Thermal-based regulation on biomineralization and biological properties of bioglass nanoparticles decorated PAN-based carbon nanofibers
Dan Chenga,
Zhiwei Rena,
Lijuan Guoa,
Cuihua Zhanga,
Xiaolong Jia*ab,
Qing Cai*ac and
Xiaoping Yangabc
aState Key Laboratory of Organic–Inorganic Composites, College of Materials Science and Engineering, Beijing University of Chemical Technology, Beijing 100029, P. R. China. E-mail: jiaxl@mail.buct.edu.cn; caiqing@mail.buct.edu.cn
bChangzhou Institute of Advanced Materials, Beijing University of Chemical Technology, Jiangsu 213164, P. R. China
cBeijing Laboratory of Biomedical Materials, Beijing University of Chemical Technology, Beijing 100029, P. R. China. Fax: +86-10-6441-2884
First published on 3rd December 2015
Abstract
Composite carbon nanofibers (CNFs) containing bioglass (BG) nanoparticles displayed different morphology and microstructures depending on the sintering temperature (800, 1000 and 1200 °C) when they were produced from an electrospun polyacrylonitrile–BG precursor blend nanofibers. Biomineralization using simulated body fluid (SBF) and biological evaluation using an osteoblast culture were performed to investigate their relationship with sintering temperature. Characterization revealed that the BG nanoparticles on CNF/BG sintered at 1000 °C (CNF/BG-1000) possessed small particle size and uniform size distribution, and the crystallinity of the BG nanoparticles increased as the sintering temperature was increased from 800 to 1200 °C. The dissolution rate of the BG nanoparticles was thus different between the cases, which enhanced the biomineralization and cell proliferation/differentiation to varying degrees. Benefiting from the homogeneous distribution and large specific surface area of the BG nanoparticles on the CNFs, the results demonstrated that CNF/BG-1000 had the strongest ability in promoting apatite deposition, proliferation and osteogenic differentiation of MC3T3-E1 pre-osteoblasts in comparison with CNF/BG sintered at 800 or 1200 °C. The results demonstrate a flexible tool to regulate the physiochemical and biological properties of CNF/BG composites by controlling the sintering temperature, which could find promising applications in skeleton repairing.
1. Introduction
Carbon nanofibers (CNFs) have attracted considerable attention from both fundamental scientific research and their potential biomedical applications due to their good mechanical properties, chemical stability, high aspect ratio and surface properties, allowing for easy functionalization with biocompatible hydrophilic groups.1–4 In particular, CNFs have been increasingly applied in bone repairing since Webster's group reported that the adhesion and proliferation of osteoblasts were selectively enhanced by nanoscaled carbon fibers.5 To achieve sufficient binding between CNFs and bone tissue, combinations of CNFs with other osteocompatible materials are envisioned as very effective approaches. Bioactive glass (BG) is a type of bioceramic material, which has been extensively investigated for applications in bone regeneration. In addition to its excellent bone binding properties, soft connective tissues have also been reported to be able to form a bond with BG, which led the BG substrates to bind firmly with living bone and surrounding tissues.6–10 These characteristics of BG implied that the addition of BG into CNFs could enhance their abilities for bone binding and bone defect repairing in comparison to pure CNFs, as found in our previous studies.11,12 Besides, composite CNFs containing BG components (CNF/BG) displayed a type of “degradation” feature with the dissolution of BG, which would break down the continuous CNFs. In addition, the obtained CNF/BG composites had the advantage of high mechanical properties derived from the CNFs and were able to offset the intrinsic drawback of the poor strength of the BG materials. Promisingly, CNF/BG composites could be used as substrates directly for cell culture or as bioactive reinforcements for other polymeric biomaterials.The mechanism of bioactivity and bone binding ability of BG was attributed to the formation of a mineralite layer (such as hydroxycarbonated apatite (HCA)) on its surface, a layer similar to the mineral part of bone, along with the chemical degradation of BG by releasing sodium, calcium and silicon ions when it was in contact with body fluid.13–15 In the cases of CNF/BG composites, the dissolution–deposition and ion exchange related to BG would be more complicated because of the combination of two completely different materials. It was reported in our previous studies16–19 and in the literature20,21 that the physicochemical properties and surface morphology of electrospun CNF/metal or CNF/metal oxide composites significantly depend on the preparation parameters such as pre-oxidation conditions and sintering conditions. Arstila H. et al. reported that the chemical composition and crystal types of BG varied remarkably with the sintering temperature.22 Ma J. et al. also found that the sintering temperature greatly influenced the structural features of pure BG, which directly affected the biomineralization and bioactivity of BG.23 Similarly, in our previous study,19 the morphology evolution, crystallization behavior and growth kinetics of the BG component in the CNF/BG composites were also found to be influenced by sintering temperature and time. The BG component in CNF/BG composites could be in the form of amorphous, wollastonite polycrystals or pseudowollastonite crystals, and could exist inside the CNFs and/or on the surface of the CNFs in different sizes and shapes. Marta G. C. et al. reported that the effects of BG on biomineralization and bone repairing were predominated by its physicochemical properties and surface morphology.24 Julie E. G. et al. stated that the formation rate of a mineralite layer was faster on BG with a rough surface than that with a smooth surface, and the large specific surface area of BG favored the adhesion and proliferation of osteoblasts.25 With all these approaches, it could be thus deduced that the biomineralization and the bioactivity of CNF/BG composites would be strongly affected by their preparation parameters. Therefore, it is quite necessary to relate key preparation parameters (e.g. sintering temperature) of CNF/BG composites to their biological features, which would be very helpful in regulating the performance of CNF-based substrates in biomedical applications.
To this end, 58S type BG (mol%: 58.0% SiO2–26.3% CaO–15.7% P2O5) was embedded into CNFs using electrospinning and carbonization techniques similar to our previous studies,11,12,16,17,19 which has been identified as an efficient way to produce CNF/BG composites in the laboratory. Briefly, a polyacrylonitrile (PAN)/N,N-dimethyl formamide (DMF) solution with the addition of the BG precursor sol–gel solution was electrospun and sintered at different temperatures to render CNFs with further flexibility in property regulation. The as-electrospun composite nanofibers were sintered at 800, 1000 or 1200 °C to obtain CNF/BG composites with different morphologies and structures following our previous study.19 Biomineralization in simulated body fluid (SBF) and in an in vitro osteoblast culture on the CNF/BG composites were performed to investigate their dependence on the sintering temperature by evaluating apatite formation, cell proliferation and osteogenic differentiation. A favorable hypothesis of the present study was that the changes in the morphology and structure of the CNF/BG composites could be a useful tool to regulate their biological features.
2. Experimental section
2.1 Materials
PAN (Mw = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1), containing 93.0 wt% acrylonitrile, 5.3 wt% methylacrylate and 1.7 wt% itaconic acid, was purchased from Courtaulds Co. (UK). BG precursors including triethyl phosphate (TEP), calcium nitrate tetrahydrate (CN) and tetraethoxysilane (TEOS) were purchased from Aldrich (USA) and used directly. DMF (99.5%) was bought from Tianjin Fine Chemical Co. (China). Salts for the preparation of SBF and other reagents required for the experiments were of analytically pure grade and obtained from Beijing Fine Chemical Co. (China).
000 g mol−1), containing 93.0 wt% acrylonitrile, 5.3 wt% methylacrylate and 1.7 wt% itaconic acid, was purchased from Courtaulds Co. (UK). BG precursors including triethyl phosphate (TEP), calcium nitrate tetrahydrate (CN) and tetraethoxysilane (TEOS) were purchased from Aldrich (USA) and used directly. DMF (99.5%) was bought from Tianjin Fine Chemical Co. (China). Salts for the preparation of SBF and other reagents required for the experiments were of analytically pure grade and obtained from Beijing Fine Chemical Co. (China).
2.2 Sample preparation
The schematic illustration of whole experimental process is presented in Fig. 1. Referring to our previous studies,11,12,19,26 the preparation of CNF/BG composites is briefly described as follows. First, TEP (10 mL) was dissolved in a mixed solvent containing absolute ethyl alcohol (10.12 mL), distilled water (3.16 mL) and ammonia water (0.12 mL) by stirring at 80 °C for 24 h to obtain a hydrolyzed TEP solution. Then, the hydrolyzed TEP solution (0.155 mL), CN (0.42 g) and TEOS (0.65 mL) were added in turn to 20 mL of DMF, in which, 10 wt% of PAN had been dissolved in advance. The system was stirred continuously at room temperature for 48 h to obtain a homogeneous solution for electrospinning. Electrospinning was conducted with parameters of 15 kV (voltage), 0.3 mL h−1 (flow rate) and receiving distance (15 cm) using a rolling rod as the collector. The as-spun nanofibers were stabilized at 280 °C in air for 0.5 h, then sintered at 800, 1000 or 1200 °C for 3 h in a N2 atmosphere to obtain the CNF/BG composites. For clarity, CNF/BG composites sintered at 800, 1000 and 1200 °C are denoted as CNF/BG-800, CNF/BG-1000 and CNF/BG-1200, respectively. The BG type in the CNF/BG composites was 58S type with a chemical composition of 58% SiO2–33% CaO–9% P2O5 in moles as shown in the aforementioned feeding dose. Pure CNF sheet was prepared in a similar way, except for the addition of the BG precursors and the sintering condition was set at 1000 °C/3 h.2.3 Biomineralization
Biomineralization in vitro is usually applied to evaluate the bone binding ability of substrates for bone repairing. Therefore, CNF/BG composites were soaked in 1.5 times SBF (1.5SBF), which contained nearly 1.5 times the inorganic ion concentrations of human blood plasma. The 1.5SBF was prepared by dissolving reagent grade salts (NaCl, NaHCO3, Na2SO4, KCl, K2HPO4·3H2O, MgCl2·6H2O, and CaCl2·2H2O) in 1 L of distilled water and the final concentrations of the ions were 213.0 mM Na+, 7.5 mM K+, 3.8 mM Ca2+, 2.3 mM Mg2+, 221.7 mM Cl−, 6.3 mM HCO3−, 1.5 mM HPO42− and 0.8 mM SO42−, respectively. The solution pH was adjusted to 7.4 at 37 ± 0.2 °C with the buffer solution (0.05 mol L−1) composed of tris(hydroxymethyl)aminomethane (Tris) and hydrochloric acid. The CNF/BG composite sheets were cut into patches (2 cm × 4 cm) and immersed in the 1.5SBF at 37 ± 0.2 °C for 0.5 day, 1 day, 3 days, 5 days, 7 days and 14 days. At each predetermined time point, the samples were retrieved and gently rinsed with distilled water and then freeze-dried for 24 h.2.4 Biological properties
To evaluate the biocompatibility and cell affinity of the CNF/BG composites, a mouse calvaria-derived osteoblast cell line (MC3T3-E1) was used for cell culture and seeding. The cells were cultured in Dulbecco's modified Eagle's medium (DMEM, Hyclone, USA) supplemented with 10% fetal bovine serum (FBS, PAA, Germany), 100 IU mL−1 penicillin (Sigma, USA) and 100 mg mL−1 streptomycin (Sigma, USA) in an incubator (Sanyo, Japan) with 5% CO2 at 37 °C and saturated humidity. Once grown to a confluence of 80%, MC3T3-E1 cells were digested using 0.25% trypsin (Sigma, USA) and 0.02% ethylene diaminetetraacetic acid (EDTA) for further use.2.5 Characterization
The morphology of both the nanofibers and cells was observed using scanning electron microscopy (SEM, S-250, UK) at an accelerating voltage of 15–20 kV and transmission electron microscopy (TEM, JEOL 2000 EX, Japan) at an accelerating voltage of 200 kV. Before SEM observation, the sample surface was coated with a thin layer of a gold alloy (30 mA, 20 s) using a sputter coater (E5600, Polaron, USA). For TEM examination, the samples were first dispersed in ethanol under ultrasonication and mounted onto formvar carbon coated 100 mesh nickel grids. The average fiber diameters and particle sizes were calculated using the SEM images using ImageJ software (National Institutes of Health, USA) to measure at least 500 nanofibers or nanoparticles. The crystalline microstructures of CNFs and BG nanoparticles were investigated using high-resolution transmission electron microscopy (HR-TEM, Hitachi H-800, Japan) at an operating voltage of 200 kV. The contents of BG component in CNF/BG composites were determined using thermogravimetric analysis (TGA, TA, Q-50) at a heating rate of 10 °C min−1 towards 750 °C in an air atmosphere. The chemical compositions were determined using energy dispersive spectroscopy (EDS, 51-ADD0011, USA) and Fourier transform infra-red spectroscopy (FT-IR, Magna 750R, Nicolet, USA). X-ray diffraction patterns were collected using an X-ray diffractometer with Cu K-radiation (XRD, Rigaku D/max 2500 VB2+/PC) operating at 40 kV and 200 mA.2.6 Statistical analysis
The experiments of biological property evaluation were performed in triplicate (n = 3) and repeated for three times. The results are presented as mean ± SD. Statistical analysis was made by t-test between two groups and the differences were considered as significant for p ≤ 0.05.3. Results and discussion
3.1 Characterization of CNF/BG composites
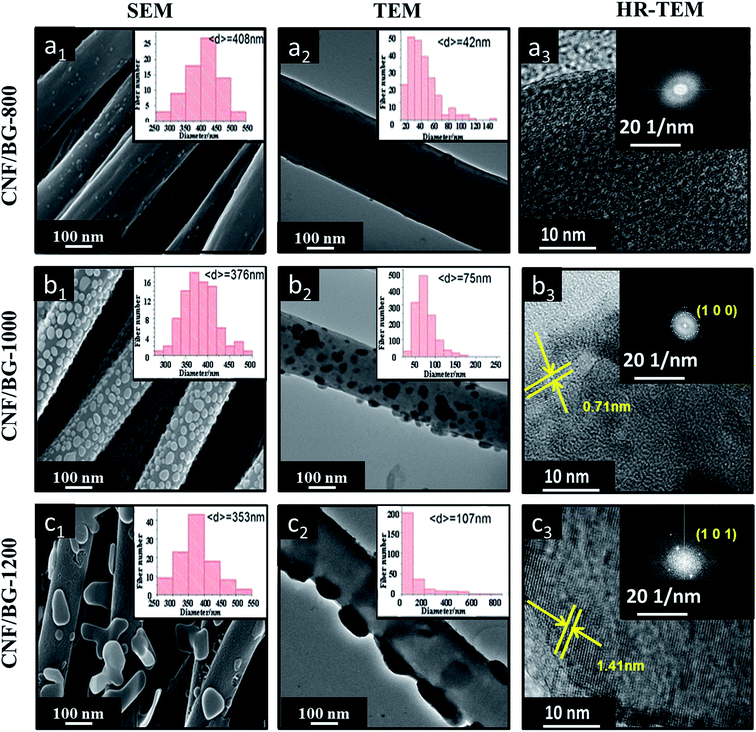
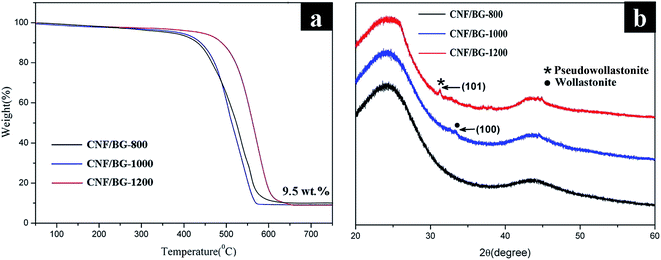
Fig. 2 shows the SEM, TEM and HR-TEM images of CNF/BG composites sintered at various temperatures. Parallel-aligned fibrous matrixes were obtained in all the cases because a rolling rod was used as the receiver in the electrospinning process. It could be seen from the images that visible nanoparticles existed on both outside and inside of the CNFs at the same time, which were the BG nanoparticles according to our previous studies.11,12,19,26 The amounts of the loaded BG nanoparticles in the three CNF composites were all around 9.5 wt%, as measured by TGA (Fig. 3(a)), and were held consistent due to their similar fabrication process. As shown in Fig. 2(a1 and a2), numerous BG nanoparticles with an average size of 42 nm were observed within the fibers and only a few nanoparticles were found on the fiber surface for CNF/BG-800. For CNF/BG-1000, a mass of spherical nanoparticles with an average size of 75 nm covered the fiber surface evenly with a small amount of nanoparticles inside the fiber (Fig. 2(b1 and b2)). A similar phenomenon was observed for CNF/BG-1200, as shown in Fig. 2(c1 and c2), but the nanoparticles had merged into a cuboid-like geometry with an average size of 107 nm and sparse particles could be found inside the fiber. As reported in our previous study,19 the BG nanoparticles in CNF/BG-800 were amorphous, whereas nanoparticles in CNF/BG-1000 were mainly composed of wollastonite (β-CaSiO3) polycrystals and those in CNF/BG-1200 mainly consisted of pseudowollastonite (Ca3(Si3O9)) single crystals. This was characterized and confirmed with HRTEM (Fig. 2(a3, b3 and c3)) and XRD (Fig. 3(b)). In FFT patterns, no diffraction spot or ring was found for CNF/BG-800, while the diffraction spots in the different patterns for CNF/BG-1000 and CNF/BG-1200 indicated that the two materials have crystalline domains but in different crystalline structures. Moreover, the HRTEM images revealed the same results that no clear lattice fringe could be found for CNF/BG-800, while well-defined lattice fringes with an average d-spacing of 0.71 or 1.41 nm were clearly seen in the cases of CNF/BG-1000 and CNF/BG-1200, respectively. These two lattice fringes suggested the formation of the (1 0 0) lattice plane of the wollastonite structure27 and the (1 0 1) lattice plane of the pseudowollastonite structure.28 The XRD results provided more solid evidence for this deduction. As shown in Fig. 3(b), no characteristic sharp peak was found in the XRD pattern of CNF/BG-800, except the broad and weak diffraction peaks at 2θ = 24.7° and 44.5° for the (0 0 2) and (1 0 1) reflections of graphitic carbon. Referring to the matching analysis of JADE software and literature,29,30 the peak at 2θ = 32.8° was assigned to the (1 0 0) reflection of wollastonite (β-CaSiO3) (JCPDS 42-0547) in CNF/BG-1000, and the peak at 2θ = 31° was assigned to the (1 0 1) reflection of pseudowollastonite (Ca3(Si3O9)) (JCPDS 74-0874) in CNF/BG-1200. Apparently, the CNF/BG composites produced at the different sintering temperatures could have different morphological and structural characteristics, which evoked the interest to relate them to biomineralization and biological response targeting for bone repairing applications.3.2 Apatite deposition on CNF/BG composites in SBF
The ability of apatite formation in SBF has been identified useful and reasonable for evaluating the osteocompatibility of biomaterials considering that the mineralized nucleation was heterogeneous instead of homogeneous precipitation.31–33 In reference to our previous studies,12,16,17,26 1.5SBF was selected to investigate the biomineralization because its supersaturation was not high enough to cause homogeneous nucleation and the SBF was able to remain transparent during the experiment. Fig. 4 shows the SEM images obtained for different CNF/BG composites after being soaked in 1.5SBF for 0.5–14 days. Within only 0.5 day, it was found that the surfaces of all the CNF/BG composites became much smoother in comparison with their original ones. The BG nanoparticles on the CNF surface had dissolved as they came in contact with the SBF aqueous solution. However, the dissolution imprint of the BG particles could be tracked as shown in Fig. 4(a1, b1 and c1), in which a spot of mineralites could be seen originating from where the original BG nanoparticles were located. After 1 day of soaking in the 1.5SBF, clusters of mineralite depositions were clearly detected on all the three CNF/BG composites, especially on CNF/BG-1000, which was almost covered completely by the newly-formed mineralites (Fig. 4(a2, b2 and c2)). In our previous studies,11,12 the CNF/BG composites had been confirmed to have a strong ability in enhancing apatite deposition in comparison with pure CNFs by providing active sites with the dissolution of the BG component. In the present case, the difference in the mineralite deposition rate was inferred from the different dissolution rates of BG component in CNF/BG composites because their morphology, particle size, distribution and crystalline structure differed from each other depending on sintering temperature. It appeared that CNF/BG-1000 displayed the fastest dissolution and ion-exchanging rate of BG and provided the most nucleation sites for mineralite deposition among the three CNF/BG composites studied, followed by CNF/BG-1200 and CNF/BG-800. As shown in Fig. 2, BG nanoparticles, being in the form of wollastonite polycrystals, were mainly on the surface of CNFs, which endowed them with a large effective contact area with the SBF solution and led to fast dissolution. The BG nanoparticles in CNF/BG-800 were mainly amorphous, which, theoretically, should dissolve faster than those in CNF/BG-1000. However, they were supposed to diffuse out of CNFs slowly because the BG component was dominantly embedded inside the fibers. However, in the case of CNF/BG-1200, the exposed BG nanoparticles were found in the form of pseudowollastonite single crystals, which would slow down the dissolution rate of the BG nanoparticles due to the higher crystallinity in comparison with CNF/BG-1000. Thus, it was expected to find the biomineralization proceeding on the CNF/BG composites in the order of CNF/BG-1000 > CNF/BG-1200 > CNF/BG-800 at the initial stage.As the biomineralization proceeds, more and more mineralites are continuously deposited onto CNF/BG composites (Fig. 4(a3, b3 and c3)). The original CNFs could be seen fully wrapped by mineralites when they were soaked in 1.5SBF for 7 days. Moreover, until the 14th day of biomineralization, the whole CNF/BG composites had been completely covered with a layer of mineralites (Fig. 4(a4, b4 and c4)). It was revealed that no distinct difference in morphology between the different CNF/BG composites could be observed after soaking for a long time.
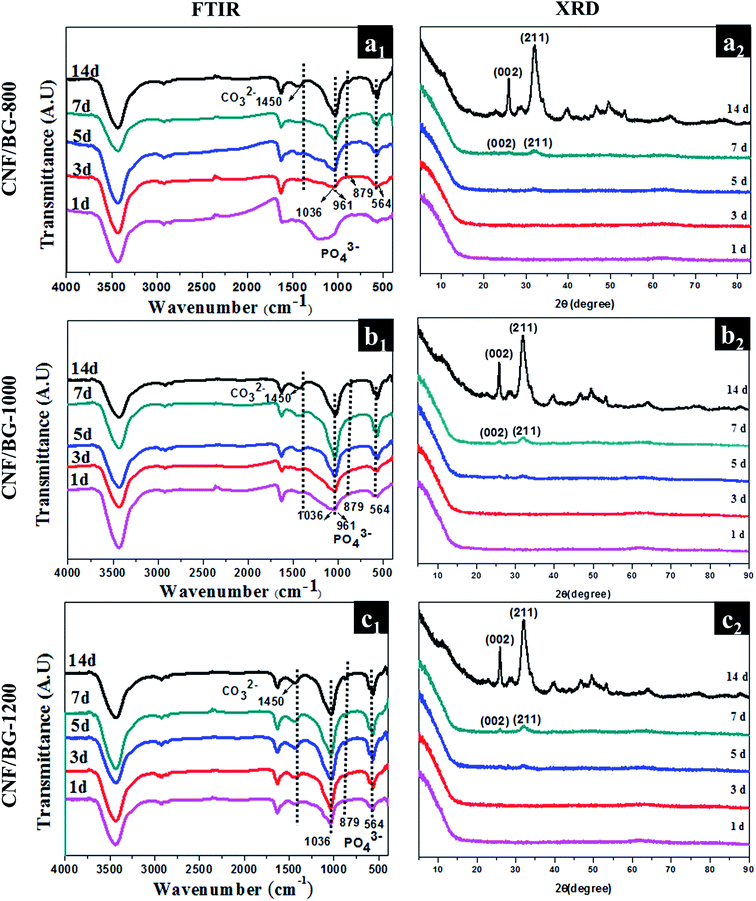
The images shown in Fig. 4 illustrate that the mineralite depositions have a kind of flaky-like structure, which was identified by the typical structure of crystalline hydroxyapatite (HA).12,34 To confirm this inference, FT-IR and XRD analyses of the biomineralized CNF/BG composites were carried out and the results are shown in Fig. 5. From the FT-IR spectra (Fig. 5(a1, b1 and c1)), changes in chemical structures could be identified for all the CNF/BG composites as the soaking time was prolonged. The strengths of the absorption bands at 564, 961, 879 and 1036 cm−1 increased gradually with a longer soaking time and the signals were ascribed to the formation of PO43− or HPO42− groups.35 The peak at 1450 cm−1 was attributed to CO32−,36 which implied that the mineralite depositions were carbonated. Normally, the crystallinity of mineral depositions from SBF was not high due to their fast nucleation and growth rate. As observed from the XRD patterns (Fig. 5(a2, b2 and c2)), two weak diffraction peaks at 2θ = 25° and 32° were detected after the materials had been soaked in the SBF for over 5 days. In addition, the peaks intensified remarkably with a longer soaking time. Inferring from the literature,37–40 the two peaks were assigned to the crystal planes of (0 0 2) and (2 1 1) of HA, and the crystallinity of HA increased with its continuous deposition from the SBF to the CNF/BG composites. Accompanied with the accumulating formation of HA on CNF/BG, changes in the chemical compositions were also detected definitely by EDS. It could be seen from Fig. 6 that the contents of Si decreased, while those of Ca and P increased rapidly upon SBF soaking. After 14 days of incubation, the deposited apatite displayed a Ca/P molar ratio of 1.63, which was close to the characteristic value (1.67) of stoichiometric HA.41
 | ||
| Fig. 6 EDS results obtained for CNF/BG-1000 after being soaked in 1.5SBF at 37 °C for various times. | ||
Taking all the aforementioned results into account, it was clear that the apatite formation ability on CNF/BG composites originated from the dissolution of BG component, whose morphology and crystalline structure could affect its dissolution rate and the apatite deposition thereof. The sintering temperature could be a tool to regulate the location, the size and the crystalline structure of BG nanoparticles on CNFs. From Fig. 4, CNF/BG-1000 demonstrated the strongest ability in inducing apatite nucleation and growth, followed by CNF-1200 and CNF-800. Generally, it is believed that the initial ion-exchanging process is essential for inducing apatite deposition when BG was soaked into the aqueous solutions.38,40 The exchange of cationic ions (e.g. Ca2+ and Na+) with H+ (or H3O+) ions would cause the solution pH to increase, which subsequently led to the hydrolysis of silica groups. Thus, active sites for apatite nucleation were generated, and an amorphous calcium phosphate (ACP) layer on the BG surface was formed during the initial stage by attracting Ca2+ and PO43− ions from surrounding solution.42 From Fig. 2 and 3, it had been known that CNF/BG-1000 displayed a homogeneous distribution of BG nanoparticles densely packed on the fiber surface, and the BG component was a type of wollastonite (β-CaSiO3) polycrystals. BG nanoparticles were exposed to SBF sufficiently and dissolved fast in the SBF, which induced the fast nucleation and growth of apatite on CNF/BG-1000. The BG component in CNF/BG-800 was mainly amorphous, which dissolved faster than the wollastonite-type BG. However, these BG nanoparticles were predominantly embedded inside the CNFs and had less chance of coming in contact with the SBF than in the case of CNF/BG-1000. As for CNF/BG-1200, the BG nanoparticles on the fiber surface had transformed into a pseudowollastonite (Ca3(Si3O9)) single crystalline structure, whose solubility was inferior to wollastonite due to its higher crystallinity. Besides, the size of the BG nanoparticles in CNF/BG-1200 was also larger than that found in CNF/BG-1000; thus, the latter would have a faster BG dissolution rate than the former due to the nanoeffect of particle size.43 It was suggested that BG nanoparticles with smaller particle size had a larger surface area, subsequently facilitating the contact of BG with SBF, which accelerated the dissolution of the BG components. Therefore, in the present study, CNF/BG-1000 was found able of enhancing the fastest apatite formation among the three CNF/BG composites.
3.3 In vitro osteoblast culture on CNF/BG composites
By culturing osteoblasts MC3T3-E1 on the composite CNFs, using pure CNFs as the control, the osteocompatibility of CNF/BG composites was evaluated by cell adhesion, proliferation and osteogenic differentiation assessments, along with cell morphology observations. As shown in Fig. 7(a), cells had adhered onto all the substrates at 2 h after cell seeding, and the cell adhesion slightly increased in all the cases 4 hours after cell seeding. In comparison with the pure CNFs, the cells could be seen more likely adhering onto the CNF/BG composites at the same time point. The cell adhesion on CNF/BG-1000 was found to be the highest among the three CNF/BG composites. As the cell culture proceeded, continuous growth in MC3T3-E1 cells was detected on all the substrates, showing that the materials did not have any obvious cytotoxicity and adverse effects on cell proliferation (Fig. 7(b)). However, the cell proliferation rate differed significantly in different cases; the cells grew the fastest on CNF/BG-1000, followed by CNF/BG-1200, CNF/BG-800 and finally pure CNFs. Cell proliferation was enhanced on the CNF/BG composites in comparison with pure CNFs due to the incorporation of the bioactive BG component. The trend in cell proliferation on the three CNF/BG composites was similar to that in biomineralization, which was also related to the dissolution rate of the BG nanoparticles. Apparently, the released ions had played an effective function in promoting cell proliferation. CNF/BG-1000, with the fastest dissolution rate of BG nanoparticles, displayed the highest cell affinity among the three CNF/BG composites.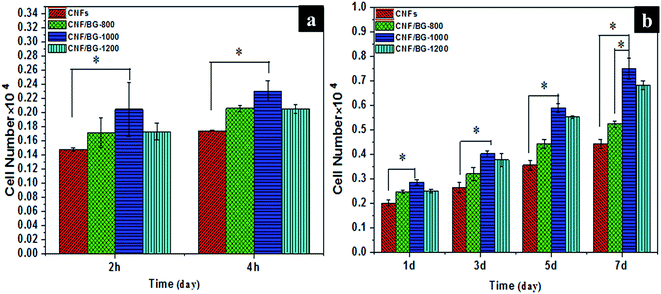 | ||
| Fig. 7 (a) Adhesion and (b) proliferation of osteoblasts (MC3T3-E1) on pure CNFs and CNF/BG for various times. The results are represented as the mean ± standard deviation for n = 3 (P < 0.05). | ||
Morphology of the cultured osteoblasts on pure CNFs and various CNF/BG composites was observed and is shown in Fig. 8. On the first day after cell seeding, MC3T3-E1 cells could be seen having adhered onto all the nanofibrous matrixes and spread. More cells were found on CNF/BG-1000 than the other three materials. When being cultured for 7 d, the proliferation of MC3T3-E1 cells was clearly identified as the images showing many confluent cells on substrates. In all the cases, the cells were in a normal spindle-like morphology, displaying oriented growth along the fiber direction. The cells tended to elongate along the fiber direction because of the contact guide effect, which is a common phenomenon when cells are cultured on parallel-aligned fibrous matrixes.44,45 A rich extracellular matrix was synthesized and secreted to connect adjacent cells together, especially in the case of CNF/BG-1000, which was almost fully covered with confluent cells on the 7th day. The results confirmed that both pure CNFs and CNF/BG composites were biocompatible and affinitive to osteoblasts. The numerous BG nanoparticles on the fiber surface of CNF/BG-1000 might have contributed to its good ability in enhancing cell adhesion by providing active sites for cell grasping.
 | ||
| Fig. 8 SEM images of osteoblasts (MC3T3-E1) cultured on CNFs and CNF/BG (from left to right) for (a1–d1) 1 and (a2–d2) 7 d. | ||
The ability of the CNF/BG composites in enhancing osteogenic differentiation was investigated by measuring an early indicator of osteogenesis, i.e. the ALP activity expression of cultured MC3T3-E1 osteoblasts.46 After the initial incubation period, as shown in Fig. 9, the ALP activity increased with a longer culture time and reached its peak value after 7 days of culture in all the cases. The results demonstrated that all the materials had certain ability in inducing osteogenic differentiation of the MC3T3-E1 osteoblasts. In comparison with TCPs, the ALP expression on pure CNFs was only slightly different from that on TCPs, suggesting that the CNFs themselves did not have a strong effect on enhancing osteogenic differentiation. In contrast, the MC3T3-E1 osteoblasts showed a distinctly high expression in ALP activity on all the BG-containing CNFs composites, especially on CNF/BG-1000 composite because the ions released from the rapidly dissolved BG nanoparticles could act on the cells.47–49 Clearly, the ALP activity on the different substrates was closely related to their chemical compositions and the dissolution rate of bioactive component.
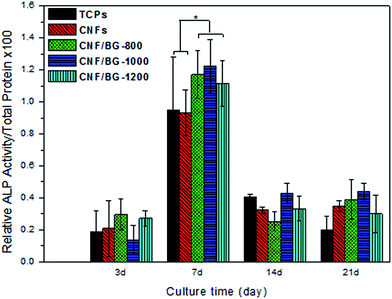 | ||
| Fig. 9 ALP activity expression of MC3T3-E1 cultured on TCPs, pure CNFs and CNF/BG for various times. The results are represented as the mean ± standard deviation for n = 3 (P < 0.05). | ||
Increasing evidence in the literature has indicated that ionic dissolution products from BG are a key factor in osteoblasts metabolism, proliferation and cell–cell and matrix–cell adhesion.13,50 The ionic dissolution products of BG nanoparticles from the CNF/BG composites into the culture medium would provide a favorable environment for MC3T3-E1 cell adhesion, proliferation and osteogenic differentiation. In other words, the incorporation of BG component into CNFs had provided the material a higher affinity to osteoblasts and a higher ability for promoting osteogenic differentiation compared to pure CNFs. The thermal-based flexibility in preparing CNF/BG composites provided an ability to regulate the morphology and structure of the resulting composites, and a type of promising substrate targeting for skeleton tissue regeneration was obtained. It should be noted that to produce this type of CNF/BG composites on a large scale for future applications in bone repairing, prospectively, methods other than electrospinning have been suggested according to some newly-published reports.51–54 Methods such as centrifugal spinning and infusion gyration appear to be more suitable for producing nanofibers with high production than electrospinning, and thus are able to accelerate the practical applications of nanofibrous substrates as biomaterials.
4. Conclusions
CNF/BG composites, which were produced from the electrospinning of a PAN–BG precursor mixed solution, enabled flexibility in regulating their physiochemical and biological properties by selecting different sintering temperatures. Under the appropriate preparation parameters, the resulting CNF/BG composites could demonstrate favorable morphologies and structures for osteoblasts by comprehensively enhancing its ability in inducing cell adhesion, proliferation, osteogenic differentiation and bone binding. The most important issue in achieving this ability was how to accelerate the ion-exchange and dissolution of BG component from CNF/BG composites. Benefiting from the small size of BG nanoparticles and the significant exposure of BG nanoparticles to surrounding medium, in this study, the CNF/BG-1000 composite displayed the strongest ability to enhance the biological behaviors of biomineralization and osteoblasts. Besides, the crystalline structure of the BG nanoparticles obtained under different sintering temperatures might also influence the solubility of BG component in an aqueous medium. Therefore, the CNF/BG composites prepared under the optimum sintering conditions would be good candidates for bone repairing applications. The future application of techniques such as centrifugal spinning or infusion gyration is envisaged to be useful for increasing the production of CNF/BG composites and to promote their practical applications.Acknowledgements
The authors are very pleased to acknowledge financial support from the National Basic Research Program of China (2012CB933904), National Natural Science Foundation of China (No. 51102008 and 51473016), Research Fund for the Doctoral Program of Higher Education (No. 20110010120014), and Fundamental Research Funds for the Central Universities (No. ZY1106).Notes and references
- R. J. Minns, D. S. Muckle and J. E. Donkin, Biomaterials, 1982, 3, 81 CrossRef CAS PubMed.
- T. J. Webster, M. C. Waid, J. L. McKenzie, R. L. Price and J. U. Ejiofor, Nanotechnology, 2004, 15, 48 CrossRef CAS.
- K. L. Elias, R. L. Price and T. J. Webster, Biomaterials, 2002, 23, 3279 CrossRef CAS PubMed.
- C. A. Bonino, L. Ji, Z. Lin, O. Toprakic, X. Zhang and S. A. Khan, ACS Appl. Mater. Interfaces, 2011, 3, 2534 CAS.
- J. L. McKenzie, M. C. Waid, R. Shi and T. J. Webster, Biomaterials, 2004, 25, 1309 CrossRef CAS PubMed.
- Q. Hu, Y. L. Li, G. H. Miao, N. R. Zhao and X. F. Chen, RSC Adv., 2014, 4, 22678 RSC.
- J. G. Tie, T. A. Hannu, Y. K. Heimo and V. Eero, Biomaterials, 2001, 22, 1475 CrossRef.
- J. Luo, Y. Ling, X. Li, B. Yuan, F. Yu, W. H. Xie and X. F. Chen, RSC Adv., 2015, 5, 79239 RSC.
- B. Michel and C. Mario, Biomaterials, 2005, 26, 3873 CrossRef PubMed.
- H. Alexander, J. Bojan, J. Djordje, F. Tobias, G. Peter, R. Stefan, S. Jochen, P. Wolfgang, L. Jonathan, J. Edouard and R. B. Aldo, ACS Appl. Mater. Interfaces, 2014, 6, 2865 Search PubMed.
- B. Han, X. H. Zheng, H. Y. Liu, X. L. Deng, Q. Cai, X. L. Jia, X. P. Yang, Y. Wei and G. Li, J. Biomater. Sci., Polym. Ed., 2014, 25, 341 CrossRef CAS PubMed.
- Q. Yang, G. Sui, Y. Z. Shi, S. Duan, J. Q. Bao, Q. Cai and X. P. Yang, Carbon, 2013, 56, 288 CrossRef CAS.
- A. Hoppe, N. S. Guldal and A. R. Boccaccini, Biomaterials, 2011, 3, 2757 CrossRef PubMed.
- M. N. Rahaman, D. E. Day, B. S. Bal, Q. Fu, S. B. Jung and L. F. Bonewald, Acta Biomater., 2011, 7, 2355 CrossRef CAS PubMed.
- S. Q. Liu, W. Y. Gong, Y. M. Dong, Q. Hu, X. F. Chen and X. J. Gao, RSC Adv., 2015, 5, 38830 RSC.
- H. Y. Liu, Q. Cai, P. F. Lian, Z. Fang, S. Duan, X. P. Yang, X. L. Deng and S. Ryu, Mater. Lett., 2010, 64, 725 CrossRef CAS.
- H. Y. Liu, Q. Cai, P. F. Lian, Z. Fang, S. Duan, S. Ryu, X. P. Yang and X. L. Deng, Carbon, 2010, 48, 2266 CrossRef CAS.
- Y. H. Yu, Q. Yang, D. H. Teng, X. P. Yang and S. Ryu, Electrochem. Commun., 2010, 12, 1187 CrossRef CAS.
- X. L. Jia, T. H. Tang, D. Cheng, L. J. Guo, C. H. Zhang, Q. Cai and X. P. Yang, RSC Adv., 2014, 4, 64299 RSC.
- L. Wang, Y. H. Yu, P. C. Chen, D. W. Zhang and C. H. Chen, J. Power Sources, 2008, 183, 717 CrossRef CAS.
- M. A. Atwater, J. Phillips and Z. C. Leseman, Carbon, 2010, 48, 1932 CrossRef CAS.
- H. Arstila, L. Hupa, K. H. Karlsson and M. Hupa, J. Non-Cryst. Solids, 2008, 35, 722 CrossRef.
- J. Ma, C. Z. Chen, D. G. Wang, X. G. Meng and J. Z. Shi, Ceram. Int., 2010, 36, 1911 CrossRef CAS.
- G. C. Marta, G. David and P. Kevin, Biomaterials, 2005, 26, 4903 CrossRef PubMed.
- E. G. Julie, R. J. Julian and L. H. Larry, Biomaterials, 2004, 25, 2039 CrossRef.
- C. H. Zhang, D. Cheng, T. H. Tang, X. L. Jia, Q. Cai and X. P. Yang, J. Mater. Chem. B, 2015, 3, 5300 RSC.
- Y. Ohashi, Phys. Chem. Miner., 1984, 10, 217 CrossRef CAS.
- P. N. de Aza, Z. B. Luklinska, M. R. Anseau, F. Guitian and S. de Aza, Biomaterials, 2003, 8, 1437 CrossRef.
- P. N. de Aza, F. Guitian, S. de Aza and J. Valle, Analyst, 1998, 123, 681 RSC.
- H. X. Yang and C. T. Prewitt, Am. Mineral., 1999, 84, 929 CAS.
- T. Kokubo and H. Takadama, Biomaterials, 2006, 27, 2907 CrossRef CAS PubMed.
- M. Y. Wu, Q. Y. Wang, X. Q. Liu and H. Q. Liu, Carbon, 2013, 51, 335 CrossRef CAS.
- L. Zhang, W. W. Liu, C. G. Yue, T. H. Zhang, P. Li, Z. W. Xing and Y. A. Chen, Carbon, 2013, 61, 105 CrossRef CAS.
- B. Marc and L. Jacques, Biomaterials, 2009, 30, 2175 CrossRef PubMed.
- R. Govindan, G. Suresh Kumar and E. K. Girij, RSC Adv., 2015, 5, 60188 RSC.
- M. P. Marlin, M. Sylvain, M. C. Jean, P. Elodie and C. Jérôme, J. Eur. Ceram. Soc., 2012, 32, 2765 CrossRef.
- N. Gupta, D. Santhiya, A. Aditya and K. Badra, RSC Adv., 2015, 5, 56794 RSC.
- P. X. Zhu, Y. Masuda and K. Koumoto, Biomaterials, 2004, 25, 3915 CrossRef CAS PubMed.
- A. Bedilu, A. S. Allo and K. M. Rizkalla, ACS Appl. Mater. Interfaces, 2012, 4, 3148 Search PubMed.
- N. Viswanadham, S. Debnath, P. Sreenivasulu, D. Nandan, S. K. Saxenaac and A. H. Al-Muhtase, RSC Adv., 2015, 5, 67380 RSC.
- T. A. Ostomel, Q. H. Shi, C. K. Tsung, H. J. Liang and G. D. Stucky, Small, 2006, 2, 1261 CrossRef CAS PubMed.
- Z. F. Lin, S. Q. Cao, X. Y. Chen, W. Wu and J. S. Li, Biomacromolecules, 2013, 14, 2206 CrossRef CAS PubMed.
- J. Y. Xin, T. C. Chen, Z. F. Lin, P. Dong, H. Tan and J. S. Li, Chem. Commun., 2014, 50, 6491 RSC.
- B. Wang, Q. Cai, S. Zhang, X. P. Yang and X. L. Deng, J. Mech. Behav. Biomed. Mater., 2011, 4, 600 CrossRef CAS PubMed.
- A. Mathur, S. W. Moore, M. P. Sheetz and J. Hone, Acta Biomater., 2012, 8, 2595 CrossRef PubMed.
- T. Olga, L. Sheyda, M. S. Molly, E. P. Alexandra and R. J. Julian, Adv. Healthcare Mater., 2014, 3, 115 CrossRef PubMed.
- E. Tsiridis, A. Bhalla, Z. Ali, N. Gurav, M. Heliotis, S. Deb and L. DiSilvio, Injury-International Journal of the Care of the Injured, 2006, 37, 25 CrossRef PubMed.
- H. A. Declercq, R. M. H. Verbeeck, L. I. F. J. M. de Ridder, E. H. Schacht and M. J. Cornelissen, Biomaterials, 2005, 26, 4964 CrossRef CAS PubMed.
- H. J. Prins, A. K. Braat, D. Gawlitta, W. J. A. Dhert, D. A. Egan, E. Tijssen-Slump, H. P. Yuan, P. J. Coffer, H. Rozemuller and A. C. Martens, Stem Cell Res., 2014, 12, 428 CrossRef CAS PubMed.
- A. C. M. Renno, M. R. Nejadnik, F. C. J. van de Watering, M. C. Crovace, E. D. Zanotto, J. P. M. Hoefnagels, J. G. C. Wolke, J. A. Jansen and J. J. J. P. van den Beucken, Biomed. Mater., 2013, 8, 2365 CrossRef PubMed.
- X. W. Zhang and Y. Lu, Polym. Rev., 2014, 54, 677 CrossRef CAS.
- S. Mahalingam and M. Edirisinghe, Macromol. Rapid Commun., 2013, 34, 1134 CrossRef CAS PubMed.
- M. Khamforoush and T. Asgari, Nano, 2015, 10, 1550016 CrossRef.
- S. Q. Zhang, B. T. Karaca, S. K. VanOosten, E. Yuca, S. Mahalingam, M. Edirisinghe and C. Tamerler, Macromol. Rapid Commun., 2015, 36, 14 Search PubMed.
| This journal is © The Royal Society of Chemistry 2016 |