Protonable pyrimidine derivative for white light emission†
Sylvain Achelle*ab,
Julián Rodríguez-López*b,
Nolwenn Cabona and
Françoise Robin-le Guena
aInstitut des Sciences Chimiques de Rennes UMR CNRS 6226, IUT de Lannion, rue Edouard Branly, BP 30219, F22302 Lannion Cedex, France. E-mail: sylvain.achelle@univ-rennes1.fr; Tel: +33 2 96 46 94 48
bFacultad de Ciencias y Tecnologías Químicas, Universidad de Castilla-La Mancha, 13071 Ciudad Real, Spain. E-mail: julian.rodriguez@uclm.es; Tel: + 34 926 295 300
First published on 11th December 2015
Abstract
White photoluminescence both in solution and the solid-state was obtained by the controlled protonation of a blue emitting 4,6-bis(arylvinyl)pyrimidine, which resulted in the formation of an orange emissive acidified form. This dye has potential applications in the fabrication of white OLEDs based on only one material.
In the context of the reduction of energy consumption, and taking into account that lighting accounts for ∼20% of energy consumption worldwide, there is great interest in research into new light emitting devices, particularly Light Emitting Diodes (LED), that consume less energy.1
In the field of light-emitting molecules, organic materials that incorporate a π-conjugated backbone are generally preferred over their inorganic counterparts owing to their low cost, ease of fine-tuning, solution processability, low toxicity and sufficient flexibility for device fabrication.2
Organic LEDs (OLEDs) that emit white light were first reported by Kido et al.3 White OLEDs (WOLEDs) can now outperform incandescent light bulbs and even fluorescent tubes in terms of luminous efficiency.4 A good white-light emitter should be a ‘warm’ white, as defined by colour coordinates close to the Planckian locus around the equi-energy white point (x = 0.33, y = 0.33 in the Commission International de l'Eclairage (CIE) 1931 diagram).2d,5
Various methods for the fabrication of WOLEDs have been reported. Such devices are generally obtained by doping mixed emitting molecules of complementary colours either in a single emitting layer6 or in multi-emitting layers.7 Unfortunately, this strategy generally requires a complex and costly fabrication process. A new strategy for WOLED fabrication has recently been proposed and this involves the use of only one emitting material that can take two forms of complementary emitting colours. Some examples have been described in the literature and these consist of monomer/excimer,8 neutral/protonated,9 neutral/deprotonated species,10 or free/complexed ligand.11
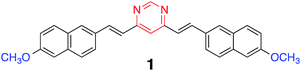
The replacement of CH groups by N atoms in π-conjugated frameworks dramatically changes the positions of the highest occupied molecular orbital (HOMO), the lowest unoccupied molecular orbital (LUMO) and the emission spectra.12 In this context, we have described a large library of push–pull pyrimidine chromophores.13 When substituted by electron-donating fragments via π-conjugated linkers, these compounds are highly fluorescent and their emission properties are highly sensitive to external stimuli such as solvent polarity,13,14 pH,13b–e,14a and metal cation complexation.15 Protonation of push–pull pyrimidine derivatives leads to a bathochromic shift in the absorption. Whereas the emission is often quenched upon protonation, methoxy-substituted pyrimidines generally exhibit a red-shifted emission.16 However, it should be noted that methoxy-substituted compounds with high emission quantum yields require an extended π-conjugated bridge.17 We recently described the synthesis and basic photophysical properties of 4,6-bis[2-(6-methoxynaphthalen-2-yl)-vinyl]pyrimidine 1 (Chart 1). The synthesis of 1 is straightforward, affordable and environmentally friendly. In solution of dichloromethane this compound emits blue light at 470 nm (ΦF = 0.38) that can be tuned to orange by protonation.13e Herein, we describe the emission properties of a mixture of neutral and protonated forms of compound 1 both in solution and in thin films. The controlled protonation of this blue emitting dye led to white photoluminescence. The electronic properties were also studied by cyclic voltammetry.
The changes in the UV-vis spectra of 1 upon addition of trifluoroacetic acid (TFA) are illustrated in Fig. 1. The spectra show the progressive attenuation of the charge transfer absorption band for the neutral compound on increasing the concentration of acid, whereas a new red-shifted band corresponding to the protonated species appeared. This bathochromic shift is explained by an enhancement of the intramolecular charge transfer (ICT) into the molecule due to the increase in the electron-attracting character of the pyrimidine ring by protonation. As far as the photoluminescence is concerned, the same trend was observed: addition of TFA led to a decrease in the emission band intensity and a new red-shifted band appeared (Fig. 2).
 | ||
| Fig. 1 Changes in the absorption spectra of 1 (c = 8.8 10−3 M) in CH2Cl2 upon addition of TFA (20 to 1000 equivalents). | ||
 | ||
| Fig. 2 Changes in the emission spectra of 1 (c = 8.8 10−3 M) in CH2Cl2 upon addition of TFA (20 to 1000 equivalents), λexc = 377 nm. | ||
When excited at 377 nm, the intensity of both bands became similar after the addition of 50–100 equivalents of TFA. With 50 equivalents of TFA, the CIE coordinates (0.30, 0.35) were very close to those of pure white light emission (0.33, 0.33) (Fig. 3). This solution showed an intense white colour under UV irradiation, whereas solutions of non-protonated and fully-protonated compound 1 appeared blue and orange, respectively (Fig. 4). It is worth noting that changes in the excitation wavelength also dramatically modified the relative intensities of both bands (see ESI, Fig. S1†).
 | ||
| Fig. 3 Chromaticity of the fluorescence colour of 1 in CH2Cl2 obtained in the absence and the presence of TFA in a 1931 CIE diagram. | ||
The large excess of acid required to see the change in the UV-vis and emission spectra is in line with the reduced basicity of the nitrogen atoms of the pyrimidine ring. Solvents such as diethyl ether and ethanol are basic enough to revert the process. In both the absorption and emission spectra, isosbestic points were observed for low concentrations of acid. Nevertheless, these isosbestic points disappeared due to the significant change in polarity when the concentration of acid increased. It should also be noted that above 200 equivalents of TFA, the intensity of the red-shifted band began to decrease for the same reason.
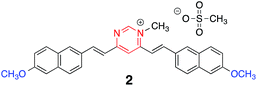
Protonation was also accompanied by a noticeable downfield shift of most of the signals in the 1H NMR spectrum of chromophore 1 (see ESI, Fig. S2†). In order to confirm that protonation took place on the pyrimidine ring, we prepared the methylated species 2 (Chart 2) that showed similar absorption and emission spectra to those of the protonated form of 1, supporting the protonation of the pyrimidine ring in the presence of TFA (see ESI and Fig. S3 and S4†).
Despite their high emission efficiency in solution, many organic materials are non-luminescent in the solid state because of the fluorescence quenching caused by intermolecular interactions occurring in the condensed phase. Thin films formed by spray deposition of a CH2Cl2 solution of 1 were luminescent but the emission spectra were significantly modified, a finding that suggests the formation of aggregates. One way to overcome this type of problem involves the incorporation of the fluorophore into a polymer matrix;18 polystyrene is particularly adapted in this context.19 Thus, thin films of polystyrene doped with 1 wt% of 1 were obtained by spray deposition on glass substrates. These films exhibited a slightly blue-shifted emission (λem = 443 nm, ΦF = 0.34) when compared with chromophore 1 in solution (λem = 470 nm). Once again, the addition of 50 equivalents of TFA in the solution used for spray deposition resulted in a film that showed two emission bands (ΦF = 0.33) with CIE coordinates of (0.31, 0.30), i.e., close to pure white (Fig. 5). The change in the emission colour could easily be seen with the naked eye, as shown in Fig. 6. In contrast to the solution, the thin film of the fully protonated compound was only weakly luminescent.
 | ||
| Fig. 5 Emission spectra of a polystyrene thin film doped with 1 wt% of 1 in the absence (blue) and the presence (red, 50 equivalents) of TFA. | ||
In an effort to gain further insights into the electronic properties of compound 1 and its protonated form, the redox properties were measured by cyclic voltammetry, in DMF-[Bu4N][PF6], in the absence and presence of p-toluenesulfonic acid (p-TsOH). Compound 1 showed an irreversible oxidation peak and a reversible one-electron reduction at E1/2 = −1.95 V vs. the ferrocenium/ferrocene couple (Fc+/0) (Fig. 7). The oxidation peak was followed by a close irreversible process, which precluded the accurate determination of the oxidation potential. Nevertheless, on using differential pulse voltammetry it was possible to determine the potential of this peak as Eoxp = 0.82 V vs. Fc+/0 (see ESI, Fig. S5†). The addition of p-TsOH led to a new irreversible reduction peak at Eredp = −1.05 V vs. Fc+/0, a potential that is less negative than the one for the reduction of 1 (Table 1). Although the shift in the reduction peak was noteworthy (0.90 V), a significant effect was not observed in the oxidation process (0.03 V).
 | ||
| Fig. 7 Cyclic voltammogram of 1 (c = 1.5 mM) before (black) and after (red) addition of p-TsOH. E (V) vs. Fc+/Fc couple. Scan rate υ = 0.1 V s−1. | ||
Thermal stability is a key parameter for OLED applications. Thermogravimetric analysis (TGA) of 1 (see ESI, Fig. S6†) showed good stability with a thermal decomposition occurring at around 400 °C, which is sufficiently high for applications in electro-optical device preparation.
In summary, a pyrimidine chromophore that exhibited white photoluminescence by controlled protonation both in solution and the solid state has been designed. The white luminescence is based on the partial protonation of this blue-light emitting dye. Protonation of the electron-deficient pyrimidine ring provides a significant enhancement of its electron-withdrawing character, as shown by the dramatic effect on the reduction potential, and leads to a red-shifted luminescence. The electroluminescence properties of this chromophore are currently under investigation.
Acknowledgements
SA acknowledges a short-term scholarship for invited professors from the Universidad de Castilla La-Mancha. JR-L thanks the Junta de Comunidades de Castilla-La Mancha/FEDER (European Union) for financial support (project PEII-2014-005 A). The authors also thank Dr G. Calvez for the quantum yield measurements in solid state. NC gratefully acknowledges Dr F. Gloaguen and UMR 6221 for their support.Notes and references
- Solid State and Other Energy Efficient Lighting Systems, Application and Market Trends (2010-2015), http://www.marketsandmarkets.com (accessed October 2015).
- (a) H. Klauk, Organic electronics : materials, manufacturing, and applications, Wiley-VCH, 2006 Search PubMed; (b) Z. Li, Z. R. Li and H. Meng, Organic light-emitting materials and devices (Optical science and engeneering), Taylor & Francis, 2006 Search PubMed; (c) T. Tsujimura, OLED display fundamentals and applications (Wiley series in display technology), Wiley-VCH, 2012 Search PubMed; (d) S. Mukherjee and P. Thilagar, Dyes Pigm., 2014, 110, 2 CrossRef CAS.
- J. Kido, K. Hongawa, K. Okuyama and K. Nagai, Appl. Phys. Lett., 1994, 64, 815 CrossRef CAS.
- M. C. Gather, R. Alle, H. Becker and K. Meerholz, Adv. Mater., 2007, 19, 4460 CrossRef CAS.
- T. Jeon, B. Geffroy, D. Tondelier, Y. Bonnassieux, S. Forget, S. Chenais and E. Ishow, Thin Solid Films, 2013, 542, 263 CrossRef CAS.
- (a) B. W. D'Andrade, R. J. Holmes and S. R. Forrest, Adv. Mater., 2004, 16, 624 CrossRef; (b) B. W. D'Andrade, M. E. Thompson and S. R. Forest, Adv. Mater., 2002, 14, 147 CrossRef; (c) T.-H. Kim, H. K. Lee, O. O. Park, B. D. Chin, S.-H. Lee and J. K. Kim, Adv. Funct. Mater., 2006, 16, 611 CrossRef CAS.
- Y. Sun, N. C. Giebink, H. Kanno, B. Ma, M. E. Thompson and S. R. Forrest, Nature, 2006, 440, 908 CrossRef CAS PubMed.
- (a) S. Tao, Y. Zhou, C.-S. Lee, S.-T. Lee, D. Huang and X. Zhang, J. Mater. Chem., 2008, 18, 3981 RSC; (b) L. Liu, F. Chen, B. Xu, Y. Dong, Z. Zhao, W. Tian and L. Ping, Synth. Met., 2010, 160, 1968 CrossRef CAS; (c) T. Fleetham, J. Ecton, Z. X. Wang, N. Bakken and J. Li, Adv. Mater., 2013, 25, 2573 CrossRef CAS PubMed.
- (a) D. Liu, Z. Zhang, H. Zhang and Y. Wang, Chem. Commun., 2013, 49, 10001 RSC; (b) H. V. Huynh, X. He and T. Baumgartner, Chem. Commun., 2013, 49, 4899 RSC; (c) C. Romero-Nieto, S. Durben, I. M. Kormos and T. Baumgartner, Adv. Funct. Mater., 2009, 19, 3625 CrossRef CAS.
- Y. Yang, M. Lowry, C. M. Schowalter, S. O. Fakayode, J. O. Escobedo, X. Xu, H. Zhang, T. J. Jensen, F. R. Fronczek, I. M. Warner and R. M. Strongin, J. Am. Chem. Soc., 2006, 128, 14081 CrossRef CAS PubMed.
- Y. Shiraishi, C. Ichimura, S. Sumiya and T. Hirai, Chem.–Eur. J., 2011, 17, 8324 CrossRef CAS PubMed.
- (a) P.-Y. Gu, Y. Zhao, J.-H. He, J. Zhang, C. Wang, Q.-F. Xu, J.-M. Lu, X. W. Sun and Q. Zhang, J. Org. Chem., 2015, 80, 3030 CrossRef CAS PubMed; (b) A. Mateo-Alonso, Chem. Soc. Rev., 2014, 43, 6311 RSC; (c) S. More, R. Bhosale and A. Mateo-Alonso, Chem.–Eur. J., 2014, 20, 10626 CrossRef CAS PubMed.
- (a) S. Achelle, A. Barsella, B. Caro and F. Robin-le Guen, RSC Adv., 2015, 5, 39218 RSC; (b) S. Achelle, J. Rodríguez-López and F. Robin-le Guen, J. Org. Chem., 2014, 79, 7564 CrossRef CAS PubMed; (c) S. Achelle, A. Barsella, C. Baudequin, B. Caro and F. Robin-le Guen, J. Org. Chem., 2012, 77, 4087 CrossRef CAS; (d) C. Hadad, S. Achelle, J. C. García-Martinez and J. Rodríguez-López, J. Org. Chem., 2011, 76, 3837 CrossRef CAS PubMed; (e) S. Achelle, I. Nouira, B. Pfaffinger, Y. Ramondenc, N. Plé and J. Rodríguez-López, J. Org. Chem., 2009, 74, 3711 CrossRef CAS PubMed.
- (a) S.-i. Kato, Y. Yamada, H. Hiyoshi, K. Umezu and Y. Nakamura, J. Org. Chem., 2015, 80, 9076 CrossRef CAS PubMed; (b) L. Skardziute, J. Dodonova, A. Voitechovicius, J. Jovaisaite, R. Komskis, A. Voitechoviciute, J. Bucevicius, K. Kazlauskas, S. Jursenas and S. Tumkevicius, Dyes Pigm., 2015, 118, 118 CrossRef CAS; (c) E. V. Verbitskiy, A. V. Schepochkin, N. I. Makarova, I. V. Dorogan, A. V. Metelitsa, V. I. Minkin, S. A. Kozykhin, V. V. Emets, V. A. Grindberg and O. N. Chupakhin, J. Fluoresc., 2015, 25, 763 CrossRef CAS PubMed.
- (a) S. Achelle, J. Rodríguez-López, F. Bureš and F. Robin-le Guen, Dyes Pigm., 2015, 121, 305 CrossRef CAS; (b) C. Hadad, S. Achelle, I. López-Solera, J. C. García-Martínez and J. Rodríguez-López, Dyes Pigm., 2013, 97, 230 CrossRef CAS.
- S. Achelle and F. Robin-le Guen, Tetrahedron Lett., 2013, 54, 4491 CrossRef CAS.
- C. Denneval, S. Achelle, C. Baudequin and F. Robin-le Guen, Dyes Pigm., 2014, 110, 49 CrossRef CAS.
- (a) H. E. Ooyama, T. Ide, H. Yamasaki, A. Harada, Y. Nagahama, A. Ono and K. Yoshida, Dyes Pigm., 2012, 94, 103 CrossRef CAS; (b) G. Martini, E. Martinelli, G. Ruggeri, G. Galli and A. Pucci, Dyes Pigm., 2015, 113, 47 CrossRef CAS; (c) A. Beneduci, S. Cospito, M. La Deda and G. Chidichimo, Adv. Funct. Mater., 2015, 25, 1240 CrossRef CAS.
- (a) G. He, Y. Li and Y. Yang, Appl. Phys. Lett., 2002, 80, 4247 CrossRef CAS; (b) T. Serevičius, R. Komskis, P. Adomėnas, O. Adomėnienė, V. Jankauskas, A. Gruodis, K. Kazlauskas and S. Juršėnas, Phys. Chem. Chem. Phys., 2014, 16, 7089 RSC.
Footnote |
| † Electronic supplementary information (ESI) available: 1H and 13C NMR spectra of 1 before and after protonation, influence of excitation wavelength on emission spectrum of 1 with 100 eq. of TFA, differential pulse voltammetry and TGA plot. See DOI: 10.1039/c5ra21514e |
| This journal is © The Royal Society of Chemistry 2015 |