Effect of carbon dioxide on pore structure characteristics of dewatered lignite and the relevance to its moisture-adsorbing capacity
Fanjing Weia,
Xiaoxia Jingab,
Yunlong Yanga,
Junjie Liaoa,
Liping Changa and
Weiren Bao*a
aState Key Laboratory Breeding Base of Coal Science and Technology Co-founded by Shanxi Province and the Ministry of Science and Technology, Taiyuan University of Technology, Taiyuan 030024, China. E-mail: baoweiren@tyut.edu.cn; Fax: +86 351 6018482; Tel: +86 351 6018482
bDepartment of Applied Chemistry, Yuncheng University, Yuncheng 044000, China
First published on 18th November 2015
Abstract
Lignite with a high moisture content is needed to effectively remove water and maximally restrain the re-adsorption capacity of dewatered coal. The ambient gas during lignite drying is an important factor influencing the physical properties of dewatered samples. CO2 is the main component of exhaust gas, which has been reused in flash drying technology. The relationship between the drying characteristics of a typical Chinese lignite and the pore structure changes of dewatered coal in a CO2 atmosphere, and the effect on the behavior of re-adsorbing moisture were studied. Drying experiments of lignite samples under an Ar atmosphere were also carried out for comparison. The moisture re-adsorption experiments of dewatered coal samples were conducted at 30 °C under relative humidity of 75%. The results show that the drying efficiency of coal samples under a CO2 atmosphere is higher than that under an Ar atmosphere due to the swelling effect caused by the adsorption of CO2, but this difference between CO2 and Ar becomes gradually smaller with increase in the drying temperature. The dewatered coal sample obtained under a CO2 atmosphere shows a stronger ability to re-adsorb moisture caused by the swelling effect, which could be attributed to the changes of the structure of the coal sample in this atmosphere and the increase of the surface area where the moisture is primarily absorbed. The total moisture content in the coal sample after re-adsorbing moisture follows a linear relationship with the specific surface area of dewatered lignite.
1. Introduction
Lignite, which is approximately 20% of the total coal reserve in China and features many obvious characteristics such as its low calorific value, high water and oxygen content, and also low sulfur content, plays an important role in supplying primary energy.1–5 At present, lignite is treated only as a low-grade fuel to supply electricity in many countries because of the high content of moisture, ease of re-absorption of moisture after drying, and difficulty of transportation.5,6 With the rapid growth of high rank coal consumption level, how to increase the utilization efficiency of lignite by reducing its moisture content and restraining the re-adsorption capacity of dewatered coal is very important.Lignite with low coal rank possesses a plentiful pore structure and numerous oxygen-containing functional groups. The transport and capillary action of channels in the pore structure and the hydrophilicity of oxygen-containing functional groups on its surface are considered to be the main reasons for a good deal of the moisture in lignite.7–9 Drying of lignite is a necessary way for its effective utilization and the drying atmosphere is one of the most important influencing factors on the dewatering behavior of lignite. Different drying atmospheres can lead to a diverse efficiency in removing moisture from lignite, resulting in the distinctly different moisture re-adsorption degrees of dewatered coal. In industry, lignite is usually dried using hot flue gas from coal combustion, which is beneficial to exhaust heat utilization. As one of the greenhouse gases, CO2 content can reach 60% of the flue gas obtained from coal oxygen-rich combustion.
The adsorption of CO2 into the coal matrix will cause significant changes in its chemical and physical structure.10,11 The changes of the pore structure in the coal matrix during drying under different atmospheres are discussed rarely in the literature. Meanwhile, the mechanism of high re-adsorbing moisture capacity for the dewatered coal under CO2 is also unclear. This study aims to investigate the relationship amongst the moisture removal of lignite, and re-adsorption of dewatered coal and the transformations of the physical structure characteristics through the experimental results under drying atmospheres of CO2 and Ar as a comparison gas.
2. Experimental
2.1 Sample preparation
Lignite from Ximeng coal mine in Inner Mongolia’s Xilingol League north of China, which is one of the largest brown coal deposits in China, was selected as the experimental sample in this study. In this region, more than 50% of the total coal reserve is suitable for opencast coal mining and the lignite used in the experiments was from dozens of meters in depth. The sample was firstly crushed and sieved under a N2 atmosphere to a particle size of 0.25–0.85 mm for the drying experiments. To make sure the experiments are well repeated, the method of coining and quartering was strictly used for each sampling.The proximate and ultimate analysis results of coal samples measured by the chemical analysis method according to Chinese national standards GB/T 212-2008, GB/T476-2008, GB/T 19227-2008 and GB/T 214-2007 are given in Table 1. Mt in Table 1 is the average result of the measured values before each experiment when the moisture content of the sample in the container was close to equilibrium. It can be seen that this sample is a representative low rank coal with a high moisture content (34.4%), oxygen content (24.7%) and volatile matter yield (42.7%).
| Proximate analysis (wt%) | Ultimate analysis (wt%, daf) | |||||||
|---|---|---|---|---|---|---|---|---|
| Mt | Aad | Vdaf | FCdaf | C | H | O* | N | S |
| a Note: ad is air dry basis; daf is dry and ash free basis; * by difference. | ||||||||
| 34.40 | 18.05 | 42.70 | 57.30 | 69.79 | 2.99 | 24.73 | 1.36 | 1.13 |
The dewatering experiments of lignite were performed in a fixed-bed reactor with a furnace temperature control system. The reactor was a glass tube with dimensions of 680 mm × 14.5 mm. Ar is selected as the comparable ambient atmosphere in the whole process of drying lignite because the kinetic diameter and thermal conductivity coefficient of Ar gas are very similar to CO2. The kinetic diameter of CO2 and Ar are 0.33 nm and 0.34 nm, respectively. The thermal conductivity coefficients of CO2 and Ar at 60 °C are 0.0192 W m−1 K−1 and 0.0196 W m−1 K−1, respectively. Thermal conductivity coefficients of the two gases at other higher temperatures are also very similar. The reactor was purged by Ar or CO2 gases for 30 min with the flow rate of 400 mL min−1 before the drying experiment. The GC analysis of the gas component in the outlet of the reactor proved that the time of 30 min is enough to entirely replace the air in it. After purging the reactor, about 2.0 g coal samples were put on the plate installed inside the glass tube and located in the isothermal zone of furnace which had been elevated to the desired temperature and kept for a preset time. The drying atmosphere was Ar or CO2 with a flow rate of 400 mL min−1 in the entire drying process. It has been proven that CO2 begins to be released from lignite when the temperature of the coal bed is above 160 °C (ref. 6 and 7) and the physico-chemical structures of lignite change greatly due to the decomposition of thermally unstable groups. In our experiments, the drying temperature was selected in the range of 60–160 °C based on the preliminary screening of operating conditions, which is mainly to avoid the influence of excessive volatiles loss and simplify the experimental results as much as possible. Raw coal and dewatered coal are denoted as RC and DC, respectively. In this process, the yield of water removed from the coal sample was calculated by the gravimetric method according to eqn (1).
 | (1) |
2.2 Moisture re-adsorption experiment
The re-adsorbing experiments of dewatered coal samples were carried out at a constant temperature of 30 °C and the relative humidity was 75%. Considering the temperature and humidity changes of summer and winter in the different regions of China, 30 °C and 75% are selected as the moderate values for representative experimental researches. The re-adsorption moisture amount of dewatered coal sample was also measured by the gravimetric method. The experimental conditions are listed in Table 2.| Parameter | Contents/conditions |
|---|---|
| Drying temperature (°C) | 60–160 |
| Drying atmosphere | CO2 or Ar |
| Gas flow rate (mL min−1) | 400 |
| Drying time (min) | 5–60 |
| Amount of sample per batch (g) | 2 |
| Particle size (mm) | 0.25–0.85 |
| Relative humidity (%) | 75 |
| Re-adsorption temperature (°C) | 30 |
| Re-adsorption time (h) | 48 |
Re-adsorbed moisture coal is denoted as AC. The re-adsorbed moisture yield of dewatered coal is denoted as M and it is defined as eqn (2).
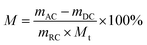 | (2) |
The residual moisture content of dewatered coal and total moisture content of re-adsorbed coal are denoted as R and Y, respectively. R and Y are calculated as follows:
| R = Mt × (1 − N) | (3) |
| Y = Mt × (1 − N + M) | (4) |
2.3 Analysis of pore structure
In order to determine the pore structure changes of lignite in the drying process under CO2 and Ar atmospheres, the pore size distributions and the surface areas of the samples before and after lignite dewatering were characterized by the nitrogen adsorption method. A JW-BK122W porosimeter (JWGB, Beijing) was used and the ultimate vacuum was 4 MPa. In addition, the changes in external morphology of dewatered samples in the drying process under two atmospheres were determined by scanning electron microscopy (SEM). A JSM-7001F thermal field emission SEM (JE01, Tokyo, Japan) with a voltage of 3.0 kV was used.2.4 Thermogravimetric analysis
The weight change experiments during the drying process were carried out with a thermal analyzer (NETZSCH STA 449 F3 Jupiter). About 20 mg of coal sample was used with the heating rate of 10 °C min−1 from room temperature to 600 °C and the carrier gas flow rate was 60 mL min−1.3. Results and discussion
3.1 Effects of drying atmosphere on the moisture removal of lignite and re-adsorbing behaviors of dewatered coal
The evolution curves of moisture removal yield of raw coal and the re-adsorbing moisture yield of dewatered coal under CO2 and Ar gas at different temperatures for the drying time of 60 min are shown in Fig. 1. It can be found that the efficiency of moisture removal under CO2 is higher than that under Ar. As the drying temperature increases, the difference between the dehydration rate of lignite in CO2 and Ar decreases. When the drying temperature was raised to 160 °C, the difference can be nearly neglected. The polarity of C–O bond and lone pair electrons of oxygen atoms in CO2 molecules result in the effortless adsorption on the lignite surface in spite of CO2 being a non-polar molecule and the replacement of moisture molecules by CO2.12–15 It means that the CO2 molecules are easier to enter into the internal pores of lignite and adsorbed on the active sites instead of H2O than Ar molecules.16–18 Besides, CO2 can absorb on the organic matrix and modify the physical and possible chemical structure of the coal matrix. This physical modification is associated with the relaxation and rearrangement of the macromolecular structure of coal and gradually changes the pore structure of the coal with holding time.15 The early crack initiation can be produced through the swelled layer induced by CO2 adsorption and then will form the early crack damage and failure points due to a lower surface energy.12,13 CO2 infiltrates to pores and makes them larger which is beneficial to heat transmission and water evaporation. When the drying temperature of lignite is elevated, the effect of CO2 weakens gradually owing to the decline of adsorption ability and thus drying efficiency becomes closer with the increase of temperature. Zhai’s19 research indicates that the CO2 swelling effect to lignite pore structure just occurs at the initial drying stage and most of the active sites are occupied by CO2 resulting in the swelling effect ending as the drying process is prolonged. Water in large pores was firstly removed by evaporation, followed by those in the larger capillaries, but the collapse of the lignite structure is relatively small during this stage. When 80% of the total moisture has been lost, the shrinkage forces caused by emptying the small capillaries induce the pore structure to completely collapse and maximum shrinkage occurs when 92% of the total moisture has been lost.20 A similar result7 was found in that the collapse of the coal structure was relatively small when the drying efficiency was less than 85%. Combined with Fig. 1, the pore structure of lignite obtained at temperatures exceeding 100 °C shrank and suffered from serious collapse. At relatively high temperatures the swelling effect of the lignite caused by CO2 adsorption just occurs at the initial drying process and is gradually covered up by the complete shrinkage and collapse of the pore structure as the drying time is prolonged. So Fig. 2 shows that the distribution curves of the pore size tended to be the same between CO2 and Ar atmospheres at 120 °C and 160 °C. | ||
| Fig. 1 Evolution curves of moisture removal yield of lignite under CO2 and Ar gases at different drying temperatures and the re-adsorbing moisture yields of dewatered coal. | ||
 | ||
| Fig. 2 Distribution curves of pore size for the dewatered sample under different temperatures and atmospheres. | ||
Distribution curves of the pore size for dewatered coal samples at different temperatures are shown in Fig. 2 and it shows that the distribution curve of the pore size for a CO2 atmosphere tends to be the same as that for an Ar atmosphere at 120 °C and 160 °C, which is consistent with the explanations above. It can be seen that the change of pore radius range of dewatered coal with drying temperature are different for the two atmospheres. At the low temperature of 60 °C, the pores of below 25 nm radius ranges in the dewatered coal under CO2 gas account for a larger proportion than that under Ar. But the pores of above 25 nm radius ranges obviously increase for the dewatered coal at 100 °C and 120 °C under CO2. And then the pore sizes are similar for the dewatered coal under CO2 and Ar when the drying temperature is raised to 160 °C. As seen in Fig. 1, the moisture removal yields of lignite under the two atmospheres are similar, both greater than 100%, when the drying temperature is higher than 120 °C. This indicates the drying temperature is the major factor affecting lignite drying at higher temperatures.
Fig. 1 also shows that the dewatered coal samples at 100 °C under CO2 and Ar atmospheres have the maximal amount of moisture re-adsorption and it is higher for dewatered coal under a CO2 than that under an Ar atmosphere. The abundant pore structure should be one of the most important reasons for the higher re-adsorbing ability of dewatered lignite. The relationship between the pore structure and the total moisture content of the re-adsorbed lignite will be discussed in detail in the following section. In the drying process of lignite, although the pore structure may vary under any gas, the degree of change under CO2 gas should be more beneficial for moisture re-adsorption than that under Ar gas. Table 3 shows the pore structure characteristic parameters of lignite and dewatered coal under different atmospheres. It can be seen that when the temperature is above 100 °C, the specific surface area, average pore size, most probable pore size and mesopore and macropore volume of dewatered coal under CO2 gas are larger than that under Ar gas, and therefore the moisture re-adsorption is easier. The morphology of raw coal and dewatered samples were characterized by SEM and the results are shown in Fig. 3. It was found that the collapse and cross-linking of pore structures in the dewatered lignite under CO2 and Ar atmospheres both take place to different degrees compared with the relatively flat morphology of raw coal. As the drying degree of lignite increases, its surface area (1.18 to 1.50 m2 g−1) clearly decreased compared with raw coal (2.06 m2 g−1), while the change in pore size and pore volume show different trends. The dewatered coal which was dried at 60 °C under a CO2 atmosphere has the lower average pore size, mesopore and macropore volume than that under Ar, but its surface area is significantly higher. When the drying temperature is above 100 °C, the average pore size, mesopore and macropore volume of the dried samples under a CO2 atmosphere significantly increase. It should be also noted that the average pore size and pore volume of dewatered coal under CO2 gas for 60 °C are smaller, while the surface area is larger than that under Ar gas at each drying temperature. The effect of CO2 perhaps acts on different pore diameters at different drying temperatures. CO2 absorption is an exothermic process,21 which means that CO2 is easier to be absorbed on lignite at low temperature. So CO2 gases are more likely to diffuse to micropores and then absorb on it at 60 °C, and this makes more micropores (<2 nm pore sizes) enlarge. Fig. 2(a) also indicates that the pores of about 2 nm markedly increase for the sample dried at 60 °C, the distributions of mesopores and macropores change slightly, which directly results in the average pore size (21.45 nm) and the most probable pore size (4.63 nm) in CO2 being lower than that (36.02 nm and 4.71 nm) in Ar. However, CO2 infiltrating micropores is relatively weak at higher temperature and more CO2 gas molecules easily touch and act on macropores and mesopores which leads to a reversal in the result.
| Structure parameter | RC | DC (60 °C) | DC (100 °C) | DC (120 °C) | DC (160 °C) | ||||
|---|---|---|---|---|---|---|---|---|---|
| Ar | CO2 | Ar | CO2 | Ar | CO2 | Ar | CO2 | ||
| Specific surface area (m2 g−1) | 2.06 | 1.41 | 1.50 | 1.23 | 1.27 | 1.24 | 1.32 | 1.20 | 1.18 |
| Average pore size (nm) | 23.45 | 36.02 | 21.45 | 26.24 | 31.41 | 30.88 | 31.90 | 31.34 | 40.13 |
| Most probable pore size (nm) | 1.91 | 4.71 | 4.63 | 4.01 | 4.61 | 3.99 | 4.59 | 3.51 | 3.74 |
| Mesopore volume (cm3 g−1) | 2.88 | 2.02 | 1.80 | 1.94 | 2.24 | 2.11 | 2.41 | 2.02 | 2.30 |
| Macropore volume (cm3 g−1) | 0.14 | 0.14 | 0.07 | 0.05 | 0.12 | 0.06 | 0.18 | 0.07 | 0.19 |
 | ||
| Fig. 3 SEM images of raw lignite and dewatered sample at 100 and 120 °C in CO2 or Ar atmosphere (A) RC, (B) DC (Ar/100 °C), (C) DC (Ar/120 °C), (D) DC (CO2/100 °C), (E) DC (CO2/120 °C). | ||
In order to verify again the behaviors of moisture removal in two atmospheres, the experiments were carried out with a thermal analyzer (NETZSCH STA 449 F3 Jupiter). Fig. 4 shows the mass decay of raw lignite under Ar and CO2 atmospheres with a heating rate of 10 °C. Under CO2 gas, the weight loss of lignite is greater than that under Ar gas and the difference is bigger with the increase of temperature,18 the difference was well proven by experiments. There are two clear regions to be identified. The first stage happens between room temperature and 250 °C and the maximum weight loss ratio occurs at around 100 °C, in which the mass decay is mainly due to the removal of H2O during coal drying. The second stage ranges from 250 °C to 600 °C and the maximum weight loss ratio occurs at around 450 °C, in which the mass decay is mainly due to the removal of volatiles during coal pyrolysis. Comparing the two DTG curves, it can be found that the rate of weight loss under the CO2 atmosphere is a little larger than that under the Ar atmosphere, which shows that CO2 gas can really enhance the drying efficiency of lignite and accelerate the dewatering rate.
3.2 Effect of drying time on the moisture removal and re-adsorbing behaviors of lignite under different atmospheres
In the drying process, the adsorption of CO2 causes a different swelling effect but the relationship between the swelling process and the drying time is unclear. To solve the problem, experiments under CO2 and Ar gas at different drying times were conducted. The results are illustrated in Fig. 5. It can be found that with the increase of drying time, the drying efficiency under CO2 gas is gradually larger than that under Ar gas, which shows that the swelling by CO2 adsorption is a gradually changing process. CO2 swelling effect to lignite pore structure just occurs at the initial drying stage and frequently causes the change of pore structure of lignite which is beneficial for water removal, thus the drying efficiency under CO2 gas becomes larger than that under Ar after a drying period. The drying efficiency under CO2 gas becomes larger than that under Ar gas at 60 °C when the drying time is 40 min, but it only needs 15 min to make the two curves intersect at 160 °C. By comparing the four figures, it can be seen that as the drying temperature rises, the cross point of CO2 curve and Ar curve appears earlier. It can be concluded that the drying efficiency in the CO2 condition improved significantly with the increase of drying temperature. | ||
| Fig. 5 Evolution curves of moisture removal yield of lignite under CO2 and Ar atmosphere for different drying times. | ||
To make the effect of drying time on the structure clear, external morphology and the BET surface area were measured by using SEM and nitrogen adsorption methods, respectively. These results are shown in Fig. 6 and Table 4, respectively. From the external morphology of dewatered lignite at 120 °C for different drying times, it can be found that the pore size of dewatered lignite surface (dried for 30 min) clearly increases and the lignite surface is seriously broken and destroyed for the samples drying for 40 min and 60 min. It shows that the swelling effect of carbon dioxide is a gradual process. It can be guessed from the previous proof that the pores swell first and then collapse during drying under a CO2 atmosphere. The drying temperature does indeed have an effect on pore swelling under a CO2 atmosphere and thus affects the variation of drying efficiency. It results in the cross point of the two curves in the four figures of Fig. 5 appearing earlier as the drying temperature rises.
 | ||
| Fig. 6 SEM images of raw lignite and dewatered sample with different drying time at 120 °C under CO2 atmosphere (a) RC, (b) DC (10 min), (c) DC (30 min), (d) DC (40 min), (e) DC (60 min). | ||
| Structure parameter | RC | DC (10 min) | DC (30 min) | DC (40 min) | DC (60 min) |
|---|---|---|---|---|---|
| Specific surface area (m2 g−1) | 2.06 | 1.66 | 1.26 | 1.46 | 1.32 |
| Average pore size (nm) | 23.45 | 40.27 | 43.86 | 41.44 | 31.90 |
| Most probable pore size (nm) | 1.91 | 4.05 | 4.01 | 4.64 | 4.59 |
| Mesopore volume (cm3 g−1) | 2.88 | 2.79 | 2.60 | 2.52 | 2.41 |
| Macropore volume (cm3 g−1) | 0.14 | 0.10 | 0.23 | 0.26 | 0.18 |
In order to more clearly see the changes of lignite surface characteristics as the drying time, the dewatered lignite were determined using the nitrogen adsorption method. It can be seen in Table 4 that the average pore size increases from 23.45 to 43.86 nm within 30 min, and then tends to decrease. Because of the existence of CO2, as soon as it probed into the dryer, the coal sample swelled instantly, so as to increase its average pore size. However, with the drying time elapsing and the drying degree deepening, the influence of CO2 weakens, and the destruction of the coal sample’s structure is aggravated resulting in the decrease of the average pore size. The changes of pore structure especially at relatively high temperature are attributed to the contributions of both CO2 cracking and pore shrinkage/collapse. The existence of CO2 makes the surface of the coal sample crack, and then the voids between the pores extend; as a result the volume content of the mesopores and macropores are increased. The change of macropores during the drying is complicated, but its proportion in the entire pore structure is very small. The increase of mesopore volume is obvious and it should be the contribution of both CO2 swelling and pore shrinkage. But the latter is dominant for mesopores.
Due to the fact that the swelling effect of the CO2 on pore structure of lignite is a gradual process during drying, the surface properties of dewatered lignite for different drying times are different, which inevitably results in a different absorption capacity. Fig. 7 and 8 show the residual moisture content and the re-adsorption capacity of dewatered lignite for different drying times, respectively.
 | ||
| Fig. 8 Effect of drying time on re-adsorption capacity of dewatered samples at different conditions. | ||
As can be seen from Fig. 7, the residual moisture content of dried samples shows the same changing trend at different drying temperatures. Under both Ar and CO2 gas, the content of residual moisture in coal decreases slowly with time in the drying process at 60 °C. When the drying temperature is higher than 100 °C, the residual moisture content decreases rapidly before 20 min, and then changes little. Different re-adsorption moisture amounts of dewatered lignite with different drying times were observed in Fig. 8. Under an Ar atmosphere, the re-adsorption amount increases first and then decreases with increased drying time. The reason is mainly due to both the residual moisture content and pore structure of the dewatered coal sample affecting the re-adsorption amount of dewatered lignite. Water in large pores was first removed by evaporation, followed by those in the larger capillaries, but collapse of the lignite structure is relatively small during this stage.7,20 At the beginning of the drying, the re-adsorption amount is mainly affected by the residual moisture content, so it increases with the continuous removal of water. As the drying process progresses, the content of the residual moisture in the coal decreases, while the collapse of the pore structure becomes more apparent, and then the re-adsorption amount constantly reduces with increased drying time due to the constant reduction of the surface area.7 However, the trend in the change of re-adsorption amount of dewatered coal under a CO2 atmosphere is quite different from that under an Ar atmosphere. Firstly, they all decrease and then increase at each drying temperature. Within 30 min, the swelling effect of CO2 makes the average pore size bigger, so the specific surface area becomes smaller which directly causes the re-adsorption moisture amount to decrease and reach a minimum value at 30 min. During the process of drying, the removal of water transfers from the surface to internally in the lignite and CO2 gas can remove the water of smaller pores. On the other hand, after drying for 30 min the pore structure suffers a serious collapse and some fragments are produced which causes a larger specific surface area. Thus many more pores and active sites are empty and the re-adsorption moisture amount begins to increase after 30 min.
Hydrophilic oxygen-containing functional groups (–COOH, Ar-OH, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) begin to decompose when the temperature is higher than 160 °C.7,22 So, it is supposed that CO2 gases make the surface area of dewatering lignite enlarge and this should be the main reason for high moisture re-adsorption capacity for the dewatered coal under CO2. Y represents the total moisture content of re-absorbed lignite on a received basis (%). S (m2 g−1) denotes the specific surface area of dewatered lignite. Fig. 9 indicates that Y decreases nearly linearly with decreasing S. A rough estimate could be obtained from the fitting formula: the total moisture content of re-adsorbed lignite is decreased about 79.18% as the specific surface area of per gram of coal is decreased to 1 m2.
O) begin to decompose when the temperature is higher than 160 °C.7,22 So, it is supposed that CO2 gases make the surface area of dewatering lignite enlarge and this should be the main reason for high moisture re-adsorption capacity for the dewatered coal under CO2. Y represents the total moisture content of re-absorbed lignite on a received basis (%). S (m2 g−1) denotes the specific surface area of dewatered lignite. Fig. 9 indicates that Y decreases nearly linearly with decreasing S. A rough estimate could be obtained from the fitting formula: the total moisture content of re-adsorbed lignite is decreased about 79.18% as the specific surface area of per gram of coal is decreased to 1 m2.
| Y = 79.18[S] − 56.05, R2 = 0.87 |
 | ||
| Fig. 9 Relationship between the total moisture content and the specific surface area of lignite dried at different conditions. | ||
4. Conclusions
The lignite shows a higher drying efficiency under a CO2 atmosphere than that under Ar. However, with increased drying temperature, the difference of drying efficiency between CO2 and Ar is gradually reducing. The coal sample obtained under a CO2 atmosphere has a higher moisture re-adsorption efficiency owing to the swelling effect. The swelling of carbon dioxide also makes the pore size of dewatered lignite larger and leads to an increased amount of moisture re-adsorption.The effect of CO2 acts differently on the various pores at different drying temperatures. At 60 °C, CO2 gas is likely to make more micropores become larger. However, it mainly acts on macropores and mesopores at higher temperature. As the drying time was prolonged, the drying efficiency under CO2 increases progressively and is greater than that under Ar at the same temperature. It requires 40 min when the drying efficiency of lignite under CO2 atmosphere exceeds that under Ar at 60 °C, while it only needs 15 min at 160 °C, which reveals CO2 changes the structure of the coal sample gradually. CO2 gas makes the lignite present a larger surface area, in which moisture is primarily absorbed. There is a linear relationship between the total moisture content and the specific surface area of dewatered lignite.
Acknowledgements
The authors gratefully acknowledge the financial supports of National Basic Research Program of China (2012CB214902), International S & T Cooperation Program of China (2013DFG61490) and National Natural Science Foundation of China (21176165).References
- Y. Ma, Y. Yuan, J. Jin, H. Zhang, X. Hu and D. Shi, Energy, 2013, 59, 105–115 CrossRef.
- H. Osman, S. V. Jangam, J. D. Lease and A. S. Mujumdar, Drying Technol., 2011, 29, 1763–1783 CrossRef CAS.
- Z. Lei, Y. Zhang, L. Wu, H. Shui, Z. Wang and S. Ren, RSC Adv., 2013, 3, 2385–2389 RSC.
- J. Cheng, X. Wang, F. Zhou, R. Huang, A. Wang, X. Chen, J. Liu and K. Cen, RSC Adv., 2015, 5, 14690–14696 RSC.
- Z. Rao, Y. Zhao, C. Huang, C. Duan and J. He, Prog. Energy Combust. Sci., 2015, 46, 1–11 CrossRef.
- S. Hu, C. Man and X. Gao, Drying Technol., 2013, 31, 1194–1205 CrossRef CAS.
- Y. Yang, X. Jing, Z. Li, X. Liu, Y. Zhang and L. Chang, Drying Technol., 2013, 31, 1430–1437 CrossRef CAS.
- Y. Zhang, X. Jing, K. Jing, L. Chang and W. Bao, Appl. Surf. Sci., 2015, 324, 90–98 CrossRef CAS.
- J. Yu, A. Tahmasebi, Y. Han, F. Yin and X. Li, Fuel Process. Technol., 2013, 106, 9–20 CrossRef CAS.
- H. L. Tay and C. Z. Li, Fuel Process. Technol., 2010, 91, 800–804 CrossRef CAS.
- M. S. A. Perera, P. G. Ranjith and M. Peter, Energy, 2011, 36, 6941–6947 CrossRef CAS.
- Z. Majewska, S. Majewski and J. Zietek, Int. J. Coal Geol., 2010, 83, 475–483 CrossRef CAS.
- S. R. Kelemen and L. M. Kwiatek, Int. J. Coal Geol., 2009, 77, 2–9 CrossRef CAS.
- G. D. Bongers, W. R. Jackson and F. Woskoboenko, Fuel Process. Technol., 2000, 64, 13–23 CrossRef CAS.
- M. Mojtaba, J. H. Peter and F. J. Hasan, J. Mater. Sci., 2010, 45, 5271–5281 CrossRef.
- M. Švábová, Z. Weishauptová and O. Přibyl, Fuel, 2012, 92, 187–196 CrossRef.
- E. Ozdemir and K. Schroeder, Energy Fuels, 2009, 23, 2821–2831 CrossRef CAS.
- Y. Bai, P. Wang, L. Yan, C. Liu, F. Li and K. Xie, J. Anal. Appl. Pyrolysis, 2013, 104, 202–209 CrossRef CAS.
- G. Zhai, L. Duan, S. Tang and Z. Xia, J. China Coal Soc., 2012, 37(5), 788–793 CAS.
- D. G. Evans, Fuel, 1973, 52, 186–190 CrossRef CAS.
- D. Zhang, Y. Cui, S. Li, W. Song and W. Lin, J. China Coal Soc., 2011, 36(10), 1696–1698 Search PubMed.
- A. Tahmasebi, J. Yu, Y. Han and X. Li, Fuel Process. Technol., 2012, 101, 85–93 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2015 |


