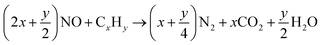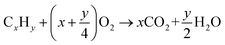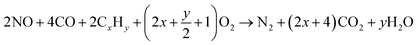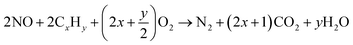Noble metal ions in CeO2 and TiO2: synthesis, structure and catalytic properties
Parthasarathi Bera
*a and
M. S. Hegde
b
aSurface Engineering Division, CSIR – National Aerospace Laboratories, Bangalore 560017, India. E-mail: partho@nal.res.in; Fax: +91-80-25210113; Tel: +91-80-25086359
bSolid State and Structural Chemistry Unit, Indian Institute of Science, Bangalore 560012, India
First published on 8th October 2015
Abstract
In the past four decades, CeO2 has been recognized as an attractive material in the area of auto exhaust catalysis because of its unique redox properties. In the presence of CeO2, the catalytic activity of noble metals supported on Al2O3 is enhanced due to higher dispersion of noble metals in their ionic form. In the last few years, we have been exploring an entirely new approach of dispersing noble metal ions on CeO2 and TiO2 matrices for redox catalysis. In this study, the dispersion of noble metal ions by solution combustion as well as other methods over CeO2 and TiO2 resulting mainly in Ce1−xMxO2−δ, Ce1−x−yTixMyO2−δ, Ce1−x−ySnxMyO2−δ, Ce1−x−yFexMyO2−δ, Ce1−x−yZrxMyO2−δ and Ti1−xMxO2−δ (M = Pd, Pt, Rh and Ru) catalysts, the structure of these materials, their catalytic properties toward different types of catalysis, structure–property relationships and mechanisms of catalytic reactions are reviewed. In these catalysts, noble metal ions are incorporated into a substrate matrix to a certain limit in a solid solution form. Lower valent noble metal-ion substitution in CeO2 and TiO2 creates noble metal ionic sites and oxide ion vacancies that act as adsorption sites for redox catalysis. It has been demonstrated that these new generation noble metal ionic catalysts (NMIC) have been found to be catalytically more active than conventional nanocrystalline noble metal catalysts dispersed on oxide supports.
1. Introduction
Catalysis has widespread applications from environment clean-up to manufacturing various chemicals, energy production, food processing and biological processes. The production of most of the useful commercial bulk and fine chemicals involves catalysis.1–9 The science and technology of heterogeneous catalysis is one of the rapidly moving frontiers in the area of catalysis as well as chemical sciences. The synthesis of ammonia, sulfuric acid and nitric acid, the production of hydrogen, the control of exhaust emissions, ammoxidation, methanol synthesis and many other important reactions of the present day are associated with heterogeneous catalysis. Metals, metal clusters, alloys, oxides, sulfides, solid acids and bases have been used as catalysts for several catalytic reactions since the early days of heterogeneous catalysis.1 Among these catalysts, supported and unsupported metals have been regarded as efficient catalysts for their usefulness in different catalytic reactions. Extensive research and investigations, such as metal–support interactions, structure–catalytic property relations, metal dispersion, redispersion, and synergistic effects, have been carried out over these catalysts for decades.Since the early days of catalysis, it has been established that metals, in general, are active sites for the adsorption of reactant molecules and subsequent catalysis, and therefore most catalysts contain nanocrystalline metals, especially noble metals dispersed on supports like Al2O3 or SiO2.1,10,11 However, in these catalysts, noble metal atoms present on the surfaces of the metal nanoparticles in their zero valent state serve as active sites for both oxidizing and reducing molecules and only 1/4 or 1/5 of the total number of noble metals are utilized for adsorption and subsequent catalytic conversion in 4–6 nm metal particles. For example, if a 5 nm Pt particle is considered, then the total number of Pt atoms in a 5 nm cubic Pt metal crystal is 8365, taking four atoms per unit cell of Pt (fcc) metal. Again, the total number of metal atoms on the 6 surfaces of a 5 nm Pt metal cube is 1962 with two atoms per unit cell surface. Pt metal particles are anchored on a solid substrate surface. Therefore, five faces of a Pt metal crystal with 1635 surface atoms are exposed. This indicates that 1/5th of the total number of Pt atoms are available for adsorption and catalysis when reactant molecules are exposed on the catalyst surface and 4/5th of the atoms inside the metal nanocrystal are not available for adsorption or they are wasted. If all the Pt atoms are utilized, then the reaction rate would be 5 times higher, and thus catalytic activity would increase with the increase in metal dispersion. However, the atomic dispersion of Pt and other noble metals on traditional supports, such as Al2O3 and SiO2, is difficult because metal atoms sinter into metal particles due to metal–metal bonding. Are there alternative ways to utilize all the Pt atoms present in the catalyst? One possible way to keep the Pt metal atoms apart is to convert Pt atoms (Pt0) into Pt ions (Pt2+) and keep them separated from O2− ions. Pt2+ ions cannot come closer to form Pt crystals. If Pt2+ ions act as adsorption centers as well as Pt atoms, then in principle, all the metal atoms become available for adsorption.
Auto exhaust catalysts are mainly dispersed noble metals, such as Pt, Pd and Rh, on oxide supports like Al2O3 in the form of nanocrystalline metal particles.12 A huge increase in the dispersion of noble metals has been observed with the addition of CeO2 to Al2O3, leading to an increase in oxygen storage capacity (OSC) and auto exhaust catalytic activity.12–18 Even though oxidized Pt, Pd and Rh are found on the Al2O3/CeO2 surface, the reasons for higher dispersion of noble metals in the form of ions in the presence of CeO2 have not been understood. In the early 1990s, Sayle and coworkers demonstrated the segregation of noble metal ions, such as Pd2+, Pt2+ and Rh3+, at the surface of CeO2 with oxygen vacancies by computer simulation studies.19 Theoretically, noble metal ions can be stable in a CeO2 matrix, leading to the formation of defects on the surface. Therefore, complete dispersion of noble metals can be achieved in terms of ions within reducible oxide supports such as CeO2 and TiO2. In this regard, entirely new generation catalysts that can be useful for multipurpose applications in different catalytic reactions have been developed in our laboratory in the last 15 years. It has been found that an increase in the dispersion of noble metals is possible through the oxidation of noble metals into their ions, which are dispersed on the reducible oxide surface, while simultaneously reducing the oxide support.20–24 In other words, direct substitution of noble metal ions in reducible supports, such as CeO2 and TiO2, results in uniform solid catalysts in the form of their solid solutions. Thus, a new idea in heterogeneous catalysis has been conceptualized wherein dispersed metal ions act as catalytically active sites for heterogeneous catalysis. In the last few years, significant studies on these types of ionically dispersed catalysts have been done by other groups. Research studies on supported gold catalysts by Corma's and Gates' groups have demonstrated that gold is mainly present in its ionic form in these catalysts. They have reviewed the significance of cationic gold in different matrices with respect to several organic reactions and CO oxidation.25,26 Flytzani-Stephanopoulos and Gates have discussed key findings of cationic noble metals as catalytic sites on solid supports.27
Noble metal oxides, such as PtO, PtO2, PdO and Rh2O3, are known, and therefore it should be possible to synthesize solid solutions between CeO2 or TiO2 and noble metal oxides. Noble metal loading for several catalytic reactions is only to the extent of 1–2 at%, and therefore substitution of only 1–2 at% of noble metal ions in CeO2 is sufficient to develop a catalyst. The underlying principle of doping aliovalent metal ions into a CeO2 or TiO2 lattice is to retain their parent structures. In this sense, new-age advanced catalysts, such as Ce1−xMxO2−δ (M = Pd, Pt, Rh, Cu, Ag and Au), Ce1−x−yAxMyO2−δ (A = Ti, Zr, Sn and Fe; M = Pt and Pd) and Ti1−xMxO2−δ (M = Pd, Pt, Rh and Ru), that retained their parent fluorite and anatase structures have been prepared in our laboratory, employing a solution combustion method.20–23 Thus, the synthesis of these single-phase oxides is a new concept that is the basis of metal ion catalysts. In these catalysts, the noble metal ions and corresponding oxide ion vacancies are supposed to be the active sites. Because the adsorption sites in these catalysts are noble metal ions, such as Pd2+, Pt2+, Rh3+ and Ru4+, we have named these materials as noble metal ionic catalysts (NMIC). In the last few years, we have been studying several catalytic exhaust reactions, such as NO reduction, CO and hydrocarbon oxidation, selective catalytic reduction of NO, three-way catalytic reactions, water gas shift (WGS) reactions, preferential oxidation of CO (CO-PROX), H2 + O2 recombination reactions, hydrogenation and Heck reaction, over these noble metal ionic catalysts and it has been demonstrated that these catalysts are much more active for several catalytic reactions compared to other conventional supported metal catalysts. In this review, at first, we mainly discuss the background, synthesis and characterization of these noble metal ionic catalysts (NMIC). Then, we emphasize on our detailed study related to exhaust catalysis and comparing activities with several catalysts reported in the literature. Furthermore, we write about our studies on the water gas shift reaction, the CO-PROX reaction, the H2 + O2 reaction, hydrogenation, Heck reaction and unique reaction mechanisms over these catalysts. Metal–support interactions and synergistic effects of the interactions that lead to the superiority of these catalysts over conventional catalysts are also discussed. We also focus on our density functional theory (DFT) studies for high oxygen storage capacity (OSC) and the catalytic properties of several CeO2 and TiO2-based materials. Finally, we conclude our review by focusing on the future prospects of these noble metal ionic catalysts.
2. Synthesis of noble metal ionic catalysts
2.1. Conventional preparation methods of catalysts
The dispersion of metals over suitable supports is one of the important issues for higher catalytic activities. Higher the dispersion higher is the catalytic activity. Dispersed noble metals and transition metal ions on a particular support are the active sites for adsorption of CO, NO, hydrocarbons and oxygen. In this sense, the method of catalyst preparation can influence the dispersion characteristics of metals on the supports and their catalytic behavior. The main objective of the preparation of a supported metal catalyst is to obtain the maximum dispersion of catalytically active species over the support material from its precursor at a particular concentration. The catalysts for fundamental studies are usually prepared by coprecipitation, sol–gel, deposition, impregnation, ion exchange, anchoring–grafting and spreading–wetting methods.28,29 In general, the dispersion of active components is carried out over the oxide support in a single-step preparation, such as a coprecipitation or sol–gel process, whereas dispersion has been done over an independently prepared oxide support by impregnation, deposition, or incipient wetness methods. Some of these preparation procedures are briefly described below.(a) Coprecipitation – this involves a reaction between a solution of two or more metal salts in the presence of a base such as hydroxide, alkaline carbonate and bicarbonate. For example, a Ni/Al2O3 catalyst is prepared from Ni(NO3)2 and AlCl3 in the presence of NaHCO3.29
(b) Sol–gel – this process describes the transition of a system from a mostly colloidal liquid into a solid or a gel phase. This is based on the polymerization of molecular precursors such as metal alkoxides. Hydrolysis and condensation of these alkoxides lead to the formation of oxopolymers, which are then transformed into a homogeneous oxide network. Pd/SiO2, Pt/SiO2 and Pd/Al2O3 catalysts are prepared by this method.30
(c) Deposition – this describes the addition of a catalytically active component to a separately produced support. There are two types of deposition.
(i) Deposition–precipitation – a precipitating agent is added to a suspension of the support in an active metal salt solution. Accordingly, the active metal species is adsorbed onto the surface of the support. A Ni/Al2O3 catalyst is prepared by the slow addition of NaOH to a suspension of Al2O3 in NiCl2 solution. This process is used to synthesize nickel supported on silica, alumina, magnesia, titania, thoria, ceria, zinc oxide and chromium oxide.31 This process is also used for the preparation of Pd(OH)2/C, Rh(OH)3/C and Ru(OH)3/C.
(ii) Deposition–reduction – addition of a reducing agent to a suspension of the support in a solution of the active metal salt provides supported reduced metal catalysts. Pd/C and Pt/C can be prepared by this method. In general, hydrogen, formaldehyde, hydrazine and borohydride are used as the reducing agents.29
(d) Impregnation – this involves introducing a precursor solution into the pore space of a support that is filled with the same solvent as the precursor solution. Herein, the concentration gradient is the driving force. It is also called wet or diffusional impregnation. Rh/TiO2, Rh/Al2O3, Pt/C and Ru/MgO are prepared by this method.29
(e) Incipient wetness – this is a type of impregnation process in which a precursor solution is introduced into contact with the previously dried support. It is also called dry or pore-volume impregnation. It is an exothermic process and it depends on capillary pressure developed in the pores and the rate of the impregnation process. Ni/Al2O3, Pt/SiO2, Pt/Al2O3, Ni/SiO2, Ni/C, Ru/SiO2, Rh/SiO2 and Ru/C are prepared by this method.29
(f) Ion exchange – in this method, an ion in an electrostatic interaction with the surface of a support is replaced by another ionic species. For example, zeolites, clays and silicates are cation exchangers, whereas hydrotalcites are anion exchangers. The overall charge of a zeolite is negative and is distributed over the oxygen atom; this charge is neutralized by various cations such as Na+ and K+.29
(g) Microemulsion – this is a physical mixture of two immiscible liquids along with a surfactant. Reverse micelles formed can act as micro-reactors. These reactors are utilized for the synthesis of nanoparticles. Nanocrystalline CeO2 was prepared by water-in-oil micro-emulsion methods.32 CeO2–ZrO2 ultrafine particles were also prepared by the micro-emulsion method.33
(h) Mechanical milling – this is a solid-to-solid synthetic method. Mechanical milling has been used extensively to synthesize CeO2–MOx (M = Zr, Hf, Tb, Cu and Mn) mixed oxide catalysts.34,35 The main feature of this method is to obtain a powder consisting of small crystallites with a size of a few nanometers along with a high concentration of lattice defects. Mechanical alloying has also been applied to prepare mixed oxides containing CeO2 to enhance the catalysis. High-energy mechanical alloying of pure CeO2 and ZrO2 at room temperature results in the formation of a single-phase fluorite-structured solid solution of Ce1−xZrxO2 in all the examined composition ranges.
In general, most of the abovementioned conventional preparation methods are quite involved and require long processing times. The post preparation steps include washing, drying, H2 reduction and removal of Cl− ions by heating in steam.
2.2. Solution combustion method
In the last few years, there has been a trend of novel chemical routes of synthesis that lead to ultra-fine and high surface area catalysts for heterogeneous catalysis. The solution combustion method has been found to be unique for obtaining nanocrystalline oxide materials.36,37 In general, combustion is a chemical reaction that is accompanied by heat and light. Many exothermic, non-catalytic, solid–solid or solid–gas reactions liberate enough heat to allow them to self-propagate after being ignited. The reactions, ignited locally by an external energy source with short term service, may propagate throughout the sample at a certain rate and occur in a narrow zone, which separates the starting substances and reactions. In the literature, a variety of names have been given to redox reactions employed for the synthesis of different materials. Some of the fashionable names are furnaceless or fire synthesis, self-propagating chemical decomposition (SCD), self-propagating high temperature synthesis (SHS) and the combustion method. Merzhanov has introduced the process of SHS wherein he has employed metals and organic fuels to produce oxides, carbides and sulfides.38 Patil and coworkers have developed a solution combustion method involving metal salts, mainly nitrates and organic fuels.39,40 Metal salts are easily available, and therefore this method provides a better alternative for the synthesis of a variety of complex oxides. Thus, the solution combustion method is useful for preparing high-surface-area oxides.The solution combustion method involves rapid heating at a particular temperature of an aqueous redox mixture containing stoichiometric amounts of the corresponding metal salts and hydrazine-based fuels and the product is formed within 5 min. Redox compounds or mixtures containing hydrazide groups has been found useful in the solution combustion synthesis of oxide materials. An ideal fuel should be water-soluble and has a low ignition temperature (<500 °C). It should be compatible with the transition metal nitrate so that the combustion can be controlled, carried out smoothly and does not lead to explosion. The merits of the solution combustion technique are as follows: (a) being a solution process, it has all the advantages of a wet chemical process such as control of stoichiometry, the doping of a desired amount of impurity ions and the formation of nano-sized particles (b) it is a low-temperature initiated process, (c) it is highly exothermic due to the redox reaction, (d) it is self-propagating, (e) it involves a transient high temperature, (f) it involves the production of a huge amount of gases, (g) it is simple, fast and economical and (h) it involves the formation of a high surface area, voluminous and homogeneous product. Using this method, a variety of oxide materials, such as α-Al2O3, ZnO, Y2O3, CeO2, γ-Fe2O3, Cr2O3, MgFe2O4, MnFe2O4, CoFe2O4, PbTiO3, PbZrO3 and BaFe12O19, have been prepared. The solution combustion method not only yields nano-sized TiO2, ZrO2 and hexaferrites, but it also yields metastable phases such as γ-Fe2O3, t-ZrO2 and anatase TiO2. Several supported, unsupported and doped oxide catalysts have been synthesized by this method. In a recent review, González-Cortés and Imbert have discussed current approaches and future possibilities for developing advanced solid catalysts using solution combustion synthesis.41 Fig. 1 shows a typical sequence of the formation of a catalyst by the solution combustion process.
 | ||
| Fig. 1 Sequences of solution combustion synthesis: initial solution of precursors, flame during combustion and final product. Reprinted from ref. 21 with the permission of American Chemical Society. | ||
The substitution of noble metal ions in CeO2, achieved for the first time by the solution combustion method, was an accidental discovery.20 When an aqueous solution containing stoichiometric amounts of Al(NO3)3, H2PtCl6 and urea (CON2H4) is heated rapidly at 500 °C, the solution boils, froths and burns with a flame temperature of 1500 °C, yielding nano-sized Pt metal particles dispersed on α-Al2O3.42 In contrast, Pt metal particles are not formed when a similar attempt has been made to disperse 1–2 at% Pt metal on CeO2 by the combustion of ceric ammonium nitrate [(NH4)2Ce(NO3)6], chloroplatinic acid (H2PtCl6) and oxalyl dihydrazide (C2H6N4O2, ODH) heated at 350 °C. Instead of nano-sized Pt metal particles dispersed on CeO2, 25–30 nm Ce1−xPtxO2−δ nanocrystallites have been formed, wherein Pt has been observed to be present in the +2 and +4 oxidation states. A typical reaction for the preparation of a Ce1−xPtxO2−x catalyst is given below:
| 5(1 − x)(NH4)2Ce(NO3)6 + 5xH2PtCl6 + 12(1 − x)C2H6N4O2 → 5Ce1−xPtxO2−x + 24(1 − x)CO2 + 44(1 − x)N2 + (56 − 66x)H2O + 10xHCl. |
Similarly, Pd, Rh, Ag and Au-substituted Ce1−xMxO2−δ catalysts have been prepared by this method using PdCl2, RhCl3, AgNO3 and HAuCl4 as respective metal precursors.20,43–46 Ce1−x−yTixMyO2−δ (M = Pt and Pd), Ce1−x−ySnxPdyO2−δ, Ce1−x−yFexPdyO2−δ, Ce1−x−yZrxPdyO2−δ and Ce1−x−yHfxPdyO2−δ catalysts have also been prepared with a combustion mixture of (NH4)2Ce(NO3)6 with TiO(NO3)2 or SnC2O4 or Fe(NO3)3 or ZrO(NO3)3 or Hf(NO3)4, H2PtCl6, PdCl2 and C2H5NO2.47–52
The combustion of TiO(NO3)2, noble metal salts and glycine (C2H5NO2) fuel has been employed for the preparation of noble metal ion-doped TiO2 catalysts in the form of a Ti1−xMxO2−x (M = Pd, Pt, Rh and Rh) solid solution.53–56 A typical reaction for the preparation of Ti1−xPdxO2−x catalyst is given below:
| 9(1 − x)TiO(NO3)2 + 10(1 − x)C2H5NO2 + 9xPdCl2 → 9Ti1−xPdxO2−x + 20(1 − x)CO2 + 14(1 − x)N2 + (25 − 34x)H2O + 18HCl. |
2.3. Hydrothermal method
The term ‘hydrothermal’ is of geological origin. Many materials, including zeolites, were formed in the Earth's crust under high temperature and high pressure. The hydrothermal method has been introduced by Barrer to synthesize zeolites.57 The hydrothermal method utilizes water under pressure and at temperatures above its boiling point as a means of speeding up the reaction between solids. Under hydrothermal conditions, the reaction occurs at a relatively low temperature (∼150–200 °C); otherwise, a much higher temperature is required. Because the hydrothermal reaction must be carried out in a closed vessel, the temperature–pressure relationship is very important. Among all these preparation methods, hydrothermal synthesis always offers an extra merit in preparing highly crystallized nanomaterials with controlled shape, size and orientation. In this method, metal salts are dissolved in water and mixed with structure-directing reagents, generally, amines that form a gel. Gel is used in a vessel called a hydrothermal bomb, which is a thick-walled steel vessel with a hermetic seal. Inside the vessel, containers made of Teflon are filled with the gel to the extent of 75%. The pressure vessel is closed and heated in an oven to 200 °C for 24–48 h. Nanostructural CeO2 and TiO2 were prepared via an alkaline hydrothermal route.58,59 The hydrothermal method is one of the most popular techniques for preparing metal-doped CeO2 materials such as Ce1−xMxO2−δ (M = Ca and Sm).60 Several Ce1−xMxO2−δ (M = Zr, Ti, Pr, Y, Fe, Cr and Ru) catalysts have been prepared using the hydrothermal method in our laboratory.61–632.4. Sonochemical method
Ultrasound radiation is used mostly for the diagnosis of diseases, whereas focused ultrasound radiation has been used to burn cancer cells. Ultrasonic radiation is utilized to break chemical bonds to produce nanoparticles. Gedanken has pioneered the sonochemical method to prepare a number of oxide nanomaterials such as CeO2, ZnO and RVO4 (R = La, Ce, Nd, Sm, Eu and Gd).64–66 Ceria nanoparticles embedded in poly methyl methacrylate (PMMA) have been synthesized using the sonochemical method.67 Pt-supported TiO2@C core–shell composites have been synthesized by a sonochemical route for methanol electrooxidation applications.68 This method is especially suitable for making Fe and noble metal ion-substituted CeO2 and TiO2 catalysts. In our laboratory, Ce1−xPtxO2−δ, Ce1−xFexO2−δ and Ce1−x−yFexPdyO2−δ catalysts have been synthesized by the sonochemical method.69,70Noble as well as transition metal ions have been substituted into CeO2 and TiO2 matrices by solution combustion, hydrothermal and sonochemical methods successfully. Table 1 summarizes the metal ion-substituted CeO2 and TiO2 materials synthesized in our laboratory.20,43–56,61–63,69–75
| Metal ions | Support oxides | |||||
|---|---|---|---|---|---|---|
| CeO2 | Ce1−xTixO2 | Ce1−xSnxO2 | Ce1−xFexO2−δ | Ce1−xZrxO2 | TiO2 | |
| Pd2+ | Ce1−xPdxO2−δ (x = 0.01, 0.02) (20, 43) | Ce0.75−xTi0.25PdxO2−δ (x = 0.01, 0.02) (48) | Ce0.78Sn0.2Pd0.02O2−δ (49, 50) | Ce1−x−yFexPdyO2−δ (x = 0.1–0.45, y = 0.02) (51, 70) | Ce1−x−yZrxPdyO2−δ (x = 0.25, y = 0.02) (52) | Ti1−xPdxO2−δ (x = 0.01, 0.03) (54, 55) |
| Rh3+ | Ce1−xRhxO2−δ (x = 0.005, 0.01) (44) | Ti1−xRhxO2−δ (x = 0.01) (54) | ||||
| Pt2+, Pt4+ | Ce1−xPtxO2−δ (x = 0.01, 0.02) (20, 69, 71, 72) | Ce0.85−xTi0.15PtxO2−δ (x = 0.01, 0.02) (47) | Ti1−xPtxO2−δ (x = 0.01) (54) | |||
| Pt2+, Rh3+ | Ce1−xRhx/2Ptx/2O2−δ (x = 0.01, 0.02) (73) | |||||
| Ru4+ | Ce1−xRuxO2−δ (x = 0.05, 0.1) (63) | Ti1−xRuxO2−δ (x = 0.01) (54) | ||||
| Cu2+ | Ce1−xCuxO2−δ (x = 0.01–0.1) (74, 75) | |||||
| Ag+ | Ce1−xAgxO2−δ (x = 0.01) (45) | |||||
| Au3+ | Ce1−xAuxO2−δ (x = 0.01) (46) | |||||
| Cr4+, Cr6+ | Ce1−xCrxO2+δ (x = 0.01–0.2) (62) | |||||
3. Structure of Ce1−xMxO2−δ and Ti1−xMxO2−δ-based catalysts
Several physical characterization techniques, such as X-ray diffraction (XRD), transmission electron microscopy (TEM), X-ray photoelectron spectroscopy (XPS) and X-ray absorption fine structure spectroscopy (XAFS), have been employed to study the crystal structure, morphology, electronic structure, local structure and oxide ion vacancies of Ce1−xMxO2−δ and Ti1−xMxO2−δ-based catalysts. The main findings of these studies are discussed in the following subsections.3.1. XRD studies
High resolution XRD data (Rigaku-2000, PANalytical X'Pert PRO) demonstrate that CeO2 crystallizes in the fluorite structure in all CeO2-based catalysts, where Ce4+ ions are cubic close-packed with all tetrahedral sites occupied by oxygen. There are no impurity peaks related to any platinum oxides in the Ce0.99Pt0.01O2−δ catalyst. However, a small, broad hump at 2θ = 39° due to Pt(111) is observed in the diffraction pattern, indicating the presence of trace amounts of Pt in the catalyst. Comparing the intensity ratio of Pt(111) in Ce0.99Pt0.01O2−δ with that in a 1% Pt + CeO2 physical mixture, it has been evaluated that at least 92 at% of the platinum used in the preparation of Ce0.99Pt0.01O2−δ is incorporated into the CeO2 lattice.71,72 The total oxygen content in Ce0.99Pt0.01O2−δ is 1.883 and that in pure CeO2 is 1.934 and a decrease in the lattice parameter is observed in Ce0.99Pt0.01O2−δ compared to pure CeO2. Similarly, diffraction lines are indexed to the fluorite structure in Ce0.99Pd0.01O2−δ and Ce0.98Pd0.02O2−δ, and lines corresponding to Pd or PdO are not observed in their XRD patterns. Even a slow scan in the Pd(111) region (2θ = 40° ± 5°) does not show any indication of a Pd metal peak.43 On the other hand, a 1% Pd/CeO2 catalyst prepared by the impregnation method shows diffraction lines due to Pd metal. The lattice parameter decreases from 5.4113(2) Å in pure CeO2 to 5.4107(3) Å in Ce0.98Pd0.02O2−δ, confirming the substitution of Pd2+ ions (0.84 Å) for Ce4+ ions (0.99 Å) in CeO2. In a Ce0.99Rh0.01O2−δ catalyst, diffraction lines corresponding to Rh metal, Rh2O3 and RhO2 are not detected. The total oxygen content in a Ce0.99Rh0.01O2−δ catalyst is 1.87, whereas it is 1.934 for pure CeO2.44 Therefore, a decrease in the oxygen content in the catalyst compared with pure CeO2 signifies the creation of an oxide ion vacancy. Thus, detailed XRD studies demonstrate the formation of Ce1−xMxO2−δ (M = Pd, Rh, Pt, Ru, Cu, Ag and Au), Ce1−x−yTixMyO2−δ (M = Pd and Pt), Ce1−x−ySnxPdyO2−δ, Ce1−x−yFexPdyO2−δ and Ce1−x−yZrxPdyO2−δ solid solution phases.43–52,63,69–75 Similarly, solution combustion-synthesized Ti1−xMxO2−δ (M = Pd, Rh, Ru and Pt) catalysts crystallize in the anatase structure.53–56 All the catalysts show broad lines in their XRD patterns. A rutile contribution has been observed to the extent of 10% only for Ti1−xRuxO2−δ. No diffraction lines due to noble metals or their oxides are observed in Ti1−xMxO2−δ. Rietveld refinement of these catalysts reveals a slight increase in cell volume for Ti1−xPtxO2−δ due to a slight increase in the ionic radius of Pt; however, there is almost no change in cell volume or cell parameters for Pd2+, Rh3+ and Ru4+ ion-doped TiO2. The particle sizes measured from the Scherrer formula are in the range of 10–15 nm for the four catalysts. Typical Rietveld-refined XRD patterns of Ce0.99Pt0.01O2−δ and Ti0.98Pd0.02O2−δ are shown in Fig. 2. | ||
| Fig. 2 Rietveld refined XRD patterns of (a) Ce0.99Pt0.01O2−δ and (b) Ti0.99Pd0.01O2−δ. Reprinted from ref. 71 with the permission of American Chemical Society and from ref. 142 with the permission of Elsevier B. V. | ||
XRD patterns of hydrothermally prepared Ce1−xMxO2−δ (M = Zr, Ti, Pr, Y, Fe and Cr) catalysts are indexed to the fluorite structure and there are no traces of the respective oxides in their patterns.61,62 Similarly, diffraction lines associated with RuO2 and Ru metal are not observed in the XRD patterns of Ce1−xRuxO2−δ (0 ≤ x ≤ 0.1) prepared by the hydrothermal method and they show the fluorite structure.63 Ru and RuO2 impurity peaks are observed in the XRD pattern of Ce1−xRuxO2−δ prepared with x = 0.15. Thus, only up to 10% Ru4+ ions can be substituted for Ce4+ in CeO2 by this method.
Diffraction lines related to Pt and Pd metals or PtO2 and PdO impurities are not observed in the XRD patterns of Ce1−xMxO2−δ (M = Pt and Pd; x = 0.02, 0.05 and 0.10) synthesized by the sonochemical method, whereas all the peaks are indexed to the cubic fluorite lattice, indicating the substitution of Pt2+ and Pd2+ ions into the CeO2 lattice to a certain extent.70
Li and coworkers have demonstrated the absence of Pd metal up to 2.5 wt% metal loading in the XRD pattern of mesoporous Pd/CeO2.76 Pt metal is not found in combustion-synthesized 1 at% Pt/CeO2, as shown by Tang et al.77 Diffraction peaks related to Pd metal are not observed in sol–gel synthesized 1 at% Pd/CeO2 in the study done by Wang and coworkers.78 Pt metal and its oxides are absent in the XRD pattern of solution combustion-synthesized 2 at% Pt/CeO2, as shown by Bisht and coauthors.79 Pd metal is not observed in the XRD pattern of a 1 wt% Pd/CeO2–SiO2 catalyst.80 Misch et al. have demonstrated the formation of a single phase Ce1−xPdxO2−δ (x = 0.025, 0.05, 0.075, and 0.1) solid solution with a fluorite structure.81
3.2. TEM studies
High resolution TEM (FEI Technai 20) images of noble metal ion-substituted CeO2 and TiO2 catalysts have been obtained to understand their morphology and lattice fringes. CeO2-based catalysts show the cubic morphology of CeO2 crystallites with average sizes of 5–30 nm. Noble metal particles are not observed on the surface of CeO2 crystallites. A HRTEM image and the electron diffraction (ED) pattern of solution combustion synthesized Ce0.73Ti0.25Pd0.02O2−δ is displayed in the top panel of Fig. 3. The fringe spacing at ∼3.12 Å corresponds to the (111) layers of Ce0.75Ti0.25O2. The ED pattern clearly demonstrates the crystalline nature of the catalyst and it is indexed to the fluorite structure. Neither a lattice fringe in the HRTEM image nor a ring in the ED pattern due to Pd metal particles at 2.25 Å is observed. In contrast, the Pd(111) ring is visible as indicated by an arrow in the ED pattern of 2% Pd/Ce0.75Ti0.25O2 prepared by the impregnation method, shown in Fig. 3C of top panel. The absence of a diffraction ring due to Pd metal in the solution combustion-synthesized material shows substitution of Pd in the lattice, leading to a Ce0.73Ti0.25Pd0.02O2−δ solid solution.48 The average particle size of CeO2 crystallites obtained from the low magnification image is ∼20 nm. Similarly, lattice fringes that are 3.2 Å apart, corresponding to the (111) plane of Ce0.85Ti0.15O2 have been observed in a high resolution image of Pt ion-substituted Ce0.85Ti0.15O2 and fringes related to Pt metal particles at 2.30 Å are absent. Energy dispersive X-ray spectroscopy (EDXS) confirms the presence of 1% Pt in the catalyst and Pt ion substitution in Ce0.85Ti0.15O2.47 Lattice fringes in the HRTEM image of Ce0.89Fe0.1Pd0.01O2−δ are 3.1 Å apart, corresponding to its (111) plane.51 On the other hand, a large number of nano-sized fine Pt metal particles can be dispersed on α-Al2O3 by the solution combustion method.42,71 | ||
| Fig. 3 Top panel: HRTEM image of Ce0.73Ti0.25Pd0.02O2−δ (A) and ED patterns of Ce0.73Ti0.25Pd0.02O2−δ (B) and impregnated 2 at% Pd/Ce0.75Ti0.25O2 (C); bottom panel: (a) bright field TEM image of Ti0.97Pd0.03O2−δ and its ED pattern (inset), (b) HRTEM image of Ti0.97Pd0.03O2−δ and (c) EDXS of Ti0.97Pd0.03O2−δ from the image. Reprinted from ref. 48 and 55 with the permission of American Chemical Society. | ||
An HRTEM image and the corresponding ED pattern of a Ti0.97Pd0.03O2−δ catalyst are shown in the bottom panel of Fig. 3. Lattice fringes in the Ti0.97Pd0.03O2−δ catalyst, shown in the bottom panel of Fig. 3 (Fig. 3b) correspond to the (101) planes of TiO2. Lattice fringes due to PdO or Pd metals are not detected in the high resolution lattice images from the Ti0.97Pd0.03O2−δ catalyst. The EDX spectrum of this image shows the presence of Pd. The ED pattern is indexed to polycrystalline TiO2 in the anatase structure in both catalysts and there is no signature of Pd metal or PdO phases in their ED patterns.55 Therefore, the absence of Pd or PdO in Ti0.97Pd0.03O2−δ in the TEM image and the presence of Pd in the EDX spectrum confirm substitution of the Pd2+ ion in the TiO2 lattice. In an another study, the Pd metal diffraction line is observed in the ED pattern of 1% Pd/TiO2 prepared by impregnation.54 The particle sizes measured from the image are in the range of 8–15 nm, which agrees well with the XRD studies. Other noble metal-doped TiO2 catalysts show similar type of characteristics.
An HRTEM image of Ce0.9Ru0.1O2−δ synthesized by the hydrothermal method shows the absence of isolated RuO2 and Ru metal and the average crystallite size is 10 nm. The ring-type ED pattern is indexed to the fluorite structure.63
3.3. XPS studies
Oxidation states of Pt, Pd, Rh, Ru, Cu, Ag, Au, Ce and Ti present in the CeO2 and TiO2-based catalysts studied herein can be obtained from XPS (ESCA-3 Mark II, Thermo Fisher Scientific). Typical Pt4f and Rh3d core level spectra in reference compounds and CeO2-based catalysts are shown in Fig. 4. In Ce0.99Pt0.01O2−δ and Ce0.83Ti0.15Pt0.02O2−δ catalysts, Pt is present in both the +2 and +4 oxidation states, along with a small amount of Pt metal.20,71,72,47 XPS of the Pt4f core level peaks of Pt metal, PtO2, Ce0.99Pt0.01O2−δ and Ce0.83Ti0.15Pt0.02O2−δ is displayed in the left panel of Fig. 4. Rh is in the +3 oxidation state in the Ce1−xRhxO2−δ catalyst as observed in the Rh3d core level spectra.44 In the right panel of Fig. 4, Rh3d core level spectra in Rh, Rh2O3, RhCl3 and Ce1−xRhxO2−δ are displayed. Similarly, a core-level Pd3d5/2 peak is observed at 337.6 eV in Ce0.98Pd0.02O2−δ, which is 2.4 and 0.9 eV higher than in Pd metal and PdO.43 The binding energies of Pd3d peaks in Ce0.98Pd0.02O2−δ are close to the Pd3d peaks of PdCl2 and Pd(NO3)2, clearly indicating that Pd2+ ions are much more ionic in the CeO2 matrix than in PdO. Pd is observed to be in the high binding energy region in Ce0.98Pd0.02O2−δ. Pd is also observed to be in the +2 state in Ce0.73Ti0.25Pd0.02O2−δ, Ce0.78Sn0.2Pd0.02O2−δ, Ce0.89Fe0.1Pd0.01O2−δ and Ce0.73Zr0.25Pd0.02O2−δ.48–52 Therefore, XPS of Ce1−xPdxO2−δ-based catalysts indicates that Pd is present in the highly ionic Pd2+ state. Similarly, XPS results demonstrate that Cu in Ce1−xCuxO2−δ, Ag in Ce1−xAgxO2−δ and Au in Ce1−xAuxO2−δ are fully dispersed in the +2, +1 and +3 oxidation states, respectively.45,46,74,75 XP spectra of Ce3d with characteristic satellites in all catalysts show that Ce is mainly present in the +4 oxidation state. Ru in Ce1−xRuxO2−δ prepared by the hydrothermal method, Pt and Pd in Ce1−xMxO2−δ (M = Pt and Pd) and Pd in doped Ce1−xFexO2−δ prepared by sonochemical method are in oxidized states.63,69,70 | ||
| Fig. 4 Left panel: XPS of Pt4f core levels of (a) Pt metal, (b) (NH3)4Pt(NO3)2, (c) PtO2, (d) Ce0.99Pt0.01O2−δ and (e) Ce0.84Ti0.15Pt0.01O2−δ; right panel: XPS of Rh3d core levels of (a) Rh metal, (b) Rh2O3, (c) Ce0.99Rh0.01O2−δ and (d) Ce0.98Rh0.02O2−δ. Reprinted from ref. 24 with the permission of Elsevier B. V. and from ref. 44 with the permission of American Chemical Society. | ||
Pd metal and highly ionic Pd2+ species are observed in Pd/CeO2 prepared by wet impregnation method.76 Pd3d5/2,3/2 core level peaks observed at 336.5 and 338.0 eV in Pd/CeO2 catalysts prepared by the coprecipitation method correspond to PdOx and highly ionic Pd2+ species of the Ce1−xPdxO2−δ solid solution.82 Li and coworkers have demonstrated a Pd3d5/2 core level peak at 336.7 eV in a coprecipitated PdCeOx solid solution.83 XPS studies have shown that Pt, Pd and Ru are in their ionic states in M/Ce0.72Zr0.18Pr0.1O2 (M = Pt, Pd and Ru) catalysts.84 There are several recent reports where Pt and Pd are present in their ionic states in a CeO2 matrix.85–89 Pt4f7/2 core level peaks are observed at 71.3, 72.3 and 73.9 eV in 3.7 wt% Pt–Ce(La)Ox catalyst synthesized by the coprecipitation method.90 A combustion-synthesized Pt/CeO2 catalyst contains Pt0, Pt2+ and Pt4+ species as demonstrated by XPS.77 Pt0, Pt2+ and Pt4+ species are found in Pt/CeO2 catalysts prepared by the deposition–reduction method.91 Ru is in the +4 oxidation state in Ru/CeO2 catalysts.92,93 Au0, Au+ and Au3+ species are present in 4.7 wt% Au–Ce(La)Ox and 1% Au/CeO2 prepared by coprecipitation and photoreduction methods.90,94 Au is in Au0, Au+ and Au3+ oxidation states in 2.8 wt% Au/CeO2 obtained from the deposition–precipitation method.95 Ag3d5/2 core level peaks at 368.3 and 367.8 eV observed in an Ag/CeO2 catalyst prepared by the impregnation method are attributed to Ag0 and Ag+ species.96
XPS studies of Ti0.99M0.01O1.99 (M = Pd, Pt, Rh and Ru) catalysts also show the ionic nature of substituted noble metals in a TiO2 matrix.54 Core level XPS of Pd3d in Ti0.99Pd0.01O1.99, Pt4f in Ti0.99Pt0.01O1.99, Rh3d in Ti0.99Rh0.01O1.99 and Ru3p in Ti0.99Ru0.01O1.99 are presented in Fig. 5. A Pd3d5/2 peak at 337.2 eV is attributed to Pd2+ ion substitution in the TiO2 support. Pd3d peaks in Ti0.99Pd0.01O1.99 are observed at higher binding energies compared to PdO, suggesting that Pd2+ ions in TiO2 are in a more ionic state than Pd2+ in PdO. Pt4f in Ti0.99Pt0.01O1.99 shows a broad spectrum of mixed valence states of Pt2+ and Pt4+. Rh3d5/2 at 310.0 eV indicates clearly that Rh is in the +3 oxidation state in Ti0.99Pt0.01O1.99. A Ru3p3/2 peak observed at 463 eV indicates that Ru is in the +4 state in Ti0.99Ru0.01O1.99 and the peak position is similar to that in RuO2. In all the cases, core level Ti2p3/2,1/2 peaks are observed at 459.0 and 464.0 eV, indicating that Ti is in the +4 oxidation state.
 | ||
| Fig. 5 XPS of (a) Pd3d in Ti0.99Pd0.01O2−δ, (b) Pt4f in Ti0.99Pt0.01O2−δ, (c) Rh3d in Ti0.99Rh0.01O2−δ and (d) Ru3p in Ti0.99Ru0.01O2. Reprinted from ref. 54 with the permission of Elsevier B. V. | ||
The stabilization of noble metal ions in CeO2 or TiO2 can be substantiated from the relative positions of metal valence levels with respect to valence levels of CeO2 and TiO2. Typical valence band XPS of CeO2, Ce0.98Pd0.02O1.98, Pd metal, PdO, TiO2 and Ti0.97Pd0.03O2−δ are shown in Fig. 6. The valence band of CeO2 consists of the O2p band spread over ∼3–9 eV that is shown in the left panel of Fig. 6. An empty Ce4f level is located at ∼2 eV below the Fermi level (EF), where binding energy is referenced to zero. Pt, Pd and Rh metals have a high electron density at the EF and the valence bands of these metals even extend up to 6 eV below the EF. When these metals are oxidized, Mn+d bands shift to higher binding energies with the effect that the Mn+d bands are located at about 2.5–3.5 eV below EF as in PdO in the figure. In Ce1−xMxO2−δ, the Mn+d band lies between the Ce4f and O2p bands; therefore, Ce in the compound remains mostly in the +4 state. When the metal ion in Ce1−xMxO2−δ is reduced to the metal in the lattice, the metal valence band is shifted towards the EF that is above the empty Ce4f band. Therefore, electron transfer from the metal to the Ce4+ ion (M0 + 2Ce4+4f0 → M2+ + 2Ce3+4f1) becomes facile and the noble metals remain ionic in CeO2.21 For example, the difference in the valence band XPS of CeO2 and Ce0.98Pd0.02O1.98 shows a small density of states at ∼3.2 eV from EF. This density of states corresponds to the Pd2+4d8 occupied state that is higher than that of the Ce4f level.51 Similarly, Pt2+ and Pd2+ ions are stabilized in TiO2, because the Pt2+5d and Pd2+4d bands are situated between Ti3+3d (0.9 eV) and O2p bands.21,53,97 The right panel of Fig. 6 shows the band positions of TiO2 and Ti0.97Pd0.03O2−δ.
 | ||
| Fig. 6 Left panel: valence band XPS of CeO2, Ce0.9Pd0.1O2−δ, PdO, Pd metal and the difference spectrum of Ce0.9Pd0.1O2−δ and CeO2 valence bands showing the Pd2+4d band position. Reprinted from ref. 23 with the permission of Springer; right panel: valence band XPS of TiO2, Ti0.97Pd0.03O2−δ and the difference spectrum of Ti0.97Pd0.03O2−δ and TiO2 valence bands showing the Pd2+4d band position. | ||
3.4. XAFS studies
Substitution of metal ions into the CeO2 lattice has been confirmed by XAFS studies (SPring-8, Japan). XAFS analysis shows unique Ce–Pd, Ce–Rh and Ce–Pt distances of 3.31, 3.16 and 3.28 Å, respectively, whereas the Ce–Ce distance is observed to be 3.84 Å, demonstrating the substitution of Pd2+, Rh3+ and Pt2+ ions into the CeO2 lattice.43,44,72 Similar distances related to Ce–Pt and Ce–Pd interactions are also observed in Ce1−x−yTixPtyO2−δ, Ce1−x−yTixPdyO2−δ and Ce1−x−ySnxPdyO2−δ catalysts.50,98 It is important to mention that these unique interactions are found in neither CeO2 nor noble metals or their oxides such as PdO, Rh2O3 and PtO2. Typical XAFS of Pt and Rh ion-substituted CeO2 catalysts along with Pt metal, PtO2, Rh metal and Rh2O3 are shown in Fig. 7. Lower coordination numbers around metal ions compared to the Ce ion indicate that the oxide ion vacancy is due to lower valent metal ion substitution. | ||
| Fig. 7 Left panel: Fourier transformed XAFS of Pt metal, PtO2, Ce0.99Pt0.01O2−δ and Ce0.98Pt0.02O2−δ, reprinted from ref. 23 with the permission of Springer; right panel: Rh metal, Rh2O3, Ce0.99Rh0.01O2−δ and Ce0.98Rh0.02O2−δ. | ||
3.5. DRIFTS studies
The DRIFTS technique has been used to understand the interaction of CO and NO with noble metal ionic catalysts, such as Ce0.98Pd0.02O2−δ and Ce0.73Sn0.25Pd0.02O2−δ, and the nature of Pd species present on their surface through CO and NO adsorption. Adsorption studies are also carried out with Pd/Al2O3, where Pd is in the metallic state. It has been demonstrated that CO and NO are molecularly adsorbed on Pd/Al2O3, whereas NO is dissociatively adsorbed on Ce0.98Pd0.02O2−δ and Ce0.73Sn0.25Pd0.02O2−δ catalysts.99 In contrast, a linear Pdδ+–CO adsorption band along with a bridging CO band has been observed over CeO2-based catalysts when CO is adsorbed first, whereas a band related to Pd2+–CO interaction is noticed on preadsorbed NO catalysts. As CO is a reducing molecule, partial surface reduction of Pd2+ occurs only when CO is adsorbed on the Ce0.98Pd0.02O2−δ and Ce0.73Sn0.25Pd0.02O2−δ catalysts. However, Pd2+ remains intact after CO adsorption in the presence of an oxidizing molecule such as NO indicating the presence of Pd2+ species on the surface. Again, a Pd2+–NO band is observed only when NO is adsorbed on their surfaces. Therefore, CO and NO adsorption studies demonstrate that Pd is in a +2 oxidation state in Ce0.98Pd0.02O2−δ and Ce0.73Sn0.25Pd0.02O2−δ catalysts. Typical DRIFT spectra of (a) NO adsorption and (b) followed by CO adsorption over Ce0.98Pd0.02O2−δ are shown in Fig. 8. Similarly, a Pt2+–CO band has been observed when CO is passed over the Ce0.99Pt0.01O2−δ catalyst.71 In a recent study, a Cu2+–CO band was observed to be present in Ce0.93Cu0.07O2−δ and Ce1−x−yMxCuyO2−δ (M = Zr, Hf and Th) catalysts with CO exposure.100 | ||
| Fig. 8 DRIFT spectra of (a) NO adsorption and (b) followed by CO adsorption over Ce0.98Pd0.02O2−δ. Reprinted from ref. 99 with the permission of Elsevier B. V. | ||
Based on extensive characterization studies employing XRD, TEM, XPS, XAFS and DRIFTS, it has been demonstrated that noble metals are ionically dispersed over CeO2-based catalysts and noble metal ions are incorporated into the CeO2 matrix in the form of a Ce1−xMxO2−δ solid solution phase in combustion-synthesized M/CeO2 catalysts. In addition to this, oxide ion vacancies are created due to aliovalent ionic substitutions for Ce4+; there seem to be additional oxide ion vacancies to the extent of 3.5% in nano CeO2. Similarly, a Ti1−xMxO2−δ type of solid solution phase has been formed in M/TiO2 catalysts by substituting noble metal ions into a TiO2 matrix.
4. Exhaust catalysis
Energy for modern civilization comes from the burning of fossil fuels such as coal, natural gas and oil. Most fossil fuels are constituted of carbon, nitrogen, hydrogen and to some extent sulfur-containing compounds. The energy stored in the fossil fuels, biomass, industrial and domestic waste is released mostly by complete as well as partial combustion of these constituents. Partial combustion leads to emission of nitrogen oxides (NOx), carbon monoxide (CO), sulfur oxides (SOx), NH3 and unburned hydrocarbons (HC) into the atmosphere, which is the main reason for environmental pollution. These pollutants are emitted every day from the exhausts of mobile and stationary sources and have harmful effects on the Earth's atmosphere, the ecological system and also on human health.22,101,102 Emissions from automobiles and aeroplanes are the mobile sources, whereas industrial processes, power plants, combustion of waste and biomass and domestic burning are the stationary sources. In the early 1950s, air pollution and automobiles were first correlated by Californian scientists who established that a huge amount of traffic was to blame for the smoggy skies over the Los Angeles area.103–107 Over the years, international concern about environmental pollution caused by polluting sources has been increasing as it becomes a global environmental issue. Consequently, several developed and developing countries have enacted legislation for the control of pollutant gases in the exhaust stream of automobiles. Each of these countries has set particular emission standards by law for different types of vehicles according to their own country's situation. Emission standards set specific limits to the amount of pollutants that can be released into the environment. It is a limit that sets threshold amounts above which different types of emission control technology must be used. Standards generally regulate the emissions of NOx, CO, HCs, SOx and particulate matter (PM) or soot. Therefore, controlling these pollutants to achieve a clean environment is an important issue in the present day.There are several technologies for controlling pollutants in exhaust emission. Among the available technologies, one of the best ways of controlling exhaust emission is to reduce or convert these pollutant gases by using a catalyst at the source itself. Therefore, the conversion of environmentally unacceptable gases, such as NOx, CO and HC to N2, CO2 and H2O using catalysts, is a challenging task, and therefore this area of heterogeneous catalysis has been developing immensely in the last four decades. Within this context, environmental catalysis related to auto exhaust has received much more attention as exhausts from automobiles have significantly contributed to global environmental pollution. Extensive studies on the scientific research and technological development in this area have been carried out in the last few years. A number of reviews, articles and books regarding various aspects of exhaust catalysis have been published to update the status of catalytic science and technological development in this area from time to time.108–115
4.1. Reactions involved in exhaust catalysis
In the last few years, enormous efforts have been made to advance scientific and technological development for the controlling of exhaust emission. Catalytic converters have been found to be the best way to control exhaust emission from automobiles among all the types of technologies developed so far.116 A catalytic converter is a device that usually reduces the toxicity of exhaust emission. A catalyst housed inside a catalytic converter performs chemical reactions by which combustion by-products of pollutants are converted to less polluting or benign substances. Since 1981, three-way catalytic converters have been used in vehicle emission control systems of roadgoing vehicles in North America and many other countries. The catalyst used in a three-way catalytic converter is called a three-way catalyst (TWC). A TWC has three simultaneous tasks, namely (i) reduction of NOx to nitrogen and oxygen, (ii) oxidation of CO to CO2 and (iii) oxidation of unburned hydrocarbons to CO2 and H2O, provided that the air to fuel (A/F) ratio in the exhaust stream is constantly kept at the stoichiometric point (14.7), where concentrations of oxidizing and reducing gases are equal.The essential requirement for a catalyst employed in present-day catalytic converters for a gasoline engine is to carry out the conversion of NOx, CO and HCs present in the auto exhaust gases to inert and harmless N2, CO2 and H2O. The overall catalytic reactions that are important for controlling auto exhaust emissions are given by the following stoichiometric equations:
(a) Reduction reactions
| 2NO + 2CO → N2 + 2CO2 |
| 2NO + 2H2 → N2 + 2H2O |
(b) Oxidation reactions
| 2CO + O2 → 2CO2 |
| 2H2 + O2 → 2H2O |
(c) Three-way catalytic reaction
(d) Decomposition reaction
| 2NO → N2 + O2 |
(e) Selective catalytic reduction
| 4NO + 4NH3 + O2 → 4N2 + 6H2O |
| 6NO2 + 8NH3 → 7N2 + 12H2O |
(f) Secondary reactions with NO
| 2NO + CO → N2O + CO2 |
| 2NO + H2 → N2O + H2O |
| 2NO + 5H2 → 2NH3 + 2H2O |
| 4NH3 + 5O2 → 4NO + 6H2O |
| 4NH3 + 4NO + 3O2 → 4N2O + 6H2O |
| 2NH3 + 2O2 → N2O + 3H2O |
| 2NO + O2 → 2NO2 |
(g) Secondary reactions with SO2
| 2SO2 + O2 → 2SO3 |
| SO2 + 3H2 → H2S + 2H2O. |
All the abovementioned reactions required some heat or temperature on the catalyst surface for the reaction to occur. In the case of auto exhaust catalysis, when an automobile first starts, both the engine and the catalyst are cold. After start up, the heat of combustion is transferred from the engine and the exhaust piping begins to heat up. Finally, a temperature is reached within the catalyst that initiates the catalytic reactions. This light-off temperature depends on the chemistry of the catalyst.
4.2. Importance of OSC in auto exhaust catalysis and role of CeO2
In the early 1980s, the discovery of a catalytic converter with a three way catalyst (TWC) was considered as a major breakthrough in the development of devices for the control of auto exhaust emissions. Pollutant gases, such as CO, HC and NOx, are converted into benign CO2, H2O and N2 simultaneously and efficiently over TWCs with a constant A/F stoichiometric ratio (14.7) in the exhaust. A high efficiency of conversion is achieved over the TWC in a very narrow A/F window that requires highly efficient control of the exhaust composition, particularly in terms of the residual oxygen concentration. Strong deviations or oscillations of A/F from the stoichiometric value lead to widening of the operating window, resulting in a below average performance of the TWC. To widen the operating window of the TWC, the exhaust gas composition on the catalyst must be controlled by storing oxygen in oxygen-rich conditions and by releasing oxygen in oxygen-lean conditions. This can be achieved by adding oxygen storage material, which shows high oxygen storage capacity (OSC), to the TWC. In this sense, OSC is defined as the extent of reversibly exchanged oxygen of an oxygen storage material under rich and lean fluctuations of the exhaust gas composition. This oxygen storage material readily undergoes reduction–oxidation cycles that provides oxygen for CO and HC oxidation in the rich region and the reduced state can remove oxygen from the gas phase when the exhaust gas runs into the lean region. Thus, an oxygen storage material not only widens the A/F ratio window, but also promotes oxidation activity. Therefore, materials for TWC applications should have redox behavior capable of releasing/uptaking rapidly as much oxygen as possible, leading to a high OSC. Utilization of lattice oxygen from reducible oxides under reducing conditions and thereafter replenishment of oxygen under an oxidizing atmosphere has played a critical role in various oxidation reactions. Therefore, a reducible oxide with redox characteristics can act as both an oxidizing and reducing agent to various reactants, depending on the reaction conditions. In this regard, CeO2 has been found to be an efficient oxygen storage material for wide application in TWCs according to the following reversible reactions:| CeO2 → CeO2−δ + δ/2O2 |
| CeO2−δ + δ/2O2 → CeO2 |
To understand the reducibility of oxygen species as well as the metal–support interaction in CeO2 and related materials, H2-TPR is extensively employed where the volume of H2 consumed by the reduction of an oxide is measured. The OSC of CeO2 for oxidation/reduction reactions was estimated for the first time in a H2-TPR experiment on CeO2 by Yao and Yao.13 A simple mechanism for the OSC from H2-TPR can be written as follows:
| CeO2 + δH2 → CeO2−δ + δH2O |
| CeO2−δ + δ/2O2 → CeO2. |
H2-TPR of CeO2 shows primarily two peaks at approximately 500 °C and 825 °C.13,103,117,118 The low temperature peak is due to the reduction of the most easily reducible surface oxygen of CeO2, whereas the removal of bulk oxygen is suggested as the cause of the high temperature peak at 825 °C. The first peak is dependent on the surface area and is responsible for higher catalytic activity at lower temperature. Thus, the unique redox property of CeO2 has made it a special component in three-ways catalysts, because it can store oxygen during lean (net oxidizing, excess of O2) conditions and release it during rich (net reducing, deficient of O2) conditions as given below:119
| CeO2 + δCO → CeO2−δ + δCO2 |
| CeO2 + CxHy → CeO2−(2x+1/2y) + xCO2 + 1/2yH2O |
| CeO2 + δH2 → CeO2−δ + δH2O |
| CeO2−δ + δNO → CeO2 + δ/2N2 |
| CeO2−δ + δH2O → CeO2 + δH2 |
| CeO2−δ + 1/2δO2 → CeO2. |
It has also been observed that substitution of isovalent cations, such as Hf4+, Pr4+ and Tb4+, into the CeO2 lattice, forming a Ce1−xMxO2−δ solid solution, enhances the OSC.117,118 Nonreducible ZrO2 does not show redox properties as there is no peak in its respective H2-TPR profile, whereas significant OSC has been observed in Ce1−xZrxO2 samples.117,118 Higher OSC has also been noticed when precious metals are doped with CeO2.117 In general, a high surface area, oxygen vacancies and movement of oxide ions from tetrahedral sites to vacant octahedral sites are the possibilities for enhancing the OSC. It is important to note that Ce1−xZrxO2 and such oxygen storage materials are not the catalysts themselves for real applications. Noble metals, such as Pt, Pd, Rh, and bimetallic Pt–Rh, are dispersed mostly in the form of nanocrystalline metal particles on such OSC materials and they are used as catalysts.
4.3. Conventional catalysts for exhaust catalysis
In auto exhaust catalysis, a high efficiency for the conversions of NOx, CO and hydrocarbons can be achieved over a TWC in a very narrow A/F window that requires highly efficient control of exhaust composition, particularly in terms of residual oxygen concentration. This has been achieved by adding oxygen storage material to the TWC, which shows high OSC. Therefore, formulating suitable catalysts for TWC is a challenging task in auto exhaust catalysis. Within this context, studies on individual reactions for controlling exhaust emission, such as NO reduction, CO and hydrocarbon oxidation and SCR of NO by NH3, CO and hydrocarbons in the presence of O2, are also important. Therefore, extensive research work has been going on in the last few decades to find efficient catalysts for these reactions.In general, in the early years of exhaust catalysis, metal, alloy and metal oxide catalysts were used for catalytic reactions to control exhaust emission, especially NOx removal.119 Basic studies on metal catalysts involve single crystal or polycrystalline forms of bulk metal such as powder, wire, foil or ribbons. Prominent metals used are Pt, Pd, Rh, Ru and Ir.120–123 CuO, Fe2O3, TiO2, V2O5, MoO3, WO3, Cr2O3 and Al2O3 are employed as oxide catalysts.124 Transition metal ion-exchanged zeolites have also been developed for exhaust catalysis.125 Copper is one of the promising elements, which has the ability to increase the SCR activity of NO with NH3. Cu2+ ion-exchanged Y zeolite, mordenite, ZSM-5 and MFI-ferrisilicate have been found to be effective SCR catalysts for NO.119,126,127 Other metal ions, such as Fe3+, Co2+, Ce4+ and Ag+-exchanged zeolites, also show good SCR activity for NO.119,128–130 Noble metal exchanged zeolites are active for the removal of NO.131 The catalytic reduction of NO and the oxidation of CO and hydrocarbons have also been carried out over pure and cation-substituted perovskite oxides such as LaFeO3, LaCoO3, LaMnO3, Ln1−xPbxMnO3 (Ln = La, Pr and Nd), and LaMn1−xCuxO3 and La1−xSrxMnO3.132–134 They have different cationic sites in the crystal structure, which are highly amenable to substitution. Usually, metals in high oxidation states can be stabilized in the perovskite structure. Similarly, a large number of spinels, AB2O4 (A = Mg2+, Co2+, Ni2+, Cu2+ and Zn2+ and B = Al3+, Cr3+, Mn3+, Fe3+ and Co3+), are also employed for NO reduction and CO and hydrocarbon oxidation.119,135
In early 1980s, supported metal catalysts came into the scenario of auto exhaust catalysis.136–138 Metal catalysts with low metal loadings are usually dispersed on the surface of a support. Highly dispersed catalysts provide a maximum surface area. As the metal loading increases, the crystallites come close together, resulting in larger particle sizes. In general, the more expensive noble metal catalysts have low metal loadings and are highly dispersed, whereas catalysts containing less expensive base metals have higher metal loadings, usually 20–40% by weight or atom. Supports, such as Al2O3, SiO2, TiO2, CeO2, ZrO2, SnO2, WO3 and MgO, are used for the dispersion of metals. In this sense, in the search for a suitable material to satisfy several requirements for exhaust catalysis, CeO2 has emerged as a unique and active oxide support in the last few years. However, a catalyst in rigorous automotive exhaust conditions should have (a) intrinsic reactivity, (b) poison resistance ability and (c) durability and it soon has become apparent that the base metal oxides of Ni, Co, Mn and Cr do not have these properties. In particular, modern-day automotive exhaust catalysts revolve around three noble metals, Pt, Pd and Rh, which have been dispersed, stabilized, promoted, alloyed and segregated in sophisticated ways to fulfill the required performance and γ-Al2O3 has been found to be the main support along with a CeO2 additive. It is observed that the addition of CeO2 increases the activity of the Al2O3 catalyst towards three-way catalytic activities. The promoting effect of CeO2 is largely attributed to the enhancement of metal dispersions, OSC, increase in oxygen mobility due to oxide ion defects and thermal stability. CeO2 is able to act as an efficient oxygen storage material by releasing/storing oxygen because of its capability to undergo effective reduction and reoxidation under fuel-rich (A/F < 14.7) and fuel-lean (A/F > 14.7) conditions. The reversible oxygen storage/release feature coupled with chemical stability in adverse conditions is a unique feature that makes CeO2 a prime material in TWC design.117,136,139
4.4. Noble metal ionic catalysts for exhaust catalysis
From the fundamental basis of TWC it is necessary to develop materials that can provide good OSC as well as active sites for redox-type catalytic exhaust reactions, because both these properties are correlated. Realizing the importance of CeO2 as an oxygen storage material as well as an active catalyst support for redox-type catalytic exhaust reactions, we have synthesized Ce1−xMxO2−δ (M = Pd, Pt, Rh, Ru, Cu, Ag and Au), Ce1−x−yAxMyO2−δ (A = Ti, Zr, Sn and Fe; M = Pt and Pd) and Ti1−xMxO2−δ (M = Pd, Pt, Rh and Ru) catalysts by solution combustion, hydrothermal and sonochemical methods, where metal ions act as active sites for adsorption as well as reaction. Promoting the action of CeO2 and TiO2, the metal–support interaction, high OSC, hydrogen spillover and synergistic interaction are attributed to the interaction of Mn+/M0, Ce4+/Ce3+ and Ti4+/Ti3+ redox couples in the noble metal ion-substituted reducible oxides.21 In the following sections, we discuss several aspects of exhaust catalysis over these novel materials. | ||
| Fig. 9 H2-TPR profiles of (a) Ce0.99Pt0.01O2−δ, (b) Ce0.84Ti0.15Pt0.01O2−δ and (c) Ce0.83Ti0.15Pt0.02O2−δ. | ||
| Materials | OSC (μmol g−1) | References |
|---|---|---|
| CeO2 | 174 | 52 |
| Ce0.98Rh0.02O1.99 | 244 | 44 |
| Ce0.75Zr0.25O2 | 1900 | 140 |
| Ce0.6Zr0.4O2 | 2429 | 52 |
| Ce0.5Zr0.5O2 | 2900 | 140 |
| Ce0.9Ti0.1O2 | 740 | 47 |
| Ce0.85Ti0.15O2 | 880 | 47 |
| Ce0.8Ti0.2O2 | 1110 | 47 |
| Ce0.75Ti0.25O2 | 1340 | 141 |
| Ce0.7Ti0.3O2 | 1590 | 47 |
| Ce0.6Ti0.4O2 | 2000 | 47 |
| Ce0.99Pt0.01O1.99 | 290 | 47 |
| Ce0.84Ti0.15Pt0.01O1.99 | 1570 | 47 |
| Ce0.83Ti0.15Pt0.02O1.98 | 2320 | 47 |
| Ce0.73Ti0.25Pd0.02O1.98 | 1286 | 52 |
| Ce0.75Hf0.25O2 | 714 | 52 |
| Ce0.73Hf0.25Pd0.02O1.98 | 1121 | 52 |
| Ce0.58Hf0.4Pd0.02O1.98 | 804 | 52 |
| Ce0.98Pd0.02O1.98 | 232 | 52 |
| Ce0.83Zr0.15Pd0.02O1.98 | 638 | 52 |
| Ce0.73Zr0.25Pd0.02O1.98 | 1509 | 52 |
| Ce0.58Zr0.4Pd0.02O1.98 | 1612 | 52 |
| Ce0.78Zr0.2Pd0.02O1.98 | 620 | 50 |
| Ce0.9Sn0.1O2 | 980 | 49 |
| Ce0.8Sn0.2O2 | 1650 | 49 |
| Ce0.7Sn0.3O2 | 2100 | 49 |
| Ce0.6Sn0.4O2 | 2545 | 49 |
| Ce0.5Sn0.5O2 | 2780 | 49 |
| Ce0.78Sn0.2Pd0.02O1.98 | 1650 | 50 |
| Ce0.5Sn0.48Pd0.02O1.98 | 1330 | 50 |
| Ce0.95Ru0.05O1.97 | 1335 | 63 |
| Ce0.9Ru0.1O1.94 | 2513 | 63 |
| Ce0.9Fe0.1O1.88 | 665 | 51 |
| Ce0.89Fe0.1Pd0.01O1.84 | 780 | 51 |
| Ce0.67Cr0.33O2.11 | 2513 | 62 |
| TiO2 | 250 | 55 |
| Ti0.99Pd0.01O1.99 | 1240 | 55 |
| Ti0.98Pd0.02O1.98 | 2220 | 55 |
| Ti0.97Pd0.03O1.97 | 5100 | 55 |
| Ti0.97Pt0.032+O1.97 | 2010 | 56 |
| Ti0.97Pt0.034+O1.97 | 2315 | 56 |
4.4.2.1. CO oxidation by O2. Among Pt2+, Pd2+ and Rh3+ ions, Pd2+ ion-substituted CeO2, Ce1−xTixO2 and TiO2 show the highest rate of CO conversion and the lowest activation energy toward CO oxidation. Within this context, it is important to note that Pd is the cheapest among the noble metals. It has also been observed that activation energy decreases with an increase in the effective charge on the Pd2+ ion in these oxides.52 It has to be noted that the rates with metal ionic catalysts are higher by 20–30 times compared with the same amount of metal-impregnated catalysts. Pt and Pd ion-substituted Ce1−xTixO2, Ce1−xSnxO2 and Ce1−xFexO2−δ catalysts show very high CO conversion.47–50 In the top panel of Fig. 10, CO conversion over different Pt2+ substituted catalysts is presented. The Ti0.99Pt0.01O2−δ catalyst is observed to show the lowest temperature 100% CO conversion. Complete conversions of CO to CO2 are achieved below 105 and 135 °C over hydrothermally prepared Ce0.9Ru0.1O2−δ and Ce0.95Ru0.05O2−δ catalysts, respectively, in the presence of feed oxygen.63 In another type of experiment, CO is oxidized to CO2 by extracting activated lattice oxygen in the absence of feed oxygen. On exposure to oxygen, the lattice oxygen is replenished. This means that the feed oxygen is adsorbed and incorporated into the oxide ion vacancy, indicating the presence of two independent sites on the catalyst that adsorb reducing and oxidizing molecules.48,49,63 A Ce0.65Fe0.33Pd0.02O2−δ catalyst prepared by sonochemical method shows very low temperature CO oxidation with an activation energy of 38 kJ mol−1.70 Low temperature CO oxidation over Ti1−xPdxO2−x catalysts has been demonstrated by our group.142 It is important to note that CO oxidation over Pt/Al2O3 and Pd/Al2O3 synthesized by the solution combustion method occurs at higher temperatures compared to CeO2 and TiO2-based catalysts.42
 | ||
| Fig. 10 Top panel: CO oxidation over Pt substituted CeO2 and TiO2-based catalysts; bottom panel: CO oxidation over Rh metal and oxidized Rh species. Reprinted from ref. 47 with the permission of American Chemical Society and from ref. 145 with the permission of Springer. | ||
In recent times, there have been reports about the CO oxidation activities of supported ionic noble metals and it has been found that these oxidized noble metals are more active compared to noble metals. Grass and coauthors have demonstrated the formation of a thin, active RhOx over-layer on polymer-stabilized Rh nanoparticles, which is responsible for low-temperature CO oxidation.143 Active RhOx phase dispersed on CeO2 has been found to be highly active for CO oxidation as reported by Ligthart et al.144 Manuel and coworkers have shown that an oxidized Rh species (Rhx+) on Ce0.75Zr0.25O2 exhibits a significant 25 times higher turnover rate in CO oxidation than the corresponding zero valent one (Rh0).145 Herein, the Ce0.75Zr0.25O2 support stabilizes Rh atoms as ions. A comparison of CO oxidation over Rhx+/Ce0.75Zr0.25O2 and Rh0/Ce0.75Zr0.25O2 is displayed in the bottom panel of Fig. 10. A solution combustion-synthesized Au/CeO2 catalyst, where Au is in the +3 oxidation state, shows a very high CO oxidation rate at low temperatures.46 Carrettin et al. have demonstrated the stabilization of Au3+ in nanocrystalline CeO2, which shows very high CO oxidation activity.146 A strong interaction between ionic gold and CeO2 is attributed to an exceptionally high CO oxidation rate over an Au/CeO2 catalyst prepared by the deposition–precipitation method.147 In Table 3, a comparison of CO conversion rates and activation energies (Ea) for the CO + O2 reaction over noble metal ion-substituted CeO2 and TiO2 catalysts with supported nanometal catalysts is presented.47–49,51,55,56,63,70,82,84,87,142,148–153
| Catalysts | Rate (μmol g−1 s−1) | Ea (kJ mol−1) | References |
|---|---|---|---|
| 5 wt% Ru/SiO2 | 1.0 (110 °C) | 94 | 148 |
| 5 wt% Rh/SiO2 | 0.0251 (110 °C) | 103 | 148 |
| 5 wt% Pd/SiO2 | 0.316 (143 °C) | 103 | 148 |
| 5 wt% Ir/SiO2 | 0.26 (180 °C) | 105 | 148 |
| 5 wt% Pt/SiO2 | 0.32 (115 °C) | 56 | 148 |
| Pd/CeO2/Al2O3 | 38 (250 °C) | 84 | 149 |
| 0.014 wt% Rh/Al2O3 | 0.6 (196 °C) | 115.5 | 150 |
| 0.014 wt% Rh/9 wt% Ce/Al2O3 | 0.95 (196 °C) | 90.3 | 150 |
| 0.5 wt% Pd/CeO2–ZrO2 | 0.7 (220 °C) | 175 | 151 |
| 1.53% Pd/SiO2 | 0.155 (140 °C) | — | 152 |
| 25Au75Pd/SiO2 | 0.128 (140 °C) | — | 152 |
| 50Au50Pd/SiO2 | 0.06 (140 °C) | — | 152 |
| 5% Au/CeO2 | 0.77 (300 °C) | 80.3 | 153 |
| 5% Pd/CeO2 | 0.76 (300 °C) | 76.9 | 153 |
| Ce0.98Pd0.02O2−δ | 7.0 (130 °C) | 67.2 | 48 |
| 1 at% Pd/Al2O3 (impregnated) | 2.73 (200 °C) | 86.88 | 142 |
| 1 at% Pd/TiO2 (impregnated) | 0.758 (100 °C) | 75.25 | 142 |
| Ce0.99Pt0.01O2−δ | 0.6 (155 °C) | 55.4 | 47 |
| Ce0.84Ti0.15Pt0.01O2−δ | 28.0 (155 °C) | 82.3 | 47 |
| Ce0.73Ti0.25Pd0.02O2−δ | 18.0 (120 °C) | 54.6 | 48 |
| 2% Pd/Ce0.75Ti0.25O2−δ | — | 132.3 | 48 |
| Ce0.78Sn0.2Pd0.02O2−δ | 1.9 (50 °C) | 84 | 49 |
| Ce0.95Ru0.05O2−δ | 2.05 (100 °C) | 94.5 | 63 |
| Ce0.9Ru0.1O2−δ | 3.3 (100 °C) | 44.8 | 63 |
| Ce0.89Fe0.1Pd0.01O2−δ | — | 52.5 | 51 |
| Ce0.9Fe0.1O2−δ | — | 46.2 | 51 |
| Ce0.99Pd0.01O2−δ | — | 63 | 51 |
| Ce0.65Fe0.33Pd0.02O2−δ | 4.1 (80 °C) | 38 | 70 |
| 0.74% Pd/CeO2 | — | 42 | 82 |
| 2% Pt/Ce0.72Zr0.18Pr0.1O2 | 1.6 (61 °C) | — | 84 |
| 2% Pd/Ce0.72Zr0.18Pr0.1O2 | 1.6 (130 °C) | — | 84 |
| 2% Ru/Ce0.72Zr0.18Pr0.1O2 | 1.6 (150 °C) | — | 84 |
| 2% Pt/CeO2 | 2.89 (40 °C) | 51 | 87 |
| Ti0.99Pd0.01O1.99 | 13.83 (120 °C) | 53.13 | 142 |
| Ti0.97Pd0.03O1.97 | 2.75 (60 °C) | 17 | 55 |
| Ti0.97Pt0.032+O1.97 | 0.76 (60 °C) | 33.6 | 56 |
| Ti0.97Pt0.034+O1.97 | 0.08 (60 °C) | 16.8 | 56 |
4.4.2.2. NO reduction by CO. High activity for the NO + CO reaction with 100% N2 selectivity has been observed over Ce0.98Pd0.02O2−δ.154,155 The decomposition rate of adsorbed NO is faster over this catalyst, leading to higher N2 selectivity. For the NO + CO + O2 reaction, N2 selectivity of 100% is observed at higher temperatures over this catalyst. Ce0.98Pd0.02O2−δ shows lower temperature NO reduction by CO in relation to Ce0.98Pt0.02O2−δ and Ce0.98Rh0.02O2−δ catalysts.155 N2 selectivity is highest in Ce0.98Pd0.02O2−δ among all these three catalysts in the temperature range of 125–350 °C; however, N2 selectivities are more or less the same over all catalysts at higher temperatures (>300 °C). Complete NO reduction by CO occurs over Ce0.99Pt0.01O2−δ and Ce0.84Ti0.15Pt0.01O2−δ at 180 °C.47 100% N2 selectivity is observed at 240 °C for NO reduction by CO over a Ce0.73Ti0.25Pd0.02O2−δ catalyst.48 Ce0.78Sn0.2Pd0.02O2−δ exhibits 70% N2 selectivity at 200 °C, whereas 100% N2 selectivity is obtained at 245 °C over this catalyst.49 Complete NO conversion into N2 is observed at 200 and 250 °C over Ce0.95Ru0.05O2−δ and Ce0.9Ru0.1O2−δ, respectively, where N2O formation occurs at low temperatures.63 NO reduction by CO over a Ti0.99Pd0.01O1.99 catalyst shows high rates of NO conversion for the NO + CO reaction with high N2 selectivity. The rate of N2O reduction by CO is very high over this catalyst.137 N2 selectivity is 80% or more at all temperatures over this catalyst for the NO + CO + O2 reaction. However, it is important to mention that complete NO reduction by CO over 1% Pt/Al2O3 and 1% Pd/Al2O3 catalysts is observed at 400 and 350 °C, respectively.42 Comparisons of NO conversion rates, activation energies and N2 selectivities for the NO + CO reaction over noble metal ion-substituted CeO2 and TiO2 catalysts with supported nanometal catalysts are presented in Table 4.47–49,51,63,142,154,156–161
| Catalysts | Rate (μmol g−1 s−1) | Ea (kJ mol−1) | SN2 | SN2O | References |
|---|---|---|---|---|---|
| 1 wt% Pt/Al2O3 | 0.1 (300 °C) | 92.4 | 27 | 73 | 156 |
| 0.2 wt% Rh/Al2O3 | 7.2 (300 °C) | 184.8 | 32 | 68 | 157 |
| 1 wt% Pt–0.02 wt% Rh/Al2O3 | 0.58 (300 °C) | 126 | 39 | 61 | 158 |
| Pt–Rh/Al2O3–CeO2 | 0.54 (300 °C) | 84 | 33 | 67 | 158 |
| Rh/Al2O3 | 0.58 (227 °C) | 86.6 | 45 | 55 | 159 |
| Pd/Rh/Al2O3 | 0.11 (227 °C) | 100 | 43 | 57 | 159 |
| Pd/Al2O3 | 0.83 (227 °C) | 59.8 | 36 | 64 | 159 |
| Pd/La2O3/Al2O3 | 4.3 (227 °C) | 100 | 30 | 70 | 159 |
| 1% Pd/γ-Al2O3 | 0.75 (287 °C) | 158 | 34 | 66 | 160 |
| Pd8Mo/Al2O3 | 2.0 (300 °C) | 84 | 39 | 61 | 161 |
| Ce0.98Pd0.02O2−δ | 0.242 (175 °C) | 70.2 | 75 | 25 | 154 |
| Ce0.73Ti0.25Pd0.02O2−δ | 2.0 (180 °C) | 52.9 | — | — | 48 |
| Ce0.99Pt0.01O2−δ | 0.2 (135 °C) | 100.8 | — | — | 47 |
| Ce0.84Ti0.15Pt0.01O2−δ | 0.2 (135 °C) | 96.6 | — | — | 47 |
| Ce0.78Sn0.2Pd0.02O2−δ | 5.0 (150 °C) | 105.4 | 100 | — | 49 |
| Ce0.89Fe0.1Pd0.01O2−δ | 3.8 (220 °C) | — | 56 | 44 | 51 |
| Ce0.95Ru0.05O2−δ | 3.8 (200 °C) | 41 | 100 | — | 63 |
| Ce0.9Ru0.1O2−δ | 3.4 (200 °C) | 53.2 | 100 | — | 63 |
| Ti0.99Pd0.01O2−δ | 1.47 (175 °C) | 64.1 | 70 | 30 | 142 |
| 1% Pd/Al2O3 (impregnated) | 0.05 (240 °C) | 86.6 | 59 | 41 | 142 |
4.4.2.3. NO reduction by H2. NO can also be reduced by H2, which is also present in the mobile and stationary sources. 100% N2 selectivity and high reaction rates at low temperature are observed over the Ce0.98Pd0.02O1.98 catalyst, therefore making it superior to other existing catalysts reported in the literature.162 100% NO conversions are observed at 160, 180 and 370 °C over Ce0.89Fe0.1Pd0.02O2−δ, Ce0.99Pd0.01O2−δ and Ce0.9Fe0.1O2−δ, respectively.51 Solution combustion-synthesized Ti0.99M0.01O2−δ (M = Pd, Pt, Rh and Ru) catalysts have been tested for the NO + H2 reaction.54 The rate of NO reduction by H2 follows the order: Ti0.99Pt0.01O2−δ > Ti0.99Pd0.01O2−δ > Ti0.99Rh0.01O2−δ > Ti0.99Ru0.01O2−δ. The formation of the lowest amount of N2O occurs over Ti0.99Pd0.01O2−δ and no NH3 formation is traced during the reaction, whereas a significant amount of N2O and NH3 formation can be observed over the Ti0.99Pt0.01O2−δ catalyst. The experimental observations demonstrate that Ti0.99Pd0.01O2−δ is the best catalyst among the four catalysts investigated. In the presence of excess O2, N2 selectivity increases over the Ti0.99Pd0.01O2−δ catalyst during the NO + H2 + O2 reaction (lean conditions).
4.4.2.4. Selective catalytic reduction (SCR) of NO by NH3. SCR of NO by NH3 has been used extensively to clean up emissions from stationary exhausts. Good SCR catalysts should show low temperature activity, high N2 selectivity and a wide SCR window. A series of metal ion-substituted TiO2 catalysts have been synthesized by the solution combustion method and their SCR activities have been tested to understand the structure–property relationship and the reaction mechanism. The SCR reaction occurs at the lowest temperature over Ti0.9Mn0.1O2−δ among the Ti0.9M0.1O2−δ (M = Cr, Mn, Fe, Co and Cu) catalysts but N2 selectivity is found to be highest over Ti0.9Fe0.1O2−δ.163 In this sense, the Ti0.9Mn0.05Fe0.05O2−δ catalyst, where both Mn and Fe are substituted in TiO2, has been synthesized to optimize low temperature SCR activity and N2 selectivity. Indeed, the SCR reaction occurs at low temperatures with a high N2 selectivity over this catalyst. SCR activities over Ti0.9Mn0.1O2−δ, Ti0.9Fe0.1O2−δ and Ti0.89Mn0.1Pd0.01O2−δ have also been investigated to understand the role of base metal and noble metal ionic catalysts.164 From H2-TPR and SCR activity studies it has been observed that a more reducible catalyst shows better NH3 oxidation and poor SCR activity. NO reduction follows the order: Ti0.9Mn0.1O2−δ > Ti0.89Mn0.1Pd0.01O2−δ > Ti0.9Fe0.1O2−δ and ammonia oxidation follows the reverse order, which agrees well with the reducibility of the catalyst. The Ti0.89Mn0.1Pd0.01O2−δ catalyst, where a base metal and a noble metal are substituted in TiO2, does not show any synergetic effect. Therefore, the catalyst should have less reducibility to achieve better SCR activity. Thus, noble metal ion (Pd2+)-substituted TiO2, which has better reducibility than the base metal ion (Mn3+)-substituted TiO2, performs poorly for SCR.
4.4.2.5. NO reduction over NMIC coated on cordierite monolith. In recent years, monolith supports with honeycomb structures have been attractive alternative carriers for catalysts in heterogeneous catalysis and have been extensively used as catalyst supports inside catalytic converters for removal of exhaust gases in the automobile industry.165–167 In general, the monolith is first coated with a high-surface-area oxide such as γ-Al2O3 and the process is called washcoating. The active catalyst phase is dispersed over the monolith support after washcoating. Several methods of washcoating and coating of the active catalyst phase on monoliths are available in the literature.168,169 Due to advances in monolith technology, simple catalyst mounting methods, flexibility in reactor design, low pressure drop and high heat and mass transfer rates, monolithic supports dominate the entire automotive market as the preferred catalyst support. Ceramic monolith materials are leaders in the market among different monolith supports and the preferred material is cordierite.
Noble metal ionic catalysts are coated on cordierite monoliths by a single-step solution combustion method and three-way catalytic reactions have been carried out over these catalysts. The coating process involves growing of γ-Al2O3 on cordierite by the solution combustion method and then further growing of the active catalyst phase containing noble metal ionic catalysts on γ-Al2O3-coated cordierite, again by the same method. Due to the closeness of unit cell lattice parameters of cordierite and γ-Al2O3 as well as γ-Al2O3 and Ce1−xMxO2−δ, polycrystalline γ-Al2O3 on cordierites and Ce0.98Pd0.02O2−δ, Ce0.98Pt0.02O2−δ and Ce0.9Cu0.1O2−δ over γ-Al2O3 coated cordierites have been grown by the solution combustion method following the principle of epitaxial growth.170,171
It has been observed that Ce0.98Pd0.02O2−δ coated on cordierite shows much more catalytic activity toward CO oxidation, NO reduction and three-way catalysis in comparison with Ce0.98Pt0.02O2−δ and Ce0.9Cu0.1O2−δ-coated cordierite catalysts. Within this context, a detailed investigation on a Ce0.98Pd0.02O2−δ-coated cordierite catalyst has been carried out. 100% CO and NO conversions are achieved at around 80 and 200 °C for CO + O2 and NO + CO reactions, respectively, at a space velocity of 880 h−1. At the same space velocity, a three-way catalytic reaction over a Ce0.98Pd0.02O2−δ-coated monolith has been carried out with a gas mixture of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppm CO, 2000 ppm NO, 2000 ppm acetylene (C2H2) and 7000 ppm O2. It has been observed that 100% conversion of all the pollutants occurs below 220 °C and CO and C2H2 conversion is observed before NO conversion.170 All pollutant gases are converted to N2, CO2 and H2O below 225 °C, even with 15% excess oxygen. CO + O2, NO + CO and three-way catalytic reactions over Ce0.98Pd0.02O2−δ coated on a cordierite monolith are displayed in Fig. 11. In another study, three-way catalysis over a noble metal ionic catalyst coated on a monolith is carried out in the presence of NO, CO, O2 and a mixture of hydrocarbons, such as acetylene (C2H2), ethylene (C2H4), propene (C3H6) and propane (C3H8), with a GHSV of 4523 h−1. All the pollutant gases are fully converted to N2, CO2 and H2O within 340 °C. NO, CO and C2H2 are converted at almost same temperature of 260 °C, whereas complete conversions of C2H4, C3H6 and C3H8 occur at 290, 315 and 340 °C, respectively.171 Thus, the Ce0.98Pd0.02O2−δ catalyst coated on a cordierite monolith by the novel solution combustion method demonstrates good low-temperature three-way catalytic activity. In this regard, it is to be noted that the solution combustion method provides a new technique of washcoating a high-surface-area oxide, such as γ-Al2O3 and dispersing an active catalyst phase, containing noble metal ions over a monolith.
000 ppm CO, 2000 ppm NO, 2000 ppm acetylene (C2H2) and 7000 ppm O2. It has been observed that 100% conversion of all the pollutants occurs below 220 °C and CO and C2H2 conversion is observed before NO conversion.170 All pollutant gases are converted to N2, CO2 and H2O below 225 °C, even with 15% excess oxygen. CO + O2, NO + CO and three-way catalytic reactions over Ce0.98Pd0.02O2−δ coated on a cordierite monolith are displayed in Fig. 11. In another study, three-way catalysis over a noble metal ionic catalyst coated on a monolith is carried out in the presence of NO, CO, O2 and a mixture of hydrocarbons, such as acetylene (C2H2), ethylene (C2H4), propene (C3H6) and propane (C3H8), with a GHSV of 4523 h−1. All the pollutant gases are fully converted to N2, CO2 and H2O within 340 °C. NO, CO and C2H2 are converted at almost same temperature of 260 °C, whereas complete conversions of C2H4, C3H6 and C3H8 occur at 290, 315 and 340 °C, respectively.171 Thus, the Ce0.98Pd0.02O2−δ catalyst coated on a cordierite monolith by the novel solution combustion method demonstrates good low-temperature three-way catalytic activity. In this regard, it is to be noted that the solution combustion method provides a new technique of washcoating a high-surface-area oxide, such as γ-Al2O3 and dispersing an active catalyst phase, containing noble metal ions over a monolith.
 | ||
| Fig. 11 Catalytic reactions over Ce0.98Pd0.02O2−δ coated on cordierite monolith: (a) CO + O2, (b) NO + CO and (c) three-way catalysis. Reprinted from ref. 170 with the permission of Springer. | ||
4.4.2.6. Hydrocarbon oxidation. Complete CH4 oxidation occurs over Ce0.99Pt0.01O2−δ and Ce0.99Pd0.01O2−δ at 400 and 330 °C, respectively, whereas complete oxidation of C3H8 over these catalysts has been observed at 110 and 230 °C, respectively.20 On the other hand, these reactions occur at much higher temperatures over 1% Pt/Al2O3 and 1% Pd/Al2O3 catalysts.172 Similarly, 100% conversion of CH4 and C3H8 has been noticed at much lower temperatures over Ce0.99Ag0.01O2−δ and Ce0.99Au0.01O2−δ catalysts in comparison with 1% Ag/Al2O3 and 1% Au/Al2O3.45,46 The rate of C2H4 oxidation over bimetallic ionic Ce1−xPtx/2 Rhx/2O2−δ is observed to be higher than the corresponding monometallic ionic Ce1−xPtxO2−δ and Ce1−xRhxO2−δ catalysts.73 Ce0.84Ti0.15Pt0.01O2−δ shows a higher catalytic activity toward the oxidation of C3H8, C2H4 and C2H2.47 Activation energies of oxidation of C2H4 and C2H2 over Ce0.98Pd0.02O2−δ and Ti0.99Pd0.01O2−δ are found to be lower in comparison with previous studies in the literature.142,154 Oxidation reactions of several hydrocarbons over Ce1−xRuxO2−δ occur at lower temperatures and complete conversion temperatures depend on the amount of Ru.63 Hydrocarbon oxidation temperatures over Ti1−xPdxO2−δ catalysts decrease with an increase in Pd content and room temperature C2H2 oxidation occurs over a Ti0.97Pd0.03O2−δ catalyst.55
5. Water gas shift reaction
The reaction of CO with steam, leading to H2 and CO2, is generally called the water gas shift reaction (WGSR). It has been used as an important step in the production of H2 as well as bringing down the CO level in the steam reformate introduced into the polymer electrolyte fuel cells (PEMFC). Fu and Flytzani-Stephanopoulos have shown that the water gas shift reaction occurs at significantly low temperatures over leached Pt/CeO2 and Au/CeO2 containing ionic Pt and Au, compared to unleached catalysts that contain metallic species.90 Ionic Pt and Au species that are strongly associated with surface cerium–oxygen groups are responsible for the high activity. CO conversion has been found to be maximized at 200 °C over Ce1−xPtxO2−δ catalysts without any methanation.173 However, Ti0.99Pt0.01O2−δ shows the highest activity among Ti0.99Pt0.01O2−δ, Ce0.83Ti0.15Pt0.02O2−δ and Ce0.98Pt0.02O2−δ catalysts without deactivation.174 There is no carbonate formation over Ti0.99Pt0.01O2−δ due to the highly acidic nature of Ti4+ in the catalyst and consequently, no deactivation is observed even after prolonged reaction. Pt2+-substituted ZrO2 shows the lowest temperature WGS reaction among Pt2+ and Pd2+-substituted ZrO2 and Ce0.85Zr0.15O2 catalysts.175 The formation of Brønsted acid–base pairs is the main cause for the high activity of ZrO2 catalysts. Ce0.78Sn0.2Pt0.02O2−δ shows excellent CO conversion due to the presence of Ce4+/Ce3+ and Sn4+/Sn2+ redox couples.176 Much higher activity for the WGS reaction is noticed over a Ce0.65Fe0.33Pt0.02O2−δ catalyst, compared to Ce0.67Fe0.33O2−δ, due to the synergistic interaction of Pt4+ with Ce4+ and Fe3+ ions.177 Ce0.95Ru0.05O2−δ prepared by the hydrothermal method shows a low activation energy and high conversion rates at low temperatures and 100% H2 selectivity is observed over this catalyst.178 A sonochemically synthesized Ce0.65Fe0.33Pd0.02O2−δ catalyst is highly active for water gas shift reaction with reaction rate of 27.2 μmol g−1 s−1 and activation energy of 46.4 kJ mol−1.706. CO-PROX reaction
Preferential oxidation of CO (CO-PROX) is widely used to reduce trace levels of CO in a H2-rich gas stream from fuel reformate that is fed to polymer electrolyte fuel cells (PEMFC).179 A CO content above 10 ppm is present in the H2-rich gas stream from fuel reformate that is fed to deactivated platinum-based electrodes in PEMFCs. An effective CO-PROX catalyst shows high activity and selectivity for CO oxidation. It should be active in two temperature ranges, 170–230 °C, which is the outlet temperature for the low temperature water gas shift reaction (WGSR) and 80–100 °C, which is the operational temperature for PEMFCs. Therefore, a wide temperature range is required to avoid precise temperature controls. There have been reports of a Pt/CeO2 catalyst for the PROX reaction, where Pt is present in the metallic state.180 Pt2+ in CeO2 in the form of a Ce1−xPtxO2−δ solid solution is a more efficient catalyst than supported Pt metal catalysts and just 15% of Ti substitution in CeO2 improves the overall PROX activity.181 CO conversion of 100% is observed over a Ce0.83Ti0.15Pt0.02O2−δ catalyst at 55 °C, which represents a much higher conversion than that over Ce0.98Pt0.02O2−δ. Thus, the CO-PROX temperature window is larger than that of the Ce0.98Pt0.02O2−δ catalyst. The higher activity of Ce0.83Ti0.15Pt0.02O2−δ is because of the higher reducibility of this catalyst in comparison with Ce0.98Pt0.02O2−δ. Lattice oxygen becomes activated because of Ti substitution and can easily be removed under the reaction conditions. Herein, reducibility is an important factor as the reaction conditions are highly reducing (35% H2), making Ce0.83Ti0.15Pt0.02O2−δ more active than Ce0.98Pt0.02O2−δ. On the other hand, Ti0.98Pt0.02O2−δ is not a good PROX catalyst for CO as it shows a good activity for the H2 + O2 recombination reaction.7. H2 + O2 reaction
H2 + O2 recombination is technologically a very important reaction. Keeping the H2 concentration lower than 4.1% in nuclear power reactors is crucial for safety. Furthermore, the catalytic combustion of H2 by O2 is the cleanest source of energy. Hydrogen fuel cells also make use of a Pt/C catalyst for the overall recombination to produce power. Ever since Michael Faraday demonstrated the H2 + O2 recombination reaction over platinum metal plates, Pt nanoparticles have remained the only room-temperature recombination catalyst for this reaction. Alternative approaches to achieving higher recombination rates are: (1) to design a catalyst possessing two different adsorption sites for H2 and O2, unlike the single Pt0 site in Pt nanoparticles for both the molecules, (2) dissociative chemisorptions of H2 and O2 so that the activation energy of recombination is brought down and (3) making the catalyst more tolerant to CO to avoid CO poisoning in fuel cell applications. We have designed new catalysts, such as Ce1−xPtxO2−δ and Ti0.99Pd0.01O2−δ, where Pt and Pd are present in their ionic state and are highly active for H2 + O2 reaction.182,183 It has been demonstrated that the Ti0.99Pd0.01O2−δ catalyst not only shows higher rates for the H2 + O2 recombination reaction, but is also more tolerant to CO than Ce0.98Pt0.02O2−δ.183 Ce0.98Pt0.02O2−δ has also been found to be an excellent hydrogen and oxygen recombinant catalyst at room temperature for the recovery of water in sealed lead-acid batteries.1848. Photocatalysis
Photocatalysis is more economical with regard to low energy consumption and operating costs. It is an alternative method of NO reduction and decomposition that has been carried out over noble metal ionic catalysts in the presence of UV light in our laboratory. The optimum Pd2+ concentration has been found to be 1 at% for NO reduction by CO and NO decomposition over Pd2+ ion-substituted TiO2 containing Ti4+, Pd2+, O2− and the oxide ion vacancy sites.53 Measureable NO conversion is not observed over pure TiO2. With an increase in the Pd2+ ion content in Ti1−xPdxO2−δ, NO conversion increases with values of x from 0.005 to 0.01, and then it decreases with an increase in the Pd2+ concentration and for 3 at% Pd there is no NO conversion. The highest NO conversion of 80% was observed for Ti0.99Pd0.01O2−δ. This shows that there is an optimum Pd2+ ion concentration for NO conversion. The Pd2+4d band becomes broader as the Pd2+ ion concentration increases and the photoluminescence decreases significantly at 2 and 3%, leading to lower conversion. A decrease in NO conversion over Ti0.98Pd0.02O2−δ and Ti0.97Pd0.03O2−δ catalysts can also be correlated with an increase in electron density due to the Pd2+4d band in the band gap region (0–3 eV) of pure TiO2. With an increase in the Pd2+ ion concentration, an increase in the h+–e− pair recombination (shorting effect) should occur that lowers the rate of the photocatalytic reaction. Again, NO is adsorbed over both pure TiO2 and Ti0.99Pd0.01O2−δ, but photodissociation of NO happens only on Ti0.99Pd0.01O2−δ, not on pure TiO2. Therefore, photodissociation of NO requires oxide ion vacancies, which are present on Ti0.99Pd0.01O2−δ, and therefore NO adsorbed on oxygen vacancy sites is dissociative NO. Therefore, Pd2+ ions for adsorption, oxide ion vacancies and UV light are essential requirements for the photo dissociation of NO.NO conversion of 45% is observed when NO decomposition is carried out in the absence of CO over a Ti0.99Pd0.01O2−δ catalyst. In contrast, there is no NO conversion over pure TiO2. NO decomposition decreases over Ti0.99Pd0.01O2−δ in light on/off mode. It has been demonstrated that NO2 formed due to a partial reaction between NO and evolved O2 from NO decomposition blocks the oxide ion vacancy sites. However, in the presence of CO, this O2 is utilized to form CO2, and therefore a high NO conversion of 80% is observed over this catalyst.
9. Hydrogenation
The hydrogenation of p-chloronitrobenzene (p-CNB) to p-chloroaniline (p-CAN) is an important reaction, because chloroanilines are key intermediates in industry for the synthesis of herbicides, pesticides, pigments, pharmaceuticals, cosmetics and other organic fine chemicals. Recently, Mistri and coworkers have demonstrated the hydrogenation of p-chloronitrobenzene to p-chloroaniline over Ce0.98Pd0.02O2−δ, Ce0.98Ru0.02O2−δ and Ce0.98Pd0.01Ru0.1O2−δ catalysts.185 It has been observed that hydrogenation is inactive over monometallic ion-substituted catalysts. On the other hand, hydrogenation is completed beyond 75 min over Ce0.98Pd0.01Ru0.1O2−δ. A little higher activity with a lower selectivity is noticed with an increase in temperature from 35 to 80 °C. The presence of Ru4+, together with Pd2+ on CeO2 plays a crucial role towards the hydrogenation of p-CNB without dechlorination, indicating remarkable Ru4+ promotion in the bimetallic ion-substituted CeO2 catalyst.Hydrogenation of benzene to cyclohexane is an important reaction from an industrial point of view. A 42% conversion of benzene to cyclohexane with 100% selectivity is observed at 100 °C over Ce0.98Pt0.02O2−δ catalysts.186 The turnover frequency is observed to be an order of magnitude higher than those of other Pt catalysts.
10. Heck reaction
Heck coupling reaction, catalyzed by Pd, is well known in organic synthesis.187,188 It is known for its high tolerance of functional groups and general applicability.189 The Pd-catalyzed coupling reaction of organic/aromatic halides with olefins allows a one-step synthesis of aromatic olefins that are used extensively as biologically active compounds, natural products, pharmaceuticals and precursors of conjugated polymers.190,191 Heck reaction proceeds in the presence of homogeneous as well as heterogeneous Pd catalysts. Ce0.98Pd0.02O1.98 synthesized by the solution combustion method catalyzes C–C cross-coupling of substituted iodobenzene, bromobenzene and bromoheteroaryls with alkyl acrylates and styrenes with good yields. There is no conversion when iodobenzene with methyl acrylate is passed over pure CeO2, indicating that Pd2+ ions in Ce0.98Pd0.02O1.98 are the active sites for Heck reaction.192 It has been observed that C–X (X = Cl, Br, and I) bond strength influences the activity towards Heck coupling reaction as the coupling reaction follows the order: C–I > C–Br > C–Cl. Temperature also affects the conversion for the coupling of iodobenzene and methyl acrylate that is presented in Fig. 12. Around 100% conversion is observed in 30 min at 150 °C, whereas the reaction requires nearly 1 h when temperature is 125 °C. However, the reaction is very slow at 80 °C, where conversion is only 60% even after 7 h. The Ce0.98Pd0.02O1.98 catalyst can also be recycled without significant loss of catalytic activity, which indicates that the catalyst is stable under the reaction conditions. The observed turnover frequency (TOF) value for the reaction of iodobenzene and methyl acrylate is higher than for other Pd-substituted ligandless heterogeneous catalysts for C–C cross coupling reactions reported in the literature. | ||
| Fig. 12 Conversion efficiency of the reaction of iodobenzene and methyl acrylate over Ce0.98Pd0.02O1.98 at different temperatures. Reprinted from ref. 192 with the permission of Springer. | ||
11. Electrocatalysis and redox coupling between noble metal ions and CeO2
Electrocatalysis has been studied by several electrochemical methods with regard to oxygen evolution, oxidation of formic acid and methanol oxidation over noble metal ionic catalysts, providing a better understanding of electronic interactions between noble metal ions and CeO2 through electron exchange among redox ions, rather than gas–solid interactions. In a potentiostatic experiment (chronoamperometry) combined with extensive XPS studies, a pure CeO2 electrode shows almost complete (70%) reduction when it is subjected to an oxygen evolution potential (1.2 V) for 1000 s.193 The envelope of the Ce3d core level spectrum of the CeO2 electrode looks more similar to Ce2O3 after the chronoamperometry experiment. In an another experiment under potentiodynamic conditions (cyclic voltammetry), when the CeO2 electrode is cycled in the potential range of 0–1.2 V, Ce4+ is reduced to Ce3+, where the Ce3+ content is 75%. Though the fluorite structure is retained after the electrochemical experiment, as demonstrated by XRD patterns, this reduced CeO2 does not oxidize back under any electrochemical conditions. On the other hand, the Ce0.98Pt0.02O2−δ electrode shows partial reduction (38%) of Ce4+ to Ce3+ in a chronoamperometry experiment at an oxygen evolution potential (1.2 V) for 1000 s and Pt remains in the +2 and +4 oxidation states. The Ce4+ species in the Ce0.98Pt0.02O2−δ electrode material is reduced to 50% Ce3+ after stopping the cycle at 1.2 V in a cyclic voltammetry experiment. However, it returns to its initial status when the Ce0.98Pt0.02O2−δ electrode is cycled under potentiodynamic conditions in the positive potential range of 0–1.2 V with a particular scan rate. A similar phenomenon is also observed for Pt component species present in the Ce0.98Pt0.02O2−δ electrode. There is not much difference between the Ce and Pt component ratios with an increase in scan rates. Thus, the Ce0.98Pt0.02O2−δ electrode is stable to redox cycles in the potential range of 0–1.2 V. Similar evidence of redox reactions is observed in Ce0.98Pd0.02O2−δ.194 The catalytic redox properties of Ce0.98Pt0.02O2−δ and Ce0.98Pd0.02O2−δ electrodes is associated with the interaction between Pt4+/Pt2+/Pt0 and Ce4+/Ce3+, Pd2+/Pd0 and Ce4+/Ce3+ redox couples within the oxide catalysts. The redox ability of Cu2+/Cu+ and Cu+/Cu0 couples in Cu/CeO2 catalysts has been studied using cyclic voltammetry to understand the catalytic redox reactions.195–197 The catalytic activities of Ti0.99M0.01O2−δ (M = Pd, Pt, Rh and Ru) catalysts are correlated with their cyclic voltammetry studies.54Substitution of the Pt2+ and Pd2+ ions in CeO2 by sonochemical method activates the lattice oxygen and hydrogen spillover or a higher H/Pt ratio ∼ 8.1 and a H/Pd ratio ∼ 4.2 is observed. Ce0.95Pt0.05O2−δ reversibly releases 0.203 mol of [O] per mol of compound below −15 °C and Ce0.95Pd0.05O2−δ releases 0.105 mol of [O] per mol of compound below 55 °C. The reversible nature of the higher oxygen storage capacity or the higher H/Pt and H/Pd ratios is due to the interaction of redox couples such as Pt4+/Pt2+ (0.91 V), Pt2+/Pt0 (1.18 V), Pd2+/Pd0 (0.92 V), and Ce4+/Ce3+ (1.61 V). Because of the participation of lattice oxygen through the reduction of Ce4+ to Ce3+, Ce0.95Pt0.05O2−δ and Ce0.95Pd0.05O2−δ have shown higher electrooxidation of methanol compared to the same molar quantities of Pt in 5% Pt/C.69
It has been observed that Pt2+ ion substitution in CeO2 and Ce1−xTixO2 improves the electrocatalytic activity of the oxygen evolution reaction.198 In a cyclic voltammetry experiment, bubbles can be observed on the Ce0.98Pt0.02O2−δ electrode surface above 1.1 V, which confirms the oxygen evolution. The CeO2 electrode shows bubbling only above 1.4 V, which confirms the catalytic effect of Pt2+ in CeO2. Steady state current density values obtained from an 1800 s chronoamperometry experiment are 0.09, 0.15 and 0.31 mA cm−2 for CeO2, Ce0.98Pt0.02O2−δ and Ce0.83Ti0.15Pt0.02O2−δ, respectively. From XPS studies, it has been demonstrated that oxygen evolution is directly related to the extent of Ce4+ reduction and the stability of CeO2. The oxygen evolution current increases with a decrease in Ce4+ reduction. Pt ions in CeO2 and Ce0.83Ti0.15O2 control the lattice oxygen in a particular way to impart the activity in Ce0.98Pt0.02O2−δ and Ce0.83Ti0.15Pt0.02O2−δ.
Electrooxidation of formic acid has been investigated over Ce0.98Pt0.02O2−δ and Ce0.83Ti0.15Pt0.02O2−δ catalysts and the results are compared with the Pt/C electrocatalyst, where Pt is in the metallic state. In cyclic voltammetry experiments, a CO poisoning effect is observed on Pt/C, which is eliminated in Ce0.98Pt0.02O2−δ and Ce0.83Ti0.15Pt0.02O2−δ catalysts. In chronoamperometry experiments, a gain in the oxidation current in the ionic catalysts is 10 times more than the corresponding Pt/C catalysts for the same amount of Pt metal.198 Pt2+/Pt4+ states remain the same after cyclic voltammetry and chronoamperometry experiments in Ce0.98Pt0.02O2−δ and Ce0.83Ti0.15Pt0.02O2−δ, as demonstrated by XPS studies of the electrode materials. The extents of reduction of Ce4+ are 50% and 25% in Ce0.98Pt0.02O2−δ and Ce0.83Ti0.15Pt0.02O2−δ, respectively, indicating that lattice oxygen present in the ionic catalysts is utilized initially and a steady state is reached with equilibrium concentrations of Pt2+/Pt4+ and Ce4+/Ce3+ states. The involvement of lattice oxygen during electrooxidation of formic acid prevents CO poisoning of the catalysts. Similar behavior by Ce0.98Pt0.02O2−δ and Ce0.83Ti0.15Pt0.02O2−δ catalysts toward methanol oxidation has been observed and their activities are much higher than the same amount of Pt0 in Pt/C, which is attributed to interaction of the Pt2+ ion with CeO2 and Ce0.83Ti0.15O2 activating their lattice oxygen.198
12. Mechanism of redox reactions over noble metal ionic catalysts
Employing several characterization techniques, it has been demonstrated that there are two independent sites on the noble metal ionic catalysts, which adsorb reducing and oxidizing molecules during redox-type reactions such as CO + O2, NO + CO, NO + H2, NO + NH3, WGS reaction, CO-PROX reaction, H2 + O2 recombination, photocatalytic reduction of NO and Heck reaction. A dual site mechanism for redox reactions, such as CO + O2 and NO + CO, based on the structure of Ce1−xMxO2−δ and Ti1−xMxO2−δ-related catalysts has been proposed that is different from Langmuir–Hinshelwood mechanism on a noble metal surface. In Fig. 13, the CO + O2 reaction mechanism over a Pt metal surface is compared with that over Ce1−xMxO2−δ. It is well established that the CO + O2 reaction over Pt follows the Langmuir–Hinshelwood mechanism, where both CO and O2 are adsorbed on the Pt metal surface. CO is a polar molecule. CO has a triple bond as it is isoelectronic to nitrogen. The third bond is a co-ordinate-covalent bond due to lone pair donation from oxygen. Thus, the carbon end of CO is relatively negative. Therefore, carbon can donate electrons to the Pt metal. Bonding of CO with Pt is described by σ-donation to the Pt metal atom from its 5σ orbital and back-donation of the Pt metal d electron to the antibonding orbital of CO. A weak chemical bond is formed between CO and Pt metal and therefore CO adsorption is a chemisorption process. Thus, adsorption of CO makes Pt metal atoms negative as a whole. The electrons received by the Pt atoms on the surface from CO adsorption are shared by all the atoms in the particle; these are referred to as delocalized electrons in modern scientific terms. On the other hand, oxygen being electronegative, it tends to acquire electrons. The O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond length is known to be 1.21 Å. Unlike CO, O2 is adsorbed horizontally on the Pt surface. The Pt–Pt distance in Pt metal is 2.74 Å, and when an oxygen atom aligns with the top of adjacent Pt atoms, the molecule is stretched from 1.21 to 2.71 Å, leading to weakening of the O
O bond length is known to be 1.21 Å. Unlike CO, O2 is adsorbed horizontally on the Pt surface. The Pt–Pt distance in Pt metal is 2.74 Å, and when an oxygen atom aligns with the top of adjacent Pt atoms, the molecule is stretched from 1.21 to 2.71 Å, leading to weakening of the O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond of molecular O2. Lengthening of O–O is favored when the anti-bonding orbital of the O2 molecule receives electrons from Pt atoms. Transfer of electrons by Pt is possible because the Pt atoms have received electrons from CO. In this sense, the O2 molecule is dissociatively adsorbed on the Pt metal surface. This eventually leads to dissociation of O2 into two O atoms or partially negative ions. Once the O atom is created, it combines with the C end of CO, forming CO2. In contrast, on the Pt ionic catalyst surface, CO is adsorbed on the electron-deficient metal ions, whereas O2 is adsorbed onto the oxide ion vacancy because the vacancy site is activated by the electron-rich environment. The size of the oxide ion vacancy is ∼2.8 Å, which can accommodate an oxygen molecule of diameter 2.42 Å. Thus, there are two distinct sites, one for reducing and another for oxidizing molecules in the Pt metal ionic catalysts, which is different from the mechanism involving Pt metal atoms. Electron transfer from a reducing molecule to oxygen is facilitated by the lattice coupling between accessible Pt2+/Pt0 and Ce4+/Ce3+ redox couples. The enhanced activity is due to the creation of redox sites leading to site-specific adsorption and electronic interaction between the noble metal ions and the lattice. In addition to this, the lattice oxygen is activated in the catalyst with long M–O and Ce–O bonds and CO can be oxidized through a Mars–van Krevelen mechanism wherein the oxide ion is continuously consumed and formed. The difference in the rate of CO oxidation over different noble metal ion-substituted catalysts is due to differences in their redox properties.
O bond of molecular O2. Lengthening of O–O is favored when the anti-bonding orbital of the O2 molecule receives electrons from Pt atoms. Transfer of electrons by Pt is possible because the Pt atoms have received electrons from CO. In this sense, the O2 molecule is dissociatively adsorbed on the Pt metal surface. This eventually leads to dissociation of O2 into two O atoms or partially negative ions. Once the O atom is created, it combines with the C end of CO, forming CO2. In contrast, on the Pt ionic catalyst surface, CO is adsorbed on the electron-deficient metal ions, whereas O2 is adsorbed onto the oxide ion vacancy because the vacancy site is activated by the electron-rich environment. The size of the oxide ion vacancy is ∼2.8 Å, which can accommodate an oxygen molecule of diameter 2.42 Å. Thus, there are two distinct sites, one for reducing and another for oxidizing molecules in the Pt metal ionic catalysts, which is different from the mechanism involving Pt metal atoms. Electron transfer from a reducing molecule to oxygen is facilitated by the lattice coupling between accessible Pt2+/Pt0 and Ce4+/Ce3+ redox couples. The enhanced activity is due to the creation of redox sites leading to site-specific adsorption and electronic interaction between the noble metal ions and the lattice. In addition to this, the lattice oxygen is activated in the catalyst with long M–O and Ce–O bonds and CO can be oxidized through a Mars–van Krevelen mechanism wherein the oxide ion is continuously consumed and formed. The difference in the rate of CO oxidation over different noble metal ion-substituted catalysts is due to differences in their redox properties.
 | ||
| Fig. 13 CO + O2 reaction: (a) Langmuir–Hinshelwood mechanism on Pt metal and (b) dual site mechanism on Ce1−xPtxO2−δ. Reprinted from ref. 21 with the permission of American Chemical Society. | ||
A similar explanation can be given for the NO + CO reaction over noble metal ionic catalysts. NO is an electron donor as well as an acceptor molecule. Therefore, on the ionic catalysts, NO can be molecularly adsorbed on the noble metal ions and dissociatively chemisorbed on the vacant oxide ion sites. Adsorption of NO over noble metal ions is facilitated by the donation of the 5σ orbital electron of NO to the metal ion, leading to molecular adsorption. On the other hand, the anti-bonding 2π* orbital of NO obtains an electron from the oxide ion vacancy site where it is adsorbed, leading to the dissociation of NO. On the contrary, CO is specifically adsorbed on noble metal ion sites by donating its 5σ orbital electron to the metal ions. Molecularly adsorbed NO on the metal ion sites leads to N2O formation, whereas N2 is formed when NO is dissociatively adsorbed on the vacant oxide ion sites. Therefore, N2 selectivity is governed by dual site adsorption of NO on the catalyst. Because both NO and CO compete for metal ion sites and NO is adsorbed on vacant oxide ion sites, overall NO reduction results in N2 and N2O formation. However, N2O formed on the metal ion sites can move to the vacant oxide ion sites, making way for CO adsorption on metal ion sites and the reaction, N2O + CO → N2 + CO2, takes over, leading to N2 formation. Based on dual site adsorption (molecular adsorption on noble metal ion and dissociative adsorption on the vacant oxide ion), N2 and N2O formation along with CO2 over NMICs such as the Ce1−xPdxO2−δ catalyst during the NO + CO reaction are schematically shown in Fig. 14. A similar mechanism can be applied to the NO + CO reaction over Ti0.99M0.01O2−δ catalysts. Again, it has been demonstrated that Ce0.98Pd0.02O2−δ shows higher N2 selectivity compared to Ce0.98Rh0.02O2−δ.155 Higher N2O formation over Ce0.98Rh0.02O2−δ at lower temperatures seems to be due to higher NO adsorption on Rh3+ sites. Furthermore, the Rh ion being in the +3 state, there are fewer oxide ion vacancies in Rh3+-substituted CeO2 compared to Pd2+-substituted CeO2. Therefore, the NO → N2O conversion is facilitated as NO is more molecularly adsorbed on Rh3+ sites and less dissociatively adsorbed on the vacant sites of Ce0.98Rh0.02O1.99 at low temperatures. However, at higher temperatures, NO as well as N2O dissociation is increased and NO → N2 takes place. In contrast, NO → N2 conversion predominates on the Ce0.98Pd0.02O1.98 catalyst over the entire range of temperatures.
 | ||
| Fig. 14 Mechanism of N2, N2O and CO2 formation during the NO + CO reaction over Ce1−xPdxO2−δ. Reprinted from ref. 23 with the permission of Springer. | ||
For the NO + H2 reaction over Ti0.99M0.01O2−δ (M = Pd, Pt, Rh and Ru) catalysts, both NO and H2 are adsorbed on noble metal ion sites and NO is also adsorbed on the oxide ion vacancy sites.54 The formation of N2O occurs because of the close proximity of the N atom of adsorbed NO and the N atom of adsorbed NO on oxide ion vacancy sites. The hydrogen atom from the noble metal ion site reacts with the oxygen in the vacant oxide ion site to form water. The N2O molecule dissociates directly into N2 and an O atom in oxide ion vacancy sites. NH3 can also be formed by the combination of an adsorbed H atom with adsorbed NO in oxygen vacancy sites.
NH3-TPD demonstrates that low temperature SCR activity of Ti0.9Mn0.1O2−δ is due to the highest Brønsted acidity observed over this catalyst, whereas the highest Lewis acidity in Ti0.9Fe0.1O2−δ corresponds to a wide SCR window.163 Therefore, NH3 can be adsorbed on the surface as NH4+ or as molecular NH3 in the cases of Brønsted acid sites and Lewis acid sites, respectively. NO can be adsorbed molecularly on the transition metal ion or it can dissociatively chemisorb in the oxide ion vacancy of the catalysts. The adsorbed NH3 or NH4+ can form an adduct with the adsorbed NO and the NH2NO adduct dissociates into N2 and H2O. This path of dissociation dominates in the cases of Ti0.9Fe0.1O2−δ and Ti0.9Mn0.05Fe0.05O2−δ. However, NH2NO dissociating into N2O is more likely to be favored over Ti0.9Mn0.1O2−δ. Therefore, a “dual site-redox” mechanism occurs on these catalysts' surfaces. However, if the adsorbed NH3 reacts with the oxygen to form a NH2OO adduct, it produces NO and the SCR window becomes narrow. This seems to be the mechanism for Ti0.9Cu0.1O2−δ. Thus, it has been shown that the SCR reaction occurs at the lowest temperature with the highest N2 selectivity over the catalysts that have Brønsted and Lewis acid sites for NH3 adsorption as well as oxide ion vacancies for NO dissociation.
With regard to the WGS reaction, it has been proposed that CO is adsorbed on noble metal ionic sites, whereas H2O is dissociatively adsorbed over oxide ion vacancy sites.199 The surface reaction between two adsorbed species results in the formation of products that restore the catalyst upon desorption. From kinetic rate expression, it has been demonstrated that the WGS reaction over noble metal ionic catalysts does not follow the Eley–Rideal and Langmuir–Hinshelwood mechanisms. Apparent activation energies are found to be less, considering the dual site mechanism for the WGS reaction over NMICs.
In case of photocatalytic reduction of NO over Ti0.99Pd0.01O1.98, the presence of Pd2+ ion sites for adsorption of CO, oxide ion vacancy sites and UV light are the primary requirements for the photodecomposition of NO. A detailed mechanism accounts for the fact that there is competitive adsorption of NO and CO on Pd2+ sites and a high rate of NO photodissociation on the vacant oxide ion site present on the catalyst surface enhances NO reduction and decomposition activity.53
The Ce0.98Pd0.02O1.98 catalyst has two distinct sites: Pd2+ ions and oxide ion vacancies. Electron-deficient Pd2+ sites are useful for the adsorption of donor molecules, whereas vacant oxide ion sites next to Pd2+ ions act as sites for the adsorption of acceptor molecules. In Heck reaction over this catalyst, olefin is adsorbed on Pd2+ ions, whereas oxide ion vacancies are nucleophilic sites that are ideal for the accommodation of –Br and –I ions of aryl halides.192 The elimination of HBr facilitated by the base leads to C–C coupling.
13. DFT studies of noble metal ionic catalysts for higher OSC, adsorption and catalytic properties
In the last few decades, an enormous amount of studies on the synthesis, structure and catalytic properties of CeO2-related materials with different aspects have been carried out. It has been found that the OSC and catalytic activities of CeO2-based materials are enhanced when CeO2 is substituted with several divalent, trivalent and tetravalent metal ions. The substitution of noble metal ions into CeO2 further augments these properties. In this sense, there should have been changes in the atomic environment of the parent CeO2 structure after substitution of metal ions, leading to enhancement of the OSC and catalytic activity. The OSC of CeO2 is observed to be enhanced by the substitution of a Zr4+ ion for the Ce4+ site in CeO2.117,118,140,200 As ZrO2 itself cannot be reduced by H2, this observation is surprising. OSC studies demonstrate that the reducibility of CeO2 is increased by Ti4+ substitution in Ce1−xTixO2−δ.47 Again, Pt ion substitution in Ce1−xTixO2 enhances its reducibility further compared to Ce1−xTixO2 and Ce1−xPtxO2−δ. The rate of CO conversion in the CO + O2 reaction over Pt ion-substituted Ce1−xTixO2 is observed to be much higher than with Ce1−xPtxO2–δ.47 Therefore, several questions can be raised about these findings. In this regard, what is the structural and chemical origin of the increased OSC in transition-metal-ion-substituted CeO2? And what is the effect of noble metal substitution on the structural and chemical origins of the higher OSC and the high catalytic activities of CeO2? A simulation of the crystal structure of CeO2-based catalysts and probe reactions with these catalysts employing DFT calculations, has provided answers to these questions.50,51,201–223The first conception of the activation of lattice oxygen of CeO2 by the substitution of a smaller ion originated from the substitution of a Zr4+ ion into a CeO2 lattice in Ce1−xZrxO2 (x = 0.0–0.5) solid solutions.140 The structure can be simulated by DFT calculations, considering a lattice containing 32Ce4+ ions and 64O2− ions in their crystal positions in a fluorite lattice. CeO2 crystallizes in the fluorite structure, where the Ce4+ ion is inside a cube formed by eight oxide ions or the coordination number of Ce4+ is 8. Each O2− is coordinated with four Ce4+ ions. Because of the smaller ionic radius of Zr4+ (0.84 Å) compared to Ce4+ (0.97 Å), the lattice parameter decreases when a Zr4+ ion is substituted into the CeO2 lattice. Ce4+ and Zr4+ ions occupy alternate cubes in the Ce16Zr16O64 ideal lattice. When the smaller Zr4+ ion occupies the place of the Ce4+ ion, the Zr4+ ion does not have complete contact with the 8 oxide ions. Accordingly, in the [ZrO8] cube, the alternate four oxide ions come closer to the Zr4+ ion, forming a tetrahedron with a shortened Zr–O distance and the other four oxide ions also form a tetrahedron with longer Zr–O distances. Distortion in the ZrO8 cube will induce distortion in the CeO8 cube, also leading to 4 + 4 coordination that creates long Ce–O and short Ce–O bonds compared to 2.34 Å in pure CeO2. The longer Ce–O bond is naturally weaker. There is a distribution of bond lengths from the mean Ce–O values when the Zr4+ ion is substituted.24 Longer Ce–O bonds are indeed weaker and therefore they can be more easily extracted by H2, leading to a higher OSC.140 Similarly, XAFS studies and DFT calculations show the presence of short and long Ce–O and Ti–O bonds in Ce1−xTixO2, giving strongly and weakly bound oxygens.141 The stronger bonds have a valency greater than 2 and the weaker ones have a valency less than 2 and weakly-bound oxygen has been found to be responsible for a higher OSC in these mixed oxides.
It has been demonstrated that CeO2 doped with several transition, noble and rare earth metals undergoes structural distortion that results in a higher oxygen storage capacity.50,51,201–203 On the other hand, this type of situation cannot be found in pure CeO2 and TiO2, therefore these pure oxides show a lower OSC compared to mixed oxides. For example, structural simulations of CeO2, Ce0.8Sn0.2O2 and Ce0.78Sn0.2Pd0.02O2−δ have been carried out by DFT calculations, using their respective Ce4O8, Ce26Sn6O64 and Ce25Sn6Pd1O63 supercells.50 Bond length distributions of Ce4O8, Ce26Sn6O64 and Ce25Sn6Pd1O63 supercells are shown in Fig. 15. The optimized Ce4O8 supercell has all the Ce–O distances equal to 2.34 Å. In contrast, Ce–O bonds are distributed in shorter and longer bonds over the range of 2.16–2.54 Å in the Ce26Sn6O64 supercell. Sn–O and Pd–O bonds become longer in Ce0.78Sn0.2Pd0.02O2−δ with oxygen vacancies where oxygen sites are highly activated and can easily be removed from the lattice, leading to a higher OSC. With an increase in the Sn content, a larger number of longer Sn–O bonds is obtained in the Ce16Sn16O64 supercell related to Ce0.5Sn0.5O2 in comparison with the Ce26Sn6O64 supercell, thus, Ce0.5Sn0.5O2 shows a higher OSC in relation to Ce0.8Sn0.2O2. With the introduction of Pd2+ ions in Ce0.5Sn0.5O2, Ce–O and Sn–O bonds exhibit a wider distribution in the Ce16Sn15Pd1O63 supercell and here Pd–O bonds are longer than in the Ce25Sn6Pd1O63 supercell. Similarly, substitution of Pd2+ in Ce1−xFexO2−δ enhances the OSC and the catalytic activity.51 It has also been observed that transition metal ion substitution in CeO2 greatly enhances its reducibility in Ce1−xMxO2−δ solid solution, leading to a higher OSC due to longer Ce–O and M–O bonds, whereas rare earth metal ion substitution has little effect on it.202 The substitution of noble metal ions further enhances the OSC due to the presence of still longer bonds. It has also been demonstrated that Pt2+, Pd2+ and Cu2+ ions are stabilized in the CeO2 matrix and CO is adsorbed only on Pt2+, Pd2+ and Cu2+ sites present on the CeO2 surface.204
 | ||
| Fig. 15 Bond length distributions in (a) Ce4O8, (b) Ce26Sn6O64 and (c) Ce25Sn6Pd1O63 supercells from DFT calculations. Reprinted from ref. 50 with the permission of American Chemical Society. | ||
Both structural and electronic factors help to make CeO2 more reducible in Pd2+, Pt2+ and Rh3+-doped CeO2 than in pure CeO2 and Zr4+-doped CeO2, as shown by Yang et al.205 Wang and coworkers have proposed a model to understand the oxygen vacancy formation in a series of Ce1−xZrxO2 materials, which consists of bond energy (Ebond) and relaxation energy (Erelax).206 It has been found that the relaxation energy plays a vital role in affecting the oxygen vacancy formation energy and Ce0.5Zr0.5O2 possesses the lowest oxygen vacancy formation energy, which is the origin of its excellent OSC performance. Metiu and coworkers have demonstrated that doping of Pt in a CeO2 surface weakens the bonds of oxygen atoms in the neighborhood by lowering the formation energy of the oxygen vacancy. The easier specific oxygen atoms are to remove, the more reactive they are when exposed to a reductant.77 This works in a similar way for other doped CeO2 materials.209 Au3+-substituted CeO2 is more active for CO oxidation than a single Au+ species supported on stoichiometric CeO2, as demonstrated by Camellone and Fabris with DFT studies.210 This higher catalytic activity is due to the formation of oxygen vacancies and the interplay between reversible Au3+/Au+ and Ce4+/Ce3+ reductions. Chen et al. have found that substitution of Fe, Ru, Os, Sm, and Pu in CeO2 results in activated oxygen in Ce1−xMxO2 compared to pure CeO2, due to its structural and electronic modifications and oxygen vacancy formation is lowered by doping of noble metals.211 Watson's group has shown with DFT calculations that increased OSC in Pt and Pd-doped CeO2 is due to a large displacement of dopant ions from the Ce lattice site. Pd2+ and Pt2+ with d8 configuration move by ∼1.2 Å to adopt a square planar coordination with four lattice oxygens due to the crystal field effect.212 This large lattice distortion leaves three oxygen atoms under-coordinated or weakly bound in the vicinity of the dopant, which are more facile to remove than an oxygen atom in pure CeO2. Similarly, several divalent dopant metal ions, such as Be2+, Mg2+, Ca2+, Ni2+, Cu2+ and Zn2+, distort to adopt the coordination of their own binary oxide, instead of cubically coordinated Ce in CeO2. Depending on the electronic structure of the dopants, the different coordinations can create weakly or under-coordinated oxygen ions that are more easily removed than in pure CeO2.213 Recently, Nolan has shown that divalent Pd and Ni in CeO2(111) and CeO2(110) surfaces distort the local atomic environment, and the most stable structure for both dopants arises through compensation of the dopant +2 valency by oxygen vacancy formation.214 Both Pd2+ and Ni2+ dopants lower the formation energy of the oxygen vacancy. In another study, it is found that strong interactions exist between the Pt and the surfaces, and Pt adsorption is stronger on the reduced surface than that on the unreduced surface.215 A Pt–O–Ce bond may be formed between the Pt adatom and the CeO2 surface. Thus, DFT calculations provide local structures, metal–oxygen distances, and density of valence states to understand OSC, adsorption and catalytic properties.
The OSC of Ti0.97Pd0.03O1.97 is as high as 5100 μmol g−1, which is more than 20 times higher than that of pure TiO2. Oxygen is extracted by CO, resulting in the formation of CO2 in the absence of feed oxygen, even at room temperature. The rate of CO oxidation is 2.75 μmol g−1 s−1 at 60 °C over Ti0.97Pd0.03O1.97. Such a high catalytic activity is due to activated lattice oxygen created by the substitution of a Pd2+ ion, as observed from DFT calculations with 96 atom supercells of Ti32O64, Ti31Pd1O63, Ti30Pd2O62 and Ti29Pd3O61 related to TiO2, Ti0.99Pd0.01O2−δ, Ti0.98Pd0.02O2−δ and Ti0.97Pd0.03O2−δ, respectively.55 Ti–O bond distribution in the Ti32O64 supercell has two types of Ti–O bonds at 1.93 and 1.97 Å. The bond length of 1.93 Å corresponds to a Ti ion that is coordinated with four oxygen atoms in a planar geometry and other one is related to a Ti ion coordinating with two axial oxygen atoms in addition to 4 planar oxygens. Upon substitution of Pd2+, there is a distortion in the structure, resulting in metal–oxygen bonds with long and short distances compared to pure TiO2. Four Pd–O bonds in the three Pd2+ ion-substituted compounds are in nearly square planar geometry with Pd–O short bonds between 1.97 and 2.0 Å. Ti–O bonds are distorted with reference to pure TiO2 by the creation of longer and shorter Ti–O bonds. Similarly, by substitution of a Pt2+ ion in TiO2, there is a large change in bond distribution, with short and long metal–oxygen bond lengths compared to Ti32O64.56 In the Ti31Pt1O63 supercell, the Pt2+ ion is coordinated with four oxygens in a square planar geometry having two Pt–O bonds of 1.98 Å and two Pt–O bonds of 2.00 Å. As a result of Pt2+ substitution, two shorter Ti–O bonds of 1.81 Å and one longer Ti–O bond of 2.18 Å occur, coordinating to the same oxygen atom. In Ti30Pt2O62, there is a slight increase in the bond length distribution compared to that in Ti31Pt1O63. Bond length distributions in the two different configurations of Ti29Pt3O61 show a similar trend and these have slightly wider distributions compared to that in Ti30Pt2O62. Chrétien and Metiu have demonstrated that doping TiO2 with Cu, Ag, Au, Ni, Pd and Pt weakens the bonds of surface oxygens and oxides, making them better oxidation catalysts.224 Strong electron transfer from Ru to TiO2(101) makes Ru particles positively charged, lowering the activation energy for CO dissociation, resulting in high catalytic activity toward CO2 methanation.225
It has been established from our studies that noble metal ion-substituted CeO2 and TiO2 catalysts show a higher OSC as well as catalytic activity towards several important reactions. DFT calculations have been carried out to understand the causes behind the higher OSC and catalytic properties in CeO2 and TiO2-based materials. From our DFT studies, along with TPR and XAFS, it has been found that a change of structure at the atomic level is the reason for higher OSC in these mixed oxides. Because of the smaller size of substituent cations, surrounding oxygen ions are displaced from their original position and the oxygens at a larger distance are easily removed by reducing gases.
14. Conclusions and future outlook
The dispersion of noble metal ions over CeO2 and TiO2 by the solution combustion method, resulting in Ce1−xMxO2−δ, Ce1−x−yTixMyO2−δ, Ce1−x−ySnxMyO2−δ, Ce1−x−yFexMyO2−δ and Ce1−x−yZrxMyO2−δ and Ti1−xMxO2−δ (M = Pd, Pt, Rh and Ru) catalysts, the structure of these materials and their catalytic properties are documented here. In these materials, noble metals are present as ions and they are incorporated into reducible oxide matrices, such as CeO2 and TiO2, to a certain limit as the solution combustion method is a redox-reaction-type of preparation procedure. Hydrothermal and sonochemical methods have also been adopted to substitute aliovalent and isovalent noble metal ions in CeO2 and TiO2 matrices. Extensive experimental and theoretical studies done by our group and other research groups have established that these ionically dispersed noble metal catalysts are much more active than conventional finely-dispersed noble metal particles toward several important reactions such as CO oxidation, NO reduction by CO, H2 and NH3, selective catalytic reduction of NO, hydrocarbon oxidation, water gas shift reaction, CO-PROX reaction, H2–O2 reaction and hydrogenation. The idea of dispersing noble metals as ions can be extended to other reducible transition metal oxide supports such as SnO2, V2O5, WO3 and MoO3. These catalysts can be probed for several catalytic reactions, obtaining more insights into metal–support interactions, redox properties and synergistic effects. In general, extensive studies on these NMIC materials for several heterogeneous catalytic reactions that are relevant to present-day requirements, other than exhaust catalytic reactions, are a worthwhile area to investigate on the basis of solid state chemistry view point.Acknowledgements
We thank our past and present laboratory members and collaborators for their significant contributions. We thank the Department of Science and Technology (DST), Government of India, for financial support. MSH is grateful to the Council of Scientific and Industrial Research (CSIR), Government of India, for an Emeritus Professor Fellowship.References
- J. T. Richardson, Principles of Catalyst Development, Plenum Press, New York, 1989 Search PubMed.
- J. M. Thomas and W. J. Thomas, Principles and Practice of Heterogeneous Catalysis, Wiley-VCH, Weinheim, 2nd edn, 2015 Search PubMed.
- I. Chorkendorff and J. W. Niemantsverdriet, Concepts of Modern Catalysis and Kinetics, Wiley-VCH, Weinheim, 2003 Search PubMed.
- J. A. Anderson and M. F. García, Supported Metals in Catalysis, Imperial College Press, London, 2005 Search PubMed.
- J. Hagen, Industrial Catalysis, Wiley-VCH, Weinheim, 2006 Search PubMed.
- G. Rothenberg, Catalysis: Concepts and Green Applications, Wiley-VCH, Weinheim, 2008 Search PubMed.
- J. R. H. Ross, Heterogeneous Catalysis: Fundamentals and Applications, Elsevier, Oxford, 2012 Search PubMed.
- R. A. Sheldon and H. van Bekkum, Fine Chemicals through Heterogeneous Catalysis, Wiley-VCH, Weinheim, 2001 Search PubMed.
- Nanoparticles and Catalysis, ed. D. Astruc, Wiley-VCH, Weinheim, 2008 Search PubMed.
- G. C. Bond, Catalysis by Metals, Academic Press, New York, 1962 Search PubMed.
- J. R. Anderson, Structure of Metallic Catalysts, Academic Press, London, 1975 Search PubMed.
- H. S. Gandhi, G. W. Graham and R. W. McCabe, J. Catal., 2003, 216, 433 CrossRef CAS.
- J. C. Summers and S. A. Ausen, J. Catal., 1979, 58, 131 CrossRef CAS.
- H. C. Yao, H. S. Gandhi and M. Shelef, Metal–Support and Metal–Additive Effects in Catalysis, ed. B. Imelik, C. Naccache, G. Coudurier, H. Praliaud, P. Meriaudeau, P. Gallezot, G. A. Martin and J. C. Vedrine, Elsevier, Amsterdam, 1982, p. 159 Search PubMed.
- H. C. Yao and Y. F. Y. Yao, J. Catal., 1984, 86, 254 CrossRef CAS.
- G. Munuera, A. Fernandez and A. R. Gonzalez-Elipe, Stud. Surf. Sci. Catal., 1991, 71, 207 CrossRef CAS.
- J. G. Nunan, H. J. Robota, M. J. Cohn and S. A. Bradley, Stud. Surf. Sci. Catal., 1991, 71, 221 CrossRef CAS.
- J. Kašpar, P. Fornasiero and N. Hickey, Catal. Today, 2003, 77, 419 CrossRef.
- T. X. T. Sayle, S. C. Parker and C. R. A. Catlow, J. Phys. Chem., 1994, 98, 13625 CrossRef CAS.
- P. Bera, K. C. Patil, V. Jayaram, G. N. Subbanna and M. S. Hegde, J. Catal., 2000, 196, 293 CrossRef CAS.
- M. S. Hegde, K. C. Patil and G. Madras, Acc. Chem. Res., 2009, 42, 704 CrossRef CAS PubMed.
- P. Bera and M. S. Hegde, J. Indian Inst. Sci., 2010, 90, 299 CAS.
- P. Bera and M. S. Hegde, Catal. Surv. Asia, 2011, 15, 181 CrossRef CAS.
- M. S. Hedge and P. Bera, Catal. Today, 2015, 253, 40 CrossRef.
- A. Corma and H. Garcia, Chem. Soc. Rev., 2008, 37, 2096 RSC.
- J. C. Fierro-Gonzalez and B. C. Gates, Chem. Soc. Rev., 2008, 37, 2127 RSC.
- M. Flytzani-Stephanopoulos and B. C. Gates, Annu. Rev. Chem. Biomol. Eng., 2012, 3, 545 CrossRef CAS PubMed.
- F. J. Janssen, Handbook of Heterogeneous Catalysis, ed. G. Ertl, H. Knözinger and J. Weitkamp, VCH, Weinheim, 1997, vol. 1, p.191 Search PubMed.
- R. L. Augustine, Heterogeneous Catalysis for the Synthetic Chemist, Marcel Dekker, Inc., New York, 1996, p. 267 Search PubMed.
- M. A. Cauqui and J. M. Rodríguez-Izquierdo, J. Non-Cryst. Solids, 1992, 147–148, 724 CrossRef CAS.
- P. Turlier, H. Praliaud, P. Moral, G. A. Martin and J. A. Dalmon, Appl. Catal., 1985, 19, 286 Search PubMed.
- A. Hadi and I. I. Yaacob, Mater. Lett., 2007, 61, 93 CrossRef CAS.
- T. Masui, K. Fujiwara, Y. Penga, T. Sakatab, K. Machida, H. Mori and G. Adachi, J. Alloys Compd., 1998, 269, 116 CrossRef CAS.
- A. Suda, T. Kandori, N. Terao, Y. Ukyo, H. Sobukawa and M. Sugiura, J. Mater. Sci. Lett., 1998, 17, 89 CrossRef CAS.
- S. Enzo, F. Delogu, R. Frattini, A. Primavera and A. Trovarelli, J. Mater. Res., 2000, 15, 1538 CrossRef CAS.
- K. C. Patil, M. S. Hegde, T. Rattan and S. T. Aruna, Chemistry of Nanocrystalline Oxide Materials: Combustion Synthesis, Properties and Applications, World Scientific, Singapore, 2008 Search PubMed.
- W. Wen and J.-M. Wu, RSC Adv., 2014, 4, 58090 RSC.
- A. G. Merzhanov, SHS Research and Development Handbook, Chernogolovka, Russia, Russian Academy of Sciences, 1999 Search PubMed.
- K. C. Patil, S. T. Aruna and S. Ekabaram, Curr. Opin. Solid State Mater. Sci., 1997, 2, 158 CrossRef CAS.
- K. C. Patil, S. T. Aruna and T. Mimani, Curr. Opin. Solid State Mater. Sci., 2002, 6, 507 CrossRef CAS.
- S. L. González-Cortés and F. E. Imbert, Appl. Catal., A, 2013, 452, 117 CrossRef.
- P. Bera, K. C. Patil, V. Jayaram, M. S. Hegde and G. N. Subbanna, J. Mater. Chem., 1999, 9, 1801 RSC.
- K. R. Priolkar, P. Bera, P. R. Sarode, M. S. Hegde, S. Emura, R. Kumashiro and N. P. Lalla, Chem. Mater., 2002, 14, 2120 CrossRef CAS.
- A. Gayen, K. R. Priolkar, P. R. Sarode, V. Jayaram, M. S. Hegde, G. N. Subbanna and S. Emura, Chem. Mater., 2004, 16, 2317 CrossRef CAS.
- P. Bera, K. C. Patil and M. S. Hegde, Phys. Chem. Chem. Phys., 2000, 2, 3715 RSC.
- P. Bera and M. S. Hegde, Catal. Lett., 2002, 79, 75 CrossRef CAS.
- T. Baidya, A. Gayen, M. S. Hegde, N. Ravishankar and L. Dupont, J. Phys. Chem. B, 2006, 110, 5262 CrossRef CAS PubMed.
- T. Baidya, A. Marimuthu, M. S. Hegde, N. Ravishankar and G. Madras, J. Phys. Chem. C, 2007, 111, 830 CrossRef CAS.
- T. Baidya, A. Gupta, P. A. Deshpandey, G. Madras and M. S. Hegde, J. Phys. Chem. C, 2009, 113, 4059 CrossRef CAS.
- A. Gupta, M. S. Hegde, K. R. Priolkar, U. V. Waghmare, P. R. Sarode and S. Emura, Chem. Mater., 2009, 21, 5836 CrossRef CAS.
- A. Gupta, A. Kumar, U. V. Waghmare and M. S. Hegde, Chem. Mater., 2009, 21, 4880 CrossRef CAS.
- T. Baidya, G. Dutta, M. S. Hegde and U. V. Waghmare, Dalton Trans., 2009, 455 RSC.
- S. Roy, M. S. Hegde, N. Ravishankar and G. Madras, J. Phys. Chem. C, 2007, 111, 8153 CrossRef CAS.
- S. Roy, M. S. Hegde, S. Sharma, N. P. Lalla, A. Marimuthu and G. Madras, Appl. Catal., B, 2008, 84, 341 CrossRef CAS.
- B. D. Mukri, G. Dutta, U. V. Waghmare and M. S. Hegde, Chem. Mater., 2012, 24, 4491 CrossRef CAS.
- B. D. Mukri, U. V. Waghmare and M. S. Hegde, Chem. Mater., 2013, 25, 3822 CrossRef CAS.
- R. M. Barrer, Hydrothermal Chemistry of Zeolites, Academic Press, London, 1982 Search PubMed.
- W.-Q. Han, L. Wu and Y. Zhu, J. Am. Chem. Soc., 2005, 127, 12814 CrossRef CAS PubMed.
- Z. Y. Zhong, T.-P. Ang, J. Z. Luo, H.-C. Gan and A. Gedanken, Chem. Mater., 2005, 17, 6814 CrossRef CAS.
- W. Hung, P. Shuk and M. Greenblatt, Chem. Mater., 1997, 9, 2240 CrossRef.
- P. Singh and M. S. Hegde, J. Solid State Chem., 2008, 181, 3248 CrossRef CAS.
- P. Singh, M. S. Hegde and J. Gopalakrishnan, Chem. Mater., 2008, 20, 7268 CrossRef CAS.
- P. Singh and M. S. Hegde, Chem. Mater., 2009, 21, 3337 CrossRef CAS.
- L. Yin, Y. Wang, G. Pang, Y. Koltypin and A. Gedanken, J. Colloid Interface Sci., 2002, 246, 78 CrossRef CAS PubMed.
- S. Bhattacharyya and A. Gedanken, Microporous Mesoporous Mater., 2008, 110, 553 CrossRef CAS.
- R. K. Selvan, A. Gedanken, P. Anilkumar, G. Manikandan and C. Karunakaran, J. Cluster Sci., 2009, 20, 291 CrossRef.
- V. K. Rangari, D. N. Srivastava and A. Gedanken, Mater. Lett., 2006, 60, 3766 CrossRef CAS.
- S. Shanmugam and A. Gedanken, J. Phys. Chem. C, 2009, 113, 18707 CrossRef CAS.
- P. Singh and M. S. Hegde, Cryst. Growth Des., 2010, 10, 2995 Search PubMed.
- P. Singh and M. S. Hegde, Dalton Trans., 2010, 39, 10768 RSC.
- P. Bera, A. Gayen, M. S. Hegde, N. P. Lalla, L. Spadaro, F. Frusteri and F. Arena, J. Phys. Chem. B, 2003, 107, 6122 CrossRef CAS.
- P. Bera, K. R. Priolkar, A. Gayen, P. R. Sarode, M. S. Hegde, S. Emura, R. Kumashiro, V. Jayaram and G. N. Subbanna, Chem. Mater., 2003, 15, 2049 CrossRef CAS.
- A. Gayen, T. Baidya, K. Biswas, S. Roy and M. S. Hegde, Appl. Catal., A, 2006, 315, 135 CrossRef CAS.
- P. Bera, S. T. Aruna, K. C. Patil and M. S. Hegde, J. Catal., 1999, 186, 36 CrossRef CAS.
- P. Bera, K. R. Priolkar, P. R. Sarode, M. S. Hegde, S. Emura, R. Kumashiro and N. P. Lalla, Chem. Mater., 2002, 14, 3591 CrossRef CAS.
- G. Li, L. Li, Y. Yuan, J. Shi, Y. Yuan, Y. Li, W. Zhao and J. Shi, Appl. Catal., B, 2014, 158–159, 341 CrossRef CAS.
- W. Tang, Z. Hua, M. Wang, G. D. Stucky, H. Metiu and E. W. McFarland, J. Catal., 2010, 273, 125 CrossRef CAS.
- B. Wang, D. Weng, X. Wu and J. Fan, Catal. Today, 2010, 153, 111 CrossRef CAS.
- A. Bisht, B. P. Gangwar, T. Anupriya and S. Sharma, J. Solid State Electrochem., 2014, 18, 197 CrossRef CAS.
- S.-Y. Wang, N. Li, R.-M. Zhou, L.-Y. Jin, G.-S. Hu, J.-Q. Lu and M.-F. Luo, J. Mol. Catal. A: Chem., 2013, 374–375, 53 CrossRef CAS.
- L. M. Misch, J. A. Kurzman, A. R. Derk, Y.-I. Kim, R. Seshadri, H. Metiu, E. W. McFarland and G. D. Stucky, Chem. Mater., 2011, 23, 5432 CrossRef CAS.
- E. M. Slavinskaya, R. V. Gulyaev, A. V. Zadesenets, O. A. Stonkus, V. I. Zaikovskii, Y. V. Shubin, S. V. Korenev and A. I. Boronin, Appl. Catal., B, 2015, 166–167, 91 CrossRef CAS.
- G. Li, L. Li and D. Jiang, J. Phys. Chem. C, 2015, 119, 12520 Search PubMed.
- A. V. Malyutin, A. I. Mikhailichenko, Y. V. Zubavichus, V. Y. Murzin, A. G. Koshkin and I. V. Sokolov, Kinet. Catal., 2015, 56, 89 CrossRef CAS.
- W. Miao, S. Meiqing, W. Jianqiang, W. Guangxi, L. Hang and W. Jun, J. Rare Earths, 2014, 32, 1114 CrossRef.
- R. V. Gulyaev, E. M. Slavinskaya, S. A. Novopashin, D. V. Smovzh, A. V. Zaikovskii, D. Yu. Osadchii, O. A. Bulavchenko, S. V. Korenev and A. I. Boronin, Appl. Catal., B, 2014, 147, 32 CrossRef.
- H.-H. Liu, Y. Wang, A.-P. Jia, S.-Y. Wang, M.-F. Luo and Z.-Q. Lu, Appl. Surf. Sci., 2014, 314, 725 CrossRef CAS.
- R. V. Gulyaev, D. Yu. Osadchii, S. V. Koscheev and A. I. Boronin, J. Struct. Chem., 2015, 56, 566 CrossRef CAS.
- F. Pilger, A. Testino, M. A. Lucchini, A. Kambolis, M. Tarik, M. E. Kazzi, Y. Arroyo, M. D. Rossell and C. Ludwig, J. Nanosci. Nanotechnol., 2015, 15, 3530 CrossRef CAS PubMed.
- Q. Fu, H. Saltsburg and M. Flytzani-Stephanopoulos, Science, 2003, 301, 935 CrossRef CAS PubMed.
- X. Tang, B. Zhang, Y. Li, Y. Xu, Q. Xin and W. Shen, Catal. Lett., 2004, 97, 163 CrossRef CAS.
- J. Lin, L. Zhang, Z. Wang, J. Ni, R. Wang and K. Wei, J. Mol. Catal. A: Chem., 2013, 366, 375 CrossRef CAS.
- M. Kurnatowska, W. Mista, P. Mazur and L. Kepinski, Appl. Catal., B, 2014, 148–149, 123 CrossRef CAS.
- L. Ge, T. Chen, Z. Liu and F. Chen, Catal. Today, 2014, 224, 209 CrossRef CAS.
- A. Karpenko, R. Leppelt, V. Plzak and R. J. Behm, J. Catal., 2007, 252, 231 CrossRef CAS.
- L. Wang, H. He, Y. Yu, L. Sun, S. Liu, C. Zhang and L. He, J. Inorg. Biochem., 2014, 135, 45 CrossRef CAS PubMed.
- M. S. Chen and D. W. Goodman, Top. Catal., 2007, 44, 41 CrossRef CAS.
- T. Baidya, K. R. Priolkar, P. R. Sarode, M. S. Hegde, K. Asakura, G. Tateno and Y. Koike, J. Chem. Phys., 2008, 128, 124711 CrossRef PubMed.
- T. Baidya, P. Bera, B. D. Mukri, S. K. Parida, O. Kröcher, M. Elsener and M. S. Hegde, J. Catal., 2013, 303, 117 CrossRef CAS.
- T. Baidya and P. Bera, Catal. Struct. React., DOI:10.1179/2055075815Y.0000000004.
- C. T. Driscoll, K. M. Driscoll, M. J. Mitchell and D. J. Raynal, Environ. Pollut., 2008, 123, 327 CrossRef.
- K. Marilena and E. Castanas, Environ. Pollut., 2008, 151, 362 CrossRef PubMed.
- A. J. Haagen-Smit, Ind. Eng. Chem., 1952, 44, 1342 CrossRef CAS.
- P. L. Magill and R. W. Benoliel, Ind. Eng. Chem., 1952, 44, 1347 CrossRef CAS.
- A. J. Haagen-Smit and M. M. Fox, Ind. Eng. Chem., 1956, 48, 1484 CrossRef CAS.
- F. E. Littman, H. W. Ford and N. Endow, Ind. Eng. Chem., 1956, 48, 1492 CrossRef CAS.
- E. R. Stephens, P. L. Hanst, R. C. Doerr and W. E. Scott, Ind. Eng. Chem., 1956, 48, 1498 CrossRef CAS.
- A. Troveralli, Catalysis by Ceria and Related Materials, Imperial College Press, London, 2002 Search PubMed.
- A. Troveralli, Catalysis by Ceria and Related Materials, Imperial College Press, London, 2nd edn, 2013 Search PubMed.
- M. V. Twigg, Philos. Trans. R. Soc., A, 2005, 363, 1013 CrossRef CAS PubMed.
- F. Klingstedt, K. Arve, K. Eränen and D. Y. Murzin, Acc. Chem. Res., 2006, 39, 273 CrossRef CAS PubMed.
- E. S. J. Lox, Handbook of Heterogeneous Catalysis, ed. G. Ertl, H. Knözinger, F. Schüth and J. Weitkamp, Wiley-VCH, Weinheim, 2nd edn, 2008, vol. 5, p. 2274 Search PubMed.
- P. Gabrielsson and H. G. Pedersen, Handbook of Heterogeneous Catalysis, ed. G. Ertl, H. Knözinger, F. Schüth and J. Weitkamp, Wiley-VCH, Weinheim, 2nd edn, 2008, vol. 5, p. 2345 Search PubMed.
- M. Bowker, Chem. Soc. Rev., 2008, 37, 2204 RSC.
- P. Granger and V. I. Parvulescu, Chem. Rev., 2011, 111, 3155 CrossRef CAS PubMed.
- R. J. Farrauto and R. M. Heck, Catal. Today, 1999, 51, 351 CrossRef CAS.
- A. Troveralli, Catal. Rev.: Sci. Eng., 1996, 38, 439 CrossRef.
- A. Troveralli, Comments Inorg. Chem., 1999, 20, 263 CrossRef.
- P. Bera, Ph. D. thesis, Indian Institute of Science, Bangalore, India, 2002.
- T. P. Kobylinski and B. W. Taylor, J. Catal., 1974, 33, 376 CrossRef CAS.
- G. L. Bauerle, S. C. Wu and K. Nobe, Ind. Eng. Chem. Prod. Res. Dev., 1975, 14, 123 Search PubMed.
- J. T. Kummer, J. Phys. Chem., 1986, 90, 4747 CrossRef CAS.
- H. Bartholomew, Ind. Eng. Chem. Prod. Res. Dev., 1975, 14, 29 Search PubMed.
- G. L. Bauerle and K. Nobe, Ind. Eng. Chem. Prod. Res. Dev., 1974, 13, 185 Search PubMed.
- M. Iwamoto, Stud. Surf. Sci. Catal., 1994, 84, 1395 CrossRef CAS.
- T. Seiyama, T. Arakawa, T. Matsuda, Y. Takita and N. Yamazoe, J. Catal., 1977, 48, 1 CrossRef CAS.
- T. Komatsu, M. Nunokawa, I. S. Moon, T. Takahara, S. Namba and T. Yashima, J. Catal., 1998, 148, 427 CrossRef.
- R. T. Yang, T. J. Pinnavaia, W. Li and W. Zhang, J. Catal., 1997, 172, 488 CrossRef CAS.
- E. Ito, Y. J. Mergler, B. E. Nieuwenhuys, H. P. A. Calis, H. van Bekkum and C. M. van den Bleek, J. Chem. Soc., Faraday Trans., 1996, 92, 1799 RSC.
- J. A. Martens, A. Cauvel, A. Francis, C. Hermans, F. Jayat, M. Remy, M. Keung, J. Lievens and P. A. Jacobs, Angew. Chem., Int. Ed., 1998, 37, 1901 CrossRef CAS.
- W. Fu, X.-H. Li, H.-L. Bao, K.-X. Wang, X. Wei, Y.-Y. Cai and J.-S. Chen, Sci. Rep., 2013, 3, 2349 CrossRef PubMed.
- W. F. Libby, Science, 1971, 176, 499 CrossRef.
- R. J. H. Voorhoeve, J. P. Remeika, P. E. Freeland and B. T. Matthias, Science, 1973, 177, 353 CrossRef.
- R. Zhang, A. Villanueva, H. Alamdari and S. Kaliaguine, J. Catal., 2006, 237, 368 CrossRef CAS.
- F. Severino, J. L. Brito, J. Laine, J. L. G. Fierro and A. L. Agudo, J. Catal., 1998, 177, 82 CrossRef CAS.
- R. K. Herz, J. B. Klela and J. A. Sell, Ind. Eng. Chem. Prod. Res. Dev., 1983, 22, 387 CrossRef CAS.
- M. C. Kung, K. A. Bethke, J. Yan, J.-H. Lee and H. H. Kung, Appl. Surf. Sci., 1997, 121–122, 261 CrossRef.
- R. Burch, Pure Appl. Chem., 1996, 68, 377 CrossRef CAS.
- G. Kim, Ind. Eng. Chem. Prod. Res. Dev., 1982, 21, 267 CrossRef CAS.
- G. Dutta, U. V. Waghmare, T. Baidya, M. S. Hegde, K. R. Priolkar and P. R. Sarode, Catal. Lett., 2006, 108, 165 CrossRef CAS.
- G. Dutta, U. V. Waghmare, T. Baidya, M. S. Hegde, K. R. Priolkar and P. R. Sarode, Chem. Mater., 2006, 18, 3249 CrossRef CAS.
- S. Roy, A. Marimuthu, M. S. Hegde and G. Madras, Appl. Catal., B, 2007, 73, 300 CrossRef CAS.
- M. E. Grass, Y. Zhang, D. R. Butcher, J. Y. Park, Y. Li, H. Bluhm, K. M. Bratlie, T. Zhang and G. A. Somorjai, Angew. Chem., Int. Ed., 2008, 47, 8893 CrossRef CAS PubMed.
- D. A. J. M. Ligthart, R. A. van Santen and E. J. M. Hensen, Angew. Chem., Int. Ed., 2011, 50, 5306 CrossRef CAS PubMed.
- I. Manuel, C. Thomas, C. Bourgeois, H. Colas, N. Matthess and G. Djéga-Mariadassou, Catal. Lett., 2001, 77, 193 CrossRef CAS.
- S. Carrettin, A. Corma, M. Iglesis and F. Sánchez, Appl. Catal., A, 2005, 291, 247 CrossRef CAS.
- A. M. Venezia, G. Pantaleo, A. Longo, G. Di Carlo, M. P. Casaletto, F. L. Liotta and G. Deganello, J. Phys. Chem. B, 2005, 109, 2821 CrossRef CAS PubMed.
- N. W. Cant, P. C. Hicks and B. S. Lennon, J. Catal., 1978, 54, 372 CrossRef CAS.
- Y.-F. Yu Yao, J. Catal., 1984, 87, 152 CrossRef.
- S. H. Oh and C. C. Eickel, J. Catal., 1988, 112, 543 CrossRef CAS.
- E. Bekyarova, P. Fornasiero, J. Kašpar and M. Graziani, Catal. Today, 1998, 45, 179 CrossRef CAS.
- A. M. Venezia, L. F. Liotta, G. Pantaleo, V. L. Parola, G. Deganello, A. Beck, Zs. Koppány, K. Frey, D. Horváth and L. Guczi, Appl. Catal., A, 2003, 251, 359 CrossRef CAS.
- Y. Park, S. K. Kim, D. Pradhan and Y. Sohn, React. Kinet., Mech. Catal., 2014, 113, 85 CrossRef CAS.
- S. Roy, A. Marimuthu, M. S. Hegde and G. Madras, Appl. Catal., B, 2007, 71, 23 CrossRef CAS.
- S. Roy and M. S. Hegde, Catal. Commun., 2008, 9, 811 CrossRef CAS.
- P. Granger, C. Dathy, J. J. Lecomte, L. Leclercq, M. Prigent, G. Mabilon and G. Leclercq, J. Catal., 1998, 173, 304 CrossRef CAS.
- P. Granger, J. J. Lecomte, C. Dathy, L. Leclercq and G. Leclercq, J. Catal., 1998, 175, 194 CrossRef CAS.
- P. Granger, L. Delannoy, J. J. Lecomte, C. Dathy, H. Praliaud, L. Leclercq and G. Leclercq, J. Catal., 2002, 207, 202 CrossRef CAS.
- J. H. Holles, M. A. Switzer and R. J. Davis, J. Catal., 2000, 190, 247 CrossRef CAS.
- D. R. Rainer, S. M. Vesecky, M. Koranne, W. S. Oh and D. W. Goodman, J. Catal., 1997, 167, 234 CrossRef CAS.
- M. Schmal, M. A. S. Baldaza and M. A. Vannice, J. Catal., 1999, 185, 138 CrossRef CAS.
- S. Roy, A. Marimuthu, M. S. Hegde and G. Madras, Catal. Commun., 2008, 9, 101 CrossRef CAS.
- S. Roy, B. Viswanath, M. S. Hegde and G. Madras, J. Phys. Chem. C, 2008, 112, 6002 CrossRef CAS.
- S. Roy, A. Marimuthu, P. A. Deshpande, M. S. Hegde and G. Madras, Ind. Eng. Chem. Res., 2008, 47, 9240 CrossRef CAS.
- M. V. Twigg, Appl. Catal., B, 2007, 70, 2 CrossRef CAS.
- P. Avila, M. Montes and E. E. Miró, Chem. Eng. J., 2005, 109, 11 CrossRef CAS.
- V. Tomašić, Catal. Today, 2007, 119, 106 CrossRef.
- T. A. Nijhuis, A. E. W. Beers, T. Vergunst, I. Hoek, F. Kapteijn and J. A. Moulijn, Catal. Rev.: Sci. Eng., 2001, 43, 345 CrossRef CAS.
- P. Jiang, G. Lu, Y. Guo, Y. Guo, S. Zhang and X. Wang, Surf. Coat. Technol., 2005, 190, 314 CrossRef CAS.
- S. Sharma and M. S. Hegde, Catal. Lett., 2006, 112, 69 CrossRef CAS.
- S. Sharma, M. S. Hegde, R. N. Das and M. Pandey, Appl. Catal., A, 2008, 337, 130 CrossRef CAS.
- P. Bera, K. C. Patil and M. S. Hegde, Phys. Chem. Chem. Phys., 2000, 2, 273 Search PubMed.
- P. Bera, S. Malwadkar, A. Gayen, C. V. V. Satyanarayana, B. S. Rao and M. S. Hegde, Catal. Lett., 2004, 96, 213 CrossRef CAS.
- S. Sharma, P. A. Deshpande, M. S. Hegde and G. Madras, Ind. Eng. Chem. Res., 2009, 48, 6535 CrossRef CAS.
- P. A. Deshpande, M. S. Hegde and G. Madras, Appl. Catal., B, 2010, 96, 83 CrossRef CAS.
- A. Gupta and M. S. Hegde, Appl. Catal., B, 2010, 99, 279 CrossRef CAS.
- N. Mahadevaiah, P. Singh, B. D. Mukri, S. K. Parida and M. S. Hegde, Appl. Catal., B, 2011, 108–109, 117 CrossRef CAS.
- P. Singh, N. Mahadevaiah, S. K. Parida and M. S. Hegde, J. Chem. Sci., 2011, 123, 577 CrossRef CAS.
- A. Martínez-Arias, D. Gamarra, M. Fernández-García, A. Hornés, P. Bera, Z. Koppány and Z. Scahy, Catal. Today, 2009, 143, 211 CrossRef.
- D. Teschner, A. Wootsch, O. Pozdnyakova-Tellinger, J. Kröhnert, E. M. Vass, M. Hävecker, S. Zafeiratos, P. Schnörch, P. C. Jentoft, A. Knop-Gericke and R. Schlögl, J. Catal., 2007, 249, 318 CrossRef CAS.
- S. Sharma, A. Gupta and M. S. Hegde, Catal. Lett., 2010, 134, 330 CrossRef CAS.
- P. Bera, M. S. Hegde and K. C. Patil, Curr. Sci., 2001, 80, 1576 CAS.
- S. Sharma and M. S. Hegde, ChemPhysChem, 2009, 10, 637 CrossRef CAS PubMed.
- B. Hariprakash, P. Bera, S. K. Martha, S. A. Gaffoor, M. S. Hegde and A. K. Shukla, Electrochem. Solid-State Lett., 2001, 4, A23 CrossRef CAS.
- R. Mistri, J. Llorca, B. C. Ray and A. Gayen, J. Mol. Catal. A: Chem., 2013, 376, 111 CrossRef CAS.
- K. Nagaveni, G. Sivalingam, A. Gayen, G. Madras and M. S. Hegde, Catal. Lett., 2003, 88, 73 CrossRef CAS.
- A. de Meijere and F. E. Meyer, Angew. Chem., Int. Ed. Engl., 1995, 33, 2379 CrossRef.
- A. P. Beletskaya and V. Cheprakov, Chem. Rev., 2000, 100, 3009 CrossRef PubMed.
- G. T. Crisp, Chem. Soc. Rev., 1998, 27, 427 RSC.
- J. G. de Vries, Can. J. Chem., 2001, 79, 1086 CrossRef CAS.
- T. R. Burke, D. G. Liu and Y. J. Gao, J. Org. Chem., 2000, 65, 6288 CrossRef CAS PubMed.
- S. R. Sanjaykumar, B. D. Mukri, S. Patil, G. Madras and M. S. Hegde, J. Chem. Sci., 2011, 123, 47 CrossRef CAS.
- S. Sharma and M. S. Hegde, J. Chem. Phys., 2009, 130, 114706 CrossRef PubMed.
- S. Sharma, B. D. Mukri and M. S. Hegde, Dalton Trans., 2011, 40, 11480 RSC.
- P. Bera, S. Mitra, S. Sampath and M. S. Hegde, Chem. Commun., 2001, 927 RSC.
- X. Tang, B. Zhang, Y. Li, Y. Xu, Q. Xin and W. Shen, Catal. Today, 2004, 93–95, 191 CrossRef CAS.
- L. Li, Y. Zhan, Q. Zheng, Y. Zheng, X. Lin, D. Li and J. Zhu, Catal. Lett., 2007, 118, 91 CrossRef CAS.
- S. Sharma, P. Singh and M. S. Hegde, J. Solid State Electrochem., 2011, 15, 2185 CrossRef CAS.
- P. A. Deshpande, M. S. Hegde and G. Madras, AIChE J., 2010, 56, 1315 CrossRef CAS.
- R. Di Monte, P. Fornasiero, M. Graziani and J. Kašpar, J. Alloys Compd., 1998, 275, 877 CrossRef.
- G. Dutta, U. V. Waghmare, T. Baidya and M. S. Hegde, Chem. Mater., 2007, 19, 6430 CrossRef CAS.
- A. Gupta, U. V. Waghmare and M. S. Hegde, Chem. Mater., 2010, 22, 5184 CrossRef CAS.
- A. Gupta, A. Kumar, U. V. Waghmare and M. S. Hegde, J. Chem. Phys., 2010, 132, 194702 CrossRef PubMed.
- G. Dutta, A. Gupta, U. V. Waghmare and M. S. Hegde, J. Chem. Sci., 2011, 123, 509 CrossRef CAS.
- Z. Yang, G. Luo, Z. Lu, T. K. Woo and K. Hermansson, J. Phys.: Condens. Matter, 2008, 20, 035210 CrossRef.
- H.-F. Wang, X.-Q. Gong, Y.-L. Guo, Y. Guo, G. Z. Lu and P. Hu, J. Phys. Chem. C, 2009, 113, 10229 CrossRef CAS.
- V. Shapovalov and H. Metiu, J. Catal., 2007, 245, 205 CrossRef CAS.
- Z. Hu and H. Metiu, J. Phys. Chem. C, 2011, 115, 17898 CrossRef CAS.
- E. W. McFarland and H. Metiu, Chem. Rev., 2013, 113, 4391 CrossRef CAS PubMed.
- M. F. Camellone and S. Fabris, J. Am. Chem. Soc., 2009, 131, 10473 CrossRef PubMed.
- H.-L. Chen, J.-G. Chang and H.-T. Chen, Chem. Phys. Lett., 2011, 502, 169 CrossRef CAS.
- D. O. Scanlon, B. J. Morgan and G. W. Watson, Phys. Chem. Chem. Phys., 2011, 13, 4279 RSC.
- A. B. Kehoe, D. O. Scanlon and G. W. Watson, Chem. Mater., 2011, 23, 4464 CrossRef CAS.
- M. Nolan, J. Mater. Chem., 2011, 21, 9160 RSC.
- Y. Zhao, B.-T. Teng, X.-D. Wen, Y. Zhao, L.-H. Zhao and M.-F. Luo, Catal. Commun., 2012, 27, 63 CrossRef CAS.
- H.-T. Chen, J. Phys. Chem. C, 2012, 116, 6239 CrossRef CAS.
- Z. Yang, Z. Lu and G. Luo, Phys. Rev. B: Condens. Matter Mater. Phys., 2007, 76, 075421 CrossRef.
- Z. Lu and Z. Yang, J. Phys.: Condens. Matter, 2010, 22, 475003 CrossRef PubMed.
- A. Bruix, K. M. Neyman and F. Illas, J. Phys. Chem. C, 2010, 114, 14202 CrossRef CAS.
- W. Song, C. Popa, A. P. J. Jansen and E. J. M. Hensen, J. Phys. Chem. C, 2010, 116, 22904 CrossRef.
- J. Paier, C. Penschke and J. Sauer, Chem. Rev., 2013, 113, 3949 CrossRef CAS PubMed.
- X.-Q. Gong, L.-L. Yin, J. Zhang, H.-F. Wang, X.-M. Cao, G. Lu and P. Hu, Adv. Chem. Eng., 2014, 44, 1 CAS.
- A. D. Mayernick and M. J. Janik, J. Catal., 2011, 278, 16 CrossRef CAS.
- S. Chrétien and H. Metiu, Catal. Lett., 2006, 107, 143 CrossRef.
- F. Wang, S. Zhang, C. Li, J. Liu, S. He, Y. Zhao, H. Yan, M. Wei, D. G. Evans and X. Duan, RSC Adv., 2014, 4, 10834 RSC.
| This journal is © The Royal Society of Chemistry 2015 |