Formulation characteristics of triacylglycerol oil-in-water emulsions loaded with ergocalciferol using microchannel emulsification
Nauman Khalidabc,
Isao Kobayashi*a,
Zheng Wangac,
Marcos A. Nevesac,
Kunihiko Uemuraa,
Mitsutoshi Nakajimaac and
Hiroshi Nabetaniab
aFood Engineering Division, National Food Research Institute, NARO, 2-1-12 Kannondai, Tsukuba, Ibaraki 305-8642, Japan. E-mail: isaok@affrc.go.jp; Fax: +81-29-838-8122; Tel: +81-29-838-8025
bGraduate School of Agricultural and Life Sciences, The University of Tokyo, 1-1-1 Yayoi, Bunkyo-ku, Tokyo 113-8657, Japan
cFaculty of Life and Environmental Sciences, University of Tsukuba, 1-1-1 Tennoudai, Tsukuba, Ibaraki 305-8572, Japan
First published on 12th November 2015
Abstract
Ergocalciferol is one important form of vitamin D that is needed for proper functioning of the human metabolic system. The study formulates monodisperse food grade ergocalciferol loaded oil-in-water (O/W) emulsions by microchannel emulsification (MCE). The primary characterization was performed with grooved MCE, while the storage stability and encapsulating efficiency (EE) were investigated with straight-through MCE. The grooved microchannel (MC) array plate has 5 × 18 μm MCs, while the asymmetric straight-through MC array plate consists of numerous 10 × 80 μm microslots each connected to a 10 μm diameter circular MC. Ergocalciferol at a concentration of 0.2–1.0% (w/w) was added to various oils and served as the dispersed phase, while the continuous phase constituted either of 1% (w/w) Tween 20, decaglycerol monolaurate (Sunsoft A-12) or β-lactoglobulin. The primary characterization indicated successful emulsification in the presence of 1% (w/w) Tween 20 or Sunsoft A-12. The average droplet diameter increased slowly with the increasing concentration of ergocalciferol and ranged from 28.3 to 30.0 μm with a coefficient of variation below 6.0%. Straight-through MCE was conducted with 0.5% (w/w) ergocalciferol in soybean oil together with 1% (w/w) Tween 20 in Milli-Q water as the optimum dispersed and continuous phases. Monodisperse O/W emulsions with a Sauter mean diameter (d3,2) of 34 μm with a relative span factor of less than 0.2 were successfully obtained from straight-through MCE. The resultant oil droplets were physically stable for 15 days (d) at 4 °C without any significant increase in d3,2. The monodisperse O/W emulsions exhibited an ergocalciferol EE of more than 75% during the storage period.
Introduction
Vitamin D plays a vital role in maintaining and developing a healthy human skeletal system, since it maintains calcium levels in the body. Deficiency of vitamin D results in an increased risk of diabetes, hypertension, cancer and autoimmune diseases.1–4 Broad spectrum deficiencies of vitamin D include rickets in children, osteomalacia in adults and osteoporosis in women; all of these lead to softening and weakening of bones.5–7 Nutritional and cultural factors leading to vitamin D deficiency include insufficient fortified food consumption, sun-block usage, limited body exposure to sun and fear for excessive intake of vitamin D.7,8 Vitamin D is synthesized in the skin and involves the phytochemical conversion of pro-vitamin D by the action of ultraviolet (UV-B) rays. This process takes place if the UV-B rays fall between 290 and 315 nm of the spectrum. These rays are only emitted in the regions that lie below 35° latitude.9,10The terminology and classification related to vitamin D is confusing and can be classified into 5 different forms and metabolites. Among these, vitamin D2 (ergocalciferol) and D3 (cholecalciferol) are important (Fig. 1).11,12 Vitamin D2 is naturally present in some plants and is produced commercially by UV irradiation of yeast, while vitamin D3 is naturally produced in human and animal bodies. Vitamin D2 is substantially used for fortification and supplementation in the food and pharmaceutical industries.12 Several researchers pointed out the rapid metabolism of vitamin D2 in comparison to vitamin D3, while the action becomes bio-equivalent if taken daily.13,14 Both forms of vitamin D are converted to 25-hydroxy vitamin D [25-(OH)D] in the liver. The quantification of 25-(OH)D in blood gives the quantitation of the vitamin D status. A cutoff value of 30 ng mL−1 is sometimes used for an optimal vitamin D status.12 Ergocalciferol was produced in the 1920s through UV-B exposure of foods, leading to the formation of the first medicinal preparation called viosterol.15 Ergocalciferol has limited natural sources and the most significant source is wild mushrooms.16 Ergocalciferol is prone to oxidation and is also isomerized to isotachysterol in the presence of sunlight and mostly under acidic conditions.16,17 Ergocalciferol is mostly supplemented and fortified in fat-based products due to its hydrophobic nature.
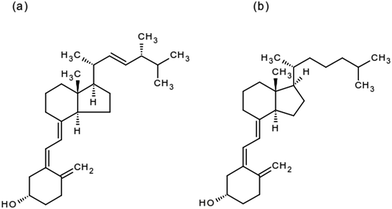 | ||
| Fig. 1 Chemical structures of important forms of vitamin D: (a) ergocalciferol and (b) cholecalciferol. | ||
Emulsification technologies play an important role in the production of encapsulated foods, pharmaceuticals, cosmetics and chemicals.18,19 The emulsions are usually either single (O/W and W/O) emulsions or double emulsions (W/O/W and O/W/O).19 These different emulsion systems are produced by either conventional devices including colloid mills, high pressure homogenizers and rotor-stator homogenizers or modern devices such as microfluidic devices (lab-on-a-chip), membrane emulsification and microchannel emulsification (MCE) devices.19,20 Conventional devices produce polydisperse emulsions with broader size distributions, which in turn reduce the emulsion stability and functionality. Microfabricated emulsification devices have the potential to produce monodisperse emulsions with the smallest coefficient of variation (CV) of less than 10% for membrane emulsification and around 5% for microfluidic devices and MCE.20
MCE is a progressive technique that enables production of monodisperse emulsions by spontaneous transformation of the oil–water interface specifically driven by interfacial tension dominant on a micron scale.21 MCE studies were comprehensively reviewed by Vladisavljević et al.20,22 Similarly, this emulsification technique allows integration of hundreds of thousands of droplet generation units on a single plate.23,24 MCE has been used for producing O/W, W/O and W/O/W emulsions with diameters ranging from 1 μm to 550 μm.22 Based on the microfabrication design, the MCE devices can either be categorized into grooved microchannel (MC) arrays each consisting of uniform microgrooves and a slit-like terrace or straight-through MC arrays each having uniform asymmetric microholes together with microslots.22 Grooved MC array plates are further classified into dead-end and cross-flow types. Grooved MCE plates of the cross-flow type are particularly useful to observe the entire droplet generation process together with droplet collection at low dispersed phase flow rates.25,26 On the other hand, straight-through MC arrays are designed to improve the throughput capacity of MCE. Straight-through MC arrays have the ability to increase the production capacity of monodisperse emulsion droplets over 2000 L m−2 h−1.22
MCE has also been used to produce monodisperse microdispersions (e.g. solid lipid microspheres,26 gel microbeads,27 and giant vesicles28). MCE has promising potential for producing uniformly sized oil droplets containing functional lipids such as β-carotene,29 γ-oryzanol,30 L-ascorbic acid,31 ascorbic acid derivatives,32 oleuropein,33 and vitamin D.34 Different food grade materials (e.g. refined vegetable oils, a medium-chain triglyceride oil, hydrophobic and hydrophilic emulsifiers, proteins, and hydrocolloids) were utilized to produce monodisperse O/W, W/O, and W/O/W emulsions and microparticles by MCE.35,36
We previously encapsulated vitamin D (both vitamin D2 and D3) in O/W emulsions using MCE and reported long-term stability studies and encapsulation efficiencies of O/W emulsions encapsulating vitamin D2 and D3.34 Ergocalciferol is a plant-based vitamin D type, and previous studies seldom report its encapsulation in different formulations. Keeping its rapid metabolism rate and importance in the human diet, the present study was conducted to encapsulate only ergocalciferol in triacylglycerol oil-in-water emulsions using MCE. The basic characterization and optimization of these emulsions were performed using a grooved MC array plate. Straight-through MCE was carried out to investigate the effect of the dispersed phase flux on the production characteristics as well as the physical and chemical stability of the ergocalciferol-loaded O/W emulsions. The effects of different triacylglycerol oils and emulsifiers on the preparation characteristics of O/W emulsions by MCE were also evaluated. The results of this research are expected to help formulate new aqueous-based functional foods.
Experimental
Chemicals
Ergocalciferol, polyoxyethylene (20) sorbitan monolaurate (Tween 20), olive oil, and soybean oil were purchased from Wako Pure Chemical Industries Ltd. (Osaka, Japan). Medium-chain triacylglycerol (MCT, Sunsoft MCT-7) with a fatty acid residue composition of 75% caprylic acid and 25% capric acid and decaglycerol monolaurate (Sunsoft A-12) were procured from Taiyo Kagaku Co. Ltd (Yokkaichi, Japan). Safflower oil was purchased from MP biomedicals (lllkirch, France). β-Lactoglobulin (β-lg) from bovine milk (>90% purity) was purchased from Sigma-Aldrich Co. LLC (St. Louis, USA). All other chemicals used in this study were of analytical grade and used as received.Preparation of solutions
The continuous phase was prepared by dissolving either 1% (w/w) Tween 20, 1% (w/w) Sunsoft A-12 or 1% (w/w) β-lg in Milli-Q water with a resistivity of 18 MΩ cm. The dispersed phase was prepared by dissolving 0.2–1.0% (w/w) ergocalciferol in MCT, soybean, olive or safflower oil at 85 °C ± 3 °C for 20 min and afterwards cooling at room temperature for 2 h before storing at 4 ± 1 °C. During storage, dissolved ergocalciferol molecules could form nuclei whose growth eventually makes crystals large enough to sediment. Before initiating the experiments, the samples were therefore shaken slightly to avoid formation of any ergocalciferol crystals.Silicon plates for MCE
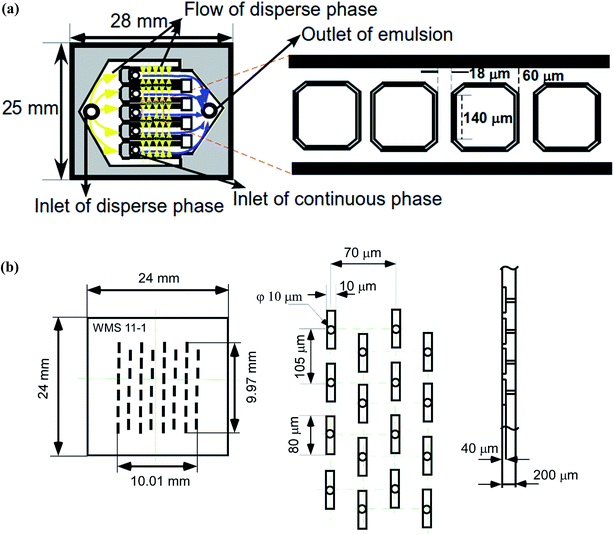
The experiments have been carried out using silicon MC array plates (model CMS 6-2 and WMS 11-1, EP. Tech Co., Ltd., Hitachi, Japan). Fig. 2a is a schematic representation of a CMS 6-2 plate with 540 parallel channels on 10 consecutive MC arrays. Each MC array contains 54 parallel MCs with a depth of 5 μm, a width of 18 μm and a length of 140 μm and a terrace with a depth of 5 μm and a length of 60 μm. Each continuous phase channel outside the terrace outlet has a depth of 100 μm. Fig. 2b is a schematic representation of a WMS 11-1 plate with 27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 MCs compactly arranged within a 10 × 10 mm square region in the plate center. Each MC consists of a cylindrical 10 μm diameter straight microhole with a depth of 200 μm and a 10 × 80 μm microslot with a depth of 40 μm. The slot aspect ratio of 8 was above the threshold value of 3 for successfully generating monodisperse emulsion droplets.37 The distance between the two adjacent MCs vertically was 105 μm, and the distance between the centers of the MCs in adjacent rows was 70 μm. The MC array plates were subjected to plasma oxidation in a plasma reactor (PR41, Yamato Science Co. Ltd., Tokyo, Japan) to activate a silicon dioxide layer on their surfaces. The activated silicon dioxide layer is capable of maintaining their hydrophilicity during MCE.
400 MCs compactly arranged within a 10 × 10 mm square region in the plate center. Each MC consists of a cylindrical 10 μm diameter straight microhole with a depth of 200 μm and a 10 × 80 μm microslot with a depth of 40 μm. The slot aspect ratio of 8 was above the threshold value of 3 for successfully generating monodisperse emulsion droplets.37 The distance between the two adjacent MCs vertically was 105 μm, and the distance between the centers of the MCs in adjacent rows was 70 μm. The MC array plates were subjected to plasma oxidation in a plasma reactor (PR41, Yamato Science Co. Ltd., Tokyo, Japan) to activate a silicon dioxide layer on their surfaces. The activated silicon dioxide layer is capable of maintaining their hydrophilicity during MCE.
Experimental procedure for MCE
For grooved MCE, the setup consists of an MC module, a 10 mL liquid chamber that contains the disperse phase and a syringe pump (Model 11, Harvard Apparatus Inc., Holliston, USA) that feeds the continuous phase with a 50 mL glass syringe (Fig. 3a). MCE was carried out for approximately 3 h and monitored through an inverted metallographic microscope equipped with an objective lens of 2.5× to 20× and a CCD camera (MS-511B, Seiwa Kougaku Sesakusho Ltd., Tokyo, Japan). The whole process was recorded with a video recorder (RDR-HX67, Sony Co., Tokyo, Japan). Droplet generation experiments were performed with the grooved MCE setup depicted in Fig. 3b. The module was initially filled with the continuous phase before mounting the CMS 6-2 plate. The pressurized dispersed phase was introduced into the module. The pressure applied to the dispersed phase (ΔPd) was gradually increased. ΔPd can be given by:| ΔPd = ρdΔhdg | (1) |
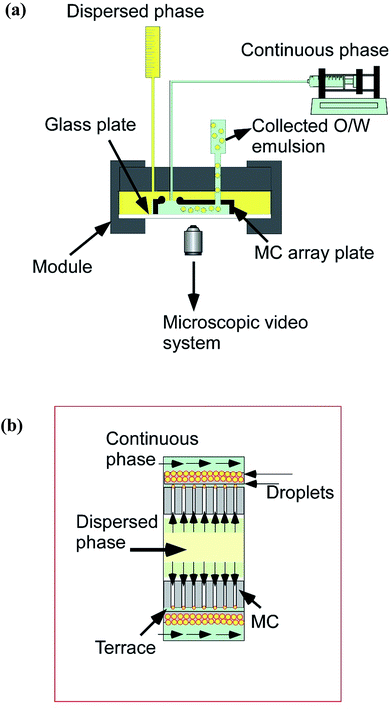 | ||
| Fig. 3 (a) Schematic drawing of a grooved MCE setup. (b) Schematic drawing of droplet generation via part of a MC array having a 5 μm depth. | ||
For straight-through MCE, the MC array plate was degassed in a continuous aqueous phase by an ultrasonic bath for 20 min and the setup consists of a MC module (comprising of six steel parts and two glass plates of different dimensions and rubber seals) and syringe pumps (Model 11, Harvard Apparatus Inc.) that feed the continuous and dispersed phases (Fig. 4a). MCE was carried out for approximately 1 h and monitored through a FASTCAM-1024 PCI high speed video system at 250 to 500 fps (Photron Ltd., Tokyo, Japan) attached to the inverted metallographic microscope. The droplet generation process is depicted in Fig. 4b. Droplet generation started with injecting the dispersed phase through the syringe pump at a dispersed phase flow rate (Qd) ranging from 0.5 to 2.0 mL h−1 (5 to 20 L m−2 h−1 in a dispersed phase flux (Jd)). The generated droplets were removed by varying the continuous phase flow rate (Qc) from 100 to 500 mL h−1 through the gap between the MC array plate and the glass plate. The shear stress (τ) in the module surrounding the WMS 11-1 plate is given by:
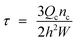 | (2) |
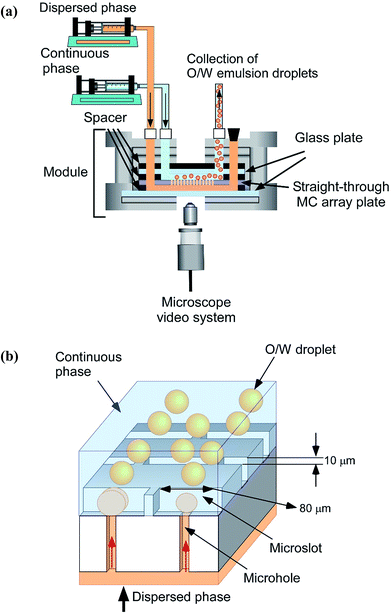 | ||
| Fig. 4 (a) Schematic representation of a straight-through MCE setup. (b) Droplet generation representation through asymmetric MCs. | ||
Determination of droplet size and droplet size distribution
The size and size distribution of the resultant O/W emulsion droplets from the grooved MCE were determined as follows. The average droplet diameter (dav) was defined by:
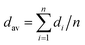 | (3) |
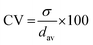 | (4) |
The droplet size distribution of the O/W emulsions obtained from straight-through MCE was measured using a laser scattering instrument that works on the principle of Polarization Intensity Differential Scattering Technology (LS 13 320, Beckman Coulter, Inc., Brea, USA). This instrument has the ability to measure the size ranging from 0.04 to 2000 μm with a resolution of 116 particle size channels. The mean droplet size was expressed as the Sauter mean diameter (d3,2), defined as the diameter of a droplet having the same area per unit volume as that of the total collection of droplets in emulsions. The width of the droplet size distribution was expressed as relative span factor (RSF), defined as:
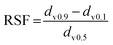 | (5) |
Measurement of fluid properties
The densities of the dispersed and continuous phases were measured using a density meter (DA-130 N, Kyoto Electronics Manufacturing Co., Ltd., Kyoto, Japan) at 25 ± 2 °C. Their viscosities were measured with a vibro viscometer (SV-10, A&D Co., Ltd., Tokyo, Japan) at 25 ± 2 °C by taking either 10 or 35 mL of samples in a measuring vessel followed by immersion of sensor plates in that vessel. Viscosity was measured by detecting the electric current needed to resonate the sensor plates. The static interfacial tension between the preceding two phases was measured with a fully automatic interfacial tensiometer (PD-W, Kyowa Interface Sciences Co., Ltd., Saitama, Japan) using a pendant drop method. The key physical properties of the dispersed and continuous phases used in this study are presented in Table 1.| Dispersed phase | Continuous phase | |||||||
|---|---|---|---|---|---|---|---|---|
| ηd (mPa s) | ρd (kg m−3) | γd (mN m−1) | ζb | Emulsifiers in Milli-Q water | ηc (mPa s) | ρc (kg m−3) | γc (mN m−1) | |
| a Dispersed phase contains 0.5% (w/w) ergocalciferol and interfacial tension was measured in the presence of 1% (w/w) Tween 20 in Milli-Q water.b Viscosity ratio (ζ) was defined as the ratio of dispersed phase viscosity over continuous phase viscosity. | ||||||||
| MCTa | 22.5 ± 0.3 | 946.9 ± 0.2 | 5.3 ± 0.4 | 24.7 | 0.5% Tween 20 | 0.89 ± 0.1 | 997.3 ± 0.6 | 5.1 ± 0.2 |
| Soybean oila | 53.0 ± 0.1 | 921.9 ± 0.4 | 5.6 ± 0.1 | 58.2 | 1.0% Tween 20 | 0.91 ± 0.1 | 998.4 ± 0.6 | 5.2 ± 0.1 |
| Olive oila | 68.2 ± 0.1 | 911.9 ± 0.2 | 6.2 ± 0.2 | 75.0 | 1.5% Tween 20 | 0.96 ± 0.1 | 999.1 ± 0.8 | 5.2 ± 0.2 |
| Safflower oila | 53.2 ± 0.1 | 918.9 ± 0.1 | 5.3 ± 0.2 | 58.5 | 2.0% Tween 20 | 0.99 ± 0.1 | 1000.1 ± 0.1 | 5.4 ± 0.3 |
| 1.0% β-lg | 0.95 ± 0.1 | 999.9 ± 0.2 | 12.6 ± 0.9 | |||||
| 1.0% Sunsoft A-12 | 0.97 ± 0.1 | 998.5 ± 0.6 | 4.8 ± 0.2 | |||||
Physical and chemical stability of O/W emulsions
The physical stability of the O/W emulsion droplets loaded with ergocalciferol was evaluated according to the method described in an earlier section. The d3,2, RSF, consistency and coalescence during 15 d of storage at 4 ± 1 °C under dark conditions were observed.The amount of ergocalciferol encapsulated in the O/W emulsions was determined spectrophotometrically. All spectral measurements of the ethanolic extracts of these O/W emulsions were carried out using a UV/VIS/NIR spectrophotometer (UV-1700, Shimadzu Co., Kyoto, Japan). First, 1 mL of the emulsion was mixed with 9 mL of ethanol, followed by ultrasonication for 20 min. The ethanolic extracts were then centrifuged (Avanti HP-25, Beckman Coulter, Inc.) at 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g for 15 min. A 1 mL aliquot of the subnatants was diluted ten times with ethanol and then injected into a quartz cell with a 10 mm pass length. The absorbance of ergocalciferol in the emulsion extract was measured at 310 nm using an appropriate blank. A representative standard curve of absorbance versus concentration gave the linear least-squares regression with a coefficient of determination (r2) of 0.9996. All experiments were repeated in triplicate and mean values were calculated. Beer’s law was obeyed in the concentration range of 0.1–0.5 mg mL−1 and the sensitivity of measurement has a relative standard deviation of 0.85% (n = 15). The molar absorptivity (ε) for ergocalciferol during this study was 2.52 mM−1 cm−1. The encapsulation efficiency (EE) of ergocalciferol in the samples were calculated with the equation:
000g for 15 min. A 1 mL aliquot of the subnatants was diluted ten times with ethanol and then injected into a quartz cell with a 10 mm pass length. The absorbance of ergocalciferol in the emulsion extract was measured at 310 nm using an appropriate blank. A representative standard curve of absorbance versus concentration gave the linear least-squares regression with a coefficient of determination (r2) of 0.9996. All experiments were repeated in triplicate and mean values were calculated. Beer’s law was obeyed in the concentration range of 0.1–0.5 mg mL−1 and the sensitivity of measurement has a relative standard deviation of 0.85% (n = 15). The molar absorptivity (ε) for ergocalciferol during this study was 2.52 mM−1 cm−1. The encapsulation efficiency (EE) of ergocalciferol in the samples were calculated with the equation:
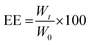 | (6) |
Results and discussion
Basic droplet generation characteristics through grooved MCE
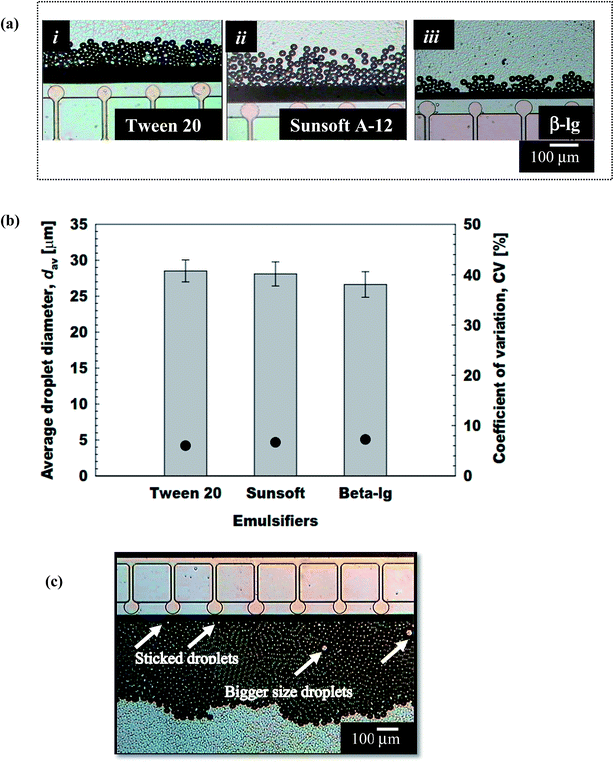
Kobayashi and Nakajima40 investigated the effect of the emulsifier type on the droplet generation characteristics using a straight-through extrusion filter. They reported Tween 20 and decaglyerol monolaurate as suitable, hydrophilic food grade emulsifiers in MCE without ergocalciferol. Patel and San Martin-Gonzalez17 also demonstrated the successful preparation of solid lipid nanoparticles loaded with ergocalciferol stabilized by Tween 20. The preceding results demonstrate that Tween 20 and Sunsoft A-12 are potential emulsifiers for generating ergocalciferol-loaded O/W emulsions, either with conventional homogenization techniques (data not shown) or MCE.
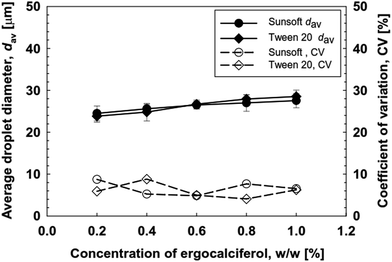
At the lowest Qd of 2 × 10−3 mL h−1, the resultant droplets with a monomodal and very narrow size distribution had a dav of 23.5 μm and a CV of 5.4%. When Qd was increased stepwise, monodisperse emulsions with a CV of 4 to 10% were produced at a Qd of 8 × 10−2 mL h−1 or less. In this Qd range, the dav of the resultant droplets ranged from 23.7 μm to 26.7 μm. The microscopic observations during MCE confirmed that the resultant droplet size hardly changed at Qc between 0 mL h−1 and 5.0 mL h−1. The generation of droplets even without external flow of the continuous phase depicts the unique spontaneous transformation of the interface in MCE. In contrast, at a Qd of 8 × 10−2 mL h−1 or more, the dav and CV of the O/W emulsions dramatically increased to >30 μm with CV values of more than 10% (Fig. 8a). Moreover the droplet size distribution became wider and shifted towards a large droplet size area. The CMS 6-2 plate used here enabled the production of O/W emulsions with uniformly sized droplets at a critical Qd of 8 × 10−2 mL h−1, which was higher than a maximum Qd (5 × 10−3 mL h−1) for the previously reported studies from grooved MCE.42
After reaching the critical Qd, some of the dispersed phase that passed through the MCs expanded instead of generating droplets, suggesting that the flow state of the dispersed phase was affected by the dispersed phase velocity inside the MC. Sugiura et al.21 reported that the droplet generation behavior inside the MCs is related to the capillary number of the dispersed phase that flows inside the MCs. The capillary number (Ca), which indicates the balance between the viscous force and interfacial force, can be determined by:
| Ca = ndUd/γ | (7) |
The influence of Qd on the droplet generation frequency per MC array plate (f) (Fig. 8b) can be estimated by:
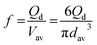 | (8) |
Stability evaluation of ergocalciferol-loaded O/W emulsions prepared by MCE
The stability and EE of the emulsion system are directly dependant on the droplet size and droplet size distribution. The more monodisperse the system is, the better the efficiency of the process parameters. Grooved MCE provides useful information regarding the basic characterization of droplet generation, whereas its drawback lies in low droplet productivity (e.g. a maximum of 1.5 × 10−3 L h−1).25 In comparison, straight-through MCE can increase the throughput capacity of droplets and work even at a Qd of 0.27 L h−1 with uniform droplet productivity.44 Straight-through MC arrays are comprised of narrow microholes and microslots that can accommodate >104 asymmetric MCs per 1 cm2.45 Here we focus on the stability and encapsulation efficiency of soybean oil loaded-ergocalciferol O/W emulsions prepared by straight-through MCE.
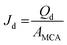 | (9) |
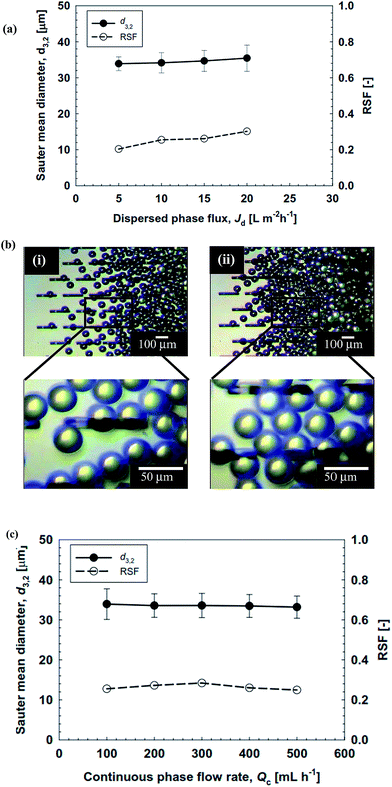
The results presented in Fig. 9 deviate from the previous findings of Vladisavljević et al.44 They reported the size stable zone of soybean oil-in-water emulsions which ranged between 0 and 50 L m−2 h−1. Moreover, they reported a critical Jd of 260 L m−2 h−1 for soybean oil loaded emulsions without loading any bioactive computed from CFD simulations. Our results are somewhat similar to the findings of Neves et al.30 They formulated soybean oil-in-water emulsions loaded with polyunsaturated fatty acid at a critical Jd of 80 L m−2 h−1. It should be noted that the flow rate of the continuous phase hardly affected the d3,2 and RSF of the O/W emulsions encapsulating ergocalciferol (Fig. 9c), which is another advantage for the stable preparation of emulsion droplets.
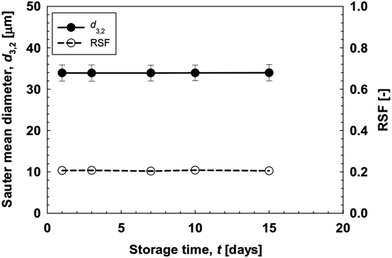
The monodisperse O/W emulsions loaded with ergocalciferol prepared using the WMS 11-1 plate were stored at 4 ± 1 °C for 15 d. MCE was performed by keeping Jd at 5 L m−2 h−1 and a Qc of 150 mL h−1. Immediately after collection, the O/W emulsion samples had a colorless turbid appearance with good flowability. Their appearance did not change with storage time. Fig. 10 depicts time changes in the d3,2 and RSF values of the resultant O/W emulsions loaded with ergocalciferol. There was hardly any increase in their d3,2 and RSF values during the evaluated storage time, indicating a high physical stability of the monodisperse O/W emulsions loaded with ergocalciferol.
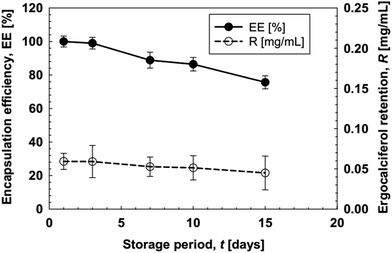 | ||
| Fig. 11 Encapsulating efficiency and retention profile of O/W emulsions with storage time. The emulsions were prepared at a Jd of 5 L m−2 h−1, and the data are presented over 15 days of storage time. | ||
The potential reason for such a decrease is the conversion of ergocalciferol to its isomeric form, isotachysterol. This conversion is accelerated in the presence of light and under slightly acidic conditions. In a conventional emulsification system this process is highly accelerated due to heat and other stress conditions, and the EE is always lower at around 60% even within one week of storage. In comparison the MCE system improves the stability as well as the encapsulation efficiency up to two weeks of storage. In our research these emulsions were stored at 4 °C and most of the emulsion samples encapsulating ergocalciferol were subjected to this optimum temperature for storage. In our previous study we found no prominent different in the EE of O/W emulsions encapsulating both ergocalciferol and cholecalciferol at 4 and 25 °C.34
Semo et al.49 encapsulated ergocalciferol in casein micelles, having an initial EE of over 85%. Moreover, the concentration of ergocalciferol in casein micelles was about 5.5 times the concentration in serum surrounding the micelles. Ron et al.50 encapsulated ergocalciferol in β-lg stabilized nanoparticles. The concentration of ergocalciferol in nanoparticles was 55 times higher in comparison to unbounded ergocalciferol. Ergocalciferol encapsulated in the above-mentioned studies performed better against oxygen diffusion, interaction with oxidizing agents and harmful effects of UV radiation. The evaluation of these mechanisms is beyond the scope of the present study.
Conclusions
Monodisperse food grade O/W emulsions loaded with ergocalciferol were successfully formulated through grooved MCE and straight-through MCE. The key point of our findings is the stable generation of uniformly sized O/W droplets that encapsulate ergocalciferol via MC arrays of asymmetric microstructures, without any coalescence or wetting of the dispersed phase during MCE. Successful grooved MCE is achieved with different food grade ingredients as dispersed and continuous phases. The high throughput studies with straight-through MCE indicate successful operating conditions under a critical dispersed phase flux as well as under 1% (w/w) Tween 20 as an optimum emulsifier in the continuous phase. There was hardly any increase in the droplet size during 15 days of storage. The resultant O/W emulsions containing ergocalciferol have an encapsulating efficiency of more than 75% after 15 days of storage time. The improved physical and chemical stability correlate well with the monodispersity of the emulsion system. Our results indicate that MCE is a promising technique for encapsulating bioactive compounds, with superior control of the processing parameters and various other physical conditions. The forthcoming scaling up of MCE devices is expected to further improve the production capacity of emulsions, to make it practical on an industrial scale through much more throughput production capacities.References
- M. T. Cantorna and B. D. Mahon, Exp. Biol. Med., 2004, 229, 1136–1142 CAS.
- E. Hypponen, E. Laara, A. Reunanen, M. R. Jarvelin and S. M. Virtanen, Lancet, 2001, 358, 1500–1503 CrossRef CAS.
- J. Kendrick, G. Targher, G. Smits and M. Chonchol, Atherosclerosis, 2009, 205, 255–260 CrossRef CAS.
- J. M. Lappe, D. Travers-Gustafson, K. M. Davies, R. R. Recker and R. P. Heaney, Am. J. Clin. Nutr., 2007, 85, 1586–1591 CAS.
- F. Bandeira, A. G. Costa, M. A. Soares Filho, L. Pimentel, L. Lima and J. P. Bilezikian, Arq. Bras. Endocrinol. Metabol., 2014, 58, 504–513 CrossRef PubMed.
- I. G. Song and C. J. Park, Blood Res., 2014, 49, 84 CrossRef PubMed.
- L. M. Ward, I. Gaboury, M. Ladhani and S. Zlotkin, Can. Med. Assoc. J., 2007, 177, 161–166 CrossRef PubMed.
- R. W. Chesney, Pediatr. Int., 2003, 45, 509–511 CrossRef PubMed.
- M. F. Holick, J. Cell. Biochem., 2003, 88, 296–307 CrossRef CAS PubMed.
- M. F. Holick, J. Invest. Med., 2011, 59, 872–880 CAS.
- R. B. Japelt and J. Jakobsen, Front. Plant Sci., 2013, 4, 136 CrossRef PubMed.
- T. D. Thacher and B. L. Clarke, Mayo Clin. Proc., 2011, 86, 50–60 CrossRef CAS PubMed.
- L. A. Armas, B. W. Hollis and R. P. Heaney, J. Clin. Endocrinol. Metab., 2004, 89, 5387–5391 CrossRef CAS PubMed.
- H. M. Trang, D. E. Cole, L. A. Rubin, A. Pierratos, S. Siu and R. Vieth, Am. J. Clin. Nutr., 1998, 68, 854–858 CAS.
- L. A. Houghton and R. Vieth, Am. J. Clin. Nutr., 2006, 84, 694–697 CAS.
- R. R. Eitenmiller, W. Landen Jr and L. Ye, Vitamin analysis for the health and food sciences, CRC Press, 2007 Search PubMed.
- M. R. Patel and M. F. San Martin-Gonzalez, J. Food Sci., 2012, 77, N8–N13 CrossRef CAS PubMed.
- L. L. Schramm, Microscience and Applications of Emulsions, Foams, Suspensions, and Aerosols, John Wiley & Sons, 2014 Search PubMed.
- D. J. McClements, Crit. Rev. Food Sci. Nutr., 2007, 47, 611–649 CrossRef CAS PubMed.
- G. T. Vladisavljević, N. Khalid, M. A. Neves, T. Kuroiwa, M. Nakajima, K. Uemura, S. Ichikawa and I. Kobayashi, Adv. Drug Delivery Rev., 2013, 65, 1626–1663 CrossRef PubMed.
- S. Sugiura, M. Nakajima, N. Kumazawa, S. Iwamoto and M. Seki, J. Phys. Chem. B, 2002, 106, 9405–9409 CrossRef CAS.
- G. T. Vladisavljević, I. Kobayashi and M. Nakajima, Microfluid. Nanofluid., 2012, 13, 151–178 CrossRef.
- I. Kobayashi, S. Mukataka and M. Nakajima, Ind. Eng. Chem. Res., 2005, 44, 5852–5856 CrossRef CAS.
- I. Kobayashi, Y. Wada, Y. Hori, M. A. Neves, K. Uemura and M. Nakajima, Chem. Eng. Technol., 2012, 35, 1865–1871 CrossRef CAS.
- I. Kobayashi, Y. Wada, K. Uemura and M. Nakajima, Microfluid. Nanofluid., 2010, 8, 255–262 CrossRef CAS.
- S. Sugiura, M. Nakajima, J. H. Tong, H. Nabetani and M. Seki, J. Colloid Interface Sci., 2000, 227, 95–103 CrossRef CAS PubMed.
- F. Ikkai, S. Iwamoto, E. Adachi and M. Nakajima, Colloid Polym. Sci., 2005, 283, 1149–1153 CAS.
- S. Sugiura, T. Kuroiwa, T. Kagota, M. Nakajima, S. Sato, S. Mukataka, P. Walde and S. Ichikawa, Langmuir, 2008, 24, 4581–4588 CrossRef CAS PubMed.
- M. A. Neves, H. S. Ribeiro, I. Kobayashi and M. Nakajima, Food Biophys., 2008, 3, 126–131 CrossRef.
- M. A. Neves, H. S. Ribeiro, K. B. Fujiu, I. Kobayashi and M. Nakajima, Ind. Eng. Chem. Res., 2008, 47, 6405–6411 CrossRef CAS.
- N. Khalid, I. Kobayashi, M. A. Neves, K. Uemura, M. Nakajima and H. Nabetani, Colloids Surf., A, 2014, 458, 69–77 CrossRef CAS.
- N. Khalid, I. Kobayashi, M. A. Neves, K. Uemura, M. Nakajima and H. Nabetani, Colloids Surf., A, 2014, 459, 247–253 CrossRef CAS.
- S. Souilem, I. Kobayashi, M. Neves, S. Sayadi, S. Ichikawa and M. Nakajima, Food Bioprocess Technol., 2014, 7, 2014–2027 CrossRef CAS.
- N. Khalid, I. Kobayashi, Z. Wang, M. A. Neves, K. Uemura, M. Nakajima and H. Nabetani, Int. J. Food Sci. Technol., 2015, 50, 1807–1814 CrossRef CAS.
- Z. Wang, M. A. Neves, I. Kobayashi and M. Nakajima, Controlling properties of micro-to nano-sized dispersions using emulsification devices, Wiley-Blackwell, Oxford, UK, 2015 Search PubMed.
- O. Brand, G. K. Fedder, C. Hierold, J. G. Korvink, O. Tabata and N. Kockmann, Micro process engineering: fundamentals, devices, fabrication, and applications, John Wiley & Sons, 2013 Search PubMed.
- I. Kobayashi, S. Mukataka and M. Nakajima, J. Colloid Interface Sci., 2004, 279, 277–280 CrossRef CAS PubMed.
- K. van Dijke, I. Kobayashi, K. Schroen, K. Uemura, M. Nakajima and R. Boom, Microfluid. Nanofluid., 2010, 9, 77–85 CrossRef CAS.
- C. Tan, I. Kobayashi and M. Nakajima, Preparation of monodispersed refined-bleached-deodorized (RBO) palm olein-in-water emulsions by microchannel emulsification, Proceedings of the International Chemical Congress of Pacific Basin Societies, 15-20, Honolulu, Hawaii, December 2005 Search PubMed.
- I. Kobayashi and M. Nakajima, Eur. J. Lipid Sci. Technol., 2002, 104, 720–727 CrossRef CAS.
- N. Khalid, I. Kobayashi, M. A. Neves, K. Uemura, M. Nakajima and H. Nabetani, Biosci., Biotechnol., Biochem., 2015, 79, 1852–1859 CrossRef CAS PubMed.
- T. Kawakatsu, G. Tragardh, Y. Kikuchi, M. Nakajima, H. Komori and T. Yonemoto, J. Surfactants Deterg., 2000, 3, 295–302 CrossRef CAS.
- I. Kobayashi, M. A. Neves, Y. Wada, K. Uemura and M. Nakajima, Procedia Food Sci., 2011, 1, 109–115 CrossRef CAS.
- G. T. Vladisavljević, I. Kobayashi and M. Nakajima, Microfluid. Nanofluid., 2011, 10, 1199–1209 CrossRef.
- I. Kobayashi, S. Mukataka and M. Nakajima, Langmuir, 2005, 21, 7629–7632 CrossRef CAS PubMed.
- S. Sugiura, M. Nakajima, S. Iwamoto and M. Seki, Langmuir, 2001, 17, 5562–5566 CrossRef CAS.
- I. Kobayashi, T. Takano, R. Maeda, Y. Wada, K. Uemura and M. Nakajima, Microfluid. Nanofluid., 2008, 4, 167–177 CrossRef.
- S. Schultz, G. Wagner, K. Urban and J. Ulrich, Chem. Eng. Technol., 2004, 27, 361–368 CrossRef CAS.
- E. Semo, E. Kesselman, D. Danino and Y. D. Livney, Food Hydrocolloids, 2007, 21, 936–942 CrossRef CAS.
- N. Ron, P. Zimet, J. Bargarum and Y. D. Livney, Int. Dairy J., 2010, 20, 686–693 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2015 |