Identifying the descriptor governing NO oxidation on mullite Sm(Y, Tb, Gd, Lu)Mn2O5 for diesel exhaust cleaning†
Abstract
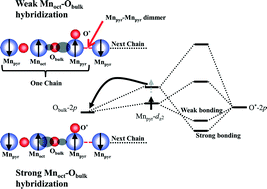
The current fast selective catalytic reduction (fast-SCR) technology shows effectiveness in converting diesel engine generated nitrogen oxides (NOx) to environmentally benign nitrogen (N2) with the aid of the precious metal catalyst platinum. Driven by the previous finding of the low-cost mullite's great superiority over Pt in terms of NO oxidation, a series of Mn-based oxides Sm(Y, Tb, Gd, Lu)Mn2O5 are synthesized to identify a general descriptor that governs the catalytic performance. Utilizing soft X-ray absorption characterization and molecular orbital theory, here, we show that the catalytic activity difference presents little dependence on the 3d electron occupancy when the A site element is varied (Sm, Tb, Y, Gd, Lu). Instead, strong p–d hybridization between lattice O and octahedral Mn leads to weak bonding strength between external O* and pyramid Mn and essentially increases the catalytic activity for converting NO to NO2.

 Please wait while we load your content...
Please wait while we load your content...