Supercritical synthesis of a magnetite-reduced graphene oxide hybrid with enhanced adsorption properties toward cobalt & strontium ions†
Ahmad Tayyebi* and
Mohammad Outokesh*
Department of Energy Engineering, Sharif University of Technology, Azadi Ave. P. O. Box: 113658639, Tehran, Iran. E-mail: Outokesh@sharif.edu; atayebi162@gmail.com
First published on 22nd January 2016
Abstract
The current study presents a supercritical synthesis of magnetite-reduced graphene oxide (M-RGO) in methanol media, in which Fe3O4 nanoparticles are simultaneously formed, surface modified and decorated on the surface of the reduced graphene oxide. Simulations using density functional theory, which were performed using the M06-2x/cc-pVDZ level of theory, indicate that upon adsorption of a Fe3O4 cluster on the graphene, the overall charge on the graphene surface becomes about −0.0236e, indicating charge transfer from the Fe3O4 cluster to the graphene surface. Instrumental and chemical analyses exhibited the formation of strong bonds between Fe3O4 and graphene, through C–O–Fe and C–Fe bridges. Based on this data the study puts forward a formation mechanism for M-RGO. The adsorption behavior of the M-RGO towards Co2+ and Sr2+ ions demonstrated an appreciably higher uptake capacity than that of magnetite-graphene oxide (M-GO), a zero zeta potential point at pH ≈ 2, endothermic and spontaneous adsorption, and fast kinetics that was controlled by the chemical reaction between the ions and the surface active sites of the Fe3O4 nanoparticles. The material also showed good reusability, quick elution by 0.5 mol dm−3 HCl, and most importantly, simple separation from the solution using a magnet. Overall, the M-RGO appears to be a promising alternative for the current magnetite-graphene oxide hybrids in the adsorption of heavy metal ions.
Introduction
In recent decades, water pollution has been one of the most important millstones around every society’s neck due to the shortage of clean water resources. This pollution, which has arisen from the deliberate disposal of organic–inorganic effluents (e.g. toxic metal ions) into fresh water, has resulted in a worldwide environmental concern, and serious health problems.1As for the treatment of the contaminated water, so far, various methods have been developed including among others: chemical precipitation, membrane technology, photocatalytic processes and adsorption.2
Adsorption is a fast, inexpensive, and universal method for the treatment and purification of water. Consequently, so far, a great number of adsorbents have been tested and commercially exploited in water treatment processes, namely activated carbon, inorganic adsorbents (e.g. zeolites), and ion exchange resins.3 Despite such efforts, the development of new, efficient and low-cost adsorbents is still favored by the water treatment industries. In particular, the demand for an available and low cost adsorbent of heavy metal ions, that encompasses both a large surface area and a simple functionalization process is quite high.
When one seeks a huge surface area and abundant functional groups, graphene oxide (GO) arises as one of the best candidates. Graphene oxide is a single sheet of carbon atoms which hosts a great number of oxygen functionalities such as epoxy (–O–) and hydroxyl (–OH) groups in its inner area,4 and carboxylic groups at its edges.5 The presence of these functionalities makes the surface of GO an ideal template for the nucleation and growth of useful nanoparticles (NPs) such as Au,6 Ag,7 Pt,8 TiO2,9 and Fe3O4.10 Among these graphene-based nanomaterials, magnetite–graphene hybrids have been recognized as promising materials for various applications including lithium-ion batteries,11 drug delivery,12 sensor fabrication,13 MRI contrast enhancement,10 photothermal therapy,14 protective surface coatings,15 and adsorbent synthesis.16
As for the synthesis of magnetite–graphene hybrids, so far a number of methods have been devised including coprecipitation,10 solvothermal,17 hydrothermal,14 and sonochemical techniques.16 However, obstacles in the preparation of magnetite nanoparticles such as the necessity for accurate pH adjustment, preparing an inert atmosphere, controlling the Fe(II)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe(III) molar ratio, and exploiting toxic organic solvents and reducants18 has given rise to the need for an alternative method to circumvent all these hurdles. This new method should additionally support the surface modification of the magnetite NPs, which is a vital stage in improving their adsorption properties.
Fe(III) molar ratio, and exploiting toxic organic solvents and reducants18 has given rise to the need for an alternative method to circumvent all these hurdles. This new method should additionally support the surface modification of the magnetite NPs, which is a vital stage in improving their adsorption properties.
In recent decades, supercritical fluids have attracted much attention as synthesis media owing to their exclusive properties including low viscosity, high diffusivity, negligible surface tension, and environmental benignity.19,20 Such properties are particularly useful for the synthesis of nanoparticles or more generally, nanomaterials.21 Amongst the oxides of iron, hematite (α-Fe2O3) was fabricated in supercritical water,22 while magnetite NPs were reportedly prepared in supercritical alcohols e.g. methanol or ethanol.19 The reducing effect of supercritical alcohols is primarily attributed to the dissociated hydroxide ions (OH−)23 that are formed under supercritical conditions. These hydroxide ions not only cause the formation of metal24,25 or metal oxides with low oxidation states (e.g. Fe3O4, ZnO),19,26 but also are able to reduce graphene oxide nanosheets.27
When extensive investigations demonstrated that the deposition of some NPs on GO brings about enhanced physicochemical properties compared to the pure graphene or pristine NPs (i.e. synergism),28–30 a lot of interest was devoted to the fabrication of GO-NPs hybrids and the elucidation of their properties. Shao et al. showed that the intercalation of NPs between the graphene sheets attenuates the π–π interaction, and in turn prevents the stacking of the graphene layers (i.e. restoration of the graphite structure).31 Other researchers emphasized the role of the metal–O–C connection in the grafting of the metal oxide NPs on the surface of graphene.32,33 Song and co-workers demonstrated that flower like ZnO is connected to the surface of GO through Zn–O–C bonds.32 According to Nethravathi et al. negative oxygen functionalities on the surface of GO sequester the positive Pt4+ ions through the C–O–Pt chemical bond, formed by consecutive transient-complex formation and dehydration steps.8 Despite such great efforts, most of the accomplished research dealt with the connection mechanism onto the GO rather than the reduced GO (RGO). Indeed, in the case of supercritical alcohols, in which NPs are connected to the RGO, less information is available.
The current study aims to elaborate on the following issues:
• The supercritical synthesis of magnetite-reduced graphene oxide (M-RGO) in methanol.
• Finding plausible mechanisms for the surface modification of magnetite NPs and the connection of them onto the graphene surface.
• Elucidation of the adsorption properties of M-RGO for Sr2+ and Co2+ ions, and the comparison with those of magnetite NPs and RGO.
The ultimate goal of this work is to synthesize a new graphene-based adsorbent with potential environmental applications.
Experimental section
Materials and methods
Natural high purity flake graphite, sulfuric acid 98%, hydrochloric acid 37%, hydrogen peroxide 30%, iron(III) nitrate hexahydrate, methanol, sodium nitrate, and potassium permanganate were all purchased from Merck AG (Darmstadt, Germany) and used without purification.Synthesis of graphene oxide (GO)
Graphene oxide (GO) was synthesized from high purity graphite using the modified Hummers and Offeman method.34 In a typical procedure, a mixture of 2 g of graphite powder and 2 g of sodium nitrate was dispersed in 45 cm3 of 98% (w/w) sulfuric acid. While the temperature was maintained below 5 °C using an ice bath, 6 g of potassium permanganate was gradually added to the dispersion within a period of 2 h. Then, the mixture was stirred for 30 min, and after that, 100 cm3 of distilled water was slowly added to it. At this stage, the temperature was increased to 95 °C, and the mixture was kept at such a temperature for 30 min. Afterward, the hot slurry was cooled to 40 °C by adding 300 cm3 of distilled water, and the reaction was terminated using 20 cm3 of a 30% (w/v) hydrogen peroxide solution. The obtained mixture was filtered, washed with dilute (5% w/v) HCl solution to remove the metal ions, and repeatedly washed (with distilled water) and centrifuged until the pH became 5. Finally the produced GO was dried at ambient temperature.Synthesis of magnetite-reduced graphene oxide (M-RGO)
About 0.1 g of graphene oxide (GO) was dispersed in 100 cm3 of methanol (1 mg ml−1) and the dispersion was sonicated using an ultrasonic bath (Elmasonic, S 30H, Branson, CT, USA) until the solution became clear. Next, 10 cm3 of a 2.5 mol l−1 iron nitrate(III) solution was added to the foregoing solution. Then, 15 cm3 of the obtained mixture was poured into a 30 cm3 stainless steel autoclave, and the autoclave was inserted into a furnace to effect the M-RGO synthesis under supercritical conditions. The employed autoclave was able to endure a working temperature and pressure of 500 °C and 550 atm, respectively, but in order to keep an adequate safety margin, it was always loaded with 50% of its capacity (Fig. S1 in ESI†).Due to the small size of the reactor and the intensive rate of heating, the warm up time for surpassing the critical point was quite short and was less than 10 min. The residence time, which is defined as the “heating period at the set temperature point”, was about 15 min. After removing the reaction vessel from the furnace, it was rapidly quenched using cold water and the obtained material was separated from the solution using a magnet. The M-RGO hybrid prepared by this method was transferred to some Petri dishes and dried under a nitrogen atmosphere. Fig. S1 in ESI† presents a schematic illustration of the M-RGO fabrication process. In addition to the M-RGO, we also prepared reduced graphene oxide (RGO) by charging of the GO–methanol solution into the autoclave, and its treatment exactly with the same manner as M-RGO.
Adsorption tests
All of the adsorption tests were carried out in a batchwise fashion. In every test, about 20 mg of adsorbent was charged into a conical flask, which contained 20 cm3 of a Co2+ or Sr2+ solution. Next, the mixture was ultrasonicated for a certain period, after which the M-RGO particles were separated from the solution using a magnet placed behind the vessel wall. Finally, the concentration of the decanted supernatant was measured using atomic adsorption spectrometry (AAnalyst, 800, Perkin-Elmer, USA).In order to investigate the effect of pH on the uptake, the pH of the Co2+ or Sr2+ solution was adjusted to the desired value between 1 and 10, by accurate addition of HCl or NaOH solution. The kinetics of the adsorption and the uptake capacity of the M-RGO were studied by conducting the above-mentioned batch experiments in different time intervals and with different target-ion concentrations. All of the experiments were conducted at pH 6.5, except the ones that investigated the effect of pH. Moreover, the initial concentrations of Sr2+ and Co2+ in all of the experiments were 50 ppm except the ones related to the adsorption isotherms.
In the analysis of the adsorption data, the adsorption capacity (Q), uptake percentage (R, %), fractional attainment to equilibrium (X), and distribution coefficient (Kd) were defined as:
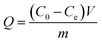 | (1) |
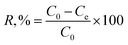 | (2) |
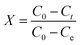 | (3) |
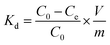 | (4) |
In order to elucidate the adsorption properties of M-RGO in a more detailed fashion, we compared them with those of magnetite NPs and magnetite-graphene oxide hybrids (M-GO) for the adsorption of Co2+ and Sr2+ ions. The required M-GO for this purpose was synthesized using the ultrasound assisted co-precipitation method, which has been described elsewhere.16
Characterization
Morphological microscopic pictures of the GO nanosheets and the M-RGO hybrid were obtained using low and high-resolution transmission electron microscopy (TEM/HRTEM, JEOL, JEM-2100, Japan). The employed HRTEM mode also supported the selected area electron diffraction (SAED) and energy dispersive X-ray (EDX) analysis of the samples. Atomic force microscopy (AFM, Park Scientific CP-Research model, VEECO) was used to find the thickness and morphology of the GO and the M-RGO hybrid. The employed AFM system worked in the tapping mode, with a frequency of 320 kHz, and by using a 20 nm thick silicone tip. Samples for AFM imaging were prepared by drop casting the dilute GO and M-RGO suspensions onto a clean mica substrate.Raman spectroscopy (SENTERRA BRUKER, Germany) was performed on the samples at room temperature using a 785 nm Nd-YAG laser excitation source to scrutinize the change in the chemical and crystalline structure during the synthesis process. Meanwhile, the X-ray photoelectron spectroscopy (XPS) revealed the chemical states of the GO and M-RGO nanosheets. The XPS instrument was equipped with a hemispherical analyzer, a data acquisition system, and an Al-Kα X-ray source (hν = 1486.6 eV) operating at pressure lower than 10−7 Pa. The XPS peaks were deconvoluted using a Gaussian components model after Shirley background subtraction. X-ray diffractometry (Rigaku Miniflex XRD, Texas, USA) and Fourier transform infrared spectroscopy (FTIR, Perkin-Elmer, SpectrumRX, USA) were respectively used for the crystallographic and chemical bond analysis of the prepared materials.
The magnetic hysteresis cycle of the powdered M-RGO was obtained using a QMPS SQUID magnetometer in the field range of −1 T to 1 T at room temperature. The optical characteristics of the diluted GO and M-RGO suspensions (0.01 mg cm−3) were studied using a UV-visible spectrophotometer (Perkin-Elmer UV-Vis-NIR model Lambda 950, USA).
A “Malvern Zetasizer nano ZS” instrument was used to measure the zeta potentials of all the prepared samples at 25 °C. Samples prepared for dynamic light scattering (DLS) measurements were loaded into a pre-rinsed folded capillary cell for zeta potential measurements.
In order to study the thermal stability and composition of the prepared materials, thermo-gravimetric analysis (TGA) was performed under an air atmosphere using a simultaneous thermal analyser (TGA/DSC 1, METTLER TOLEDO, Switzerland). A heating rate of 10 °C min−1 was utilized in those experiments.
Density functional theory calculations
To better understand the nature of the interaction between graphene and the Fe3O4 cluster, we undertook a density functional calculation using circumcoronene (C54H18), composed of 19 aromatic rings, as a model of graphene.35 In our model, all the boundary carbon atoms of the circumcoronene have been saturated with hydrogen atoms (see Fig. 8) and thus, the Fe3O4 cluster just interacts and is sequestered on the carbon atoms of the circumcoronene. Full optimization and property calculations for the adsorption of the Fe3O4 cluster on the graphene surface (circumcoronene model) were done at the M06-2X36 level using the Dunning cc-pVDZ basis set with the Gaussian 03 program.37 All of the calculations were carried out without symmetry restrictions. The harmonic vibrational frequencies and the corresponding zero-point vibrational energies (ZPVEs) were calculated for all of the optimized geometries, and real frequencies were obtained in all cases.Results and discussion
Fig. 1a exhibits an AFM image of the prepared GO, in which the thickness of the sheet corresponds to the height of the white-marked line, depicted in Fig. 1a. The height profile shows two monolayer GO sheets with a thickness of around 0.8 nm. Due to the presence of the oxygen containing groups on both sides of the GO surface, it is broadly accepted that the thickness of a single GO layer is around 0.9 nm which is 0.5 nm thicker than a 0.4 nm typical graphene sheet.38According to Fig. 1b, the UV-vis absorption peak of the GO suspension appears at 227 nm, while for the reduced graphene oxide (RGO), it emerges at 253 nm, indicating a red-shift arising from the electronic conjugation of the graphene sheets, restored upon reduction. For M-RGO the absorption peak appears at 244 nm as a result of the intercalation of magnetite nanoparticles between the graphene sheets that results in the attenuation of the restoration process.
Fig. S2a in ESI† shows the XRD patterns of the synthesized materials in which the characteristic peak of graphite emerges at 2θ = 26 (interlayer space, d ∼ 0.4 nm). As a result of the intercalation of the oxygen functionalities and water, the position of the XRD peak of GO shifted to a lower degree (2θ = 11) corresponding to a larger interlayer space (d ∼ 0.8 nm). The XRD patterns of the reduced graphene oxide (RGO) present a broad and low intensity peak at 2θ = 24 indicating the amorphous nature of the RGO (Fig. S2a†), obtained at the high temperature and pressure conditions of supercritical ethanol.
The XRD pattern of the M-RGO hybrid depicts seven characteristic peaks at 30.1°, 35.4°, 37.1°, 43.1°, 53.4°, 56.9° and 62.5°, that respectively correspond to the (220), (311), (222), (400), (422), (511) and (440) planes of the magnetite (Fe3O4) crystal. No peak for hematite (α-Fe2O3) or any other impurities is detected in the XRD pattern of the M-RGO. In addition, the presence of a broad and low intensity peak at around 2θ = 24° demonstrates the incorporation of graphene sheets in the M-RGO structure.
Fig. S2b in ESI† presents the Raman spectra of the as-prepared materials with a D-band at 1350 cm−1 and a G-band at 1580 cm−1. In the M-RGO sample, 5 characteristic peaks including A1g(1) + Eg(1), Eg(2) + Eg(3), Eg(4), A1g(2) and Eg(5) define the finger print of Fe3O4 NPs at 223.5 cm−1, 289 cm−1, 404 cm−1, 491 cm−1 and 604.5 cm−1, respectively.
Morphology and crystalline structures
It seems to be worthwhile to study the effect of supercritical methanol on the morphology and structure of GO, RGO, and M-RGO. In this regard, low magnification TEM images and SAED patterns of the prepared materials are illustrated in Fig. 2. The wrinkled sheets of GO are depicted in Fig. 2a, in which the transparent regions indicated by the arrows most likely correspond to monolayer graphene sheets. The SAED pattern of GO in the inset of Fig. 2a shows the diffraction spots and resolved ring along the [001] zone axis of GO confirming the hexagonal crystal structure of GO. According to Fig. 2b, increasing the temperature over 350 °C and reaching the supercritical condition leads to the disappearance of diffraction spots and resolved rings in the SAED pattern of GO, as a result of the formation of the reduced graphene (RGO) with a mild crystalline structure. This is in a good agreement with the XRD results (Fig. S2a†) in which RGO exhibits a broad and low intensity peak, corresponding to the amorphous structure. In the case of the M-RGO hybrid, the grafting of Fe3O4 nanoparticles (NPs) onto the graphene surface (Fig. 2c), resulted in the random distribution of diffraction spots between the SAED rings. | ||
| Fig. 2 (a) Low magnification TEM image of GO, (b) RGO, and (c) M-RGO; inset figures show the corresponding SAED patterns of the prepared materials. | ||
Fig. 3 presents more accurate SEM and TEM images of the Fe3O4 NPs that were decorated on the GO surface using supercritical methanol. Oxygen functionalities on the GO surface act as active sites for the nucleation and growth of Fe3O4 NPs. In addition, a large number of oxygen-containing groups means more available space for the deposition of the Fe3O4 NPs, and thus less agglomeration of the magnetite particles. Fig. 3a shows the SEM image of the Fe3O4 NPs on the surface of the graphene sheets. Fig. 3b displays a TEM image of M-RGO, according to which the average size of the magnetite nanoparticles was found to be about 15 nm (Fig. S3†). Elemental analysis of GO, RGO, and M-RGO was further investigated using energy dispersive X-ray analysis (EDX) (Fig. S4 in ESI†), which demonstrated the absence of any elemental impurity other than Fe, C, and O in the M-RGO hybrid, as well as the reductive effect of supercritical methanol on GO.
 | ||
| Fig. 3 (a) SEM image of the M-RGO hybrid, (b) low magnification TEM image of M-RGO, and (c) high resolution TEM image of Fe3O4 NPs synthesized in supercritical methanol. | ||
Fig. 3c shows the HRTEM image of the Fe3O4 NPs whose 0.2 nm inter-planar spacing corresponds to the distance between two (222) planes of the magnetite.
Aggregation of carbon-based nanomaterials such as graphene reduces the surface area of their particles in the aquatic system, and results in reduced adsorption ability. In this respect, we characterized the aqueous dispersions of GO, M-RGO, and RGO using dynamic light-scattering (DLS). As can be seen in Fig. 4, the nominal effective diameters which were estimated using the spherical particle model for GO, M-RGO, and RGO were about 750, 850, and 1150 nm, respectively. Due to the aggregation of GO sheets during their reduction in supercritical methanol (SCMeOH), the average diameter of RGO was 1.5 times larger than that of GO. The deposition of magnetite nanoparticles between graphene layers lowered the aggregation rate of M-RGO, and consequently reduced the average size.
Magnetic properties
Fig. 5 shows the magnetization hysteresis curves of the magnetite NPs and the M-RGO hybrid, synthesized in SCMeOH (the inset photograph shows M-RGO dispersed in aqueous solution (left) and that in the presence of a magnet (right)). As can be seen in the upper inset of Fig. 5, the saturation magnetization (MS), and remanence (MR) for M-RGO are 60 and 12.5 emu g−1, respectively. In comparison with the pure Fe3O4 NPs, the M-RGO hybrid demonstrates lower magnetic intensities due to the presence of the graphene as a diamagnetic material that can reduce the superparamagnetic property of the Fe3O4.Another phenomenon that may stand as a reason for the reduction of the magnetic properties of M-RGO compared to those of pure magnetite is the smaller size of the Fe3O4 NPs that are formed on the surface of the M-RGO. As was reported by Mahmoudi et al. the magnetic saturation decreases appreciably as the size of the Fe3O4 NPs decreases.39 It was demonstrated in the morphology section that the presence of an enormous surface area and oxygen functionalities on the surface of GO causes the formation of Fe3O4 NPs of a very small size in the M-RGO hybrid. In order to have a quantitative evaluation of the size effect, we measured the size distribution of the Fe3O4 NPs in both pure magnetite nanoparticles and M-RGO, and compared them with each other. Diffraction light scattering (DLS) analysis of the pure Fe3O4 NPs in water (Fig. S5 in ESI†) gives an average size of 25 nm. The TEM image of M-RGO (Fig. S3 in ESI†), when analyzed using Microstructure Measurement Software, resulted in a histogram of Fe3O4 NPs on the surface of the graphene sheets from which the average size of the Fe3O4 NPs was obtained as 15 nm. Besides the above direct methods, there is another indirect method for the measurement of the size of the Fe3O4 NPs. This second method is based on the famous Langevin equation, which correlates the average size of the Fe3O4 NPs to their saturated magnetization and magnetic susceptibility as follows:
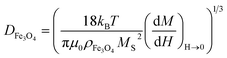 | (5) |
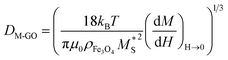 | (6) |
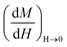 is the susceptibility of the M-RGO at zero field obtained from the magnetization curve (Fig. 5), and finally T = 298 K is the measurement temperature. For the M-RGO composites, the effective saturated magnetization (M*S, eqn (6)) was obtained from the measurement of the Fe3O4 content of its samples using the wet chemical method (M*S = mass percentage of Fe3O4 in the M-RGO hybrid). The wet chemical analysis was performed by dissolving about 0.01 g of the M-RGO hybrid in 20 cm3 of concentrated hydrochloric acid, and measuring its iron content using atomic absorption spectrometry (AAS).16
is the susceptibility of the M-RGO at zero field obtained from the magnetization curve (Fig. 5), and finally T = 298 K is the measurement temperature. For the M-RGO composites, the effective saturated magnetization (M*S, eqn (6)) was obtained from the measurement of the Fe3O4 content of its samples using the wet chemical method (M*S = mass percentage of Fe3O4 in the M-RGO hybrid). The wet chemical analysis was performed by dissolving about 0.01 g of the M-RGO hybrid in 20 cm3 of concentrated hydrochloric acid, and measuring its iron content using atomic absorption spectrometry (AAS).16
Table 1 shows the results of the estimation of the size of the Fe3O4 NPs in pure magnetite and in M-RGO by both direct and Langevin methods. Evidently, there is a fair amount of consistency between the two methods. The accuracy of the modified Langevin equation in the evaluation of the size of Fe3O4 NPs in the magnetite–graphene hybrid, and the application of the wet chemical method in its implementation were previously reported elsewhere.16
| Sample | MS | Mass fraction of Fe3O4 | Dtheoretical (nm) from eqn (5) | Dexperimental (nm) |
|---|---|---|---|---|
| M-RGO | 60 | 0.92 | 19 | 15 (Fig. S3) |
| Magnetite (Fe3O4) | 76 | 1 | 27 | 25 (Fig. S5) |
Chemical composition
XPS analysis was performed to find out the compositions of the GO, RGO, and M-RGO samples. In order to study the XPS results, the binding energies were standardized using the carbon peak at 284.8 eV as a reference peak, and the relative intensity of the other peaks were estimated from the area under the curves by integrating the peak areas.40 The full scan survey of GO and RGO in Fig. 6a shows only two peaks at 285 and 532 eV (which are assigned to the C1s and O1s core levels).41 The intensity of the O1s core level peak of RGO is decreased notably relative to that of GO, as a result of the deoxygenation of the GO in the supercritical methanol. The full scan survey of M-RGO indicates the presence of Fe3p and Fe2p core level peaks that confirms the formation of Fe3O4 NPs (Fig. 6a). The XPS peaks corresponding to the Fe2p core level of Fe3O4 NPs is shown in Fig. S6† in which the Fe2p energy level of the iron atoms is divided into two distinct levels of Fe2p3/2 at 711.8 eV and Fe2p1/2 at 724.4 eV. It should be noted that the energy splitting width is around 12.6 eV that corresponds to a spin–orbit coupling effect, and it is within the standard reference value of the Fe3O4 NPs. | ||
| Fig. 6 (a) The full scan XPS survey of GO, RGO, and M-RGO, and the C1s spectra of (b) GO, (c) RGO, and (d) M-RGO. | ||
Fig. 6b–d show the C1s core level peak deconvolution of GO, RGO, and M-RGO, respectively. The noticeable presence of oxygen functionalities in the structure of GO were indicated in Fig. 6b. The oxygen-containing functional groups were attenuated on the surface of RGO, compared to in GO, as a result of the reducing effect of SCMeOH (Fig. 6b).
As can be seen in Fig. 6b and d, there are two significant differences between the C1s core levels of M-RGO and GO: first, the increase of the C/O ratio in M-RGO through the reduction of GO, and second, the emergence of Fe–C, Fe–O–C, and pi–pi peaks in the M-RGO hybrid, which indicates the formation of Fe3O4 NPs, and their connection to the surface of the graphene via chemical bonding (Fig. 6d).
The O1s core level peak also provides valuable information about the nature of the chemical bonds on the surface of the M-RGO hybrid. As can be seen in Fig. S7 in ESI,† the O1s peak in the M-RGO hybrid is broader than that of GO. This phenomenon is presumably attributed to the change in the number of chemical bonds with oxygen atoms. Indeed, due to some sort of chemical interaction (bonding) between iron or oxygen atoms and the RGO surface, (see “Density Functional Theory” section), the full width at half maximum (FWHM) of O1s in M-RGO (6 eV) is increased compared to that of GO (4.1 eV). To prove the presence of the new chemical bonds in the M-RGO composites, the O1s spectra of GO and M-RGO were deconvoluted and are shown in Fig. 7a and b. In Fig. 7a, the O1s peak of GO includes two spectral peaks at 532.2 and 533.3 eV, that are attributed to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (carbonyl and carboxyl) and C–O (epoxy and hydroxyl) groups, respectively. The O1s spectrum of M-RGO is deconvoluted to four peaks at 530.2, 531.2, 532.2, and 533.3 eV (Fig. 7b). The peaks at 532.2 and 533.3 eV refer to the remaining oxygen functionalities of M-RGO. In addition, the emerging peak at 530.2 is attributed to Fe–O bonds in the crystalline structure of the Fe3O4 NPs. The peak appearing at 531.2 eV is assigned to the C–O–Fe bond formed at the high temperature and pressure conditions of supercritical methanol.
O (carbonyl and carboxyl) and C–O (epoxy and hydroxyl) groups, respectively. The O1s spectrum of M-RGO is deconvoluted to four peaks at 530.2, 531.2, 532.2, and 533.3 eV (Fig. 7b). The peaks at 532.2 and 533.3 eV refer to the remaining oxygen functionalities of M-RGO. In addition, the emerging peak at 530.2 is attributed to Fe–O bonds in the crystalline structure of the Fe3O4 NPs. The peak appearing at 531.2 eV is assigned to the C–O–Fe bond formed at the high temperature and pressure conditions of supercritical methanol.
Proposed formation mechanism
Finding an acceptable mechanism that could account for the simultaneous formation of Fe3O4 NPs, the reduction of GO by methanol, and eventually the interaction of Fe3O4 NPs with graphene or methanol is a formidable task that is evidently out of the scope of the current study. Nevertheless, we tried to summarize the obtained evidence, and address some general guidelines that are helpful for devising such a mechanism in the future. In this regard, first, the interaction of the Fe3O4 NPs with the graphene sheets was investigated using DFT. Then, the interaction of methanol with the Fe3O4 NPs for surface modification was studied.Adsorption of Fe3O4 NPs onto the graphene surface (density functional theory)
In order to find the most stable geometry of the graphene–(Fe3O4) complexes, we placed a Fe3O4 cluster in all possible states on the graphene surface. Then, these structures were optimized at the M06-2X/cc-pVDZ level of theory. The most stable geometry for the adsorption of the Fe3O4 cluster on the graphene surface is displayed in Fig. 8. As is seen in this figure, the sequestration of the Fe3O4 cluster on the graphene surface can take place through either the iron or oxygen atoms in the Fe3O4 structure. | ||
| Fig. 8 The most stable geometry for the adsorption of the (Fe3O4)1 cluster on the graphene surface optimized at the M06-2X/cc-pVDZ level of theory. | ||
Assuming that the Fe and O atoms were 2.430 Å and 3.00 Å away from the graphene surface, the binding energy was determined as the difference between the energy of the “G⋯Fe3O4” complex and the sum of the energies of the graphene surface and the Fe3O4 cluster (ΔEb = E(G⋯(Fe3O4)) − (E(Fe3O4) + E(G))). The counterpoise procedure (CP) by Boys and Bernardi was used to calculate the basis set superposition errors (BSSEs),42 and then the binding energy for the adsorption of Fe3O4 on the graphene surface was corrected using the calculated BSSEs. The calculated binding energy (ΔEb) was about −14.60 kcal mol−1 at the M06-2X/cc-pVDZ level of theory.
The energy difference between the Highest Occupied Molecular Orbital (HOMO) and Lowest Unoccupied Molecular Orbital (LUMO) is termed the HOMO–LUMO energy gap. The magnitude of the HOMO–LUMO energy gap depends on the relative magnitude of the respective orbital energies. The HOMO–LUMO energy gap of the graphene surface in our circumcoronene model was about 4.32 eV. With the adsorption of the (Fe3O4)1 on the graphene surface, the HOMO–LUMO band gap decreased from 4.32 eV to 4.09 eV in the “G⋯Fe3O4” complex. Another useful technique for investigating the changes in the HOMO–LUMO energy gap due to molecular interactions is the DOS. The density of states (DOS) of a system describes the number of states per interval of energy at each energy level that is available for occupation by electrons. The DOS spectra for the graphene surface and the “G⋯Fe3O4” complex is shown in Fig. 9. As seen in this figure, the energies of the HOMO and LUMO orbitals of graphene shift to more positive values upon adsorption of Fe3O4 on the graphene surface. These changes cause a decrease in the HOMO–LUMO energy gap of about 0.23 eV.
In order to evaluate the charge transfer mechanism between the Fe3O4 cluster and the graphene surface, Mulliken population analysis on the optimized “G⋯Fe3O4” complex was performed at the M06-2X/cc-pVDZ level of theory. Upon adsorption of the Fe3O4 cluster on the graphene surface, the overall charge on the graphene surface becomes about −0.0236e, indicating charge transfer from the Fe3O4 cluster to the graphene surface. The bond properties between each pair of atoms were systematically analyzed using the quantum theory of atoms in molecules (QTAIM).43 Based on this theory, the electron density (ρ(r)) value at the bond critical point (BCP) formed between two atoms is used to describe the strength of a bond, and a stronger bond is associated with a larger ρ(r) value. The electron density values at the BCPs formed between the Fe and O atoms of the Fe3O4 cluster and the graphene surface are 0.0293 and 0.0092, respectively. These values show that the bond formed between Fe and C (Fe–C) is stronger than that between O and C (C–O bond). On the other hand, based on Bader’s theory, the binding energies “EFe/O⋯C” were calculated using the equation EFe/O⋯C = 1/2V(r) (V(r) = potential energy density at the BCPs).35 Our calculations showed that the binding energy values for the interaction of the Fe and O atoms of the Fe3O4 cluster with the graphene surface are −9.08 and −1.89 kcal mol−1, respectively.
Surface modification of Fe3O4 NPs using methanol
It is important to figure out the mechanisms for the simultaneous reactions of supercritical methanol with Fe3O4 NPs and GO. In this regard, FTIR analysis was used to support the proposed surface interaction of the magnetite nanoparticles and RGO sheets. As shown in Fig. 10, a broad hydroxyl (–OH) bond stretch appeared on the surface of M-RGO, while no peak is detected in RGO. Thus, the aforementioned peak likely refers to the modification of the Fe3O4 surface with OH groups (Fig. 12a). Furthermore, a doublet peak at around 2900 cm−1 that corresponds to methylene (–CH2–) and methyl (–CH3) asymmetric and symmetric stretching vibrations became stronger in M-RGO compared to that in GO and RGO. In addition, the intensity of a band at around 1037 cm−1 assigned to the –C–O stretching vibration increased in comparison with that in GO and especially RGO. Logically the intensity of the –OH and –C–O stretching peaks should be decreased in M-RGO, as it was attenuated in RGO after the SCMeOH treatment. But the higher intensity of the aforementioned peaks in M-RGO represents the presence of oxygenated functional groups on the surface of the Fe3O4 NPs. The three described pieces of evidence reveal that the surface of the Fe3O4 NPs in M-RGO is covered with hydroxyl (OH) and alkoxide (–C–OH) groups (Fig. 12a). Similar effects of supercritical or near critical alcohols on the surfaces of ZnO, CeO2, Al2O3, TiO2, and ZrO2 nanoparticles were reported previously.44–46 Combining the results of these reports with our own observations will lead us to the outcome that methanol acts as a strong hydroxylation and alkoxylation agent at supercritical conditions, resulting in the surface modification of different NPs.Another useful tool to study the thermal stability of the prepared materials is thermogravimetric analysis. The TG curve (Fig. 11) illustrates three major weight losses for GO. The first one, an 8% weight loss occurring up to 110 °C corresponds to the absorbed water on the GO nanosheets. The second weight loss at 225 °C (31%) is attributed to the loss of CO and CO2 due to the decomposition of the oxygenated functional groups. The third and main weight loss (50%) refers to the oxidation of carbon atoms at high temperatures and in the presence of air. The TG curve of the M-RGO hybrid demonstrates just one major weight loss (16%) which is attributed to the oxidation of carbon atoms and the decomposition of functional groups on the surface of M-RGO. According to the wet chemical method, Fe3O4 NPs comprise about 92% M-RGO (Table 1), and the remaining 8% is mainly made of graphene.
Fig. 12 proposes a few probable mechanisms for the interaction of SCMeOH with the Fe3O4 NPs according to our FTIR, XPS, and TG experiments, and the results of the previous studies. The first step in these mechanisms is the adsorption of methanol on the surface of the Fe3O4 NPs. If the adsorbed methanol acts as an acid, its hydrogen atom can be connected to the oxygen atoms of the Fe3O4 NPs. The disproportionation of H+ from methanol leaves the methoxide group with a negative charge, and forces it to connect to the surface Fe3+ sites of the Fe3O4 (Fig. 12a). The final outcome is the creation of two alkoxide and hydroxide groups on the surface of the Fe3O4 NPs as shown in Fig. 12a and S8a.† According to the FTIR spectra, the carboxylic acid peak (1740 cm−1) disappeared in M-RGO, while a new peak at 1540 cm−1 assigned to the carboxylate emerged. Such a species formation was formerly recognized by Idriss and co-workers.47 Furthermore, according to Zaki and co-workers, with an increase in the temperature of supercritical alcohols, the absorbed alkoxide groups undergo a dehydrogenation process that is followed by demethylation in the next step.48 The aforementioned dehydrogenation–demethylation reactions are completed by the consequent formation of the bridged carboxylate species on the surface of Fe3O4 as shown in Fig. 12b and S8b.† It can be observed in Fig. 12c and S8c† that a chelating type carboxylate is possibly formed using two successive dehydrogenation processes.
 | ||
| Fig. 12 One of the possible pathways for the simultaneous interaction of methanol with the Fe3O4 NP surface. | ||
Adsorption properties
According to Fig. 13a and b, the adsorption capacity of M-RGO for Co2+ ions was about two folds higher than that for the Sr2+ ions, possibly due to the greater affinity of the Fe3O4 NPs toward the Fe(II)-like cations.16 In other words, the close similarity between the cobalt and iron ions allows a more effective sequestering of the Co2+ ions on the Fe3O4 by ion exchange or other mechanisms. To prove this argument, the adsorption ability of pure Fe3O4 NPs toward Sr2+ and Co2+ ions was examined and depicted in Fig. 13a and b. In contrast to the RGO whose maximum adsorption capacity for both Sr2+ and Co2+ ions is nearly identical (Fig. 13a and b), the Fe3O4 NPs show a much higher affinity toward Co2+ ions.
 | ||
| Fig. 13 Adsorption isotherms of (a) Co2+, (b) Sr2+ ions onto RGO, Fe3O4 NPs and the M-RGO hybrid, and the effect of temperature on the adsorption capacity of M-RGO for (c) Co2+, and (d) Sr2+ ions. | ||
The aforementioned Fe3O4 NPs were prepared in SCMeOH media and thus were rich in different functional groups such as hydroxyl, alkoxide, and carboxylate on their surfaces (Fig. 12). The existence of such oxygen functionalities enhances the adsorption capacity of the Fe3O4 NPs appreciably, and gives an evident advantage to those particles in comparison to the Fe3O4 NPs which are prepared using the ultrasound method in aqueous media16 (Fig. S9†).
Another significant phenomenon which is observed in Fig. 13a–d is the higher adsorption capacity of M-RGO in comparison to both the RGO and Fe3O4 NPs. According to the wet chemical analysis and the TG method, the M-RGO nanocomposite comprises 92% wt. Fe3O4 and 8% wt RGO. Thus, it is expected to have an adsorption capacity equal to the arithmetic mean of the adsorption capacities of its constituents. Surprisingly, this is not true, and the actual behavior of M-RGO displays a synergetic effect in the adsorption of Sr2+ and Co2+ ions. This interesting phenomenon can be explained using the discussions of the last paragraph as follows: as it was shown, it is not the body of the Fe3O4 NPs, but indeed their active surfaces that is responsible for the uptake of the metal ions. As a result, any factor that causes agglomeration of the Fe3O4 NPs will lead to a reduction in their uptake ability. The M-RGO and pure Fe3O4 NPs were both synthesized in methanol media, and thus both must have been rich in surface functional groups. However, as the TEM images reveal (Fig. S10†) the pure Fe3O4 NPs are agglomerated, while the M-RGO consists of Fe3O4 NPs that are homogeneously and separately distributed on the surface of the graphene sheet (Fig. S10b†). As was discussed in the morphology section, the huge surface area and numerous active sites of graphene oxide as a starting material causes the homogeneous dispersion of Fe3O4 NPs and inhibits their agglomeration.
In order to achieve more information about the adsorption mechanisms, the Langmuir and Freundlich models were fitted to the experimental data. The Langmuir model is expressed by:
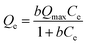 | (7) |
The Freundlich model is represented by the following equation:
| Qe = KCen | (8) |
| T (K) | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| Qmax (mmol g−1) | b (l mmol−1) | R2 | K2 | n | R2 | |
| 298 | 0.71 | 0.21 | 0.99 | 15 | 0.28 | 0.9 |
| 318 | 0.9 | 0.32 | 0.99 | 16.7 | 0.31 | 0.89 |
| 338 | 1.1 | 0.24 | 0.97 | 22 | 0.33 | 0.83 |
| T (K) | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| Qmax (mmol g−1) | b (l mmol−1) | R2 | K2 | n | R2 | |
| 298 | 0.36 | 0.15 | 0.95 | 10 | 0.28 | 0.82 |
| 318 | 0.5 | 0.27 | 0.96 | 16 | 0.26 | 0.84 |
| 338 | 0.6 | 0.64 | 0.95 | 26 | 0.21 | 0.79 |
According to Tables 2 and 3, the higher the temperature is, the greater the adsorption capacity of M-RGO. For the Co2+ and Sr2+ ions, the maximum values achieved for the Qmax were equal to 1.1 and 0.6 mmol g−1, respectively, and they were obtained at 338 K. Interestingly, the maximum adsorption capacity of M-RGO is two times greater than that for M-GO which indicates the positive effect of SCMeOH in increasing the number of the acidic (–COOH) as well as hydroxyl (–OH) groups on the surface of the Fe3O4 NPs which results in a higher affinity toward the metal cations.
Thermodynamic parameters such as ΔG0, ΔH0, and ΔS0 provide valuable information about the endothermic or exothermic nature of the reaction and its degree of spontaneity. These parameters can be estimated from the temperature-dependent uptake data using the following equations:
ΔG0 = −RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K0 K0
| (9) |
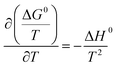 | (10) |
| ΔG0 = ΔH0 − TΔS0 | (11) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kd curves versus Ce (Fig. S11 in ESI†) and extrapolating Ce to zero, ln
Kd curves versus Ce (Fig. S11 in ESI†) and extrapolating Ce to zero, ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K0 values can be obtained for every temperature. The ln
K0 values can be obtained for every temperature. The ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K0 values then are used for the calculation of ΔG0 using eqn (9). Thereafter, the
K0 values then are used for the calculation of ΔG0 using eqn (9). Thereafter, the 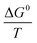 values, which are obtained from ln
values, which are obtained from ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K0, can be re-plotted against
K0, can be re-plotted against 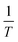 to give ΔH0 by application of eqn (10) on their line slopes (Fig. S12 in ESI†). Also, the entropy difference ΔS0 is obtained from eqn (11).
to give ΔH0 by application of eqn (10) on their line slopes (Fig. S12 in ESI†). Also, the entropy difference ΔS0 is obtained from eqn (11).
The results of the calculation of the above thermodynamic parameters are presented in Table 4. The positivity of the ΔH0 values reveals the endothermic nature of the sorption process. This result is in good agreement with the previous observation about the improvement of the maximum uptake rate (i.e. Qmax) with an increase in the temperature.
| Ions | ΔG0 (kJ mol−1) | ΔS0 (kJ mol−1 K−1) | ΔH0 (kJ mol−1) | ||
|---|---|---|---|---|---|
| 298 (K) | 318 (K) | 338 (K) | |||
| Co2+ | −4.2 | −4.9 | −5.9 | 0.1 | 14.2 |
| Sr2+ | −2.4 | −3.9 | −5.8 | 0.1 | 10.7 |
In order to acquire information regarding the kinetics data and find out the rate controlling step of the kinetics, two different kinds of kinetics models were utilized.
First, the kinetics data were fitted with a pseudo-second order reaction model, represented by:
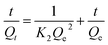 | (12) |
Furthermore, the kinetics data were fitted using the well-known shrinking core model (SCM).49 There are three different types of SCM which are liquid film resistance, intraparticle resistance, and reaction control. Mathematically, these are represented by the following formulae:
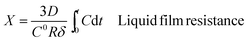 | (13) |
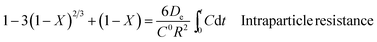 | (14) |
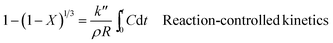 | (15) |
Besides the above-mentioned fitting results, there are three pieces of evidence that show that the rate determining step of the adsorption is more likely to be the chemical reaction of the ions with the M-RGO hybrid: (1) nano sized magnetite or graphene particles cannot present an intraparticle resistance; (2) the uptake kinetics greatly depends on the nature of the ions so that for Co2+ it is about two times faster. Indeed in a series of experiments that were conducted on the adsorption of Th4+ ions by graphene oxide, the present authors observed an instantaneous rate of adsorption. This facet reveals the importance of the nature of the ion or, in other words, the role of the chemical reaction in kinetics; and (3) the diffusion coefficient of Sr2+ ions in water which is about 7.9 × 10−6 cm2 s−1 is higher than the 6.5 × 10−6 cm2 s−1 of Co2+, thus if liquid film resistance had been the rate controlling factor, the adsorption of Sr2+ ions should take place faster than that of Co2+.
Fig. 16 displays the uptake percentage of Co2+ and Sr2+ ions onto the M-RGO hybrid. The adsorption capacity of M-RGO for Co2+ ions over the whole range of pH is higher than that of M-GO as a result of: (1) the presence of more abundant active sites and functional groups on the surface of M-RGO (Fig. 12) which is result of the surface modification of the Fe3O4 NPs by the supercritical methanol media, and (2) the higher Fe3O4 NP content in M-RGO (92% wt) in comparison with that in M-GO (58% wt) which plays an important role in the adsorption of Sr2+ and Co2+ ions.
 | ||
| Fig. 16 The effect of pH on the adsorption of M-GO and M-RGO for (a) Co2+, and (b) Sr2+ ions from aqueous solution. | ||
For M-GO, the uptake percentage remains quite small up to pH ≈ 5, while in the case of M-RGO this break point occurs at a lower pH (pH ≈ 2.1). This behavior is attributed to the emergence of the PZC for M-RGO at a lower pH (pH = 1.9) compared to the PZC of M-GO (pH = 3.7). Taking into consideration the simplicity of the magnetic separation of M-RGO from the solution (Fig. S1†) and the high adsorption capacity in acidic solutions, M-RGO appears to be a promising material for the removal of toxic cations from industrial effluents.
Another important characteristic of a commercial adsorbent which reduces the operational cost of the adsorption process is the reusability, meaning that the uptake capacity should be maintained in successive adsorption–desorption cycles. As the outcome of the experiments of the last paragraph, we used a solution of 0.5 M HCl for elution, and conducted a 4-cycle adsorption–desorption test on the prepared samples. The data obtained in Fig. 17a reveal that M-GO loses around 30% and 25% of its initial capacity for Sr2+ and Co2+ ions after four cycles of adsorption, respectively. In contrast, M-RGO could keep its adsorption capacity nearly intact even after four cycles of adsorption–desorption (Fig. 17b). This could be attributed to the numerous functional groups such as hydroxyl, alkoxide, and carboxylate on the Fe3O4 NP surfaces (Fig. 12) resulting in the retention of the adsorption capacity of M-RGO in comparison with M-GO. These experiments clearly demonstrate the superiority of the M-RGO hybrid over M-GO in the practical separation of Sr2+ and Co2+ ions.
Conclusion
A green, quick, and flexible method for the simultaneous synthesis and grafting of Fe3O4 NPs onto the surface of graphene was introduced in the current study. Supercritical methanol, in which such fabrication processes were conducted, provides a unique reaction medium that supports both reduction of GO and surface modification of Fe3O4 NPs by alkoxide, hydroxyl, and carboxylate groups.The result of different instrumental analyses and a theoretical density functional simulation allowed us to propose a formation mechanism for the M-RGO hybrid according to which, (1) after grafting of the magnetite nanoparticles onto the surface of graphene, the overall surface charge becomes about −0.0236e, exhibiting a charge transfer from the Fe3O4 cluster to the graphene surface. In addition, it was shown that the Fe3O4 cluster is connected to the surface of graphene via C–O–Fe and C–Fe bonds, and (2) supercritical methanol significantly modifies the surface of the Fe3O4 NPs through negative functional groups, and results in a higher adsorption affinity toward Co2+ and Sr2+ ions.
As a result of the aforementioned surface modification, the adsorption capacity of M-RGO is appreciably higher than that of the previously synthesized ultrasound-assisted M-GO composite. Due to the similarity of Co2+ and Fe2+ ions, the uptake capacity of M-RGO for Co2+ ions was about two fold higher than that for Sr2+ ions, but for both of the ions, the uptake isotherm closely followed Langmuir’s monolayer model, with a maximum capacity of 1.1 and 0.6 mmol g−1 for Co2+ and Sr2+, respectively.
The kinetics data were modelled using different forms of the shrinking core model. The results showed that for both Co2+ and Sr2+ ions, the chemical reaction SCM provided the best fitting. Furthermore, the adsorption kinetics obeyed a pseudo-second order reaction model for both of the studied ions, indicating that the chemical reaction of the ions with the functional groups on the surface of M-RGO is the rate controlling step of the adsorption.
Thermodynamic data indicated that the adsorption of both of the Sr2+ and Co2+ ions was endothermic, and the entropy slightly increased during the adsorption process.
The examination of the pH-dependent sorption of the Sr2+ and Co2+ ions, along with the zeta potential measurement, showed that by increasing the pH, the surface charge of M-RGO changes from positive to negative at pHPZC = 1.9, and consequently, the adsorption ability for metal cations is initiated at pH > 2, even though an abrupt jump in the adsorption ability takes place at pH > 4. Reusability tests indicated that the M-RGO hybrid could be used in at least 4 cycles of adsorption–desorption processes without considerable losses of the capacity. Therefore, M-RGO could be considered as a promising alternative of the well-known magnetite-graphene oxide hybrid in the uptake of heavy metal ions from aqueous solutions.
References
- F. K. Shieh, C.-T. Hsiao, H.-M. Kao, Y.-C. Sue, K.-W. Lin, C.-C. Wu, X.-H. Chen, L. Wan, M.-H. Hsu and J. R. Hwu, RSC Adv., 2013, 3, 25686–25689 RSC.
- V. K. Gupta, I. Ali, T. A. Saleh, A. Nayak and S. Agarwal, RSC Adv., 2012, 2, 6380–6388 RSC.
- S. Pollard, G. Fowler, C. Sollars and R. Perry, Sci. Total Environ., 1992, 116, 31–52 CrossRef CAS.
- K. A. Mkhoyan, A. W. Contryman, J. Silcox, D. A. Stewart, G. Eda, C. Mattevi, S. Miller and M. Chhowalla, Nano Lett., 2009, 9, 1058–1063 CrossRef CAS PubMed.
- W. Gao, L. B. Alemany, L. Ci and P. M. Ajayan, Nat. Chem., 2009, 1, 403–408 CrossRef CAS PubMed.
- Y. K. Kim, H. K. Na, S. Kim, H. Jang, S. J. Chang and D. H. Min, Small, 2015, 21, 2527–2535 CrossRef PubMed.
- S. Dutta, C. Ray, S. Sarkar, M. Pradhan, Y. Negishi and T. Pal, ACS Appl. Mater. Interfaces, 2013, 5, 8724–8732 CAS.
- C. Nethravathi, E. Anumol, M. Rajamathi and N. Ravishankar, Nanoscale, 2011, 3, 569–571 RSC.
- P. Fernández-Ibáñez, M. Polo-López, S. Malato, S. Wadhwa, J. Hamilton, P. Dunlop, R. D’Sa, E. Magee, K. O’Shea and D. Dionysiou, Chem. Eng. J., 2015, 261, 36–44 CrossRef.
- S. Moradi, O. Akhavan, A. Tayyebi, R. Rahighi, M. Mohammadzadeh and H. S. Rad, RSC Adv., 2015, 5, 47529–47537 RSC.
- M. Srivastava, J. Singh, T. Kuila, R. K. Layek, N. H. Kim and J. H. Lee, Nanoscale, 2015, 7, 4820–4868 RSC.
- A. K. Swain, L. Pradhan and D. Bahadur, ACS Appl. Mater. Interfaces, 2015, 7, 8013–8022 CAS.
- G. Bharath, V. Veeramani, S.-M. Chen, R. Madhu, M. Manivel Raja, A. Balamurugan, D. Mangalaraj, C. Viswanathan and N. Ponpandian, RSC Adv., 2015, 5, 13392–13401 RSC.
- R. Justin, K. Tao, S. Román, D. Chen, Y. Xu, X. Geng, I. M. Ross, R. T. Grant, A. Pearson, G. Zhou, S. MacNeil, K. Sun and B. Chen, Carbon, 2016, 97, 54–70 CrossRef CAS.
- J. C. Spear, J. P. Custer and J. D. Batteas, Nanoscale, 2015, 7, 10021–10029 RSC.
- A. Tayyebi, M. Outokesh, S. Moradi and A. Doram, Appl. Surf. Sci., 2015, 353, 350–362 CrossRef CAS.
- T. Qi, C. Huang, S. Yan, X.-J. Li and S.-Y. Pan, Talanta, 2015, 144, 1116–1124 CrossRef CAS PubMed.
- M. Faraji, Y. Yamini and M. Rezaee, J. Iran. Chem. Soc., 2010, 7, 1–37 CrossRef CAS.
- B. Veriansyah, J.-D. Kim, B. K. Min and J. Kim, Mater. Lett., 2010, 64, 2197–2200 CrossRef CAS.
- A. Baiker, Chem. Rev., 1999, 99, 453–474 CrossRef CAS PubMed.
- T. Adschiri, Y. Hakuta and K. Arai, Ind. Eng. Chem. Res., 2000, 39, 4901–4907 CrossRef CAS.
- C. Xu and A. S. Teja, J. Supercrit. Fluids, 2006, 39, 135–141 CrossRef CAS.
- D. T. Sawyer and J. L. Roberts Jr, Acc. Chem. Res., 1988, 21, 469–476 CrossRef CAS.
- H. Choi, B. Veriansyah, J. Kim, J.-D. Kim and J. W. Kang, J. Supercrit. Fluids, 2010, 52, 285–291 CrossRef CAS.
- J. Kim, D. Kim, B. Veriansyah, J. W. Kang and J.-D. Kim, Mater. Lett., 2009, 63, 1880–1882 CrossRef CAS.
- B. Veriansyah, J.-D. Kim, B. K. Min, Y. H. Shin, Y.-W. Lee and J. Kim, J. Supercrit. Fluids, 2010, 52, 76–83 CrossRef CAS.
- A. Hadi, J. Karimi-Sabet, S. M. A. Moosavian and S. Ghorbanian, J. Supercrit. Fluids, 2016, 107, 92–105 CrossRef CAS.
- A. Tayyebi, M. M. Tavakoli, M. Outokesh, A. Shafiekhani and A. Simchi, Ind. Eng. Chem. Res., 2015, 54, 7382–7392 CrossRef CAS.
- B. Li and H. Cao, J. Mater. Chem., 2011, 21, 3346–3349 RSC.
- P. T. Yin, S. Shah, M. Chhowalla and K.-B. Lee, Chem. Rev., 2015, 115, 2483–2531 CrossRef CAS PubMed.
- Y. Shao, J. Wang, H. Wu, J. Liu, I. A. Aksay and Y. Lin, Electroanalysis, 2010, 22, 1027–1036 CrossRef CAS.
- N. Song, H. Fan and H. Tian, Appl. Surf. Sci., 2015, 353, 580–587 CrossRef CAS.
- P. V. Kamat, J. Phys. Chem. Lett., 2010, 1, 520–527 CrossRef CAS.
- W. S. Hummers and R. E. Offeman, J. Am. Chem. Soc., 1958, 80, 1339 CrossRef CAS.
- M. Shakourian-Fard, Z. Jamshidi, A. Bayat and G. Kamath, J. Phys. Chem. C, 2015, 119, 7095–7108 CAS.
- Y. Zhao and D. G. Truhlar, Theor. Chem. Acc., 2008, 120, 215–241 CrossRef CAS.
- R. A. MacPhail, Gaussian09, Inc., Wallingford CT, 2009 Search PubMed.
- A. Nourmohammadi, R. Rahighi, O. Akhavan and A. Moshfegh, J. Alloys Compd., 2014, 612, 380–385 CrossRef CAS.
- M. Mahmoudi, A. Simchi, M. Imani, A. S. Milani and P. Stroeve, J. Phys. Chem. B, 2008, 112, 14470–14481 CrossRef CAS PubMed.
- M. Tayebi, S. Ramazani, M. Hamed Mosavian and A. Tayyebi, Polym. Adv. Technol., 2015, 26, 1083–1090 CrossRef CAS.
- M. M. Tavakoli, A. Tayyebi, A. Simchi, H. Aashuri, M. Outokesh and Z. Fan, J. Nanopart. Res., 2015, 17, 1–13 CrossRef CAS.
- S. F. Boys and F. d. Bernardi, Mol. Phys., 1970, 19, 553–566 CrossRef CAS.
- R. F. Bader, Atoms in molecules, Wiley Online Library, 1990 Search PubMed.
- H. Idriss and E. Seebauer, J. Mol. Catal. A: Chem., 2000, 152, 201–212 CrossRef CAS.
- C. d. Slostowski, S. Marre, O. Babot, T. Toupance and C. Aymonier, Langmuir, 2012, 28, 16656–16663 CrossRef CAS PubMed.
- M. Hasan, M. Zaki and L. Pasupulety, J. Mol. Catal. A: Chem., 2002, 178, 125–137 CrossRef CAS.
- H. Idriss, Platinum Met. Rev., 2004, 48, 105–115 CrossRef CAS.
- M. I. Zaki and N. Sheppard, J. Catal., 1983, 80, 114–122 CrossRef CAS.
- A. Tayyebi, A. Khanchi, M. Ghofrani and M. Outokesh, Sep. Sci. Technol., 2010, 45, 288–298 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra19057f |
| This journal is © The Royal Society of Chemistry 2016 |










