Double polymer sheathed carbon nanotube supercapacitors show enhanced cycling stability†
Wenqi
Zhao
a,
Shanshan
Wang
b,
Chunhui
Wang
a,
Shiting
Wu
c,
Wenjing
Xu
c,
Mingchu
Zou
c,
An
Ouyang
c,
Anyuan
Cao
*c and
Yibin
Li
*a
aCentre for Composite Materials and Structures, Harbin Institute of Technology, Harbin 150080, P. R. China. E-mail: liyibin@hit.edu.cn
bSchool of Marine Science and Technology, Harbin Institute of Technology at Weihai, Weihai 264209, P. R. China
cDepartment of Materials Science and Engineering, College of Engineering, Peking University, Beijing 100871, P. R. China. E-mail: anyuan@pku.edu.cn
First published on 20th November 2015
Abstract
Pseudo-materials are effective in boosting the specific capacitance of supercapacitors, but during service their degradation may also be very strong, causing reduced cycling stability. Here, we show that a carbon nanotube sponge grafted by two conventional pseudo-polymer layers in sequence can serve as a porous supercapacitor electrode with significantly enhanced cycling stability compared with single polymer grafting. Creating conformal polymer coatings on the nanotube surface and the resulting double-sheath configuration are important structural factors leading to the enhanced performance. Combining different polymers as double sheaths as reported here might be a potential route to circumvent the dilemma of pseudo-materials, and to simultaneously improve the capacitance and stability for various energy storage devices.
1 Introduction
Supercapacitors represent an important type of energy storage device which can offer a large specific capacitance and high power density.1–5 It has been a common strategy to improve the capacitance of supercapacitors by introducing pseudo-materials which have redox reactions with electrolytes during charging and discharging cycles.6–9 To this end, numerous hybrid or composite electrodes that contain pseudo-materials grafted on a conductive scaffold have been proposed, including those based on carbon nanotube (CNT) networks10–12 or graphene oxide aerogels.13–15 Introduction of pseudo-polymers and oxides in general shows a considerable enhancement in the specific capacitance, but the presence of these materials also may cause reduced cycling stability due to degradation in various electrolytic environments and undesired structural evolution over many reaction cycles.16,17 Although both the specific capacitance and cycling stability are important criteria for evaluating new electrode systems, most of the research in the field have been focused on the former (achieving maximum specific capacitance) whereas studies on solving the stability issue are relatively few. Recently, Liu et al. utilized a deposited thin carbonaceous shell to enhance the cycling stability of conductive polymer electrodes.18Our group has investigated the supercapacitor properties of CNT sponges by coating conventional polymers (e.g. polypyrrole, PPy) or oxides (e.g. MnO2) onto the surface of CNT networks.19,20 A noticeable increase of the specific capacitance was observed in the CNT–PPy and CNT–PPy–MnO2 electrodes, owing to the synergistic effects stemming from the embedded conductive CNT network and the as-coated pseudo-layer interfacing with electrolytes. Furthermore, the porous CNT sponges can be compressed to large strains (up to 80% volume reduction) with enhanced volume-specific capacitance.21 These results prove that our pseudo-material-grafted CNT sponges have strong potential in developing high performance and flexible supercapacitors.
Here, we show that the cycling stability, one of the most important considerations of supercapacitors, could be significantly enhanced by creating smooth and uniform double-layer polymer coatings throughout the CNT sponge. Although individual polymer layers do not show high stability, a simple combination of two different such polymers leads to much higher capacitance retention after thousands of cycles without sacrificing the specific capacitance. We attribute the enhanced stability to two structurally related factors: (1) uniform polymer coating throughout the CNT network and (2) the unique double-sheath configuration obtained by sequential coating of two polymers.
2 Results and discussion
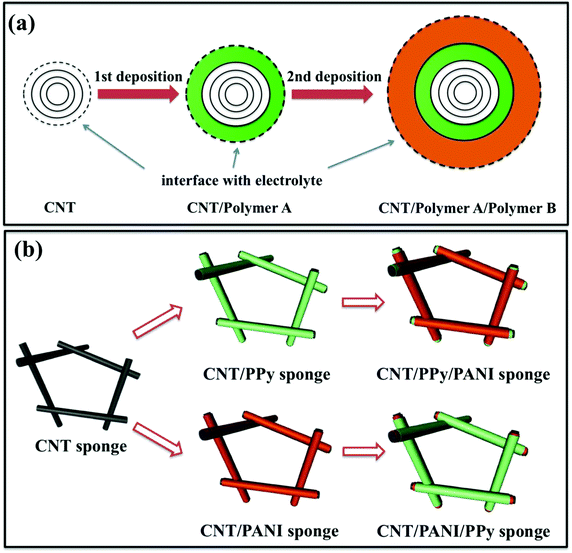
A CNT sponge is a bulky, highly porous material containing three-dimensionally overlapped multi-walled nanotubes with diameters of 20–40 nm and lengths of tens of μm, as reported by our group a few years ago.22 These light-weight and conductive CNT sponges are ideal bulky electrode materials for various energy devices such as supercapacitors and batteries. Here, our basic idea is to utilize the bare CNT surfaces among the as-grown sponge and to deposit the desired materials onto the CNTs for surface modification. As illustrated in Fig. 1a, first one polymer (A) was coated onto the surface of a CNT and then another polymer (B) was coated onto polymer A, forming a double-layer coating with a certain thickness in each layer. This sequential deposition ensures that only the polymer A can be in direct contact with the inside of the CNTs, while the polymer B only makes contact with the polymer A, creating a strict double-layer configuration without any mixing in the respective layers. The resulting structure containing two different polymer layers is treated as a double-sheathed CNT. Correspondingly, the outermost surface (interface with the electrolyte) changes from the CNT surface (for as-grown sponge) to the polymer A (after the first coating) and the polymer B in the final double-sheathed structure.Both polymer layers (A and B) were coated onto the CNTs by the electrodeposition method using the sponge as one electrode immersed in solution containing targeted monomers in a three-electrode system (see the Experimental section for details). This ensures that the entire sponge can be deposited by the polymers uniformly, and also the deposition process does not disturb the initial CNT network. Finally, we obtained a three dimensional (3D) CNT network covered by a uniform double-sheath. By depositing PPy and polyaniline (PANI) respectively in different sequences, we have obtained two resulting configurations including a CNT/PPy/PANI and a CNT/PANI/PPy composite sponge with a reversed sheath structure, as illustrated in Fig. 1b.
Fig. 2a and b show the scanning electron microscopy (SEM) images of an original CNT sponge and a double-sheathed CNT/PPy/PANI sponge, respectively. To examine the internal structure, the samples were broken and inner cross-sections were imaged. Due to the random overlapping of CNTs, every nanotube is suspended in the 3D network, which ensures a uniform polymer coating around the nanotube surface. As a result, all CNTs were coated by polymers and the tube diameters increased from 20–40 nm to 60–140 nm (Fig. 2b). Corresponding to the diameter increase, the mass loadings of polymers were controlled to be about 40 wt% PPy (1.86 mg) and 60 wt% PANI (2.8 mg), resulting in a total polymer loading of ∼70 wt% (4.6 mg) relative to the initial sponge weight (6.6 mg). The same polymer loadings were adopted in samples for subsequent electrochemical tests (both CNT/PPy/PANI and CNT/PANI/PPy sponges). Because the sponge has an open-porous structure, monomer solutions could infiltrate through the channels between CNTs and access the inner part, leading to uniform deposition throughout the bulk sponge.
Enlarged view of the broken sponge reveals distinct characteristics of the double-sheathed structure. Many flexible thin wires are exposed in the cross-section; these are the inner CNTs protruding from the polymer-wrapped segments (CNT/PPy/PANI) with diameters of about 130 nm (Fig. 2c and d) and the schematic of these ternary nanotubes is shown in Fig. 2e. It is interesting to note that on some CNTs, close to their exposed tips, there is a short segment with an intermediate diameter (∼60 nm), corresponding to the CNT/PPy structure after the first deposition process. Appearing of such step-wise structure (having different diameters) is due to different broken regions in the PPy or PANI layer, along the axis of CNTs (illustrated in Fig. 2e). This distinct micro morphology also proves the formation of the double-sheath configuration consisting of two different polymer layers coated along the CNTs. SEM characterization of bare CNT, CNT/PANI and CNT/PPy/PANI sponges reveals distinct structures of single polymer-sheathed (a uniform smooth coating throughout the CNT network) and double-sheathed sponges (stepwise double-layer coating) in which the second layer is prevented from coming in direct contact with inner CNTs (ESI, Fig. S1†). Similarly, we also obtained a reversed double-sheath sample, the CNT/PANI/PPy sponge, with the PANI layer inside and the PPy layer outside (ESI, Fig. S2a and S2b†). Transmission electron microscopy (TEM) images of the as-grown CNTs show a typical multi-walled structure with outer diameters of 33 nm and inner cavities of 15 nm (Fig. S3a†). After depositing a single PANI layer, the diameter increases to 78 nm (Fig. S3b†). It can be seen that the PANI layer was coated on the CNTs smoothly without forming gaps at the interface. For the CNT/PANI/PPy sponge, one cannot distinguish the interface between the two polymer layers, but sometimes a necklace like structure was observed possibly due to the non-uniform deposition of PPy on the PANI layer (Fig. S3c and S3d†). Also, the double polymer sheathed CNT sponges still possess high flexibility (can be bent or rolled up), indicating potential applications as flexible or compressible supercapacitors (Fig. S2a†).
Successful coating of polymers has been confirmed by X-ray diffraction (XRD) and Raman characterization. The original CNT sponge only shows a broad band at about 20° coming from the multi-walled nanotubes (Fig. S4†). For CNT/PANI and CNT/PPy/PANI sponges, we observe pronounced peaks centered at 25.8°, which are attributed to the periodicity parallel and perpendicular to the polymer chains.23 Raman spectra of the composites show characteristic peaks of PANI and PPy in the CNT/PANI and CNT/PPy/PANI sponges, respectively (Fig. S5†). For example, in the CNT/PANI sponge, there are several distinct peaks corresponding to PANI including those centered at 1571 cm−1 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching), 1348 cm−1 (C–N+ stretching) and 1169 cm−1 (in plane C–H bending), while in the CNT/PPy/PANI sponge, peaks at 1004 cm−1 (ring deformation), 1082 cm−1 (in-plane C–H bending), 1355 and 1410 cm−1 (C–N stretching) and 1583 cm−1 (C
C stretching), 1348 cm−1 (C–N+ stretching) and 1169 cm−1 (in plane C–H bending), while in the CNT/PPy/PANI sponge, peaks at 1004 cm−1 (ring deformation), 1082 cm−1 (in-plane C–H bending), 1355 and 1410 cm−1 (C–N stretching) and 1583 cm−1 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching) appear due to the presence of PPy. The Raman spectra of the two composite sponges are in well accordance with previous characterizations on pure PANI and PPy, respectively.24,25
C stretching) appear due to the presence of PPy. The Raman spectra of the two composite sponges are in well accordance with previous characterizations on pure PANI and PPy, respectively.24,25
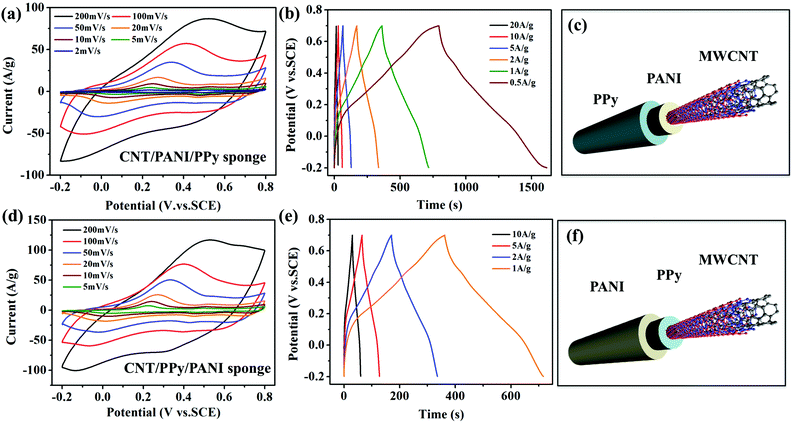
The electrochemical properties of two double-sheathed sponges in different sequences (CNT/PANI/PPy and CNT/PPy/PANI) have been characterized by cyclic voltammograms (CVs) and galvanostatic charge/discharge curves, respectively (Fig. 3). First of all, polymer coatings in either sequence can make an excellent supercapacitor electrode with large-area CV curves (scan rates of 2 to 200 mV s−1, voltage window of 1 V) and long charge/discharge periods at different current densities. Meanwhile, the CV curves of the two samples show similar shapes with redox peaks, indicating that the two polymers give rise to pseudo-capacitance of the electrode (Fig. 3a and d). Negligible voltage drop (IR drop) can be seen from the galvanostatic curves, indicating a low internal resistance which is important for energy electrodes (Fig. 3b and e). With decreasing current density, the discharge time increases clearly indicating a good rate capability. Based on galvanostatic discharge curves, the calculated specific capacitance reaches 395 F g−1 and 596 F g−1 (at 1 A g−1) for the CNT/PANI/PPy and CNT/PPy/PANI sponges, respectively. Compared with bare CNT sponges typically having capacitances below 30 F g−1,21 coating the two polymers has significantly improved the electrode performance. The above results also suggest that the specific capacitance also depends on which polymer is coated outside and in contact with the electrolyte.
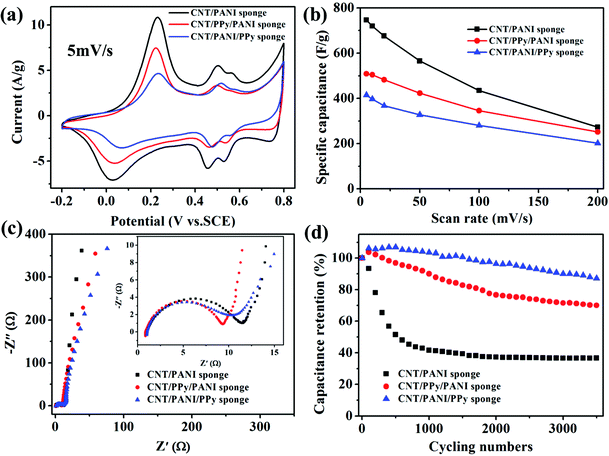
So what is the advantage of making a double-sheathed structure versus single polymer coating? To investigate this, we have compared the CV characteristics of a single-sheath sponge (CNT/PANI) and the above mentioned double-sheath samples (with the same loadings of PPy and PANI). At a scan rate of 5 mV s−1, the redox peaks appear more pronounced since electron transportation at a slow scan rate is more efficient (Fig. 4a). All three samples have similar redox peaks, but the CNT/PANI sponge shows the largest areas enclosed by the CV curve and hence the highest specific capacitance (746 F g−1 at 5 mV s−1). Meanwhile, the CNT/PPy/PANI and CNT/PANI/PPy sponges exhibit lower capacitances of 509 F g−1 and 415 F g−1, respectively. If we compare the CNT/PANI and CNT/PPy/PANI sponges, it shows that introducing a PPy intermediate layer has decreased the capacitance to about the average value of the CNT/PANI and CNT/PPy sponges (ca. 300 F g−1).19 Interestingly, the double-layer sponge shows an average effect, with an overall specific capacitance in between the single PPy (lower bound) and PANI coating (higher bound), due to the physical combination of these two polymers (with different pseudo-capacitances).18,26 Moreover, we studied the relationship between the deposition time of PANI on the CNT/PPy sponge (50 wt%) and the electrochemical performance (Fig. S6†). The results show that the specific capacitance depends on the PANI deposition time, in which the capacitance increases to about 509 F g−1 at 160 min (∼40 wt% PANI), and then becomes saturated upon excessive PANI loading due to the increase in material weight and less efficient charge collection.19 However, when the scan rate increases from 5 to 200 mV s−1, their specific capacitances become close, and the capacitance retention at 200 mV s−1 of the double-sheathed samples (48.9% of CNT/PPy/PANI and 48.7% of CNT/PANI/PPy sponge) is higher than the single-sheathed CNT/PANI sponge (36.2%) (Fig. 4b).
We have conducted electrochemical impedance spectroscopy (EIS) tests to characterize the internal resistances of polymer-sheathed CNT sponges. Fig. 4c shows the Nyquist plots of EIS spectra for the CNT/PANI, CNT/PPy/PANI and CNT/PANI/PPy sponges, respectively. All spectra are composed of a typical semicircle in the high frequency region and a straight line in the low frequency region. The CNT/PANI sponge shows the biggest diameter of the semicircle in the high frequency region, indicating a relatively large charge transfer resistance (Rct). In comparison, the two double-sheathed samples show a decreased semicircle diameter and hence lowered internal resistances. The results suggest that the intermediate PPy layer at the CNT–polymer interface could improve the electrode conductivity by providing superb highways for fast electron transportation and electrolyte ions diffusion. This also leads to increased capacitance retention in the CNT/PPy/PANI and CNT/PANI/PPy sponges at different scan rates (Fig. 4b).
Notably, we found that the double-sheath structure could improve the cycling stability, which is an important criterion for supercapacitor applications. Composite sponges were tested at a scan rate of 200 mV s−1 over 3500 cycles and their CV curves were recorded (Fig. 4d). Although the CNT/PANI sponge has the highest specific capacitance, it shows a quick decay during initial cycles, with a capacitance retention of 41.5% at the 1000th cycle and 36.5% at the 3500th cycle, respectively. In comparison, samples with two polymer layers exhibit much enhanced cycling stability. Specifically, the CNT/PPy/PANI sponge shows a capacitance retention of 90.1% (1000th cycle) and 70.0% (3500th cycle), much higher than that of the CNT/PANI sponge. For the CNT/PANI/PPy sponge with reversed sheath sequence, the capacitance retention even reaches 103.6% (1000th cycle) and 88.0% (3500th cycle). This is because the PPy layer, although with lower pseudo-capacitance than PANI, has better stability as reported in the literature.26 Extended cycling tests on a CNT/PANI/PPy sponge showed excellent stability with nearly 60% retention of original capacitance even after 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles (Fig. S7†).
000 cycles (Fig. S7†).
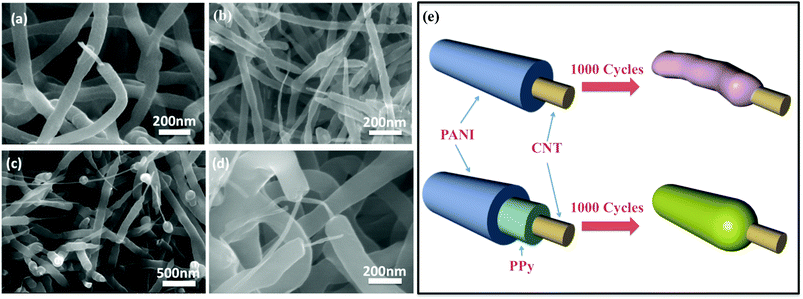
To study the mechanism for enhanced cyclic stability, the structural evolution in the polymer-sheathed sponges before and after 1000 CV cycles was characterized by SEM (Fig. 5 and S8†). We can see obvious difference in the final structure of the sponges coated with single or double-layer polymers. For the CNT/PANI sponge which showed quick degradation, the diameter of core–shell nanostructures decreased from about 100 nm to 50 nm, due to the consumption of the PANI sheath (Fig. 5a and b). Also, the PANI surface appears rougher than the original smooth core–shell structures. However, for the CNT/PPy/PANI sponge, the polymer surface remains smooth with negligible decrease in the core–shell diameter (Fig. 5c and d). Such good maintenance of the micro structure and polymer morphology should be the underlying factor for the improved cycling stability in the CNT/PPy/PANI sponge. This phenomenon suggests that introducing an intermediate PPy layer is favorable for limiting the consumption of pseudo-polymers and retaining their original morphology (Fig. 5e).
3 Conclusion
We have synthesized double polymer-sheathed CNT sponges adopting two conventional pseudo-polymers (PANI and PPy) with a controlled layer sequence, and obtained CNT/PPy/PANI and CNT/PANI/PPy composite sponges correspondingly. Systematic electrochemical tests show that the double-sheath structure significantly improves the cycling stability, with much higher capacitance retention than the single-sheath sample. The mechanism study reveals that the maintenance of the microstructure in the double-sheathed sponge is an underlying factor for improved stability. Combining different polymers and designing double or multiple sheath structures in the porous electrodes might be a potential route to simultaneously improve the capacitance and stability for various energy storage devices.4 Experimental section
4.1. Synthesis of CNT sponges
CNT sponges were synthesized by chemical vapor deposition (CVD) using ferrocene and 1,2-dichlorobenzene as the catalyst and carbon precursor. A solution of ferrocene in dichlorobenzene (0.06 g mL−1) was injected into the CVD furnace by using a syringe pump at a feeding rate in the range of 0.1–0.25 mL min−1, and carried into the reaction zone by Ar/H2 (2000 and 300 mL min−1, respectively) to grow sponges. The reaction temperature was set as 860 °C, and the CVD time was 4 h. A quartz sheet was placed in the reaction zone to deposit and collect sponge samples. The as-synthesized sponges were peeled off from the substrate.4.2. Fabrication of CNT/PANI core–shell sponges
An as-grown bulk CNT sponge block was directly used as the working electrode in the potential range from −0.2 to 0.8 V at a sweep rate of 40 mV s−1 for 100–500 cycles in 1 M H2SO4 and 0.05 M aniline aqueous solution. The electrodeposition of PANI was performed using a three-electrode electrochemical workstation (CHI660D Instruments, Shanghai, China). A Pt wire and a saturated calomel electrode (SCE) were employed as the counter and reference electrodes, respectively. Subsequent to electrodeposition, the electrode was washed with distilled water several times until the pH of the water is more than 5, and then freeze-dried to maintain the porous structure.4.3. Fabrication of CNT/PPy/PANI core–double-shell sponges
An as-grown bulk CNT sponge block was immersed in a Py/acetone solution with a concentration in the range 0.1–1 M for 0.5 h. Then the CNT sponge with Py monomers adsorbed on the surface of each individual CNT was directly used as the working electrode under a potential of 0.8 V in 0.3 M NaClO4 aqueous solution. The electropolymerization of Py was performed using a three-electrode electrochemical workstation (CHI660D Instruments, Shanghai, China). A Pt wire and Ag/AgCl were used as the counter and reference electrodes, respectively. A typical electropolymerization time was 5–10 min. After the electropolymerization process, the crosslinked CNT/PPy core–shell sponge was rinsed with distilled water and then attached the PANI shell also through electrodeposition according to step 2.4.4. Fabrication of CNT/PANI/PPy core–shell sponges
According to step 2 CNT/PANI sponge can be obtained, then according to step 3 PPy can be electrodeposited on the CNT/PANI sponge directly.4.5. Material characterization
The microstructure and morphology of samples including CNT sponges before and after compression, and AC-CNT sponges before and after testing, were characterized using SEM (Hitachi S4800) and TEM (FEI G2 T20, 200 kV). Raman spectra were recorded with a micro-Raman spectrometer (Renishaw inVia plus). The structures of the prepared sponges were analyzed by X-ray diffraction in 2θ range from 10° to 80° at room temperature.4.6. Electrochemical measurements
Electrochemical characterization of the sponges including cyclic voltammetry (CV), galvanostatic charge/discharge and electrochemical impedance spectroscopy (EIS) was carried out in 1 M H2SO4 electrolyte with the same equipment in a three-electrode cell as in the electrodeposition. The CV curves were measured with different scan rates from 2–200 mV s−1 between −0.2 and 0.8 V. EIS measurements were carried out in the frequency range from 100 kHz to 0.01 Hz at open circuit potential with an ac perturbation of 5 mV. The specific capacitance of the sponge electrode (Cs) was calculated from the CV curves and discharging curves according to eqn (1) and (2), respectively.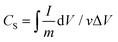 | (1) |
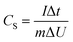 | (2) |
Acknowledgements
This work was financially supported by the National Nature Science Foundation of China (NO. 51325202). Y. Li acknowledges financial support from the Natural Science Foundation of China (NSFC 11272109) and the PhD. Programs Foundation of Ministry of Education of China (20122302110065). S. Wang acknowledges funding support from Beijing National Laboratory for Molecular Sciences (BNLMS), NSFC 21301039, HIT.NSRIF.2011096, Promotive Research Fund for Young and Middle-aged Scientists of Shandong Province (BS2013HZ021) and Technology and Development Program of Weihai.Notes and references
- L. Zhang and X. Zhao, Carbon-based materials as supercapacitor electrodes, Chem. Soc. Rev., 2009, 38, 2520–2531 RSC
.
- M. Winter and R. J. Brodd, What are batteries, fuel cells, and supercapacitors?, Chem. Soc. Rev., 2004, 104, 4245–4269 CrossRef CAS
.
- Y. Gogotsi, Energy storage wrapped up, Nature, 2014, 509, 568–570 CrossRef CAS PubMed
.
- Z. Yang, J. Deng, X. Chen, J. Ren and H. Peng, A Highly Stretchable, Fiber-Shaped Supercapacitor, Angew. Chem., Int. Ed., 2013, 52, 13453–13457 CrossRef CAS PubMed
.
- X. Chen, L. Qiu, J. Ren, G. Guan, H. Lin, Z. Zhang, P. Chen, Y. Wang and H. Peng, Novel Electric Double-Layer Capacitor with a Coaxial Fiber Structure, Adv. Mater., 2013, 25, 6436–6441 CrossRef CAS PubMed
.
- X. Lu, T. Zhai, X. Zhang, Y. Shen, L. Yuan, B. Hu, L. Gong, J. Chen, Y. Gao, J. Zhou, Y. Tong and Z. Wang, WO3−x@Au@MnO2 core–shell nanowires on carbon fabric for high-performance flexible supercapacitors, Adv. Mater., 2012, 24, 938–944 CrossRef CAS PubMed
.
- Q. Wu, Y. Xu, Z. Yao, A. Liu and G. Shi, Graphene/polyaniline nanofiber composite films, ACS Nano, 2010, 4, 1963–1970 CrossRef CAS PubMed
.
- B. Choi, S. Chang, H. Kang, C. Park, H. Kim, W. Hong, S. Lee and Y. Huh, High performance of a solid-state flexible asymmetric supercapacitor based on grapheme films, Nanoscale, 2012, 4, 4983–4988 RSC
.
- T. Brezesinski, J. Wang, S. H. Tolbert and B. Dunn, Ordered mesoporous α-MoO3 with iso-oriented nanocrystalline walls for thin-film pseudocapacitors, Nat. Mater., 2010, 9, 146–151 CrossRef CAS PubMed
.
- H. Lin, L. Li, J. Ren, Z. Cai, L. Qiu, Z. Yang and H. Peng, Conducting polymer composite film incorporated with aligned carbon nanotubes for transparent, flexible and efficient supercapacitor, Sci. Rep., 2013, 3, 1353–1358 Search PubMed
.
- C. Peng, S. Zhang, X. Zhou and G. Z. Chen, Unequalisation of electrode capacitances for enhanced energy capacity in asymmetrical supercapacitors, Energy Environ. Sci., 2010, 3, 1499–1502 Search PubMed
.
- Y. Hou, Y. Cheng, T. Hobson and J. Liu, Design and synthesis of Hierarchical MnO2 nanospheres/carbon nanotubes/conducting polymer ternary composite for high performance electrochemical electrodes, Nano Lett., 2010, 10, 2727–2733 CrossRef CAS PubMed
.
- Y. Zhao, J. Liu, Y. Hu, H. Cheng, C. Hu, C. Jiang, L. Jiang, A. Cao and L. Qu, Highly compression-tolerant supercapacitor based on polypyrrole-mediated grapheme foam electrodes, Adv. Mater., 2013, 25, 591–595 CrossRef CAS PubMed
.
- B. G. Choi, M. Yang, W. H. Hong, J. W. Choi and Y. S. Huh, 3D macroporous grapheme frameworks for supercapacitors with high energy and power densities, ACS Nano, 2012, 6, 4020–4028 CrossRef CAS PubMed
.
- G. Yu, L. Hu, N. Liu, H. Wang, M. Vosgueritchian, Y. Yang, Y. Cui and Z. Bao, Enhancing the supercapacitor performance of grapheme/MnO2 nanostructured electrodes by conductive wrapping, Nano Lett., 2011, 11, 4438–4442 CrossRef CAS PubMed
.
- L. Li, H. Song, Q. Zhang, J. Yao and X. Chen, Effect of compounding process on the structure and electrochemical properties of ordered mesoporous carbon/polyaniline composites as electrodes for supercapacitors, J. Power Sources, 2009, 187, 268–274 CrossRef CAS
.
- G. Yu, X. Xie, L. Pan, Z. Bao and Y. Cui, Hybrid nanostructured materials for high-performance electrochemical capacitors, Nano Energy, 2013, 2, 213–234 CrossRef CAS
.
- T. Liu, L. Finn, M. Yu, H. Wang, T. Zhai, X. Lu, Y. Tong and Y. Li, Polyaniline and polypyrrole pseudocapacitor electrodes with excellent cycling stability, Nano Lett., 2014, 14, 2522–2527 CrossRef CAS PubMed
.
- P. Li, E. Shi, Y. Yang, Y. Shang, Q. Peng, S. Wu, J. Wei, K. Wang, H. Zhu, Q. Yuan, A. Cao and D. Wu, Carbon nanotube-polypyrrole core–shell sponge and its application as highly compressible supercapacitor electrode, Nano Res., 2014, 7, 209–218 CrossRef CAS
.
- P. Li, Y. Yang, E. Shi, Q. Shen, Y. Shang, S. Wu, J. Wei, K. Wang, H. Zhu, Q. Yuan, A. Cao and D. Wu, Core-double-shell, carbon nanotube@polypyrrole@MnO2 sponge as freestanding, compressible supercapacitor electrode, ACS Appl. Mater. Interfaces, 2014, 6, 5228–5234 CAS
.
- P. Li, C. Kong, Y. Shang, E. Shi, Y. Yu, W. Qian, F. Wei, J. Wei, K. Wang, H. Zhu, A. Cao and D. Wu, Highly deformation-tolerant carbon nanotube sponges as supercapacitor electrodes, Nanoscale, 2013, 5, 8472–8479 RSC
.
- X. Gui, J. Wei, K. Wang, A. Cao, H. Zhu, Y. Jia, Q. Shu and D. Wu, Carbon Nanotube Sponges, Adv. Mater., 2010, 22, 617–621 CrossRef CAS PubMed
.
- S. O. Vilela, M. A. Soto-Oviedo, A. P. F. Albers and R. Faez, Polyaniline and mineral clay-based conductive composites, Mater. Res., 2007, 10, 297–300 CrossRef CAS
.
- M. Li, Z. Wei and L. Jiang, Polypyrrole nanofiber arrays synthesized by a biphasic electrochemical strategy, J. Mater. Chem., 2008, 18, 2276–2280 RSC
.
- T. Lindfors, C. Kvarnström and A. Ivaska, Raman and UV-vis spectroscopic study of polyaniline membranes containing a bulky cationic additive, J. Electroanal. Chem., 2002, 518, 131–138 CrossRef CAS
.
- Z. Wang, X. He, S. Ye, Y. Tong and G. Li, Design of polypyrrole/polyaniline double-walled nanotube arrays for electrochemical energy storage, ACS Appl. Mater. Interfaces, 2014, 6, 642–647 CAS
.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5nr05978j |
| This journal is © The Royal Society of Chemistry 2016 |