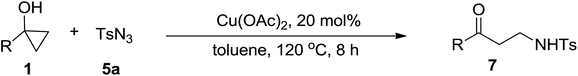Copper(II) acetate catalysed ring-opening cross-coupling of cyclopropanols with sulfonyl azides†
Mei-Hua Shen*,
Xiao-Long Lu and
Hua-Dong Xu*
School of Pharmaceutical Engineering and Life Science, Changzhou University, Changzhou, Jiangsu Province 213164, China. E-mail: shenmh@cczu.edu.cn; hdxu@cczu.edu.cn
First published on 9th November 2015
Abstract
A copper(II) acetate catalyzed ring-opening cross-coupling of cyclopropanol with sulfonyl azide has been developed. By this method, various β-amino ketones have been made efficiently in medium to high yields and venerable functional groups such as benzylic C–H, alkyl and aryl bromides, alkyl sulfonate, silyl ether and alkene are compatible with these reaction conditions. Control experiments have precluded the involvement of both radical and simple copper nitrene intermediates and a possible mechanism featuring key steps of ring-opening metalation and alkyl group migratory insertion into copper nitrene has been proposed.
The energy favouring ring-opening of the cyclopropanyl group has been exploited to develop a plethora of elegant synthetic methodologies.1 Factors that induce the C–C bond breaking in this small ring are extremely diverse depending on the nature of the ring itself, which is defined by the substitution pattern on the ring, and reaction conditions. For simple cyclopropanol, the ring opening event normally occurs in a high regioselective fashion due to the effect of the hydroxyl group.2 Roughly, these reactions fall into three categories in the mechanistic respect: (a) base and electrophile promoted heterolytic cleavage of cyclopropanol ring through the intermediacy of homoenolate I or oxonium ion II (Scheme 1, a);3 (b) single electron oxidation induced homolytic breaking of C–C bond resulting in β-carbonyl radical III which can be intercepted by various carbon radical receptors (Scheme 1, b);4 (c) activation and removal of the hydroxyl group to give rise to cyclopropanyl cation IV, which would open up to form more stable allylic cation V for further reactions (Scheme 1, c).5
The incorporation of a transition metal would expand the reaction scope and improve their usefulness via the intermediary of metal homoenolate M-I. Indeed, both palladium and copper are reported to mediate ring-opening coupling reactions of cyclopropanol with carbon-based partners.6 Very recently, transition metal catalysed conversion of cyclopropanol to β-F, β-CF3/SCF3 and β-NR2 substituted ketones have appeared in literature.7 On the other hand, sulfonyl azide has been used in various metal catalysed transformations via metal nitrene M-VI, metal-nitrene radical M-VII or other reactive species as key reaction intermediates.8 In line with our interests in azide chemistry,9 we are curious that if the chemistry of cyclopropanol and sulfonyl azide could interwoven through transition metal catalysis (Fig. 1).
 | ||
| Fig. 1 Conceptual depiction of interweaving of cyclopropanol chemistry with sulfonyl azide chemistry by metal catalysis. | ||
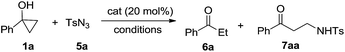
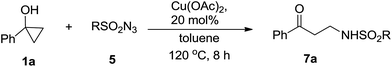
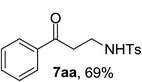
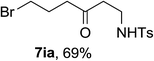
Our investigation started with catalyst screening for reaction of phenyl cyclopropanol 1a with tosyl azide 5a (Table 1). In most cases, propiophenone 6a was the only observable product (see ESI-Table 1†). To our delight, when a solution of 1a and 5a in dichloroethane (DCE) was heated to reflux in the presence of a catalytic amount Cu(OAc)2, ring-opening cross coupling product 7aa was isolated in 40% yield along with 6a (Table 1, entry 1). Interestingly, while Cu(acac)2 was slightly less effective in terms of yield when DCE was used for reaction media (entry 1 vs. 2), CuBr2 was totally ineffective for the cross-coupling reaction (entry 3) indicating the importance of ligand effect. Common copper(I) complexes all failed to deliver 7aa (entries 4–6) and only 6a was obtained. When the reaction was performed in reflux toluene using Cu(OAc)2 as catalyst, the yield of cross-coupling product 7aa increased to 69% (entry 7). More electron deficient Cu(hfacac)2 showed much poorer activity than Cu(acac)2 in toluene (entry 8 vs. 9). Toluene was superior solvent than DCE, THF and chloroform (entries 10 and 11). Interestingly, in polar solvents such as acetonitrile, DMF and DMSO, 1a remained intact under the same conditions (entries 12–14).
| Entry | Cat | Solvent | Temp | Productsb,c (yield) |
|---|---|---|---|---|
| a Conditions 1a (0.5 mmol), 5a (0.75 mmol), cat (20% mol%), solvent (2 mL), heating, 12 h.b Isolated yields.c Yields for 7aa are reported. | ||||
| 1 | Cu(OAc)2 | DCE | Reflux | 6a + 7aa (40%) |
| 2 | Cu(acac)2 | DCE | Reflux | 6a + 7aa (37%) |
| 3 | CuBr2 | DCE | Reflux | 6a only |
| 4 | CuCl | DCE | Reflux | 6a only |
| 5 | CuBr | DCE | Reflux | 6a only |
| 6 | CuI | DCE | Reflux | 6a only |
| 7 | Cu(OAc)2 | Toluene | Reflux | 6a + 7aa (69%) |
| 8 | Cu(acac)2 | Toluene | Reflux | 6a + 7aa (51%) |
| 9 | Cu(hfacac)2 | Toluene | Reflux | 6a + 7aa (15%) |
| 10 | Cu(OAc)2 | THF | Reflux | 6a + 7aa (29%) |
| 11 | Cu(OAc)2 | CHCl3 | Reflux | 6a + 7aa (27%) |
| 12 | Cu(OAc)2 | CH3CN | Reflux | No reaction |
| 13 | Cu(OAc)2 | DMF | 120 °C | No reaction |
| 14 | Cu(OAc)2 | DMSO | 120 °C | No reaction |
The effect of sulfonyl azide was also checked with Cu(OAc)2 as catalyst and toluene as solvent (Table 2). TsN3, MbsN3 and MsN3 afforded corresponding sulfonyl amides in comparable yields (entries 1–3). MtsN3, TcesN3 and CamsN3 were much inferior reaction partners for this reaction (entries 4–6), and electron deficient NsN3 reacted with 1a to give a complex mixture.‡
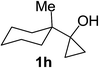
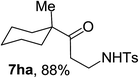
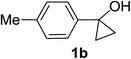
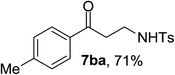
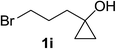
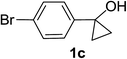
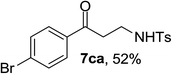
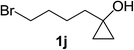
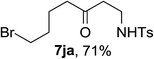
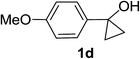
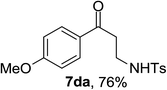
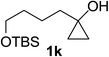
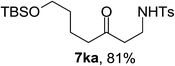
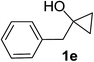
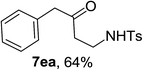
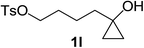
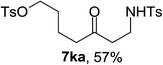
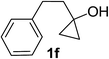
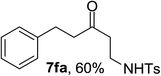
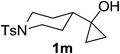
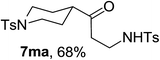
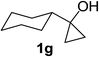
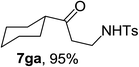
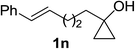
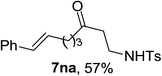
Next the scope of cyclopropanol was investigated with TsN3 as the amination agent (Table 3). For aryl cyclopropanols 1a–d, the yield of corresponding β-tosylamidylaryl ketones increased from 52% to 76%, suggesting that the electron-donating group on the phenyl ring favor the ring open/coupling reaction (from entry 3 to 1, 2 and 4). Substrates with benzylic C–H group which is potential reactive site in metal catalyzed reactions involving sulfony azide were also viable for this reaction, as both 7ea and 7fa were obtained smoothly (entries 5 and 6) in comparable yields. 1-Cyclohexylcyclopropanol 1g reacted with TsN3 in the same conditions to give 7ga in an excellent yield (entry 7, 95%). Methyl cyclohexylcyclopropanol 1h afford 7ha in 88% yield (entry 8). The slight decrease in yield for 7ha than for 7ga might reflect the effect of increased bulkiness at the alkyl group in 1h. Functional groups such as alkyl/aryl bromide, alkyl silyl ether, alkylsulfonate and alkylsulfonamide were all tolerated in this reaction conditions and related β-tosylamide aryl ketones 7ia-ma were all generated in medium to high yields (entries 9–13). It is worth to note that 1n bearing a vulnerable alkene group was also a good substrate to give 7na in 57% (entry 14).
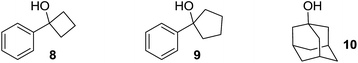
Cyclobutanol 8, cyclopentanol 9 and adamantanol 10 failed to undergo analogous ring breaking/coupling reaction when subjected under the same reaction conditions. These experiments underscore the necessity of three-membered ring for the reaction.
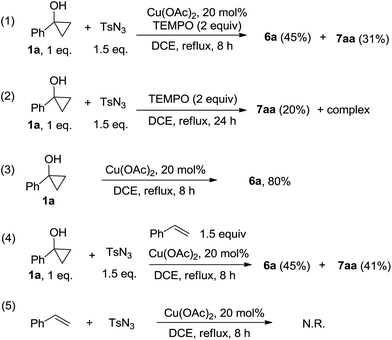
To gain more information about this reaction, control experiments have been carried out (Scheme 2, eqn 1–4). It was found that TEMPO has neglectable effect on this reaction. When a mixture of 1a (1 equiv.), 5a (1.5 equiv.), TEMPO (2 equiv.) and Cu(OAc)2 was heated in DCE for 8 hours, 7aa and 6a were isolated as sole products (Scheme 2, 1), while increasing the amount of TEMPO to 10 equiv., the formation of 7aa was not observed. Interestingly, in the presence of 2 equiv. TEMPO but free of Cu(OAc)2, 7aa could be isolated in 20% yield (Scheme 2, 2). Reaction without sulfonyl azide, phenyl cyclopropanol 1a was converted to ketone 6a in a high isolated yield (Scheme 2, 3). In the presence of 1.5 equiv. styrene, the reaction took place as normal and no phenyl tosylaziridine was observed (Scheme 2, 4). No reaction happened when styrene and TsN3 was heated with catalytic amount of Cu(OAc)2 (Scheme 2, 5). These results suggested there was no simple copper nitrene species TsN = Cu(OAc) in the reaction system.10
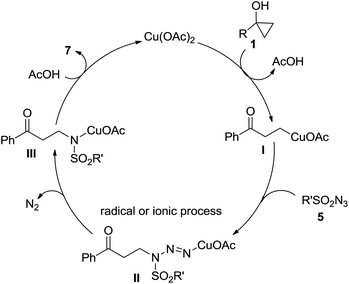
In view our results and literature precedence,11 a mechanism depicted in Scheme 3 was proposed even though a free radical alternate couldn't be excluded at this stage.12 The catalytic circle starts with a Cu(OAc)2 promoted ring-opening metalation of cyclopropanol 1 to give alkyl copper(II) homoenolate I. Protonation of this species would lead to the side product ketone 6. On the other hand, Cu(II) species I could be harnessed by sulfonyl azide 5 either through a transition metal mediated mechanism or a copper coupled radical process to construct the key C–N bond giving rise to intermediate II which, upon release of N2, would produce intermediate III. Subsequently, ligand exchange would take place with AcOH to release product 7 and Cu(OAc)2 to complete the catalytic circle. This mechanism is also in line with the ineffectiveness of Cu(I) salts as catalyst for this reaction.
Conclusions
In summary, we have described a copper(II) acetate catalysed ring-opening cross-coupling of cyclopropanol with sulfonyl azide to produce β-amino ketone. The conditions of this reaction are mild enough to be compatible with a number of fragile functionalities.Acknowledgements
The authors wish to thank the Natural Science Foundation of China (21402014 and 21272077), the Natural Science Foundation of Jiangsu Province (BK20131143), the Priority Academic Program Development of Jiangsu Higher Education Institutions (PADA), and Jiangsu Key Laboratory of Advanced Catalytic Materials and Technology (BM2012110). We also thank Prof. Lei Zhu at Florida State University for mechanism discussion.References
- (a) M. A. Cavitt, L. H. Phun and S. France, Chem. Soc. Rev., 2014, 43, 804 RSC; (b) T. F. Schneider, J. Kaschel and D. B. Werz, Angew. Chem., Int. Ed., 2014, 53, 5504 CrossRef CAS PubMed; (c) L. Jiao and Z.-X. Yu, J. Org. Chem., 2013, 78, 6842 CrossRef CAS PubMed; (d) M. H. Shaw, R. A. Croft, W. G. Whittingham and J. F. Bower, J. Am. Chem. Soc., 2015, 137, 8054 CrossRef CAS; (e) H. Xu, J.-L. Hu, L. Wang, S. Liao and Y. Tang, J. Am. Chem. Soc., 2015, 137, 8006 CrossRef CAS PubMed; (f) J. Zhu, Y. Liang, L. Wang, Z.-B. Zheng, K. N. Houk and Y. Tang, J. Am. Chem. Soc., 2014, 136, 6900 CrossRef CAS PubMed; (g) P. A. Wender, D. N. Fournogerakis, M. S. Jeffreys, R. V. Quiroz, F. Inagaki and M. Pfaffenbach, Nat. Chem., 2014, 6, 448 CrossRef CAS PubMed; (h) T. Hashimoto, Y. Kawamata and K. Maruoka, Nat. Chem., 2014, 6, 702 CAS; (i) H. Xu, W. Zhang, D. Shu, J. B. Werness and W. Tang, Angew. Chem., Int. Ed., 2008, 47, 8933 CrossRef CAS PubMed.
- O. G. Kulinkovich, Chem. Rev., 2003, 103, 2597 CrossRef CAS PubMed.
- N. H. Werstiuk, Tetrahedron, 1983, 39, 205 CrossRef CAS.
- (a) J. Jiao, L. X. Nguyen, D. R. Patterson and R. A. Flowers, Org. Lett., 2007, 9, 1323 CrossRef CAS; (b) A. Ilangovan, S. Saravanakumar and S. Malayappasamy, Org. Lett., 2013, 15, 4968 CrossRef CAS PubMed.
- C. H. DePuy, L. G. Schnack and J. W. Hausser, J. Am. Chem. Soc., 1966, 88, 3343 CrossRef CAS.
- (a) R. V. N. S. Murali, N. N. Rao and J. K. Cha, Org. Lett., 2015, 17, 3854 CrossRef CAS PubMed; (b) K. Cheng and P. J. Walsh, Org. Lett., 2013, 15, 2298 CrossRef CAS PubMed; (c) N. N. Rao, B. B. Parida and J. K. Cha, Org. Lett., 2014, 16, 6208 CrossRef CAS PubMed; (d) I. Ryu, K. Matsumoto, Y. Kameyama, M. Ando, N. Kusumoto, A. Ogawa, N. Kambe, S. Murai and N. Sonoda, J. Am. Chem. Soc., 1993, 115, 12330 CrossRef CAS; (e) D. Rosa and A. Orellana, Chem. Commun., 2013, 49, 5420 RSC; (f) D. Rosa and A. Orellana, Org. Lett., 2011, 13, 110 CrossRef CAS PubMed; (g) H. B. Lee, M. J. Sung, S. C. Blackstock and J. K. Cha, J. Am. Chem. Soc., 2001, 123, 11322 CrossRef CAS.
- (a) H. Zhao, X. Fan, J. Yu and C. Zhu, J. Am. Chem. Soc., 2015, 137, 3490 CrossRef CAS PubMed; (b) S. Ren, C. Feng and T.-P. Loh, Org. Biomol. Chem., 2015, 13, 5105 RSC; (c) Y. Li, Z. Ye, T. M. Bellman, T. Chi and M. Dai, Org. Lett., 2015, 17, 2186 CrossRef CAS PubMed; (d) Z. Ye and M. Dai, Org. Lett., 2015, 17, 2190 CrossRef CAS PubMed; (e) S. Bloom, D. D. Bume, C. R. Pitts and T. Lectka, Chem.–Eur. J., 2015, 21, 8060 CrossRef CAS PubMed.
- (a) M. Goswami, V. Lyaskovskyy, S. R. Domingos, W. J. Buma, S. Woutersen, O. Troeppner, I. Ivanović-Burmazović, H. Lu, X. Cui, X. P. Zhang, E. J. Reijerse, S. DeBeer, M. M. van Schooneveld, F. F. Pfaff, K. Ray and B. de Bruin, J. Am. Chem. Soc., 2015, 137, 5468 CrossRef CAS PubMed; (b) H. Lu, C. Li, H. Jiang, C. L. Lizardi and X. P. Zhang, Angew. Chem., Int. Ed., 2014, 53, 7028 CrossRef CAS PubMed; (c) H. Han, S. B. Park, S. K. Kim and S. Chang, J. Org. Chem., 2008, 73, 2862 CrossRef CAS PubMed; (d) J. Peng, Z. Xie, M. Chen, J. Wang and Q. Zhu, Org. Lett., 2014, 16, 4702 CrossRef CAS PubMed; (e) T. Kang, Y. Kim, D. Lee, Z. Wang and S. Chang, J. Am. Chem. Soc., 2014, 136, 4141 CrossRef CAS PubMed; (f) H. Kim, J. Park, J. G. Kim and S. Chang, Org. Lett., 2014, 16, 5466 CrossRef CAS PubMed; (g) K. Shin and S. Chang, J. Org. Chem., 2014, 79, 12197 CrossRef CAS PubMed; (h) K. Shin, H. Kim and S. Chang, Acc. Chem. Res., 2015, 48, 1040 CrossRef CAS; (i) D.-G. Yu, M. Suri and F. Glorius, J. Am. Chem. Soc., 2013, 135, 8802 CrossRef CAS.
- (a) H.-D. Xu, Z.-H. Jia, K. Xu, M. Han, S.-N. Jiang, J. Cao, J.-C. Wang and M.-H. Shen, Angew. Chem., Int. Ed., 2014, 53, 9284 CrossRef CAS PubMed; (b) M.-H. Shen, K. Xu, C.-H. Sun and H.-D. Xu, Org. Lett., 2015, 17, 3654 CrossRef CAS PubMed.
- (a) P. Brandt, M. J. Södergren, P. G. Andersson and P.-O. Norrby, J. Am. Chem. Soc., 2000, 122, 8013 CrossRef CAS; (b) M. M. Díaz-Requejo, P. J. Pérez, M. Brookhart and J. L. Templeton, Organometallics, 1997, 16, 4399 CrossRef; (c) D. A. Evans, M. M. Faul, M. T. Bilodeau, B. A. Anderson and D. M. Barnes, J. Am. Chem. Soc., 1993, 115, 5328 CrossRef CAS; (d) Z. Li, K. R. Conser and E. N. Jacobsen, J. Am. Chem. Soc., 1993, 115, 5326 CrossRef CAS; (e) L. D. Amisial, X. Dai, R. A. Kinney, A. Krishnaswamy and T. H. Warren, Inorg. Chem., 2004, 43, 6537 CrossRef CAS PubMed.
- (a) S. D. McCann and S. S. Stahl, Acc. Chem. Res., 2015, 48, 1756 CrossRef CAS PubMed; (b) A. Casitas and X. Ribas, Chem. Sci., 2013, 4, 2301 RSC.
- H.-S. Dang and B. P. Roberts, J. Chem. Soc., Perkin Trans. 1, 1996, 1493 RSC.
Footnotes |
| † Electronic supplementary information (ESI) available: Experimental procedures, characterization of compounds and copies of spectra. See DOI: 10.1039/c5ra20729k |
| ‡ Sulfonyl azides are known explosive compounds and therefore special precautions are paid for their preparation and purification. In some reactions, significant amount of both TsN3 and TsNH2 were observed after 8 h heating, suggesting that some sulfonyl azides are safe for laboratory use at least in small scale. |
| This journal is © The Royal Society of Chemistry 2015 |