Photophysical properties of luminescent silicon nanoparticles surface-modified with organic molecules via hydrosilylation†
Mari
Miyano
,
Yuichi
Kitagawa
*,
Satoshi
Wada
,
Akira
Kawashima
,
Ayako
Nakajima
,
Takayuki
Nakanishi
,
Junya
Ishioka
,
Tamaki
Shibayama
,
Seiichi
Watanabe
and
Yasuchika
Hasegawa
*
Faculty of Engineering, Hokkaido University, N13 W8, Kita-ku, Sapporo, Hokkaido 060-8628, Japan. E-mail: y-kitagawa@eng.hokudai.ac.jp; hasegaway@eng.hokudai.ac.jp; Fax: +81-11-706-7114; Tel: +81-11-706-7114
First published on 21st December 2015
Abstract
Luminescent silicon nanoparticles have attracted considerable attention for their potential uses in various applications. Many approaches have been reported to protect the surface of silicon nanoparticles and prevent their easy oxidation. Various air-stable luminescent silicon nanoparticles have been successfully prepared. However, the effect of interactions of the π-electron system with the silicon surface on the excited state properties of silicon nanoparticles is unclear. In this study, we have successfully prepared silicon nanoparticles protected with three organic compounds (styrene, 1-decene, and 1-vinyl naphthalene) and have examined their photophysical properties. The ligand π-electron systems on the silicon surface promoted the light harvesting ability for the luminescence through a charge transfer transition between the protective molecules and silicon nanoparticles and also enhanced the radiative rate of the silicon nanoparticles.
1. Introduction
As novel luminescent materials, quantum dots (QDs) have attracted considerable attention for their potential use in applications such as light-emitting diodes, solar cells, biosensors, and photodynamic therapy.1–12 Because of the quantum size effect, the energy gap in QDs can, in general, be manipulated by controlling the particle size, making it possible to control the luminescence wavelength from near ultraviolet (UV) to near infrared.13,14 At present, various nanoparticles and their luminescence properties have been reported. Shirahata reported the size tunable luminescence for Ge nanoparticles.15 Tanaka reported the aggregation of EuS nanoparticle through the interdigitation of protective molecules induced red-shifted luminescence band.16 Balaz reported the size dependent circular polarized luminescence for CdSe nanoparticles.17 In particular, silicon nanoparticles have major advantages such as being non-toxic, abundant natural resources, and inexpensive.18The visible red luminescence of silicon nanoparticles after UV irradiation was first reported by Canham in 1990.19–21 The luminescence of silicon nanoparticles is based on a quasi-direct electronic transition, which comes from the direct recombination of excited electrons and positive holes, based on the wavefunction overlap in the nanometer range, i.e., the quantum confinement effect.22 To observe quantum confinement and size effects in silicon nanoparticles, we need to prepare the silicon nanoparticles with diameters of less than 6 nm. The luminescent properties of silicon nanoparticles with this size are strongly affected by the large numbers of Si atoms with dangling bonds on the surfaces of the particles. These Si atoms can be rapidly oxidized, which induces low luminescent quantum yields.23 Therefore, the creation of air-stable luminescent silicon nanoparticles requires a surface protection method.
At present, various types of approaches to the surface protection of silicon nanoparticles have been reported, including termination strategies using chlorides,24 amines,25 and esters.26 Recently, we reported prepared luminescent silicon nanoparticles using a photochemical reaction of styrene molecules with Si–H bonds on the surface in a deoxidizing atmosphere.27 We also induced large chiroptical properties in silicon nanoparticles modified by chiral molecules.28 However, the effect of the resulting π-electron system with the silicon surface on the excited states properties of silicon nanoparticles is unclear, although the control of the excited properties of silicon nanoparticles with protective molecules is very important.
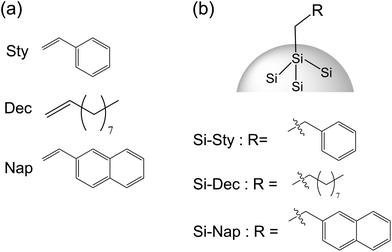
In this paper, we report the photophysical properties of silicon nanoparticles that have protective molecules bonded to surface Si atoms via hydrosilylation. These surface-modified silicon nanoparticles using three protective organic molecules were prepared (Fig. 1). The light harvesting abilities of the silicon nanoparticles were obtained by the π-electron system on their surface. Based on the light harvesting properties, we found that the nanoparticles provide luminescence related to their effective quantum size. This quantum size was observed via transmission electron microscopy (TEM) images. We found the π-electron systems on the silicon surface not only promote the light harvesting ability for the luminescence using a charge transfer transition between organic compounds and silicon nanoparticles, and but also enhance the radiation rate of silicon nanoparticles.
2. Experimental section
2.1 Materials
Tetra-ethoxysilane ES40 as a source for silicon was purchased from COLCOAT CO. LTD. Phenol resin SR-101 as a source of carbon was obtained from AIR WATER INC. Aqueous solution of maleic acid (70%) as a catalyst was purchased from Nippon Shokubai. These materials were starting materials for a synthesis of silicon nanoparticles. Hydrofluoric acid HF (48%) was purchased from Wako Pure Chemical Industries, Ltd. Nitric acid HNO3 (62%) and styrene (Sty) were purchased from Kanto Chemical Co., Inc. Chloroform was purchased from Wako Pure Chemical Industries, Ltd. 1-Decene (Dec) and 1-vinylnaphthalene (Nap) were purchased from Sigma-Aldrich. Polyethylene filter was obtained by Japan Entegris Inc. All other chemicals and solvents were reagent grade and were used without further purification.2.2 Apparatus
UV light source (wavelength at 365 nm) was used a LED365-SPT/L (Optocode Corp) for observation of acid-etching process. UV cross-linker, as a photo-assisted reactor (Funakoshi Co.) was used for the photo-chemical reaction of each organic compound with silicon surface. TEM measurements were performed by 200 kV FE-TEM (JEOL JEM-2010F) with multi-scan CCD camera (Gatan Co.).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) methanol = 3
methanol = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v, 20 ml) to the solution. After the acid etching process, the solution was filtered using a polyethylene filter (20 nm mesh) and the residue (silicon nanoparticles) was washed with 5 mL of 2% HF to eliminate the thin surface oxidation layer, followed by rinsing twice with 5 mL of purified water to completely wash off the acid. This filter with silicon nanoparticle capped by hydrogen was cut into three equal parts carefully. These three pieces were put in glass vessels and vacuum-dried at room temperature for 60 min to completely remove water.
1 v/v, 20 ml) to the solution. After the acid etching process, the solution was filtered using a polyethylene filter (20 nm mesh) and the residue (silicon nanoparticles) was washed with 5 mL of 2% HF to eliminate the thin surface oxidation layer, followed by rinsing twice with 5 mL of purified water to completely wash off the acid. This filter with silicon nanoparticle capped by hydrogen was cut into three equal parts carefully. These three pieces were put in glass vessels and vacuum-dried at room temperature for 60 min to completely remove water.
3. Results and discussion
3.1 TEM images of silicon nanoparticles with organic compounds
TEM images of prepared silicon nanoparticles with organic compounds are shown in Fig. 2. We observed these silicon nanoparticles with clear lattice fringes and the quantum size. These lattice spacing are estimated to be about 0.31 nm from TEM image, which is consistent with the typical spacing value of the Si (111) plane (0.312 nm).3.2 Electronic absorption properties of silicon nanoparticles with organic compounds
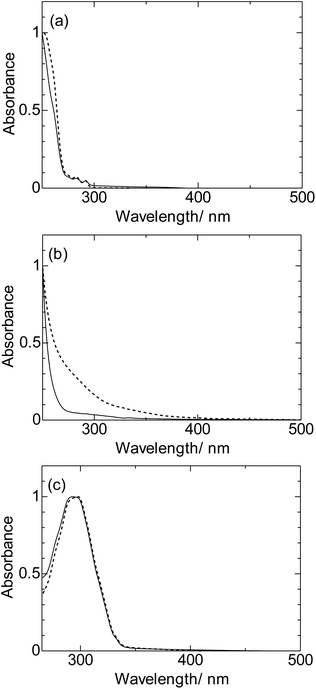
The electronic absorption spectra of organic compounds (Sty, Dec, and Nap) are shown in Fig. 3. The absorption peaks were observed for Sty (282 nm, 291 nm) and Nap (298 nm). On the other hand, that of Dec couldn't be observed at wavelength longer than 250 nm. These differences can be well explained by the effects of π-conjugation length and substituents using TD-DFT calculations. These differences can be well explained by the effects of π-conjugation length and substituents using TD-DFT calculations. The lowest excited states (S1) of Sty and Nap are composed of two electronic configurations.31 For the S1 states, the weak band for Sty and intense band for Nap can be explained by the differences between the configuration interaction degrees (Tables S1 and S3†). S1 state of Dec was expressed as a single electronic configuration, whose energy is higher than the extended π-conjugated molecule such as Sty and Nap (Table S2†).On the other hand, the intense absorption bands of surface modified silicon nanoparticles were blue-shifted (Si-Sty, Si-Dec, and Si-Nap) (Fig. 3). The blue shifted bands originate from the decrease of π-conjugation length of the organic compounds, which indicate the formation of the new electronic states due to the covalent bonds with the surface silicon atoms.
3.3 Luminescence properties of silicon nanoparticles with organic compounds
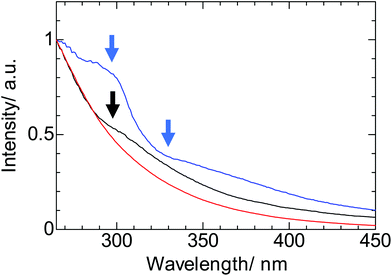
Luminescence spectra of surface-modified silicon nanoparticles (Si-Sty, Si-Dec, and Si-Nap) were shown in Fig. 4. We observed luminescence at around 700 nm for Si-Sty, Si-Dec, and Si-Nap. In the previous report, the aggregation of EuS nanoparticle through the interdigitation of protective molecules induced red-shifted luminescence band.16 On the other hand, the luminescence band of silicon nanoparticle was also slightly red-shift and broadened after four days (Fig. S6,† Si-Sty: 692 nm → 721 nm, FWHM: 3880 cm−1 → 3990 cm−1). The characteristic red-shift band may reflect the aggregation of silicon nanoparticles through the interactions between protective molecules as previously reported.16In the excitation spectra (Fig. 5, λem = 690 nm), the intense signals were observed in the ultraviolet regions. The band at 300 nm for Si-Nap is assigned to π–π* transition of Nap moiety. The characteristic shoulder bands were also observed for Si-Sty (300 nm) and Si-Nap (330 nm), respectively. These shoulder peaks can't be explained by the direct-band gap transition of silicon nanoparticles, because the absorption wavelengths are completely different from the theoretical prediction value.32 These bands are the CT transitions between silicon nanoparticles and protective molecules. This interpretation is consistent with the experimental fact that these bands are broad (FWHM: Si-Sty: 8860 cm−1, Si-Nap: 9860 cm−1), and supported by TD-DFT calculations of silicon cluster with Sty in the ESI (Fig. S4, S5 and Table S4†). This calculation suggests that the oscillator strength of the charge transfer bands is much larger than that32 of direct-band gap transition of silicon nanoparticles, in spite of the small overlapping orbitals between organic compounds and silicon nanoparticles. These facts indicate that the control of the coupling scheme and the orientation between the organic moieties and silicon nanoparticles may be one of the key factor in enhancing the light harvesting ability for the luminescence. In addition, the orientations induced the transition between the nonplanar orbitals33 of the aromatic compounds and silicon nanoparticles, which is consistent with the previous results of the large chiroptical properties with the CT transitions between chiral organic compounds and silicon nanoparticles.28 Therefore, the control of the CT transition is expected to be an important factor for not only the enhancement of light harvesting ability for the luminescence and but also a creation of a new photo-functional materials.
In order to study the influence of the protective molecules to the excited states properties of silicon nanoparticles, the luminescence quantum yields and time-resolved luminescence decays (Fig. 6) were measured for the red-luminescent silicon nanoparticles (Si-Sty, Si-Dec, and Si-Nap). The luminescence quantum yields of Si-Sty and Si-Nap were estimated to be 4.3% and 4.4%, respectively (λex = 380 nm). These quantum yields are based on both the excitations corresponding to CT transitions and the direct transitions of silicon nanoparticles (Fig. 5). On the other hand, the smaller luminescence quantum yield was observed for Si-Dec excited by the direct transitions of silicon nanoparticles. The luminescence decay profiles of silicon nanoparticles exhibit multiexponential decay curves (Fig. S10†). The lifetimes are summarized in Table 1. Although the multiexponential decays indicate the presence of distinct populations of emitting nanoparticles, which may originate from distinct nanoparticles size or the ratio of organic compounds on the surface to silicon nanoparticles, the results indicate that the calculated lifetimes of Si-Dec are longer than those of Si-Sty and Si-Nap.
 | ||
| Fig. 6 Luminescence decay profiles of surface-modified silicon nanoparticles in CHCl3 (Si-Sty: black line, Si-Dec: red line, Si-Nap: blue line). | ||
These results indicates the radiation rate of Si-Dec is slower than those of Si-Sty and Si-Nap. We consider that surface modification by π-electron system as Sty or Nap promotes the enhancement of the radiation rate of silicon nanoparticle. The detail analysis of specific interaction of π-electron orbitals and silicon orbitals are now in progress.
4. Conclusions
We successfully prepared silicon nanoparticles with three organic compounds, and examined their photophysical properties. The π-electron systems on the silicon surface promote the light harvesting ability for the luminescence and enhance the radiation rate. The results indicate that luminescent silicon nanoparticles with a π-electron system are expected to be an important factor for creating strongly luminous silicon nanoparticles.Acknowledgements
This work was party supported by Grants-in-Aid for Scientific Research on Innovative Areas of New Polymeric Materials Based on Element-Blocks (No. 2401) (24102012) of the Ministry of Education, Culture, Sports, Science and Technology (MEXT) of Japan.References
- M. K. Kwon, J. Y. Kim, B. H. Kim, I. K. Park, C. Y. Cho, C. C. Byeon and S. J. Park, Surface-Plasmon-Enhanced Light-Emitting Diodes, Adv. Mater., 2008, 20, 1253–1257 CrossRef CAS.
- L. Qian, Y. Zheng, J. Xue and P. H. Holloway, Stable and Efficient Quantum-Dot Light-Emitting Diodes based on Solution-Processed Multilayer Structures, Nat. Photon., 2011, 5, 543–548 CrossRef CAS.
- H. S. Jung, Y. J. Kim, S. W. Ha and J. K. Lee, White Light-Emitting Diodes Using Thermally and Photochemically Stable Fluorescent Silica Nanoparticles as Color-Converters, J. Mater. Chem. C, 2013, 1, 5879–5884 RSC.
- P. Reineck, G. P. Lee, D. Brick, M. Karg, P. Mulvaney and U. Bach, A Solid-State Plasmonic Solar Cell via Metal Nanoparticle Self-Assembly, Adv. Mater., 2012, 24, 4750–4755 CrossRef CAS PubMed.
- L. Yang, S. Pillai and M. A. Green, Can Plasmonic Al Nanoparticles Improve Absorption in Triple Junction Solar Cells?, Sci. Rep., 2015, 5, 11852 CrossRef CAS PubMed.
- W. Zhang, M. Saliba, S. D. Stranks, Y. Sun, X. Shi, U. Wiesner and H. J. Snaith, Enhancement of Perovskite-Based Solar Cells Employing Core–Shell Metal Nanoparticles, Nano Lett., 2013, 13, 4505–4510 CrossRef CAS PubMed.
- S. Myung, A. Solanki, C. Kim, J. Park, K. S. Kim and K. B. Lee, Graphene-Encapsulated Nanoparticle-Based Biosensor for the Selective Detection of Cancer Biomarkers, Adv. Mater., 2011, 23, 2221–2225 CrossRef CAS PubMed.
- J. N. Anker, W. P. Hall, O. Lyandres, N. C. Shah, J. Zhao and R. P. Van Duyne, Biosensing with Plasmonic Nanosensors, Nat. Mater., 2008, 7, 442–453 CrossRef CAS PubMed.
- T. Sun, Y. Zhang, B. Pang, D. Hyun, M. Yang and Y. Xia, Engineered Nanoparticles for Drug Delivery in Cancer Therapy, Angew. Chem., Int. Ed., 2014, 53, 12320–12364 CAS.
- B. Cao, M. Yang, Y. Zhu, X. Qu and C. Mao, Stem Cells Loaded With Nanoparticles as a Drug Carrier for in Vivo Breast Cancer Therapy, Adv. Mater., 2014, 26, 4627–4631 CrossRef CAS PubMed.
- K. P. Tamarov, L. A. Osminkina, S. V. Zinovyev, K. A. Maximova, J. V. Kargina, M. B. Gongalsky, Y. V. Ryabchikov, A. Al-Kattan, A. P. Sviridov, M. Sentis, A. V. Ivanov, V. N. Nikiforov, A. V. Kabashin and V. Y. Timoshenko, Radio Frequency Radiation-induced Hyperthermia using Si Nanoparticle-based Sensitizers for Mild Cancer Therapy, Sci. Rep., 2014, 4, 7034 CrossRef CAS PubMed.
- M. G. Bobo, Y. Mir, C. Rouxel, D. Brevet, I. Basile, M. Maynadier, O. Vaillant, O. Mongin, M. B. Desce, A. Morere, M. Garcia, D. Jean-Olivier and R. Laurence, Mannose-Functionalized Mesoporous Silica Nanoparticles for Efficient Two-Photon Photodynamic Therapy of Solid Tumors, Angew. Chem., Int. Ed., 2011, 50, 11425–11429 CrossRef PubMed.
- J. Zheng, P. R. Nicovich and R. M. Dickson, Highly Fluorescent Noble-Metal Quantum Dots, Annu. Rev. Phys. Chem., 2007, 58, 409–431 CrossRef CAS PubMed.
- V. K. A. Sreenivasan, A. V. Zvyagin and E. M. Goldys, Luminescent Nanoparticles and their Applications in the Life Sciences, J. Phys.: Condens. Matter, 2013, 25, 194101 CrossRef PubMed.
- N. Shirahata, D. Hirakawa, Y. Masuda and Y. Sakka, Size-Dependent Color Tuning of Efficiency luminescent Germanium nanoparticles, Langmuir, 2013, 29, 7401–7410 CrossRef CAS PubMed.
- A. Tanaka, H. Kamikubo, M. Kataoka, Y. Hasegawa and T. Kawai, Size-Controlled Aggregation of Cube-Shaped EuS Nanocrystals with Magneto-Optic Properties in Solution Phase, Langmuir, 2011, 27, 104–108 CrossRef CAS PubMed.
- U. Tohgha, K. Deol, A. Porter, S. Bartko, J. K. Choi, B. M. Leonard, K. Varga, J. Kubelka, G. Muller and M. Balaz, Ligand Induced Circular Dichroism and Circularly Polarized Luminescence in CdSe Quantum Dots, ACS Nano, 2013, 7, 11094–11102 CrossRef CAS PubMed.
- J. L. Pelley, A. S. Daar and M. A. Saner, State of Academic Knowledge on Toxicity and Biological Fate of Quantum Dots, Toxicol Sci., 2009, 112, 276–296 CrossRef CAS PubMed.
- L. T. Canham, Silicon Quantum Wire Array Fabrication by Electrochemical and Chemical Dissolution of Wafers, Appl. Phys. Lett., 1990, 57, 1046–1048 CrossRef CAS.
- X. Cheng, S. B. Lowe, P. J. Reece and J. J. Gooding, Colloidal silicon quantum dots: from preparation to the modification of self-assembled monolayers (SAMs) for bio-applications, Chem. Soc. Rev., 2014, 43, 2680–2700 RSC.
- M. Montalti, A. Cantelli and G. Battistelli, Nanodiamonds and silicon quantum dots: ultrastable and biocompatible luminescent nanoprobes for long-term bioimaging, Chem. Soc. Rev., 2015, 44, 4853–4921 RSC.
- N. Koshida and N. Matsumoto, Fabrication and Quantum Properties of Nanostructured Silicon, Mater. Sci. Eng., R, 2003, 40, 169–205 CrossRef.
- D. Kovalev, E. Gross, J. Diener, V. Timoshenko and Y. Fujii, Photodegradation of Porous Silicon induced by Photogenerated Singlet Oxygen Molecules, Appl. Phys. Lett., 2004, 85, 3590–3592 CrossRef CAS.
- M. A. Islam, T. K. Purkait and J. G. C. Veinot, Chloride Surface Terminated Silicon Nanocrystal Mediated Synthesis of Poly(3-hexylthiophene), J. Am. Chem. Soc., 2014, 136, 15130–15133 CrossRef CAS PubMed.
- J. H. Ahire, Q. Wang, P. R. Coxon, G. Malhotra, R. Brydson, R. Chen and Y. Chao, Highly Luminescent and Nontoxic Amine-Capped Nanoparticles from Porous Silicon: Synthesis and Their Use in Biomedical Imaging, ACS Appl. Mater. Interfaces, 2012, 4, 3285–3292 CAS.
- W. Biesta, B. van Lagen, V. S. Gevaert, A. T. M. Marcelis, J. M. J. Paulusse, M. W. F. Nielen and H. Zuilhof, Preparation, Characterization, and Surface Modification of Trifluoroethyl Ester-Terminated Silicon Nanoparticles, Chem. Mater., 2012, 24, 4311–4318 CrossRef CAS.
- M. Miyano, S. Endo, H. Takenouchi, S. Nakamura, Y. Iwabuti, O. Shiino, T. Nakanishi and Y. Hasegawa, Novel Synthesis and Effective Surface Protection of Air-Stable Luminescent Silicon Nanoparticles, J. Phys. Chem. C, 2014, 118, 19778–19784 CAS.
- M. Miyano, T. Nakanishi, S. Wada, Y. Kitagawa, A. Kawashima, K. Fushimi, Y. Morisaki, Y. Chujo and Y. Hasegawa, Luminescent Silicon Nanoparticles Surface-Modified with Chiral Molecules, J. Photopolym. Sci. Technol., 2015, 28, 255–260 CrossRef CAS.
- C. T. Lee, W. T. Yang and R. G. Parr, Development of the Colle–Salvetti Correlation-Energy formula into a Functional of the Electron-Density, Phys. Rev. B: Condens. Matter, 1988, 37, 785–789 CrossRef CAS.
- A. D. Becke, Density-Functional Thermochemistry 0.3. The Role of Exact Exchange, J. Chem. Phys., 1993, 98, 5648–5652 CrossRef CAS.
- A. L. Sklar, Rev. Electronic Absorption Spectra of Benzene and Its Derivatives, Mod. Phys., 1942, 14, 232 CrossRef CAS.
- C. Meier, A. Gondorf, S. Lüttjohann, A. Lorke and H. Wiggers, Silicon Nanoparticles: Absorption, Emission, and the Nature of the Electronic Bandgap, J. Appl. Phys., 2007, 101, 103112 CrossRef.
- L. Bednárová, P. Malon and P. Bour, Spectroscopic Properties of the Nonplanar Amide Group: a Computational Study, Chirality, 2007, 10, 775–786 CrossRef PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5pp00364d |
| This journal is © The Royal Society of Chemistry and Owner Societies 2016 |