What can electrospray mass spectrometry of paratungstates in an equilibrating mixture tell us?†
Linyuan Fan,
Jie Cao* and
Changwen Hu*
Key Laboratory of Cluster Science, Ministry of Education of China, Beijing Key Laboratory of Photoelectronic/Electrophotonic Conversion Materials, School of Chemistry, Beijing Institute of Technology, Beijing 100081, P. R. China. E-mail: jcao@bit.edu.cn; cwhu@bit.edu.cn; Fax: +86-10-68912631; Tel: +86-10-68912631
First published on 25th September 2015
Abstract
An acidified aqueous solution of Na2WO4 at nearly neutral conditions, in which paratungstates A ([W7O24]6−) and B ([H2W12O42]10−) were coexistent in an equilibrating mixture, was firstly investigated by a combination of ESI-MS, 183W NMR and Raman spectroscopy. [W7O24]6− was proved to be the main species in the equilibrating mixtures at pH ≤ 7 by NMR and Raman spectroscopies, however, it failed to be detected by ESI-MS due to its limited stability in the gas phase. The distinctive composition of the precipitate from its precipitating solution suggests that the product isolated in its crystalline form may not necessarily be the one with the highest abundance in solution. This is the first representative example to demonstrate non-compliant structures derived from ESI-MS and NMR studies, illustrative of the limitation of ESI-MS analysis for labile POMs, and to show different speciation in the solid and its corresponding solution.
Introduction
Polyoxometalates (POMs) are a vast class of anionic oxide clusters of the early transition metals which have been extensively studied due to tunable structures and virtually unmatched range of physical properties.1 A family of isopolyoxotungstates, e.g. [W6O19]2−,2 [W6O22]8−,3 [W7O24]6−,4 [W10O32]4−,5 [H2W12O40]6−,6 [H2W12O42]10−,7 [H4W19O62]6−,8 [H4W22O74]12−,9 [H10W34O116]18−,9 [H12W36O120]12−,10 has been synthesized using a routine “one-pot” reaction by acidification of an aqueous tungstate solution to different pH values. However, the complex solution speciation processes behind the simple method arising from multiple equilibria are still not completely understood.Electrospray ionisation mass spectrometry (ESI-MS) has been used to analyze a range of polyoxoanions, as well as complex mixtures of oxoanions.11,12 The approach has recently been extended to directly probe the reaction solution, thereby simplifying the conventional crystallization-based procedure to cluster discovery.13–15 However, people seldom scrutinize this method. There are cases to show failure on detection of intact POMs by ESI-MS16 and subtle speciation changes due to ESI-induced reactions.17–20
It is known that paratungstate A ([W7O24]6−) is rapidly formed when sodium tungstate is acidified to nearly neutral pH. It will then convert to thermodynamically stable paratungstate B ([H2W12O42]10−) slowly. The kinetics to establish the equilibrium between the two is quite slow.21,22 Usually the salt based on [H2W12O42]10−, not [W7O24]6−, crystallizes upon standing of the equilibrating solution23 unless special measures (i.e., the use of organic amine or transition-metal-complex cations4d,24–28) were taken. The difficulty in the crystallization of heptatungstate is still a great challenge in POM synthesis. Yamase and coworkers29 characterized a pH 6.8 Na2WO4 solution by 183W NMR spectroscopy in which three peaks (δ = 268.3, −178.0 and −91.8 ppm) with an intensity ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 can be assigned to paratungstate A anion as a main species. This result tells us the equilibrium between [W7O24]6− and [H2W12O42]10− is largely shifted to [W7O24]6− at the pH. Our recent work on the MS of an unusual compound Na[Cu(en)2]2[HW7O24]·5H2O (en = ethanediamine)4d indicated that the main species is [W6O19]2− (W6), not [W7O24]6− (W7). The contradictory result from NMR and MS measurements based on the same system begs a question: Is the W7 anion itself not stable in solution or it is simply not survival the ionization process?
4 can be assigned to paratungstate A anion as a main species. This result tells us the equilibrium between [W7O24]6− and [H2W12O42]10− is largely shifted to [W7O24]6− at the pH. Our recent work on the MS of an unusual compound Na[Cu(en)2]2[HW7O24]·5H2O (en = ethanediamine)4d indicated that the main species is [W6O19]2− (W6), not [W7O24]6− (W7). The contradictory result from NMR and MS measurements based on the same system begs a question: Is the W7 anion itself not stable in solution or it is simply not survival the ionization process?
Herein we report a complete investigation on the speciation of an aqueous Na2WO4 solution at nearly neutral pH by a combination of ESI-MS, 183W NMR and Raman spectroscopy. This work is highlighted by the characterization of paratungstates in an equilibrating mixture in solution and solid phases. The validity of ESI-MS method is also addressed by taking this system as a test case.
Experimental
Sample preparation
Na2WO4·2H2O and formic acid were purchased from Sinopharm Chemical Reagent Beijing Co., Ltd and used without further purification. The measurements of MS, NMR and Raman spectroscopy were conducted in triplicate for each sample. A Hanna HI 8521 pH meter (Hanna Instruments, Italy) was used to measure pH values. All aqueous solutions of tungstate for MS analysis were prepared with a concentration of 10−3 M using HPLC-grade water. After adjusting the pH to about 6, 7 and 8 with formic acid, the solutions were allowed to equilibrate for different lengths of time, which were then analyzed. The precipitate from the aqueous solution of tungstate was obtained by adding aqueous solution of tetrabutylammonium bromide with stirring. The resultant precipitates were filtered and washed with water and dried under the ambient environment. The tungstate solutions used for Raman spectroscopy were prepared with concentrations of 0.1 M, 0.5 M, 1 M, 4 M, respectively. TBA2W6O19 and Na[Cu(en)2]2[HW7O24]·5H2O were prepared according to ref. 2 and 4d. The solutions used for 183W NMR were prepared by adjusting pH values of the 0.6 M Na2WO4 (1 g Na2WO4 dissolved in 5 mL of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 H2O
1 H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) D2O) to 5.8, 6.8, 7.8 with H2SO4 (33 wt%).
D2O) to 5.8, 6.8, 7.8 with H2SO4 (33 wt%).
Methods
Mass spectra were recorded on an Agilent 6520 Q-TOF LC/MS in the negative ion mode. Each scan or data point on the total ion chromatogram is an average of 5000 transients, producing a scan every 500 ms. Spectra were created by averaging the scans across each peak. The dual-spray electrospray ionization source conditions: Vcap: 3500 V; skimmer: 65 V; OCT RfV: 750 V; nebulizer: 30 psi; drying gas flow: 10 L min−1; drying gas temperature: 300 °C; fragmentor: 80 V; scan range 100–3000 m/z. Sample solutions were made to approximately 10−3 M in water (solutions) or in acetonitrile (precipitates) and analyzed by direct injection using an automatic sampler with a flow rate of 0.2 mL min−1. CID experiments were performed using N2 as the target gas. Collisional energy voltage was 40 V for [W6O19]2− precursors. 183W NMR spectra of the Na2WO4 solution (in H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) D2O = 1
D2O = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) in nearly neutral condition at 20 °C were obtained on a 400 MHz JEOL ECX NMR system (equipped with 10 mm multinuclear probe head) at 12.5 MHz using external Na2WO4 (2.0 mol L−1 in D2O) as reference. The spectra were recorded over the region between −300 and 500 ppm. Raman spectra were recorded on a RM (Renishaw) spectrophotometer equipped with a liquid-nitrogen-cooled CCD detector. The argon line at 514.5 nm was used for excitation. The measurements were carried out at room temperature (25 ± 3 °C) three times for each sample solution after preparation. The spectra were recorded over the region between 500 and 1500 cm−1. The grating grooves were 1800 L mm−1 and the exposure time was 30 s.
1) in nearly neutral condition at 20 °C were obtained on a 400 MHz JEOL ECX NMR system (equipped with 10 mm multinuclear probe head) at 12.5 MHz using external Na2WO4 (2.0 mol L−1 in D2O) as reference. The spectra were recorded over the region between −300 and 500 ppm. Raman spectra were recorded on a RM (Renishaw) spectrophotometer equipped with a liquid-nitrogen-cooled CCD detector. The argon line at 514.5 nm was used for excitation. The measurements were carried out at room temperature (25 ± 3 °C) three times for each sample solution after preparation. The spectra were recorded over the region between 500 and 1500 cm−1. The grating grooves were 1800 L mm−1 and the exposure time was 30 s.
Results and discussion
Analyses of the acidified solutions of Na2WO4 at nearly neutral condition (pH = 5.8, 6.8) by 183W NMR spectroscopy (Fig. 1) show three groups of W centers, which correspond to a single tungsten center (a) and two types of tungsten centers (consisting of four “cap” (b) and two “belt” (c) W atoms) typically found in a paratungstate A anion [W7O24]6−, related to three peaks at δ = 270.7, −90.6, −177.5 ppm with an intensity ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, respectively. This result suggests that paratungstate A anion [W7O24]6− is a main species in the solutions (pH = 5.8, 6.8), consistent with that of pH 6.8 reported in the literature.29 However, this anion starts a noticeable dissociation (into [WO4]2−) at pH 7.8, indicated by the abundant peak of [WO4]2− in Fig. 1(c).
2, respectively. This result suggests that paratungstate A anion [W7O24]6− is a main species in the solutions (pH = 5.8, 6.8), consistent with that of pH 6.8 reported in the literature.29 However, this anion starts a noticeable dissociation (into [WO4]2−) at pH 7.8, indicated by the abundant peak of [WO4]2− in Fig. 1(c).
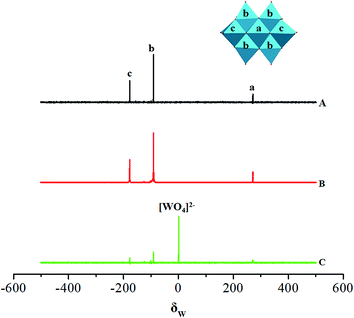 | ||
Fig. 1 183W NMR spectra of Na2WO4 solution (in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 H2O 1 H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) D2O) as a function of pH (equilibrating time = 4 h): (A), pH 5.8; (B), pH 6.8; (C), pH 7.8. D2O) as a function of pH (equilibrating time = 4 h): (A), pH 5.8; (B), pH 6.8; (C), pH 7.8. | ||
Raman spectra of the Na2WO4 solutions show the characteristic peaks for [W7O24]6− at 962 and 903 cm−1 and for [WO4]2− at 931 cm−1, respectively, indicating that [W7O24]6− is the dominant species at pH values of 5.8 and 6.8 except for 7.8 in which [WO4]2− is prevalent (Fig. 2). The characteristic Raman frequencies of isopolytungstates determined to be 931 cm−1 (intense) for [WO4]2−, 962 cm−1 (intense) for [W7O24]6−, and 996 cm−1 (intense) for [W6O19]2− (Fig. S1 in the ESI†), respectively, via using a few compounds with known structures (e.g. an aqueous solution of Na2WO4 with adjusted and unadjusted pH, Na[Cu(en)2]2[HW7O24]·5H2O and TBA2[W6O19] in DMF) were in excellent agreement with those reported in the literature.30,31 Also, it was found that the relative abundances of [W7O24]6− were in direct proportion to its concentrations in solution (Fig. S2 in the ESI†).
 | ||
| Fig. 2 Raman spectra of Na2WO4 solution as a function of pH (equilibrating time = 4 h): (A), pH 5.8; (B), pH 6.8; (C), pH 7.8. | ||
The ESI-MS mass spectra of the Na2WO4 solutions at pH 5.8, 6.8 and 7.8 after equilibrating for 4 hours were shown in Fig. 3, where the major species changes noticeably with the pH. At pH values of 5.8 and 6.8, [W6O19]2− exist as main species which were assigned on the basis of the exact mass and isotopic distribution of the clusters. At a slightly basic pH (7.8), however, [W6O19]2− was barely detectable and [HWO4]− became the dominant species. The reason leading to the conflicting results between NMR, Raman and MS is the stability of [W7O24]6−. Two points should be addressed with regard to the limited stability of [W7O24]6−: (1) [W7O24]6− has a lower symmetry in the structure than the Lindqvist analogue [W6O19]2−; (2) it is very difficult to crystallize heptatungstate from solution because paratungstates A and B are formed simultaneously during the acidification of [WO4]2− at pH 6–8 and establishment of the equilibrium is very slow. To date, only five crystals based on [W7O24]6− have been successfully isolated, Na6[W7O24]·21H2O,4a (C5H10NH3)6[W7O24],4b [(t-C4H9)NH3]6[W7O24]·2H2O,4c Na[Cu(en)2]2[HW7O24]·5H2O (en = ethanediamine),4d and [Co(en)3]2[H2W7O24]·8H2O.4e The limited stability of [W7O24]6− during ESI process was firstly reported by our group for the compound of Na[Cu(en)2]2[HW7O24]·5H2O,4d in which the quasi-molecular ion, [H5W7O24]− (m/z 1675.8376, 67%) partially dissociates into [W6O19]2− (m/z 703.8175, 100%). The subsequent CID experiments on [W6O19]2− generated from two precursors (Fig. 4), one is TBA2[W6O19] (dissolved in pure CH3CN) and the other is an aqueous Na2WO4 solution at pH 6.8, confirms the [W6O19]2− anions generated from different sources have the same Lindqvist structure based on the identical CID spectra.
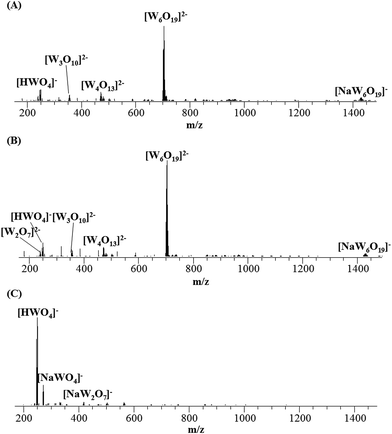 | ||
| Fig. 3 Negative-ion ESI mass spectra of Na2WO4 solution as a function of pH (equilibrating time = 4 h): (A), pH 5.8; (B), pH 6.8; (C), pH 7.8. | ||
The complete absence vs. partial presence of intact molecular ion [W7O24]6− in the mass spectra of aqueous solutions of Na2WO4 at pH 6.8 and Na[Cu(en)2]2[HW7O24]·5H2O may be attributed to the nature of cations. It was mentioned by many studies that cations affect the solution chemistry and also the gas-phase dissociation chemistry of polyoxoanions.32–34 That the copper-complex cation (Cu(en)22+) stabilizes [W7O24]6− to a greater extent than Na+ does allows a lot more portion of intact molecular ion to be detected in presence of Cu(en)22+. As for the same alkali metal cations, Li+ has a stronger stabilizing effect on the polyanion than Na+. Howarth et al.35,36 studied aqueous Li2WO4 solution at pH ca. 6.0 and showed by ESI-MS and 183W NMR spectroscopy to be very largely paratungstate B ([H2W12O42]10−). This result states that the equilibrium between paratungstates A and B is substantially dependent on the cations. The Li+ cation, having the least ionic radius, can effectively shift the balance from paratungstate A to paratungstate B.
The MS analyses of Na2WO4 and Na[Cu(en)2]2[HW7O24]·5H2O in aqueous solutions suggest that the heptatungstate anion can hardly retain its structural integrity in the gas phase, which can be explained by the following reasons. First, the intrinsic structural weakness of the asymmetric anion [W7O24]6−. Second, it was reported by a few papers17–20 that the electrospray ionization process can induce a series of reactions including protonation/desolvation and fragmentation, which make the species in solution more or less different from those detected in the mass spectrum. The ESI-induced dissociation of [W7O24]6− might result from collisions between the analyte and nitrogen gas during the desolvation of electrospray ionization process. This is a special case of non-compliant structures derived from solution and gas phases for the same system. By monitoring the whole equilibrium process of an aqueous Na2WO4 solution at pH 6.8, we found out that the W6 cluster started to be generated in large quantity at 1 hour and kept in growth for longer equilibrium time. No abundant polymerization products other than W6 clusters were detected during the whole equilibrium period (Fig. S3 in the ESI†).
Finally, we tempted to analyze the constitution of the precipitate isolated from the aqueous Na2WO4 solution by adding tetrabutylammonium bromide (TBABr) into the solution at final equilibrating time (48 h). The resultant precipitate was redissolved in CH3CN for ESI-MS analysis after filtration and a few washings with water. By comparing the mass spectrum of the precipitate with that of the corresponding pH 5.8 aqueous solution of Na2WO4 (Fig. 5), it can be seen that a range of W12 species appeared in addition to W6 clusters, e.g., [TBANaxH8−xW12O42]3− (x = 1 and 2, m/z 1049.8479 and 1057.1756, 30% and 77%), [TBA2Na4H3W12O42]3− (m/z 1152.5854, 54%), [TBANaxH9−xW12O42]2− (x = 2 and 3, m/z 1586.2871 and 1597.2804, 19% and 23%), [TBA2NaxH8−xW12O42]2− (x = 4 and 5, m/z 1729.3899 and 1740.3744, 13% and 25%) [TBA3Na6W12O42]2− (m/z 1871.9984, 12%) and [H4W12O40]4− (m/z 712.3118, 100%), [Na3HW12O40]4− (m/z 728.7865, 91%), [NaxH5−xW12O40]3− (x = 0–4, m/z 950.0780, 957.4084 and 964.7362, 972.1154 and 979.3961, 17%, 24%, 33%, 3% and 16%); [W6O19]2− (m/z 703.8073, 12%). This result manifest a fact that most often the product isolated in crystalline form may not necessarily be the one with highest abundance in solution, suggesting that the crystallisation process itself may cause the selective formation of a particular product out of vast library of candidates available in solution. At this end, it is summarized that [W7O24]6− is the main species in the aqueous Na2WO4 solution at pH 5.8 and 6.8, however, this anion can not be isolated from solutions unless special measures were taken. Usually [W12O42]12− (mostly in a high degree of cationized and/or dehydrated form due to the instability of highly charged polyoxoanions) is the solid form isolated from the solution. The results in the study are summarized in Scheme 1.
 | ||
| Fig. 5 Comparison of negative-ion ESI-MS mass spectra of (A) Na2WO4 solution at pH 5.8 (equilibrating time = 48 h); (B) precipitate isolated from (A). | ||
 | ||
| Scheme 1 The equilibrium between paratungstates A and B in an aqueous solution of Na2WO4 at pH ≈ 7 and the actual species detected by ESI-MS. | ||
In summary, this study highlights a combined use of ESI-MS, 183W NMR and Raman spectroscopy to address the speciation in an aqueous solution of Na2WO4 at nearly neutral conditions. In general, [W7O24]6− is proved to be the main species in an equilibrating mixture at pH ≤ 7 by NMR and Raman spectroscopies. This fragile anion, however, fails to be detected due to its ESI-induced dissociation into Lindqvist [W6O19]2−. The distinctive composition of the precipitate from its precipitating solution suggests that the product isolated in crystalline form may not necessarily be the one with the highest abundance in solution. This study also shows that ESI-MS is applicable mostly to stable polyanions. [W7O24]6− polyanion is a representative of unstable polyanions whose structures are labile, cautions must be taken when correlating the MS data with the solution speciation in this case.
Acknowledgements
The authors thank the National Natural Science Foundation of China (21173021, 21231002, 21276026, 21371025), 973 Program (2014CB932103), the 111 Project (B07012) and the Fundamental Research Grant (20121942006) by Beijing Institute of Technology. We thank Dr Zhengguo Lin for performing the W-NMR analysis.References
- M. T. Pope, Heteropoly and Isopoly Oxometalates, Springer-Verlag, Berlin, 1983 Search PubMed.
- A. P. Ginsberg, Inorganic Syntheses, Wiley-Interscience Publication, New York, 1990, vol. 27, pp. 80–81 Search PubMed.
- (a) H. Hartl, R. Palm and J. Fuchs, A New Type of Paratungstate, Angew. Chem., Int. Ed. Engl., 1993, 32, 1492–1494 CrossRef; (b) S. Reinoso, M. H. Dickman and U. Kortz, A Novel Hexatungstate Fragment Stabilized by Dimethyltin Groups: [{(CH3)2Sn}2(W6O22)]4−, Inorg. Chem., 2006, 45, 10422–10424 CrossRef CAS PubMed.
- (a) K. G. Burtseva, T. S. Chernaya and M. I. Sirota, Determination of the Crystal and Molecular Structure of Sodium Paratungstate, Sov. Phys. Dokl., 1978, 23, 784–786 Search PubMed; (b) J. Fuchs and E. P. Flindt, Preparation and Structure Investigation of Polytungstates. A Contribution to the Paratungstate A Problem, Z. Naturforsch., B: Anorg. Chem., Org. Chem., 1979, 34, 412–422 Search PubMed; (c) S. Ikenoue, M. Mikuriya, O. Miyauchi, R. Nukada and A. Yagasaki, Synthesis and Characterization of Tungstates Soluble in Nonaqueous Media, Bull. Chem. Soc. Jpn., 1994, 67, 2590–2592 CrossRef CAS; (d) X. F. Wang, J. Cao, K. L. Huang, Y. Q. Xu, Y. N. Chi and C. W. Hu, 3D Coordination Polymer of [HW7O24]5− Stabilized by a Copper(II) Complex and Sodium Cations: Structure, Solid-State Stability, and Aqueous Solution Behavior, Eur. J. Inorg. Chem., 2013, 1788–1792 CrossRef CAS; (e) G. Yan, X. Wang, Y. Y. Ma, X. Cheng, Y. H. Wang and Y. G. Li, A New Paratungstate-A-Based Organic–Inorganic Hybrid Compound: Synthesis, Structure and Photocatalytic Property of [Co(en)3]2[H2W7O24]·8H2O, Solid State Sci., 2013, 17, 146–150 CrossRef CAS.
- Y. Sasaki, T. Yamase, Y. Ohashi and Y. Sasada, Structural Retention of Decatungstates upon Photoreduction, Bull. Chem. Soc. Jpn., 1987, 60, 4285–4290 CrossRef CAS.
- (a) R. Singer and H. Gross, Über den Bau einiger Heteropolysäuren, Helv. Chim. Acta, 1934, 17, 1076–1080 CrossRef; (b) S. Himeno, K. Nakajima and K. Eda, Facile Preparation of An α–Keggin–Type [H3W12O40]5− Complex: Does It Exist in Aqueous Solution, Polyhedron, 2010, 29, 2595–2599 CrossRef CAS.
- A. Chrissafidou, J. Fuchs, H. Hartl and R. Palm, Crystallization and Structure Determination of Alkaline Metal-Paratungstates, Z. Naturforsch., B: J. Chem. Sci., 1995, 50, 217–222 CAS.
- D. L. Long, P. Kögerler, A. D. C. Parenty, J. Fielden and L. Cronin, Discovery of a Family of Isopolyoxotungstates [H4W19O62]6− Encapsulating a {WO6} Moiety within a {W18} Dawson–like Cluster Cage, Angew. Chem., Int. Ed., 2006, 45, 4798–4830 CrossRef CAS PubMed.
- H. N. Miras, J. Yan, D. L. Long and L. Cronin, Structural Evolution of “S”-Shaped [H4W22O74]12− and “§”-Shaped [H10W34O116]18- Isopolyoxotungstate Clusters, Angew. Chem., Int. Ed., 2008, 47, 8420–8423 CrossRef CAS PubMed.
- D. L. Long, H. Abbas, P. Kögerler and L. Cronin, A High-Nuclearity “Celtic-Ring” Isopolyoxotungstate, [H12W36O120]12−, That Captures Trace Potassium Ions, J. Am. Chem. Soc., 2004, 126, 13880–13881 CrossRef CAS PubMed.
- M. T. Ma, T. Waters, K. Beyer, R. Palamarczuk, P. J. S. Richardt, R. A. J. O'Hair and A. G. Wedd, Gas-Phase Fragmentation of Polyoxotungstate Anions, Inorg. Chem., 2009, 48, 598–606 CrossRef CAS PubMed.
- C. S. Truebenbach, M. Houalla and D. M. Hercules, Characterization of Isopoly Metal Oxyanions Using Electrospray Time-of-flight Mass Spectrometry, J. Mass Spectrom., 2000, 35, 1121–1127 CrossRef CAS.
- J. Yan, D. L. Long, E. F. Wilson and L. Cronin, Discovery of Heteroatom-“Embedded” Te ⊂ {W18O54} Nanofunctional Polyoxometalates by Use of Cryospray Mass Spectrometry, Angew. Chem., Int. Ed., 2009, 48, 4376–4380 CrossRef CAS PubMed.
- Q. D. Jia, J. Cao, Y. P. Duan and C. W. Hu, The Solution Chemistry and Reactivity of Lacunary Keggin Silicotungstates Monitored in Real-time by a Combination of Mass Spectrometry and Electrochemistry, Dalton Trans., 2015, 44, 553–559 RSC.
- E. F. Wilson, H. Abbas, B. J. Duncombe, C. Streb, D. L. Long and L. Cronin, Probing the Self-Assembly of Inorganic Cluster Architectures in Solution with Cryospray Mass Spectrometry: Growth of Polyoxomolybdate Clusters and Polymers Mediated by Silver(I) Ions, J. Am. Chem. Soc., 2008, 130, 13876–13884 CrossRef CAS PubMed.
- S. G. Mitchell, P. I. Molina, S. Khanra, H. N. Miras, A. Prescimone, G. J. T. Cooper, R. S. Winter, E. K. Brechin, D. L. Long, R. J. Cogdell and L. Cronin, A Mixed-Valence Manganese Cubane Trapped by Inequivalent Trilacunary Polyoxometalate Ligands, Angew. Chem., Int. Ed., 2011, 50, 9154–9157 CrossRef CAS PubMed.
- M. Bonchio, O. Bortolini, V. Conte and A. Sartorel, Electrospray Behavior of Lacunary Keggin-Type Polyoxotungstates [XW11O39]p− (X = Si, P): Mass Spectrometric Evidence for a Concentration-Dependent Incorporation of an MOn+ (M = WVI, MoVI, VV) Unit into the Polyoxometalate Vacancy, Eur. J. Inorg. Chem., 2003, 699–704 CrossRef CAS.
- T. H. Bray, R. Copping, D. K. Shuh and J. K. Gibson, Electrospray Ionization Mass Spectrometry of a Cerium (III) Phosphomolybdate Complex: Condensed and Gas-Phase Cluster Chemistry, Int. J. Mass Spectrom., 2011, 299, 35–46 CrossRef CAS.
- P. Yang, Y. X. Xiang, Z. G. Lin, B. S. Bassil, J. Cao, L. Y. Fan, Y. X. Fan, M. X. Li, P. Jiménez-Lozano, J. J. Carbó, J. M. Poblet and U. Kortz, Alkaline Earth Guests in Polyoxopalladate Chemistry: From Nanocube to Nanostar via An Open-Shell Structure, Angew. Chem., Int. Ed., 2014, 53, 11974–11978 CrossRef CAS PubMed.
- Z. G. Lin, B. Wang, J. Cao, B. K. Chen, Y. Z. Gao, Y. N. Chi, C. Xu, X. Q. Huang, R. D. Han, S. Y. Su and C. W. Hu, Cation-Induced Synthesis of New Polyoxopalladates, Inorg. Chem., 2012, 51, 4435–4437 CrossRef CAS PubMed.
- J. F. Duncan and D. L. Kepert, Polyanion Equilibria in Aqueous Solution. Part I. The Quantitative Analysis of AcidiJied Tungstate Solutions, J. Chem. Soc., 1961, 5317–5325 RSC.
- J. F. Duncan and D. L. Kepert, Polyanion Equilibria in Aqueous Solution. Part II. A Thermodynamic Study of the Paratungstate A Anion, J. Chem. Soc., 1962, 205–214 RSC.
- R. Allmann, Die Struktur des Ammoniumparawofframates (NH4)10[H2W12O42]·10H2O, Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem., 1971, 27, 1393–1404 CrossRef CAS.
- D. L. Long, P. Kögerler, A. D. C. Parenty, J. Fielden and L. Cronin, Discovery of a Family of Isopolyoxotungstates [H4W19O62]6− Encapsulating a {WO6} Moiety within a {W18} Dawson-like Cluster Cage, Angew. Chem., Int. Ed., 2006, 45, 4798–4803 CrossRef CAS PubMed.
- D. L. Long, H. Abbas, P. Kögerler and L. Cronin, A High-Nuclearity “Celtic-Ring” Isopolyoxotungstate, [H12W36O120]12−, That Captures Trace Potassium Ions, J. Am. Chem. Soc., 2004, 126, 13880–13881 CrossRef CAS PubMed.
- L. Shen, C. H. Li, Y. N. Chi and C. W. Hu, Zn(2,2′-bipy)2/Co(2,2′-bipy)2 Linked Decaniobate [Nb10O28]6− Clusters–Zigzag Neutral Chains, Inorg. Chem. Commun., 2008, 11, 992–994 CrossRef CAS.
- F. Y. Cui, K. L. Huang, X. Y. Ma, C. Li, Z. G. Han, Y. Z. Gao, X. Liu, Y. N. Chi and C. W. Hu, Transformation from [W6O19]2− to [W6O22]8− stabilized by Cu(II) complexation, Dalton Trans., 2010, 39, 5080–5083 RSC.
- G. L. Guo, Y. Q. Xu, J. Cao and C. W. Hu, An Unprecedented Vanadoniobate Cluster With ‘Trans-Vanadium’ Bicapped Keggin-type {VNb12O40(VO)2}, Chem. Commun., 2011, 47, 9411–9413 RSC.
- C. W. Hu, B. Yue and T. Yamase, Photoassisted Dehalogenation of Organo-Chlorine Compounds by Paratungstate A in Aqueous Solutions, Appl. Catal., A, 2000, 194–195, 99–107 CrossRef CAS.
- P. Haufe, Raman-Spectrophotometric Determination of the Tungstate Anion and Its Isopolyanions in Aqueous Systems, Fresenius' Z. Anal. Chem., 1982, 310, 388–391 CrossRef CAS.
- S. Himeno, M. Yoshihara and M. Maekawa, Formation of Voltammetrically-Active Isopolyoxotungstate Complexes in Aqueous CH3CN Media, Inorg. Chim. Acta, 2000, 298, 165–171 CrossRef CAS.
- C. P. Pradeep, D. L. Long and L. Cronin, Cations in Control: Crystal Engineering Polyoxometalate Clusters Using Cation Directed Self-Assembly, Dalton Trans., 2010, 39, 9443–9457 RSC.
- J. Cao, C. C. Li, Z. X. Zhang, C. Xu, J. Yan, F. Y. Cui and C. W. Hu, Intriguing Role of a Quaternary Ammonium Cation in the Dissociation Chemistry of Keggin Polyoxometalate Anions, J. Am. Soc. Mass Spectrom., 2012, 23, 366–374 CrossRef CAS PubMed.
- J. Cao, C. Xu, Y. X. Fan, L. Y. Fan, X. H. Zhang and C. W. Hu, Selective Production of Electrostatically-Bound Adducts of Alkyl Cations/Polyoxoanions by the Collision-Induced Fragmentations of Their Quaternary Ammonium Counterparts, J. Am. Soc. Mass Spectrom., 2013, 24, 884–894 CrossRef CAS PubMed.
- J. J. Hastings and O. W. Howarth, A 183W, 1H and 17O Nuclear Magnetic Resonance Study of Aqueous Isopolytungstates, J. Chem. Soc., Dalton Trans., 1992, 209–215 RSC.
- M. J. Deery, O. W. Howarth and K. R. Jennings, Application of Electrospray Ionisation Mass Spectrometry to the Study of Dilute Aqueous Oligomeric Anions and Their Reactions, J. Chem. Soc., Dalton Trans., 1997, 4783–4788 RSC.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra18059g |
| This journal is © The Royal Society of Chemistry 2015 |

