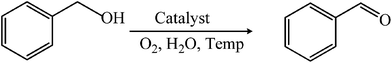
A novel iron(III)-based heterogeneous catalyst for aqueous oxidation of alcohols using molecular oxygen†
Debojeet Sahua,
Ana Rosa Silva b and
Pankaj Das*a
b and
Pankaj Das*a
aDepartment of Chemistry, Dibrugarh University, Dibrugarh, Assam, India. E-mail: pankajd29@yahoo.com; Fax: +91-373-2370323; Tel: +91-373-2370210
bCICECO-Aveiro Institute of Materials, University of Aveiro, Aveiro, Portugal
First published on 7th September 2015
Abstract
A novel silica supported iron(III)-based heterogeneous catalyst (FeCl3-imine@SiO2) has been synthesized and characterized by several techniques, such as FTIR, nitrogen adsorption analysis (BET), SEM-EDX, XRD, EPR, AAS and XPS analyses. The catalyst showed excellent activity for selective oxidation of alcohols to corresponding carbonyl compounds in water under atmospheric pressure of oxygen without using any additive. Notably, the heterogeneous catalyst showed remarkable improvement over its homogeneous counterpart, which might be attributable to the synergic effect between the metal and the supported ligand. The catalyst could be easily recovered by simple filtration and reused several times without compromising its activity. A wide range of primary and secondary alcohols including heterocyclic alcohols are tolerated as substrates.
Introduction
Selective oxidation of alcohols to corresponding carbonyl compounds is one of the most industrially significant reactions in organic synthesis. The use of transition metal-based homogeneous1 and heterogeneous2 catalysts is a good precedent for this reaction mainly because of their ability to replace conventional oxidation methods of using stoichiometric oxidants such as chromate or permanganate with molecular oxygen. From both economic and green chemistry perspectives, molecular oxygen is an ideal oxidant as it is readily available and produces water as the only by-product.1,3 Unfortunately, majority of the reported protocols for efficient oxidation of alcohols using molecular oxygen require noble metals such as palladium,4 rhodium,5 ruthenium,6 gold,7 platinum,8 etc. Apart from their costs, these noble metals also have serious toxicity issues which make these catalytic systems impractical for industrial use.1d Hence, over the years, considerable efforts have been made towards replacement of these expensive precious metal catalysts with cheaper base metals like copper or iron.1a–c Although, copper-based systems already displayed tremendous potential to act as an alternate to platinum metal catalysts in oxidation of alcohols,9 iron-based systems remain relatively underexplored. It is fair to say that from sustainability point of view iron is in much more advantageous position over copper due to its lower price, and high environmental acceptability.10 It is surprising to note that the first iron-catalyzed oxidation of alcohols was reported only in 2002,11 while copper-catalyzed alcohol oxidation was first reported in 1966.12 Since the first report, iron-catalyzed systems have been increasingly investigated in alcohol oxidation studies.13,14 Typical examples are Wang's FeCl3–TEMPO–NaNO2 system,13a Repo's FeCl3–thyamine system,13b Liu's Fe(NO3)–TEMPO–NaCl system,13c,d etc. However, majority of these successful systems are homogeneous in nature where separation and recovery of the catalyst is extremely difficult which pose significant limitations to their industrial scale applications. Thus, in recent years, strategies have been adopted to immobilize iron(III) salts/compounds into/onto different supports such as aluminosilicates,14b carbon,14c mesoporous silica,14i graphene-oxide,14g polymer,14e etc. and applied in alcohol oxidation reaction. Although notable progress was achieved in some cases with respect to development of stable and reusable catalysts of high efficiency, such catalysts usually showed lower activities compared to their homogeneous counterparts. Moreover, except one report of iron-oxide nanoparticles as catalyst,14b in all the cases the reactions were performed in organic solvents that are not only expensive but also toxic in nature.In recent years, because of economical and environmental issues, organic reactions in water have gained much attention. However, to the best of our knowledge, there is no report on the use of iron-based heterogeneous catalysts for alcohol oxidation reaction in water with molecular oxygen as terminal oxidant. In this work, we disclose a simple procedure for the preparation of a new iron(III)-based heterogeneous catalyst supported on silica and its application for alcohol oxidation reaction with molecular oxygen in neat water.
Experimental section
General information
3-Aminopropyl-functionalized silica gel (APTES@SiO2: 1 mmol g−1 of silica gel; 200–400 mesh) was purchased from Sigma-Aldrich. Acetamide and anhydrous FeCl3 was purchased from Thermo Fisher scientific. The substrates used in the alcohol oxidation reactions were purchased from Sigma-Aldrich, Spectrochem and Rankem. All other chemicals and solvents were purchased from different Indian firms. The solvents were distilled and dried prior to use.The Fourier transform infrared (FTIR) spectra (KBr; 1800–1400 cm−1) of the materials were recorded on a Shimadzu IR spectrophotometer (prestige-21). X-ray powder diffraction (XRD) was performed with a Bruker diffractometer (model AXS D8) with Cu-Kα (1.541 °A) radiation. The scanning electron microscope (SEM) images were obtained from JEOL Model JSM - 6390LV and energy dispersive X-ray (EDX) peaks were recorded in JEOL Model JED – 2300. The iron content was determined by atomic absorption spectroscopy (AAS) using a Thermo Scientific spectro photometer (IEC 3000). Electron spin resonance (ESR) spectra was recorded on a JEOL instrument (JES-FA200) at liquid N2 temperature. Nitrogen adsorption studies were carried out at 77 K using a Quantachrome, ASIQA 2010 analyzer. Prior to analysis the samples were degassed by heating at 200 °C for 4 h under vacuum. Specific surface area was calculated following the Brunauer–Emmett–Teller (BET) calculation and pore-size distribution was obtained by using the Barrett–Joyner–Halenda (BJH) method. X-ray photoelectron spectroscopy (XPS) was performed at “Centro de Materiais da Universidade do Porto” (Portugal), in a Kratos Axis Ultra HSA spectrometer using a non-monochromatized Mg-Kα radiation (1253.6 eV). Gas chromatography-mass spectrometry (GC-MS) studies were done using Agilent 7820A GC system with Agilent 5975 Series MSD Mass detector equipped with a 30 m × 0.25 mm × 0.5 μm HP-5MS capillary column.
Synthesis of the silica based materials
General procedure for the alcohol oxidation reactions
A 50 mL two necked round bottomed flask fitted with a condenser and gas bubbler was charged with alcohol (1 mmol), catalyst (46 mg, 2 mol% Fe) and water (6 mL). Then O2 (1 atm.) was bubbled into the mixture for the required time and the reaction mixture was heated at 80 °C with continuous stirring. On completion of the reaction, the catalyst was separated from the reaction mixture by simple filtration. The filtrate was extracted with ether and dried over Na2SO4. After evaporation of the solvent under reduced pressure the concentrated organic product is then extracted with ether and analyzed by GC-MS. The product was isolated by column chromatography technique using ethyl acetate/hexane (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9) as eluent.
9) as eluent.
Results and discussion
Synthesis and characterization of the catalyst
The catalyst, FeCl3-imine@SiO2, has been prepared in two steps (Scheme 1). Firstly, commercially available 3-aminopropyltriethoxysilane functionalized silica gel (APTES@SiO2) underwent a condensation reaction with acetamide to get an imine-functionalized silica gel (imine@SiO2) which subsequently treated with anhydrous FeCl3 to get the iron-catalyst. The FTIR spectra (Fig. S1, ESI†) of the silica-based material are in good agreement with the proposed coordination environment. In particular, the bidentate bonding mode of the supported ligand in FeCl3-imine@SiO2 was confirmed by the shift of νCH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N band from 1646 cm−1 to 1630 cm−1 and νNH2 band from 1531 cm−1 to 1511 cm−1. The nitrogen adsorption–desorption isotherms of the silica-based materials displayed typical type IV patterns with a sharp hysteresis loop, which is characteristics of mesoporous materials (Fig. 1). The surface area and pore size distribution of the support (APTES@SiO2) studies reveals a high specific surface area of about 416 m2 g−1 with specific pore volume of 0.65 cm3 g−1 (Table S1, ESI†). These values upon metal immobilization (FeCl3-imine@SiO2) decreases significantly to 283 m2 g−1 and 0.43 cm3 g−1 which is consistent with successful immobilization of iron onto silica matrix.
N band from 1646 cm−1 to 1630 cm−1 and νNH2 band from 1531 cm−1 to 1511 cm−1. The nitrogen adsorption–desorption isotherms of the silica-based materials displayed typical type IV patterns with a sharp hysteresis loop, which is characteristics of mesoporous materials (Fig. 1). The surface area and pore size distribution of the support (APTES@SiO2) studies reveals a high specific surface area of about 416 m2 g−1 with specific pore volume of 0.65 cm3 g−1 (Table S1, ESI†). These values upon metal immobilization (FeCl3-imine@SiO2) decreases significantly to 283 m2 g−1 and 0.43 cm3 g−1 which is consistent with successful immobilization of iron onto silica matrix.
The XRD patterns (Fig. 2) of the imine@SiO2 material exhibit a broad peak centered at 2θ = 22.48° for silica which upon immobilization of FeCl3 does not show any prominent shift (2θ = 22.13°), except a slight decrease in intensity. No crystalline phases related to metallic iron or iron oxides were observed in the spectra of FeCl3-imine@SiO2. However, a prominent peak at 2θ = 35.9° was observed which matched well with the XRD patterns of FeCl3 or FeCl3 doped polymer,15 suggesting the presence of FeCl3 crystallites within silica matrix. The SEM image of the fresh catalyst material shows silica particles are of different dimensions (Fig. 3a). The existence of iron in the catalyst was further confirmed by SEM-EDX analysis. The ESR spectra of FeCl3-imine@SiO2 displays six lines (Fig. 4) with an average g value of 2.01, typical for a Fe3+ high spin (I = 5/2) system with hyperfine splitting. The iron content determined by AAS analysis was found to be 0.44 mmol g−1.
 | ||
| Fig. 3 (a) SEM-EDX spectra of FeCl3-imine@SiO2 (before catalysis). (b) SEM-EDX spectra of FeCl3-imine@SiO2 (after 5th use). | ||
The XPS survey spectrum (Fig. S2, ESI†) of the iron-based material clearly shows the presence of iron, chloride and nitrogen, besides of carbon, silica and oxygen. In the N 1s region (Fig. 5a) the high resolution spectrum exhibits two peaks at 398.0 eV and 400.2 attributable to amine and imine group respectively.16 The Fe(2p) band (Fig. 5b) splits into two peaks, 2p3/2 and 2p5/2, with corresponding binding energies at 711.1 eV and 724.8 eV respectively and the values are very much close to the Fe2p binding energies of reported iron(III) compounds.14c,16
Catalytic activity
The catalytic activity of the material FeCl3-imine@SiO2 was investigated for alcohol oxidation reaction using water as solvent. Initial experiments for determining comparative efficacy and optimization study (e.g. temperature, catalyst quantity) was performed by considering oxidation of benzyl alcohol as model reaction (Table 1). Initially, oxidation reaction was performed under aerobic condition by bubbling air into the reaction mixture at room temperature. After 24 h only 42% benzaldehyde formation was detected by GC-MS (entry 1). However, when the same reaction was performed by bubbling molecular O2 (1 atm.) for 8 h moderate improvement in benzaldehyde formation was noticed (entry 2). On increasing temperature to 80 °C, significant improvement in the product yield was observed (entry 4). Interestingly, under similar experimental condition, homogeneous FeCl3 as catalyst gave only 36% benzaldehyde (entry 5). It may be worth to mention that usually immobilized catalysts show lesser activity compared to their homogeneous counterparts, however in our case a significant improvement in yield was observed by the catalyst FeCl3-imine@SiO2 compared to homogeneous FeCl3 possibly suggesting a cooperative effect between the metal and supported ligand which plays a crucial role in promoting oxidation reaction.| Entry | Catalyst | Oxidant | Catalyst (mol%) | Temp (°C) | Time (h) | Yieldb (%) |
|---|---|---|---|---|---|---|
| a Reaction conditions: benzylalcohol (1 mmol), O2 (1 atm., bubbling), iron-catalyst, water (6 mL).b GC yield. | ||||||
| 1 | FeCl3@imine-SiO2 | Air | 3 | 30 | 24 | 42 |
| 2 | FeCl3@imine-SiO2 | O2 | 3 | 30 | 8 | 64 |
| 3 | FeCl3@imine-SiO2 | O2 | 3 | 60 | 8 | 85 |
| 4 | FeCl3@imine-SiO2 | O2 | 3 | 80 | 8 | 96 |
| 5 | FeCl3 | O2 | 3 | 80 | 8 | 36 |
| 6 | FeCl3@imine-SiO2 | O2 | 2 | 80 | 8 | 96 |
| 7 | FeCl3@imine-SiO2 | O2 | 1 | 80 | 8 | 84 |
| 8 | FeCl3@imine-SiO2 | O2 | 2 | 80 | 6 | 96 |
Encouraged by our initial result, we extended the scope of our iron-based catalyst for a range of other representative primary and secondary alcohols including heterocyclic alcohol (Table 2). Under the optimized condition (80 °C, 2.0 mol% Fe), primary benzylic alcohols bearing electron-donating groups at para position were effectively converted to their corresponding aldehydes in excellent yields (entries 2 and 4). However, conversions of benzylic alcohol bearing electron-withdrawing substituents at para position were found to be moderate (entry 5). Our result also revealed that the positional isomers of the substituents have a significant influence on the catalytic performances. For instance, oxidation of m-methylbenzylalcohol gave 73% product while under similar experimental condition p-methylbenzylalcohol gave 92% aldehyde (entries 3 and 2). The secondary benzyl alcohols like benzhydrol (entry 10) or 1-phenyl ethanol (entry 9) also gave excellent yields of corresponding ketones. In fact, our protocol was also amenable to allylic alcohols such as cinnamyl alcohol (entry 7) or heterocyclic alcohol like furfuryl alcohol (entry 8) which are usually considered to be difficult substrates for alcohol oxidation reaction.4e In fact, our catalytic system was also found to be effective for oxidations of aliphatic alcohols like cyclohexanol (entry 12) or 1-octanol (entry 13), however only moderate yields were obtained.
| Entry | Substrate | Product | Time (h) | Yieldb,c (%) |
|---|---|---|---|---|
| a Reaction conditions: alcohol (1 mmol), O2 (1 atm.), H2O (6 mL), catalyst (2 mol%), 80 °C.b GC yield.c Isolated yield in the parenthesis. | ||||
| 1 | 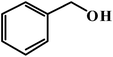 |
 |
6 | 96 (93) |
| 2 | 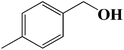 |
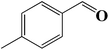 |
6 | 92 (88) |
| 3 | 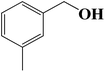 |
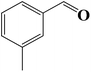 |
6 | 73 (68) |
| 4 | 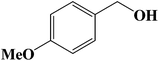 |
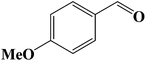 |
6 | 89 (86) |
| 5 | 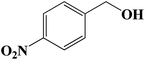 |
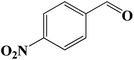 |
7 | 65 (63) |
| 6 | 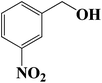 |
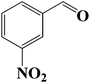 |
7 | 46 (44) |
| 7 | 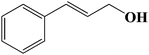 |
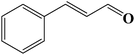 |
6 | 89 (87) |
| 8 | 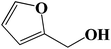 |
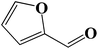 |
6 | 78 |
| 9 | 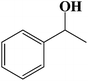 |
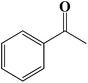 |
6 | 90 (87) |
| 10 | 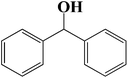 |
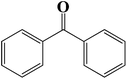 |
6 | 91 (90) |
| 11 | 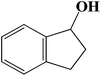 |
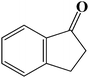 |
7 | 73 |
| 12 | 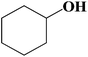 |
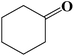 |
7 | 51 |
| 13 |  |
 |
8 | 62 |
| 14 |  |
 |
8 | 56 |
Reusability and heterogeneity test
Our study (Fig. S3, ESI†) reveals that the catalyst could be reused at least five cycles without significant loss in activity. The AAS analysis of the five times used catalyst shows a very negligible decrease in the Fe content (<2%). The SEM picture of the used catalyst (Fig. 4b) is very much similar to that of the fresh catalyst (Fig. 4a) except slight decrease in silica particle sizes, clearly indicating the surface morphology of the catalyst remained unaffected even after five consecutive uses. The SEM-EDX spectra of the used catalyst also shows the presence of Fe suggesting that the catalyst could be reused for further runs. In order to confirm the heterogeneity of our system we have performed hot filtration test by taking benzyl alcohol as a model substrate. For this purpose the oxidation reaction was stopped after 2 h and the solid catalyst was separated from the mixture by filtration under hot condition (conversion 46%) and the filtrate was then allowed to continue stirring under identical conditions. After 4 h of stirring no further change in the product concentration was noticed (Fig. 6), indicating that no leaching of catalyst was occurred.Conclusion
In conclusion, an iron(III)-based heterogeneous catalyst has been developed for the first time that effectively oxidizes primary and secondary alcohols to corresponding carbonyl compounds in water with molecular oxygen without using any additive or base. The low costs coupled with environment friendly properties of the materials used in our protocol (e.g. iron, silica, O2, water) will make this system a sustainable alternative to expensive platinum metal based catalysts.Acknowledgements
The CSIR, New Delhi, is gratefully acknowledged for financial support (Grant no: 02/0081/12). The UGC, New Delhi was acknowledged SAP-DRS programme and BSR-RFSMS fellowship. DST, New Delhi has also been acknowledged for providing Surface Area Analyzer to the Department under FIST programme. SAIF, NEHU, Shillong and SAFE, STIC, Cochin are acknowledged for TEM and SEM analysis respectively.References
- (a) R. A. Sheldon, Catal. Today, 2015, 247, 4 CrossRef CAS PubMed; (b) C. Parmeggiani and F. Cardona, Green Chem., 2012, 14, 547 RSC; (c) Z. Shi, C. Zhang, C. Tang and N. Jiao, Chem. Soc. Rev., 2012, 41, 3381 RSC; (d) Q. Cao, L. M. Dornan, L. Rogan, N. L. Hughes and M. J. Muldoon, Chem. Commun., 2014, 50, 4524 RSC; (e) M. J. Schultz and M. S. Sigman, Tetrahedron, 2006, 62, 8227 CrossRef CAS PubMed.
- (a) Z. Guo, B. Liu, Q. Zhang, W. Deng, Y. Wang and Y. Yang, Chem. Soc. Rev., 2014, 43, 3480 RSC; (b) S. E. Devis, M. S. Lde and R. J. Davis, Green Chem., 2013, 15, 17 RSC.
- M. Hajimohammadi, N. Safari, H. Mofakham and F. Deyhimi, Green Chem., 2011, 13, 991 RSC.
- (a) K. M. Gligorich and M. S. Sigman, Chem. Commun., 2009, 3854 RSC; (b) Y. Ye, M. D. Johnson, T. Diao, M. H. Yates and S. S. Stahl, Green Chem., 2010, 12, 1180 RSC; (c) B. Karimi, S. Abedi, J. H. Clark and V. Budarin, Angew. Chem., Int. Ed., 2006, 45, 4776 CrossRef CAS PubMed; (d) B. Karimi, A. Zamani, S. Abedi and J. H. Clark, Green Chem., 2009, 11, 109 RSC; (e) D. Sahu, C. Sarmah and P. Das, Tetrahedron Lett., 2014, 55, 3422 CrossRef CAS PubMed; (f) S. F. J. Hackett, R. M. Brydson, M. H. Gass, I. Harvey, A. D. Newman, K. Wilson and A. F. Lee, Angew. Chem., Int. Ed., 2007, 46, 8593 CrossRef CAS PubMed; (g) H. Wu, Q. Zhang and Y. Wang, Adv. Synth. Catal., 2005, 347, 1536 Search PubMed; (h) X. Ye, M. D. Johnson, T. Diao, M. H. Yates and S. S. Stahl, Green Chem., 2010, 12, 1180 RSC; (i) B. Karimi, D. Elhamifar, J. H. Clark and A. J. Hunt, Org. Biomol. Chem., 2011, 9, 7420 RSC; (j) H. Yang, X. Han, Z. Ma, R. Wang, J. Liu and X. Ji, Green Chem., 2010, 12, 441 RSC; (k) C. M. A. Parlett, D. W. Bruce, N. S. Hondow, A. F. Lee and K. Wilson, ACS Catal., 2011, 1, 636 CrossRef CAS; (l) J. Chen, Q. Zhang, Y. Wang and H. Wan, Adv. Synth. Catal., 2008, 350, 453 CrossRef CAS PubMed; (m) B. Karimi, A. Zamani and J. H. Clark, Organometallics, 2005, 24, 4695 CrossRef CAS; (n) M. J. Schultz, C. C. Park and M. S. Sigman, Chem. Commun., 2002, 3034 RSC.
- (a) A. Wusiman and C.-D. Lu, Appl. Organomet. Chem., 2015, 29, 254 CrossRef CAS PubMed; (b) J. Olguin, H. Muller-Bunz and M. Albrecht, Chem. Commun., 2014, 50, 3488 RSC; (c) L. Liu, M. Yu, B. B. Wayland and X. Fu, Chem. Commun., 2010, 46, 6353 RSC.
- (a) K. Yamaguchi and N. Mizuno, Angew. Chem., Int. Ed., 2002, 41, 4538 CrossRef CAS; (b) Z.-W. Yang, X. Zhao, T.-J. Li, W.-L. Chen, Q.-X. Kang, X.-Q. Xu, X.-X. Liang, Y. Feng, H.-H. Duan and Z.-Q. Li, Catal. Commun., 2015, 65, 34 CrossRef CAS PubMed; (c) D. Canseco-Gonzalez and M. Albrecht, Dalton Trans., 2013, 42, 7424 RSC.
- (a) P. Miedziak, M. Sankar, N. Dimitratos, J. A. Lopez-Sanchez, A. F. Carley, D. W. Knight, S. H. Taylor, C. J. Kiely and G. J. Hutchings, Catal. Today, 2011, 164, 315 CrossRef CAS PubMed; (b) Y. Li, Y. Gao and C. Yang, Chem. Commun., 2015, 51, 7721 RSC; (c) X. Yu, Y. Huo, J. Yang, S. Chang, Y. Ma and W. Huang, Appl. Surf. Sci., 2013, 280, 450 CrossRef CAS PubMed; (d) R. H. Adnan, G. G. Andersson, M. I. J. Polson, G. F. Metha and V. B. Golovko, Catal. Sci. Technol., 2015, 5, 1323 RSC; (e) T. Wang, X. Yuan, S. Li, L. Zeng and J. Gong, Nanoscale, 2015, 7, 7593 RSC; (f) C. D. Pina, E. Falletta, L. Prati and M. Rossi, Chem. Soc. Rev., 2008, 37, 2077 RSC.
- (a) Y. Hong, X. Yan, X. Liao, R. Li, X. Xu, L. Xiao and J. Fan, Chem. Commun., 2014, 50, 9679 RSC; (b) T. Lu, Z. Du, J. Liu, H. Ma and J. Xu, Green Chem., 2013, 15, 2215 RSC; (c) T. Wang, H. Shou, Y. Kou and H. Liu, Green Chem., 2009, 11, 562 RSC; (d) K. Kon, S. M. A. H. Siddiki and K.-i. Shimizu, J. Catal., 2013, 304, 63 CrossRef CAS PubMed; (e) T. Wang, C.-X. Xiao, L. Yan, L. Xu, J. Luo, H. Shou, Y. Kou and H. Liu, Chem. Commun., 2007, 4375 RSC.
- (a) B. Xu, J.-P. Lumb and B. A. Arndtsen, Angew. Chem., Int. Ed., 2015, 54, 4208 CrossRef CAS PubMed; (b) L. Vanoye, M. Pablos, C. D. Bellefon and A. Favre-Reguillon, Adv. Synth. Catal., 2015, 357, 739 CrossRef CAS PubMed; (c) B.-T. Chen, K. V. Bukhryakov, R. Sougrat and V. O. Rodionov, ACS Catal., 2015, 5, 1313 CrossRef CAS; (d) G. Zhang, C. Yang, E. Liu, L. Li, J. A. Golen and A. L. Rheingold, RSC Adv., 2014, 4, 61907 RSC; (e) P. Gamez, I. W. C. E. Arends, J. Reedijk and R. A. Sheldon, Chem. Commun., 2003, 2414–2415 RSC; (f) G. Zhang, X. Han, Y. Luan, Y. Wang, X. Wen, L. Xu, C. Ding and J. Gao, RSC Adv., 2013, 3, 19255 RSC; (g) S. Samanta, S. Das, P. K. Samanta, S. Dutta and P. Biswas, RSC Adv., 2013, 3, 19455 RSC.
- K. Junge, K. Schroder and M. Beller, Chem. Commun., 2011, 47, 4849 RSC.
- S. E. Martin and D. F. Suarez, Tetrahedron Lett., 2002, 43, 4475 CrossRef CAS.
- W. Brackman and C. Gaasbeek, Recl. Trav. Chim. Pays-Bas, 1966, 85, 257 CrossRef CAS PubMed.
- (a) N. Wang, R. Liu, J. Chen and X. Liang, Chem. Commun., 2005, 5322 RSC; (b) A. Al-Hunaiti, T. Niemi, A. Sibaouih, P. Pihko, M. Leskela and T. Repo, Chem. Commun., 2010, 46, 9250 RSC; (c) J. Liu and S. Ma, Org. Lett., 2013, 15, 5150 CrossRef CAS PubMed; (d) J. Liu and S. Ma, Tetrahedron, 2013, 69, 10161 CrossRef CAS PubMed; (e) M. Lenze and E. B. Bauer, Chem. Commun., 2013, 49, 5889 RSC; (f) S. Chakraborty, P. O. Lagaditis, M. Forster, E. A. Bielinski, N. Hazari, M. C. Holthausen, W. D. Jones and S. Schneider, ACS Catal., 2014, 4, 3994 CrossRef CAS; (g) C.-X. Miao, J.-Q. Wang, B. Yu, W.-G. Cheng, J. Sun, S. Chanfreau, L.-N. He and S.-J. Zhang, Chem. Commun., 2011, 47, 2697 RSC; (h) V. V. Namboodiri, V. Polshettiwar and R. S. Varma, Tetrahedron Lett., 2007, 48, 8839 CrossRef CAS PubMed; (i) J. Liu and S. Ma, Org. Biomol. Chem., 2013, 11, 4186 RSC; (j) J. Liu and S. Ma, Tetrahedron, 2013, 69, 10161 CrossRef CAS PubMed; (k) D. Sahu, B. Banik, M. Borah and P. Das, Lett. Org. Chem., 2014, 11, 671 CrossRef CAS.
- (a) A. Rezaeifard, M. Jafarpour and A. Naeimi, Catal. Commun., 2011, 16, 240 CrossRef CAS PubMed; (b) F. Rajabi, A. Pineda, S. Naserian, A. M. Balu, R. Luque and A. A. Romero, Green Chem., 2013, 15, 1232 RSC; (c) L. Geng, X. Zhang, W. Zhang, M. Jia and G. Liu, Chem. Commun., 2014, 50, 2965 RSC; (d) G. S. Machado, F. Wypych and S. Nakagaki, Appl. Catal., A, 2012, 413–414, 94 CrossRef CAS PubMed; (e) S. Fan, Y. Luan, J. Wang, H. Gao, X. Zhang and G. Wang, J. Mol. Catal. A: Chem., 2015, 404, 186 CrossRef PubMed; (f) D. Obermayer, A. M. Balu, A. A. Romero, W. Goessler, R. Luque and C. O. Kappe, Green Chem., 2013, 15, 1530 RSC; (g) M. Mahyari and A. Shaabani, Appl. Catal., A, 2014, 469, 524 CrossRef CAS PubMed; (h) F. Shi, M. K. Tse, M.-M. Pohl, J. Radnik, A. Bruckner, S. Zhang and M. Beller, J. Mol. Catal. A: Chem., 2008, 292, 28 CrossRef CAS PubMed; (i) L. Wang, J. Li, X. Zhao, Y. Lv, H. Zhang and S. Gao, Tetrahedron, 2013, 69, 6041 CrossRef CAS PubMed; (j) A. Rezaeifard, M. Jafarpour and A. Naeimi, Catal. Commun., 2011, 16, 240 CrossRef CAS PubMed; (k) P. Shejwalkar, N. P. Rath and E. B. Bauer, Dalton Trans., 2011, 40, 7617 RSC; (l) G. S. Machado, O. J. de Lima, K. J. Ciuffi, F. Wypych and S. Nakagaki, Catal. Sci. Technol., 2013, 3, 1094 RSC; (m) G. S. Machado, F. Wypych and S. Nakagaki, Appl. Catal., A, 2012, 413–414, 94 CrossRef CAS PubMed.
- E. M. Abdelrazek, Phys. B, 2007, 400, 26 CrossRef CAS PubMed.
- A. R. Silva, M. M. A. Freitas, C. Freire, B. de Castro and J. L. Figueiredo, Langmuir, 2002, 18, 8017 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Textural data of BET studies, FTIR spectra, XPS survey spectra, reusability diagram and GC-MS spectra of the carbonyl products. See DOI: 10.1039/c5ra16734e |
| This journal is © The Royal Society of Chemistry 2015 |