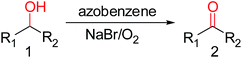Highly practical sodium(I)/azobenzene catalyst system for aerobic oxidation of benzylic alcohols†
Chengkou Liu‡
a,
Zheng Fang‡a,
Zhao Yangb,
Qingwen Lia,
Shiyu Guoa and
Kai Guo *ac
*ac
aCollege of Biotechnology and Pharmaceutical Engineering Nanjing Tech University, 30 Puzhu South Road, Nanjing, 211816, China
bCollege of Engineering China Pharmaceutical University, 24 Tongjiaxiang, Nanjing, 210003, China
cState Key Laboratory of Materials-Oriented Chemical Engineering Nanjing Tech University, 30 Puzhu South Road, Nanjing, 211816, China. E-mail: guok@njtech.edu.cn; Fax: +86 2558139926; Tel: +86 2558139935
First published on 15th September 2015
Abstract
An economic, environmental and practical aerobic oxidation of benzylic alcohols and hetero aryl alcohols to the corresponding carbonyl compounds with good substrate scope is disclosed for the first time. Good to excellent yields were obtained by employing economic and commercially available sodium bromide and a catalytic amount of azobenzene under metal-free and ligand-free conditions. Moreover, aldehydes and acids, the oxidation products of benzylic 1° alcohols, could be obtained using sodium bromide and sodium hydroxide as the co-catalyst respectively in high yields.
Introduction
From both economic and environmental viewpoints, the selective oxidation of benzylic alcohols to the corresponding carbonyl compounds is one of the most challenging transformations. Many traditional and novel methods have been developed with the efforts of many researchers over the past few decades. Manganese1–4 and chromium oxides5–7 may be still the most used oxidants in the oxidation of alcohols, such as KMnO4, MnO2 and PCC. Unfortunately, stoichiometric heavy metal waste was produced at the same time, which limits these reagents into the small-scale. Besides, iodine reagent,8–10 such as Dess–Martin reagent, and activated-DMSO reagent,11,12 such as Swern reagent were built, and the oxidation products of alcohols were obtained smoothly. However, either economics (the iodine reagents were usually costly) or the difficulty of the operation (low temperature should be kept when Swern reagent was used), limits the use of these reagents. Besides these reagents, DDQ (2,3-dichloro-5,6-dicyano-1,4-benzoquinone),13–16 TEMPO (2,2,6,6-tetramethylpiperidinooxy)17–19 and NaClO20–22 were also used as the oxidant of benzylic alcohols. Unfortunately, some drawbacks were existent, such as high cost, using complex ligands and danger. In summary, dioxygen should be the most practical oxidant. In recent years, many oxidation systems of dioxygen have been developed. The most common systems were dioxygen together with noble metals (e.g., Pd, Pt, Au, Ru)23–26 or complex and commercially unavailable ligands.27,28 Consequently, the development of green, economical and practical oxidation process is still a big challenge.Azo compounds have been used as the oxidant of alcohols for decades.29 And, researchers were devoted to finding efficient catalysts and co-catalysts because of the low-reactivity and high cost of azo compounds. Many oxidation systems have been developed, such as azo compounds together with Grignard reagent30 or AZADO (2-azaadamantane-N-oxyl).31 And, many metal compounds were proved to be efficient catalyst for the oxidation of alcohols by azo compounds, such as Cu,32–34 Mg,35 Ni35 and Zn.35 Moreover, copper was used mostly. Lately, the azo compounds were often used as the ligands of the copper to catalyse the oxidation reaction.
Results and discussion
Within the context, we desired to develop a novel oxidation system using catalytic amount of azo compounds together with dioxygen without any heavy metal or complex co-catalyst. The 4-nitrobenzyl alcohol was chosen as the model substrate for our initial study. The detailed results were summarized inTable 1.| Entry | Catalyst (mol%) | Co-catalyst | T (°C) | Solvent | t (h) | Yieldb (%) |
|---|---|---|---|---|---|---|
| a Reaction conditions: 1a (1 mmol), catalyst, co-catalyst (2 mmol), solvent (3 mL),T (°C), under O2 (O2 balloon), DIAD: diisopropyl azodicarboxylate.b Yield: isolated yield.c Under N2. | ||||||
| 1 | DIAD(200) | — | 80 | DMSO | 24 | Trace |
| 2 | DIAD(200) | — | 80 | Toluene | 24 | Trace |
| 3 | DIAD(200) | — | 80 | Dioxane | 24 | Trace |
| 4 | Azobenzene(200) | — | 80 | DMSO | 24 | Trace |
| 5 | Azobenzene(200) | — | 80 | Toluene | 24 | Trace |
| 6 | Azobenzene(200) | — | 80 | Dioxane | 24 | Trace |
| 7 | DIAD(200) | NaBr | 80 | Dioxane | 24 | Trace |
| 8 | Azobenzene(200) | NaBr | 80 | Dioxane | 24 | 90 |
| 9 | Azobenzene(5) | NaBr | 80 | Dioxane | 48 | 88 |
| 10 | Azobenzene(5) | NaBr | 80 | DMF | 48 | 5 |
| 11 | Azobenzene(5) | NaBr | 80 | DMSO | 48 | 8 |
| 12 | Azobenzene(5) | NaBr | 80 | Acetonitrile | 48 | 8 |
| 13 | Azobenzene(5) | NaBr | 80 | Toluene | 48 | 42 |
| 14 | Azobenzene(5) | NaBr | 65 | Dioxane | 48 | 78 |
| 15 | Azobenzene(5) | NaBr | 50 | Dioxane | 48 | 22 |
| 16 | Azobenzene(5) | TBAB | 80 | Dioxane | 48 | Trace |
| 17 | Azobenzene(5) | KBr | 80 | Dioxane | 48 | Trace |
| 18 | Azobenzene(5) | NH4Cl | 80 | Dioxane | 48 | Trace |
| 19 | Azobenzene(5) | NaCl | 80 | Dioxane | 48 | 35 |
| 20 | Azobenzene(5) | NaHCO3 | 80 | Dioxane | 48 | Trace |
| 21 | Azobenzene(5) | Na2CO3 | 80 | Dioxane | 48 | 64 |
| 22 | Azobenzene(5) | NaOH | 80 | Dioxane | 48 | — |
| 23 | Azobenzene(5) | Na2SO4 | 80 | Dioxane | 48 | 82 |
| 24c | Azobenzene(5) | NaBr | 80 | Dioxane | 48 | — |
Firstly, the reaction did not occur when azo compounds were used alone, which indicated that these azo compounds showed lower reactivity (Table 1, entries 1–6). To our delight, the expected oxidation product was obtained when NaBr, the inexpensive and readily available reagent, was added (Table 1, entry 8). However, the reaction did not proceed when DIAD was used. The instability of DIAD may be the main reason (Table 1, entry 7). Fortunately, the decrease of the equivalent of the catalyst azobenzene did not cause the obvious drop of the yield (Table 1, entry 9). Further solvent screening indicated that 1,4-dioxane was the best one amongN,N-dimethylformamide, dimethyl sulfoxide, acetonitrile and toluene (Table 1, entries 9–13). An obvious lower yield was obtained when polar solvent was used, which indicated possible critical complex was unstable in these polar solvent.
When the temperature was decreased to 65 °C, a little drop of the yield was obtained even though the reaction time was extended (Table 1, entry 14). Even worse, the yield was dropped to 22% when the temperature was decreased to 50 °C, which indicated that temperature affected this oxidation reaction obviously (Table 1, entries 9, 14 and 15). In addition, further co-catalyst screened showed that this catalytic process was promoted by the Na+ rather than Br− or K+ (Table 1, entries 9, 16 and 17). And, different Na+ source have different catalytic activity (Table 1, entries 9, 19–23). NaBr was the best choice. It was possible that the catalytic performance of different Na-based co-catalysts was influenced by pKb. And, neutral co-catalysts showed higher catalytic activity. So, relatively good yields were obtained when NaBr, Na2SO4 and NaCl were involved. Moreover, further investigation of the equivalent of NaBr showed the amount of the co-catalyst influenced the reaction process obviously (Table SI†). It might be the reason why Na2CO3 gave a higher yield compared with NaHCO3. No product was detected when the reaction was conducted under Ar atmosphere, which suggested dioxygen was the actual oxidant (Table 1, entries 9 and 24).
We set out to explore the methodology with respect to the substitution of the aryl ring and aliphatic alcohols (Table 2). In general, the oxidation reaction is affected by steric hindrance and electronic factors moderately.18,36And, aliphatic alcohols often show lower reactivity. One set of alcohols oxidation was performed following the optimized procedure above. It was observed that both electron-rich and electron-deficient benzylic alcohols could be oxidized smoothly in moderate to good yields (Table 2, entries 1–10). Nevertheless, electron-withdrawing groups attached to the phenyl rings of substrate showed lower reactivity obviously (Table 2, entries 1–3). As, more reaction time should be needed to reach the full conversion. To our delight, the efficiency of this reaction was not affected obviously by substituents at different positions of the aryl ring and steric hindrance (Table 2, entries 1–3, 9 and 10). Moreover, there was no obvious effect observed between 1° benzylic alcohols and 2° benzylic alcohols and hetero aryl alcohols were oxidized to afford the corresponding products in good yields (Table 2, entries 1–13). Unfortunately, almost no desired product was obtained when aliphatic alcohols were texted (Table 2, entries 14 and 15).
| Entry | Substrates R1 | R2 | t (h) | Product no. | Yieldb (%) |
|---|---|---|---|---|---|
| a Reaction conditions: 1 (1 mmol), azobenzene (0.05 mmol), NaBr (2 mmol), 1,4-dioxane (3 mL), 80 °C, under O2 (O2 balloon).b Yield: isolated yield. | |||||
| 1 | o-NO2Ph | H | 48 | 2b | 86 |
| 2 | m-NO2Ph | H | 48 | 2c | 85 |
| 3 | p-NO2Ph | H | 48 | 2a | 88 |
| 4 | p-OMePh | H | 24 | 2d | 94 |
| 5 | p-ClPh | H | 24 | 2e | 96 |
| 6 | p-ClPh | CH3 | 24 | 2f | 96 |
| 7 | p-BrPh | H | 24 | 2g | 94 |
| 8 | p-BrPh | CH3 | 24 | 2h | 95 |
| 9 | Ph | Ph | 24 | 2i | 92 |
| 10 | 2,6-Dichloro-Ph | H | 24 | 2j | 93 |
| 11 | 2-Furan | H | 24 | 2k | 92 |
| 12 | 2-Thiophene | H | 24 | 2l | 93 |
| 13 | 3-Pyridine | H | 36 | 2m | 89 |
| 14 | Hexyl alcohol | 48 | 2n | Trace | |
| 15 | Cyclohexanol | 48 | 2o | Trace | |
The experimental results above showed that this oxidation system was applicable to all kinds of 1° benzylic alcohols, 2° benzylic alcohols and hetero aryl alcohols. The limitation of steric hindrance and electronic nature could be ignored. A library of aldehydes and ketones were synthesized from various alcohols in good yields (Scheme 1).
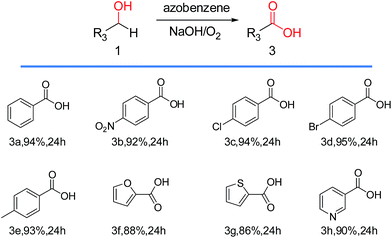
As we all know, the dismutation reaction could occur under the strong alkaline condition when the aldehyde did not involve any α-H.37–39 And, the corresponding alcohol and acid were got. In Table 1, when NaOH was chosen as the co-catalyst, the oxidation of alcohol and the cannizzaro reaction occurred smoothly. And, the acids were obtained in good yields as the final product. To our knowledge, a special base (sodium pyrazolide) could promote the oxidation of 1° benzylic alcohol to the corresponding acid successfully. Afterwards, NaH was found to be a useful replacer in this oxidation system.40 Similarly, NaOH here may be used as the promoter just like the NaH before. In other words, we guessed the transformation could occur normally without any azobenzene. Finally, we found that lei and his co-workers41 reported on aerobic oxidation of benzylic alcohols into the corresponding acids under NaOH/air/THF conditions in 2013. Nevertheless, the oxidation system of azobenzene/NaOH/O2 was still meaningful. As, higher yields of heterocyclic alcohols were obtained compared with oxidation system without azobenzene. Especially, 2,5-furandicarboxylic acid was obtained in excellent isolated yield from oxidation of 5-hydroxymethylfurfural under azobenzene/sodium methanolate/O2. However, an obvious decrease of the yield was observed in the absence of azobenzene, which would be discussed detailed in another paper later.
Following this new protocol, a wide range of acids were obtained from the oxidation of 1° alcohols in excellent yields (Scheme 2). The 1° benzylic alcohols could be oxidized to acid efficiently. Fortunately, the effect of different electronic properties (electron-donating or electron-withdrawing) of the substituents could be ignored. Moreover, the desired products could be got when hetero aryl 1° alcohols were investigated, which indicated that the Na+/azobenzene oxidation system was very practical.
Conclusions
In summary, a practical and economical oxidation system of Na+/azobenzene/O2 was built firstly, although some oxidation systems of Cu/azo compounds (CuCl·Phen·DEAD-H2·K2CO3/CuCl·dpPhen·DBAD·Cs2CO3(ref. 42)) were reported before. A series of benzylic alcohols and hetero aryl alcohols could be oxidized to the corresponding carbonyl compounds smoothly. Moreover, the products of 1° alcohols could be controlled by the different co-catalyst. And, the aldehydes and acids were obtained accordingly. Although the exact mechanism was uncertain, it was easy to believe that the Na+ here played a similar role as Cu42 above.Acknowledgements
The research has been supported by National Key Basic Research Program of China (973 Program) 2012CB725204; the National High Technology Research and Development Program of China (863 Program) 2014AA022101; the National Natural Science Foundation of China (Grant No. U1463201 and 81302632); the National Science and Technology Major Projects for “Major New Drugs Innovation and Development” (2013ZX09103001-004).Notes and references
- J. W. Ladbury and C. F. Cullis, Chem. Rev., 1958, 58, 403–438 CrossRef CAS
.
- H. Du, P. K. Lo, Z. Hu, H. Liang, K. C. Lau, Y. N. Wang, W. W. Lam and T. C. Lau, Chem. Commun., 2011, 47, 7143–7145 RSC
.
- C. S. Kovash Jr, E. Pavlacky, S. Selvakumar, M. P. Sibi and D. C. Webster, ChemSusChem, 2014, 7, 2289–2294 CrossRef PubMed
.
- A. Kamimura, Y. Nozaki, S. Ishikawa, R. Inoue and M. Nakayama, Tetrahedron Lett., 2011, 52, 538–540 CrossRef CAS PubMed
.
- E. Corey and J. W. Suggs, Tetrahedron Lett., 1975, 16, 2647–2650 CrossRef
.
- G. Piancatelli, A. Scettri and M. Dauria, Synthesis, 1982, 245–258 CrossRef CAS
.
- A. De, J. Sci. Ind. Res., 1982, 41, 484–494 CAS
.
- D. B. Dess and J. C. Martin, J. Org. Chem., 1983, 48, 4155–4156 CrossRef CAS
.
- M. Frigerio and M. Santagostino, Tetrahedron Lett., 1994, 35, 8019–8022 CrossRef CAS
.
- M. Uyanik and K. Ishihara, Chem. Commun., 2009, 2086–2099 RSC
.
- A. J. Mancuso, S. L. Huang and D. Swern, J. Org. Chem., 1978, 43, 2480–2482 CrossRef CAS
.
- A. J. Mancuso, D. S. Brownfain and D. Swern, J. Org. Chem., 1979, 44, 4148–4151 CrossRef CAS
.
- Z. Shen, J. Dai, J. Xiong, X. He, W. Mo, B. Hu, N. Sun and X. Hu, Adv. Synth. Catal., 2011, 353, 3031–3038 CrossRef CAS PubMed
.
- L. Wang, J. Li, H. Yang, Y. Lv and S. Gao, J. Org. Chem., 2012, 77, 790–794 CrossRef CAS PubMed
.
- C. C. Cosner, P. J. Cabrera, K. M. Byrd, A. M. A. Thomas and P. Helquist, Org. Lett., 2011, 13, 2071–2073 CrossRef CAS PubMed
.
- K. Walsh, H. F. Sneddon and C. J. Moody, Org. Lett., 2014, 16, 5224–5227 CrossRef CAS PubMed
.
- B. L. Ryland and S. S. Stahl, Angew. Chem., Int. Ed., 2014, 53, 8824–8838 CrossRef CAS PubMed
.
- M. M. Hossain and S.-G. Shyu, Adv. Synth. Catal., 2010, 352, 3061–3068 CrossRef CAS PubMed
.
- J. Hou, Y. Luan, J. Tang, A. M. Wensley, M. Yang and Y. Lu, J. Mol. Catal. A: Chem., 2015, 407, 53–59 CrossRef CAS PubMed
.
- A. B. Leduc and T. F. Jamison, Org. Process Res. Dev., 2012, 16, 1082–1089 CrossRef CAS
.
- Y. Zhang, S. C. Born and K. F. Jensen, Org. Process Res. Dev., 2014, 18, 1476–1481 CrossRef CAS
.
- Y. Zhang, Q. Zhou, W. Ma and J. Zhao, Catal. Commun., 2014, 45, 114–117 CrossRef CAS PubMed
.
- S. M. Alia, K. Duong, T. Liu, K. Jensen and Y. Yan, ChemSusChem, 2014, 7, 1739–1744 CrossRef CAS PubMed
.
- S. E. Davis, M. S. Ide and R. J. Davis, Green Chem., 2013, 15, 17–45 RSC
.
- T. Mallat and A. Baiker, Chem. Rev., 2004, 104, 3037–3058 CrossRef CAS PubMed
.
- D. I. Enache, J. K. Edwards, P. Landon, B. Solsona-Espriu, A. F. Carley, A. A. Herzing, M. Watanabe, C. J. Kiely, D. W. Knight and G. J. Hutchings, Science, 2006, 311, 362–365 CrossRef CAS PubMed
.
- Z. Hu and F. M. Kerton, Appl. Catal., A, 2012, 413–414, 332–339 CrossRef CAS PubMed
.
- M. Mahyari, M. S. Laeini and A. Shaabani, Chem. Commun., 2014, 50, 7855–7857 RSC
.
- F. Yoneda, K. Suzuki and Y. Nitta, J. Org. Chem., 1967, 32, 727–729 CrossRef CAS
.
- K. Narasaka, A. Morikawa, K. Saigo and T. Mukaiyama, Bull. Chem. Soc. Jpn., 1977, 50, 2773–2776 CrossRef CAS
.
- M. Hayashi, M. Shibuya and Y. Iwabuchi, J. Org. Chem., 2012, 77, 3005–3009 CrossRef CAS PubMed
.
- I. E. Marko, A. Gautier, J. L. Mutonkole, R. Dumeunier, A. Ates, C. J. Urch and S. M. Brown, J. Organomet. Chem., 2001, 624, 344–347 CrossRef CAS
.
- T. Nishii, T. Ouchi, A. Matsuda, Y. Matsubara, Y. Haraguchi, T. Kawano, H. Kaku, M. Horikawa and T. Tsunoda, Tetrahedron Lett., 2012, 53, 5880–5882 CrossRef CAS PubMed
.
- M. Hayashi, M. Shibuya and Y. Iwabuchi, J. Org. Chem., 2012, 77, 3005–3009 CrossRef CAS PubMed
.
- H. T. Cao and R. Grée, Tetrahedron Lett., 2009, 50, 1493–1494 CrossRef CAS PubMed
.
- J. M. Hoover and S. S. Stahl, J. Am. Chem. Soc., 2011, 133, 16901–16910 CrossRef CAS PubMed
.
- P. Wang, W.-J. Tao, X.-L. Sun, S. Liao and Y. Tang, J. Am. Chem. Soc., 2013, 135, 16849–16852 CrossRef CAS PubMed
.
- S. Subbiah, S. P. Simeonov, J. M. S. S. Esperanca, L. P. N. Rebelo and C. A. M. Afonso, Green Chem., 2013, 15, 2849–2853 RSC
.
- J. Akhigbe, C. Ryppa, M. Zeller and C. Bruckner, J. Org. Chem., 2009, 74, 4927–4933 CrossRef CAS PubMed
.
- X. Wang and D. Z. Wang, Tetrahedron, 2011, 67, 3406–3411 CrossRef CAS PubMed
.
- J. Wang, C. Liu, J. Yuan and A. Lei, New J. Chem., 2013, 37, 1700 RSC
.
- I. E. Marko, P. R. Giles, M. Tsukazaki, I. Chelle-Regnaut, A. Gautier, S. M. Brown and C. J. Urch, J. Org. Chem., 1999, 64, 2433–2439 CrossRef CAS
.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI:10.1039/c5ra15286k |
| ‡ C.-K. Liu and Z. Fang contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2015 |