Dual functionalized task specific ionic liquid promoted in situ generation of palladium nanoparticles in water: synergic catalytic system for Suzuki–Miyaura cross coupling†
Jayavant D. Patila,
Suyog N. Koradea,
Supriya A. Patilb,
Dipak S. Gaikwada and
Dattaprasad M. Pore*a
aDepartment of Chemistry, Shivaji University, Kolhapur, 416 004, India. E-mail: p_dattaprasad@rediffmail.com; Fax: +91 231 2692333; Tel: +91 231 2690571
bDepartment of Chemistry, Hanyang University, Sungdong-Ku, Haengdang-dong 17, Seoul, 133-791, Republic of Korea
First published on 9th September 2015
Abstract
Herein we report the synthesis of a novel, dual functionalized task specific ionic liquid possessing cation with hydroxyl functionality which induces reaction in water and acts as a reducing agent as well as an anion with prolinate functionality serves as ligand and stabilizes and/or activates the in situ formed palladium nanoparticles (Pd-Nps) thus act as palladium(0) reservoirs. The generated palladium nanoparticles were characterized by UV-visible spectroscopy, high resolution transmission electron microscopy (HR-TEM), selected area electron diffraction (SAED) and energy-dispersive X-ray spectroscopy (EDX) analysis. These in situ synthesized palladium nanoparticles with particle size between 3 to 9 nm exhibited a high catalytic activity in the Suzuki–Miyaura cross-coupling of aryl halide with aryl boronic acid without using external phosphine ligand. Less activated aryl chlorides also coupled smoothly with aryl boronic acid. It is noteworthy that, isolation of the products were performed by the simple extraction in diethyl ether, and the aqueous system containing ionic liquid along with Pd-NPs was recycled for seven times, without significant loss of catalytic activity.
Introduction
In recent years, the palladium-catalyzed Suzuki–Miyaura cross-coupling has been one of the most studied catalytic reactions due to its aids of functional group compatibility.1 In 1981, biaryls were synthesized by Suzuki–Miyaura cross-coupling of aryl boranes with haloarenes.2 After this discovery, traditionally phosphine ligands along with inert atmosphere and organic solvents were employed for this coupling to avoid the side products formed due to use of phosphine ligands. Pd(0) or Pd(II) derivatives associated with appropriate ligands in conventional organic or biphasic media is basic need of this coupling. In this context, significant innovations have been made through modifications of traditional phosphine ligands and the discoveries of novel phosphine systems,3 but most of the phosphine ligands are sensitive to air and moisture, also their synthesis is laborious. These problems were overcome by exploring non-phosphine alternatives for the Suzuki–Miyaura coupling. A number of phosphine-free ligands have been developed such as palladacycles,4 N-heterocyclic carbenes,5 imines,6 diazabutadienes,7 amines,8 pyrazoles,9 triazoles10 and bipyridine-based systems.11 Although, complexes containing such ligands often show excellent activity, in the majority of cases, the principal drawback is the availability, stability, and cost of the palladium complexes as well as related ligands and in most cases reactions were performed in organic solvents.Transition metal nanoparticles (NPs) have attracted attention and became a best choice over other alternatives in coupling chemistry due to their unique physicochemical properties.12–19 High surface-to-volume ratio of NPs provide larger number of active sites per unit area and show excellent catalytic activity in the cross-coupling. As they are thermodynamically unstable, stabilizers should be chosen to prevent their aggregation. Various non-phosphine stabilizers of palladium NPs, such as surfactants,13 polymers,14 dendrimers,15 PEG16 have been reported. In contrast to the heterogeneous supported metal NPs which are restricted by support surfaces, soluble metal NPs have much superiority, such as controllable size, more rotational degree of freedom and more active sites accessible to reactants. Moreover, the optimum balance between activity and stability of palladium NPs can be easily tuned through the careful choice and modification of ligands, which enhances the scope of NPs for different catalytic applications.20
Recently, innovations in IL have been explored as ILs are coupled with nanotechnology to produce nano-materials with improved catalytic, optical, electrical, magnetic, biological, or biomedical properties.21 Ionic liquids (ILs) have paved the way for alternative and new synthetic strategies, because this methodology allows not only high reaction activity but also the possibility of recycling the catalyst.22 Regrettably, these processes suffer from some limitations viz. use of toxic, expensive and air-sensitive phosphine23 and use sophisticated methodology, such as ultrasound or microwave.24 These limits are of significant concern in large-scale organic syntheses. Moreover, this reaction system requires a large amount of expensive ionic liquid. It would be desirable to reduce the amount of ionic liquid phase from an economic and toxicological viewpoint.25 As a result; the search for new readily available ILs addressing these drawbacks remains an impending goal.
Hydroxyl functionalized ILs possess higher solvent polarities, high dielectric constants which remarkably increases the hydrophilicity of these ILs.26 Anderson group27 suggested that IL cation appending a hydroxyl moiety greatly increases its H-bond acidity with a small increase in H-bond basicity and no change in dipolarity. The Weingartner group28 reported that, hydroxyl-containing ILs exhibited higher dielectric constant than non-functionalized ILs. The Welton group29 suggested that hydroxyl-containing ILs have a higher hydrogen-bond acidity (the value based on the Kamlet–Taft scale) than non-functionalized ILs. Solvatochromic study by Chiappe et al.30 of variety of hydroxyl-functionalized ILs confirm that the introduction of –OH groups in the cation increases polarity. The Dyson group31 reported that, hydroxyl-functionalized ILs act as a reducing agent and formed highly stable, small PdNPs with a narrow size distribution. Amino acid (AA) is one of the most versatile class of natural, bio-renewable, and non-toxic raw materials; it is easy to obtain pure AAs in low cost as well as further advantages of AAs, including biodegradability32 and biological activity33 induce utilization of amino acids (AA) as ionic liquid precursors for designer ionic liquid synthesis. Due to these reasons, amino acid-functionalized ILs were developed as reagents and catalysts to mediate chemical transformations.34 A variety of prolinate functionalized precursors have been used to form organometallic catalysts or as a stabilizer for metal catalyst in organic transformation.35 Hence, there should be an optimum combination of cations and anions for an IL with a specific function that facilitates the solvation of the catalyst, the formation and stabilization of the active catalytic species.
Herein, we report the use of easily accessible, cheap and nontoxic, hydroxyl functionalized TSIL as an excellent promoter in palladium catalyzed Suzuki–Miyaura cross-coupling of aryl halides with arylboronic acids in water.
Results and discussion
Initial attempts were focused on synthesis of novel dual functionalised task specific ionic liquid incorporated cation with hydroxyl functionality which not only induces reaction in water but also act as reducing agent while anion with prolinate functionality serves as ligand and able to stabilize and/or activate the in situ formed palladium nanoparticles thus act as palladium(0) reservoirs. Thus, this ionic liquid can facilitate Suzuki–Miyaura coupling in water and provide an efficient, eco-friendly, recyclable system for said coupling (Fig. 1). | ||
| Fig. 1 Dual functionalized task specific ionic liquid (TSIL): 1-(2-hydroxyethyl)-1-methylpyrrolidinium prolinate [HEMPy][Pro]. | ||
In this perspective, we designed 1-(2-hydroxyethyl)-1-methylpyrrolidinium prolinate [HEMPy][Pro] as a dual functionalized task specific ionic liquid (TSIL). Synthetic route for its synthesis is outlined in Scheme 1. Initially, quaternisation of 1-methylpyrrolidine (1) was carried out with chloroethanol (2) in toluene employing reflux conditions for 24 h to form 3. 3 on metathesis reaction with potassium hydroxide in dichloromethane carried out at room temperature for 24 h furnished 4. Finally, desired TSIL 6 was achieved through acid–base reaction of 4 and L-proline 5 in water initially at 0–5 °C and then stirring at room temperature for 12 h.
The confinement of hydroxyl functionality and prolinate functionality in the TSIL; [HEMPy][Pro] was confirmed by IR spectroscopy (Fig. 2). The IR spectrum of TSIL [HEMPy][Pro] displayed characteristic vibrational bands at 3361 and 3260 cm−1 due to N–H and O–H stretching vibrations, respectively while bands at 1670, 1584 and 774 cm−1 relevant to the carboxylate functionality of prolinate anion. The thermal stability profile of the TSIL; [HEMPy][Pro] was studied using TGA-DSC analysis in the temperature range of 25–400 °C. The TGA-DSC profile is shown in Fig. 3. The initial weight loss of 14.51% may be due to the elimination of water of crystallization or water associated with the ionic liquid through hydrogen bonding. The steep weight loss of 65.29% at temperature range 100–350 °C was mainly attributed to the decomposition of organic groups of TSIL.
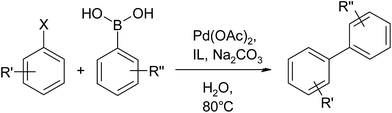
Next, we focused our attention to explore this task specific ionic liquid for Suzuki–Miyaura coupling (Scheme 2). While optimization of reaction conditions, initially screening of ionic liquids was done for model reaction of bromobenzene (1 mmol) and phenylboronic acid (1.2 mmol), base and palladium acetate in water at 80 °C.
Ionic liquids with hydrophilic, hydrophobic, prolinate functionalized and hydroxyl functionalized were employed (Table 1; entries 1–7). Pleasingly, in ionic liquid viz. 1-(2-hydroxyethyl)-1-methylpyrrolidinium prolinate; [HEMPy][Pro] desired biaryl was obtained in high yield (98%) within short time duration (2 h) as compared to other utilized ionic liquids (Table 1, entry 7). When L-proline (20 mol%) was employed without ionic liquid under the identical conditions the desired product was obtained with 50% yield (Table 1; entry 8), while without TSIL; [HEMPy][Pro] only 40% yield of product was obtained (Table 1; entry 9) indicating the influence of ionic liquid on the activity of the coupling reaction. After this success, attempts were then focused on effect of amount of ionic liquid on model reaction, it is found that 20 mol% of [HEMPy][Pro] provided the maximum yield. Exceeding the catalyst quantity than 20 mol% did not affect the yield of product and reaction time (Table 1; entries 10–13).
| Entry | IL | Loading (mol%) | Time (h) | Isolated yield (%) |
|---|---|---|---|---|
| a Reaction conditions: bromobenzene (1 mmol), phenylboronic acid (1.2 mmol), Pd(OAc)2 (2 mol%), IL (mol%), Na2CO3 (1 mmol), water (5 mL), temp = 80 °C. [BMIM] = 1-butyl-3-methylimidazolium, [HEMPy] = 1-(2-hydroxyethyl)-1-methylpyrrolidinium, [Cl] = chloride, [BF4] = tetrafluoroborate, [PF6] = hexafluorophosphate, [Pro] = prolinate. | ||||
| 1 | [BMIM][Cl] | 20 | 2 | 65 |
| 2 | [BMIM][BF4] | 20 | 1 | 76 |
| 3 | [BMIM][Pro] | 20 | 1 | 80 |
| 4 | [HEMPy][Cl] | 20 | 2 | 80 |
| 5 | [HEMPy][BF4] | 20 | 1 | 84 |
| 6 | [HEMPy][PF6] | 20 | 2 | 84 |
| 7 | [HEMPy][Pro] | 20 | 1 | 98 |
| 8 | L-Proline | 20 | 2 | 50 |
| 9 | — | — | 2 | 40 |
| 10 | [HEMPy][Pro] | 10 | 1 | 84 |
| 11 | [HEMPy][Pro] | 15 | 1 | 90 |
| 12 | [HEMPy][Pro] | 25 | 1 | 98 |
| 13 | [HEMPy][Pro] | 30 | 1 | 98 |
Then screening of base for Suzuki–Miyaura coupling was carried out employing Na2CO3, K2CO3, K3PO4, NaOH, KOH, NaOAc, and TEA (Table 2). In the course of this study, it was found that Na2CO3 is the most effective base in terms of the yield (98%) as compared with studied organic and inorganic bases (Table 2, entries 1–7). Further study for effect of amount of base on model reaction revealed that 1 mmol of Na2CO3 was sufficient.
| Entry | Base | Base (mmol) | Time (h) | Yield (%) |
|---|---|---|---|---|
| a Reaction conditions: bromobenzene (1 mmol), phenylboronic acid (1.2 mmol), Pd(OAc)2 (2 mol%), IL (20 mol%), base, water (5 mL), temp = 80 °C. | ||||
| 1 | Na2CO3 | 2 | 1 | 98 |
| 2 | K2CO3 | 2 | 1 | 98 |
| 3 | K3PO4 | 2 | 1 | 96 |
| 4 | NaOH | 2 | 1 | 92 |
| 5 | KOH | 2 | 1 | 94 |
| 6 | NaOAc | 2 | 1 | 84 |
| 7 | TEA | 2 | 1 | 80 |
| 8 | Na2CO3 | 1 | 1 | 98 |
We next investigated suitable palladium source for model reaction by employing Pd/C, Pd(PPh)4, Pd2(dba)3, PdCl2, and Pd(OAc)2 (Table 3, entries 1–5). We observed that simple palladium inorganic salts viz. Pd(OAc)2 and PdCl2 were proved to be the best choice as desired biaryl was obtained in 98% and 86% yield, respectively while Pd/C, Pd2(dba)3, Pd(PPh3)4 were found to be inferior. From further study for effect of amount of Pd(OAc)2 on model reaction revealed that 2 mol% of Pd(OAc)2 was enough.
| Entry | Palladium source | Loading (mol%) | Isolated yield (%) |
|---|---|---|---|
| a Reaction conditions: bromobenzene (1.0 mmol), phenylboronic acid (1.2 mmol), palladium source (mol%), IL (20 mol%), Na2CO3 (1 mmol), water (5.0 mL), temp = 80 °C. | |||
| 1 | Pd/C | 2 | 32 |
| 2 | Pd(PPh)4 | 2 | 55 |
| 3 | Pd2(dba)3 | 2 | 50 |
| 4 | PdCl2 | 2 | 86 |
| 5 | Pd(OAc)2 | 2 | 98 |
| 6 | Pd(OAc)2 | 1.5 | 94 |
| 7 | Pd(OAc)2 | 1 | 82 |
| 8 | Pd(OAc)2 | 0.5 | 78 |
| 9 | Pd(OAc)2 | 3 | 98 |
From these experiments for optimization of reaction conditions, we found optimized condition as TSIL; [HEMPy][Pro] (20 mol%), base; Na2CO3 (1 mmol), Pd(OAc)2 (2 mol%) and water (5 mL). After optimization of reaction conditions, we examined the scope of the substrates to illustrate generality of the method. It was found that aryl iodide, bromide as well as less activated aryl chlorides also get coupled with aryl boronic acid under optimized reaction conditions. A variety of functional groups can be tolerated for aryl halides including methyl, carbonyl, amino, phenyl, formyl and cyano group as well as for aryl boronic acids bearing methoxy, nitro or chloro groups. The results are summarized in Table 4. These results conclude that the electronic characters of aryl halides or aryl boronic acids have no obvious effect on the Suzuki–Miyaura coupling under optimized reaction conditions. It is noticeable that less activated aryl chlorides bearing either electron-donating or electron-withdrawing substituents afford the corresponding products in excellent yields (entries 9–12; Table 4). As one-pot double coupling of aryl 1,4-dihalide is highly valuable for the synthesis of functionalized terphenyls, we proceed to investigate the double coupling of 1-bromo-4-chlorobenzene with phenyl boronic acid under the optimized reaction conditions (entry 12; Table 4). Gratifyingly, terphenyl was obtained in good yield illustrate generality of the method.
| Entry | R′ | X | R′′ | Time (h) | Yieldb (%) |
|---|---|---|---|---|---|
| a Reaction conditions: aryl halide (1 mmol), phenylboronic acid (1.2 mmol), Pd(OAc)2 (2 mol%), IL (20 mol%), Na2CO3 (1 mmol), water (5 mL), temp = 80 °C.b Isolated yield. | |||||
| 1 | H | I | 4-Meo | 1 | 98 |
| 2 | H | Br | H | 1 | 98 |
| 3 | 4-CN | Br | H | 2 | 94 |
| 4 | H | Br | 4-NO2 | 2 | 96 |
| 5 | 4-NH2 | Br | 4-Cl | 1 | 95 |
| 6 | 4-Me | Br | H | 1 | 94 |
| 7 | 4-Ph | Br | 2,4-Cl | 2 | 90 |
| 8 | 4-CHO | Br | H | 3 | 92 |
| 9 | H | Cl | H | 2 | 90 |
| 10 | 4-NH2 | Cl | H | 3 | 86 |
| 11 | 4-Ac | Cl | H | 3 | 80 |
| 12 | 4-Br | Cl | H | 3 | 80 |
After the completion of reaction, we have extracted the reaction mixture by ethyl acetate to separate the synthesized biaryls. Then the aqueous phase have been analysed by recording UV-vis spectrum in acetone medium and compared the same with that of Pd(OAc)2 and ionic liquid (Fig. 4). The absorption at 405 nm due to Pd(OAc)2 disappeared from the reaction mixture due to the formation of palladium nanoparticles. These obtained Pd-Nps is characterized by high resolution transmission electron microscopy (HR-TEM), selected area electron diffraction (SAED) and energy-dispersive X-ray spectroscopy (EDX) analysis as shown in Fig. 5. HR-TEM image, Fig. 5b, shows Pd-Nps having d-spacing 0.651 nm. The selected area electron diffraction (SAED) pattern of the Pd-NPs is shown in Fig. 5c, exhibiting sharp diffraction rings corresponding to the specific crystalline form, confirming the formation of nano-crystalline Pd-NPs. The HRTEM and SAED pattern also indicate that the Pd-NPs are randomly orientated. A surface-averaged Pd-NPs having particle size of 4.4 ± 0.20 nm was derived from the corresponding size histogram Fig. 6a. From the histogram uneven distribution of the Pd-NPs particles exhibited in the range of 3 nm to 9 nm (Fig. 6a). These obtained Pd-Nps with variable size in the range of 3–9 nm are confirmed by high resolution transmission electron microscopy (HR-TEM) analysis (Fig. 6b and c). Thus palladium nanoparticles with high catalytic activity are in situ generated and efficiently stabilized due to the synergetic effect of hydroxyl groups containing cation and the prolinate anion of task specific ionic liquid.
 | ||
| Fig. 6 Histogram of the particle diameters as measured from TEM analysis (b and c) of ∼22 particles shows that the particles are primarily between 3 to 9 nm. | ||
In order to ascertain the catalytic role of in situ formed Pd-NPs, we performed a mercury poisoning experiment using the coupling reaction of 4-bromotoluene (1 mmol) with phenylboronic acid (1.2 mmol) in the presence 1 mol% of Pd(OAc)2, 20 mol% of ionic liquid, Na2CO3 (1 mmol), one drop of mercury and 5 mL of water at 80 °C for 2 h. No product was detected due to formation of Pd(0) amalgam, which exhibits no catalytic activity.
Fig. 7a, shows a fast Fourier transform images for single-Pd-NPs. From the FFT images, the measured lattice spacing of the Pd-NPs 0.672 nm, 0.464 nm, respectively and confirmed that Pd-NPs are crystalline in nature. In the inset of Fig. 7a, the intensity pattern of the Pd-NPs layer is presented. Fig. 7b and c represents the TEM images of Pd-NPs and energy-dispersive spectrometer (EDS) scanning transmission electron microscopy (STEM) mapping images of the same Pd-NPs.
Comparison of catalytic system was carried out in Table 5. Table 5 highlights merits as well as demerits of the literature methods. It is noteworthy that the in situ generated Pd-NPs using TSIL in present method has a comparatively similar/excellent activity as compared to other known Pd catalytic systems. However, from view point of use of water as a universal solvent, cost, easy isolation of product from the reaction mixture, operational simplicity, and reusability developed protocol is superior and easily adaptable.
| Entry | Catalyst | Conditions | Time (h) | Yielda (%) | Ref. |
|---|---|---|---|---|---|
| a Isolated yield.b GC yield. | |||||
| 1 | Pd-NPs | Pd(OAc)2 (2 mol%), TSIL (20 mol%), Na2CO3 (1 mmol), water (5 mL), 80 °C | 1–3 | 98 | This work |
| 2 | Pd(OAc)2 (1–3 mol%) | Na2CO3 (1–3 mmol), 80 °C. H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [BMIM]PF6 = (3 [BMIM]PF6 = (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 g) 3 g) |
2–6 | 99 | 36a |
| 3 | PdCl2 (2 mol%) | [C4-DABCO] [dca] (7.4 mol), 5 M LiCl (0.4 mL), 110 °C. | 3–6 | 90 | 36b |
| 4 | Pd(OAc)2 (5 mol%) | Cs2CO3 (2.0 mmol) PEG1000–[BMIM]PF6 (2.0 g), 80 °C | 2–8 | 95 | 36c |
| 5 | Mag-IL-Pd (0.25–0.50 mol%) | K2CO3 (0.75 mmol), H2O (3 mL), 80 °C, argon atmosphere | 6–12 | 95b | 36d |
| 6 | Pd-NPs | PdCl2 (0.015 mmol) TBAF (1.5 mmol) at 40–100 °C, argon atmosphere | 0.5–12 | 90 | 36e |
| 7 | Pd-NPs | Pd(OAc)2 (2 mol%) K2CO3 (1 mmol), PEG (4 g), 45 °C | 1–5 | 92 | 36f |
| 8 | PdCl2 (0.05–0.5 mol%) | Ligand NaL2 (0.05–0.5 mol%), K3PO4 (2 mmol), H2O (3 mL), 100 °C | 6 | 96 | 36g |
| 9 | Pd-NPs | PdCl2 (1.2 mol%), IL (1.0 mL), Na2CO3 (1.05 mmol), water (0.5 mL), 110 °C | 4 | 96b | 36h |
| 10 | Zwitterionic palladium complexes (2.0 mol%) | KOH (2.0 equiv.), 1,4-dixoxane/H2O (4/1, 2.0 mL), RT | 2–12 | 88b | 36i |
| 11 | Pd(NHC) complexes (1 mol%) | K2CO3 (1.5 mmol), H2O (5 mL), 100 °C | 4–12 | 99b | 36j |
| 12 | Palladium nanocomposite xerogel (0.15 mol% Pd) | K2CO3 (0.98 mmol), H2O (1.0 mL), 90 °C | 12 | 90 | 36k |
| 13 | Pd-NPs | NaOAc (2 mmol), DMF/H2O (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v, 6 mL), 80 °C 1, v/v, 6 mL), 80 °C |
1–3 | 95 | 36l |
| 14 | Pd(acac)2 (2–3 mg) | KOH (2 mmol), ethanol (2 mL), 70 °C, nitrogen atmosphere | 0.5–4 | 99b | 36m |
In coupling chemistry, the recycle or reusability of the catalytic system is an important aspect from economical point of view. Hence, last attempt was focused on reusability of the catalytic system. As the products are soluble in ethyl acetate, and can be readily separated by extraction from the aqueous catalytic system, the remaining catalytic system can be recycled for fresh cross-coupling of aryl boronic acid with aryl halide. The reusability study of the catalytic system was carried out with the model cross-coupling of 4-bromotoluene (1 mmol), phenyl boronic acid (1.2 mmol), Na2CO3 (1 mmol), Pd(OAc)2 (2 mol%), and IL (20 mol%) in water (5 mL) at 80 °C for 2 h. After completion of reaction, the mixture was cooled to room temperature and extracted by ethyl acetate (2 × 10 mL) upper organic phase containing the product was easily separated using simple liquid–liquid extraction. The residual aqueous catalytic phase containing the IL-Pd-NPs was reused for the next run under the same reaction conditions and found reusable for 7 subsequent runs without any loss of catalytic activity (Fig. 8). Thus the use of this catalytic system meets the trend of green chemistry. HR-TEM analysis of Pd-Nps after seventh recycle is as shown in Fig. 9.
Conclusions
A dual functionalized task specific ionic liquid, 1-(2-hydroxyethyl)-1-methylpyrrolidinium prolinate was designed and synthesized from inexpensive and commercially available precursors. Palladium nanoparticles were in situ formed and synergistically stabilized, due to the hydroxyl functionality in cation and the prolinate anion of task specific ionic liquid. 1-(2-Hydroxyethyl)-1-methylpyrrolidinium prolinate provided excellent catalytic activity towards the Suzuki–Miyaura cross-coupling of arylboronic acid with aryl halide in water. Noticeably, less activated aryl chlorides also coupled efficiently. TEM analysis, mercury drop test showed Pd-Nps were involved in catalysis. The ionic ligand served as an efficient protective agent for the stabilization of palladium NPs in water. The catalytic system can also be reused several times and has potential for industrial applications.Experimental
General information
Phenylboronic acids (Spectrochem, Mumbai) aryl halides (Sigma Aldrich, India) and palladium sources (Sigma Aldrich, India) were used as received. All reactions were carried out aerobic condition in predried glassware. Infrared spectra were recorded on a Perkin-Elmer FT-IR-783 spectrophotometer. 1H NMR and 13C NMR spectra were recorded on a Bruker AC spectrometer (300 MHz for 1H NMR and 75 MHz for 13C NMR), using CDCl3 as solvent and tetramethylsilane (TMS) as an internal standard. Chemical shifts (δ) are expressed in parts per million (ppm) and coupling constants are expressed in hertz (Hz). Mass spectra were recorded on a Shimadzu QP2010 GCMS. HR-TEM, FFT were taken on TEM 2100F, JEOL. Elemental analyses were performed on a EURO EA3000 vector model. The thermal gravimetric analysis (TGA) curves were obtained using the instrument TA SDT Q600 in the presence of static air at a linear heating rate of 10 °C min−1 from 25 °C to 400 °C.Typical procedure for the preparation of 1-(2-hydroxyethyl)-1-methylpyrrolidinium prolinate [HEMPy][Pro] ionic liquid
To a vigorously stirred solution of 1-methylpyrrolidine (10 mmol) in toluene (25 mL), 2-chloroethanol (12 mmol) was slowly added at room temperature and quaternisation was carried out at 80 °C for 24 h, after which it was placed in a freezer at 0 °C for 4 h. Toluene was decanted and the remaining viscous oil was repeatedly washed with diethyl ether to yield white viscous ionic liquid, which was dried in vacuum furnished 1-(2-hydroxylethyl)-1-methylpyrrolidinium chloride; [HEMPy][Cl]. 1-(2-Hydroxylethyl)-1-methylpyrrolidinium chloride (10 mmol) was then dissolved in dichloromethane followed by addition of potassium hydroxide (12 mmol) and stirred it for 24 h at room temperature. The suspension was filtered to remove the precipitated potassium chloride salt and the solvent was evaporated under reduced pressure furnished 1-(2-hydroxylethyl)-1-methylpyrrolidine hydroxide; [HEMPy][OH]. Finally, 1-(2-hydroxylethyl)-1-methylpyrrolidine hydroxide (5 mmol) was dissolved in water and L-proline (5 mmol) was added slowly at 0–5 °C after addition it was stirred at room temperature for 12 h. The solvent was removed in vacuum to furnish desire ionic liquid viz. 1-hydroxylethyl-1-methylpyrrolidine prolinate; [HEMPy][Pro].General procedure for the Suzuki–Miyaura coupling
To a mixture of aryl halide (1 mmol), arylboronic acid (1.2 mmol), Pd(OAc)2 (2 mol%), IL (20 mol%) and Na2CO3 (1 mmol), water (5 mL) was added and stirred at 80 °C under aerobic conditions. After completion of reaction (monitored by TLC), the reaction mixture was cooled. The product was extracted with ethyl acetate (2 × 10 mL) while the IL-Pd-Nps were remained in aqueous phase. The aqueous phase containing IL-Pd-NPs washed with ethyl acetate. The organic layer was dried with MgSO4 and concentrated on a rotary evaporator to afford the desired biaryl in excellent yield. The crude products were then purified by column chromatography (5% ethyl acetate). Synthesized products were confirmed from physical constant,37 IR, 1H, 13C NMR and mass analysis.General procedure for the reusability of catalytic system in the Suzuki–Miyaura coupling
A mixture of 4-bromotoluene (1 mmol), phenylboronic acid (1.2 mmol), Na2CO3 (1 mmol), Pd(OAc)2 (2 mol%), and IL (20 mol%) in water (5 mL) was stirred at 80 °C under aerobic condition. After completion of reaction, reaction mixture was cooled to room temperature and extracted by ethyl acetate (2 × 10 mL). The combined organic layer was concentrated under reduced pressure, and the residue was isolated by chromatography on a silica gel (300–400 mesh) column using petroleum and ethyl acetate to afford the product. The aqueous phase containing the IL-Pd-Nps were loaded with the fresh reactants and base, for the next run under the same reaction conditions. The aqueous catalytic system was reused for 7 subsequent runs without any loss of catalytic activity.Acknowledgements
We gratefully acknowledge DST New Delhi for financial assistance under Fast Track Scheme [No. SB/FT/CS-154/2012].Notes and references
- (a) A. Suzuki, Angew. Chem., Int. Ed., 2011, 50, 6722 CrossRef CAS PubMed; (b) N. Miyaura and A. Suzuki, Chem. Rev., 1995, 95, 2457 CrossRef CAS; (c) A. Suzuki and Y. Yamamoto, Chem. Lett., 2011, 40, 894 CrossRef CAS; (d) F. A. Littke and G. C. Fu, Angew. Chem., Int. Ed., 2002, 41, 4176 CrossRef; (e) J. Dupont, C. S. Consorti and J. Spencer, Chem. Rev., 2005, 105, 2527 CrossRef CAS PubMed; (f) A. Balanta, C. Godard and C. Claver, Chem. Soc. Rev., 2011, 40, 4973 RSC; (g) A. Fihri, M. Bouhrara, B. Nekoueishahraki, J.-M. Basset and V. Polshettiwar, Chem. Soc. Rev., 2011, 40, 5181 RSC; (h) J. D. Sellars and P. G. Steel, Chem. Soc. Rev., 2011, 40, 5170 RSC.
- N. Miyaura, T. Yanagi and A. Suzuki, Synth. Commun., 1981, 11, 513 CrossRef CAS PubMed.
- (a) R. B. Bedford, C. S. J. Cazin and D. Holder, Coord. Chem. Rev., 2004, 248, 2283 CrossRef CAS PubMed; (b) F. E. Hahn and M. C. Jahnke, Angew. Chem., Int. Ed., 2008, 47, 3122 CrossRef CAS PubMed; (c) P. L. Arnold and S. Pearson, Coord. Chem. Rev., 2007, 251, 596 CrossRef CAS PubMed; (d) A. F. Christoph and H. Plenio, Green Chem., 2007, 9, 1287 RSC; (e) N. Jiang and A. J. Ragauskas, Tetrahedron Lett., 2006, 47, 197 CrossRef CAS PubMed; (f) K. H. Shaughnessy, Chem. Rev., 2009, 109, 643 CrossRef CAS PubMed; (g) D. J. M. Snelders, R. Kreiter, J. Firet, G. van Koten and R. J. M. K. Gebbink, Adv. Synth. Catal., 2008, 350, 262 CrossRef CAS PubMed; (h) R. Huang and K. H. Shaughnessy, Chem. Commun., 2005, 4484 RSC; (i) M. Lombardo, M. Chiarucci and C. Trombini, Green Chem., 2009, 11, 574 RSC.
- (a) I. D. Kostas, F. J. Andreadaki, D. Kovala-Demertzi, C. Prentjas and M. A. Demertzis, Tetrahedron Lett., 2005, 46, 1967 CrossRef CAS PubMed; (b) J. L. Zhang, L. Zhao, M. P. Song, T. C. W. Mak and Y. J. Wu, J. Organomet. Chem., 2006, 691, 1301 CrossRef CAS PubMed; (c) J. Ma, X. L. Cui, B. Zhang, M. P. Song and Y. J. Wu, Tetrahedron, 2007, 63, 5529 CrossRef CAS PubMed.
- (a) C. Zhang, J. Huang, M. L. Trudell and S. P. Nolan, J. Org. Chem., 1999, 64, 3804 CrossRef CAS; (b) C. W. K. Gstottmayr, V. P. W. Bohm, E. Herdtweck, M. Grosche and W. Herrmann, Angew. Chem., Int. Ed., 2002, 41, 1363 CrossRef CAS; (c) D. M. Pore, D. S. Gaikwad and J. D. Patil, Monatsh. Chem., 2013, 144, 1355 CrossRef; (d) Z. Wang, Y. Yu, Y. X. Zhang, S. Z. Li, H. Qian and Z. Y. Lin, Green Chem., 2015, 17, 413 RSC.
- (a) H. Weissman and D. Milstein, Chem. Commun., 1999, 1901 RSC; (b) R. B. Bedford and C. S. J. Cazin, Chem. Commun., 2001, 1540 RSC; (c) K. M. Wu, C. A. Huang, K. F. Peng and C. T. Chen, Tetrahedron, 2005, 61, 9679 CrossRef CAS PubMed.
- (a) G. A. Grasa, A. C. Hillier and S. P. Nolan, Org. Lett., 2001, 3, 1077 CrossRef CAS PubMed; (b) C. Zhou, J. Wang, L. Li, R. Wang and M. Hong, Green Chem., 2011, 13, 2100 RSC.
- (a) J. H. Li and W. J. Liu, Org. Lett., 2004, 6, 2809 CrossRef CAS PubMed; (b) J. H. Li, W. J. Liu and Y. X. Xie, J. Org. Chem., 2005, 70, 5409 CrossRef CAS PubMed; (c) H. Yi, J. B. Liu, Q. Li and J. Tang, Chin. Chem. Lett., 2005, 16, 1173 CAS; (d) Y. X. Xie, J. H. Li and D. L. Yin, Chin. J. Org. Chem., 2006, 26, 1155 CAS.
- (a) A. Mukherjee and A. Sarkar, Tetrahedron Lett., 2005, 46, 15 CrossRef CAS PubMed; (b) A. d. Leon, M. Guerrero, J. Garcıa-Anton, J. Ros, M. F. Bardı and J. Pons, CrystEngComm, 2013, 15, 1762 RSC.
- (a) G. Zhang, Y. Luan, X. Han, Y. Wang, X. Wen, C. Dingand and J. Gao, Green Chem., 2013, 15, 2081 RSC; (b) C. Shen, H. Shen, M. Yang, C. Xia and P. Zhang, Green Chem., 2015, 17, 225 RSC.
- (a) T. Yoshidomi, Y. Segawa and K. Itam, Chem. Commun., 2013, 49, 5648 RSC; (b) C.-M. Hsu, C.-B. Li and C.-H. Sun, J. Chin. Chem. Soc., 2009, 56, 873 CrossRef CAS PubMed.
- (a) A. Corma and H. Garcıa, Chem. Soc. Rev., 2008, 37, 2096 RSC; (b) L. Starkey Ott and R. G. Finke, Coord. Chem. Rev., 2007, 251, 1075 CrossRef PubMed; (c) D. Astruc, F. Lu and J. R. Aranzaes, Angew. Chem., Int. Ed., 2005, 44, 7852 CrossRef CAS PubMed; (d) M. Moreno-Manas and R. Pleixats, Acc. Chem. Res., 2003, 36, 638 CrossRef CAS PubMed.
- (a) D. Saha, K. Chattopadhyay and B. C. Ranu, Tetrahedron Lett., 2009, 50, 1003 CrossRef CAS PubMed; (b) S. Xu and Q. Yang, J. Phys. Chem. C, 2008, 112, 13419 CrossRef CAS; (c) M. Shiri and M. A. Zolfigol, Tetrahedron, 2009, 65, 587 CrossRef CAS PubMed; (d) R. Liu, C. Wu, Q. Wang, J. Ming, Y. Hao, Y. Yu and F. Zhao, Green Chem., 2009, 11, 979 RSC; (e) D. S. Gaikwad and D. M. Pore, Synlett, 2012, 23, 2631 CrossRef CAS; (f) G. Zhang, H. Zhou, J. Hu, M. Liua and Y. Kuang, Green Chem., 2009, 11, 1428 RSC; (g) R. Zhong, A. Pöthig, Y. Feng, K. Riener, W. A. Herrmann and F. E. Kühn, Green Chem., 2014, 16, 4955 RSC.
- (a) C. Ornelas, A. K. Diallo, J. Ruiz and D. Astruc, Adv. Synth. Catal., 2009, 351, 2147 CrossRef CAS PubMed; (b) H. Oyamada, R. Akiyama, H. Hagio, T. Naito and S. Kobayashi, Chem. Commun., 2006, 4297 RSC; (c) B. P. S. Chauhan, J. S. Rathore and T. Bandoo, J. Am. Chem. Soc., 2004, 126, 8493 CrossRef CAS PubMed; (d) H. Veisi, A. A. Manesh, N. Eivazi and A. R. Faraji, RSC Adv., 2015, 5, 20098 RSC; (e) R. L. Oliveira, W. He, R. J. M. K. Gebbink and K. P. de Jong, Catal. Sci. Technol., 2015, 5, 1919 RSC; (f) J. Yang, D. Wang, W. Liu, X. Zhang, F. Bian and W. Yu, Green Chem., 2013, 15, 3429 RSC; (g) H. P. Hemantha and V. V. Sureshbabu, Org. Biomol. Chem., 2011, 9, 2597 RSC.
- (a) K. R. Gopidas, J. K. Whitesell and M. A. Fox, Nano Lett., 2003, 3, 1757 CrossRef CAS; (b) C. Ornelas, L. Salmon, J. R. Aranzaes and D. Astruc, Chem. Commun., 2007, 4946 RSC; (c) L. Wu, B. L. Li, Y. Y. Huang, H. F. Zhou, Y. M. He and Q. H. Fan, Org. Lett., 2006, 8, 3605 CrossRef CAS PubMed; (d) T. R. Hendricks, E. E. Dams, S. T. Wensing and I. Lee, Langmuir, 2007, 23, 7404 CrossRef CAS PubMed; (e) A. Garcia-Bernabe, C. C. Tzchucke, W. Bannwarth and R. Haag, Adv. Synth. Catal., 2005, 347, 1389 CrossRef CAS PubMed; (f) M. Qi, P. Z. Tan, F. Xue, H. S. Malhi, Z.-X. Zhang, D. J. Young and T. S. Andy Hor, RSC Adv., 2015, 5, 3590 RSC.
- (a) S. Sawoo, D. Srimani, P. Dutta, R. Lahiri and A. Sarkar, Tetrahedron, 2009, 65, 4367 CrossRef CAS PubMed; (b) B. Feng, Z. Hou, H. Yang, X. Wang, Y. Hu, H. Li, Y. Qiao, X. Zhao and Q. Huang, Langmuir, 2010, 26, 2505 CrossRef CAS PubMed; (c) X. Wang, H. Yang, B. Feng, Z. Hou, Y. Hu, Y. Qiao, H. Li and X. Zhao, Catal. Lett., 2009, 132, 34 CrossRef CAS; (d) W. Han, N. Liu, C. Liu and Z. L. Jin, Chin. Chem. Lett., 2010, 21, 1411 CrossRef CAS PubMed; (e) N. Mejıas, A. Serra-Muns, R. Pleixats, A. Shafir and M. Tristany, Dalton Trans., 2009, 7748 RSC.
- (a) A. Corma, H. Garcıa and A. Leyva, J. Catal., 2006, 240, 87 CrossRef CAS PubMed; (b) Z. Hou, N. Theyssen and W. Leitner, Green Chem., 2007, 9, 127 RSC; (c) D. Srimani, S. Sawoo and A. Sarkar, Org. Lett., 2007, 9, 3639 CrossRef CAS PubMed.
- (a) N. Mejıas, R. Pleixats, A. Shafir, M. Medio-Simon and G. Asensio, Eur. J. Org. Chem., 2010, 5090 CrossRef PubMed.
- (a) R. Wang, J. C. Xiao, B. Twamley and J. M. Shreeve, Org. Biomol. Chem., 2007, 5, 671 RSC; (b) R. Wang, M. M. Piekarski and J. M. Shreeve, Org. Biomol. Chem., 2006, 4, 1878 RSC.
- N. Yan, Y. Yuan and P. J. Dyson, Dalton Trans., 2013, 42, 13294 RSC.
- (a) N. Yan, C. Xiao and Y. Kou, Coord. Chem. Rev., 2010, 254, 1179 CrossRef CAS PubMed; (b) Z. Li, Z. Jia, Y. Luan and T. Mu, Curr. Opin. Solid State Mater. Sci., 2008, 12, 1 CrossRef CAS PubMed.
- (a) R. Sheldon, Chem. Commun., 2001, 2399 RSC; (b) J. Dupont, R. F. D. Souza and P. A. Z. Suarez, Chem. Rev., 2002, 102, 3667 CrossRef CAS PubMed; (c) P. Wasserscheid and W. Keim, Angew. Chem., Int. Ed., 2000, 39, 3772 CrossRef CAS; (d) H. Hagiwara, K. Sato, T. Hoshi and T. Suzuki, Synlett, 2011, 2545 CrossRef CAS.
- (a) J. Mathews, P. J. Smith and T. Welton, Chem. Commun., 2000, 1249 RSC; (b) H. Yang, C. C. Tai, Y. T. Huang and I. W. Sun, Tetrahedron, 2005, 61, 4857 CrossRef PubMed; (c) M. M. Manas, R. Pleixats and A. S. Muns, Synlett, 2006, 3001 CrossRef.
- (a) Z. Y. Du, W. W. Zhou, L. Bai, F. Wang and J. X. Wang, Synlett, 2011, 369 CrossRef CAS; (b) K. S. A. Vallin, P. Emilsson, M. Larhed and A. Hallberg, J. Org. Chem., 2002, 67, 6243 CrossRef CAS PubMed; (c) R. Hajipour, K. Karami and F. Rafiee, Appl. Organomet. Chem., 2012, 26, 27 CrossRef PubMed.
- (a) A. Taher, J.-B. Kim, J.-Y. Jung, W.-S. Ahn and M.-J. Jin, Synlett, 2009, 2477 CAS; (b) R. H. Wang, B. Twamley and J. M. Shreeve, J. Org. Chem., 2006, 71, 426 CrossRef CAS PubMed; (c) J. D. Revell and A. Ganesan, Org. Lett., 2002, 4, 3071 CrossRef CAS PubMed.
- S. Tang, G. A. Baker and H. Zhao, Chem. Soc. Rev., 2012, 41, 4030 RSC.
- (a) P. Twu, Q. Zhao, W. R. Pitner, W. E. Acree, G. A. Baker and J. L. Anderson, J. Chromatogr., A, 2011, 1218, 5311 CrossRef CAS PubMed; (b) Q. Zhao, J. Eichhorn, W. R. Pitner and J. L. Anderson, Anal. Bioanal. Chem., 2009, 395, 225 CrossRef CAS PubMed.
- M. M. Huang, Y. Jiang, P. Sasisanker, G. W. Driver and H. Weingartner, J. Chem. Eng. Data, 2011, 56, 1494 CrossRef CAS.
- M. A. Ab Rani, A. Brant, L. Crowhurst, A. Dolan, M. Lui, N. H. Hassan, J. P. Hallett, P. A. Hunt, H. Niedermeyer, J. M. Perez-Arlandis, M. Schrems, T. Welton and R. Wilding, Phys. Chem. Chem. Phys., 2011, 13, 16831 RSC.
- C. Chiappe, C. S. Pomelli and S. Rajamani, J. Phys. Chem. B, 2011, 115, 9653 CrossRef CAS PubMed.
- (a) N. Yan, X. Yang, Z. Fei, Y. Li, Y. Kou and P. J. Dyson, Organometallics, 2009, 28, 937 CrossRef CAS; (b) X. Yuan, N. Yan, S. A. Katsyuba, E. E. Zvereva, Y. Kou and P. J. Dyson, Phys. Chem. Chem. Phys., 2012, 14, 6026 RSC; (c) X. Yang, N. Yan, Z. Fei, R. M. Crespo-Quesada, G. Laurenczy, L. Kiwi-Minsker, Y. Kou, Y. Li and P. J. Dyson, Inorg. Chem., 2008, 47, 7444 CrossRef CAS PubMed.
- N. Gathergood, M. T. Garcia and P. J. Scammells, Green Chem., 2004, 6, 166 RSC.
- K. M. Docherty and C. F. Kulpa Jr, Green Chem., 2005, 7, 185 RSC.
- (a) P. Moriel, E. J. García-Suárez, M. Martínez, A. B. García, M. A. Montes-Morán, V. Calvino-Casilda and M. A. Bañares, Tetrahedron Lett., 2010, 51, 4877 CrossRef CAS PubMed; (b) N. A. Larionova, A. S. Kucherenko, D. E. Siyutkin and S. G. Zlotin, Tetrahedron, 2011, 67, 1948 CrossRef CAS PubMed; (c) N. Ferlin, M. Courty, A. N. Van Nhien, S. Gatard, M. Pour, B. Quilty, M. Ghavre, A. Haiß, K. Kümmerer, N. Gathergood and S. Bouquillon, RSC Adv., 2013, 3, 26241 RSC; (d) K. R. Roshan, T. Jose, D. Kim, K. A. Cherian and D. W. Park, Catal. Sci. Technol., 2014, 4, 963 RSC.
- (a) A. Morel, E. Silarska, A. M. Trzeciak and J. Pernak, Dalton Trans., 2013, 42, 1215 RSC; (b) J. Yan and L. Wang, Synthesis, 2010, 447 CAS; (c) W. Liu and F. Hou, Appl. Organomet. Chem., 2015, 29, 368 CrossRef CAS PubMed; (d) K. Azizi and A. Heydari, RSC Adv., 2014, 4, 6508 RSC; (e) G. Zhang, Y. Luan, X. Han, Y. Wang, X. Wen and C. Ding, Appl. Organomet. Chem., 2014, 28, 332 CrossRef CAS PubMed; (f) A. R. Hajipour, E. Boostania and F. Mohammadsaleh, RSC Adv., 2015, 5, 24742 RSC; (g) K. Rajender Reddy, C. V. Rajasekhar and G. Gopi Krishna, Synth. Commun., 2007, 37, 1971 CrossRef PubMed; (h) M. M. Heravi, M. H. Tehrani, K. Bakhtiari and H. A. Oskooie, Catal. Commun., 2007, 8, 1341 CrossRef CAS PubMed.
- (a) L. Liu, Y. Dong, B. Pang and J. Ma, J. Org. Chem., 2014, 79, 7193 CrossRef CAS PubMed; (b) P. R. Boruah, M. J. Koiri, U. Bora and D. Sarma, Tetrahedron Lett., 2014, 55, 2423 CrossRef CAS PubMed; (c) X. Liu, Y. Mao and M. Lu, Appl. Organomet. Chem., 2012, 26, 305 CrossRef CAS PubMed; (d) B. Karimi, F. Mansouria and H. Valib, Green Chem., 2014, 16, 2587 RSC; (e) L. Tao, Y. Xie, C. Deng and J. Li, Chin. J. Chem., 2009, 27, 1365 CrossRef CAS PubMed; (f) W. Han, C. Liu and Z.-L. Jin, Org. Lett., 2007, 9, 4005 CrossRef CAS PubMed; (g) Q. Lia, L.-M. Zhang, J.-J. boa, H.-X. Li, J.-B. Xie and J.-P. Lang, Appl. Organomet. Chem., 2014, 28, 861 CrossRef PubMed; (h) Y. Cui, I. Biondi, M. Chaubey, X. Yang, Z. Fei, R. Scopelliti, C. G. Hartinger, Y. Li, C. Chiappe and P. J. Dyson, Phys. Chem. Chem. Phys., 2010, 12, 1834 RSC; (i) J.-Y. Lee, D. Ghosh, J.-Y. Lee, S.-S. Wu, C.-H. Hu, S.-D. Liu and H. M. Lee, Organometallics, 2014, 33, 6481 CrossRef CAS; (j) F. Godoy, C. Segarra, M. Poyatos and E. Peris, Organometallics, 2011, 30, 684 CrossRef CAS; (k) M. Maity and U. Maitra, J. Mater. Chem. A, 2014, 2, 18952 RSC; (l) S. Liu, Q. Zhou and H. Jiang, Chin. J. Chem., 2010, 28, 589 CrossRef CAS PubMed; (m) B. Luo, J. Q. Wang, D. H. Ge, X. M. Li, X. Q. Cao, Y. Pan and H. W. Gu, Sci. China: Chem., 2014, 57, 1310 CrossRef CAS.
- (a) N. S. Nandurkar and B. M. Bhanage, Tetrahedron, 2008, 64, 3655 CrossRef CAS PubMed; (b) C. Desmarets, R. O. Amrani, A. Walcarius, J. Lambert, B. Champagne, Y. Fort and R. Schneider, Tetrahedron, 2008, 64, 372 CrossRef CAS PubMed; (c) S. J. Sabounchei and A. Hashemi, Inorg. Chem. Commun., 2014, 47, 123 CrossRef CAS PubMed; (d) S. J. Hoseini, V. Heidari and H. Nasrabadi, J. Mol. Catal. A: Chem., 2015, 396, 90 CrossRef CAS PubMed; (e) R. Huang and K. H. Shaughnessy, Organometallics, 2006, 25, 4105 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra17186e |
| This journal is © The Royal Society of Chemistry 2015 |