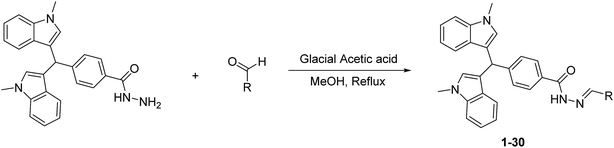
Identification of bisindolylmethane–hydrazone hybrids as novel inhibitors of β-glucuronidase, DFT, and in silico SAR intimations†
Muhammad Taha*ab,
Nor Hadiani Ismailab,
Syahrul Imranab,
El Hassane Anouarc,
Muhammad Alide,
Waqas Jamilf,
Nizam Udding and
Syed Muhammad Kashiff
aAtta-ur-Rahman Institute for Natural Product Discovery, Universiti Teknologi MARA (UiTM), Puncak Alam Campus, 42300, Bandar Puncak Alam, Selangor, Malaysia. E-mail: taha_hej@yahoo.com; muhamm9000@puncakalam.uitm.edu.my; Tel: +60193098141
bFaculty of Applied Science Universiti Teknologi MARA (UiTM), 40450, Shah Alam, Selangor, Malaysia
cChemistry Department, College of Sciences and Humanities, Prince Sattam bin Abdulaziz University, P.O. Box 83, Al-Kharij 11942, Saudi Arabia
dCenter for Advanced Drug Research, COMSATS Institute of Information Technology, University Road, Abbottabad-22060, KPK, Pakistan
eDepartment of Chemistry, COMSATS Institute of Information Technology, University Road, Abbottabad-22060, KPK, Pakistan
fInstitute of Advance Research Studies in Chemical Sciences, University of Sindh Jamshoro, Hyderabad 76080, Pakistan
gBatterje Medical College for Science & Technology, Jeddah-21442, Kingdom of Saudi Arabia
First published on 15th December 2015
Abstract
The present study involves the synthesis of bisindolylmethane–hydrazone hybrids, 1–30, in a three-step reaction sequence, followed by evaluation against β-glucuronidase enzyme. The IC50 values for the potent compounds were in the range from 0.10 to 83.50 μM. Compound, a trihydroxy analog was found to be the most potent derivative, having an IC50 value of 0.10 ± 0.001 μM. Molecular docking revealed that the active compounds could fit perfectly into the binding groove of β-D-glucuronidase. The presence of hydroxyl groups on the aromatic side chain proved to be the single most important factor that contributed toward the inhibitory potential of these compounds. On the other hand, the imino group of the hydrazone linkage displayed interactions with the side chain carboxyl oxygen (Oε2) of Asp207. The high inhibitory potential of these compounds could be associated with these strong hydrogen bonds. Structures of all the synthesized compounds were confirmed using modern spectroscopic methods.
1. Introduction
A number of indole alkaloids are found naturally in marine and terrestrial sources with unique chemical structures and diverse biological potentials such as antibacterial, cytotoxic,1 and protection against DNA damage in human colon cell lines.2 Some commercial drugs contain an indole moiety as either a fundamental unit or as an attached part to invoke specific biological activities.3 Bisindole is a special class of the indole family applauded as an important scaffold in pharmaceutical chemistry.4–9 Bisindole and its derivatives have been widely identified as a pharmacophore that can exhibit antiviral,10 anticancer,11 antitubercular,12 antihypertension,13 free radical induced lipid peroxidation,14 antioxidant,15 Alzheimer disease, and antihyperlipidemic potentials.16 A number of bis(indolyl)methanes were reported to have antioxidant potential,17 anti-inflammatory with analgesic properties, and ulcerogenic activities in mice and rat models.18 Their interesting biological profiles, distinctive chemical structures, and low availability make the bisindole alkaloids attractive targets for use in drug discovery.β-Glucuronides are the final metabolites of hydrophobic xenobiotics,19 and their hydrolysis is catalyzed by the β-glucuronidase enzyme, which is commonly found in animals, plants, and bacteria.20 In animals, low β-glucuronidase activity is maintained through localization in lysosomes.21 However, β-glucuronidase produced by intestinal bacteria hydrolyzes glucuronide to liberate xenobiotics. These xenobiotics exhibit toxicity in the intestine and lower the rate of excretion of xenobiotic by reabsorption. Thus, to decrease toxicity, the inhibition of bacterial β-glucuronidase in the intestine is required to promote the excretion of xenobiotics. A number of β-glucuronidase inhibitors have been isolated from nature and categorized as terpenoids and their glucuronides,22–25 flavonoids and their glucuronides,26,27 and pseudo-sugars that contain nitrogen,28 whereas various synthetic compounds have also been found to be potent β-glucuronidase inhibitors.26
In continuation of our work on the development of new enzyme inhibitors for drug discovery, we produced ongoing work on the synthesis and biological evaluation of potent heterocyclic scaffolds.27–29 Recently, we published bisindolylmethane analogs as a potent inhibitor of β-D-glucuronidase enzyme. This effort resulted in the identification of some novel inhibitors of β-D-glucuronidase enzyme.30 However, these bisindolylmethane derivatives seriously lacked the sites for further modifications for the identification of more potential leads. For a more potent analog identification, we adopted the technique of utilizing hybrid molecules, in which we combined two or more biologically active scaffolds. This methodology proved extremely successful and led us to identifying some distinctly potent inhibitors, such as the bisindolylmethane–hydrazone hybrids 1–30 of the β-D-glucuronidase enzyme in the present study. The synthesized hybrids were evaluated in vitro against β-D-glucuronidase enzyme and an SAR was developed, whereas the mode of binding action was evaluated by molecular modeling and DFT studies.
2. Results and discussion
2.1. Chemistry
Bisindolylmethane ester I was obtained by reacting 1-methyl-1H-indole and methyl 4-formylbenzoate in refluxing acetic acid. Intermediate I was transformed into an advanced hydrazide intermediate II by refluxing it in ethanolic hydrazine hydrate solution (Scheme 1). In the present study, various methods were used (I: NaBrO3/NaHSO3, EAN, and BF3·Et2O) to synthesize the starting material methyl 4-(bis(1-methyl-1H-indol-3-yl)methyl)benzoate (I),31 but in very low yield. When the same reaction was carried out in refluxing acetic acid, we managed to recover up to 93% yield by handling the reaction carefully. The other conversions from ester to hydrazide and hydrazide to hydrazone are well documented, with yields up to 70–85%.The advanced intermediate hydrazide II was then reacted with various aryl aldehydes in the presence of acetic acid to yield the targeted bisindolylmethane–hydrazone hybrids 1–30 (Scheme 2; Table 1) in good to excellent yields. The structures of all the synthesized compounds 1–30 were confirmed using various spectroscopic techniques, such as NMR and EIMS, and were further confirmed through CHN analysis.
| S. no. | R | IC50 (μM ± SEMa) | S. no. | R | IC50 (μM ± SEMa) |
|---|---|---|---|---|---|
| a SEM is the standard error of the mean.b N.A. no activity. | |||||
| 1 | 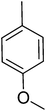 |
83.50 ± 1.70 | 16 | 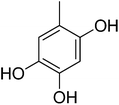 |
0.10 ± 0.001 |
| 2 | 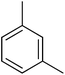 |
N. A.b | 17 | 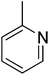 |
18.60 ± 0.30 |
| 3 | 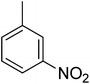 |
73.50 ± 1.40 | 18 | 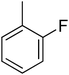 |
9.60 ± 0.16 |
| 4 | 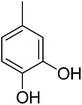 |
0.20 ± 0.01 | 19 | 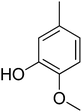 |
32.36 ± 0.64 |
| 5 | 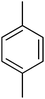 |
51.10 ± 1.29 | 20 | 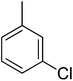 |
16.10 ± 0.29 |
| 6 | 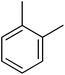 |
23.12 ± 0.45 | 21 | 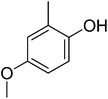 |
11.28 ± 0.23 |
| 7 | 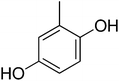 |
3.50 ± 0.05 | 22 | 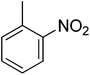 |
13.38 ± 0.22 |
| 8 | 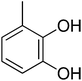 |
0.50 ± 0.3 | 23 | 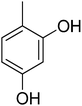 |
6.30 ± 0.10 |
| 9 | 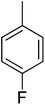 |
3.80 ± 0.08 | 24 | 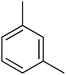 |
N. A.b |
| 10 | 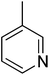 |
88.56 ± 1.88 | 25 | 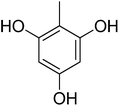 |
2.20 ± 0.06 |
| 11 | 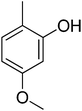 |
13.60 ± 0.26 | 26 | 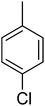 |
26.63 ± 0.38 |
| 12 | 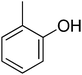 |
12.10 ± 0.24 | 27 | 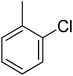 |
15.20 ± 0.28 |
| 13 | 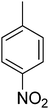 |
N. A.b | 28 | 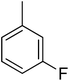 |
26.20 ± 0.39 |
| 14 | 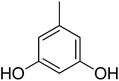 |
22.18 ± 0.45 | 29 | 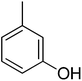 |
16.25 ± 0.29 |
| 15 | 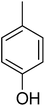 |
15.26 ± 0.26 | 30 | 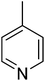 |
N. A.b |
| D-Saccharic acid 1,4-lactone | 48.4 ± 1.25 | ||||
2.2. β-Glucuronidase inhibition studies
Herein, we evaluated bis-indole bearing hydrazones 1–30 for their β-glucuronidase inhibitory potential according to the protocol reported in the literature.30 These compounds demonstrated varying degrees of inhibitory potentials, whereas the IC50 values for the potent compounds were found to be in the range from 0.10 to 83.50 μM (Table 1) when compared with standard D-saccharic acid 1,4-lactone (IC50 value 48.4 ± 1.25 μM). In addition to the determination of the inhibitory potentials, molecular dockings and DFT calculations were also performed to estimate the binding modes of the current series of compounds. The results are shown in Table 1. These compounds do not show toxicity, which may be due to their higher carbon content and are expected to have lower toxicity levels compared to compounds with small molecules as reported.32Compound 16, a trihydroxy derivative, with an IC50 value of 0.10 ± 0.001, was found to be ∼500 times more active than the standard D-saccharic acid 1,4-lactone. Compounds 6 (IC50 = 18.46 ± 0.65 μM), 7 (IC50 = 34.46 ± 0.85 μM), 8 (IC50 = 29.15 ± 0.75 μM), 9 (IC50 = 15.14 ± 0.55 μM), and 18 (IC50 = 21.14 ± 1.05 μM) were found to be more potent than the standard. Compounds 25 (IC50 = 42.26 ± 1.16 μM) and 11 (IC50 = 44.16 ± 1.20 μM) displayed inhibitory potentials comparable to the standard. It was shown that the presence of hydroxyl and halogen groups, in general, proved to be the decisive factor that resulted in the enhanced inhibition of β-glucuronidase, as depicted in our previous studies.33,34 Furthermore, some stunning similarities in inhibition potential between the fluoro-, chloro-, hydroxyl, and nitro-substituted derivatives were observed. For example, all the ortho analogs, 12 with an OH group, 27 with a chloro group, 18 with fluoro substitution, and finally compound 22 with a nitro group, displayed a similar trend in their inhibitory potential (9.60 ± 0.16 to 13.38 ± 0.22 μM) toward β-glucuronidase. Similarly, the meta-hydroxy and meta-chloro analogs, 29 and 20, showed a similar potential of inhibition. These findings suggest that electronic factors are also operating and play a role in inhibition in addition to hydrogen bonding effects of the hydroxyl groups. However, it is difficult to argue the strong inhibitory potentials of the di- and trihydroxy substituted analogs and their interactions with receptor binding sites. To better understand the inhibition mechanisms and binding modes of the compounds 1–30 with β-glucuronidase, molecular modeling studies became imperative to understand the underlying phenomenon.
2.3. Molecular docking analysis
In order to obtain more insights into the binding mode of bisindolylmethane derivatives within the active site of β-D-glucuronidase and to obtain additional validations for the experimental results, molecular docking studies were performed. Herein, the X-ray crystal structure of the human β-glucuronidase enzyme at 2.6 Å resolution (PDB ID: 1BHG)36 was employed to further identify the binding modes involved in the inhibition activity. Human β-D-glucuronidase 3D structure was used for our structure–activity relationship (SAR) studies due to the absence of the bovine β-D-glucuronidase structure.Prior to the docking of the bisindolylmethane derivatives, the known substrate molecule p-nitrophenyl β-D-glucuronide was first docked into the active site of β-D-glucuronidase using the docking program AutoDock 4.2.37 The modeled substrate-bound structure of human β-D-glucuronidase showed that the glycoside bond of p-nitrophenyl β-D-glucuronide was properly oriented toward the catalytic residues, Glu451 and Glu540 (Fig. 1). It has been proposed for human β-D-glucuronidase that during catalysis, Glu451 acts as the acid/base catalyst, whereas Glu540 serves as the nucleophilic residue.36 As shown in Fig. 1, the substrate neatly fits in the active site, forming various hydrogen bonding interactions with the active site residues, including Asp207, His385, Glu451, Tyr507, and Glu540.
 | ||
| Fig. 1 Native β-D-glucuronide in the active site of β-glucuronidase surrounded by the amino acid residues Asp207, His385, Glu451, Tyr507, and Glu540. | ||
All the residues play an important role in binding the substrate molecule in the active site of β-D-glucuronidase.36 This modeled protein structure was used in the prediction of a favorable binding mode for the newly synthesized bisindolylmethane derivatives. The predicted binding models of the biologically active compounds are shown in Fig. 2. The docking studies showed that the active compounds are well accommodated in the binding cavity of the enzyme. Analysis of the predicted binding conformations of our most active compound 16 (IC50 = 0.10 ± 0.001 μM) revealed that compound 16 can fit straight into the binding groove of β-D-glucuronidase. Visually inspecting the best binding position for compound 16 showed that the hydroxyl group at the C-3 position formed a strong hydrogen bond interaction with the side chain carboxyl oxygen (Oε1) of Glu451 (1.94 Å), whereas the hydroxyl group at the C-5 position formed a hydrogen bond with the side chain nitrogen (Nε2) of His385 (2.43 Å). However, the hydroxyl group at the C-4 position did not form any interaction with the surrounding amino acid residues. The amino group of the hydrazone linkage is involved in strong hydrogen bonding interaction with the side chain carboxyl oxygen (OD2) of Asp207 (1.82 Å). The high inhibition activity for compound 16 toward β-D-glucuronidase can be explained by these strong hydrogen bonds. The predicted binding mode of compound 16 is shown in Fig. 2c. His385, Asn450, Asn484, Glu451, and Tyr508 are involved in stabilizing the binding of the compound in the active site of β-D-glucuronidase through π-sigma, π-anion, and T-shaped π–π interactions.
In contrast, the binding mode of compound 4 (IC50 = 0.20 ± 0.01 μM), which is the second most active compound among the series, revealed that the hydroxyl group at the meta position is involved in hydrogen bond interaction with the side chain carboxyl oxygen (Oε1) of Glu540 (1.98 Å) (Fig. 2a). Another hydroxyl group of compound 4 at the para position was observed to form a hydrogen bond with Asn450 (2.68 Å). The binding mode of compound 8 (IC50 = 0.50 ± 0.3 μM) revealed that the hydroxyl group at the ortho position forms a hydrogen bond with the side chain carboxyl of Glu451 (1.93 Å), whereas the hydroxyl at the para position did not adopt a favorable conformation for binding with the surrounding residues (Fig. 2b). The observed binding mode of compound 25 showed that it possesses three hydroxyl groups at the ortho and para positions. The docking study showed that the hydroxyl at the para position was able to interact well with Glu540 (2.03 Å), Asn450 (2.94 Å), and His385 (2.59 Å). Both hydroxyl groups at the ortho position of compound 25 formed strong interactions with the side chain hydroxyl oxygen (OD2) of both Asp207 (1.67 Å) and Glu451 (1.56 Å) (Fig. 2d). Hydrazone linkage formed weak hydrogen bonding with some important residues like Arg600 (3.03 Å), Lys606 (2.81 Å), and Tyr504 (2.36 Å).
In the present study, it was observed that the compounds are positioned in such a way that enabled the meta hydroxyl substituent to form a better interaction with the important residues, such as Glu451 and Glu540, than to the hydroxyls at the ortho and para positions.
The docking studies on the inactive compounds 1 (IC50 = 83.50 ± 1.70 μM), 3 (IC50 = 73.50 ± 1.40 μM), and 10 (IC50 = 88.56 ± 1.88 μM) (Fig. 3) clearly demonstrated the reason for their inactivity against the enzyme. These compounds were unable to form any noteworthy hydrogen bond interactions with the surrounding amino acid residues. It was observed that their hydrazone linkage generally formed hydrogen bond interactions with Asn207 and Arg600 with the distance between 1.81 and 2.65 Å. Hence, the activity of these compounds largely depends on the maximum hydrophilic interactions with the active site amino acids of β-D-glucuronidase.
2.4. Density functional predictions for hydrogen bonding
In order to emphasize the role of the hydrogen bonds (intramolecular or intermolecular) of the synthesized bisindolylmethane–hydrazone hybrids in β-glucuronidase inhibition, DFT calculations were performed at the B3LYP/6-31+G(d,p) level. The potency of the β-glucuronidase inhibition of the synthesized bisindolylmethane derivatives can be related to the number of intramolecular hydrogen bonds occurring in the substrates (bisindolylmethane derivatives) and to the number of intermolecular hydrogen bonds that occur at the substrate–receptor complex. Herein, we focused on the stability and effects of the number of intramolecular hydrogen bonds of the bisindolylmethane derivatives in β-glucuronidase inhibition. A high number of hydrogen bonds mean that bisindolylmethane is more stable and the inhibition is more profound (low IC50). The gap energies (eV), electronic energies, number of electronic donor groups, number of withdrawing groups, number of intramolecular hydrogen bonds, and number of intramolecular hydrogen bonds of the bisindolylmethane hybrids are reported in Table 2. The bisindolylmethane hybrids that show high β-glucuronidase inhibition (0.1 ≤ IC50 ≤ 10 μM) are those with a high number of electron donating groups. For instance, bisindolylmethane derivative 16, with two intramolecular hydrogen bonds (Table 1S†) and three electron donating groups (i.e., the ability to form three intermolecular hydrogen bonds) showed potent β-glucuronidase inhibition (IC50 = 0.1 μM) compared with the bisindolylmethane derivative 7, which possesses one intramolecular hydrogen bond (Table 1S, ESI†) and two electron donor groups (IC50 = 3.50 μM). The bisindolylmethane derivative 16 is relatively stable compared to 7 (ΔE = 4.7 kcal mol−1), which confirms the role of hydrogen bonding in the stability of the substrate and its role (i.e., via hydrogen bonding) in inhibiting β-glucuronidase. The 3D-structure of the optimized bisindolylmethane–hydrazone hybrids are reported in Fig. 1S in the ESI.† Based on the results from the docking and density functional theory studies, some discussion on the ability of these compounds to mainly interact with the target enzyme through hydrogen bonding or van der Waals interactions is included. As the results showed that the compounds interact quite well through these interactions, which involve no chemical interaction, these compounds could possibly be reversible inhibitors.| Compounds | EDG | EWG | HB | HB′ | OH | Electronic energy, E | Gap energy | IC50 (μM ± SEMa) |
|---|---|---|---|---|---|---|---|---|
| a SEM is the standard error of the mean.b N.A. no activity. | ||||||||
| 1 | 1 | 0 | 0 | 1 | 0 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 056 056![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 229.52 229.52 |
3.69 | 83.5 ± 1.70 |
| 2 | 1 | 0 | 0 | 0 | 0 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 009 009![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 033.76 033.76 |
3.60 | N. A.b |
| 3 | 0 | 1 | 0 | 0 | 0 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 112 112![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 695.66 695.66 |
2.43 | 73.5 ± 1.40 |
| 4 | 2 | 0 | 1 | 2 | 2 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 078 078![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 770.62 770.62 |
3.67 | 0.2 ± 0.01 |
| 5 | 1 | 0 | 0 | 0 | 0 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 009 009![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 034.12 034.12 |
3.62 | 51.1 ± 1.29 |
| 6 | 1 | 0 | 0 | 0 | 0 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 009 009![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 032.00 032.00 |
3.55 | 23.12 ± 0.45 |
| 7 | 2 | 0 | 1 | 2 | 2 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 078 078![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 773.53 773.53 |
3.52 | 3.5 ± 0.05 |
| 8 | 2 | 0 | 1 | 2 | 2 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 078 078![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 768.97 768.97 |
3.57 | 0.5 ± 0.3 |
| 9 | 0 | 1 | 1 | 0 | 0 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 046 046![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 636.60 636.60 |
3.56 | 3.8 ± 0.08 |
| 10 | 0 | 0 | 0 | 0 | 0 | −994![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 424.90 424.90 |
3.42 | 88.56 ± 1.88 |
| 11 | 2 | 0 | 1 | 2 | 1 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 103 103![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 439.57 439.57 |
3.67 | 13.6 ± 0.26 |
| 12 | 1 | 0 | 1 | 1 | 1 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 031 031![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 570.87 570.87 |
3.55 | 12.1 ± 0.24 |
| 13 | 0 | 1 | 0 | 0 | 0 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 112 112![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 696.32 696.32 |
2.31 | N. A.b |
| 14 | 2 | 0 | 1 | 2 | 2 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 078 078![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 773.53 773.53 |
3.52 | 22.18 ± 0.45 |
| 15 | 1 | 0 | 1 | 1 | 1 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 031 031![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 566.53 566.53 |
3.69 | 15.26 ± 0.26 |
| 16 | 3 | 0 | 2 | 3 | 3 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 125 125![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 978.56 978.56 |
3.65 | 0.1 ± 0.008 |
| 17 | 0 | 0 | 0 | 0 | 0 | −994![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 424.73 424.73 |
3.36 | 18.6 ± 0.30 |
| 18 | 0 | 1 | 0 | 0 | 0 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 046 046![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 636.25 636.25 |
3.46 | 9.6 ± 0.16 |
| 19 | 2 | 0 | 1 | 2 | 1 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 103 103![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 434.15 434.15 |
3.69 | 32.36 ± 0.64 |
| 20 | 0 | 1 | 0 | 0 | 0 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 272 272![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 761.43 761.43 |
3.44 | 16.1 ± 0.29 |
| 21 | 2 | 0 | 1 | 2 | 1 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 103 103![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 436.46 436.46 |
3.54 | 11.28 ± 0.23 |
| 22 | 0 | 1 | 0 | 0 | 0 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 112 112![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 690.60 690.60 |
2.44 | 13.38 ± 0.22 |
| 23 | 2 | 0 | 1 | 2 | 2 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 078 078![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 776.66 776.66 |
3.67 | 6.3 ± 0.10 |
| 24 | 1 | 0 | 0 | 0 | 0 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 009 009![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 033.85 033.85 |
3.59 | N. A.b |
| 25 | 3 | 0 | 1 | 3 | 3 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 125 125![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 980.82 980.82 |
3.69 | 2.2 ± 0.06 |
| 26 | 0 | 1 | 0 | 0 | 0 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 272 272![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 761.84 761.84 |
3.47 | 26.63 ± 0.38 |
| 27 | 0 | 1 | 0 | 0 | 0 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 272 272![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 757.00 757.00 |
3.47 | 15.2 ± 0.28 |
| 28 | 0 | 1 | 0 | 0 | 0 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 046 046![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 636.45 636.45 |
3.45 | 26.2 ± 0.39 |
| 29 | 1 | 0 | 1 | 1 | 1 | −1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 031 031![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 565.89 565.89 |
3.53 | 16.25 ± 0.29 |
| 30 | 0 | 0 | 0 | 0 | 0 | −994![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 425.20 425.20 |
3.24 | N. A.b |
In conclusion, the binding of the active compounds with the target site is mainly dependent on the nature and position of the polar groups, such as the hydroxyl and halogen groups. In addition to the identification of the hydroxyl and halogen substituted analogs as the most potent compounds, it was also observed that the compounds in which hydroxyl groups were positioned adjacently were found to interact exclusively with the important residues, such as Glu451 and Glu540, as compared to hydroxyl groups that were far apart from each other. Furthermore, DFT studies confirmed the role of hydrogen bonding along with the role of the electronic nature of the substituents; these factors played pivotal roles in the inhibition potential of β-glucuronidase.
3. Experimental
3.1. General
NMR experiments were performed on an UltraShield Bruker FT NMR 500 MHz; CHN analysis was performed on a Carlo Erba Strumentazion-Mod-1106, Italy. Electron impact mass spectra (EI-MS) were obtained on a Finnegan MAT-311A, Germany. Thin layer chromatography (TLC) was performed on pre-coated silica gel aluminum plates (Kieselgel 60, 254, E. Merck, Germany). Chromatograms were visualized by UV at 254 and 365 nm.3.2. β-Glucuronidase inhibition activity
β-Glucuronidase (E.C. 3.2.1.31 from bovine liver, G-0251) inhibition activity of the compounds was evaluated using the method reported in the ref. 30 and 37. In the present study, D-saccharic acid 1,4-lactone was used as the standard drug.38,393.3. Synthesis of methyl 4-(bis(1-methyl-1H-indol-3-yl)methyl)benzoate (I)
Methyl 2-hydroxybenzoate (1) (7.60 g, 53 mmol) and 20 mL of hydrazine hydrate were mixed in methanol (50 mL). The mixture was refluxed for 6 h. Methanol was then evaporated and the product formed was rinsed with plenty of water to remove excess hydrazine hydrate. The product formed was left to dry at room temperature and its yield was recorded. Yield: 9.68 g (94.9%). White solid. mp 239–241 °C; IR (cm−1, ATR): 3082, 2975, 1738, 1627, 1546, 1468, 1126; 1H NMR (500 MHz, DMSO-d6): δ 7.97 (d, J = 7.5 Hz, 2H), 7.83 (dd, J = 7.5, 1.6 Hz, 2H), 7.76 (dd, J = 7.4, 1.5 Hz, 2H), 7.63 (s, 2H), 7.44 (d, J = 7.5 Hz, 2H), 7.32 (dtd, J = 29.3, 7.5, 1.5 Hz, 4H), 5.74 (s, 1H), 3.92 (s, 3H), 3.78 (s, 6H); 13C-NMR (DMSO-d6, 125 MHz): δ 167.45, 141.55, 141.55, 140.27, 130.69, 130.37, 130.37, 128.65, 128.65, 128.49, 128.49, 128.20, 128.20, 124.69, 124.69, 123.42, 123.42, 122.72, 122.72, 111.63, 111.63, 111.08, 111.08, 52.18, 38.43, 35.19, 35.19; HREI-MS: m/z calcd for C27H24N2O2 [M]+ 408.1838; found 408.1838; anal. calcd for C27H24N2O2, C, 79.39; H, 5.92; N, 6.86; O, 7.83; found C, 79.39; H, 5.92; N, 6.86; O, 7.83.3.4. Synthesis of 4-(bis(1-methyl-1H-indol-3-yl)methyl)benzohydrazide (II)
A mixture of compound 2 (7.00 g, 46 mmol), methyl 4-formylbenzoate (7.56 g, 46 mmol), and a catalytic amount of acetic acid in methanol (50 mL) was refluxed for 3 h. The solvent was evaporated and the residue (3) was washed with diethyl ether, filtered, dried, and then crystallized from ethanol to give a brownish solid. Yield: 5.69 g, 93.0%. mp 269–271 °C; IR (cm−1, ATR): 3423, 3352, 3137, 2997, 1655, 1612, 1558, 1469, 1341, 1268; 1H NMR (500 MHz, DMSO-d6): δ 8.33 (s, 1H), 7.94–7.84 (m, 4H), 7.76 (dd, J = 7.5, 1.4 Hz, 2H), 7.49 (d, J = 7.5 Hz, 2H), 7.42 (s, 2H), 7.34 (td, J = 7.4, 1.5 Hz, 2H), 7.26 (td, J = 7.4, 1.5 Hz, 2H), 5.73 (s, 1H), 4.08 (s, 2H), 3.76 (s, 6H); 13C-NMR (150 MHz, DMSO-d6): δ 167.32, 142.28, 141.57, 141.57, 135.10, 135.10, 130.34, 130.34, 129.66, 129.66, 128.20, 128.20, 126.76, 126.76, 124.79, 124.79, 123.56, 123.56, 122.85, 122.75, 111.78, 111.78, 111.28, 111.28, 38.48, 35.99, 35.99; HREI-MS: m/z calcd for C26H24N4O [M]+ 408.1950; found 408.1950; anal. calcd for C26H24N4O, C, 76.45; H, 5.92; N, 13.72; found C, 76.45; H, 5.92; N, 13.72.3.4.1.1. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(4-methoxybenzylidene)benzohydrazide (1). Brown solid. Yield: 75.2%. mp: 259–261 °C. IR (cm−1, ATR): 3425, 3339, 3143, 1643, 1609, 1254. 1H NMR (500 MHz, DMSO-d6): δ 11.62 (s, 1H), 8.36 (s, 1H), 7.81 (d, J = 8.2 Hz, 2H), 7.66 (d, J = 8.3 Hz, 2H), 7.50 (d, J = 8.0 Hz, 2H), 7.40 (d, J = 8.2 Hz, 2H), 7.33 (d, J = 7.9 Hz, 2H), 7.13 (t, J = 7.6 Hz, 2H), 7.02 (d, J = 8.4 Hz, 2H), 6.93 (t, J = 7.5 Hz, 2H), 6.88 (s, 2H), 5.94 (s, 1H), 3.81 (s, 3H), 3.72 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.32, 160.20, 149.54, 142.36, 141.70, 141.70, 134.31, 130.25, 130.25, 129.63, 129.63, 129.22, 129.22, 128.19, 128.19, 128.09, 128.09, 127.15, 124.79, 124.79, 123.42, 123.42, 122.71, 122.71, 114.42, 114.42, 111.78, 111.78, 111.02, 111.02, 56.13, 38.32, 35.92, 35.92; HREI-MS: m/z calcd for C34H30N4O2 [M]+ 526.2369; found 526.2373; anal. calcd for C34H30N4O2, C, 77.54; H, 5.74; N, 10.64; found C, 77.56; H, 5.73; N, 10.65.
3.4.1.2. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(3-methylbenzylidene)benzohydrazide (2). Pale yellow solid. Yield: 82.9%. mp: 262–263 °C. IR (cm−1, ATR): 3324, 3260, 3064, 1656, 1590, 1539, 1469, 1357, 1277, 1180, 752; 1H NMR (500 MHz, DMSO-d6): δ 11.73 (s, 1H), 8.38 (s, 1H), 7.82 (d, J = 7.9 Hz, 2H), 7.55 (s, 1H), 7.50 (d, J = 8.0 Hz, 2H), 7.40 (d, J = 8.2 Hz, 2H), 7.33 (d, J = 7.9 Hz, 2H), 7.25 (d, J = 8.1 Hz, 1H), 7.13 (t, J = 7.6 Hz, 2H), 6.93 (t, J = 7.5 Hz, 2H), 6.88 (s, 2H), 5.95 (s, 1H), 3.72 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.38, 148.78, 142.56, 141.40, 141.40, 137.86, 136.61, 134.31, 130.45, 130.45, 129.81, 129.63, 128.74, 128.74, 128.59, 128.31, 128.09, 128.09, 124.79, 124.79, 123.51, 123.51, 123.49, 123.49, 122.73, 122.73, 111.78, 111.78, 111.15, 111.15, 38.48, 35.69, 35.69, 21.35; HREI-MS: m/z calcd for C34H30N4O [M]+ 510.2420; found 510.2424; anal. calcd for C34H30N4O, C, 79.97; H, 5.92; N, 10.97; found C, 79.98; H, 5.93; N, 10.98.
3.4.1.3. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(3-nitrobenzylidene)benzohydrazide (3). Red solid. Yield: 71.3%. mp: 262–264 °C. IR (cm−1, ATR): 3335, 3260, 1560, 3138, 1657, 1609, 1275, 1125; 1H NMR (500 MHz, DMSO-d6): δ 12.03 (s, 1H), 8.55 (s, 2H), 8.26 (d, J = 7.4 Hz, 1H), 8.15 (d, J = 7.0 Hz, 1H), 7.84 (d, J = 7.8 Hz, 2H), 7.76 (t, J = 7.8 Hz, 1H), 7.52 (d, J = 8.1 Hz, 2H), 7.40 (d, J = 8.2 Hz, 2H), 7.34 (d, J = 7.9 Hz, 2H), 7.13 (t, J = 7.5 Hz, 2H), 6.94 (t, J = 7.5 Hz, 2H), 6.89 (s, 2H), 5.96 (s, 1H), 3.73 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.52, 148.78, 147.38, 142.71, 141.47, 141.47, 137.78, 134.11, 132.69, 130.31, 130.31, 129.96, 129.51, 129.51, 128.25, 128.25, 128.20, 128.20, 124.79, 124.79, 124.12, 123.44, 123.44, 122.72, 122.72, 122.43, 111.78, 111.78, 111.26, 111.26, 38.37, 35.92, 35.92; HREI-MS: m/z calcd for C33H27N5O3 [M]+ 541.2114; found 541.2118; anal. calcd for C33H27N5O3, C, 73.18; H, 5.02; N, 12.93; found C, 73.19; H, 5.00; N, 12.94.
3.4.1.4. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(3,4-dihydroxybenzylidene)benzohydrazide (4). Red solid. Yield: 78.2%. mp: 278–280 °C. IR (cm−1, ATR): 3423, 3299, 3072, 1636, 1617, 1545, 1277, 1180; 1H NMR (500 MHz, DMSO-d6): δ 11.50 (s, 1H), 9.35 (s, 1H), 9.23 (s, 1H), 8.23 (s, 1H), 7.80 (d, J = 8.2 Hz, 2H), 7.49 (d, J = 8.2 Hz, 2H), 7.39 (d, J = 8.3 Hz, 2H), 7.33 (d, J = 7.9 Hz, 2H), 7.23 (s, 1H), 7.13 (t, J = 7.6 Hz, 2H), 6.91–6.95 (m, 3H), 6.88 (s, 1H), 6.78 (d, J = 8.1 Hz, 1H), 5.94 (s, 1H), 3.72 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.38, 148.69, 148.09, 145.47, 142.76, 141.48, 141.48, 134.18, 130.32, 130.32, 129.73, 129.73, 128.49, 128.49, 128.20, 128.20, 127.87, 124.69, 124.69, 123.36, 123.36, 122.65, 122.65, 121.12, 116.73, 115.93, 111.58, 111.58, 111.18, 111.18, 38.52, 35.92, 35.92; HREI-MS: m/z calcd for C33H28N4O3 [M]+ 528.2161; found 528.2161; anal. calcd for C33H28N4O3, C, 74.98; H, 5.34; N, 10.60; found C, 75.01; H, 5.32; N, 10.58.
3.4.1.5. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(4-methylbenzylidene)benzohydrazide (5). Dark brown solid. Yield: 79.4%. mp: 246–248 °C. IR (cm−1, ATR): 3347, 3352, 3043, 1631, 1591, 1555, 1357, 1270, 1143, 752; 1H NMR (500 MHz, DMSO-d6): δ 11.68 (s, 1H), 8.38 (s, 1H), 7.81 (d, J = 8.1 Hz, 2H), 7.61 (d, J = 7.4 Hz, 2H), 7.50 (d, J = 7.9 Hz, 2H), 7.40 (d, J = 8.2 Hz, 2H), 7.33 (d, J = 7.6 Hz, 2H), 7.27 (d, J = 7.5 Hz, 2H), 7.13 (t, J = 7.6 Hz, 2H), 6.93 (t, J = 7.5 Hz, 2H), 6.88 (s, 2H), 5.95 (s, 1H), 3.72 (s, 6H), 2.35 (s, 3H); 13C NMR (150 MHz, DMSO-d6): δ 164.38, 149.24, 142.56, 141.47, 141.47, 138.49, 134.15, 131.96, 130.38, 130.38, 129.43, 129.43, 129.25, 129.25, 128.28, 128.28, 128.07, 128.07, 127.30, 127.30, 124.82, 124.82, 123.36, 123.36, 122.63, 122.63, 111.67, 111.67, 111.04, 111.04, 38.38, 35.72, 35.72, 21.09; HREI-MS: m/z calcd for C34H30N4O [M]+ 510.2420; found 510.2425; anal. calcd for C34H30N4O, C, 79.97; H, 5.92; N, 10.97; found C, 79.98; H, 5.90; N, 10.95.
3.4.1.6. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(2-methylbenzylidene)benzohydrazide (6). Light brown solid. Yield: 89.7%. mp: 246–248 °C. IR (cm−1, ATR): 3325, 3299, 3043, 1631, 1568, 1357, 1277, 1180, 752; 1H NMR (500 MHz, DMSO-d6): δ 11.73 (s, 1H), 8.72 (s, 1H), 7.83 (t, J = 7.7 Hz, 2H), 7.51 (d, J = 7.9 Hz, 2H), 7.40 (d, J = 8.2 Hz, 2H), 7.33 (d, J = 7.9 Hz, 2H), 7.32–7.23 (m, 3H), 7.13 (t, J = 7.6 Hz, 2H), 6.94 (t, J = 7.5 Hz, 2H), 6.88 (s, 2H), 5.95 (s, 1H), 3.72 (s, 6H), 2.43 (s, 3H); 13C NMR (150 MHz, DMSO-d6): δ 164.28, 145.62, 142.69, 141.45, 141.45, 136.27, 134.26, 133.49, 130.51, 130.51, 130.31, 129.50, 129.50, 128.49, 128.31, 128.31, 128.12, 128.12, 126.79, 126.27, 124.69, 124.69, 123.48, 123.48, 122.79, 122.79, 111.58, 111.58, 111.18, 111.18, 38.32, 35.91, 35.91, 20.15; HREI-MS: m/z calcd for C34H30N4O [M]+ 510.2420; found 510.2415; anal. calcd for C34H30N4O, C, 79.97; H, 5.92; N, 10.97; found C, 79.99; H, 5.90; N, 10.96.
3.4.1.7. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(2,5-dihydroxybenzylidene)benzohydrazide (7). Red solid. Yield: 92.6%. mp: 284–285 °C. IR (cm−1, ATR): 3353, 3139, 2994, 1657, 1611, 1556, 1495, 1466, 1259, 1166, 749; 1H NMR (500 MHz, DMSO-d6): δ 11.90 (s, 1H), 10.40 (s, 1H), 8.95 (s, 1H), 8.53 (s, 1H), 7.84 (d, J = 8.0 Hz, 2H), 7.51 (d, J = 8.1 Hz, 2H), 7.40 (d, J = 8.2 Hz, 2H), 7.33 (d, J = 7.9 Hz, 2H), 7.13 (t, J = 7.6 Hz, 2H), 6.97–6.91 (m, 3H), 6.88 (s, 2H), 5.95 (s, 1H), 3.72 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.46, 152.63, 151.39, 149.69, 142.52, 141.47, 141.47, 134.18, 130.34, 130.34, 129.57, 129.57, 128.16, 128.16, 128.09, 128.09, 124.76, 124.76, 123.26, 123.26, 122.65, 122.65, 122.16, 121.52, 118.28, 116.47, 111.78, 111.78, 111.04, 111.04, 38.47, 35.93, 35.93; HREI-MS: m/z calcd for C33H28N4O3 [M]+ 528.2161; found 528.2157; anal. calcd for C33H28N4O3, C, 74.98; H, 5.34; N, 10.60; found C, 74.99; H, 5.32; N, 10.58.
3.4.1.8. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(2,3-dihydroxybenzylidene)benzohydrazide (8). Orange solid, Yield: 86.5%. mp: 257–259 °C. IR (cm−1, ATR): 3381, 3062, 1613, 1611, 1269, 1205, 750; 1H NMR (500 MHz, DMSO-d6): δ 12.02 (s, 1H), 11.18 (s, 1H), 9.17 (s, 1H), 8.56 (s, 1H), 7.85 (d, J = 6.4 Hz, 2H), 7.52 (d, J = 6.6 Hz, 2H), 7.40 (d, J = 8.1 Hz, 2H), 7.34 (d, J = 7.7 Hz, 2H), 7.13 (t, J = 6.5 Hz, 2H), 6.94 (t, J = 7.3 Hz, 2H), 6.89 (s, 1H), 6.86 (d, J = 7.1 Hz, 1H), 6.74 (t, J = 6.6 Hz, 1H), 5.96 (s, 1H), 3.73 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.37, 149.97, 146.93, 144.72, 142.68, 141.57, 141.57, 134.19, 130.34, 130.34, 129.52, 129.52, 128.18, 128.18, 128.08, 128.08, 124.91, 124.91, 123.49, 123.49, 122.85, 122.85, 122.09, 121.42, 119.90, 119.73, 111.61, 111.61, 111.18, 111.18, 38.39, 35.94, 35.94; HREI-MS: m/z calcd for C33H28N4O3 [M]+ 528.2161; found 528.2165; anal. calcd for C33H28N4O3, C, 74.98; H, 5.34; N, 10.60; found C, 74.99; H, 5.35; N, 10.58.
3.4.1.9. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(4-fluorobenzylidene)benzohydrazide (9). Brown solid. Yield: 85.8%. mp: 248–250 °C. IR (cm−1, ATR): 3354, 3212, 3021, 1626, 1606, 1519, 1470, 1372, 1269, 1186, 760; 1H NMR (500 MHz, DMSO-d6): δ 11.77 (s, 1H), 8.42 (s, 1H), 7.86–7.73 (m, 4H), 7.50 (d, J = 7.9 Hz, 2H), 7.40 (d, J = 8.2 Hz, 2H), 7.33 (d, J = 7.9 Hz, 2H), 7.32–7.26 (m, 2H), 7.13 (t, J = 7.6 Hz, 2H), 6.94 (t, J = 7.4 Hz, 2H), 6.88 (s, 2H), 5.95 (s, 1H), 3.73 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.52, 160.29 (d, J = 261.5 Hz), 149.43, 142.51, 141.62, 141.62, 134.74, 130.69, 130.40, 130.40, 130.25, 130.25, 129.43, 129.43, 128.31, 128.31, 128.15, 128.15, 124.82, 124.82, 123.36, 123.36, 122.72, 122.72, 115.51, 115.51, 111.78, 111.78, 111.18, 111.18, 38.25, 35.69, 35.69; HREI-MS: m/z calcd for C33H27FN4O [M]+ 514.2169; found 514.2172; anal. calcd for C33H27FN4O, C, 77.02; H, 5.29; N, 10.89; found C, 77.01; H, 5.31; N, 10.90.
3.4.1.10. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(pyridin-3-ylmethylene)benzohydrazide (10). Light brown solid. Yield: 89.6%. mp: 258–260 °C. IR (cm−1, ATR): 3373, 3159, 2835, 1609, 1558, 1470, 1445, 1257, 1186, 754; 1H NMR (500 MHz, DMSO-d6): δ 11.93 (s, 1H), 8.85 (s, 1H), 8.61 (s, 1H), 8.48 (s, 1H), 8.13 (s, 1H), 7.83 (d, J = 7.3 Hz, 2H), 7.51 (d, J = 7.8 Hz, 2H), 7.40 (d, J = 8.0 Hz, 2H), 7.34 (d, J = 7.7 Hz, 2H), 7.13 (t, J = 7.3 Hz, 2H), 6.94 (t, J = 7.5 Hz, 2H), 6.89 (s, 2H), 5.95 (s, 1H), 3.72 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 165.32, 150.15, 146.51, 145.72, 142.46, 141.70, 141.70, 135.59, 134.17, 133.82, 130.45, 130.45, 129.33, 129.33, 128.18, 128.18, 128.07, 128.07, 124.91, 124.91, 124.31, 123.45, 123.45, 122.85, 122.85, 111.72, 111.72, 111.02, 111.02, 38.52, 35.79, 35.79; HREI-MS: m/z calcd for C32H27N5O [M]+ 497.2216; found 497.2213; anal. calcd for C32H27N5O, C, 77.24; H, 5.47; N, 14.07; found C, 77.25; H, 5.48; N, 14.05.
3.4.1.11. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(2-hydroxy-4-methoxybenzylidene)benzohydrazide (11). Red solid. Yield: 89.6%. mp: 255–257 °C. IR (cm−1, ATR): 3389, 3249, 3002, 1684, 1652, 1585, 1467, 1496, 1282, 1205, 750; 1H NMR (500 MHz, DMSO-d6): δ 11.90 (s, 1H), 11.64 (s, 1H), 8.51 (s, 1H), 7.83 (d, J = 8.1 Hz, 2H), 7.51 (d, J = 8.2 Hz, 2H), 7.40 (t, J = 4.2 Hz, 2H), 7.39 (s, 1H), 7.33 (d, J = 7.9 Hz, 2H), 7.13 (t, J = 7.6 Hz, 2H), 6.93 (t, J = 7.5 Hz, 2H), 6.88 (s, 2H), 6.52 (d, J = 8.6 Hz, 1H), 6.50 (s, 1H), 5.95 (s, 1H), 3.78 (s, 3H), 3.72 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.41, 162.10, 161.25, 151.29, 142.69, 141.72, 141.72, 134.31, 130.92, 130.32, 130.32, 129.58, 129.58, 128.26, 128.26, 128.18, 128.18, 124.79, 124.79, 123.36, 123.36, 122.62, 122.62, 113.87, 111.69, 111.69, 111.18, 111.18, 107.39, 102.25, 56.23, 38.39, 35.92, 35.92; HREI-MS: m/z calcd for C34H30N4O3 [M]+ 542.2318; found 542.2323; anal. calcd for C34H30N4O3, C, 75.26; H, 5.57; N, 10.33; found C, 75.27; H, 5.56; N, 10.35.
3.4.1.12. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(2-hydroxybenzylidene)benzohydrazide (12). Brown solid. Yield: 79.7%. mp: 272–274 °C. IR (cm−1, ATR): 3347, 3140, 3054, 1650, 1603, 1489, 1465, 1260, 1184, 750; 1H NMR (500 MHz, DMSO-d6): δ 12.02 (s, 1H), 11.30 (s, 1H), 8.60 (s, 1H), 7.85 (d, J = 8.0 Hz, 2H), 7.52 (d, J = 7.9 Hz, 2H), 7.40 (d, J = 8.2 Hz, 2H), 7.33 (d, J = 7.9 Hz, 2H), 7.13 (t, J = 7.6 Hz, 2H), 6.96–6.91 (m, 3H), 6.89 (s, 2H), 5.96 (s, 1H), 3.73 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.51, 158.09, 151.32, 142.50, 141.66, 141.66, 134.34, 130.20, 130.20, 129.86, 129.43, 129.43, 128.51, 128.32, 128.32, 128.08, 128.08, 124.79, 124.79, 123.35, 123.35, 122.86, 122.86, 121.23, 120.42, 117.31, 111.64, 111.64, 111.28, 111.28, 38.45, 35.69, 35.69; HREI-MS: m/z calcd for C33H28N4O2 [M]+ 512.2212; found 512.2209; anal. calcd for C33H28N4O2, C, 77.32; H, 5.51; N, 10.93; found C, 77.33; H, 5.53; N, 10.95.
3.4.1.13. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(4-nitrobenzylidene)benzohydrazide (13). Yellow solid. Yield: 91.7%. mp: 284–285 °C. IR (cm−1, ATR): 3429, 3254, 2835, 1642, 1614, 1584, 1561, 1520, 1470, 1440, 1275, 1236, 1180, 755; 1H NMR (500 MHz, DMSO-d6): δ 12.06 (s, 1H), 8.52 (s, 1H), 8.30 (d, J = 8.2 Hz, 2H), 7.99 (d, J = 6.7 Hz, 2H), 7.84 (d, J = 7.9 Hz, 2H), 7.52 (d, J = 8.0 Hz, 2H), 7.40 (d, J = 8.2 Hz, 2H), 7.34 (d, J = 7.9 Hz, 2H), 7.13 (t, J = 7.6 Hz, 2H), 6.94 (t, J = 7.5 Hz, 2H), 6.89 (s, 2H), 5.96 (s, 1H), 3.73 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.34, 149.22, 148.66, 142.14, 141.95, 141.95, 139.64, 134.35, 130.26, 130.26, 129.21, 129.21, 128.52, 128.52, 128.12, 128.12, 127.87, 127.87, 124.95, 124.95, 124.58, 124.58, 123.47, 123.47, 122.65, 122.65, 111.78, 111.78, 111.06, 111.06, 38.52, 35.78, 35.78; HREI-MS: m/z calcd for C33H27N5O3 [M]+ 541.2114; found 541.2117; anal. calcd for C33H27N5O3, C, 73.18; H, 5.02; N, 12.93; found C, 73.19; H, 5.01; N, 12.95.
3.4.1.14. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(3,5-dihydroxybenzylidene)benzohydrazide (14). Brown solid. Yield: 86.2%. mp: 262–263 °C. IR (cm−1, ATR): 3143, 2962, 3062, 1603, 1583, 1490, 1462, 1266, 1164, 756; 1H NMR (500 MHz, DMSO-d6): δ 11.61 (s, 1H), 9.41 (s, 2H), 7.81 (d, J = 8.3 Hz, 2H), 7.50 (d, J = 8.3 Hz, 2H), 7.40 (s, 1H), 7.40–7.27 (m, 4H), 7.13 (t, J = 7.3 Hz, 2H), 6.93 (t, J = 7.3 Hz, 2H), 6.88 (s, 2H), 6.59 (s, 2H), 6.26 (s, 1H), 5.95 (s, 1H), 3.72 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.30, 158.28, 158.28, 147.66, 142.91, 141.52, 141.52, 138.42, 134.29, 130.25, 130.25, 129.52, 129.52, 128.29, 128.29, 128.12, 128.12, 124.89, 124.89, 123.46, 123.46, 122.45, 122.45, 111.78, 111.78, 111.28, 111.28, 107.45, 107.45, 104.27, 38.32, 35.49, 35.49; HREI-MS: m/z calcd for C33H28N4O3 [M]+ 528.2161; found 528.2158; anal. calcd for C33H28N4O3, C, 74.98; H, 5.34; N, 10.60; found C, 75.01; H, 5.32; N, 10.58.
3.4.1.15. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(4-hydroxybenzylidene)benzohydrazide (15). Dark brown solid. Yield: 86.2%. mp: 276–278 °C. IR (cm−1, ATR): 3394, 3296, 3064, 1607, 1587, 1507, 1462, 1266, 1205, 1157, 736; 1H NMR (500 MHz, DMSO-d6): δ 11.53 (s, 1H), 9.88 (s, 1H), 8.31 (s, 1H), 7.80 (d, J = 8.2 Hz, 2H), 7.54 (d, J = 8.3 Hz, 2H), 7.49 (d, J = 8.1 Hz, 2H), 7.39 (d, J = 8.2 Hz, 2H), 7.33 (d, J = 7.9 Hz, 2H), 7.13 (t, J = 7.6 Hz, 2H), 6.93 (t, J = 7.4 Hz, 2H), 6.88 (s, 2H), 6.83 (d, J = 8.4 Hz, 2H), 5.94 (s, 1H), 3.72 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.28, 158.34, 149.44, 142.56, 141.32, 141.32, 134.41, 130.25, 130.25, 129.72, 129.72, 129.43, 129.43, 128.25, 128.25, 128.18, 128.18, 126.72, 124.69, 124.69, 123.75, 123.75, 122.46, 122.46, 115.68, 115.68, 111.59, 111.59, 111.23, 111.23, 38.89, 35.42, 35.42. HREI-MS: m/z calcd for C33H28N4O2 [M]+ 512.2212; found 512.2215; anal. calcd for C33H28N4O2, C, 77.32; H, 5.51; N, 10.93; found C, 77.34; H, 5.48; N, 10.90.
3.4.1.16. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(2,4,5-trihydroxybenzylidene)benzohydrazide (16). Light brown solid. Yield: 85.3%. mp: 291–293 °C. IR (cm−1, ATR): 3352, 3182, 3075, 1633, 1591, 1276, 1246, 1171, 752; 1H NMR (500 MHz, DMSO-d6): δ 11.70 (s, 1H), 10.65 (s, 1H), 9.51 (s, 1H), 8.52 (s, 1H), 8.41 (s, 1H), 7.82 (d, J = 8.2 Hz, 2H), 7.50 (d, J = 8.1 Hz, 2H), 7.40 (d, J = 8.2 Hz, 2H), 7.33 (d, J = 7.9 Hz, 2H), 7.13 (t, J = 7.6 Hz, 2H), 6.93 (t, J = 7.4 Hz, 2H), 6.88 (s, 2H), 6.85 (s, 1H), 6.33 (s, 1H), 5.94 (s, 1H), 3.72 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.51, 153.78, 150.93, 149.71, 142.51, 141.78, 141.78, 140.25, 134.35, 130.53, 130.53, 129.32, 129.32, 128.72, 128.72, 128.18, 128.18, 124.83, 124.83, 123.31, 123.31, 122.85, 122.85, 117.22, 112.87, 111.64, 111.64, 111.06, 111.06, 102.76, 38.37, 35.86, 35.86; HREI-MS: m/z calcd for C33H28N4O4 [M]+ 544.2111; found 544.2107; anal. calcd for C33H28N4O4, C, 72.78; H, 5.18; N, 10.29; found C, 72.79; H, 5.20; N, 10.31.
3.4.1.17. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(pyridin-2-ylmethylene)benzohydrazide (17). Light red solid. Yield: 86.3%. mp: 251–252 °C. IR (cm−1, ATR): 3433, 3253, 2915, 1660, 1557, 1466, 1277, 755; 1H NMR (500 MHz, DMSO-d6): δ 11.94 (s, 1H), 8.61 (d, J = 4.3 Hz, 1H), 8.45 (s, 1H), 7.98 (d, J = 8.9 Hz, 1H), 7.86 (T, J = 8.9 Hz, 1H), 7.83 (d, J = 8.2 Hz, 2H), 7.52 (d, J = 8.2 Hz, 2H), 7.40 (d, J = 8.2 Hz, 2H), 7.34 (d, J = 7.9 Hz, 2H), 7.13 (t, J = 7.6 Hz, 2H), 6.94 (t, J = 7.4 Hz, 2H), 6.89 (s, 2H), 5.96 (s, 1H), 3.73 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.32, 154.70, 148.07, 146.05, 142.66, 141.50, 141.50, 137.60, 134.21, 130.35, 130.35, 129.53, 129.53, 128.21, 128.21, 128.10, 128.10, 124.89, 124.89, 123.46, 123.46, 123.00, 122.75, 122.75, 119.09, 111.68, 111.68, 111.08, 111.08, 38.42, 35.89, 35.89; HREI-MS: m/z calcd for C32H27N5O [M]+ 497.2216; found 497.2221; anal. calcd for C32H27N5O, C = 77.24; H = 5.47; N = 14.07; found C = 77.25; H = 5.49; N = 14.05.
3.4.1.18. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(2-fluorobenzylidene)benzohydrazide (18). Light pink solid, Yield: 80.7%. mp: 243–245 °C. IR (cm−1, ATR): 3432, 3183, 3065, 1625, 1542, 1469, 1253, 743; 1H NMR (500 MHz, DMSO-d6): δ 11.88 (s, 1H), 8.68 (s, 1H), 7.95 (s, 1H), 7.83 (d, J = 7.9 Hz, 2H), 7.50 (t, J = 8.7 Hz, 3H), 7.40 (d, J = 8.2 Hz, 2H), 7.34 (d, J = 7.9 Hz, 2H), 7.32–7.26 (m, 2H), 7.13 (t, J = 7.6 Hz, 2H), 6.94 (t, J = 7.5 Hz, 2H), 6.89 (s, 2H), 5.95 (s, 1H), 3.73 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.41, 161.35, 149.37, 142.76, 141.41, 141.41, 134.27, 130.83, 130.55, 130.55, 129.63, 129.32, 129.32, 128.18, 128.18, 128.09, 128.09, 125.35, 124.75, 124.62, 124.54, 123.41, 123.41, 122.85, 122.85, 117.38, 111.52, 111.52, 111.16, 111.16, 38.38, 35.86, 35.86; HREI-MS: m/z calcd for C33H27FN4O [M]+ 514.2169; found 514.2172; anal. calcd for C33H27FN4O, C, 77.02; H, 5.29; N, 10.89; found C, 77.03; H, 5.26; N, 10.86.
3.4.1.19. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(3-hydroxy-4-methoxybenzylidene)benzohydrazide (19). Dark brown solid. Yield: 86.4%. mp: 279–281 °C. IR (cm−1, ATR): 3427, 3126, 2947, 1609, 1464, 1270, 749; 1H NMR (500 MHz, DMSO-d6): δ 11.56 (s, 1H), 9.26 (s, 1H), 8.27 (s, 1H), 7.80 (d, J = 8.2 Hz, 2H), 7.49 (d, J = 8.2 Hz, 2H), 7.40 (d, J = 8.3 Hz, 2H), 7.33 (d, J = 7.9 Hz, 2H), 7.26 (s, 1H), 7.13 (t, J = 7.3 Hz, 2H), 7.03 (t, J = 8.4 Hz, 1H), 6.97 (d, J = 8.5 Hz, 1H), 6.93 (t, J = 7.5 Hz, 2H), 6.88 (s, 2H), 5.94 (s, 1H), 3.81 (s, 3H), 3.72 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.48, 149.80, 148.58, 146.65, 142.46, 141.32, 141.32, 134.28, 130.25, 130.25, 129.76, 129.31, 129.31, 128.34, 128.34, 128.09, 128.09, 124.93, 124.93, 123.41, 123.41, 122.62, 122.62, 120.75, 115.34, 115.21, 111.78, 111.78, 111.26, 111.26, 56.69, 38.46, 35.79, 35.79; HREI-MS: m/z calcd for C34H30N4O3 [M]+ 542.2318; found 542.2321; anal. calcd for C34H30N4O3, C, 75.26; H, 5.57; N, 10.33; found C, 75.27; H, 5.56; N, 10.31.
3.4.1.20. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(3-chlorobenzylidene)benzohydrazide (20). Red solid. Yield: 81.7%. mp: 275–277 °C. IR (cm−1, ATR): 3406, 3293, 3153, 1623, 1543, 1467, 1280, 757; 1H NMR (500 MHz, DMSO-d6): δ 11.89 (s, 1H), 8.41 (s, 1H), 7.82 (d, J = 7.7 Hz, 2H), 7.77 (s, 1H), 7.68 (s, 1H), 7.54–7.47 (m, 4H), 7.40 (d, J = 8.2 Hz, 2H), 7.33 (d, J = 7.9 Hz, 1H), 7.13 (t, J = 7.2 Hz, 2H), 6.93 (t, J = 7.2 Hz, 2H), 6.88 (s, 2H), 5.95 (s, 1H), 3.72 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.31, 148.58, 142.54, 141.68, 141.68, 135.32, 134.18, 134.14, 130.45, 130.45, 130.26, 129.57, 129.57, 128.65, 128.29, 128.29, 128.15, 128.15, 127.68, 125.23, 124.91, 124.91, 123.59, 123.59, 122.65, 122.65, 111.51, 111.51, 111.16, 111.16, 38.46, 35.72, 35.72; HREI-MS: m/z calcd for C33H27ClN4O [M]+ 530.1873; found 530.1877; anal. calcd for C33H27ClN4O, C, 74.64; H, 5.12; N, 10.55; found C, 74.65; H, 5.09; N, 10.53.
3.4.1.21. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(2-hydroxy-5-methoxybenzylidene)benzohydrazide (21). Brown solid. Yield: 91.4%. mp: 263–265 °C. IR (cm−1, ATR): 3273, 3120, 3021, 1608, 1589, 1309, 1256, 747; 1H NMR (500 MHz, DMSO-d6): δ 11.99 (s, 1H), 10.71 (s, 1H), 8.59 (s, 1H), 7.84 (d, J = 7.9 Hz, 2H), 7.52 (d, J = 8.0 Hz, 2H), 7.40 (d, J = 8.2 Hz, 2H), 7.34 (d, J = 7.9 Hz, 2H), 7.15–7.11 (m, 3H), 6.95–6.92 (m, 2H), 6.89 (s, 2H), 6.86 (d, J = 9.0 Hz, 1H), 5.95 (s, 1H), 3.74 (s, 3H), 3.73 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.26, 154.31, 154.34, 149.71, 142.56, 141.13, 134.25, 130.53, 129.35, 128.47, 128.21, 124.96, 123.56, 122.43, 121.78, 116.67, 116.23, 112.86, 111.38, 111.18, 56.25, 38.48, 35.94; HREI-MS: m/z calcd for C34H30N4O3 [M]+ 542.2318; found 542.2314; anal. calcd for C34H30N4O3, C, 75.26; H, 5.57; N, 10.33; found C, 75.27; H, 5.58; N, 10.31.
3.4.1.22. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(2-nitrobenzylidene)benzohydrazide (22). Light yellow solid. Yield: 72.5%. mp: 232–233 °C. IR (cm−1, ATR): 3422, 3382, 3242, 1609, 1546, 1466, 1273, 752; 1H NMR (500 MHz, DMSO-d6): δ 12.12 (s, 1H), 8.85 (s, 1H), 8.14 (d, J = 6.9 Hz, 1H), 8.08 (d, J = 8.2 Hz, 1H), 7.84 (d, J = 7.8 Hz, 2H), 7.71–7.64 (m, 1H), 7.52 (d, J = 8.1 Hz, 2H), 7.40 (d, J = 8.2 Hz, 2H), 7.34 (d, J = 7.9 Hz, 2H), 7.13 (t, J = 7.4 Hz, 2H), 6.94 (t, J = 7.4 Hz, 2H), 6.89 (s, 1H), 5.96 (s, 1H), 3.72 (s, 6H); 13C NMR (150 MHz, DMSO-d6): δ 164.39, 148.03, 142.84, 142.62, 141.47, 141.47, 134.18, 133.79, 130.49, 130.31, 130.31, 129.43, 129.43, 129.38, 129.38, 128.16, 128.16, 128.07, 128.07, 125.16, 124.69, 124.69, 123.42, 123.42, 122.71, 122.71, 111.69, 111.69, 111.18, 111.18, 38.32, 35.81, 35.81; HREI-MS: m/z calcd for C33H27N5O3 [M]+ 541.2114; found 541.2110; anal. calcd for C33H27N5O3, C, 73.18; H, 5.02; N, 12.93; found C, 73.17; H, 5.03; N, 12.91.
3.4.1.23. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(2,4-dihydroxybenzylidene)benzohydrazide (23). Brown solid. Yield: 72.5%. mp: 262–264 °C. IR (cm−1, ATR): 3431, 3255, 3023, 1613, 1589, 1543, 1519, 1468, 1274, 755; 1H NMR (500 MHz, DMSO-d6): δ 11.82 (s, 1H), 11.49 (s, 1H), 9.93 (s, 1H), 8.47 (s, 1H), 7.82 (d, J = 7.9 Hz, 2H), 7.51 (d, J = 8.0 Hz, 2H), 7.40 (d, J = 8.2 Hz, 2H), 7.33 (d, J = 7.9 Hz, 2H), 7.28 (d, J = 8.5 Hz, 1H), 7.13 (t, J = 7.6 Hz, 2H), 6.93 (t, J = 7.5 Hz, 2H), 6.88 (s, 2H), 6.36 (d, J = 8.3 Hz, 1H), 6.32 (s, 1H), 5.95 (s, 1H), 3.72 (s, 6H); 13C NMR (150 MHz, DMSO-d6): 164.36, 160.58, 160.52, 151.66, 142.09, 141.53, 141.53, 134.86, 130.35, 130.21, 130.21, 129.21, 129.21, 128.53, 128.53, 128.34, 128.34, 124.46, 124.46, 123.89, 123.89, 122.85, 122.85, 113.19, 111.58, 111.58, 111.18, 111.18, 109.25, 103.14, 38.43, 35.94, 35.94; HREI-MS: m/z calcd for C33H28N4O3 [M]+ 528.2161; found 528.2161; anal. calcd for C33H28N4O3, C, 74.98; H, 5.34; N, 10.60; found C, 74.98; H, 5.34; N, 10.60.
3.4.1.24. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(3-methylbenzylidene)benzohydrazide (24). Dark brown solid. Yield: 84.8%. mp: 254–256 °C. IR (cm−1, ATR): 3539, 3252, 3075, 3143, 1640, 1564, 1469, 1342, 1274, 1145, 732; 1H NMR (500 MHz, DMSO-d6): δ 11.76 (s, 1H), 8.40 (s, 1H), 7.82 (d, J = 7.9 Hz, 2H), 7.51 (d, J = 7.9 Hz, 2H), 7.40 (d, J = 8.2 Hz, 2H), 7.34 (d, J = 7.9 Hz, 2H), 7.28 (s, 1H), 7.13 (t, J = 7.6 Hz, 2H), 6.94 (t, J = 7.5 Hz, 2H), 6.89 (s, 1H), 5.95 (s, 1H), 3.81 (s, 3H), 3.73 (s, 6H); 13C-NMR (150 MHz, DMSO-d6): δ 164.52, 148.78, 142.56, 141.52, 141.52, 137.89, 136.76, 134.31, 130.28, 130.28, 129.61, 129.33, 129.33, 128.47, 128.47, 128.13, 128.13, 128.09, 127.89, 124.53, 124.53, 123.51, 123.51, 123.26, 122.71, 122.71, 111.58, 111.58, 111.04, 111.04, 38.39, 35.86, 35.86, 21.18; HREI-MS: m/z calcd for C34H30N4O [M]+ 510.2420; found 510.2423; anal. calcd for C34H30N4O, C, 79.97; H, 5.92; N, 10.97; found C, 79.98; H, 5.94; N, 10.96.
3.4.1.25. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(2,4,6-trihydroxybenzylidene)benzohydrazide (25). Dark red solid. Yield: 89.4%; mp: 313–315 °C, IR (cm−1, ATR): 3413, 1466, 1341, 3051, 1657, 1519, 1269, 1113, 756; 1H NMR (500 MHz, DMSO-d6): δ 11.77 (s, 1H), 11.07 (s, 1H), 9.77 (s, 1H), 8.77 (s, 1H), 7.83 (d, J = 7.8 Hz, 2H), 7.50 (d, J = 8.0 Hz, 2H), 7.39 (d, J = 8.3 Hz, 2H), 7.33 (d, J = 8.0 Hz, 2H), 7.13 (t, J = 7.6 Hz, 2H), 6.93 (t, J = 7.4 Hz, 2H), 6.88 (s, 1H), 5.94 (s, 1H), 5.84 (s, 1H), 3.72 (s, 6H); 13C-NMR (150 MHz, DMSO-d6): δ 163.92, 163.12, 161.47, 161.47, 144.68, 142.35, 141.47, 141.47, 134.18, 130.25, 130.25, 129.50, 129.50, 128.38, 128.38, 128.09, 128.09, 124.69, 124.69, 123.56, 123.56, 122.85, 122.85, 111.69, 111.69, 111.18, 111.18, 106.29, 95.87, 95.87, 38.32, 35.86, 35.86; HREI-MS: m/z calcd for C33H28N4O4 [M]+ 544.2111; found 544.2108; anal. calcd for C33H28N4O4, C, 72.78; H, 5.18; N, 10.29; found C, 72.79; H, 5.15; N, 10.26.
3.4.1.26. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(4-chlorobenzylidene)benzohydrazide (26). Red solid. Yield: 88.3%; mp: 237–239 °C, IR (cm−1, ATR): 3434, 3232, 3019, 1633, 1514, 1469, 1233, 1187, 752; 1H NMR (500 MHz, DMSO-d6): δ 11.82 (s, 1H), 8.41 (s, 1H), 7.82 (d, J = 8.1 Hz, 2H), 7.74 (d, J = 7.2 Hz, 2H), 7.51 (t, J = 8.0 Hz, 4H), 7.40 (d, J = 8.3 Hz, 2H), 7.33 (d, J = 7.9 Hz, 2H), 7.13 (t, J = 7.6 Hz, 2H), 6.93 (t, J = 7.4 Hz, 2H), 6.88 (s, 2H), 5.95 (s, 1H), 3.72 (s, 6H); 13C-NMR (150 MHz, DMSO-d6): δ 164.42, 149.37, 142.56, 141.51, 141.51, 135.23, 134.82, 134.20, 130.37, 130.37, 129.51, 129.51, 129.48, 129.48, 129.16, 129.16, 128.31, 128.31, 128.13, 128.13, 124.97, 124.97, 123.47, 123.47, 122.85, 122.85, 111.58, 111.58, 111.09, 111.09, 38.43, 35.83, 35.83; HREI-MS: m/z calcd for C33H27ClN4O [M]+ 530.1873; found 530.1877; anal. calcd for C33H27ClN4O, C, 74.64; H, 5.12; N, 10.55; found C, 74.66; H, 5.13; N, 10.57.
3.4.1.27. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(2-chlorobenzylidene)benzohydrazide (27). Red solid. Yield: 79.4%; mp: 247–249 °C, IR (cm−1, ATR): 3119, 3026, 2828, 1658, 1539, 1449, 1269, 745; 1H NMR (500 MHz, DMSO-d6): δ 11.99 (s, 1H), 8.84 (s, 1H), 8.02 (s, 1H), 7.84 (d, J = 7.7 Hz, 2H), 7.52 (t, J = 7.3 Hz, 2H), 7.45 (d, J = 4.0 Hz, 2H), 7.40 (d, J = 8.2 Hz, 2H), 7.33 (d, J = 7.9 Hz, 2H), 7.13 (t, J = 7.4 Hz, 2H), 6.94 (t, J = 7.4 Hz, 2H), 6.89 (s, 2H), 5.95 (s, 1H), 3.72 (s, 6H); 13C-NMR (150 MHz, DMSO-d6): δ 164.48, 149.92, 142.32, 141.58, 141.58, 134.31, 132.59, 131.20, 130.93, 130.37, 130.37, 129.73, 129.73, 128.69, 128.69, 128.41, 128.41, 128.20, 128.20, 127.73, 124.92, 124.92, 123.51, 123.51, 122.79, 122.79, 111.58, 111.58, 111.28, 111.28, 38.38, 35.72, 35.72; HREI-MS: m/z calcd for C33H27ClN4O [M]+ 530.1873; found 530.1871; anal. calcd for C33H27ClN4O, C, 74.64; H, 5.12; N, 10.55; found C, 74.62; H, 5.10; N, 10.56.
3.4.1.28. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(3-fluorobenzylidene)benzohydrazide (28). Light orange solid. Yield: 81.9%; mp: 263–264 °C, IR (cm−1, ATR): 3421, 3282, 3061, 1660, 1608, 1519, 1466, 1360, 1272, 752; 1H NMR (500 MHz, DMSO-d6): δ 11.86 (s, 1H), 8.43 (s, 1H), 7.82 (d, J = 7.5 Hz, 2H), 7.52 (t, J = 13.1 Hz, 3H), 7.40 (d, J = 8.2 Hz, 2H), 7.33 (d, J = 7.8 Hz, 2H), 7.13 (t, J = 7.6 Hz, 2H), 6.93 (t, J = 7.5 Hz, 2H), 6.88 (s, 2H), 5.95 (s, 1H), 3.72 (s, 6H); 13C-NMR (150 MHz, DMSO-d6): δ 164.51, 162.78 (d, J = 261.5 Hz), 148.79, 142.46, 141.51, 141.51, 138.63, 134.31, 130.37, 130.37, 129.87, 129.43, 129.43, 128.31, 128.31, 128.12, 128.12, 124.91, 123.51, 123.51, 122.74, 122.05, 117.14, 116.92, 114.35, 114.19, 111.58, 111.58, 111.09, 111.09, 38.54, 35.91, 35.91; HREI-MS: m/z calcd for C33H27FN4O [M]+ 530.1873; found 530.1869; anal. calcd for C33H27FN4O, C, 77.02; H, 5.29; N, 10.89; found C, 77.01; H, 5.31; N, 10.90.
3.4.1.29. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(3-hydroxybenzylidene)benzohydrazide (29). Brown solid. Yield: 72.4%; mp: 233–234 °C, IR (cm−1, ATR): 3312, 3179, 2833, 1633, 1609, 1505, 1467, 1253, 1141, 747; 1H NMR (500 MHz, DMSO-d6): δ 11.69 (s, 1H), 9.92 (s, 1H), 9.60 (s, 1H), 8.33 (s, 1H), 7.81 (d, J = 8.0 Hz, 2H), 7.50 (d, J = 8.1 Hz, 2H), 7.40 (d, J = 8.3 Hz, 2H), 7.33 (d, J = 7.9 Hz, 2H), 7.25 (t, J = 7.8 Hz, 2H), 7.19 (s, 1H), 7.16–7.09 (m, 3H), 6.93 (t, J = 7.5 Hz, 2H), 6.88 (s, 2H), 5.95 (s, 1H), 3.72 (s, 6H); 13C-NMR (150 MHz, DMSO-d6): δ 164.52, 156.37, 148.69, 142.51, 141.40, 141.40, 137.07, 134.31, 130.72, 130.38, 130.38, 129.52, 129.52, 128.31, 128.31, 128.15, 128.15, 124.91, 124.91, 123.56, 123.56, 122.73, 122.73, 120.07, 119.35, 114.82, 111.48, 111.48, 111.18, 111.18, 38.40, 35.92, 35.92; HREI-MS: m/z calcd for C33H28N4O2 [M]+ 512.2212; found 512.2216; anal. calcd for C33H28N4O2, C, 77.32; H, 5.51; N, 10.93; found C, 77.30; H, 5.52; N, 10.94.
3.4.1.30. (E)-4-(Bis(1-methyl-1H-indol-3-yl)methyl)-N′-(pyridin-4-ylmethylene)benzohydrazide (30). Red solid. Yield: 82.6%; mp: 253–254 °C, IR (cm−1, ATR): 3423, 3352, 3138, 1657, 1611, 1269; 1H NMR (500 MHz, DMSO-d6): δ 12.02 (s, 1H), 8.65 (d, J = 4.2 Hz, 2H), 8.42 (s, 1H), 7.83 (d, J = 8.0 Hz, 2H), 7.66 (s, 2H), 7.52 (d, J = 8.1 Hz, 2H), 7.40 (d, J = 8.2 Hz, 2H), 7.34 (d, J = 7.9 Hz, 2H), 7.13 (t, J = 7.6 Hz, 2H), 6.94 (t, J = 7.5 Hz, 2H), 6.89 (s, 2H), 5.96 (s, 1H), 3.72 (s, 6H); 13C-NMR (150 MHz, DMSO-d6): δ 164.52, 150.19, 150.19, 149.46, 142.58, 141.79, 141.79, 140.75, 134.31, 130.38, 130.38, 129.63, 129.63, 128.32, 128.32, 128.20, 128.20, 124.93, 124.93, 123.56, 123.56, 122.71, 122.71, 122.45, 122.45, 111.98, 111.98, 111.18, 111.18, 38.52, 35.79, 35.79; HREI-MS: m/z calcd for C32H27N5O [M]+ 497.2216; found 497.2221; anal. calcd for C32H27N5O, C, 77.24; H, 5.47; N, 14.07; found C, 77.25; H, 5.45; N, 14.06.
3.5. Cytotoxicity assays using 3T3-L1 and CC-1 cell lines
In vitro cytotoxicity assays were performed as described in Taha et al. 2015,35 using the 3T3-L1 mouse embryo fibroblast cell line (American Type Culture Collection ‘ATCC’, Manassas, VA 20108, USA), and CC-1 cells, a rat Wistar hepatocyte cell line (European Collection of Cell Cultures, Salisbury, UK). The CC-1 cells were suspended in Minimum Essential Medium Eagle (MEM) supplemented with 10% FBS, 2 mM glutamine, 1% non-essential amino acids, and 20 mM HEPES. The 3T3-L1 cells were suspended in Dulbecco's Modified Eagle's Medium (DMEM) formulated with 10% FBS. Using flat-bottomed plates, both cell lines were plated at a concentration of 6 × 104 cells per mL and incubated for 24 h at 37 °C in a 5% CO2 environment. After removal of the media, the cells were challenged with three different concentrations (1.0, 5.0, and 20 μg mL−1) of compounds in triplicates and were then further incubated for 48 h at 37 °C in a CO2 incubator. Following exposure to each compound, the cells' viability was assessed using 0.5 mg mL−1 of MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) for 4 h, followed by removal of the supernatant and the addition of DMSO to solubilize the formazan complex. Plates were read at 540 nm after one minute shaking and the readings were processed using the MS Excel software. The results were expressed as means ± SD of the triplicate readings.3.6. Docking studies
The structures of all the compounds were prepared using Chem3D by CambridgeSoft. The human β-D-glucuronidase crystal structure was first retrieved from the protein data bank (PDB code: 1BHG).36 Docking was carried out using AutoDock 4.2.37 Using the Genetic Algorithm (GA) search parameter, the number of runs was set at 150, whereas the other settings were left as the default. The docking parameter was also left at its default settings. The 3D docking results were visualized using Discovery Studio Visualizer 4.1.3.7. DFT studies
The geometry optimization and frequency calculations of the synthesized novel bisindolylmethane derivatives were carried out at the B3LYP/6-31+G(d,p) level of theory,40 as implemented in the Gaussian 09 package.41 The optimized minima were confirmed by the absence of imaginary frequencies. The solvent effects were taken into account implicitly using an integral equation formalism (IEF) version of the polarizable continuum model (PCM). In PCM, the molecule (e.g., the bisindolylmethane derivative) is embedded into a cavity surrounded by the solvent, which is described by its dielectric constant ε.424. Conclusions
We synthesized novel bisindolylmethane derivatives containing a benzohydrazone moiety. In conclusion, molecular docking studies evidently demonstrated the interaction pattern of the synthetic compounds in correspondence with a biological inhibitory assay. Analysis of the binding mode clearly showed the presence of a hydrophilic group, i.e., that the hydroxyl moiety plays a key role in the activity profile. It was also observed that the compounds are positioned in the binding site in such a way that two or more hydroxyl groups have to be substituted on the carbon adjacent to each other for good interaction to take place with important residues like Glu451 and Glu540, compared to hydroxyls that are substituted far apart.Acknowledgements
The authors would like to acknowledge Universiti Teknologi MARA for the financial support under the Research Intensive Faculty grant scheme with reference number UiTM 600-RMI/DANA 5/3/RIF (347/2012).References
- X.-H. Gu, X.-Z. Wan and B. Jiang, Bioorg. Med. Chem. Lett., 1999, 9, 569 CrossRef CAS PubMed.
- C. Bonnesen, I. M. Eggleston and J. D. Hayes, Cancer Res., 2001, 61, 6120 CAS.
- Indoles: Part I, ed. W. Houlihan, W. Remers and R. Brown, 1992 Search PubMed.
- R. J. Sundberg, The Chemistry of Indoles, New York, Academic Press, 1996 Search PubMed.
- A. Casapullo, G. Bifulco, I. Bruno and R. Riccio, J. Nat. Prod., 2000, 63, 447 CrossRef CAS.
- B. Bao, Q. Sun, X. Yao, J. Hong, C.-O. Lee, C. J. Sim, K. S. Im and J. H. Jung, J. Nat. Prod., 2005, 68, 711 CrossRef CAS PubMed.
- L. Gupta, A. Talwar, S. Palne, S. Gupta and P. M. Chauhan, Bioorg. Med. Chem. Lett., 2007, 17, 4075 CrossRef CAS PubMed.
- K. Kaniwa, M. A. Arai, X. Li and M. Ishibashi, Bioorg. Med. Chem. Lett., 2007, 17, 4254 CrossRef CAS PubMed.
- S.-L. Zhu, S.-J. Ji, X.-M. Su, C. Sun and Y. Liu, Tetrahedron Lett., 2008, 49, 1777 CrossRef CAS.
- G. La Regina, A. Coluccia, F. Piscitelli, A. Bergamini, A. Sinistro, A. Cavazza, G. Maga, A. Samuele, S. Zanoli and E. Novellino, J. Med. Chem., 2007, 50, 5034 CrossRef CAS PubMed.
- S. Routier, P. Peixoto, J.-Y. Mérour, G. Coudert, N. Dias, C. Bailly, A. Pierré, S. Léonce and D.-H. Caignard, J. Med. Chem., 2005, 48, 1401 CrossRef CAS PubMed.
- A. Andreani, S. Burnelli, M. Granaiola, A. Leoni, A. Locatelli, R. Morigi, M. Rambaldi, L. Varoli, L. Landi and C. Prata, J. Med. Chem., 2008, 51, 4563 CrossRef CAS PubMed.
- V. S. Velezheva, P. J. Brennan, V. Y. Marshakov, D. V. Gusev, I. N. Lisichkina, A. S. Peregudov, L. N. Tchernousova, T. G. Smirnova, S. N. Andreevskaya and A. E. Medvedev, J. Med. Chem., 2004, 47, 3455 CrossRef CAS PubMed.
- K. E. Knott, S. Auschill, A. Jäger and H.-J. Knölker, Chem. Commun., 2009, 1467 RSC.
- M. I. Rodríguez-Franco, M. I. Fernández-Bachiller, C. Pérez, B. Hernández-Ledesma and B. Bartolomé, J. Med. Chem., 2006, 49, 459 CrossRef PubMed.
- K. V. Sashidhara, A. Kumar, M. Kumar, A. Srivastava and A. Puri, Bioorg. Med. Chem. Lett., 2010, 20, 6504 CrossRef CAS PubMed.
- S. H. Benabadji, R. Wen, J.-B. Zheng, X.-C. Dong and S.-G. Yuan, Acta Pharmacol. Sin., 2004, 25, 666 CAS.
- K. Sujatha, P. T. Perumal, D. Muralidharan and M. Rajendran, Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem., 2009, 48, 267 Search PubMed.
- R. Kato and T. Kamataki, Drug Metabolism, Tokyo Kagaku Dojin, Tokyo, 1996, in Japanese Search PubMed.
- G. A. Levvy and J. Conchie, β-glucuronidase and the hydrolysis of glucuronides, glucuronic acid, ed. G. J. Dutton, Academic Press, New York, 1966, pp. 301–364 Search PubMed.
- S. Yasuoka, Rinsho Kensa Mook, 1982, 11, 183 Search PubMed.
- H. R. Nawaz, A. Malik, P. M. Khan, S. Shujaat and A. Rahman, Chem. Pharm. Bull., 2000, 48, 1771 CrossRef CAS PubMed.
- S. B. Shim, N. J. Kim and D. H. Kim, Planta Med., 2000, 66, 40 CrossRef CAS PubMed.
- D. H. Ki, B. Shim, N. J. Kim and I. S. Jang, Biol. Pharm. Bull., 1999, 22, 162 Search PubMed.
- T. Hayashi, M. Kawasaki, K. Okamura, Y. Tamada and N. Morita, J. Nat. Prod., 1992, 55, 1748 CrossRef CAS.
- D. B. I. Cenci, P. Dorling, L. Fellows and B. Winchester, FEBS Lett., 1984, 176, 61 CrossRef.
- J. Waqas, P. Shagufta, A. A. S. Syed, T. Muhammad, H. I. Nor, P. Shahnaz, A. Nida, M. K. Khalid and I. C. Muhammad, Molecules, 2014, 19, 8788 CrossRef PubMed.
- S. Imran, M. Taha, N. H. Ismail, S. Fayyaz, K. M. Khan and M. I. Choudhary, Bioorg. Chem., 2015, 62, 83 CrossRef CAS PubMed.
- M. K. Khan, F. Rahim, N. Ambreen, M. Taha, M. Khan, H. Jahan, A. Shaikh, S. Iqbal, S. Perveen and I. M. Choudhary, Med. Chem., 2013, 9, 588 CrossRef.
- K. M. Khan, M. Taha, M. Ali and S. Perveen, Lett. Org. Chem., 2009, 6, 319 CrossRef CAS.
- K. M. Khan, F. Rahim, A. Wadood, M. Taha, M. Khan, S. Naureen, N. Ambreen, S. Hussain, S. Perveen and M. I. Choudhary, Bioorg. Med. Chem. Lett., 2014, 24, 1825 CrossRef CAS PubMed.
- Bioactive Natural Products Part E, ed. Atta-ur-Rahman, Elsevier, Amsterdam, 2000, vol. 24, p. 333 Search PubMed.
- (a) B. P. Bandgar and K. A. Shaikh, Tetrahedron Lett., 2003, 44, 1959 CrossRef CAS; (b) K. M. Khan, M. Taha, F. Rahim, W. Jamil, S. Perveen and M. I. Choudhary, J. Iran. Chem. Soc., 2012, 9, 81 CrossRef CAS; (c) S. A. Mulla, A. Sudalai, M. Y. Pathan, S. A. Siddique, S. M. Inamdar, S. S. Chavan and R. S. Reddy, RSC Adv., 2012, 2, 3525 RSC; (d) X. F. Xu, Y. Xiong, X. G. Ling, X. M. Xie, J. Yuan, S. T. Zhang and Z. R. Song, Chin. Chem. Lett., 2014, 25, 406 CrossRef CAS.
- K. M. Khan, A. Karim, S. Saied, N. Ambreen, X. Rustamova, S. Naureen, S. Mansoor, M. Ali, S. Perveen, M. I. Choudhary and G. A. Morales, Mol. Diversity, 2014, 18, 295 CrossRef CAS PubMed.
- M. Taha, N. H. Ismail, S. Imran, A. Wadood, F. Rahim, M. Ali and A. U. Rehman, MedChemComm, 2015, 6, 1826 RSC.
- S. Jain, W. B. Drendel, Z. W. Chen, F. S. Mathews, W. S. Sly and J. H. Grubb, Nat. Struct. Biol., 1996, 3, 375 CrossRef CAS PubMed.
- G. M. Morris, R. Huey, W. Lindstrom, M. F. Sanner, R. K. Belew, D. S. Goodsell and A. J. Olson, J. Comput. Chem., 2009, 16, 2785 CrossRef PubMed.
- K. M. Khan, F. Rahim, S. A. Halim, M. Taha, M. Khan, S. Perveen, Z. -ul-Haq, M. A. Mesaik and M. I. Choudhary, Bioorg. Med. Chem., 2011, 19, 4286 CrossRef CAS PubMed.
- (a) O. Lauren and M. H. Court, J. Pharmacogenomics Pharmacoproteomics, 2008, 60, 1175 Search PubMed; (b) K. M. Khan, S. M. Saad, N. N. Shaikh, S. Hussain, M. I. Fakhri, S. Perveen, M. Taha and M. I. Choudhary, Bioorg. Med. Chem., 2014, 22, 3449 CrossRef CAS PubMed; (c) K. M. Khan, N. Ambreen, M. Taha, S. A. Halim, Z. -ul-Haq, S. Naureen, S. Rasheed, S. Perveen, S. Ali and M. I. Choudhary, J. Comput.-Aided Mol. Des., 2014, 28, 577 CrossRef CAS PubMed; (d) N. K. N. Z. Abdullah, M. Taha, N. Ahmat, A. Wadood, N. H. Ismail, F. Rahim, M. Ali, N. Abdullah and K. M. Khan, Bioorg. Med. Chem., 2015, 23, 3119 CrossRef PubMed.
- A. D. Becke, Density-functional thermochemistry. III. The role of exact exchange, J. Chem. Phys., 1993, 98, 5648–5652 CrossRef CAS.
- M. Andersson and P. Uvdal, New scale factors for harmonic vibrational frequencies using the B3LYP density functional method with the triple-ζ basis set 6-311+G(d,p), J. Phys. Chem. A, 2005, 109, 2937–2941 CrossRef CAS PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. F. J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr, J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 09, Revision A.02, 2009 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra19513f |
| This journal is © The Royal Society of Chemistry 2016 |