Facile preparation of a SiO2–Al2O3 aerogel using coal gangue as a raw material via an ambient pressure drying method and its application in organic solvent adsorption
Jinmeng
Zhu
a,
Shaohui
Guo
a and
Xuanhua
Li
*ab
aState Key Laboratory of Solidification Processing, Center for Nano Energy Materials, School of Materials Science and Engineering, Northwestern Polytechnical University, Xi’an, 710072, China. E-mail: lixh32@nwpu.edu.cn
bKey Scientific and Technological Team from Shaanxi Province, Mural Research Center, Shaanxi History Museum, China
First published on 23rd November 2015
Abstract
Because of its outstanding properties such as a large specific area and Brønsted acidity, the SiO2–Al2O3 aerogel has been used for various applications such as adsorption, catalysis, and in synthetic chemistry. Regarding preparation of the SiO2–Al2O3 aerogel, however, expensive raw materials are always used. In addition, the traditional preparation method is supercritical drying, which is complex and has safety issues. Here, we successfully prepare a SiO2–Al2O3 aerogel utilizing coal gangue as a raw material, which is a zero-cost mining waste. More importantly, ambient pressure drying used in this work overcomes the drawbacks of supercritical drying. Characterizations show that the specific surface area of the SiO2–Al2O3 aerogel prepared from coal gangue can be comparable to that prepared from relative costly raw materials such as Al(NO3)3·9H2O or Al isopropoxide and tetraethoxysilane with supercritical drying conditions. In addition, adsorption tests show that the SiO2–Al2O3 aerogel demonstrates good adsorbability of an organic solvent. Our work provides an effective route to synthesize a SiO2–Al2O3 aerogel in mass quantities and opens a new direction for the comprehensive utilization of coal gangue.
1. Introduction
After the discovery of the M41S family, mesoporous materials have attracted a large number of investigations because of their outstanding properties.1–5 An aerogel, a typical mesoporous material, is a kind of nanoporous material with a continuous random network structure.6 Because of its excellent properties such as high porosity, high specific surface area and low density, the aerogel has attracted a growing number of studies and has been used in a wide range of applications such as adsorption, catalysis, environmental purification and in biochemistry.7–12 As a unique kind of aerogel, the SiO2–Al2O3 aerogel shows strong Brønsted acidity and is a promising material for absorption and catalysis, which is different from pure aerogels such as silica aerogel and alumina aerogel.13,14 However, the production of SiO2–Al2O3 aerogel has been restricted because of its expensive precursors, such as Al(NO3)3·9H2O and tetraethoxysilane.15,16 Therefore, it is highly desirable to find a kind of cheap material to synthesize the SiO2–Al2O3 aerogel.It is well known that coal gangue is one of the major solid wastes generated in the process of coal mining and washing.17,18 Coal gangue accounts for 10–20% of the original coal production and production is also increasing every year with high speed in China.19,20 Large accumulating stockpiles of coal gangue not only occupy farmland but also contaminate the groundwater, soil, and atmosphere if they are not treated timely and properly.21,22 By far, the comprehensive utilization of coal gangue is mainly focused on its fundamental application, such as for power generation,23,24 building material,25 pavement,26,27 and so on. Here, coal gangue is adopted as a starting material for synthesizing the SiO2–Al2O3 aerogel, which not only allows for the production of the SiO2–Al2O3 aerogel using cheap material, but provides a new direction for the comprehensive utilization of coal gangue.
In addition, the drying method is very important in the synthesis of an aerogel. Usually, three drying methods can be used: supercritical drying, freeze drying and ambient pressure drying.28,29 The supercritical drying method can produce an aerogel with uniform particle distribution, high porosity and minimal shrinkage, however, issues including complicated processing, safety problems, high cost and equipment requirements restrict its application.30 As a facile method, the freeze-drying method can obtain monolithic and high-quality aerogels. However, the gel network may eventually be destroyed by the nucleation and growth of solvent crystals, which tend to produce very large pores.31 Besides, the time-consuming process makes its commercial application unfeasible. Compared to the aforementioned drying methods, ambient pressure drying is a facile, relatively fast and very cheap way to realize the drying of aerogels.32 More importantly, the properties of the aerogel after ambient pressure drying could be comparable to those prepared using supercritical drying.33,34 Herein, comparable properties of SiO2–Al2O3 aerogels are assessed by taking advantage of the ambient pressure drying method.
In the current work, the SiO2–Al2O3 aerogel is successfully synthesized using coal gangue as the starting material via ambient pressure drying. To the best of our knowledge, few studies have reported the synthesis of a SiO2–Al2O3 aerogel using coal gangue via ambient pressure drying. In addition, organic solvent absorption of the SiO2–Al2O3 aerogel prepared from coal gangue has also been studied in detail.
2. Experimental
2.1 Materials
Commercial sodium carbonate (Na2CO3), hydrochloric acid (HCl), anhydrous ethanol (EtOH), cyclohexane, n-hexane and trimethylchlorosilane (TMCS) were purchased from Beijing, China. All of them are of analytical reagent grade and no further purification was carried out. The coal gangue sample used in the experiments was obtained from Shenmu, Xi’an, China. The chemical composition of the air dried basis (Aad) of coal gangue was examined using a chemical method (Table 1).2.2 Methods
The specific preparation procedure of the SiO2–Al2O3 aerogel is shown in Fig. 1(a). Firstly, gangue samples and Na2CO3 were prepared by screening the ground raw materials to a mesh of 100. Then, coal gangue was mixed with Na2CO3 at different weight ratios and calcined at a certain temperature (870–930 °C). Subsequently, the sinter was blended with excess HCl solution (ensures that the pH of the solution would be less than 3, preventing the production of a precipitate) and stirred for 5–10 min at 100 °C. After that, the resulting mixture was filtered. The filtrate was composed entirely of silica–alumina sol, which could be demonstrated by the Tyndall phenomenon (Fig. 1(b)) and the insoluble solid was dried (the weight of the insoluble solid is set as m′). Silica–alumina sol was kept motionless to gelatinize at 80 °C. After gelation, the wet gel was aged for 3 h at 50 °C to strengthen the gel network. The aged gel was then washed with distilled water until the color of the product was transformed from yellow to white (Fig. 1(c) and (d)), which indicated that FeCl3 trapped inside the system was eliminated completely. Next, the SiO2–Al2O3 gel was immersed into EtOH for solvent exchange at 50 °C for 24 h. Surface modification was then carried out by soaking the gel in a mixture of TMCS: hexane with a volume ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 for 24 h. After a complete surface modification, the SiO2–Al2O3 gel was suspended in a liquor and bailed out. The modified SiO2–Al2O3 gel was dried at ambient pressure for 24 h and then heated at 100 °C and 200 °C for 1 h respectively. The final SiO2–Al2O3 aerogel product can be obtained after cooling in an oven to room temperature.
1 for 24 h. After a complete surface modification, the SiO2–Al2O3 gel was suspended in a liquor and bailed out. The modified SiO2–Al2O3 gel was dried at ambient pressure for 24 h and then heated at 100 °C and 200 °C for 1 h respectively. The final SiO2–Al2O3 aerogel product can be obtained after cooling in an oven to room temperature.
For the absorption test of an organic solvent, 0.1 g of SiO2–Al2O3 aerogel was added to a mixture solution of EtOH and cyclohexane (10 ml) with a volume ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. 20 minutes later, the resulting aerogel was fetched and weighed.
1. 20 minutes later, the resulting aerogel was fetched and weighed.
2.3 Calculation and characterization
The decomposition rate of coal gangue (DG) and the organic solvent adsorption rate of the SiO2–Al2O3 aerogel (AR) were calculated using the following equations: | (1) |
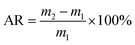 | (2) |
The density of the SiO2–Al2O3 aerogel is determined by eqn (3) as follows:
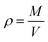 | (3) |
Various experimental tools were used to characterize the as-prepared SiO2–Al2O3 aerogel. Transmission electron microscopy (TEM) measurements were performed on a Tecnai G2 F20 S-TWIN apparatus. Images using field emission scanning electron microscopy (FESEM) were taken on a FESEM (Quanta 600 FEG). X-ray scattering patterns were obtained using an X-ray diffractometer (XRD) with Cu Kα radiation at 0.145 nm. Specific surface area data was collected using an ASAP 2020 HD88 analyzer by nitrogen adsorption and desorption. The hydrophobicity was measured using a hanging drop method on an optical contact angle & interface tension meter (SL200KB). The thermal stability was examined by thermogravimetric differential scanning calorimetry (TG-DSC) thermal analysis under an air atmosphere with a heating speed of 10 °C min−1 from room temperature to 900 °C. The spectrum produced by Fourier transform infrared spectroscopy (FTIR) was recorded on a Bruker Tensor 27 spectrometer with a wavenumber range of 4000–400 cm−1 at a resolution of 4 cm−1 by averaging over 100 scans.
3. Results and discussions
3.1 Effects of different factors on DG
To obtain a high decomposition rate of coal gangue (DG), three experimental parameters are studied, including the weight ratio of coal gangue to Na2CO3 (m(coal gangue)/m(Na2CO3)), holding time, and calcination temperature. The first factor investigated for affecting the DG is m(coal gangue)/m(Na2CO3) by keeping a holding time of 2 h and a temperature of 900 °C (Fig. 2(a)). Reactions that happened at a high temperature are as follows:| 3Al2O3·2SiO2 + 3Na2CO3 = 2NaAlSiO4 + 4NaAlO2 + 3CO2 | (4) |
| SiO2(liq) + NaAlO2 = NaAlSiO4 | (5) |
| Al2O3(liq) + Na2CO3 = 2NaAlO2 + CO2 | (6) |
| Al2O3(liq) + 2SiO2(liq) + Na2CO3 = 2NaAlSiO4 + CO2 | (7) |
These reactions testify that Na2CO3 mainly reacts with Al2O3 and SiO2 inside coal gangue. The main product is NaAlSiO4 that is easily dissolved by the HCl solution, which causes the decomposition of coal gangue. Fig. 2(a) demonstrates that the DG increases first and then reduces along with the decrease of m(coal gangue)/m(Na2CO3). When m(coal gangue)/m(Na2CO3) is 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.6, the DG reaches its maximum (72.5%). Therefore, with regards to the weight ratio of coal gangue to Na2CO3, 1
0.6, the DG reaches its maximum (72.5%). Therefore, with regards to the weight ratio of coal gangue to Na2CO3, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.6 is optimal to obtain a high DG.
0.6 is optimal to obtain a high DG.
In addition, the experimental parameter, holding time, was studied. As can be seen from Fig. 2(b), the DG increases along with an increase of the holding time when the temperature is set to 900 °C and m(coal gangue)/m(Na2CO3) is set to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.6. When the holding time is 3 h, the DG is 85% and only increases by 0.41% at the sacrifice of extending the holding time by 1 h. It can be seen from Table 1 that Al2O3 plus SiO2 account for 87.35% of the Aad of coal gangue, so the DG will not increase significantly even if the holding time is further prolonged. That is to say, almost all SiO2 and Al2O3 have been extracted from coal gangue. Therefore, 3 h is the optimal holding time to obtain a high DG.
0.6. When the holding time is 3 h, the DG is 85% and only increases by 0.41% at the sacrifice of extending the holding time by 1 h. It can be seen from Table 1 that Al2O3 plus SiO2 account for 87.35% of the Aad of coal gangue, so the DG will not increase significantly even if the holding time is further prolonged. That is to say, almost all SiO2 and Al2O3 have been extracted from coal gangue. Therefore, 3 h is the optimal holding time to obtain a high DG.
The third experimental parameter is the calcination temperature. With a holding time of 3 h and m(coal gangue)/m(Na2CO3) set to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.6, the effect of the calcination temperature on the DG is studied. As shown in Fig. 2(c), the optimal calcination temperature is 900 °C to obtain a higher DG.
0.6, the effect of the calcination temperature on the DG is studied. As shown in Fig. 2(c), the optimal calcination temperature is 900 °C to obtain a higher DG.
XRD analysis is carried out to identify the crystal structure of insoluble impurities from the reaction producing the calcination mixture and the HCl solution. As illustrated in Fig. 2(d), the XRD pattern is a precise match for NaAlSi2O6 (standard pdf card 46-0012). That is to say, at high temperature, the reaction of coal gangue with Na2CO3 produces NaAlSi2O6 (eqn (8)). Unlike NaAlSiO4 (eqn (4), (5) and (7)), NaAlSi2O6 cannot react with an acid. Thus, this causes the decline of the DG.
| Al2O3(liq) + 4SiO2(liq) + Na2CO3 = 2NaAlSi2O6 + CO2 | (8) |
In summary, the optimal parameters to maximize the DG are as follows: m(coal gangue)/m(Na2CO3) set to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.6, a holding time of 3 h, and a calcination temperature of 900 °C.
0.6, a holding time of 3 h, and a calcination temperature of 900 °C.
3.2 Characteristics of the SiO2–Al2O3 aerogel
The final product is synthesized using an ambient pressure drying method under the optimal parameters. Firstly, to study the crystal structure of the final product, the XRD pattern is used. According to the XRD pattern (Fig. 3(a)), a strong diffuse peak around 25° of 2θ is due to amorphous SiO2. As observed between 30° and 80° of 2θ in Fig. 3(a), the XRD pattern is a precise match with Al2O3 (standard pdf card 75-0278). Therefore, the XRD result demonstrates that the as-prepared product is the SiO2–Al2O3 aerogel. Furthermore, the morphology of the SiO2–Al2O3 aerogel has been studied. Fig. 3(b) is the SEM image of the final product. As can be seen from the image, the sample mainly consists of nanoparticles. Particles of the sample are uniformly distributed and the size is approximately less than 50 nm. Importantly, the inter-particle connection forms clusters, and then the clusters are linked together with each other. As a result, a large quantity of pores are formed because of the continuous inter-particle connection, as shown in the TEM image in Fig. 3(c). It is noted that the ambient pressure drying method is adopted in the preparation of the SiO2–Al2O3 aerogel. Ambient pressure drying is a facile, relatively fast and very cheap way to realize the drying of an aerogel. Based on Fig. 3(b) and (c), the pore size and particle distribution of the as-prepared SiO2–Al2O3 aerogel using coal gangue as the raw material via ambient pressure drying is comparable to that of a SiO2–Al2O3 aerogel prepared using the supercritical drying method (Fig. 3(d)), which is complex and high-cost.35 | ||
| Fig. 3 (a) XRD pattern of the SiO2–Al2O3 aerogel, (b) SEM image of the SiO2–Al2O3 aerogel, and (c) TEM image of the SiO2–Al2O3 aerogel. (d) SEM image of a SiO2–Al2O3 aerogel prepared using a supercritical drying method.35 All of the scale bars indicate 1 µm. Reproduced with permission from ref. 35. Copyright 2005 Elsevier Inc. | ||
3.3 Properties of the SiO2–Al2O3 aerogel
Considering the fact that the hydrophilic–hydrophobic property plays an important role in organic solvent absorption, this property is studied first. As shown in Fig. 4(a), the contact angle is 134°, which demonstrates that the as-prepared SiO2–Al2O3 aerogel shows strong hydrophobicity. Thus, the SiO2–Al2O3 aerogel has a potential application in organic solvent adsorption. To further clarify the hydrophobicity, the surface functional groups of the SiO2–Al2O3 aerogel are studied using FTIR, as shown in Fig. 4(a). A peak around 3480 cm−1 is attributed to the antisymmetric stretching vibration of –OH, which is caused by the physical absorption of H2O in air. Peaks at 2960 cm−1 and 1260 cm−1 are due to the stretching and bending vibration of C–H respectively, which demonstrate that the SiO2–Al2O3 aerogel surface is connected with –CH3.36 The hydrophobicity is caused by the –CH3 group connected with the SiO2–Al2O3 aerogel. Because of the rotational vibration of H–O–H, an absorption peak at 1640 cm−1 appears.37 On the other hand, other peaks are also observed. Peaks around 1095 cm−1 and 460 cm−1 are ascribed to Si–O–Si bonds. A low intensity absorption peak of Si–OH near 960 cm−1 shows that the SiO2–Al2O3 aerogel is relatively uniform.38 More importantly, the peak at 846 cm−1 is due to the vibration of the Al–O bond, which proves that the Si–O–Al bond exists in the sample as well as the successful preparation of the SiO2–Al2O3 aerogel using coal gangue.To study the thermal stability of the SiO2–Al2O3 aerogel, TG-DSC is performed. Fig. 4(b) is the TG-DSC curve of the SiO2–Al2O3 aerogel with a heating rate of 10 °C min−1 under an air atmosphere. From 0 °C to 431 °C, the mass loss is approximately 4%, which is primarily ascribed to the removal of a small amount of physically absorbed water trapped in the sample. Obviously, there is a rapid weight loss (about 4.5%) at 431 °C, which is attributed to the oxidation of –CH3 connected to the surface of the SiO2–Al2O3 aerogel. With the increase of temperature, the weight of the sample continues to decrease. The mass loss from 431 °C to 900 °C results from a crystallization change of SiO2. As a result, the SiO2–Al2O3 aerogel prepared from coal gangue can be maintained up to 431 °C in air, which implies that the coal gangue based SiO2–Al2O3 aerogel possesses high thermal stability.
The specific surface area also plays a key role in organic solvent absorption. Therefore, the characterization of the specific surface area is performed. N2 adsorption–desorption isotherms and the Barrett–Joyner–Halenda (BJH) pore size distribution curve are shown in Fig. 4(c). The isotherm is type IV and the hysteresis loop belongs to H1, which agree well with the characteristics of mesoporous materials.39,40 The SiO2–Al2O3 aerogel has a specific surface area as large as 493.5 m2 g−1 with a pore volume over 0.59 cm3 g−1 and the average pore diameter is 4.8 nm. In addition, the calculation result of eqn (3) shows that the density of the SiO2–Al2O3 aerogel is 0.3475 g cm−3. The low density of the SiO2–Al2O3 aerogel agrees well with a large specific surface area. As shown in Fig. 4(d), two pore size distribution ranges are observed: 2–10 nm and 10–50 nm. The former has the overwhelming majority of the distribution compared to the latter, explaining the small average pore diameter. On one hand, a large number of inter-particle links forming small pores causes the 2–5 nm pore size distribution. On the other hand, connections of clusters composed of nanoparticles are inclined to form large pores, which explain the 10–50 nm pore size distribution. The above results agree well with the characteristics of the TEM image. Therefore, the SiO2–Al2O3 aerogel prepared from coal gangue has a large specific surface area, small pore size and low density. The specific surface area also can be comparable to SiO2–Al2O3 aerogels prepared under supercritical drying conditions. For instance, using supercritical drying conditions, C. Hernandez et al. obtained SiO2–Al2O3 aerogels with a specific surface area range of 253–729 m2 g−1.41
3.4 Application in the absorption of an organic solvent
The application of a SiO2–Al2O3 aerogel for the treatment of organic solvents is a significant aspect. Its absorption rate of an organic solvent (mixture of EtOH and cyclohexane) was examined following exposure to atmosphere for 0, 10, 20, and 30 days. As can be seen from Fig. 5, the freshly produced SiO2–Al2O3 aerogel can absorb an organic solvent three times its own weight, which is in accordance with the BET data. With the extension of time, the adsorption capacity will slightly decline. However, though exposed to air for 30 days, the absorption rate of the SiO2–Al2O3 aerogel for an organic solvent only decreases by 40%. Hence, the SiO2–Al2O3 aerogel prepared from coal gangue not only exhibits good adsorbability, but can maintain high stability in air, which also agrees with the TG-DSC result.4. Conclusions
In summary, we synthesized a SiO2–Al2O3 aerogel using coal gangue as the raw material via ambient pressure drying. The optimal parameters to obtain a maximum DG are: m(coal gangue)/m(Na2CO3) set to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.6, a holding time of 3 h, and a calcination temperature of 900 °C. Under these conditions, almost all SiO2 and Al2O3 can be extracted from coal gangue (the DG reaches up to 85%). Residues from the reaction of the HCl solution and calcination mixture are detected by XRD. Results show that the residue is NaAlSi2O6, which cannot react with an acid and should be responsible for the decrease of the DG. More importantly, the final product has strong hydrophobicity (contact angle with water is 134°), high thermal stability that cannot be oxidized before 431 °C in air, a large specific surface area (493.5 m2 g−1), a small pore size (average pore diameter 4.8 nm) and low density (0.3475 g cm−3). In addition, adsorption tests show that the SiO2–Al2O3 aerogel demonstrates good adsorbability of an organic solvent. Our work can provide an effective route to synthesize SiO2–Al2O3 aerogels in mass quantities and open a new direction for the comprehensive utilization of coal gangue.
0.6, a holding time of 3 h, and a calcination temperature of 900 °C. Under these conditions, almost all SiO2 and Al2O3 can be extracted from coal gangue (the DG reaches up to 85%). Residues from the reaction of the HCl solution and calcination mixture are detected by XRD. Results show that the residue is NaAlSi2O6, which cannot react with an acid and should be responsible for the decrease of the DG. More importantly, the final product has strong hydrophobicity (contact angle with water is 134°), high thermal stability that cannot be oxidized before 431 °C in air, a large specific surface area (493.5 m2 g−1), a small pore size (average pore diameter 4.8 nm) and low density (0.3475 g cm−3). In addition, adsorption tests show that the SiO2–Al2O3 aerogel demonstrates good adsorbability of an organic solvent. Our work can provide an effective route to synthesize SiO2–Al2O3 aerogels in mass quantities and open a new direction for the comprehensive utilization of coal gangue.
Acknowledgements
This research is supported by the National Natural Science Foundation of China (No. 51472204, 51571166, and 61505167). We are also thankful for the support of Key Scientific and Technological Team from Shaanxi Province (No. 2015KCT-12) and start-up funds from Northwestern Polytechnical University.References
- K. Moller and T. Bein, Chem. Soc. Rev., 2013, 42, 3689–3707 RSC.
- Y. Li, W. Zhang, L. Zhang, Q. Yang, Z. Wei, Z. Feng and C. Li, J. Phys. Chem. B, 2004, 108, 9739–9744 CrossRef CAS.
- J. S. Beck, J. C. Vartuli, W. J. Roth, M. E. Leonowicz, C. T. Kresge, K. D. Schmitt, C. T. W. Chu, D. H. Olson and E. W. Sheppard, J. Am. Chem. Soc., 1992, 114, 10834–10838 CrossRef CAS.
- K. Na, C. Jo, J. Kim, K. Cho, J. Jung, Y. Seo, R. J. Messinger, B. F. Chmelka and R. Ryoo, Science, 2011, 333, 328–332 CrossRef CAS PubMed.
- X. Li, Y. Zhang, Z. Liu, Q. Liu, B. Li, P. Zhu and K. Dai, RSC Adv., 2014, 4, 62209–62214 CAS.
- L. W. Hrubesh, J. Non-Cryst. Solids, 1998, 225, 335–342 CrossRef CAS.
- H. Liu, W. Sha, A. T. Cooper and M. Fan, Colloids Surf., A, 2009, 347, 38–44 CrossRef CAS.
- S. S. Kistler, Nature, 1931, 127, 741 CrossRef CAS.
- Y. Liu, L. Zhang, X. Yao and C. Xu, Mater. Lett., 2001, 49, 102–107 CrossRef CAS.
- Y. Zhang, X. Zhou, Z. Liu, Q. Liu, G. Zhu, K. Dai, B. Li, B. Sun, Z. Jin and X. Li, RSC Adv., 2015, 5, 21925–21930 RSC.
- Y. Yu, M. Zhu, W. Liang, S. Rhodes and J. Fang, RSC Adv., 2015, 72437–72443 RSC.
- B. Yuana, S. Ding, D. Wang, G. Wang and H. Li, Mater. Lett., 2012, 75, 204–206 CrossRef.
- H. Nishihara, S. R. Mukai, Y. Fujii, T. Tago, T. Masuda and H. Tamon, J. Mater. Chem., 2006, 16, 3231–3236 RSC.
- P. Demontis and G. B. Suffritti, Chem. Rev., 1997, 97, 2845–2878 CrossRef CAS PubMed.
- J. P. Randall, M. A. Meador and S. C. Jana, ACS Appl. Mater. Interfaces, 2011, 3, 613–626 CAS.
- F. Cao, L. Ren and X. Li, RSC Adv., 2015, 5, 18025–18028 RSC.
- Y. Li, Y. Yao, X. Liu, H. Sun and W. Ni, Fuel, 2013, 109, 527–533 CrossRef CAS.
- R. M. Izatt, S. R. Izatt, R. L. Bruening, N. E. Izatt and B. A. Moyer, Chem. Soc. Rev., 2014, 43, 2451–2475 RSC.
- H. Liu and Z. Liu, Resour., Conserv. Recycl., 2010, 54, 1331–1340 CrossRef.
- F. Meng, J. Yu, A. Tahmasebi and Y. Han, Energy Fuels, 2013, 27, 2923–2932 CrossRef CAS.
- X. Querol, M. Izquierdo, E. Monfort, E. Alvarez, O. Font, T. Moreno, A. Alastuey, X. Zhuang, W. Lu and Y. Wang, Int. J. Coal Geol., 2008, 75, 93–104 CrossRef CAS.
- B. Zhou, M. Shao, M. Wen, Q. Wang and R. Horton, Clean: Soil, Air, Water, 2010, 38, 1031–1038 CrossRef CAS.
- C. Zhou, G. Liu, S. Wu and P. K. Lam, Chemosphere, 2014, 95, 274–280 CrossRef CAS PubMed.
- C. Zhou, G. Liu, Z. Yan, T. Fang and R. Wang, Fuel, 2012, 97, 644–650 CrossRef CAS.
- X. Luo, J. Xu and W. Li, RSC Adv., 2015, 5, 9283–9289 RSC.
- D. Li, X. Song, C. Gong and Z. Pan, Cem. Concr. Res., 2006, 36, 1752–1759 CrossRef CAS.
- N. Zhang, H. Sun, X. Liu and J. Zhang, J. Hazard. Mater., 2009, 167, 927–932 CrossRef CAS PubMed.
- L. Ren, S. Cui, F. Cao and Q. Guo, Angew. Chem., Int. Ed., 2014, 53, 10147–10149 CrossRef CAS PubMed.
- A. C. Pierre and G. M. Pajonk, Chem. Rev., 2002, 102, 4243–4265 CrossRef CAS PubMed.
- J. B. Peri, J. Phys. Chem., 1966, 70, 2937–2945 CrossRef CAS.
- R. Kocklenberg, B. Mathieu, R. P. S. Blachera, J. P. Pirard, R. Sobry and G. V. D. Bossche, J. Non-Cryst. Solids, 1998, 225, 8–13 CrossRef CAS.
- D. M. Smith, D. Stein, J. M. Anderson and W. Ackerman, J. Non-Cryst. Solids, 1995, 186, 104–112 CrossRef CAS.
- Y. D. Tretyakov and O. A. Shlyakhtin, J. Mater. Chem., 1999, 9, 19–24 RSC.
- C. Roger and M. J. Hampden-Smith, J. Mater. Chem., 1992, 2, 1111–1112 RSC.
- Y. Li, Q. Yang, J. Yang and C. Li, Microporous Mesoporous Mater., 2006, 91, 85–91 CrossRef CAS.
- S. Kabiri, D. N. Tran, S. Azari and D. Losic, ACS Appl. Mater. Interfaces, 2015, 7, 11815–11823 CAS.
- Y. Sheng and H. C. Zeng, ACS Appl. Mater. Interfaces, 2015, 7, 13578–13589 CAS.
- P. R. Aravind, P. Mukundan, P. Krishna Pillai and K. G. K. Warrier, Microporous Mesoporous Mater., 2006, 96, 14–20 CrossRef CAS.
- A. Kondo, H. Kajiro, H. Noguchi, L. Carlucci, D. M. Proserpio, G. Ciani, K. Kato, M. Takata, H. Seki, M. Sakamoto, Y. Hattori, F. Okino, K. Maeda, T. Ohba, K. Kaneko and H. Kanoh, J. Am. Chem. Soc., 2011, 133, 10512–10522 CrossRef CAS PubMed.
- Q. Wei, D. Wang, S. Zhang and C. Chen, J. Alloys Compd., 2001, 325, 223–229 CrossRef CAS.
- C. Hernandez and A. C. Pierre, J. Sol-Gel Sci. Technol., 2000, 20, 227–243 CrossRef.
| This journal is © The Royal Society of Chemistry 2015 |




