Growth of Fe3O4 nanosheet arrays on graphene by a mussel-inspired polydopamine adhesive for remarkable enhancement in electromagnetic absorptions†
Fanbin Meng*,
Wei Wei,
Jingjing Chen,
Xiangnan Chen,
Xiaoling Xu,
Man Jiang,
Yong Wang,
Jun Lu and
Zuowan Zhou*
Key Laboratory of Advanced Technologies of Materials (Ministry of Education), School of Materials Science and Engineering, Southwest Jiaotong University, Chengdu 610031, China. E-mail: mengfanbin_wing@126.com; zwzhou@home.swjtu.edu.cn
First published on 18th November 2015
Abstract
Fe3O4 nanosheet arrays grown on both surfaces of graphene can be achieved by combining a mussel-inspired polydopamine adhesive and ethylene glycol-mediated process. Polydopamine is utilized not only as an efficient linker molecule that binds Fe3O4 nanosheets to the graphene, but also as a carbon source during heat treatment to yield the three dimensional (3D) graphene@carbon@Fe3O4 nanosheet arrays architecture. After the growth of Fe3O4 nanosheet arrays with accompanying reduction of graphene oxide into graphene, the 3D architecture exhibits outstanding microwave absorption properties. The simulated value of maximum reflection loss can reach −52.8 dB at 9.5 GHz with a sample thickness of 2.7 mm. The improved absorption capacity arises from the synergy of dielectric loss and magnetic loss, as well as the enhancement of multiple interfaces among graphene, carbon and Fe3O4 nanosheets. Furthermore, the synthesis strategy presented here can be expended as a facile approach to synthesizing related graphene-based 3D nanostructures for functional design and applications.
1. Introduction
Recently, great efforts have been made towards the development of high efficiency and lightweight microwave absorbents to effectively eliminate the increasing adverse electromagnetic interference and radiation problems.1–4 In particular, graphene-based nanostructures are becoming more and more attractive for utilization in electromagnetic (EM) absorptions, including graphene-supported or -wrapped nanostructures.5–10 Firstly, graphene promises to be a superior building block for constructing electromagnetic wave absorbers, owing to its remarkable physical properties including large surface area, high conductivity and good thermal conductivity.11 Taking advantage of the benefits of conductive graphene and the introduced other lossy materials, as well as the interfacial and synergistic effects, the graphene-based nanostructures may result in strong EM absorption properties. Ren et al. developed a new strategy, i.e., a seed-assistant method, to fabricate three dimensional (3D) SiO2@Fe3O4 core/shell nanorod array/graphene architecture, and the maximum reflection loss reached −31.9 dB with a sample thickness of 2.5 mm.12 Li et al. reported a single polyol method to fabricate graphene/Fe3O4 hybrids with a maximum reflection loss of −30.1 dB at 1.48 mm matching thickness.13 Chen et al. introduced a new method for the preparation of graphene/polyaniline hybrids using a one-step intercalation polymerization of aniline inside the expanded graphite, and the results showed that the poor EM absorption properties of graphene could be significantly improved after the separation of graphite into graphene based hybrids. The maximum reflection loss reached −36.9 dB at 10.3 GHz with the thickness of 3.5 mm.14 However, the intrinsic poor impedance matching in such graphene/polymer systems hinders further improvement of application performances. Similarly, graphene decorated with inorganic nanocrystals generally aggregate with uneven distribution, leading to possible “dead areas” with null components, which also reduces the EM absorption properties. Therefore, design of a facile synthesis that ensures lossy nanocrystals uniformly decorated on the whole of the graphene nanosheets without aggregation is urgently needed.Herein, we developed an efficient multi-step conversion approach to growing Fe3O4 nanosheets on the surfaces of graphene to form 3D graphene@Fe3O4 nanosheet architectures. In order to ensure the Fe3O4 nanosheets grow uniformly on the both surfaces of graphene, polydopamine (PDA) was utilized to act as an efficient linker molecule that binds Fe3O4 nanosheets to the graphene, inspired by the adhesion mechanism of mussels.15 Furthermore, the PDA can also act as a carbon source during heat treatment to yield 3D graphene@carbon@Fe3O4 nanosheet arrays architectures. Due to the special morphology, multi-interfacial polarization and impedance matching, the 3D graphene@carbon@Fe3O4 nanosheet arrays exhibited very good EM absorption properties, including strong attenuation to EM wave and lightweight characteristics.
2. Experimental section
2.1 Preparation of graphene@carbon@Fe3O4 nanosheet arrays
RGO@PDA was prepared by the simultaneous reduction of GO with dopamine hydrochloride and the self-polymerization of dopamine according to the ref. 15. Then, RGO@PDA (10 mg) was dispersed into ethylene glycol (100 mL) with ultrasonication, and then FeCl3·6H2O (0.6 g), urea (1.5 g) were added. This mixture was stirred at 50 °C for 2 h. Next, the solution was subsequently heated and refluxed at ∽195 °C. When the mixture turned green, indicating the formation of iron alkoxide on the RGO@PDA. After further 5 min, the reaction was stopped and the mixture was cooled to room temperature. The precipitates were separated by centrifugation, washed with pure ethanol and dried in vacuum at 40 °C for 12 h. Last, the precipitates were calcined at 500 °C for 3 h under an argon atmosphere to obtain the final composites of graphene@carbon@Fe3O4.2.2 Characterization
The morphologies were characterized on a field emission scanning electron microscopy (FE-SEM, JEOL, JSM-7001F) and a transmission electron microscopy (TEM, JEOL, JEM-2100F). X-ray diffraction (XRD) analyses were carried out on a Philips X'Pert PRO X-ray diffractometer with a CuKα radiation. The Raman spectra for the samples were obtained on a Laser Raman spectroscopy (InVia, RENISHAW) using a 514 nm argon ion laser. The complex permeability and permittivity were measured on a vector network analyzer (AV3618, CETC, China) in the frequency of 2–18 GHz. The samples were mixed with wax and prepared as the toroidal shape with an outer diameter of 7.0 mm, an inner diameter of 3.04 mm and a thickness of 3.0 mm. The mass ratios of the samples to wax were set to be 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7.
7.
3. Results and discussion
The processes involved in the formation of the 3D architecture, are illustrated in Scheme 1. First, graphene oxide (GO) was modified by PDA, leading to the reduction of GO into reduced graphene oxide (RGO). Importantly, the coated PDA can absorb and immobilize the Fe3+ by the strong coordination ability of the catechol groups,16,17 resulting in forming metal nanoparticles by an in situ nucleation (step 1). Next, these nanoparticles served as “seeds” for the growth of iron alkoxide nanosheets on the surfaces of RGO@PDA under the ethylene glycol (EG)-mediated process (step 2). Finally the 3D graphene@carbon@Fe3O4 nanosheet arrays can be obtained by a calcination of the RGO@PDA@iron alkoxide (step 3).Fig. 1a and b shows the morphologies of RGO@PDA@iron alkoxide after the EG-mediated process by scanning electron microscopy (SEM). Growing nanosheets on both surfaces of RGO@PDA can be clearly found, forming interesting 3D architectures (Fig. 1a), and the iron alkoxide nanosheets are mostly grown upright with close and homogeneous on the RGO@PDA support as shown in Fig. 1b. The further investigations also revealed that the PDA used in the current system played an important role in the formation of such sheet-like structures, since only the aggregation of Fe3O4 nanosheets with flower-like morphology would be formed without the PDA coating on the GO surface (Fig. S1, ESI†). Additionally, each reaction stage was also traced to investigate the growth mechanism of such architecture by SEM (Fig. S2, ESI†). After thermal treatment, the 3D graphene@carbon@Fe3O4 nanosheet arrays were obtained and still maintain their original morphology (Fig. 1c). However, the size of the nanosheets decreases obviously, and many pores present in the nanosheets, due to the phase transformation of iron alkoxide to Fe3O4 (Fig. 1d). Such an interesting structure was also viewed under transmission electron microscopy (TEM; Fig. 1e), and the diffraction rings in the SAED pattern (inset of Fig. 1e) correspond to (220), (311), (400), (511) and (440) crystalline planes of Fe3O4. Furthermore, clear lattice fringes are observed in the high resolution TEM (HRTEM) image (Fig. 1f), and the spacing labeled in the image is about 0.253 nm, corresponding to (311) plane of Fe3O4. Also the inset in Fig. 1f shows the fast Fourier transform pattern of HRTEM image, further proving that the Fe3O4 nanosheets are single crystalline.
The phase structures evolution of the intermediate products obtained at various stages were characterized by X-ray power diffraction (XRD) (Fig. 2a), after GO modified by PDA, the sharp diffraction peak in GO (2θ = 10.7) disappears, and a new broad diffraction peak (2θ = 25.3) appears in the RGO@PDA, indicating the successful introduction of PDA, with accompanied reduction of GO.18 After EG-mediated process, a strong diffraction peak appears (2θ = 18.7), which agrees well with the reported iron alkoxide.19 For graphene@carbon@Fe3O4, the detected diffraction peaks can be indexed in the cubic inverse spinel structure of Fe3O4 (JCPDS card, file no. 19-0629), indicating the phase transformation from iron alkoxide to Fe3O4. Additionally, no characteristic peaks of graphene are observed, suggesting that the Fe3O4 could prevent the restacking of the graphene, and the graphene nanosheets are disordered in the graphene@carbon@Fe3O4 hybrids. Moreover, the significant structural changes of the carbon framework occurring during each stage were also reflected in their Raman spectra (Fig. 2b). Except two characteristic peaks, corresponding to the D band at ∼1340 cm−1 and G band at ∼1590 cm−1, a new peak at 667 cm−1 can be clearly identified. It corresponds to the A1g mode of Fe3O4,20 proving the presence of Fe3O4 in the hybrids. The intensity ratio of D and G bands (ID/IG) in GO (0.76), RGO@PDA (0.84) and graphene@carbon@Fe3O4 (0.89) increases gradually, suggesting that GO is reduced to graphene.21,22 The G band shift in carbon-based composite is related to the charge transfer between carbon and other compounds.23,24 Therefore, the observed G band shifts by 5 cm−1 from 1593 cm−1 (RGO@PAD) to 1598 cm−1 (graphene@carbon@Fe3O4) can be attributed to the charge transfer from graphene to Fe3O4, supporting the formation of Fe–O–C bonds between Fe3O4 and graphene sheets.22
The electromagnetic wave absorption properties were investigated by mixing 30 wt% of the samples with paraffin. The reflection loss (RL) curves of the as-obtained products were calculated by the measured values of the relative complex permittivity and permeability at a given frequency and layer thickness according to the transmission line theory, which is summarized as the following equations:25
 | (1) |
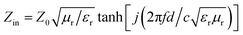 | (2) |
To understand the EM absorption enhancement mechanism of the graphene@carbon@Fe3O4 nanosheet arrays, the dependence of the complex permittivity and permeability on frequency was investigated. Where the real parts of the complex permittivity (ε′) and permeability (μ′) represent the storage capability of electric and magnetic energy, and imaginary parts the complex permittivity (ε′′) and permeability (μ′′) associate with the loss capability of electric and magnetic energy.28 The complex permittivity and permeability behaviors of graphene@carbon@Fe3O4 were studied as shown in Fig. 3. It is noting that the ε′ and ε′′ values of graphene@carbon@Fe3O4 are larger than those of GO and RGO@PDA (Fig. S3, ESI†), indicating the high storage and loss ability of the microwave energy. Furthermore, ε′ is an expression of polarization ability of a material which mainly arises from dipolar polarization and interfacial polarization at microwave frequency.29,30 In this case, the increasing ε′ may be ascribed to the dipolar polarization provided mainly by abundant surface functions and the defects in the graphene@carbon, and the interfacial polarization mainly from the interfaces in Fe3O4–carbon–graphene and a large amount of interfaces among Fe3O4 nanosheets. These contribute to the dielectric loss according to Debye theory.31 Another contribution to the dielectric loss is from conductivity loss originating from the 3D graphene@carbon@Fe3O4, which benefits from the excellent conductivity of graphene@carbon. Fig. 4b showed the frequency dependence of the μ′ and μ′′ of complex permeability of graphene@carbon@Fe3O4. Generally, the imaginary part μ′′ is related to the magnetic loss. Thus higher μ′′ of graphene@carbon@Fe3O4 lead to greater magnetic loss compared with those of GO and RGO@PDA (Fig. S3b, ESI†). The enhanced magnetic loss of materials originates mainly from natural resonance, exchange resonance and eddy current loss.32 The values of μ′′(μ′)−2f−1 are almost constant in 10–18 GHz (Fig. S4, ESI†). Therefore the magnetic loss of graphene@carbon@Fe3O4 is mainly originated from natural resonance, exchange resonance in 2–10 GHz and eddy current loss in 10–18 GHz. Moreover, the dielectric loss tangent (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δε = ε′′/ε′) and the magnetic loss tangent (tan
δε = ε′′/ε′) and the magnetic loss tangent (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δμ = μ′′/μ′) are two important factors to evaluate microwave absorption properties for an absorber. The largest value of loss tangent indicated the higher capacity of converting electromagnetic waves to other forms of energy.33 As shown in Fig. 4, the values of tan
δμ = μ′′/μ′) are two important factors to evaluate microwave absorption properties for an absorber. The largest value of loss tangent indicated the higher capacity of converting electromagnetic waves to other forms of energy.33 As shown in Fig. 4, the values of tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δε and tan
δε and tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δμ for graphene@carbon@Fe3O4 are obviously improved compared to these of GO and RGO@PDA shown in Fig. S3,† indicating the high capacity of converting electromagnetic waves to other forms of energy.
δμ for graphene@carbon@Fe3O4 are obviously improved compared to these of GO and RGO@PDA shown in Fig. S3,† indicating the high capacity of converting electromagnetic waves to other forms of energy.
 | ||
| Fig. 4 Frequency dependences of complex permittivity and dielectric loss (a), and complex permeability and magnetic loss (b) for the graphene@carbon@Fe3O4 nanosheet arrays. | ||
Based on the above analysis, the dipolar polarization and interfacial polarization will result in strong dielectric losses and take place at high frequency, and eddy current losses which lead to high magnetic losses also take place at high frequency, while natural resonance, exchange resonance work at low frequency. This complementary effect is helpful for the electromagnetic absorption properties, due to the better impedance matching, which is the important key factor for the enhanced microwave absorption properties. The impedance matching degree can be validated by a delta-function method by means of an equation: Δ = |sinh2(Kfd) − M|, where K and M can be determined by the relative complex permittivity and complex permeability.34 The smaller delta value implies better EM impedance matching. The delta values, smaller than 0.2, correspond to the RL below −8 dB. Based on the method proposed in ref. 34, the calculated delta value maps of graphene@carbon@Fe3O4 is shown in Fig. 5. The large dark blue area indicates the excellent microwave absorption properties.
In addition, EM wave attenuation in the interior of the absorber is another one of the important key factors for a remarkable absorption material. The attenuation constant α, determining the dissipation properties of materials, can be expressed by34
 | (3) |
On the basis of the above discussion, a plausible electromagnetic wave absorption mechanism for the 3D graphene@carbon@Fe3O4 nanosheet arrays is proposed, as depicted in Fig. 6. When the graphene@carbon@Fe3O4 is subjected to electromagnetic wave radiation, the Fe3O4 nanosheets grown on the surfaces of graphene@carbon are expected to act as antennae receivers allowing electromagnetic waves to penetrate into the interior of the absorber as much as possible the electromagnetic wave energy transfers in the form of microcurrent.35 The Fe3O4 nanosheets can convert more and more electromagnetic energy to microcurrent, due to the increase in impedance matching. The microwave current generated is then required to transmit along one sheet-like structure to another. During transmission, the graphene@carbon serves as an electrically conductive network. A large part of the electric energy could be attenuated due to the resistance of the graphene@carbon. Moreover, the special structure leads to defects and multi-interfaces (carbon–Fe3O4, Fe3O4–Fe3O4 and carbon–graphene), which can cause additional space charge polarization and interfacial polarization with the associated relaxation.36–38 These also could stimulate the electron polarization, favoring the attenuation electromagnetic wave.
 | ||
| Fig. 6 Schematic illustration of a plausible electromagnetic wave absorption mechanism for the graphene@carbon@Fe3O4 nanosheet arrays. | ||
4. Conclusions
In summary, we developed an efficient strategy to fabricate a 3D architecture of graphene@carbon@Fe3O4 nanosheet arrays by a mussel-inspired polydopamine adhesive, in which Fe3O4 nanosheet were almost grown perpendicularly on both surfaces of graphene sheets. After introducing Fe3O4 nanosheet arrays with accompanied reduction of GO into graphene, the 3D architectures exhibit outstanding EM absorption properties. The maximum reflection loss value can reach −52.8 dB at 9.5 GHz with the sample thickness of 2.7 mm. The improved EM wave absorption properties is realized by the synergistic effects of magnetic loss in Fe3O4 nanosheet arrays and dielectric loss in graphene@carbon. Furthermore, the synthesis strategy presented here can be expended as a facile approach to synthesizing related graphene-based 3D nanostructures for functional design and applications.Notes and references
- Y. Zhang, Y. Huang, T. F. Zhang, H. C. Chang, P. S. Xiao, H. H. Chen, Z. Y. Huang and Y. S. Chen, Adv. Mater., 2015, 12, 2049 CrossRef PubMed.
- M. Q. Ning, M. M. Lu, J. B. Li, Z. Chen, Y. K. Dou, C. Z. Wang, F. Rehman, M. S. Cao and H. B. Jin, Nanoscale, 2015, 15734 RSC.
- B. Wen, M. S. Cao, M. M. Lu, W. Q. Cao, H. L. Shi, J. Liu, X. X. Wang, H. B. Jin, X. Y. Fang, W. Z. Wang and J. Yuan, Adv. Mater., 2014, 26, 3484 CrossRef CAS PubMed.
- J. W. Liu, R. C. Che, H. J. Chen, F. Zhang, F. Xia, Q. S. Wu and M. Wang, Small, 2012, 8, 1214 CrossRef CAS PubMed.
- X. H. Li, J. Feng, Y. P. Du, J. T. Bai, H. M. Fan, H. L. Zhang, Y. Peng and F. S. Li, J. Mater. Chem. A, 2015, 3, 5535 CAS.
- L. Wang, Y. Huang, C. Li, J. J. Chen and X. Sun, Phys. Chem. Chem. Phys., 2015, 17, 5818 Search PubMed.
- L. Wang, Y. Huang, X. Sun, H. J. Huang, P. B. Liu, M. Zong and Y. Wang, Nanoscale, 2014, 6, 3157 RSC.
- H. Zhang, A. J. Xie, C. P. Wang, H. S. Wang, Y. H. Shen and X. Y. Tian, J. Mater. Chem. A, 2013, 1, 8547 CAS.
- Y. L. Ren, H. Y. Wu, M. M. Lu, Y. J. Chen, C. L. Zhu, P. Gao, M. S. Cao, C. Y. Li and Q. Y. Ouyang, ACS Appl. Mater. Interfaces, 2012, 4, 6436 CAS.
- H. L. Xu, H. Bi and R. B. Yang, J. Appl. Phys., 2012, 111, 07A522 Search PubMed.
- V. K. Singh, A. Shukla, M. K. Patra, L. Saini, R. K. Jani, S. R. Vadera and N. Kumar, Carbon, 2012, 50, 2202 CrossRef CAS.
- Y. L. Ren, C. L. Zhu, S. Zhang, C. Y. Li, Y. J. Chen, P. Gao, P. P. Yang and Q. Y. Ouyang, Nanoscale, 2013, 5, 12296 RSC.
- X. H. Li, H. B. Yi, J. W. Zhang, J. Feng, F. S. Li, D. S. Xue, H. L. Zhang, Y. Peng and N. J. Mellors, J. Nanopart. Res., 2013, 15, 1472 CrossRef.
- X. N. Chen, F. C. Meng, Z. W. Zhou, X. Tian, L. M. Shan, S. B. Zhu, X. L. Xu, M. Jiang, L. Wang, D. Hui, Y. Wang, J. Lu and J. H. Gou, Nanoscale, 2014, 6, 8140 RSC.
- L. Q. Xu, W. J. Yang, K. Neoh, E. Kang and G. D. Fu, Macromolecules, 2010, 43, 8336 CrossRef CAS.
- H. Lee, S. M. Dellatore, W. M. Miller and P. B. Messersmith, Science, 2007, 318, 426 CrossRef CAS PubMed.
- J. Ryu, S. H. Ku, H. Lee and C. B. Park, Adv. Funct. Mater., 2010, 20, 2132 CrossRef CAS.
- L. Q. Xu, W. J. Yang, K. G. Neoh, E. T. Kang and G. D. Fu, Macromolecules, 2010, 43, 8336 CrossRef CAS.
- L. S. Zhong, J. S. Hu, H. P. Liang, A. M. Cao, W. G. Song and L. J. Wan, Adv. Mater., 2006, 18, 2426 CrossRef CAS.
- G. Zhou, D. W. Wang, F. Li, L. Zhang, N. Li, Z. S. Wu, L. Wen, G. Q. Lu and H. M. Cheng, Chem. Mater., 2010, 22, 5306 CrossRef CAS.
- K. Singh, A. Ohlan, V. H. Pham, R. Balasubramaniyan, S. Varshney, J. Jang, S. H. Hur, W. M. Choi, M. Kumar, S. K. Dhawan, B. S. Kong and J. S. Chung, Nanoscale, 2013, 5, 2411 RSC.
- X. Xu, H. Li, Q. Q. Zhang, H. Hu, Z. B. Zhao, J. H. Li, J. Y. Li, Y. Qiao and Y. Gogotsy, ACS Nano, 2015, 9, 3969 CrossRef CAS PubMed.
- J. Zhou, H. Song, L. Ma and X. Chen, RSC Adv., 2011, 1, 782 RSC.
- N. Zubir, C. Yacou, J. Motuzas, X. Zhang and J. Costa, Sci. Rep., 2014, 4, 4594 CrossRef PubMed.
- E. Michielssen, J. M. Sajer, S. Ranjithan and R. Mittra, IEEE Trans. Microwave Theory Tech., 1993, 41, 1024 CrossRef CAS.
- C. Wang, X. Han, X. Zhang, S. Hu, T. Zhang, J. Wang, Y. Du, X. Wang and P. Xu, J. Phys. Chem. C, 2010, 114, 14826–14830 CAS.
- B. Zhao, G. Shao, B. Fan, W. Zhao and R. Zhang, Phys. Chem. Chem. Phys., 2015, 17, 2531 RSC.
- Y. C. Du, W. W. Liu, R. Qiang, Y. Wang, X. J. Han, J. Ma and P. Xu, ACS Appl. Mater. Interfaces, 2014, 6, 12997 CAS.
- Q. L. Liu, D. Zhang and T. X. Fan, Appl. Phys. Lett., 2008, 93, 013110 CrossRef.
- P. C. P. Watts, W. K. Hsu, A. Barnes and B. Chambers, Adv. Mater., 2003, 15, 600 CrossRef CAS.
- B. Wen, M. S. Cao, Z. L. Hou, W. L. Song, L. Zhang, M. M. Lu, H. B. Jin, X. Y. Fang, W. Z. Wang and J. Yuan, Carbon, 2013, 65, 124 CrossRef CAS.
- F. S. Wen, F. Zhang and Z. Y. Liu, J. Phys. Chem. C, 2011, 115, 14025 CAS.
- X. M. Zhang, G. B. Ji, W. Liu, B. Quan, X. H. Liang, C. M. Shang, Y. Cheng and Y. W. Du, Nanoscale, 2015, 7, 12932 RSC.
- Z. Ma, C. T. Cao, Q. F. Liu and J. B. Wang, Chin. Phys. Lett., 2012, 29, 038401 CrossRef.
- R. F. Zhuo, L. Qiao, H. T. Feng, J. T. Chen and D. Yan, J. Appl. Phys., 2008, 104, 094101 CrossRef.
- M. Zong, Y. Huang, Y. Zhao, X. Sun, C. H. Qu, D. D. Luo and J. B. Zheng, RSC Adv., 2013, 3, 23638 RSC.
- M. Zong, Y. Huang, H. W. Wu, Y. Zhao, Q. F. Wang and X. Sun, Mater. Lett., 2014, 114, 52 CrossRef CAS.
- M. Zong, Y. Huang, N. Zhang and H. W. Wu, J. Alloys Compd., 2015, 644, 491 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: SEM images of GO@iron alkoxide, the details about the formation of RGO@PDA@iron alkoxide nanosheet arrays, XRD pattern and Raman spectra of samples, frequency dependence of complex permeability and complex permittivity for GO and RGO@PDA, respectively, frequency dependence of μ′′(μ′)−2f−1 for graphene@carbon@Fe3O4. See DOI: 10.1039/c5ra21546c |
| This journal is © The Royal Society of Chemistry 2015 |





