Light triggered addition/annulation of 2-isocyanobiphenyls toward 6-trifluoromethyl-phenanthridines under photocatalyst-free conditions†
Xiangyang Tanga,
Shuang Songa,
Cuibo Liu*ab,
Rongjiao Zhua and
Bin Zhang*abc
aDepartment of Chemistry, School of Science, Tianjin University, Tianjin 300072, China. E-mail: bzhang@tju.edu.cn; chemliucuibo@163.com
bCollaborative Innovation Center of Chemical Science and Engineering (Tianjin), Tianjin 300072, China
cThe Key Lab of Systems Bioengineering, Ministry of Education, Tianjin University, Tianjin 300072, China
First published on 4th September 2015
Abstract
A photocatalyst-free, light promoted sequential radical addition/annulation of 2-isocyanobiphenyls to 6-trifluoromethyl phenanthridines is presented. Wide substrate scopes and scale-up experiment demonstrate the promising efficiency and utility of this strategy.
Isonitriles are privileged motifs in organic synthesis because of their vast potentials for synthetic versatility, which have aroused intensive and continuing interest from chemists.1 Recently, the radical addition/intramolecular homolytic aromatic substitution (HAS) reactions of 2-isocyanobiphenyls have been extensively investigated. By such a cascade procedure, a rapid assembly of 6-substituted phenanthridine frameworks is accomplished with high synthetic efficiency and atom economy. To date, various types of radical precursors including boronic acids,2 halides,3 aldehydes,4 diphenylphosphine oxide,5 a-oxocarboxylic acids,6 silanes,7 peroxides,8 amides,9 ethers,10 alkanes,11 hydrazines,12 and carbazates13 have been gradually explored for such transformations and a myriad of phenanthridine scaffolds with diverse functional groups have been successfully produced. Due to the wide applications in pharmaceutical science and drug research of phenanthridines and their derivatives,14 the synthesis 6-trifluoromethyl phenanthridines may be of great significance, and their potential biological activities may be profoundly enhanced to some extent. Because the incorporation of trifluoromethyl group can alert the physicochemical properties of the parent compounds, such as electronegativity, hydrophobicity, metabolic stability, and bioavailability compared to their non-trifluoromethylated counterparts.15 Although various methods leading to 6-trifluoromethyl phenanthridines have emerged,16 there are only three examples involving the somophilic isocyanide insertion by the electrophilic CF3 radical (Scheme 1). In 2013, Studer and co-workers reported a novel Bu4NI-catalyzed formation of 6-trifluoromethyl phenanthridines by using Togni's reagent as the precursor of CF3 radical in 1,4-dioxane at 80 °C.17 Almost simultaneously, Zhou et al. described a PhI(OAc)2-mediated oxidative cyclization of 2-isocyanobiphenyls with CF3SiMe3 in combination with BQ as the additive.18 In 2014, the Studer group again made an advance on finding a NiBr2 assisted and CF3I participated facile synthesis of 6-trifluoromethyl phenanthridines.19 Despite of the high effectiveness for the above strategies, some disadvantages are existed, such as the use of expensive CF3 reagents and environmentally unfriendly oxidants, the relatively high temperature, which restrict their practical applications to some extent. Therefore, the development of a mild, economic, environmentally benign and efficient alternative to realize the direct construction of 6-trifluoromethyl phenanthridines via the radical addition followed by HAS process is still an urgent need.
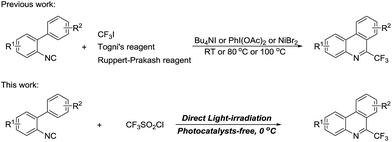 | ||
| Scheme 1 The cascade addition/annulation of 2-isocyanobiphenyls to 6-trifluoromethyl phenanthridines. | ||
Typically, the CF3 radical can be generated in three ways: the reduction, oxidation and homolysis of CF3 reagents using reducing agents, oxidizing agents and light-excitation or at high temperature or using initiators. The former two are preferred methods for the trifluoromethylation of a wide range of interesting scaffolds,20 while the reactions involving the homolysis of CF3 reagents are still limited.21 Recently, we have investigated the light-induced BiOBr nanosheets accelerated trifluoromethylation/arylation of N-aryl acrylamide substrates employing CF3SO2Cl as CF3 source.22 It is observed that small amount of the product is still formed without the addition of BiOBr photosensitizer. We reason this result may be ascribed to the homolysis of CF3SO2Cl under light-excitation generating the CF3 radical that engages this tandem transformation. This speculation inspired us to take advantage of these unexpected CF3 radicals, produced from the direct light-irradiation of CF3SO2Cl, for the trifluoromethylation of organic molecules. Isonitriles are isoelectronic with carbon monoxide, which can act as well-established C-radical acceptors. Herein, we wish to disclose our recent endeavor in light-mediated sequential arylation/trifluoromethylation of 2-isocyanobiphenyls to synthesize 6-trifluoromethyl-phenanthridines. Compared with the aforementioned protocols on the synthesis of 6-trifluoromethyl-phenanthridines, this procedure own three unique features: (1) the use of easily available and high active CF3SO2Cl as trifluoromethylating reagent; (2) direct light-induced transformation under photocatalyst-free conditions; (3) gram-scale synthesis. This promising and green chemical transformation constitutes an important complementary to the current light-driven organic reactions.23
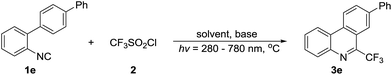
The light-induced isonitrile insertion reaction is initiated by treating 2-isocyanobiphenyl (1e) and CF3SO2Cl (2) with the addition of K2HPO4 in DMF under light irradiation (280–780 nm) for 6 h. As expected, this reaction indeed occurs, and 33% yield of the desired product is obtained (Table 1, entry 1). Delightedly, the yield of 3e dramatically increased to 76% when the reaction proceeds in DMAC (Table 1, entry 2). Encouraged by this exciting result, we further screen the other solvents including the polar/non-polar and protonic/non-protonic solvents, and observe that the others are inferior to DMAC, or even useless (Table 1, entries 3–10), which indicates a significant solvent effect of this radical addition/annulation of 2-isocyanobiphenyl. Additionally, examination of bases reveals that this one-pot tandem reaction proceeds more effectively in the presence of inorganic base, and K2HPO4 is the optimal one (Table 1, entries 11–15). However, the reaction efficiency is remarkablely decreased when K2HPO4 is removed from the reaction media, demonstrates that the base plays a stimulative role for the creation of trifluoromethylated phenanthridines (Table 1, entry 16). In order to identify the effective wavelength range for this reaction clearly, the UV-visible absorption test of CF3SO2Cl in DMAC is conducted (Fig. S1 in ESI†). From the spectrogram we can see that the optimal wavelength absorption range of CF3SO2Cl is approximately between 270 and 350 nm. It means that only this wavelength range is effective for the activation of CF3SO2Cl, and the rest section that from 350 nm to 780 nm is almost useless. Noting that, the reaction does not occur in the dark, showing that the CF3 radical is indeed generated by light-illumination, and that is the light that triggers this cascade transformation (Table 1, entry 17).
| Entry | Solvent | Base | Yieldb (%) |
|---|---|---|---|
| a Reaction conditions: 1a (0.2 mmol), 2 CF3SO2Cl (0.6 mmol), solvent (1.0 mL), base (0.6 mmol), 300 W Xe lamp, 0 °C. b Isolated yield is reported. c No reaction. d In the dark. | |||
| 1 | DMF | K2HPO4 | 33 |
| 2 | DMAC | K2HPO4 | 76 |
| 3 | DMSO | K2HPO4 | N.R.c |
| 4 | CH3CN | K2HPO4 | 23 |
| 5 | NMP | K2HPO4 | N.R.c |
| 6 | EtOAc | K2HPO4 | 43 |
| 7 | CH3OH | K2HPO4 | N.R.c |
| 8 | THF | K2HPO4 | N.R.c |
| 9 | CH2Cl2 | K2HPO4 | Trace |
| 10 | Toluene | K2HPO4 | N.R.c |
| 11 | DMAC | K2CO3 | 68 |
| 12 | DMAC | K3PO4 | 57 |
| 13 | DMAC | KOH | 67 |
| 14 | DMAC | tBuOK | 49 |
| 15 | DMAC | Et3N | 34 |
| 16 | DMAC | — | 30 |
| 17d | DMAC | K2HPO4 | N.R.c |
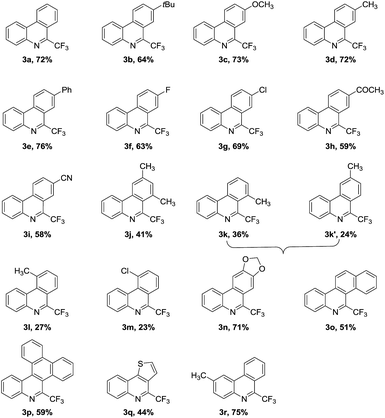
With the optimized reaction conditions in hand, the substrate scope is evaluated to test the universality, as listed in Table 2. Firstly, we investigate the influence of R2 on the nonisonitriles phenyl ring on the reaction efficiency. The substrates bearing the electron-rich or electron-deficient groups at para-position could all work smoothly to deliver the corresponding products in middle to good yields (Table 2, 3a–i). Noting that, even the potentially sensitive functional groups acetyl and cyano are both tolerated, and 59% and 58% yields of the phenanthridine derivatives are obtained (Table, 3h and 3i). The existence of acetyl and cyano gives an opportunity for further transformations by transition-metal catalyzed strategies or other technologies. In contrast, the ortho- and meta-substituted 2-isocyanobiphenyls suppress the reaction and a relatively low yields of the products are formed (Table 2, 3j–m). When meta-methyl substituted substrate is used, two regioisomers 3k and 3k′ are formed with a ratio of 1.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The dioxy heterocyclic group functionalized isocyanide is also a good candidate, and under the present reaction conditions we mainly get one isomer compound as the final product with the isolated yield of 71% (Table 2, 3n). The obtained molecule 3n containing a trisphaeridine framework, which can function as DNA intercalator.14a,c According to the significant characteristics of the trifluoromethyl-containing compounds, the biological activity of the synthesized trifluoromethylated trisphaeridine may be obviously improved. When the aromatic ring is replaced with 2-naphthyl, 9-phenanthrene and electron-deficient 2-thienyl moiety, radical addition/cyclization reactions still occur, giving rise to the desired products 3o–q in acceptable yields. Next, the substrate with a methyl group on the aromatic ring of the isocyano group is amenable to the selected reaction conditions, and 75% yield of the corresponding product is fabricated (Table 2, 3r).
1. The dioxy heterocyclic group functionalized isocyanide is also a good candidate, and under the present reaction conditions we mainly get one isomer compound as the final product with the isolated yield of 71% (Table 2, 3n). The obtained molecule 3n containing a trisphaeridine framework, which can function as DNA intercalator.14a,c According to the significant characteristics of the trifluoromethyl-containing compounds, the biological activity of the synthesized trifluoromethylated trisphaeridine may be obviously improved. When the aromatic ring is replaced with 2-naphthyl, 9-phenanthrene and electron-deficient 2-thienyl moiety, radical addition/cyclization reactions still occur, giving rise to the desired products 3o–q in acceptable yields. Next, the substrate with a methyl group on the aromatic ring of the isocyano group is amenable to the selected reaction conditions, and 75% yield of the corresponding product is fabricated (Table 2, 3r).
| a Reaction conditions: 1a (0.2 mmol), 2 CF3SO2Cl (0.6 mmol), DMAC (1.0 mL), K2HPO4 (0.6 mmol), 300 W Xe arclamp, 0 °C. |
|---|
 |
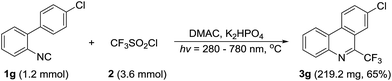
To highlight the utility of this photochemical protocol, an amplifying reaction of 1.2 mmol of 1g with 3.6 mmol CF3SO2Cl is conducted under the standard conditions (Scheme 2). A comparable yield of the product 3g with that of 0.2 mmol scale experiment is afforded, hints the potential applicability of our method.
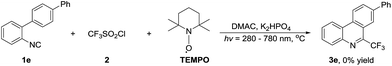
To confirm that this tandem transformation is originated from the addition of CF3 radical to the terminal carbon of 2-isocyanobiphenyl, the control experiment is carried out. Adding radical trapping agent, 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO), to the reaction system under otherwise identical conditions leads to no desired product, which supports that a radical pathway is involved in this radical addition/annulation of 2-isocyanobiphenyl (Scheme 3). This process is distinguished from Yu's work that discloses a concise and efficient synthetic approach to 6-(trifluoromethyl)phenanthridine through the ionic isocyanide insertion with no use of catalyst.24
Based on the above observations and the previous reports,2–13 a plausible mechanism is presented (Scheme 4). Homolysis of CF3SO2Cl by photoexcitation generates CF3 radical with the release of SO2 and Cl radical. Subsequent addition of CF3 radical to the terminal carbon of isocyanide affords the imidoyl radical I, which experiences a intramolecular homolytic aromatic substitution to give the cyclized radical intermediate II. The product is finally created with the leave of H radical, which is abstracted by Cl radical to get HCl. The formed HCl can be neutralized by K2HPO4 at the end.
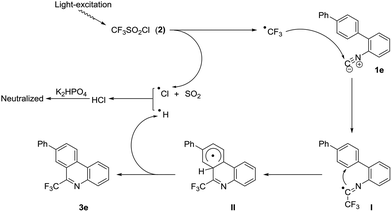 | ||
| Scheme 4 Proposed mechanism for the radical isocyanide insertion of 2-isocyanobiphenyl with CF3SO2Cl. | ||
In conclusion, we have developed a direct light-initiated one-pot consecutive radical addition/annulation of 2-isocyanobiphenyls using cheap and high active CF3SO2Cl as the precursor of CF3 radical under photocatalysts-free conditions. With this method, a variety of the corresponding 6-trifluoromethyl-phenanthridines derivatives bear diverse functional groups are constructed in acceptable to good yields. The scale-up experiment illustrates the potential applicability of our methodology. The simple, economic and environmentally friendly procedure may be applied to the future industrial processes. Further mechanistic researches and more light-induced cascade reactions for molecules with structural complexity are underway in our lab.
Acknowledgements
This research was financially supported by the National Natural Science Foundation of China (No. 21422104).Notes and references
- Selected examples see: (a) G. Y. S. Qiu, Q. P. Ding and J. Wu, Chem. Soc. Rev., 2013, 42, 5257 RSC; (b) S. Lang, Chem. Soc. Rev., 2013, 42, 4867 RSC; (c) A. Dömling, Chem. Rev., 2006, 106, 17 CrossRef PubMed; (d) B. Zhang and A. Studer, Org. Lett., 2014, 16, 1216 CrossRef CAS PubMed; (e) J. Zhu, Eur. J. Org. Chem., 2003, 1133 CrossRef CAS; (f) Z. H. Xia, J. B. Huang, Y. M. He, J. J. Zhao, J. Lei and Q. Zhu, Org. Lett., 2014, 16, 2546 CrossRef CAS PubMed; (g) J.-J. Cao, X. Wang, S.-Y. Wang and S.-J. Ji, Chem. Commun., 2014, 50, 12892 RSC; (h) X.-L. Yang, F. Chen, N.-N. Zhou, W. Yu and B. Han, Org. Lett., 2014, 16, 6476 CrossRef CAS PubMed; (i) J. Lei, X. X. Wu and Q. Zhu, Org. Lett., 2015, 17, 2322 CrossRef CAS PubMed.
- M. Tobisu, K. Koh, T. Furukawa and N. Chatani, Angew. Chem., Int. Ed., 2012, 51, 11363 CrossRef CAS PubMed.
- (a) H. Jiang, Y. Cheng, R. Wang, M. Zheng, Y. Zhang and S. Yu, Angew. Chem., Int. Ed., 2013, 52, 13289 CrossRef CAS PubMed; (b) L. J. Gu, C. Jin, J. Y. Liu, H. Y. Ding and B. M. Fan, Chem. Commun., 2014, 50, 4643 RSC; (c) W. J. Fu, M. Zhu, C. Xu, G. L. Zou, Z. Q. Wang and B. M. Ji, J. Fluorine Chem., 2014, 168, 50 CrossRef CAS; (d) I. Lenoir and M. L. Smith, J. Chem. Soc., Perkin Trans. 1, 2000, 641 RSC.
- D. Leifert, C. G. Daniliuc and A. Studer, Org. Lett., 2013, 15, 6286 CrossRef CAS PubMed.
- (a) B. Zhang, C. G. Daniliuc and A. Studer, Org. Lett., 2014, 16, 250 CrossRef CAS PubMed; (b) Y. W. Li, G. Y. S. Qiu, Q. P. Ding and J. Wu, Tetrahedron, 2014, 70, 4652 CrossRef CAS.
- J. Liu, C. Fan, H. Yin, C. Qin, G. Zhang, X. Zhang, H. Yi and A. Lei, Chem. Commun., 2014, 50, 2145 RSC.
- L. Wang, H. Zhu, S. J. Guo, J. Cheng and J.-T. Yu, Chem. Commun., 2014, 50, 10864 RSC.
- (a) C. D. Pan, H. L. Zhang, J. Han, Y. X. Cheng and C. J. Zhu, Chem. Commun., 2015, 51, 3786 RSC; (b) Q. Dai, J.-T. Yu, X. M. Feng, Y. Jiang, H. T. Yang and J. Cheng, Adv. Synth. Catal., 2014, 356, 3341 CrossRef CAS.
- H.-Y. Tu, Y.-R. Liu, J.-J. Chu, B.-L. Hu and X.-G. Zhang, J. Org. Chem., 2014, 79, 9907 CrossRef CAS PubMed.
- (a) C. D. Pan, J. Han and C. J. Zhu, Sci. China: Chem., 2014, 57, 1172 CrossRef CAS; (b) J.-J. Cao, T.-H. Zhu, S.-Y. Wang, Z.-Y. Gu, X. Wang and S.-J. Ji, Chem. Commun., 2014, 50, 6439 RSC; (c) L. Wang, W. X. Sha, Q. Dai, X. M. Feng, W. T. Wu, H. B. Peng, B. Chen and J. Cheng, Org. Lett., 2014, 16, 2088 CrossRef CAS PubMed.
- (a) W. X. Sha, J.-T. Yu, Y. Jiang, H. T. Yang and J. Cheng, Chem. Commun., 2014, 50, 9179 RSC; (b) Z.-Q. Zhu, T.-T. Wang, P. Bai and Z.-Z. Huang, Org. Biomol. Chem., 2014, 12, 5839 RSC.
- (a) T. B. Xiao, L. Y. Li, G. L. Lin, Q. L. Wang, P. Zhang, Z.-W. Mao and L. Zhou, Green Chem., 2014, 16, 2418 RSC.
- (a) C. D. Pan, J. Han, H. L. Zhang and C. J. Zhu, J. Org. Chem., 2014, 79, 5374 CrossRef CAS PubMed; (b) X. Q. Li, M. W. Fang, P. Z. Hu, G. Hong, Y. C. Tang and X. S. Xu, Adv. Synth. Catal., 2014, 356, 2103 CrossRef CAS; (c) G. Wang, S.-Y. Chen and X.-Q. Yu, Tetrahedron Lett., 2014, 55, 5338 CrossRef CAS.
- (a) T. Nakanishi, A. Masuda, M. Suwa, Y. Akiyama, N. Hoshino-Abe and M. Suzuki, Bioorg. Med. Chem. Lett., 2000, 10, 2321 CrossRef CAS PubMed; (b) O. B. Abdel-Halim, T. Morikawa, S. Ando, H. Matsuda and M. Yoshikawa, J. Nat. Prod., 2004, 67, 1119 CrossRef CAS PubMed; (c) T. Ishikawa, Med. Res. Rev., 2001, 21, 61 CrossRef CAS PubMed; (d) N. Stevens, N. O’Connor, H. Vishwasrao, D. Samaroo, E. R. Kandel, D. L. Akins, C. M. Drain and N. J. Turro, J. Am. Chem. Soc., 2008, 130, 7182 CrossRef CAS PubMed; (e) L. Sripada, J. A. Teske and A. Deiters, Org. Biomol. Chem., 2008, 6, 263 RSC.
- Selected examples see: (a) T. Furuya, A. S. Kamlet and T. Ritter, Nature, 2010, 473, 470 CrossRef PubMed; (b) K. Müller, C. Faeh and F. Diederich, Science, 2007, 317, 1881 CrossRef PubMed; (c) X.-H. Xu, K. Matsuzaki and N. Shibata, Chem. Rev., 2015, 115, 731 CrossRef CAS PubMed; (d) J. Nie, H. C. Guo, D. Cahard and J.-A. Ma, Chem. Rev., 2011, 111, 455 CrossRef CAS PubMed; (e) O. Tomashenko and V. V. Grushin, Chem. Rev., 2011, 111, 4475 CrossRef CAS PubMed; (f) S. Purser, P. R. Moore, S. Swallow and V. Governeur, Chem. Soc. Rev., 2008, 37, 320 RSC; (g) V. Matoušek, A. Togni, V. Bizet and D. Cahard, Org. Lett., 2011, 13, 5762 CrossRef PubMed; (h) X. Mu, T. Wu, H. Y. Wang, Y. L. Guo and G. S. Liu, J. Am. Chem. Soc., 2012, 134, 878 CrossRef CAS PubMed; (i) E. Merino and C. Nevado, Chem. Soc. Rev., 2014, 43, 6598 RSC; (j) Y. L. Liu, T. D. Shi, F. Zhou, X. L. Zhao, X. Wang and J. Zhou, Org. Lett., 2011, 13, 3826 CrossRef CAS PubMed; (k) L. L. Chu and F.-L. Qing, J. Am. Chem. Soc., 2010, 132, 7262 CrossRef CAS PubMed; (l) M. Y. Hu, C. F. Ni and J. B. Hu, J. Am. Chem. Soc., 2012, 134, 15257 CrossRef CAS PubMed; (m) X. Wang, Y. Xu, F. Y. Mo, G. J. Ji, D. Qiu, J. J. Feng, Y. X. Ye, S. N. Zhang, Y. Zhang and J. B. Wang, J. Am. Chem. Soc., 2013, 135, 10330 CrossRef CAS PubMed; (n) H. Egami and M. Sodeoka, Angew. Chem., Int. Ed., 2014, 53, 8294 CrossRef CAS PubMed.
- (a) Y. J. Li, J. T. Zhu, L. S. Zhang, Y. M. Wu and Y. F. Gong, Chem.–Eur. J., 2013, 19, 8294 CrossRef CAS PubMed; (b) W.-Y. Wang, X. Feng, B.-L. Hu, C. L. Deng and X.-G. Zhang, J. Org. Chem., 2013, 78, 6025 CrossRef CAS PubMed; (c) W. J. Fu, M. Zhu, F. J. Xu, Y. Q. Fu, C. Xu and D. P. Zou, RSC Adv., 2014, 4, 17226 RSC.
- B. Zhang, C. Mück-Lichtenfeld, C. G. Daniliuc and A. Studer, Angew. Chem., Int. Ed., 2013, 52, 10792 CrossRef CAS PubMed.
- Q. L. Wang, X. C. Dong, T. B. Xiao and L. Zhou, Org. Lett., 2013, 15, 4846 CrossRef CAS PubMed.
- B. Zhang and A. Studer, Org. Lett., 2014, 16, 3990 CrossRef CAS PubMed.
- (a) Y. Ji, T. Brueckl, R. D. Baxter, Y. Fujiwara, I. B. Seiple, S. Su, D. G. Blackmond and P. S. Baran, Proc. Natl. Acad. Sci. U. S. A., 2011, 108, 14411 CrossRef CAS PubMed; (b) Y. D. Ye, S. H. Lee and M. S. Sanford, Org. Lett., 2011, 13, 5464 CrossRef CAS PubMed; (c) A. Hafner and S. Bräse, Angew. Chem., Int. Ed., 2012, 51, 3713 CrossRef CAS PubMed; (d) C. P. Zhang, Z. L. Wang, Q. Y. Chen, C. T. Zhang, Y. C. Gu and J. C. Xiao, Angew. Chem., Int. Ed., 2011, 50, 1896 CrossRef CAS PubMed; (e) C. P. Zhang, Z. L. Wang, Q. Y. Chen, C. T. Zhang, Y. C. Gu and J. C. Xiao, Chem. Commun., 2011, 47, 6632 RSC; (f) D. A. Nagib, M. E. Scott and D. W. C. MacMillan, J. Am. Chem. Soc., 2009, 131, 10875 CrossRef CAS PubMed; (g) A. T. Parsons and S. L. Buchwald, Angew. Chem., Int. Ed., 2011, 50, 9120 CrossRef CAS PubMed.
- (a) S. I. Troyanov, A. Dimitrov and E. Kemnitz, Angew. Chem., Int. Ed., 2006, 45, 1971 CrossRef CAS PubMed; (b) M. Oudrhiri-Hassani, D. Brunel, A. Germain and A. Commeyras, J. Fluorine Chem., 1984, 25, 491 CrossRef CAS.
- C. B. Liu, W. W. Zhao, Y. Huang, H. M. Wang and B. Zhang, Tetrahedron, 2015, 71, 4344 CrossRef CAS.
- Selected examples see: (a) Q.-Y. Meng, J.-J. Zhong, Q. Liu, X.-W. Gao, H.-H. Zhang, T. Lei, Z.-J. Li, K. Feng, B. Chen, C.-H. Tung and L.-Z. Wu, J. Am. Chem. Soc., 2013, 135, 19052 CrossRef CAS PubMed; (b) C. K. Prier, D. A. Rankic and D. W. C. MacMillan, Chem. Rev., 2013, 113, 5322 CrossRef CAS PubMed; (c) J. M. R. Narayanam and C. R. J. Stephenson, Chem. Soc. Rev., 2011, 40, 102 RSC; (d) L. J. Allen, P. J. Cabrera, M. Lee and M. S. Sanford, J. Am. Chem. Soc., 2014, 136, 5607 CrossRef CAS PubMed; (e) S. Q. Zhu, A. Das, L. Bui, H. J. Zhou, D. P. Curran and M. Rueping, J. Am. Chem. Soc., 2013, 135, 1823 CrossRef CAS PubMed; (f) N. Iqbal, J. Jung, S. Park and E. J. Cho, Angew. Chem., Int. Ed., 2014, 53, 539 CrossRef CAS PubMed; (g) D. J. Wilger, N. J. Gesmundo and D. A. Nicewicz, Chem. Sci., 2013, 4, 3160 RSC; (h) M. Fagnoni, D. Dondi, D. Ravelli and A. Albini, Chem. Rev., 2007, 107, 2725 CrossRef CAS PubMed; (i) J. Xuan and W. J. Xiao, Angew. Chem., Int. Ed., 2012, 51, 6828 CrossRef CAS PubMed; (j) Y. Yasu, T. Koike and M. Akita, Org. Lett., 2013, 15, 2136 CrossRef CAS PubMed; (k) H. Jiang, Y. Z. Cheng, R. Z. Wang, M. M. Zheng, Y. Zhang and S. Y. Yu, Angew. Chem., Int. Ed., 2013, 52, 13289 CrossRef CAS PubMed; (l) S. Fukuzumi and K. Ohkubo, Chem. Sci., 2013, 4, 561 RSC; (m) J. Xie, Q. C. Xue, H. M. Jin, H. M. Li, Y. X. Cheng and C. J. Zhu, Chem. Sci., 2013, 4, 1281 RSC; (n) E. Arceo, I. D. Jurberg, A. Álvarez-Fernández and P. Melchiorre, Nat. Chem., 2013, 5, 750 CrossRef CAS PubMed; (o) L. W. Zheng, C. Yang, Z. Z. Xu, F. Gao and W. J. Xia, J. Org. Chem., 2015, 80, 5730 CrossRef CAS PubMed.
- Y. Z. Cheng, H. Jiang, Y. Zhang and S. Y. Yu, Org. Lett., 2013, 15, 5520 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental procedures and full spectroscopic data for all compounds. See DOI: 10.1039/c5ra16645d |
| This journal is © The Royal Society of Chemistry 2015 |