Silyl-assisted 1,2-cis-α-glucosylation for the synthesis of a triglucoside moiety in high-mannose-type oligosaccharides†
Kiichiro Totani*a,
Yuki Shinodaa,
Masaaki Shibaa,
Shogo Iwamotoa,
Akira Koizumia,
Yuji Matsuzakib and
Makoto Hiranoa
aDepartment of Materials and Life Science, Seikei University, 3-3-1 Kichijoji-kitamachi, Musashino-shi, Tokyo 180-8633, Japan. E-mail: ktotani@st.seikei.ac.jp
bGlyco Synthetic Lab., Tokyo Chemical Industry Co., Ltd., 6-15-9 Toshima, Kita-ku, Tokyo 114-0003, Japan
First published on 2nd September 2015
Abstract
A highly stereoselective 1,2-cis-α-glucosylation reaction was developed, in which the use of a strongly electron-donating tert-butyldimethylsilyl protecting group on the C-2 hydroxy group of a glycosyl donor enhanced the α-favoured transition state, and thus resulted in high yield and α-selectivity. Synthetic utility of this α-glucosylation was demonstrated by the generation of a triglucoside moiety in high-mannose-type oligosaccharides.
1,2-cis-α-Glycoside linkages represent a major subject in oligosaccharide synthesis,1 as they are often encountered in bioactive natural glycoconjugates,2 e.g. α-glucoside and α-fucoside in N-linked glycoproteins, or α-N-acetyl-galactosamine in O-linked glycoproteins. Moreover, glycolipids frequently contain 1,2-cis-α-glycosides. In the synthesis of oligosaccharides, the formation of most 1,2-cis-α-glycosides has been explained by the so-called thermodynamic anomeric effect,3 even though the origin of this effect is still controversial.4 Since the anomeric effect is still under ongoing investigation, it is often difficult to achieve 1,2-cis-α-glycosylation in high yield and stereoselectivity. The optimization of glycosylation conditions is usually a time-consuming task, because yield and stereoselectivity are affected by numerous factors such as protecting groups, promoters, solvents, and temperature. In this context, Ishiwata et al. proposed high-throughput screening for optimized solvent systems in 1,2-cis-α-glycosylation using Bn-d7 protecting groups.5 Their results showed that yield and the α/β selectivity fluctuated between 0 and 100%, as well as between 1
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 20
1 and 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, respectively. These findings illustrate the limitations of the practical use of the anomeric effect for the formation of 1,2-cis-α-glycoside linkages.
1, respectively. These findings illustrate the limitations of the practical use of the anomeric effect for the formation of 1,2-cis-α-glycoside linkages.
We have previously reported the synthesis of Glc3Man9GlcNAc2 (ref. 6) (Scheme 1), which is a glycoprotein oligosaccharide that regulates the glycoprotein quality control.7 However, the stereoselective construction of a Glcα1-2Glcα1-3Glc segment with sequential 1,2-cis-α-glucoside linkages in high yield was non-trivial, as glycosylation to generate the triglucoside could only be obtained in a maximum yield of 45%. Therefore, we developed a new 1,2-cis-α-glucosylation reaction that is mediated by enhancing the α-favoured transition state via an electron-donating tert-butyldimethylsilyl (TBS) group8 at the O2-position of the glycosyl donor. To demonstrate the efficiency of this glycosylation reaction, we also synthesized Glcα1-2Glcα1-3Glc through sequential silyl-assisted α-glucosylation.
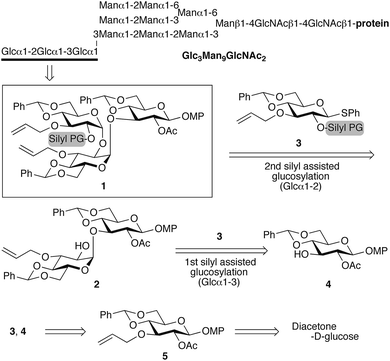 | ||
| Scheme 1 Retrosynthetic analysis for the formation of triglucoside 1 by sequential silyl-assisted glucosylation. | ||
For that purpose, we designed glycosyl donor 3 with an electron-donating protecting group, as well as efficient synthetic pathways to 3 and acceptor 4 from the common key intermediate 5 (Scheme 1). Computational studies by Nukada et al. showed that the conformational effect of the 4,6-O-benzylidene group should induce a 4H3 chair conformation for the gluco-type oxocarbenium ion intermediate.9 We decided to use benzylidene glycosyl donor 3 to reduce the number of intermediates, as this thioglycoside should exhibit advantageous stability in such a multi-step oligosaccharide synthesis. A series of glycosyl donors, 3a–d (Scheme 3), with different electron-donating groups at the O2-position should allow the examination of their influence on the reactivity and stereoselectivity in the glycosylation with 4. Combining these results with further investigations regarding the solvent effect on the glycosylation should allow a discussion of the steric and electronic effects of the protecting group. Deprotection at the O2′-position of the resulting disaccharide should afford disaccharide acceptor 2, followed by a second glycosylation with donor 3 to furnish the targeted trisaccharide 1.
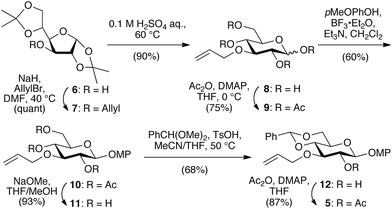
First, we synthesized key intermediate 5 as a glycosyl donor and acceptor (Scheme 2). Allylation of 3-OH in diacetone-D-glucose 6 provided 7, which was subjected to acidic cleavage of the isopropylidene groups to afford pyranosidic 8. After peracetylation of the hydroxyl groups to furnish 9, conversion of the anomeric acetate into a methoxy phenyl (MP) derivative yielded 10. After deprotection of the Ac groups in 10, resulting in the formation of 11, the 4,6-OH groups were protected with a benzylidene group to generate 12. Finally, acetylation of the 2-OH group in 12 yielded key intermediate 5. This regioselectively deprotectable compound is also commercially available from Tokyo Chemical Industry Co. (Tokyo, Japan).
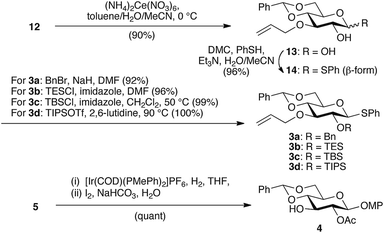
Subsequently, we prepared a series of glycosyl donors (3a–d) and one acceptor (4; Scheme 3). Although 3a–d can be obtained from 5, a one-step reduction from 12 would be more practical. Oxidative deprotection of the MP group in 12 provided 13, followed by the formation of a β-selective thioglycoside using 2-chloro-1,3-dimethyl-2-imidazolinium chloride (DMC)10 to give 14. For the protection of the 2-OH group in 14, we selected several benign electron-donating ethers as protecting groups. Initially, we introduced a benzyl group as a typical ether-type protecting group to afford glycosyl donor 3a. Subsequently, we focused on silyl ether groups, as we expected a strong electron donation from such groups. As the steric bulk is another tunable feature of silyl ethers, we introduced triethylsilyl (TES), TBS, and triisopropylsilyl (TIPS) groups with varying degrees of steric demand to afford glycosyl donors 3b, 3c, and 3d, respectively. Although the introduction of bulky silyl groups imposes structural strain on the pyranose ring conformation in some cases,11 3b, 3c, and 3d maintained a 4C1 conformation as evident from the J couplings of the pyranose ring protons in the 1H NMR spectra. Finally, allyl deprotection of key intermediate 5 provided glycosyl acceptor 4, and thus the substrate synthesis for the glycosylation was completed.
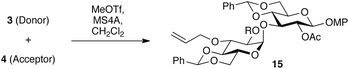
We then proceeded to examine the influence of protecting the O2 groups of donor 3 on the reactivity and stereoselectivity of the glycosylation reaction with acceptor 4 (Table 1). The promoter was selected from a range of metal free reagents, in order to avoid the possibility of affecting the stereoselectivity through coordination of the reaction intermediate with a metal. Although DMTST12 and NIS–TfOH13 are well known, strong, metal-free activation agents, these active species are prepared in situ, and small amounts of their unreacted precursors might be still present in the reaction mixture. To eliminate potential side reactions with these precursors, we chose MeOTf as a promoter.14 Considering that the resulting stereoselectivity originates from the donor structure, non-coordinative CH2Cl2 was selected as the reaction medium. A reaction mixture containing O2-benzylated donor 3a, acceptor 4, and MeOTf at 10 °C provided disaccharide 15a in 85% yield (entry 1). Although the desired α-linkage was generated preferentially as reported for similar 4,6-O-benzylidene-2-O-benzylatedglucosyl donors,15 the observed stereoselectivity was only moderate (α/β = 82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18) under our reaction conditions. In case of TES-substituted donor 3b, deprotection of the TES group was observed, and hence this donor was considered unsuitable for the glycosylation conditions (entry 2). Subsequently, the glycosylation with TBS-substituted 3c was examined, and the formation of the desired disaccharide 15c was observed in excellent yield (96%) and α-selectivity (α/β ≥ 95
18) under our reaction conditions. In case of TES-substituted donor 3b, deprotection of the TES group was observed, and hence this donor was considered unsuitable for the glycosylation conditions (entry 2). Subsequently, the glycosylation with TBS-substituted 3c was examined, and the formation of the desired disaccharide 15c was observed in excellent yield (96%) and α-selectivity (α/β ≥ 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5; entry 3). Although steric congestion around the anomeric position by the bulky TBS group might induce decreased reactivity, the required reaction temperature (0 °C) was lower than that for benzylated donor 3a (10 °C). This result suggested that the steric congestion imposed by the TBS group does not block the anomeric position. In contrast, the presence of the TBS group enhances the reactivity through stabilization of the cationic intermediate by electron donation. Using low temperature NMR spectroscopy based on the primary 13C kinetic isotope effect in combination with computational methods, Crich and co-workers revealed that the activation of a 4,6-O-benzylidene-glucosyl and mannosyl donors with Tf2O initially provide stable α-triflates in equilibrium with an α-contact ion pairs (α-CIP), before a subsequent SN2-like attack of a nucleophile provides the β-glucosides.16 Conversely, formation of the α-glucoside must arise from a β-triflate, which was not observed by VT-NMR. They also showed that this can occur, if the α-triflate exists in a rapidly exchanging equilibrium with its less stable, but more reactive β-CIP or the β-triflate via the solvent separated ion pair (SSIP), i.e. the oxocarbenium ion.16 Based on these results, the α-selectivity observed for entry 3 may result in a shift of the equilibrium towards the β-CIP through stabilization of a SSIP by the electron donation from the TBS group. In fact, Crich and co-workers reported the opposite result for the mannosyl donor, i.e. that a non-participating electron-withdrawing group at the O2-position destabilizes the formation of a SSIP and results in a β-selective glycosylation through a stable α-triflate,17 which is not in conflict with our hypothesis. However, the question of how the steric bulk of the silyl group affects the glycosylation still requires to be answered. Therefore, we examined the effect of the more bulky TIPS group in donor 3d (entry 4). For the corresponding reaction, a highly α-selective glucosylation to give disaccharide 15d was observed, even though the decreased reactivity caused a lower yield (50%) after comparable reaction time. In spite of the steric congestion around the anomeric position induced by the TIPS group, which caused the decreased reactivity, the observed highly stereoselective generation of the 1,2-cis-α-glucoside instead of the 1,2-trans-β-glucoside is interesting. The electronic effect of the silyl group at the O2-position on the stereoselectivity seemed to be unexpectedly strong.
5; entry 3). Although steric congestion around the anomeric position by the bulky TBS group might induce decreased reactivity, the required reaction temperature (0 °C) was lower than that for benzylated donor 3a (10 °C). This result suggested that the steric congestion imposed by the TBS group does not block the anomeric position. In contrast, the presence of the TBS group enhances the reactivity through stabilization of the cationic intermediate by electron donation. Using low temperature NMR spectroscopy based on the primary 13C kinetic isotope effect in combination with computational methods, Crich and co-workers revealed that the activation of a 4,6-O-benzylidene-glucosyl and mannosyl donors with Tf2O initially provide stable α-triflates in equilibrium with an α-contact ion pairs (α-CIP), before a subsequent SN2-like attack of a nucleophile provides the β-glucosides.16 Conversely, formation of the α-glucoside must arise from a β-triflate, which was not observed by VT-NMR. They also showed that this can occur, if the α-triflate exists in a rapidly exchanging equilibrium with its less stable, but more reactive β-CIP or the β-triflate via the solvent separated ion pair (SSIP), i.e. the oxocarbenium ion.16 Based on these results, the α-selectivity observed for entry 3 may result in a shift of the equilibrium towards the β-CIP through stabilization of a SSIP by the electron donation from the TBS group. In fact, Crich and co-workers reported the opposite result for the mannosyl donor, i.e. that a non-participating electron-withdrawing group at the O2-position destabilizes the formation of a SSIP and results in a β-selective glycosylation through a stable α-triflate,17 which is not in conflict with our hypothesis. However, the question of how the steric bulk of the silyl group affects the glycosylation still requires to be answered. Therefore, we examined the effect of the more bulky TIPS group in donor 3d (entry 4). For the corresponding reaction, a highly α-selective glucosylation to give disaccharide 15d was observed, even though the decreased reactivity caused a lower yield (50%) after comparable reaction time. In spite of the steric congestion around the anomeric position induced by the TIPS group, which caused the decreased reactivity, the observed highly stereoselective generation of the 1,2-cis-α-glucoside instead of the 1,2-trans-β-glucoside is interesting. The electronic effect of the silyl group at the O2-position on the stereoselectivity seemed to be unexpectedly strong.
| Entry | Glycosyl donor | R | Product | Yield (%) | α/βd |
|---|---|---|---|---|---|
| a Reagents and conditions: MeOTf, MS4A, CH2Cl2, 10 °C (for entry 1), 0 °C (for entries 2–4), 32 h.b 56% of 4 was recovered. Deprotection of the TES group was observed.c 38% of 4 was recovered.d The stereochemistry and stereoselectivity were determined by 1H NMR. | |||||
| 1 | 3a | Bn | 15a | 85 | 82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 18 |
| 2 | 3b | TES | 15b | —b | — |
| 3 | 3c | TBS | 15c | 96 | >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
| 4 | 3d | TIPS | 15d | 50c | >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
Therefore, we examined the effect of the TBS group in detail. For this silyl protecting group, applications in carbohydrate chemistry have been reported by Yang and co-workers, who demonstrated that the reactivity of arabinofranosyl thioglycoside was enhanced by the introduction of TBS groups at the O3- and O5-position,18 even though they did not comment on the origin of the increased reactivity. The groups of Bols and Yamada reported independently the conformational arming effect of glycopyranosyl donors via conformational changes of the donor, mediated by the introduction of multiple bulky silyl groups.19 As our reaction system does not include any conformational changes of the donor, the increased reactivity should be due to a stabilization of the oxocarbenium ion by the electron donation from the TBS group. Conversely, the effect of the TBS group on the α-selectivity should lead to (1) a shift of the equilibrium of the active intermediate towards the β-CIP due to the electron donation from the TBS group, and (2) an induced α-glycoside formation due to the steric congestion of the TBS group at the β-face of the anomeric position. Therefore, we examined a potential TBS-induced steric congestion around the anomeric position by comparison of solvent effects on the glycosylation reaction (Table 2). For that purpose, we determined the stereoselectivity of the glycosylation reaction between donor 3c and acceptor 4 using benign solvents (CH2Cl2, toluene), a β-participating solvent (Et2O),20 and an α-participating solvent (MeCN).21 The reaction in CH2Cl2 provided the desired disaccharide 15c in high yield and α-selectivity (entry 1). Toluene also furnished the α-product in high stereoselectivity, albeit in lower yield (entry 2). Although differences in substrate solubility should decrease the reactivity to some extend, generally an α-enhancing effect of the TBS group was observed in nonpolar and aprotic solvents. Subsequently, we used the polar, aprotic, β-participating solvent Et2O (entry 3), which resulted in high α-selectivity and low yield (20%). This can be explained by a β-orientation of Et2O with respect to the oxocarbenium ion. This orientation would block the β-face as well as decrease the electrophilicity and thus the reactivity. Accordingly, there seems to be sufficient space around the β-face at the anomeric position to coordinate a solvent molecule, which suggested that the conformation of the TBS group does not induce steric over-congestion. Using the α-participating, polar, and aprotic solvent MeCN, the reactivity was decreased on account of a diminished electrophilicity similar to Et2O, and a yield of 21% was observed (entry 4). Moreover, the stereoselectivity was decreased as a result of the α-coordination of MeCN to provide an α/β ratio of 48![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 52. Results from entries 3 and 4 indicated that enough space is available around both the α- and β-face at the anomeric position, suggesting no apparent correlation between steric bulk of the TBS group and the stereoselectivity.
52. Results from entries 3 and 4 indicated that enough space is available around both the α- and β-face at the anomeric position, suggesting no apparent correlation between steric bulk of the TBS group and the stereoselectivity.
| Entry | Glycosyl donor | Solvent | Product | Yield (%) | α/βf |
|---|---|---|---|---|---|
| a Reagents and conditions: MeOTf, MS4A, CH2Cl2, 0 °C (for entries 1–4), 10 °C (for entries 5 and 6), 32 h.b 45% of 4 was recovered.c 58% of 4 was recovered.d 63% of 4 was recovered.e 83% of 4 was recovered.f The stereochemistry and stereoselectivity were determined by 1H NMR. | |||||
| 1 | 3c | CH2Cl2 | 15c | 96 | >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
| 2 | 3c | Toluene | 15c | 41b | >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
| 3 | 3c | Et2O | 15c | 20c | >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
| 4 | 3c | CH3CN | 15c | 21d | 48![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 52 52 |
| 5 | 3a | CH2Cl2 | 15a | 85 | 82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 18 |
| 6 | 3a | Et2O | 15a | 11e | 93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 7 |
Based on these results, a reaction mechanism for the 1,2-cis-α-glucosylation is proposed (Fig. 1). Treatment of 3c with MeOTf should initially generate the α-triflate B via the temporary sulfonium salt A. Electron donation from the TBS group should stabilize the cationic intermediates C, D and E, and a subsequent SN2-like attack of acceptor 4 at the more reactive β-CIP E should provide the α-glucoside. We further examined the solvent effect on the glycosylations using 2-O-Bn donor 3a (Table 2). The observed stereoselectivity in CH2Cl2 (α/β = 82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18; entry 5) was lower than that in Et2O (α/β = 93
18; entry 5) was lower than that in Et2O (α/β = 93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7; entry 6). However, using Et2O decreased the reactivity and yielded 15a in only 11%. Thus, the introduction of glucosylation compared to an optimization of the solvent system. For generality of the TBS-assisted glucosylation reaction, a TBS group at the 2-OH-position of the glycosyl donor can be considered as a more effective approach to induce 1,2-cis-α-glycosylation of 3c with several glycosyl acceptors were also examined (Table 3). All the examples of glycosylation with galactosyl acceptor 16 (entry 1), mannosyl acceptor 17 (entry 2) glucosyl acceptor 18 (entry 3) and 19 (entry 4) gave the good to excellent α-selectivities.
7; entry 6). However, using Et2O decreased the reactivity and yielded 15a in only 11%. Thus, the introduction of glucosylation compared to an optimization of the solvent system. For generality of the TBS-assisted glucosylation reaction, a TBS group at the 2-OH-position of the glycosyl donor can be considered as a more effective approach to induce 1,2-cis-α-glycosylation of 3c with several glycosyl acceptors were also examined (Table 3). All the examples of glycosylation with galactosyl acceptor 16 (entry 1), mannosyl acceptor 17 (entry 2) glucosyl acceptor 18 (entry 3) and 19 (entry 4) gave the good to excellent α-selectivities.
 | ||
| Fig. 1 Proposed mechanism for the glycosylation of 3c, wherein electron donation from the silyl ether of 3c should stabilize cationic intermediates. | ||
| Entry | Glycosyl acceptor | Product | Yield (%) | α/βd |
|---|---|---|---|---|
a Reagents and conditions: MeOTf, MS4A, CH2Cl2, 10 °C, 32 h.b 22% of the glycosyl acceptor 16 was recovered.c 50% of the glycosyl acceptor 17 including the Ac-migrated 2,3,4,6-tetra-O-Ac form (ca. 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) were recovered.d The stereochemistry and stereoselectivity were determined by 1H NMR. 1) were recovered.d The stereochemistry and stereoselectivity were determined by 1H NMR. |
||||
| 1 | 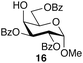 |
20 | 77b | 93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 7 |
| 2 | 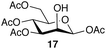 |
21 | 43c | 92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 8 |
| 3 | 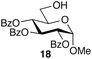 |
22 | 82 | >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
| 4 | 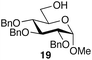 |
23 | 96 | >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
Finally, we used a sequential silyl-assisted α-glucosylation to synthesize triglucoside 1 (Scheme 4). The first silyl-assisted glycosylation to form an α-1,2-linkage between donor 3c and acceptor 4 provided disaccharide 15c in 96% yield. A subsequent deprotection of the TBS group with TBAF/AcOH to give acceptor 2 proceeded without acetyl migration. The following second silyl-assisted glycosylation of donor 3c with acceptor 2 generated an α-1,3-linkage to afford the desired trisaccharide 1 in 80% yield.
In conclusion, we developed a novel 1,2-cis-α-glucosylation reaction that is mediated by a TBS-assisted stabilization of the cationic intermediate, which is favourable for an α-attack of the glycosyl acceptor. This method was used to generate the Glcα1-2Glcα1-3Glc moiety of a high-mannose tetradecasaccharide. In previous synthetic approaches, generation of Glcα1-2Glc via glycosyl fluoride yielded in 90%, whereas subsequent glycosylation using thioglycoside with the optimized reaction conditions to obtain such trisaccharides did not exceed yields of 45%.6 Conversely, the present method allows the sequential use of the same silyl-assisted donor to form both α-1,3 and α-1,2-linkages with high yield and high 1,2-cis-α-selectivity to increase the efficiency of this synthetic pathway. Thus, regioselectively deprotectable feature of donor 3c was useful for further glycosylation at any positions of the OH-groups. Moreover, 2-OH, 3-OH and 6-OH gluco-type, 4-OH galacto-type and 2-OH manno-type acceptors were widely acceptable for this silyl-assisted α-glucosylation. In principle, our silyl-assisted approach could also be applied to construct α-fucoside and α-galactoside. Further studies in these areas are in progress and will be reported in due course.
Acknowledgements
This work was supported by a Grant-in-Aid for Young Scientists (A) and a Grant-in-Aid for Challenging Exploratory Research from the Japan Society for the Promotion of Science to K. T. (No. 23681049 and 25560420).Notes and references
- S. S. Nigudkar and A. V. Demchenko, Chem. Sci., 2015, 6, 2687–2704 RSC.
- A. Varki, R. D. Cummings, J. D. Esko, H. H. Freeze, P. Stanley, C. R. Bertozzi, G. W. Hart and M. E. Etzler, Essentials of Glycobiology, CSH Laboratory Press, New York, 2nd edn, 2009 Search PubMed.
- G. F. Bauerfeldt, T. M. Cardozo, M. S. Pereira and C. O. da Silva, Org. Biomol. Chem., 2013, 11, 299–308 CAS.
- I. Cumpstey, Org. Biomol. Chem., 2012, 10, 2503–2508 CAS.
- A. Ishiwata and Y. Ito, Tetrahedron Lett., 2005, 46, 3521–3524 CrossRef CAS PubMed.
- (a) I. Matsuo, T. Kashiwagi, K. Totani and Y. Ito, Tetrahedron Lett., 2005, 46, 4197–4200 CrossRef CAS PubMed; (b) I. Matsuo, K. Totani, A. Tatami and Y. Ito, Tetrahedron, 2006, 62, 8262–8277 CrossRef CAS PubMed.
- (a) K. Totani, Trends Glycosci. Glycotechnol., 2010, 22, 296–307 CrossRef CAS; (b) R. Benyair, E. Ron and G. Z. Lederkremer, Int. Rev. Cell Mol. Biol., 2011, 292, 197–280 CAS.
- R. Rathore and J. K. Kochi, Tetrahedron Lett., 1994, 35, 8577–8580 CrossRef CAS.
- T. Nukada, A. Berses and D. M. Whitfield, Carbohydr. Res., 2002, 337, 765–774 CrossRef CAS.
- T. Tanaka, T. Matsumoto, M. Noguchi, A. Kobayashi and S. Shoda, Chem. Lett., 2009, 38, 458–459 CrossRef CAS.
- (a) C. Walford, R. F. W. Jackson, N. H. Rees, W. Clegg and S. L. Heath, Chem. Commun., 1997, 1855–1856 RSC; (b) H. Yamada, M. Nakatani, T. Ikeda and Y. Marumoto, Tetrahedron Lett., 1999, 40, 5573–5576 CrossRef CAS; (c) C. H. Marzabadi, J. E. Anderson, J. Gonzalez-Outeirino, P. R. J. Gaffney, C. G. H. White, D. A. Tocher and L. J. Todaro, J. Am. Chem. Soc., 2003, 125, 15163–15173 CrossRef CAS PubMed.
- P. Fugedi, and P. J. Garegg, Carbohydr. Res., 1986, 149, C9–C12 Search PubMed.
- G. H. Veeneman, S. H. van Leeuwen and J. H. Boom, Tetrahedron Lett., 1990, 31, 1331–1334 CrossRef CAS.
- H. Lönn, Carbohydr. Res., 1985, 139, 105–113 Search PubMed.
- (a) D. Crich and W. Cai, J. Org. Chem., 1999, 64, 4926–4930 CrossRef CAS PubMed; (b) B. S. Komarova, M. V. Orekhova, Y. E. Tsvetkov and N. E. Nifantiev, Carbohydr. Res., 2014, 384, 70–86 CrossRef CAS PubMed.
- M. Huang, G. E. Garrett, N. Birlirakis, L. Bohe, D. A. Pratt and D. Crich, Nat. Chem., 2013, 4, 663–667 CrossRef PubMed.
- D. Crich, T. K. Hutton, A. Banerjee, P. Jayalath and J. Picione, Tetrahedron: Asymmetry, 2005, 16, 105–119 CrossRef CAS PubMed.
- X.-Y. Liang, H.-C. Bin and J.-S. Yang, Org. Lett., 2013, 15, 2834–2837 CrossRef CAS PubMed.
- (a) Y. Okada, T. Mukae, K. Okajima, M. Taira, M. Fujita and H. Yamada, Org. Lett., 2007, 9, 1573–1576 CrossRef CAS PubMed; (b) C. M. Pedersen, L. U. Nordstrøm and M. Bols, J. Am. Chem. Soc., 2007, 129, 9222–9235 CrossRef CAS PubMed.
- R. Edy and C. Scherch, Carbohydr. Res., 1974, 34, 79–90 CrossRef.
- R. R. Schmidt, M. Behrendt and A. Toepfer, Synlett, 1990, 694–696 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental details and characterization data for new compounds. See DOI: 10.1039/c5ra16659d |
| This journal is © The Royal Society of Chemistry 2015 |