Influence of a urea–formaldehyde resin adhesive on pyrolysis characteristics and volatiles emission of poplar particleboard
Y. Zhang,
Z. B. He,
L. Xue,
D. M. Chu and
J. Mu*
Beijing Key Laboratory of Wood Science and Engineering, MOE Key Laboratory of Wooden Material Science and Application, College of Materials Science and Technology, Beijing Forestry University, No. 35, Qing Hua East Road, Hai Dian District, Beijing100083, P. R. China. E-mail: mujun222@sina.com; Tel: +86-0101-62336490
First published on 21st January 2016
Abstract
To investigate the influence of urea–formaldehyde resin (UF resin) adhesive on the thermal utilization of wood waste, the pyrolysis of particleboard and its main components (poplar and UF resin) are studied in this paper. Thermogravimetric analysis combined with Fourier transform infrared spectrometry (TG-FTIR), gas chromatography coupled with mass spectrometry (GC-MS) and ultimate analysis were used to investigate the pyrolysis process and product characteristics of poplar and UF resin and particleboard. The thermogravimetric curves show that, compared to poplar and UF resin, pyrolysis of particleboard produces more char and less volatile matter. A distributed activation energy method is used to retrieve kinetic parameters from the measurements. The activation energy of particleboard is lower than that of poplar during the main pyrolysis stage. The activation energies of poplar and particleboard are 140–200 kJ mol−1, while that of UF resin is 150–300 kJ mol−1. Evolved gaseous products during the pyrolysis of poplar, such as CH4, CO, CO2, are found. N-Compounds are the main volatile products of UF resin. Moreover, N-components of particleboard are mainly produced by UF resin. The influence of UF resin on the pyrolysis liquid of particleboard is mainly on the nitrogen compounds. More nitrogenous compounds are detected in the pyrolysis liquid of particleboard, and the relative amount of nitrogenous compounds is 23.40% more than that in poplar pyrolysis liquid.
1. Introduction
With the development of building decoration and the furniture industry, the development of the wood-based panel industry has become more and more rapid. Since the start of the 21st century, the output of wood-based panels has shown an average annual growth of 20% in China;1 the production of wood-based panels was 273 million m3 in 2014. Each year, large amounts of wood-based panels are consumed and abandoned.2 The inadequate processing of waste wood-based composites, such as landfill and incineration, doesn't just create pollution; it is also the cause of significant waste.Waste wood-based panels are a kind of lignocellulosic bioresource and show great potential for providing raw materials for the production of renewable fuels and chemical feedstocks.3 Biomass pyrolysis has proven itself to be a new type of biomass energy utilization technique, and has broad prospects for application in the utilization of forest rejectamenta and agricultural residues.4 With this process, the low energy density in biomass can be converted into products with high energy density and many chemicals can be obtained.5 Pyrolysis technology provides an effective way for the utilization of recycled waste wood-based panels.6
However, the pyrolysis process of waste wood-based boards is not the same as that of conventional biomass materials due to the adhesive present in waste wood-based boards, which influences the pyrolysis process and products. Compared with wood, waste wood-based panels perform differently in pyrolysis. They may produce more nitrogenous products in pyrolysis because of the presence of a urea–formaldehyde resin (UF resin) adhesive.7
The pyrolysis of waste wood boards was reported by Mu et al.8 Research shows that the pyrolysis liquid of wood-based panels is neutral to alkaline due to the influence of UF resin adhesives. Component analysis shows that there are more nitric compounds in the pyrolysis liquid of wood boards. The liquid products of the pyrolysis of wood and bamboo have extensive applications. Wood vinegar has been found to promote plant growth and has antibacterial and anticorrosion functions, and also can be used for wood preservation.9 There are some differences between the pyrolysis condensate liquid of wood-based panels and wood vinegar in terms of composition and properties. The pyrolysis liquids of wood-based panels can be used as basic solvents and additives in pesticides and in many other ways. A study on the characteristics and bio-efficiencies of pyrolysis liquids from wood, particleboard, plywood and fiberboard showed that the inhibitions of these liquids on fungi are different due to their different compositions.10 However, research on wood-based panel pyrolysis liquids has not so far lead to their practical application.11 Therefore, study of the formation mechanism of pyrolysis liquid is of great significance in the study of the utilization of waste wood-based panels.
Furthermore, the UF resin adhesive used in wood-based panels will produce a great quantity of nitrogen-containing gases, such as NH3, HNCO, and HCN, during the pyrolysis process.12 Nitrogen oxides will be generated by the oxidation of ammonia and hydrogen.13 Nitric oxides will do serious harm to the environment and human health.14 In order to effectively control the emission of nitrogen-containing gases, the transformation rules of nitrogen during wood-based panel pyrolysis must be studied.
Poplar particleboard, which has a high content of UF resin, was selected to observe the effect of UF resin on the pyrolysis process and characterize the volatile products. In this paper, pyrolysis analysis was performed by TG-FTIR, ultimate analysis, and GC-MS, and pyrolysis kinetics analysis was performed to evaluate the thermal behavior of particleboard and its main components.
2. Experimental
2.1. Samples preparation
Poplar and UF resin were provided by a wood board factory. Particleboard was formulated in our own laboratory. The composition in wt% (on dry basis) of each component is: wood; 92–93%, UF resin; 7–8%. Samples were dried in an oven for 24 h at 376 K before experiments were performed.2.2. TG-FTIR analysis
Experimental work was carried out using a computerized thermobalance (Netzsch STA449F3). Thermobalance configuration gives a sensitivity of ±0.1 μg thus allowing the use of small samples (10–15 mg). In order to establish an inert atmosphere during all experiments, a controlled argon flow swept the measurement cell that was purged for 20 min before starting the heating program. During the experiments, argon flow was fixed at 20 mL min−1. All sample materials were heated to 1173 K for pyrolysis, employing three different heating rates (5, 20 and 50 K min−1).As the sample was heated, the evolved volatile products were carried out of the furnace and sent, via a heated line, to a gas cell of an FTIR spectrometer (Bruker Vertex70v), where they were analyzed in a semi-continuous way. The configuration of the FTIR spectrometer (resolution of 2 cm−1, 5 scans per measurement) allowed a qualitative analysis of many compounds: carbon dioxide and monoxide, light hydrocarbons (ethane, ethylene, etc.), formaldehyde, N-compounds (ammonia, isocyanic acid, hydrocyanic acid and nitric oxides), and methanol.
This was accomplished through the use of small sample and particle sizes as well as relatively fast carrier-gas flow rates so that the volatile products of pyrolysis were swept into the gas cell immediately after they were formed. The instrument and the measurements were designed in such a way so that secondary reactions were minimized. The system continuously monitored the time dependent evolution of the gases and weight of the non-volatile material (residue).
2.3. Pyrolysis reactor
Poplar, UF resin and particleboard were pyrolyzed in a laboratory scale tubular furnace reactor at a linear heating rate of 5 K min−1 from ambient temperature to 873 K, and maintained at 873 K for 30 minutes. The carrier gas was argon with a flow rate of 100 mL min−1 in order to achieve an inert atmosphere. Pyrolysis liquids were collected by using cold water to trap the condensable products. The test apparatus is shown in Fig. 1.2.4. GC-MS analysis
Pyrolysis liquids were obtained from the pyrolysis of poplar, UF resin and particleboard by a laboratory scale tubular furnace reactor. The components of the pyrolysis liquids were analyzed by GC-MS (TueboMatrix 650 TD-Clarus600 GC-MS). A DB-5MS column (30 m × 0.250 mm × 0.250 μm) was maintained at 333 K for 2 min, then the temperature was raised to 453 K at a heating rate of 5 K min−1, after which the temperature was raised to 553 K at a heating rate of 20 K min−1, and maintained at 553 K for 5 min. Helium was used as a carrier gas. The mass spectrometer was operated in an electron ionization (EI) mode with an ionization energy of 70 eV. The interface temperature was 523 K. The scan interval was 0.5 s and the scan range for m/z was 29 to 600.2.5. Elementary analysis and components analysis
The ultimate analysis of virgin samples, char of poplar, UF resin adhesive, and particleboard was carried out on an Elementar Vario EL III. The content of C, H and N was calculated by the mean value of two tests. Content of O was obtained by difference.The content of cellulose, hemicellulose, lignin and ash was measured using National Renewable Energy Laboratory's (NREL) standard analytical procedure. In brief, the cellulose and hemicellulose content of poplar was determined based on monomer content after a two-step acid hydrolysis. The acid insoluble lignin was collected by filtration, washed with deionized water until a neutral pH eluate was obtained, and dried at 378 K to a constant weight. Measurement of ash content was carried out using a muffle furnace at 848 ± 25 K until a constant weight was obtained. All analyses were run in triplicate.
Table 1 shows the ultimate analysis of poplar, UF resin and particleboard (dry basis).
| Sample | Elementary analysis (wt% on dry basis) | Chemical compositions (wt%) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| C | H | N | Oa | Holocellulose | Cellulose | Hemicellulose | Lignin | Ash | |
| a Determined by difference. | |||||||||
| Poplar | 48.44 | 5.49 | 0.13 | 45.95 | 77.19 | 48.76 | 28.43 | 21.54 | 4.4 |
| UF resin | 34.69 | 5.59 | 33.72 | 26.01 | — | — | — | — | — |
| Particleboard | 48.04 | 5.78 | 2.00 | 44.18 | — | — | — | — | — |
2.6. Pyrolysis kinetics analysis method
The distributed activation energy model (DAEM) has been employed successfully for describing coal pyrolysis. Most recent research has applied this model to the thermal decomposition of biomass and its components.15 The kinetic parameters can be calculated through TGA without considering complex chemical reactions during the thermal decomposition process.16 In this study, it is assumed that the whole thermal conversion process of particleboard and its components is composed of a set of irreversible single (first-order) reactions occurring successively.17The representative equation for the Miura–Maki model is:18
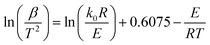 | (1) |
The method consists of four simple steps and becomes a procedure to estimate f(E) and k0: (1) measure experimentally α/T at a minimum of three different heating rates, (2) calculate the values of ln(β/T2) and −1/(RT) at the same α for the different heating rates, (3) plot ln(β/T2) vs. −1/(RT) at the selected α and then graphically determine E and k0 (eqn (1)), and (4) plot α vs. E and differentiate the α vs. E relationship to obtain f(E).19
3. Results and discussion
3.1. The influence of UF resin on the pyrolysis characteristics of particleboard
Fig. 2 shows the weight loss curves (TG) and differential thermogravimetric curves (DTG) for all the samples studied in this work at three different heating rates. The poplar, UF resin and particleboard all show three steps of weight loss at elevated temperature, as seen from the TG curves. The DTG curves are derived from the measured TG curves numerically. Here, also, the three steps of the weight loss of samples are clearly visible. | ||
| Fig. 2 TG curves of poplar (a), UF resin (c) and particleboard (e) and DTG curves of poplar (b), UF resin (d) and particleboard (f) at three different heating rates. | ||
A summary of the TGA experiments for poplar, UF resin and particleboard is presented in Table 2. The similar char yields at different heating rates could imply that cross-linking reactions are relatively unimportant. It also means that bond breaking and cross-linking are in balance with each other.20
| Sample | Poplar | UF resin | Particleboard | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Heating rate (K min−1) | 5 | 20 | 50 | 5 | 20 | 50 | 5 | 20 | 50 |
| Volatile matter (wt%) | 80.87 | 80.36 | 79.83 | 86.01 | 86.72 | 86.47 | 79.77 | 79.99 | 79.67 |
| Char (wt%) | 19.13 | 19.64 | 20.17 | 13.99 | 13.28 | 13.53 | 20.23 | 20.01 | 20.33 |
The TG curves and DTG curves during the pyrolysis of poplar at heating rates of 5, 10 and 50 K min−1 are presented in Fig. 2(a) and (b), respectively. The first stage is attributed to moisture evaporation. The second stage is the main weight loss between 473 and 673 K. A sharp peak and a smaller shoulder, attributed respectively to cellulose and hemicellulose decomposition, are identified at heating rates of 20 and 50 K min−1. This behavior is quite common in biomass samples. The sample presents a single broad peak of decomposition around 604 K at a heating rate of 5 K min−1. In this case, two peaks collide into a broader one.14 The last stage in pyrolysis is the further cracking process of poplar residues due to lignin decomposition, and the weight loss is slight during this stage. Finally, the poplar sample remains a solid residue (carbonaceous residue within inorganic solid particles) equal to around 19.5% of its original weight.
The majority weight loss of UF resin occurs during an active step over a range of about 453–603 K. Furthermore, small peaks are observed in the DTG curves. The second stage of weight loss of the UF resin forms the deepest main DTG valley, seen in Fig. 2(d). Pyrolysis of UF resin produces a higher yield of volatile matter compared to poplar. This result may be caused by a structural difference between them. The UF resin has a network which mainly consists of imino, carbonyl, and methylene groups. Parts of typical structures of UF resins are shown in a study by Hirata.21 When UF resin is heated, an end group of hydroxymethyl is first split to form formaldehyde, which is then followed by the cleavage of C–N linkages to produce volatiles containing nitrogen.21 Furthermore, the results of elemental analysis support this mechanism, indicating a reduction in the percentages of hydrogen, oxygen, and nitrogen, but the percentage of carbon increases significantly, as seen from the examples of the ultimate analysis of sample residues in Table 3. The higher yield of volatile matter may be attributable to the pyrolysis of unstable structures of the UF resin. However, hemicellulose, cellulose and lignin are the three main components of poplar; the lignin content in poplar is more than 20% (Table 1). Generally, in biomass, lignin makes a relatively large contribution to the production of fixed carbon than the other main constituents such as cellulose or hemicellulose. Therefore, the yield of char of poplar is higher than that of UF resin.
| Sample | Poplar (wt%) | UF resin (wt%) | Poplar particle board (wt%) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Heating rate (K min−1) | 5 | 20 | 50 | 5 | 20 | 50 | 5 | 20 | 50 |
| a Determined by difference. | |||||||||
| N | 0.795 | 1.253 | 0.737 | 16.325 | 16.940 | 18.330 | 2.134 | 2.418 | 2.310 |
| C | 84.840 | 86.830 | 84.040 | 77.770 | 75.970 | 74.830 | 88.265 | 88.730 | 89.295 |
| H | 1.488 | 0.427 | 0.562 | 1.018 | 0.645 | 0.721 | 0.696 | 0.561 | 0.656 |
| Oa | 12.877 | 11.490 | 14.661 | 4.887 | 6.445 | 6.119 | 8.904 | 8.291 | 7.739 |
The curves show some differences between poplar (Fig. 2(a) and (b)) and particleboard (Fig. 2(e) and (f)). Pyrolysis of poplar particleboard produces more char and less volatile matter compared to its two components, which means that the thermal behavior of poplar particleboard is not a reflection of the sum of the behavior of poplar and UF resin.13 Therefore, further studies of the gases produced during this pyrolysis process will help to understand the pyrolysis mechanism of particleboard.
3.2. The influence of UF resin on kinetics parameters of particleboard
Fig. 3 shows the plots of ln(β/T2) versus 1/T at selected α for poplar, UF resin and particleboard. For every value of α, the three points corresponding to the three heating rates are positioned in a straight line, as the method predicts. Through Fig. 3, E and k0 are graphically obtained, and the plots of ln(k0) versus E are shown in Fig. 4. | ||
| Fig. 3 Plots of ln(β/T2) versus 1/T of poplar (a), UF resin (b) and particleboard (c) at different conversion rates. | ||
Fig. 5 shows the plot of E versus α. It can be seen that the activation energy of poplar and UF resin are both higher than the activation energy of particleboard during the main pyrolysis stage. This illustrates that the thermal behavior of poplar particleboard is not just a reflection of the sum of the behaviors of poplar and UF resin. The activation energy of particleboard is lower than that of poplar during the main pyrolysis stage because of the promoting effect of UF resin. The activation energies of poplar and particleboard are 140–200 kJ mol−1. However, the activation energy of UF resin is 150–300 kJ mol−1.
3.3. The influence of UF resin on volatiles emission of particleboard
The volatiles emitted during the pyrolysis of samples were analyzed using an FTIR spectrometer. This allowed us to investigate in detail the mass loss behavior shown by TGA measurements. The infrared absorption band assignment for the evolved gases from pyrolysis of poplar, UF resin and particleboard in the TG-FTIR is shown in Table 4. According to the widely used Lambert–Beer law, the absorption spectrum at a specific wavenumber is linearly dependent on gas concentration.22 Thus, the variation of absorbance in the whole pyrolysis process reflects the concentration trend of the gas species.23Fig. 6 shows the FTIR spectra of the pyrolysis gases of poplar at the relevant temperatures. Qualitative analysis during the pyrolysis of poplar reveals the production of compounds which could be classified as follows. The non-condensable gases contain CO (2177 cm−1, 2114 cm−1), CO2 (2360 cm−1, 669 cm−1) and CH4 (3000–2700 cm−1). CO is one of the decomposition products of ether groups, and originates from the ether bridges of lignin subunit connections and/or the ether compounds in the secondary cracking of volatiles. CH4 forms from the cracking of weakly bonded methoxy groups.23 The condensable gases contain water (1900–1300 cm−1, 3950–3500 cm−1) and primary tars. The water comes from both free and bound water. When the pyrolysis temperature is increased, a C–O asymmetric stretching absorbance appears in the wavenumber range of 1131–1077 cm−1, indicating the existence of alcohols. The C–O stretching absorbance peaks in the region of 1900–1650 cm−1 are representative of aldehydes or organic acids. Light tars containing carboxyl groups such as acetic and formic acid come from the degradation of cellulose and hemicelluloses, and methanol may come from the methoxy groups (–OCH3) of lignin.13
The shift to higher temperatures has been reported and has been normally attributed to the combined effects of the heat transfer at different heating rates and the kinetics of decomposition, resulting in a delayed decomposition.24
After a numerical treatment of the different spectra recorded during each experiment, the levels of CO, CH4 and CO2 evolved in the pyrolysis gases can be shown on graphs for different heating rates (see Fig. 7). The curve shows that the temperature range of CO production extends from 500 to 680 K, and a maximum absorbance is reached at around 630 K at a heating rate of 20 K min−1. The evolution of CH4 is mainly concentrated between 497 and 681 K at a heating rate of 20 K min−1. In the case of CO2, the temperature range extends from 483 to 673 K with a peak at 640 K at a heating rate of 20 K min−1. It can be observed that the signal intensity of CO2 is stronger than for CO and CH4. The release of CO, CH4, CO2 is in agreement with their weight loss thermogravimetric data.
 | ||
| Fig. 7 Levels of CO (a), CH4 (b) and CO2 (c) evolved in pyrolysis gas during the pyrolysis of poplar at three different heating rates. | ||
A different trend of CO2 evolution at a heating rate of 5 K min−1 is observed, and the content increases with an increase in temperature and reaches a maximum peak at 618 K. As the temperature exceeds 647 K, in contrast with the decreased gaseous products in the temperature range of 618–647 K at the two faster heating rates, an increased evolution tendency is seen up to 1173 K. This is due to CO2 release from volatile matter below 618 K. As the temperature increases, the polymerization reaction of coking in solid phase is the primary reaction, and this process accompanies the emission of CO2.23
At higher heating rates, progressive broadening of the signals is observed. Since heat transfer phenomena become more important at higher temperature, the material degrades over a wider temperature range and the actual partial pressure of the volatile fraction gets lower, accounting for the decrease of secondary reactions into the gas phase.24
Fig. 8 shows the FTIR spectra of the pyrolysis gases of UF resin at the relevant temperatures. It can be observed that the breakdown products of the UF resin are mainly N-compounds such as isocyanic acid, hydrogen cyanide and ammonia. Indeed, the results given by FTIR spectrometry reveal the presence of the three nitrogenized gases. From Fig. 8, we can distinguish peaks corresponding to HNCO, NH3, CO2 and HCN.
 | ||
| Fig. 8 FTIR spectrum of the pyrolysis gaseous products of UF resin at three different heating rates. | ||
The evolution profiles of CO2, HNCO, NH3 and HCN are reported for different heating rates in Fig. 9. The curve shows that the temperature range of CO2 production extends from 496 to 643 K at a heating rate of 20 K min−1. The temperature range of HNCO extends from 507 to 631 K with a peak at 564 K. The evolution of NH3 starting at 496 K is lower than that of HNCO and HCN (514 K), and the maximum absorbance that appears at around 556 K is lower than that of HNCO and HCN (564 K).
 | ||
| Fig. 9 Evolution levels of CO2 (a), HNCO (b), NH3 (c) and HCN (d) in pyrolysis gaseous products during the pyrolysis of UF resin at three different heating rates. | ||
Furthermore, the signals of NH3 and HCN are weaker than that of HNCO. Two or three main peaks in the curves of HNCO, NH3 and HCN could be found at low heating rate conditions, while they will become one peak as the heating rate of the pyrolysis process increases from 5 to 50 K min−1. This may be attributed to the effect of heat transfer at the different heating rates.
A different trend in the evolution of CO2 at a heating rate of 5 K min−1 is observed, and the content of CO2 presents an increasing tendency up to 1173 K. This is due to CO2 release from volatile matter at the first and second pyrolysis stages. As the temperature increases, the polymerization reaction of coking in the solid phase accompanies the emission of CO2.23
The curves of particleboard (Fig. 10) are similar to those of poplar because particleboard contains around 92% wood. The small difference is probably due to the 8% UF resin present. Therefore, the gases released during the pyrolysis experiments were analyzed in order to find the evolution of N-compounds, which are derived from the UF resin present in the particleboard.
 | ||
| Fig. 10 FTIR spectra of the pyrolysis gaseous products of UF resin at three different heating rates. | ||
The evolution profiles of CO, CH4, CO2, HNCO, NH3, and HCN are shown in Fig. 11. The curves show that the production of CO2 starts at about 473 K and is practically complete at 665 K at a heating rate of 20 K min−1. The maximum absorbance of CO2 appears at around 616 K, at which CO shows a peak of reactivity. The temperature range of CO production extends from 501 to 668 K at a heating rate of 20 K min−1. The signals of these two gases produced by particleboard are stronger than those of poplar. The evolution of CH4 is mainly concentrated between 504 and 667 K at 20 K min−1.
 | ||
| Fig. 11 Evolution levels of CO (a), CH4 (b), CO2 (c), HNCO (d), NH3 (e) and HCN (f) in pyrolysis products during the pyrolysis of particleboard at three different heating rates. | ||
The curve shows that the temperature range of HNCO production extends from 440 to 650 K at a heating rate of 20 K min−1. HNCO shows a pronounced release at 549 K, as shown in Fig. 11(d). The signals of HNCO are much stronger than those of the other two N-components. Furthermore, compared with UF resin, signals of N-components are wider and stronger.
3.4. The influence of UF resin on the components of the pyrolysis liquid of particleboard
The main components of UF resin, poplar and particleboard from GC-MS results are shown in Tables 5–7, respectively.| Retention time/min | Designation | Molecular formula | Relative amount/% |
|---|---|---|---|
| 3.30 | Acetonitrile | C4H8N2 | 0.54 |
| 3.51 | N-Ethylformamide | C3H7ON | 54.52 |
| 5.12 | Pyrimidine, 5-methyl- | C5H6N2 | 4.50 |
| 6.59 | 1H-Imidazole, 1-methyl- | C4H6N2 | 1.18 |
| 8.66 | Cyclohexanol, 3-methyl- | C7H14O | 0.30 |
| 9.92 | Urea, formyltrimethyl- | C5H10O2N2 | 4.30 |
| 10.16 | Pentanamide, N-(aminocarbonyl)- | C6H12O2N2 | 0.88 |
| 10.72 | 3,4-Pyridinediamine | C5H7N3 | 1.81 |
| 11.07 | N-N′-Dimethyloxamide | C4H8O2N2 | 1.26 |
| 13.45 | 1H-Imidazole[1,2-b]pyrazole, 2,3-dihydro- | C5H7N3 | 0.19 |
| 13.62 | (1H)-Pyrrole-3-carbonitrile, 2-methyl- | C6H6N2 | 0.13 |
| 13.79 | Hydrouracil, 1-methyl- | C5H8O2N2 | 1.40 |
| 14.22 | 2,3-Pyridinediamine | C5H7N3 | 6.79 |
| 15.68 | Piperazine, 1-methyl- | C5H12N2 | 0.86 |
| 16.01 | Hexahydroindole | C8H13N | 1.63 |
| 17.07 | 2-Acetyl-3-methyl-3,4,5,6-tetrahydropyridine | C8H13ON | 0.50 |
| 18.36 | 1,3,5-Triazine-2,4,6(1H,3H,5H)-trione, 1,3,5-trimethyl- | C6H9O3N3 | 2.38 |
| 18.49 | S-Triazolo[4,3-a]pyridine, 8-amino- | C6H6N4 | 2.03 |
| 19.81 | 1-Methyl-2,4,5-trioxoimidazolidine | C4H4O3N2 | 7.65 |
| 21.20 | 1,2,4-Trizolo[4,3-a]pyridin-8-amine, 3-methyl- | C7H8N4 | 2.70 |
| 22.94 | 6-Methyl-7,8-dihydro-2(1H)-pteridinone | C7H8ON4 | 1.75 |
| 24.46 | 2-Pyrrolidinophenol | C10H13ON | 2.69 |
| Retention time/min | Designation | Molecular formula | Relative amount/% |
|---|---|---|---|
| 4.37 | 2,5-Dimethylfuran | C7H13O | 27.96 |
| 5.94 | 1-Pyrrolidinecarbonitrile | C5H8N2 | 6.44 |
| 6.32 | 2-Cyclopenten-1-one, 2-hydroxy | C5H6O2 | 1.84 |
| 7.25 | 2-Cyclohexen-1-one, 4-(2-oxopropyl)- | C9H12O2 | 7.12 |
| 7.63 | Phenol | C6H6O | 4.08 |
| 8.13 | 7-Octenal, 3,7-dimethyl | C10H18O | 1.19 |
| 8.31 | Tetrahydrofuran, 2-propyl- | C7H14O | 1.61 |
| 9.10 | 2-Cyclopenten-1-one, 2-hydroxy-3-methyl- | C6H8O2 | 7.14 |
| 9.26 | Spiro[2,4]heptan-4-one | C7H10O | 0.98 |
| 9.62 | Phenol, 2-methyl- | C7H8O | 1.90 |
| 10.29 | Phenol, 4-methyl- | C7H8O | 2.30 |
| 10.60 | Phenol, 2-methoxy- | C7H8O2 | 4.12 |
| 11.36 | Maltol | C6H6O3 | 0.89 |
| 11.53 | 2-Cyclopenten-1-one, 3-ethyl-2-hydroxy | C7H10O2 | 1.03 |
| 11.95 | Phenol, 2-ethyl- | C8H10O | 0.28 |
| 12.29 | Phenol, 2,4-dimethyl- | C8H10O5 | 0.83 |
| 12.88 | Phenol, 3,5-dimethyl- | C8H10O | 0.29 |
| 13.09 | 2-Methoxy-6-methylphenol | C8H10O2 | 0.45 |
| 13.49 | Phenol, 2-methoxy-4-methyl- | C8H10O2 | 2.74 |
| 13.58 | 1,2-Benzenediol | C6H6O2 | 0.44 |
| 14.01 | 2-Hydroxy-3-propyl-2-cyclopenten-1-one | C8H12O2 | 0.14 |
| 14.33 | 1,4:3,6-Dianhydro-alpha-D-glucopyranose | C6H8O4 | 0.45 |
| 14.48 | Phenol, 3-(1-methylethyl)- | C9H12O | 0.30 |
| 14.61 | 2,3-Anhydro-D-mannosan | C6H8O4 | 0.72 |
| 15.38 | 1,2-Benzenediol, 3-methoxy | C7H8O3 | 2.85 |
| 15.84 | Phenol, 4-ethyl-2-methoxy | C9H12O2 | 1.20 |
| 16.08 | 1-H-Inden-1-one, 2,3-dihydro- | C9H8O | 0.16 |
| 16.22 | 1,2-Benzenediol, 4-methyl- | C7H8O2 | 0.48 |
| 16.75 | 5-Acetoxymethyl-2-furaldehyde | C8H8O4 | 0.31 |
| 17.61 | 3-Furancarboxylic acid, 2,5-dimethyl-, methyl ester | C8H10O3 | 0.39 |
| 17.91 | Phenol, 2,6-dimethoxy- | C8H10O3 | 9.61 |
| 18.06 | Phenol, 3,4-dimethoxy | C8H10O3 | 0.46 |
| 18.64 | 4-Ethylcatechol | C8H10O2 | 0.32 |
| 19.11 | Benzaldehyde, 3-hydroxy-4-methoxy | C8H8O3 | 0.38 |
| 20.31 | 1,2,4-Trimethoxybenzene | C9H12O3 | 5.55 |
| 21.29 | Ethanone, 1-(4-hydroxy-3-methoxyphenyl)- | C9H10O3 | 0.31 |
| 22.16 | Benzene, 1,2,3-trimethoxy-5-methyl- | C10H14O3 | 0.12 |
| 22.31 | 2-Propanone, 1-(4-hydroxy-3-methoxyphenyl)- | C10H12O3 | 0.51 |
| 22.44 | 4-Hexenoic acid, 5-hydroxy-3-oxo-2-propionyl-, D-lactone | C9H10O4 | 0.12 |
| 22.67 | 1,3-Di-O-acetyl-alpha-beta-D-ribopyranose | C9H14O7 | 0.15 |
| 23.11 | 4-Methyl-2,5-dimethoxybenzaldehyde | C10H12O3 | 0.14 |
| 24.1 | 3,5-Dimethoxy-4-methybenzoic acid | C10H12O4 | 0.31 |
| 25.28 | Benzaldehyde, 4-hydroxy-3,5-dimethoxy- | C9H10O4 | 0.37 |
| 26.04 | 1,6;2,3-Dianhydro-4-O-acetyl-beta-D-gulopyranose | C8H10O5 | 0.05 |
| 26.24 | Phenol, 2,6-dimethoxy-4-(2-propenyl)- | C11H14O3 | 0.46 |
| Retention time/min | Designation | Molecular formula | Relative amount/% |
|---|---|---|---|
| 4.55 | 2-Furanmethanol | C5H6O2 | 14.22 |
| 5.29 | Acetamide, N,N-dimethyl- | C4H9ON | 1.84 |
| 5.68 | 2-Cyclopenten-1-one, 2-methyl- | C6H8O | 0.14 |
| 6.03 | 1-Pyrrolidinecarbonitrile | C5H8N2 | 11.24 |
| 7.39 | 2-Cyclopenten-1-one, 3-methyl- | C6H8O | 2.33 |
| 7.58 | 4-Aminopyrimidine | C4H5N3 | 0.50 |
| 8.09 | Pyrimidine, 5-methyl- | C5H6N2 | 3.19 |
| 8.98 | 2-Cyclopenten-1-one, 2-hydroxy-3-methyl- | C6H8O2 | 1.45 |
| 9.22 | 2-sec-Butyl-3-methyl-1-pentene | C10H2O | 3.19 |
| 9.27 | 2-Cyclopenten-1-one, 2,3-dimethyl- | C7H10O | 0.77 |
| 9.51 | DL-Citrulline | C6H13O3N3 | 3.25 |
| 9.64 | Piperidine, 1-methyl- | C6H13N | 0.02 |
| 9.88 | Phenol, 2-methyl- | C7H8O | 0.92 |
| 10.68 | Ethanone, 1-(2-methyl-1-cyclopenten-1-yl)- | C8H12O | 6.64 |
| 10.82 | 1,3-Cyclopentanedione, 4-ethyl- | C7H10O2 | 0.71 |
| 10.94 | 2,5-Pyrrolidinedione, 1-methyl- | C5H7O2N | 0.62 |
| 11.05 | 1-H-Pyrazole, 1,3,5-trimethyl- | C6H10N2 | 0.21 |
| 11.37 | 1,3-Cyclopentanedione, 2,2-diethyl- | C7H10O2 | 2.12 |
| 11.56 | Maltol | C6H6O3 | 1.91 |
| 11.69 | 2-Cyclopenten-1-one, 3-ethyl-2-hydroxy- | C7H10O2 | 1.17 |
| 11.85 | Bicyclo(2,2,2)octane, 2-methyl- | C9H16 | 0.10 |
| 12.18 | 4-(1H)-Pyridinone, 2,3-dihydro-1-methyl- | C6H9ON | 2.12 |
| 12.99 | 1H-Imidazole, 2,4,5-trimethyl- | C6H10N2 | 0.03 |
| 13.36 | 4,5-Octanedione | C8H14O2 | 2.04 |
| 13.6 | Phenol, 2-methoxy-4-methyl- | C8H10O2 | 0.68 |
| 13.68 | Quinuclidine-3-ol | C7H13ON | 0.15 |
| 13.83 | S-Triazolo[4,3-a]pyrazine, 3-methyl- | C6H6N4 | 1.31 |
| 14.1 | 2-Hydroxy-3,5-diethyl-5-methylcyclopent-2-en-1-one | C10H16O2 | 0.55 |
| 14.39 | 1,2-Benzenediol | C6H6O2 | 1.39 |
| 14.51 | 2-Isopropoxyphenol | C9H12O2 | 0.68 |
| 14.69 | Oxazole, 4-ethyl-2,5-dimethyl- | C7H11ON | 0.95 |
| 14.81 | 2-Methyl-4-(1-methylethyl)-2-cyclohexenone | C10H16O | 0.57 |
| 15.01 | Phenol, 2,6-dimethoxy- | C8H10O3 | 0.22 |
| 15.64 | 1,2-Benzenediol, 3-methoxy- | C7H8O3 | 3.53 |
| 15.89 | 1,2-Benzenediol, 3-methyl- | C7H8O2 | 1.15 |
| 16.27 | Pyridine, 4-(1-pyrrolidinyl) | C9H12N2 | 0.45 |
| 16.86 | 1,2-Benzenediol, 4-methyl- | C7H8O2 | 1.73 |
| 18.07 | Phenol, 2,6-dimethoxy- | C8H10O3 | 11.67 |
| 18.22 | Benzenemethanol, 3-hydroxy-5-methoxy- | C8H10O3 | 1.58 |
| 18.65 | 1,3,5-Triazine-2,4,6(1H,3H,5H)-trione, 1,3,5-trimethyl- | C6H9O3N3 | 2.28 |
| 19.07 | 2,3-Dimethylhydroquinone | C8H10O2 | 0.36 |
| 19.14 | 1,3-Benzenediol, 4-ethyl- | C8H10O2 | 0.27 |
| 20.36 | 1,2,3-Trimethoxybenzene | C9H10O3 | 3.11 |
| 20.44 | Ethanone, 1-(2,3,4-trihydroxyphenyl) | C8H8O4 | 1.14 |
| 21.41 | Ethanone, 1-(4-hydroxy-3-methoxyphenyl)- | C9H10O3 | 0.18 |
| 21.64 | 8-Quinolinol, 5-amino- | C9H8ON | 0.27 |
| 22.21 | Benzene, 1,2,3-trimethoxy-5-methyl- | C10H14O3 | 1.52 |
| 22.38 | 2-Propanone, 1-(4-hydroxy-3-methoxyphenyl)- | C10H12O3 | 0.49 |
| 22.54 | 2-(2-Aminoethyl)-4-amino-6-dimethylamino-s-triazine | C7H14N6 | 0.14 |
| 22.82 | 4(1H)-Pteridinone, 2-amino- | C5H5ON5 | 0.23 |
| 23.98 | Phenol, 2,6-dimethoxy-4-(2-propenyl)- | C11H14O3 | 0.23 |
| 24.13 | Benzene, 1,1′-propylidenebis | C15H16 | 0.20 |
| 24.53 | 5-Oxohexanenitrile | C6H9ON | 0.18 |
| 25.1 | Butan-2-one, 4-(3-hydroxy-2-methoxyphenyl)- | C11H14O3 | 0.33 |
| 25.44 | 2-Pyrrolidinophenol | C10H13ON | 0.72 |
| 26.26 | Phenol, 2,6-dimethoxy-4-(2-propenyl)- | C11H1403 | 0.40 |
| 26.79 | Ethanone, 1-(4-hydroxy-3,5-dimethoxyphenyl)- | C10H12O4 | 0.45 |
| 27.34 | Benzoic acid, 4-hydroxy-3,5-dimethoxy-, hydrazide | C9H12O4N2 | 0.14 |
From Table 5, it can be seen that nitrogenous substances are the main components of UF resin pyrolysis liquid. The relative content of N-ethylformamide reaches up to 54.52%. In addition, other nitrogenous substances, such as 1-methyl-2,4,5-trioxoimidazolidine (7.75%), 2,3-pyridinediamine (6.79%), pyrimidine, 5-methyl- (4.50%), urea, and formyltrimethyl- (4.30%), can also be detected in the pyrolysis liquid of UF resin. Nitrogen-containing heterocyclic compounds in the pyrolysis liquid of UF resin, such as pyridine, pyrimidine and piperazine, are alkaline and have an irritating smell.
Poplar mainly consists of cellulose, hemicellulose and lignin, and therefore its pyrolysis behavior can be considered to be the sum of the behaviors of these three components.25 From Table 6, it can be seen that the components of pyrolysis liquid of poplar are very complex. It consists of multiple components, such as alcohols, ketones, aldehydes, furan derivatives and carbohydrates. The major constituent is 2,5-dimethylfuran and the relative content is 27.96%. Other furan derivatives, such as tetrahydrofuran, 2-propyl- (1.61%) and 5-acetoxymethyl-2-furaldehyde (0.31%) can also be detected in the pyrolysis liquid of poplar. These furan derivatives are generated by the pyrolysis of holocellulose. A glycosidic bond cleavage of cellulose and an aldol condensation reaction lead to the formation of 1,6;2,3-dianhydro-4-O-acetyl-beta-D-gulopyranose (0.45%) and 2,3-anhydro-D-mannosan (0.72%). Cyclopentenones are produced in the process of hemicellulose decomposition. The relative content of 2-cyclopenten-1-one, 2-hydroxy-3-methyl-, 2-cyclohexen-1-one, 4-(2-oxopropyl)-, and 2-cyclopenten-1-one, 2-hydroxy is 7.14%, 7.12% and 1.84%, respectively. During the pyrolysis process, lignin molecule chains are cleaved and fragments are rearranged, and the pyrolysis products are mainly phenolic compounds. The relative content of phenol, 2,6-dimethoxy- in the pyrolysis liquid of poplar reaches up to 9.61%. In addition, other phenolic compounds, such as phenol, 2-methoxy- (4.12%), phenol (4.08%), phenol, 2-methoxy-4-methyl- (2.74%), phenol, 4-methyl- (2.30%), phenol, 2-methyl- (1.90%) and phenol, 4-ethyl-2-methoxy (1.20%), can also be detected. Poplar is a kind of broadleaf timber, and the lignin in poplar is mainly composed of syringyl lignin (S-lignin) and guaiacyl lignin (G-lignin). Methoxy groups are abundant in the lignin structure. Therefore, most of phenolic compounds in pyrolysis liquid of poplar have a methoxy chain.
From Table 7, it can be seen that the influence of UF resin for particleboard is mainly on the nitrogen compounds. There is only one kind of nitric compound (1-pyrrolidinecarbonitrile) in the pyrolysis liquid of poplar, and the relative content of 1-pyrrolidinecarbonitrile is 6.44%. However, the relative content of nitric compounds in the pyrolysis liquid of particleboard is 29.84%. The pyrolysis liquid of particleboard has a higher relative content of 1-pyrrolidinecarbonitrile (11.24%), when compared with the pyrolysis liquid of poplar. From Tables 5 and 7, it can be seen that some nitric compounds in the pyrolysis liquid of particleboard, such as pyrimidine, 5-methyl- and 1,3,5-triazine-2,4,6(1H,3H,5H)-trione, 1,3,5-trimethyl-, just generated by the pyrolysis of UF resin. But others are generated by the concerted reaction of poplar and UF resin, such as 4-(1H)-pyridinone, 2,3-dihydro-1-methyl-. Thus, it can be seen that the transformation of nitrogen-containing compounds in the process of poplar pyrolysis is influenced by the interactions between UF resin and poplar. According to the existing literature and practices, the pyrolysis liquid of wood could improve the physical and chemical properties and fertility of soil, and promote plant growth. The pyrolysis liquid of particleboard contains a high content of nitric compounds, except for the main components of wood pyrolysis liquid. Nitrogen is one of a large number of elements which a plant needs, and plays an important role in plant growth. But the application of the pyrolysis liquid of particleboard in agriculture is still a new subject, worth exploring.
4. Conclusions
Pyrolysis of particleboard produces more char and less volatile matter compared to its two components, which indicates that the thermal behavior of poplar particleboard is not just a reflection of the sum of the behavior of poplar and UF resin. The activation energies of poplar and particleboard are 140–200 kJ mol−1, while the activation energy of UF resin is 150–300 kJ mol−1. The evolved gaseous products during the pyrolysis of particleboard, such as H2O, CH4, CO, and CO2 are found, and, compared to UF resin, signals of N-components produced by particleboard are wider and stronger. The influence of UF resin on the pyrolysis liquid of particleboard is mainly on nitrogen compounds. More nitrogenous compounds are detected in the pyrolysis liquid of particleboard, and the relative amount of nitrogenous compounds is 23.40% more than that in poplar pyrolysis liquid.Acknowledgements
Financial support for this work has been provided by the National Natural Science Foundation of China (31170533).References
- Y. Feng, J. Mu, S. Chen, Z. Huang and Z. Yu, BioResources, 2012, 7, 4600–4613 CrossRef CAS.
- X. C. He and Y. P. Mu, Wood Processing Machinery, 2008, 1, 50–53 Search PubMed.
- P. Girods, A. Dufour, V. Fierro, Y. Rogaume, C. Rogaume, A. Zoulalian and A. Celzard, J. Hazard. Mater., 2009, 166, 491–501 CrossRef CAS PubMed.
- K. B. Cantrell, T. Ducey, K. S. Ro and P. G. Hunt, Bioresour. Technol., 2008, 99, 7941–7953 CrossRef CAS PubMed.
- J. Chen, X. Fan, B. Jiang, L. Mu, P. Yao, H. Yin and X. Song, Bioresour. Technol., 2015, 192, 592–602 CrossRef CAS PubMed.
- P. Girods, A. Dufour, Y. Rogaume, C. A. Rogaume and A. Zoulalian, J. Hazard. Mater., 2008, 159, 210–222 CrossRef CAS PubMed.
- S. H. Chen, S. J. Li, J. Mu and Y. S. Feng, Wood Res., 2015, 60, 113–124 CAS.
- J. Mu, Z. Yu, D. Zhang and X. Jin, J. Beijing For. Univ., 2011, 33, 125–128 CAS.
- H. Wang, G. Yang and D. Zhou, J. Northeast For. Univ., 2004, 32, 55–57 Search PubMed.
- T. Nakai, S. N. Kartal, T. Hata and Y. Imamura, Build. Environ., 2007, 42, 1236–1241 CrossRef.
- X. Jiang, C. Li, Y. Chi and J. Yan, J. Hazard. Mater., 2010, 173, 205–210 CrossRef CAS PubMed.
- S. Yuan, Z. J. Zhou, J. Li, X. L. Chen and F. C. Wang, J. Anal. Appl. Pyrolysis, 2011, 92, 463–469 CrossRef CAS.
- K. Han, S. Nui and C. Lu, Process Saf. Environ. Prot., 2010, 88, 425–430 CrossRef CAS.
- J. Giuntoli, S. Arvelakis, H. Spliethoff, W. de Jong and A. H. M. Verkooijen, Energy Fuels, 2009, 23, 5695–5706 CrossRef CAS.
- D. Shen, R. Xiao, S. Gu and K. Luo, RSC Adv., 2011, 1, 1641–1660 RSC.
- F. Ferrara, A. Orsini, A. Plaisant and A. Pettinau, Bioresour. Technol., 2014, 171, 433–441 CrossRef CAS PubMed.
- L. Fiori, M. Valbusa, D. Lorenzi and L. Fambri, Bioresour. Technol., 2012, 103, 389–397 CrossRef CAS PubMed.
- K. Miura, Energy Fuels, 1995, 9, 302–307 CrossRef CAS.
- K. Miura and T. Maki, Energy Fuels, 1998, 12, 864–869 CrossRef CAS.
- J. W. De, A. Pirone and M. A. Wojtowicz, Fuel, 2003, 82, 1139–1147 CrossRef.
- T. Hirata, S. Kawamoto and A. Okuro, J. Appl. Polym. Sci., 1991, 42, 3147–3163 CrossRef CAS.
- R. Bassilakis, R. M. Carangelo and M. A. Wojtowicz, Fuel, 2011, 80, 1765–1786 CrossRef.
- N. B. Gao, A. M. Li, C. Quan, L. Du and Y. Duan, J. Anal. Appl. Pyrol., 2013, 100, 26–32 CrossRef CAS.
- S. Galvagno, S. Casu and M. Martino, J. Therm. Anal. Calorim., 2007, 88, 507–514 CrossRef CAS.
- S. Xu, J. Chen and D. Cao, Guangzhou Chem., 2006, 31, 28–31 CAS.
| This journal is © The Royal Society of Chemistry 2016 |




![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration
O stretching vibration
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N stretching vibration
N stretching vibration