Highly efficient one-pot four-component Kabachnik–Fields synthesis of novel α-amino phosphonates under solvent-free and catalyst-free conditions†
Zahra Rashida,
Hossein Naeimi*b and
Ramin Ghahremanzadeh*a
aNanobiotechnology Research Center, Avicenna Research Institute, ACECR, Tehran, Iran. E-mail: r.ghahremanzadeh@avicenna.ac.ir; Fax: +98-02122432021; Tel: +98-02122432020
bDepartment of Organic Chemistry, Faculty of Chemistry, University of Kashan, Kashan, 87317, I.R. Iran. E-mail: Naeimi@kashanu.ac.ir; Fax: +98-03615511121; Tel: +98-03615912388
First published on 10th November 2015
Abstract
In the present study, a simple and highly efficient protocol for the preparation of novel α-amino phosphonates is achieved based on the one-pot reaction of ninhydrin, 1,2-phenylenediamines, aniline derivatives, and dialkyl or diaryl phosphites, without using any catalyst under solvent-free conditions. The main advantages of the reaction are short reaction time, high yields, simple workup, and no need for further purification.
Introduction
During the past few years, much attention has been focused on the development of new protocols in organic synthesis for environmentally benign processes due to increasing green chemistry concerns.1,2 As a result, many reactions are designed to proceed cleanly and efficiently such as catalyst- and solvent-free synthesis and multicomponent reactions (MCRs).3–5 MCRs are special type of synthetically useful organic reactions that have emerged as a powerful tool to construct complex molecular architecture in a single chemical step to produce a product that incorporates substantial portions of all the reactants with high atom efficiency.6 They are basically different from two component stepwise reactions such as: the simplicity of one-pot procedure, possible structural variation, complicated synthesis, and large number of accessible compounds.7 Multicomponent reactions under solvent-free conditions have gained significant interest due to several advantages over the conventional methods including less chemical pollution, low cost, simplicity in process and handling and, easier workup that these factors are vital especially in industry.8,9The chemistry of organic phosphorus compounds has shown a remarkable growth throughout the past 5 decades.10 α-Aminophosphonates are phosphorus analogs of α-amino acids, constitute an important class of compounds in pharmaceutical chemistry with wide range of useful pharmacological and biological activities such as anticancer agents,11 antiviral agents,12 enzyme inhibitors,13 peptide mimics,14 antibiotic,15 antithrombotic agent,16 anti-inflammatory agent,17 and etc. Due to that, in recent years, significant efforts have been devoted for preparation of these valuable compounds. One of the most common and convenient methods for the synthesis of these compounds is Kabachnik–Fields reaction.18 This reaction is a one-pot, three-component reaction of carbonyl compound, amines and phosphites. A wide range of catalysts has been employed for the Kabachnik–Fields reaction to obtain phosphorylated derivatives such as; Lewis acids,19–22 Brønsted acids,23–25 nano catalysts,26,27 and heterogeneous catalysts.28 But most of the reported procedures suffer from several shortcomings such as; long reaction times, the use of hazardous and expensive solvents and catalysts, low yields of the products, harsh reaction conditions, and tedious workup procedures that are harmful to environment, and limit the use of these methods. Hence, the development of an effective and simple method under green reaction conditions for the synthesis of mentioned structures would extend the important scope of the synthesis of these compounds.
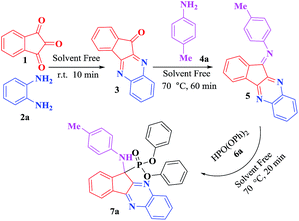
In continuation of our program that purposes to develop environmentally friendly methods for the preparation of heterocyclic compounds,29–31 the present work was aimed to synthesize novel α-aminophosphonates in a simple and efficient method from a four-component condensation reaction of ninhydrin, 1,2-phenylenediammines, various anilines, and dialkyl or diaryl phosphites without using catalyst under solvent-free condition (Scheme 1).
 | ||
| Scheme 1 One-pot synthesis of novel α-aminophosphonates under solvent-free and catalyst-free conditions. | ||
Experimental
The chemicals used in this work were obtained from Fluka and Merck and were used without purification. Melting points were measured on an Electrothermal 9200 apparatus. IR spectra were recorded as KBr pellets on a Perkin-Elmer 781 spectrophotometer and an Impact 400 Nicolet FT-IR spectrophotometer. 1H NMR and 13C NMR spectra were recorded in DMSO-d6 solvents on a Bruker DRX-400 spectrometer with tetramethylsilane as internal reference. The elemental analyses (C, H, N) were obtained from a Carlo ERBA Model EA 1108 analyzer. The purity determination of the substrates and reaction monitoring were accomplished by TLC on silica-gel polygram SILG/UV 254 plates (from Merck Company).Typical general procedure for synthesis of α aminophosphonates
A mixture of ninhydrin 1 (1 mmol) and 1,2-phenylenediamines 2 (1 mmol) was stirred by a magnet in a test tube under solvent free conditions at room temperature for 10 min, followed by the addition of aniline derivatives (1 mmol) temperature was slowly increased until 70 °C. The reaction mixture was stirred for appropriate time, after nearly complete conversion into an intermediate presumed to be the corresponding intermediate 5 (as indicated by TLC), alkyl/aryl phosphite (1 mmol) was then added to the reaction mixture which was heated at 70 °C. After completion of the reaction (as assessed by TLC), the reaction mixture was cooled to room temperature, and EtOH (5 mL) was added to the crude products and stirred for a while. Then, the mixture was filtered and the precipitate washed with EtOH to afford the pure products. The spectroscopic and analytical data for synthesized compounds are presented below.Results and discussion
In recent years, the green situation has become an outstanding topic. The reactions under catalyst- and solvent-free conditions are substantially safe, nontoxic, environmentally friendly, and inexpensive. In this research, we have planned one-pot and four component condensation reaction in order to synthesis of novel α-amino phosphonates under solvent free conditions without using catalyst with the hope that new biological activities could be developed. To attain this goal, at first an equimolar mixture of ninhydrin (1) and o-phenylenediamine (2) was taken in a 5 mL glass test tube. The reaction mixture was stirred at room temperature for the synthesis of 11H-indeno[1,2-b]quinoxalin-11-one (3) as a yellow powder. Then p-methyl aniline (4) was added and temperature was slowly increased until 70 °C. After completion of the reaction (as indicated by TLC and reaction mixture color changed to red), diphenyl phosphites was added. The reaction proceeded efficiently with completely conversion to afford the corresponding 7a (90% yields) within short reaction time (20 min) at 70 °C. The steps of synthesis are shown in Scheme 2.The structure of 7a was confirmed by Fourier transform infrared (FT-IR), 1H NMR, and 13C NMR. The FT-IR spectra of 7a shows the presence of absorption band at 3293 cm−1 region corresponding to the stretching vibrations of the NH groups. While the absorption bands at the 1381–1383 cm−1 region are due to the symmetric stretching vibrations of the P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group and absorption bands within the 896–905 cm−1 region are attributed to the P–O–C groups. In the 1H NMR spectrum, the signal at δ = 1.88 ppm with three integral values indicate the presence of –CH3 protons of –CH3 group of aromatic ring. The doublet signal at δ = 6.94 ppm (J = 11.6 Hz) (D2O exchangeable) confirmed the presence of –NH proton. The aromatic protons exhibited multiplet in the region δ = 6.08–8.29 ppm with 22 integral values (ESI†). In the 13C NMR, the peak at δ = 20.28 ppm shows the presence of methyl carbon of aromatic ring. The carbon banded to the phosphorus resonated as doublet at δ = 67.85 ppm (J = 148 Hz). Also this spectra exhibit expected 26 peaks of substituted aromatic carbons at in the 116.34–153.78 ppm region. The results agree well with our expected product (ESI†).
O group and absorption bands within the 896–905 cm−1 region are attributed to the P–O–C groups. In the 1H NMR spectrum, the signal at δ = 1.88 ppm with three integral values indicate the presence of –CH3 protons of –CH3 group of aromatic ring. The doublet signal at δ = 6.94 ppm (J = 11.6 Hz) (D2O exchangeable) confirmed the presence of –NH proton. The aromatic protons exhibited multiplet in the region δ = 6.08–8.29 ppm with 22 integral values (ESI†). In the 13C NMR, the peak at δ = 20.28 ppm shows the presence of methyl carbon of aromatic ring. The carbon banded to the phosphorus resonated as doublet at δ = 67.85 ppm (J = 148 Hz). Also this spectra exhibit expected 26 peaks of substituted aromatic carbons at in the 116.34–153.78 ppm region. The results agree well with our expected product (ESI†).
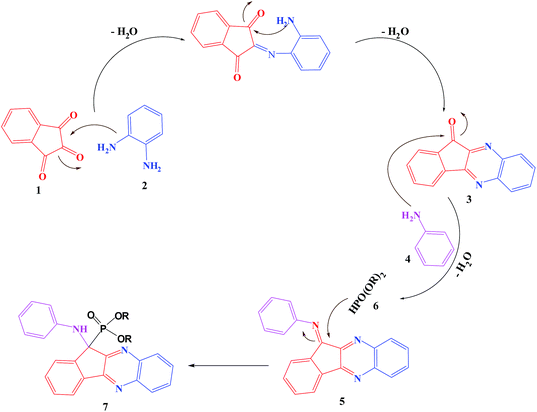
The multicomponent reaction proceeds through cyclocondensation of ninhydrin (1) and o-phenylenediamine (2) to give indenoquinoxline-11-one (3), which further condensed with aniline (4) to produce intermediate (9), then the dialkyl phosphite is added on the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N band of the intermediate (9) to furnish the α-aminophosphonate. A probable mechanistic pathway has been proposed in Scheme 3.
N band of the intermediate (9) to furnish the α-aminophosphonate. A probable mechanistic pathway has been proposed in Scheme 3.
Encouraged by this success, we extended the reaction of ninhydrin and 1,2-phenylenediammines, with a range of aromatic amines, and dialkyl or diaryl phosphites under same conditions for the synthesis of libraries of these compounds. The structure of prepared products is shown in Scheme 4. All products were obtained in high to excellent yields in short reaction times with high purity. The influence of electron-withdrawing and electron-donating substituents on the aromatic ring of aniline upon the reaction yields and time of reaction was investigated. The results showed that anilines with electron-donating group on the aromatic ring reacted successfully. Based on results, type, size, and steric effect of electron-donating substituents didn't have effect on time of the reaction and yields. While anilines bearing electron withdrawing groups such as o-NO2, m-NO2, and p-NO2 groups decreased the reactivity of anilines, and even after 24 h the corresponding imines (5) were not formed and the reaction was stopped at this stage.
Conclusions
In the present research, an efficient and operationally simple Kabachnik–Fields reaction for the synthesis of novel α-aminophosphonates under solvent-free and catalyst-free conditions has been developed. Mild, non-hazardous, environment friendly reaction conditions and short reaction times are significant advantages of this method. The work up is easy and furnished the desired products in excellent yields.Acknowledgements
We are thankful from University of Kashan for supporting this work by grant number 159148/36, and also gratefully acknowledge financial support from the Avicenna Research Institute.References
- R. C. Cioc, E. Ruijter and R. V. A. Orru, Green Chem., 2014, 16, 2958–2975 RSC.
- N. Poomathi, S. Mayakrishnan, D. Muralidharan, R. Srinivasan and P. T. Perumal, Green Chem., 2015, 17, 3362–3372 RSC.
- S. Kumar, P. Sharma, K. K. Kapoor and M. S. Hundal, Tetrahedron, 2008, 64, 536–542 CrossRef CAS.
- P. M. Habib, V. Kavala, C. W. Kuo, M. J. Raihan and C. F. Yao, Tetrahedron, 2010, 66, 7050–70565 CrossRef CAS.
- R. Ghahremanzadeh, G. Imani Shakibaei, S. Ahadi and A. Bazgir, J. Comb. Chem., 2010, 12, 191–194 CrossRef CAS PubMed.
- R. C. Cioc, E. Ruijter and R. V. A. Orru, Green Chem., 2014, 16, 2958–2975 RSC.
- (a) G. Frapper, C. Bachmann, Y. Gu, R. C. de Sousa and F. Jérôme, Phys. Chem. Chem. Phys., 2011, 13, 628–636 RSC; (b) J. P. Wan and Y. Liu, RSC Adv., 2012, 2, 9763–9777 RSC.
- E. T. Tigineh, Y. S. Wen and L. K. Liu, Tetrahedron, 2015, 71, 170–175 CrossRef.
- M. Ghorbania, H. R. Shaterian, S. Noura, F. Khammar, K. Behbodi, B. Reisi and M. Oftadeh, J. Mol. Liq., 2015, 204, 15–20 CrossRef.
- F. H. Osman and F. A. El-Samahy, Chem. Rev., 2002, 102, 629–677 CrossRef CAS PubMed.
- (a) L. Gu and C. Jin, Org. Biomol. Chem., 2012, 10, 7098–7102 RSC; (b) L. Jin, B. Song, G. Zhang, R. Xu, S. Zhang, X. Gao, D. Hu and Y. Song, Bioorg. Med. Chem. Lett., 2006, 16, 1537–1543 CrossRef CAS PubMed; (c) P. Kafarski and B. Lejczak, Curr. Med. Chem.: Anti-Cancer Agents, 2001, 1, 301–312 CrossRef CAS PubMed.
- (a) N. Long, X. J. Cai, B. A. Song, S. Yang, Z. Chen, P. S. Bhaduri, D. Y. Hu, L. H. Jin and W. J. Xue, Agric. Food. Chem., 2008, 56, 5242–5246 CrossRef CAS PubMed; (b) J. Zhou, B. A. Fan, L. H. Song, P. S. Jin, D. Y. Bhaduri, S. Hu and H. T. Yang, Phosphorus, Sulfur Silicon Relat. Elem., 2010, 186, 81–87 CrossRef.
- S. D. Lombaert, L. Blanchard, J. Tan, Y. Sakane, C. Berry and R. D. Ghai, Bioorg. Med. Chem. Lett., 1995, 5, 145–150 CrossRef.
- B. Lejczak, P. Kafarski, H. Sztajer and P. Mastarlerz, J. Med. Chem., 1986, 29, 2212–2217 CrossRef CAS PubMed.
- H. V. Patel, K. V. Vyas and P. S. Fernandes, Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem., 1990, 29, 836–842 Search PubMed.
- F. R. Atherton, C. H. Hassall and R. W. Lambert, J. Med. Chem., 1986, 29, 29–40 CrossRef CAS PubMed.
- R. D. Dillard, D. E. Pavey and D. N. Benslay, J. Med. Chem., 1973, 16, 251–253 CrossRef CAS PubMed.
- (a) P. S. Reddy, P. V. G. Reddy and S. M. Reddy, Tetrahedron Lett., 2014, 55, 3336–3339 CrossRef CAS; (b) K. Azizi, M. Karimi and A. Heydari, Tetrahedron Lett., 2014, 55, 7236–7239 CrossRef CAS; (c) E. Alcalde, N. Mesquida, C. Alvarez-Rúa, R. Cuberes, J. Frigola and S. García-Granda, Molecules, 2008, 13, 301–318 CrossRef CAS PubMed; (d) N. S. Zefirov and E. D. Matveeva, ARKIVOC, 2008, 1–17 CAS.
- M. V. N. Reddy, S. D. Dindulkar and Y. T. Jeong, Tetrahedron Lett., 2011, 52, 4764–4767 CrossRef CAS.
- Z. Rezaei, H. Firouzabadi, N. Iranpoor, A. Ghaderi, M. Reza Jafari, A. A. Jafari and H. R. Zare, Eur. J. Med. Chem., 2009, 44, 4266–4275 CrossRef CAS PubMed.
- S. Bhagat and A. K. Chakraborti, J. Org. Chem., 2007, 72, 1263–1270 CrossRef CAS PubMed.
- A. K. Bhattacharya and Y. Kaur, Synlett, 2007, 5, 745–748 CrossRef.
- T. Akiyama, Chem. Rev., 2007, 107, 5744–5758 CrossRef CAS PubMed.
- (a) S. M. Vahdat, R. Baharfar, M. Tajbakhsh, A. Heydari, S. M. Baghbanian and S. Haksar, Tetrahedron Lett., 2008, 49, 6501–6504 CrossRef CAS; (b) B. Kaboudin and E. Jafari, J. Iran. Chem. Soc., 2008, 5, 97–102 CrossRef.
- S. D. Mitragotri, D. M. Pore, U. V. Desai and P. P. Wadgaonkar, Catal. Commun., 2008, 9, 1822–1826 CrossRef CAS.
- (a) D. Saberi, S. Cheragi, S. Mahdudi, J. Akbari and A. Heydari, Tetrahedron Lett., 2013, 54, 6403–6406 CrossRef CAS; (b) B. V. Subba Reddy, A. Siva Krishna, A. V. Ganesh and G. G. K. S. Narayana Kumar, Tetrahedron Lett., 2011, 52, 1359–1362 CrossRef.
- S. M. Agawane and J. M. Nagarkar, Tetrahedron Lett., 2011, 52, 3499–3504 CrossRef CAS.
- A. Heydari, H. Hamadi and M. Pourayoubi, Catal. Commun., 2007, 8, 1224–1226 CrossRef CAS.
- R. Ghahremanzadeh, T. Amanpour and A. Bazgir, J. Heterocycl. Chem., 2009, 46, 1266–1270 CrossRef CAS.
- A. Bazgir, G. hosseini and R. Ghahremanzadeh, ACS Comb. Sci., 2013, 15, 530–534 CrossRef CAS PubMed.
- G. Imani Shakibaei, S. Samadi, R. Ghahremanzadeh and A. Bazgir, ACS Comb. Sci., 2010, 12, 295–297 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra18311a |
| This journal is © The Royal Society of Chemistry 2015 |