Influence of silver grain size, roughness, and profile on the extraordinary fluorescence enhancement capabilities of grating coupled surface plasmon resonance†
A. J. Wooda,
B. Chenb,
S. Pathanb,
S. Bokb,
C. J. Mathaib,
K. Gangopadhyayb,
S. A. Granta and
S. Gangopadhyay*b
aDepartment of Bioengineering, University of Missouri, 254 Agricultural Engineering, Columbia, MO 65211, USA. E-mail: grantsa@missouri.edu
bDepartment of Electrical and Computer Engineering, University of Missouri, 349 Engineering Building West, Columbia, MO 65211, USA. E-mail: gangopadhyays@missouri.edu
First published on 9th September 2015
Abstract
Since the development of fluoroimmunoassays, researchers have sought a method of substantially enhancing fluorescence intensity to extend the limits of detection to new levels of sensitivity. Surface plasmon resonance (SPR) and metal enhanced fluorescence has long been a topic of research and has led to the development of prism- and grating-based SPR systems. However, with the wide coupling range and ease of exciting SPR on plasmonic gratings with a simple microscope objective, they have tremendous potential for revolutionizing the fields of plasmonics, fluorescence, and sensors. In an effort to better understand the influence of grating profile and metal film properties on the extraordinary fluorescence enhancement capabilities of plasmonic gratings, a novel microcontact printing process and different metal deposition techniques were used to fabricate silver gratings with varying grain diameters, roughnesses, heights, and duty cycles using thermal evaporation and RF sputtering. The resulting plasmonic gratings exhibited fluorescence enhancements up to 116× that of dye-coated glass slides using an epifluorescence microscope, much higher than more expensive prism-based SPR systems. This silver grating represents an extraordinary opportunity to quickly and easily enhance fluorescence and widen the detection limits of common fluorescence based assays with little to no equipment modification.
Introduction
Surface Plasmon Resonance (SPR) research has grown substantially over the past several years and led to many substantial discoveries in nanotechnology including plasmonic nanoantennas and nanoresolution imaging techniques.1–6 SPR is essentially a phenomenon where light can be coupled into a metal/dielectric interface to form a propagating charge oscillation called a surface plasmon polariton (SPP). This method of light coupling only occurs if the wave vector of the incident light matches the wave vector of a SPP. Unfortunately, this does not occur under natural situations, which has led to the development of elaborate systems to form matching conditions that utilize structures such as nanoscale gratings, such as Fig. 1a.7–9 The expense of manufacturing platforms such as nanoscale gratings is currently cost prohibitive as it commonly involves the use of electron beam or interference lithography techniques.10,11 To reduce the cost of fabricating nanogratings, a novel soft lithography process was developed that can accurately replicate the gratings found on commercially available HD DVDs in polymethylsilsesquioxane (PMSSQ).12 To form SPR, a plasmonic metal coating, such as gold or silver, is required. While gold is typically chosen over silver due to its corrosion resistance, silver can support much higher field strengths due its inherently lower dielectric losses at optical frequencies.13 Using Maxwell's equations, a wave vector matching equation can be derived for a particular metal grating/dielectric interface, seen in eqn (1).12
 | (1) |
 | ||
| Fig. 1 (a) Illustration of the wave vector matching between incident EM radiation (transverse-magnetic (TM) light) on a grating and a propagating surface plasmon polariton. (b) Theoretical frequency vs. momentum plot for a metallic grating (red and black), and incident EM radiation (blue line).14,15 (c) Experimental frequency vs. momentum for a silver grating in air based on the SPR peak location from the reflectivity scans in Fig. 5. | ||
As a better understanding of SPR has been obtained, a unique interaction was observed between the evanescent electromagnetic (EM) field formed during SPR and nearby fluorescent molecules. It was observed that the emission intensity of fluorophores could be drastically enhanced when located between 10 and 100 nm to the metal surface and a phenomenon later called metal enhanced fluorescence.17 If the frequency of the surface charge oscillation matched the excitation frequency of the fluorophore, the fluorophore would be excited to a higher energy state. However, due to the proximity to metal surface, the fluorophore would non-radiatively transfer energy to the metal and form a radiative plasmon with directed photon emission, also known as surface plasmon coupled emission (SPCE).18 To maximize fluorescent enhancement of this process, the EM field strength, which is closely tied to the properties of the metal/dielectric layers, must be tuned to be strong in the intended fluorophore excitation spectrum. Grating profile features such as height, pitch, and duty cycle as well as defects in the metal such as large grain boundaries and high roughness can have a strong effect on efficiency on SPR generation and must be further studied.
The objective of this study was to examine the effect of grating structure and silver film properties on SPR generation and fluorescence enhancement. Silver gratings with varying metal film and structural properties were fabricated using the previously mentioned microcontact stamping process. The surface structure, extent of light coupling, and fluorescence enhancement capabilities of the different silver gratings was then characterized with atomic force microscopy (AFM), variable angle spectroscopic ellipsometry (VASE), and fluorescence microscopy. To better understand the effect of surface structure on electric field strength, the grating structures were also reproduced in finite difference time domain (FDTD) simulations using FullWave software.
Results and discussion
Silver grating fabrication
HD DVD gratings (height: 60 nm, Λ: 400 nm) were replicated in polymethylsilsesquioxane (PMSSQ) “ink” with an added crosslinker, 3-aminopropyltriethoxysilane (APTES), using a previously developed microcontact stamping process depicted in Fig. 2 and further described in the Experimental section.12 As the gratings were fabricated using the fixed patterns of the HD-DVD, the capability to change the height, pitch and duty cycle of the polymer layer (PMSSQ) was limited. To study the effect of various parameters discussed above on light coupling and concentration, our novel approach was to control the surface energy of the polymer layer, deposition method (RF sputtering vs. thermal evaporation), and the use of different adhesion layers (chromium (Cr) for thermal evaporation, germanium (Ge) or titanium (Ti) for sputtering). The nucleation and growth of thin films relies on the energy of the incoming atoms and their adsorption, diffusion, coalescence/chemical binding to the surface.19 This was controlled by varying the energy of the incoming atoms, surface energy of the polymer film, and binding energy of Ge, Ti, and Cr (adhesion layer) to the polymer surface with and without plasma treatment, as well as, binding energy of Ag–Ag vs. Ag–Ti, Ag–Ge and Ag–Cr. While varying these parameters, it is important to pay attention to the grain size which should not be too large to increase the scattering losses, but small enough to concentrate SPR field (less than 50 nm). The surface energy of as prepared PMSSQ polymer layer was low (0.024 J m−2) due to inherent hydrophobicity of the film. Oxygen plasma treatment changes the film to hydrophilic with high surface energy (0.056 J m−2). The hydrophobic and hydrophilic nature of PMMSQ or wettability of the film is then used to control the interaction of the incoming atoms of the seed layers with PMMSQ surface and growth of the adhesion layer. Subsequently, silver atoms will interact with the seed layer and control the growth of silver films. Metal film growth in general can be described by three growth modes, the Frank–van der Merve growth seen in Fig. 3a (layer by layer growth), Volmer–Weber growth seen in Fig. 3b (island growth), and Stranski–Krastanov growth seen in Fig. 3c (layer and island growth).20 The deposition parameters and surface energy of PMMSQ were varied to control the nucleation and growth processes to achieve films with different silver grain sizes and are discussed in further depth below. | ||
| Fig. 3 Grain and island growth mechanisms for silver films deposited onto (a) germanium, (b) titanium, and (c) chromium adhesion layers. | ||
| Surface pre-treatment | Deposition type | Mean grating pitch, Λ [nm] | Mean grating height [nm] | RMS roughness [nm] | Mean grain diameter [nm] | Duty cycle |
|---|---|---|---|---|---|---|
| No | Sputtered Ge/Ag | 401.4 ± 3.1 | 52.8 ± 1.9 | 1.6 | 31.7 ± 8.2 | 0.533 ± 0.018 |
| Oxygen plasma | Sputtered Ge/Ag | 400.2 ± 3.5 | 55.7 ± 1.7 | 1.7 | 35.6 ± 9.1 | 0.552 ± 0.022 |
| No | Sputtered Ti/Ag | 401.8 ± 3.4 | 53.1 ± 0.5 | 1.9 | 33.2 ± 9.3 | 0.512 ± 0.016 |
| Oxygen plasma | Sputtered Ti/Ag | 396.7 ± 3.7 | 54.8 ± 1.6 | 2.5 | 39.0 ± 9.9 | 0.521 ± 0.018 |
| No | Therm. evap. Cr/Ag | 403.3 ± 5.1 | 43.9 ± 1.9 | 4.0 | 40.1 ± 15.7 | 0.568 ± 0.027 |
| Oxygen plasma | Therm. evap. Cr/Ag | 397.3 ± 8.2 | 42.9 ± 3.5 | 5.1 | 55.8 ± 17.5 | 0.569 ± 0.046 |
Surface and optical characterization
 | ||
| Fig. 4 Topographical AFM scans (1.5 μm × 1.5 μm) of gratings with (a and b) sputtered germanium and silver, (c and d) sputtered titanium and silver, and (e and f) thermally evaporated chromium and silver. Oxygen plasma treated samples (b, d and f) on average exhibited larger average grain diameters than the (a, c and e) untreated counterparts. All scan heights have been plotted from 0 to 60 nm. (g) Grain diameter histograms from the previous AFM scans. (h) Real and (i) imaginary permittivity components taken from flat silver films with the same deposition parameters and PMSSQ/APTES film surface energy with bulk silver values from Johnson and Christy.25 | ||
Fluorescent dye enhancement
NA = n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sin(±θ) sin(±θ)
| (2) |
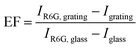 | (3) |
These fluorescence enhancements are also higher than previously observed values for similar fluorescent dyes on plasmonic grating systems. Table 2 has been provided to summarize several recent fluorescence enhancement results on silver and gold gratings with varied parameters. The lower fluorescent enhancement seen in previous studies is thought to arise from the small grain sizes obtained through sputter coating, exciting the platform at a narrow angle range or too far away from the optimal objective illumination range, and incorporation of overly thick capping layers between the silver and the fluorophore. Previously published literature also rarely studied more than two grating parameters and even fewer characterized the metal film properties where, as this article has demonstrated, several parameters must be properly tuned to obtain high fluorescence enhancements.26,29,30
| Grating pitch (nm) | Height (nm) | Duty cycle | Metal, thickness, deposition method, capping layer | Fluorescent dye (Ex. λ/Em. λ, nm) and thickness | Enhancement factor | Source |
|---|---|---|---|---|---|---|
| 400–1000 | 40 | ∼0.5 | Gold, 50 nm, not provided, no cap | R6G (530/550), not provided | 1.7–36× | 28 |
| 480 | 31 | 0.40 | Silver, 200 nm, RF sputter, 20 nm SiO2 cap | Cy5 (630/650), surface conjugated | 24× | 29 |
| 480 | 20–50 | 0.40 | 10× | 30 | ||
| 400 | 10–30 | 0.33–0.72 | 10–40× | 26 | ||
| 300 | 65 | ∼0.50 | Silver, 50 nm, evaporation, no cap | R6G (530/550), 70 nm film | 9–10× | 9 |
| 375 | 65 | 30–60× | ||||
| 400 | 65 | ∼0.5 | Silver, 100 nm, RF sputter, no cap | R6G (530/550), 35 nm film | 26–36× | 12 |
| 400 | 55 | ∼0.5 | Gold, 100 nm, evaporation, no cap | Cy5 (630/650), surface conjugated | 13.6× | 31 |
| 434 | 20–80 | ∼0.5 | Gold, 100 nm, evaporation, no cap | AF647 (633/670), 6 nm film | ∼100× | 16 |
| 400 | 52–55 | 0.51–0.55 | Silver, 100 nm, RF sputter, no cap | R6G (530/550), 35 nm film | 55–90× | This article |
| 400 | 42–44 | 0.57 | Silver, 100 nm, evaporation, no cap | 100–116× |
Influence of grating properties on SPR
It is important to note that the wide distribution of grain sizes present on the various samples encompassed the estimated 30 nm penetration depth of a photon into silver.17 As each photon has a high probability of interacting with grain boundaries for all samples, the boundaries play an important role in the losses associated with each silver film and could be studied using the complex dielectric function. In Fig. 4h, it can be seen that increasing grain size had little effect on the real component of permittivity which is often associated with the energy storage of the metal. However, the imaginary component which is related to the loss properties of a metal film shown in Fig. 4i decreased with increasing grain size. The reduction on the imaginary component primarily arises from the reduction of grain boundary density found on larger grain films.34 Grain boundaries near the metal/dielectric interface can also cause greater internal dampening and increased elastic scattering of SPPs.35 This can have a detrimental effect on SPR propagation length, for example, on copper films and gold nanorods.3,36 In both examples, a reduction in grain boundary density, resulted in higher intensity SPR fields, longer propagation lengths, and higher fluorescent enhancements which is consistent with the results seen in Fig. 6d and 7.
Duty cycle can also have a large effect on the field localization as well as the symmetricity of the electric field profile on the ridges or pits of the grating. The gratings deposited by thermal evaporation have a grating height of ∼43 nm and duty cycle of 0.56 while the samples prepared by sputtering have a grating height of ∼53 nm and duty cycle of 0.52. To better understand the effect that this variation in duty cycle and other grating profile parameters, finite difference time domain simulations were conducted to obtain reflectivity and electric field profiles in the following section.
Electric field simulations
Simulations were also conducted with the addition of a 35 nm thick conformal PMSSQ film to determine the electric field strength experienced by the dye molecules. In these simulated structures, the maximum electric field (Fig. 8b) developed at θ = 5° was more intense over a larger range of coupling wavelengths on the large grain size gratings than the small grain size grating. This is primarily due to the localized field enhancement on the sharp asperities and peaks of the larger grains. Similar electric field localization was also observed experimentally as higher fluorescence intensity of the thermally evaporated silver sample seen in Fig. 7e.
PMSSQ layers on top of silver gratings result in red-shift of the SPR wavelength (Fig. S3†), which makes the plasmonic gratings platforms match better with the excitation and emission of a fluorophore in the range of 530–550 nm as indicated in dispersion curve (Fig. 6b). These grating-based SPR systems also have broad reflectivity coupling peaks, leading to less angle-dependency in electric field strength (Fig. S3 and S4†) as compared with prism-based SPR systems. This especially beneficial for fully utilizing the broad excitation and emission peaks of fluorescent dyes for enhancement.
As shown in Fig. 8e and f, duty cycle, in a similar manner as grating height, must be optimized at a particular wavelength range and grating height to obtain the highest enhancement factors. Reflectivity and electric field intensity plots were obtained on simulated silver gratings with duty cycles between 0.25 and 0.85. The ideal duty cycle to obtain the highest electric field intensity is 0.4 according to the simulation results but at the duty cycles found on the experimental gratings the electric field intensity should be stronger for the thermally evaporated samples than the sputtered silver samples. The reflectivity plots and electric field profiles provided in Fig. S7,† further suggest that gratings with 40 nm grating height exhibit higher electric field strengths at most duty cycles.
Experimental
HD DVD grating replication
HD DVDs are composed of a metal and light sensitive dye layer laminated between two 0.6 mm-thick polycarbonate (PC) sheets with imprinted gratings (Memorex HD DVDs: 400 nm pitch, 60 nm height). PC gratings were exposed by splitting the HD DVD in half followed by sequential rinses in isopropanol and DI water to remove the organic dye layer. Once exposed, the disc was cast in 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 base/cure agent polydimethylsiloxane (PDMS) (Sylgard® 184 silicone elastomer kit, Dow Corning, Inc.) and allowed to cured over 24 hours at 21 °C and 50% relative humidity. PDMS stamps were cut from the mold (Fig. 2a) and spin-coated at 3000 rpm for 30 seconds in a rapidly curing polymer “ink” made from a 3% wt/wt polymethylsilsesquioxane (PMSSQ, GR650F Techneglass, Inc.) and 0.05% vol 3-aminopropyltriethoxysilane (APTES) in 100% ethanol (Fig. 2b). APTES was added to further crosslink the stamped grating and obtain better adhesion to the substrate. The PDMS was immediately stamped onto a glass slide and removed (Fig. 2c). Post stamping, all gratings were heat treated at 60 °C for 30 min to further crosslink the PMSSQ/APTES grating. Half of the gratings were treated with oxygen plasma for 30 seconds at 7 watts using a plasma enhanced chemical vapor deposition (PECVD) system immediately before metal deposition.
1 base/cure agent polydimethylsiloxane (PDMS) (Sylgard® 184 silicone elastomer kit, Dow Corning, Inc.) and allowed to cured over 24 hours at 21 °C and 50% relative humidity. PDMS stamps were cut from the mold (Fig. 2a) and spin-coated at 3000 rpm for 30 seconds in a rapidly curing polymer “ink” made from a 3% wt/wt polymethylsilsesquioxane (PMSSQ, GR650F Techneglass, Inc.) and 0.05% vol 3-aminopropyltriethoxysilane (APTES) in 100% ethanol (Fig. 2b). APTES was added to further crosslink the stamped grating and obtain better adhesion to the substrate. The PDMS was immediately stamped onto a glass slide and removed (Fig. 2c). Post stamping, all gratings were heat treated at 60 °C for 30 min to further crosslink the PMSSQ/APTES grating. Half of the gratings were treated with oxygen plasma for 30 seconds at 7 watts using a plasma enhanced chemical vapor deposition (PECVD) system immediately before metal deposition.
RF sputtering of germanium/silver and titanium/silver films
An AJA International OTC Orion magnetron sputtering system was used to deposit titanium and silver films. Three inch titanium and silver targets (99.999% purity – Kurt J. Lesker) were mounted to two sputtering guns. Prior to deposition, the chamber was pumped down to 2.4 × 10−7 Torr and titanium and silver targets were pre-sputtered for 600 s at 100 W to clean the target and to remove any pre-existing oxide layer from the target surface. A 5 nm thick germanium or titanium adhesion layer followed by a 100 nm thick silver layer was deposited with a 100 W RF power at a working pressure of 4 mTorr, ambient temperature (∼300 K), six inch throw distance, and 20 rpm sample rotation. Germanium and titanium was deposited at 0.1 nm s−1 while silver was deposited at 0.17 nm s−1.Thermal evaporation of chromium/silver films
Chromium and silver films were thermally depositing using a Nano 38 Thermal evaporator from Kurt J. Lesker. The chamber was first pumped down to 2.8 × 10−8 Torr. A 5 nm thick chromium adhesion layer and 100 nm silver layer were deposited sequentially. Chromium was deposited at the rate of 0.005 nm s−1 while silver, initially is deposited at the rate of 0.005–0.01 nm s−1 for the first 5 nm and then increased to 0.05 nm s−1 for the rest of silver deposition. Deposition thicknesses for the RF sputtering and thermal evaporator were calibrated prior to deposition and confirmed with an optical profilometer (Wyko NT 9100, Veeco Instruments, Inc.) and step profilometer (Alpha-Step 200, TENCOR Instruments, Inc.).Surface and optical characterization
Surface structure was examined with a Bruker© Innova AFM equipped with an open-loop, small area scanner and AppNano ACTA silicon SPM tips (k: 40 N m−1, f: 295 kHz, tip radius: ∼6 nm). Reflectivity measurements at different wavelengths and incidence angles as well as spectroscopic scans to obtain the dielectric properties were acquired using a variable angle spectroscopic ellipsometer with a Xeon broadband source (VASE®/HS-190, J. A. Woollam Co, Inc.). Grating ridges were aligned vertically and scanned with transverse-magnetic (TM) light at wavelengths from 200–2000 nm and at angles of incidence from 15–60°.Fluorescence enhancement
Silver grating samples were spin-coated at 3000 rpm for 30 seconds with a 1% wt PMSSQ solution with 10 μM Rhodamine 6G (excitation: 530 nm, emission: 550 nm) in pure ethanol. This produced a conformal, 30 nm-thick layer of fluorophore-laced PMSSQ on top of the gratings. The addition of this layer immediately after metal deposition is also thought to reduce the short term corrosion of the silver layer. The gratings were imaged with an Olympus BX51WI epifluorescence microscope equipped an ORCAflash 2.8 CCD camera, 10/20/40× objectives, 60× oil immersion objective, and illuminated with either a xenon broadband light source or high power 455 nm LED light source.Electric field and reflectivity simulations
Electric field strengths and reflectivity at different wavelengths for different silver grain sizes, grating heights, and duty cycles were simulated using Finite Difference Time Domain (FDTD) software (FullWave by Synopsys). The configuration used for two-dimensional (2D) models was based on HD DVDs grating with a grating pitch of Λ = 400 nm with and without a conformal, 35 nm PMSSQ coating. A monitor located above the launch field was used to measure reflectivity while another monitor encompassing the computational window was used to measure the EM field strength. In the x-direction, a periodic boundary condition was selected due to the periodicity of gratings. In the z-direction, PML (perfectly matched layer) and PEC (perfect electric conductor) boundary conditions were used for the maximum and minimum z value boundary, respectively. A uniform grid size of 2 nm × 2 nm was used for the computational domain for reflectivity measurements and 5 nm × 5 nm for grain size, grating height, and duty cycle simulations, respectively. A computational stop time of 40 μm (in units of cT) was chosen to obtain stability and was excited with a continuous wave (CW) excitation source.Conclusions
In summary, this paper offers an inexpensive method of fabricating gratings that are capable of enhancing fluorescence emission intensity using a simple epifluorescence microscope. It was found that the silver film properties, such as larger grain sizes, can substantially widen the range of coupled wavelengths, increase the coupling efficiency of light into the grating from 30.9% to 84.9%, and reduce the dielectric losses of the metal film. The sharpness and location of the silver grains also played an important role in concentrating the surrounding electric field formed during SPR. The grating structure, such as duty cycle and ridge height, also played a critical role in the formation of intense, symmetrical electric field profiles that were critical in exciting nearby fluorescent molecules. The wider range and more efficient coupling of light into the grating was better utilized for fluorescence enhancement by illumination the grating with the light cone of microscope objectives instead of single angle illumination techniques. The results of this paper also demonstrate the importance of characterizing the metal film properties of a plasmonic grating, properties that are often overlooked in many plasmonic grating design literature. By combining the low loss, field localizing properties of large silver grains with wide range, light coupling properties of plasmonic gratings can result in fluorescence enhancements up to 116× that of dye-coated glass samples. By developing a high fluorescence enhancement plasmonic grating for use in a conventional fluorescence microscope and reducing the expense of its manufacture, it enables the use of this platform in laboratories with little to no system modifications and can be used to replace glass slides and other generic substrates in fluorescence-based assays.Acknowledgements
The authors would like to acknowledge support from the National Science Foundation grant number 1102070 and the Center for Micro/Nano Systems and Nanotechnology at the University of Missouri, Columbia.References
- J. Chen, P. Albella, Z. Pirzadeh, P. Alonso-González, F. Huth, S. Bonetti, V. Bonanni, J. Åkerman, J. Nogués, P. Vavassori, A. Dmitriev, J. Aizpurua and R. Hillenbrand, Small, 2011, 7, 2341–2347 CrossRef CAS PubMed.
- D. K. Gramotnev and S. I. Bozhevolnyi, Nat. Photonics, 2010, 4, 83–91 CrossRef CAS PubMed.
- M. Bosman, L. Zhang, H. Duan, S. F. Tan, C. A. Nijhuis, C. W. Qiu and J. K. W. Yang, Sci. Rep., 2014, 4, 5537 CAS.
- T. Ming, L. Zhao, M. Xiao and J. Wang, Small, 2010, 6, 2514–2519 CrossRef CAS PubMed.
- M. Kuttge, E. J. R. Vesseur, J. Verhoeven, H. J. Lezec, H. A. Atwater and A. Polman, Appl. Phys. Lett., 2008, 93, 113110 CrossRef PubMed.
- W. L. Barnes, D. AlainEbbesen and W. Thomas, Nature, 2003, 424, 824 CrossRef CAS PubMed.
- E. Kretschmann and H. Raether, Z. Naturforsch., A: Phys. Sci., 1968, 23, 2135 CAS.
- A. Otto, Z. Phys., 1968, 216, 398–410 CrossRef CAS.
- Y. Jiang, H.-Y. Wang, H. Wang, B.-R. Gao, Y.-w. Hao, Y. Jin, Q.-D. Chen and H.-B. Sun, J. Phys. Chem. C, 2011, 115, 12636–12642 CAS.
- H. Hori, K. Tawa, K. Kintaka, J. Nishii and Y. Tatsu, J. Appl. Phys., 2010, 107, 114702 CrossRef PubMed.
- X. D. Hoa, A. G. Kirk and M. Tabrizian, Biosens. Bioelectron., 2009, 24, 3043–3048 CrossRef CAS PubMed.
- K. Bhatnagar, A. Pathak, D. Menke, P. V. Cornish, K. Gangopadhyay, V. Korampally and S. Gangopadhyay, Nanotehnology, 2012, 23, 12 Search PubMed.
- P. R. West, S. Ishii, G. V. Naik, N. K. Emani, V. M. Shalaev and A. Boltasseva, Laser Photonics Rev., 2010, 4, 795–808 CrossRef CAS PubMed.
- H. Shinji and O. Takayuki, J. Phys. D: Appl. Phys., 2012, 45, 433001 CrossRef.
- W. Knoll, Annu. Rev. Phys. Chem., 1998, 49, 569–638 CrossRef CAS PubMed.
- M. Bauch, S. Hageneder and J. Dostalek, Opt. Express, 2014, 22, 32026–32038 CrossRef CAS PubMed.
- J. R. Lakowicz, Anal. Biochem., 2005, 337, 171–194 CrossRef CAS PubMed.
- J. R. Lakowicz, Anal. Biochem., 2004, 324, 153–169 CrossRef CAS PubMed.
- J. A. Venables, G. D. T. Spiller and M. Hanbucken, Rep. Prog. Phys., 1984, 47, 399 CrossRef.
- J. E. Greene, in Handbook of Deposition Technologies for Films and Coatings, ed. P. M. Martin, William Andrew Publishing, Boston, 3rd edn, 2010, pp. 554–620 Search PubMed.
- K.-H. Müller, Surf. Sci., 1987, 184, L375–L382 CrossRef.
- Y.-R. Luo, in CRC Handbook of Chemistry and Physics, ed. W. M. Haynes, CRC Press, 96 edn, 2015, ch. 9 Search PubMed.
- W. Chen, M. D. Thoreson, S. Ishii, A. V. Kildishev and V. M. Shalaev, Opt. Express, 2010, 18, 5124–5134 CrossRef CAS PubMed.
- R. Lazzari and J. Jupille, Surf. Sci., 2001, 482–485(2), 823–828 CrossRef CAS.
- P. B. Johnson and R. W. Christy, Phys. Rev. B: Solid State, 1972, 6, 4370–4379 CrossRef CAS.
- X. Cui, K. Tawa, H. Hori and J. Nishii, Adv. Funct. Mater., 2010, 20, 546–553 CrossRef CAS PubMed.
- J.-Y. Laluet, A. Drezet, C. Genet and T. W. Ebbesen, New J. Phys., 2008, 10, 105014 CrossRef.
- Y.-J. Hung, I. I. Smolyaninov, C. C. Davis and H.-C. Wu, Opt. Express, 2006, 14, 10825–10830 CrossRef CAS.
- K. Tawa, H. Hori, K. Kintaka, K. Kiyosue, Y. Tatsu and J. Nishii, Opt. Express, 2008, 16, 9781–9790 CrossRef CAS.
- A. Naoko, T. Keiko, T. Yoshiro, K. Kenji and N. Junji, Jpn. J. Appl. Phys., 2009, 48, 06FH17 Search PubMed.
- A. Wood, S. Grant, S. Basuray, A. Pathak, S. Bok, C. Mathai, K. Gangopadhyay and S. Gangopadhyay, Sensors, 2014 IEEE, 2014, pp. 1479–1482, DOI:10.1109/ICSENS.2014.6985294.
- V. Lozanova, J. Tasseva and R. Todorov, Bulg. Chem. Commun., 2013, 45, 43–46 Search PubMed.
- Z. Liu, S. Durant, H. Lee, Y. Pikus, Y. Xiong, C. Sun and X. Zhang, Opt. Express, 2007, 15, 6947–6954 CrossRef.
- J. R. Sambles, Solid State Commun., 1984, 49, 343–345 CrossRef CAS.
- P. Dawson, K. B. Alexander, J. R. Thompson, J. W. Haas and T. L. Ferrell, Phys. Rev. B: Condens. Matter Mater. Phys., 1991, 44, 6372–6381 CrossRef CAS.
- H. S. Lee, C. Awada, S. Boutami, F. Charra, L. Douillard and R. E. de Lamaestre, Opt. Express, 2012, 20, 8974–8981 CrossRef CAS PubMed.
- P. Berini, Adv. Opt. Photonics, 2009, 1, 484–588 CrossRef CAS.
- D. Sarid, R. T. Deck, A. E. Craig, R. K. Hickernell, R. S. Jameson and J. J. Fasano, Appl. Opt., 1982, 21, 3993–3995 CrossRef CAS PubMed.
- J. Jaewook, S. Kim, C. Junhee and H. Yongtaek, IEEE Electron Device Lett., 2009, 30, 1284–1286 CrossRef.
- H. Raether, Surf. Sci., 1983, 125, 624–634 CrossRef CAS.
- H. Raether, Surface Plasmons on Smooth and Rough Surfaces and on Gratings, Springer, 1988 Search PubMed.
- T. Shibata, H. Ikeda, H. Nishiyama, K. Tawa and J. Nishii, Phys. Procedia, 2013, 48, 179–183 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental details and data from characterization of gratings and FullWave Field Simulations. See DOI: 10.1039/c5ra17228d |
| This journal is © The Royal Society of Chemistry 2015 |





