation time, respectively. The self-degradation of MB can be negligible in the absence of photocatalyst. An enhancement of photodegradation efficiency over Ag/N-TiO2 is observed compared with that over bare TiO2 and N-TiO2 nanoparticles. The effects of amount of Ag on the photocatalytic activity were determined from the results shown in Fig. 5C. The reaction rate constants of MB degradation are 0.0046, 0.0118, 0.0213, and 0.0071 min−1 for N-TiO2, Ag/N-TiO2 (0.05), Ag/N-TiO2 (0.10) and Ag/N-TiO2 (0.15), respectively. It was found that the photocatalytic activity of N-TiO2 can be enhanced via loading an appropriate Ag content. The photocatalyst Ag/TiO2 (0.10) achieves the highest efficiency of MB photodegradation. Excess amount of Ag is detrimental to the photocatalytic process. As shown in Fig. 5D, the degradation rates for the as-prepared samples are about 9%, 21%, 45%, 68%, and 31% for TiO2, N-TiO2, Ag/N-TiO2 (0.05), Ag/N-TiO2 (0.10) and Ag/N-TiO2 (0.15) after 50 min illumination, respectively. The highest photocatalytic performance is observed for Ag/N-TiO2 (0.10), being about 6.5 times and 3 times larger than that of bare TiO2 and N-TiO2 nanoparticles, respectively. The improved photocatalytic performance further demonstrates the multiple synergetic effects of N and Ag modification on TiO2.
The transient photocurrent and open circuit potential for the samples were measured under visible light irradiation via several on–off cycles (Fig. 6). Fig. 6A displays the transient photocurrent density of TiO2, N-TiO2 and Ag/N-TiO2 (0.10). It can be seen that photocurrent density is almost zero in the dark for all the samples. Under visible light irradiation, the photocurrent densities of bare TiO2, N-TiO2 and Ag/N-TiO2 were 0.010, 0.018 and 0.026 mA cm−2, respectively. The photocurrent density increased about 1.5 times with the modification of Ag on the surface of N-TiO2 nanoparticles. A higher transient photocurrent density was obtained for Ag/N-TiO2 electrode compared to N-TiO2 electrode under visible light. The remarkably enhanced photocurrent may be ascribed to faster electron transport and more efficient separation of the photogenerated holes and electrons in Ag. Fig. 6B shows the transient open-circuit potential of the bare TiO2, N-TiO2 and Ag/N-TiO2 electrodes under the intermittent illumination of visible light. It is noticed that the Ag/N-TiO2 electrode exhibited a photopotential of −0.485 V, which was more negative than that of the bare TiO2 electrode (−0.158 V) and the N-TiO2 electrode (−0.352 V). The photovoltage further increased about 1.4 times for Ag/N-TiO2 compared with N-TiO2, thus demonstrating the synergy effects of Ag and N on the photoelectrochemical properties of TiO2. The results are in accord with the result of DRS.
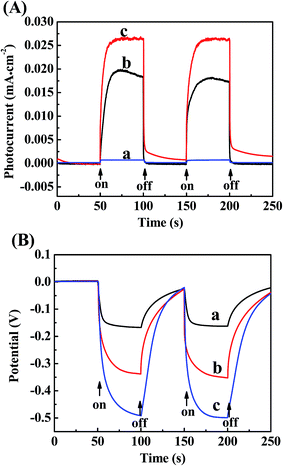 | ||
| Fig. 6 Photocurrent response (A) and open-circuit potential (B) of (a) bare TiO2, (b) N-TiO2 and (c) Ag/N-TiO2 (0.10) in 0.5 M Na2SO4 solution under visible light irradiation. | ||
To confirm the enhancement of charge transfer and separation at the surface of the Ag/N-TiO2 electrode, we investigated the EIS of as-prepared samples, and typical EIS Nyquist plots of the as-prepared samples are illustrated in Fig. 7. It is observed that the semicircle diameter in the plot of Ag/N-TiO2 is largely reduced compared to the bare TiO2 and N-TiO2, indicating that the interface resistance and the charge transfer resistance decrease with the presence of Ag in N-TiO2. Thus, the charge transfer across the interface between the semiconductor and solution becomes easy and the value of the photocurrent can be improved.49 It can be concluded from the EIS results that Ag was helpful for the charge transfer. This is consistent with the photoelectrochemical and photodegradation results.
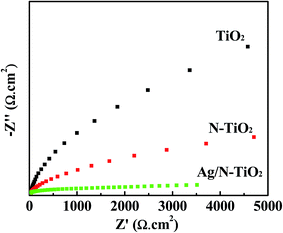 | ||
| Fig. 7 Nyquist plots of bare TiO2, N-TiO2 and Ag/N-TiO2 (0.10) electrodes in 0.5 M Na2SO4 under dark condition. | ||
Scheme 2 shows the mechanism of the photocatalytic degradation of MB over Ag/N-TiO2 nanomaterials under visible light irradiation. The enhanced photoactivity of Ag/N-TiO2 composites under visible light irradiation can be attributed to the synergistic effect of nitrogen element and Ag nanoparticles. On the one hand, the substitution of N element in the lattice of TiO2 is a more effective approach to extend the absorption edge to the visible light region due to the induced N 2p state.50,51 Meanwhile, some interstitial defects, such as substitutional N or O vacancies, also play a vital positive role in the visible response of TiO2.52 On the other hand, Ag nanoparticles in Ag/N-TiO2 composites can serve as efficient electron traps to inhibit electron–hole recombination.53 Under visible light irradiation, electrons are excited from the N impurity energy level to the conduction band of TiO2. Subsequently, they are captured by deposited Ag and then transferred rapidly to the adsorbed O2 on the surface of TiO2 to form superoxide anion radicals (˙O2−), which can further initiate the degradation reaction of MB. Simultaneously, the photogenerated holes which are left in the valence band react with OH− to form highly active oxygen species ˙OH. The generated ˙OH and ˙O2− are responsible for the degradation of MB. Overall, both Ag loading and N doping are responsible for the enhancement of photocatalytic activity of TiO2.
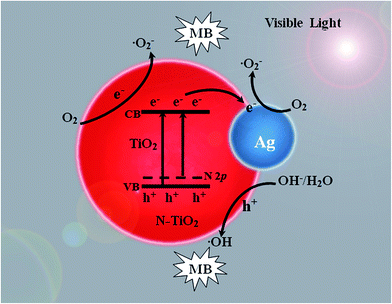 | ||
| Scheme 2 Mechanism of the photocatalytic degradation over Ag/N-TiO2 nanomaterials under visible light irradiation. | ||
4. Conclusions
In the study reported in this paper, Ag/N-TiO2 composites were successfully prepared by an ingenious in situ calcination process. The loading amount of Ag can be adjusted by controlling the concentration of AgNO3 solution in the process of preparing the Ag/N-TiO2 nanocomposites. About 1.5 and 1.4 times increase in the photocurrent density and open circuit potential were observed for the Ag/N-TiO2 nanoparticles compared with N-TiO2 under visible light irradiation. Also, the highest photocatalytic performance was confirmed over Ag/N-TiO2 composites, about 6.5 and 3 times larger than that over bare TiO2 and N-TiO2, respectively. The enhanced photoactivity of Ag/N-TiO2 is associated with an enhanced visible light response and high electron–hole separation efficiency, due to the synergetic interactions between TiO2, loaded Ag and doped N. The excellent photocatalytic performance of Ag/N-TiO2 offers promising potential for applications in solar energy conversion and environmental remediation.Acknowledgements
This work was financially supported by Innovation Program of Shanghai Municipal Education Commission (15ZZ092), Training Program for Young Teachers in Shanghai Colleges and Universities (ZZgcd14010), Startup Foundation of Shanghai University of Engineering Science (no. XiaoQi 2014-22), and Shanghai University of Engineering Science Innovation Fund (15KY0516). We thank Minjie Yan and Fang Li (School of Materials Engineering, Shanghai University of Engineering Science) for EDS measurements. The authors also appreciate the referee's very valuable comments, which have greatly improved the quality of the paper.References
- W. H. Chiu, K. Lee and W. Hsieh, J. Power Sources, 2011, 196, 3683–3687 CrossRef CAS.
- Y. H. Zhang, Z. R. Tang, X. Z. Fu and Y. J. Xu, ACS Nano, 2010, 4, 7303–7314 CrossRef CAS PubMed.
- G. M. Wang, H. Y. Wang, Y. C. Ling, Y. C. Tang, X. Y. Yang, R. C. Fitzmorris, C. C. Wang, J. Z. Zhang and Y. Li, Nano Lett., 2011, 11, 3026–3033 CrossRef CAS PubMed.
- D. H. Wang, D. W. Choi, J. Li, Z. G. Yang, Z. M. Nie, R. Kou, D. H. Hu, C. M. Wang, L. V. Saraf, J. G. Zhang, I. A. Aksay and J. Liu, ACS Nano, 2009, 3, 907–914 CrossRef CAS PubMed.
- G. H. Liu, M. Zhang, D. Z. Zhang, J. R. Zhou, F. X. Meng and S. P. Ruan, J. Alloys Compd., 2014, 616, 155–158 CrossRef CAS.
- M. X. Sun and X. L. Cui, Electrochem. Commun., 2012, 20, 133–136 CrossRef CAS.
- M. X. Sun, X. Y. Zhang, J. Li, X. L. Cui, D. L. Sun and Y. H. Lin, Electrochem. Commun., 2012, 16, 26–29 CrossRef CAS.
- R. Klaysri, S. Wichaidit, T. Tubchareon, S. Nokjan, S. Piticharoenphun, O. Mekasuwandumrong and P. Praserthdam, Ceram. Int., 2015, 41, 11409–11417 CrossRef CAS.
- L. G. Devi and R. Kavitha, RSC Adv., 2014, 4, 28265–28299 RSC.
- J. Y. Lei, Y. Chen, F. Shen, L. Z. Wang, Y. D. Liu and J. L. Zhang, J. Alloys Compd., 2015, 631, 328–334 CrossRef CAS.
- M. Y. Wang, L. Sun, Z. Q. Lin, J. H. Cai, K. P. Xie and C. Q. Lin, Energy Environ. Sci., 2013, 6, 1211–1220 CAS.
- F. Dong, W. R. Zhao, Z. B. Wu and S. Guo, J. Hazard. Mater., 2009, 162, 763–770 CrossRef CAS PubMed.
- V. Caratto, L. Setti, S. Campodonico, M. M. Carnasciali, R. Botter and M. Ferretti, J. Sol-Gel Sci. Technol., 2012, 63, 16–22 CrossRef CAS.
- Z. F. Jiang, X. M. Lv, D. L. Jiang, J. M. Xie and D. J. Mao, J. Mater. Chem. A, 2013, 1, 14963–14972 CAS.
- R. Asahi, T. Morikawa, T. Ohwaki, K. Aoki and Y. Taga, Science, 2001, 293, 269–271 CrossRef CAS PubMed.
- H. Li, Y. B. Hao, H. Q. Lu, L. P. Liang, Y. Y. Wang, J. H. Qiu, X. C. Shi, Y. Wang and J. F. Yao, Appl. Surf. Sci., 2015, 344, 112–118 CrossRef CAS.
- Z. Jiang, L. Kong, F. S. Alenazey, Y. D. Qian, L. France, T. C. Xiao and P. P. Edwards, Nanoscale, 2013, 5, 5396–5402 RSC.
- G. H. Tian, Y. J. Chen, K. Pan, D. J. Wang, W. Zhou, Z. Y. Ren and H. G. Fu, Appl. Surf. Sci., 2010, 256, 3740–3745 CrossRef CAS.
- W. Zhou, C. L. Yu, Q. Z. Fan, L. F. Wei, J. C. Chen and J. C. Yu, Chin. J. Catal., 2013, 34, 1250–1255 CrossRef CAS.
- Y. Cong, B. Z. Tian and J. L. Zhang, Appl. Catal., B, 2011, 101, 376–381 CrossRef CAS.
- Y. F. Ma, J. L. Zhang, B. Z. Tian, F. Chen and L. Z. Wang, J. Hazard. Mater., 2010, 182, 386–393 CrossRef CAS PubMed.
- J. L. Zhang, Y. M. Wu, M. Y. Xing, S. A. K. Leghari and S. Sajjad, Energy Environ. Sci., 2010, 3, 715–726 CAS.
- Y. M. Wu, H. B. Liu, J. L. Zhang and F. Chen, J. Phys. Chem. C, 2009, 113, 14689–14695 CAS.
- R. Ravindranath, P. Roy, A. Periasamy and H. –T. Chang, RSC Adv., 2014, 4, 57290–57296 RSC.
- N. F. Jaafar, A. A. Jalil, S. Triwahyono, J. Efendi, R. R. Mukti, R. Jusoh, N. W. C. Jusoh, A. H. Karim, N. F. M. Salleh and V. Suendo, Appl. Surf. Sci., 2015, 338, 75–84 CrossRef CAS.
- A. Zielińska-Jurek and J. Hupka, Catal. Today, 2014, 230, 181–187 CrossRef.
- A. Lehoux, L. Ramos, P. Beaunier, D. B. Uribe, P. Dieudonné, F. Audonnet, A. Etcheberry, M. José-Yacaman and H. Remita, Adv. Funct. Mater., 2012, 22, 4900–4908 CrossRef CAS.
- X. Li, Z. Zhuang, W. Li and H. Pan, Appl. Catal., A, 2012, 429–430, 31–38 CrossRef CAS.
- C. W. Dunnill, Z. Ansari, A. Kafizas, S. Perni, D. J. Morgan, M. Wilson and I. P. Parkin, J. Mater. Chem., 2011, 21, 11854–11861 RSC.
- C. Liu, D. Yang, Y. Jiao, Y. Tian, Y. Wang and Z. Jiang, ACS Appl. Mater. Interfaces, 2013, 5, 3824–3832 CAS.
- W. J. Tseng, S. M. Kao and J. H. Hsieh, Ceram. Int., 2015, 41, 10494–10500 CrossRef CAS.
- F. Bensouici, T. Souier, A. A. Dakhel, A. Iratni, R. Tala-Ighil and M. Bououdinac, Superlattices Microstruct., 2015, 85, 255–265 CrossRef CAS.
- W. G. Zhang, Y. M. Liu, D. Y. Zhou, J. Wen, W. Liang and F. Q. Yang, RSC Adv., 2015, 5, 57155–57163 RSC.
- Z. F. Jiang, W. Wei, D. J. Mao, C. Chen, Y. F. Shi, X. M. Lv and J. M. Xie, Nanoscale, 2015, 7, 784–797 RSC.
- X. F. Zhou, J. Lu, J. L. Gao, M. F. Xu and Z. S. Wang, Ceram. Int., 2014, 40, 3975–3979 CrossRef CAS.
- D. S. Dhabbe, A. N. Kadam, M. B. Suwarnkar, M. R. Kokate and K. M. Garadkar, J. Mater. Sci.: Mater. Electron., 2014, 25, 3179–3189 CrossRef.
- Y. P. Gao, P. F. Fang, F. T. Chen, Y. Liu, D. H. Wang and Y. Q. Dai, Appl. Surf. Sci., 2013, 265, 796–801 CrossRef CAS.
- M. Miyauchi, M. Takashio and H. Tobimatsu, Langmuir, 2004, 20, 232–236 CrossRef CAS PubMed.
- J. B. Yin and X. P. Zhao, Chem. Mater., 2004, 16, 321–328 CrossRef CAS.
- H. Q. Wang, J. P. Yan, W. F. Chang and Z. M. Zhang, Catal. Commun., 2009, 10, 989–994 CrossRef CAS.
- X. Chen and C. Burda, J. Phys. Chem. B, 2004, 108, 15446–15449 CrossRef CAS.
- Y. Cong, J. Zhang, F. Chen and M. Anpo, J. Phys. Chem. C, 2007, 111, 6976–6982 CAS.
- M. X. Sun, P. Song, J. Li and X. L. Cui, Mater. Res. Bull., 2013, 48, 4271–4276 CrossRef CAS.
- Z. Wu, F. Dong, W. Zhao and S. Guo, J. Hazard. Mater., 2008, 157, 57–63 CrossRef CAS PubMed.
- H. Irie, Y. Watanabe and K. Hashimoto, J. Phys. Chem. B, 2003, 107, 5483–5486 CrossRef CAS.
- J. X. He, P. J. Yang, H. Sato, Y. Umemura and A. Yamagishi, J. Electroanal. Chem., 2004, 566, 227–233 CrossRef CAS.
- F. Zhang, Y. Pi, J. Cui, Y. Yang, X. Zhang and N. Guan, J. Phys. Chem. C, 2007, 111, 3756–3761 CAS.
- T. Tuac, R. Grigorovici and A. Vancu, Phys. Status Solidi B, 1966, 15, 627–637 CrossRef.
- S. Ameen, M. S. Akhtar, Y. S. Kim, O. B. Yang and H. S. Shin, J. Phys. Chem. C, 2010, 114, 4760–4764 CAS.
- C. D. Valentin, G. Pacchioni, A. Selloni, S. Livraghi and E. Giamello, J. Phys. Chem. B, 2005, 109, 11414–11419 CrossRef PubMed.
- R. Nakamura, T. Tanaka and Y. Nakato, J. Phys. Chem. B, 2004, 108, 10617–10620 CrossRef CAS.
- Z. W. Luo, H. Jiang, D. Li, L. Z. Hu, W. H. Geng, P. Wei and P. K. Ouyang, RSC Adv., 2014, 4, 17797–17804 RSC.
- Y. Y. Wen, H. M. Ding and Y. K. Shan, Nanoscale, 2011, 3, 4411–4417 RSC.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2016 |