Facile and cost-effective synthesis of CNT@MCo2O4 (M = Ni, Mn, Cu, Zn) core–shell hybrid nanostructures for organic dye removal†
Haizhen Li,
Zebin Sun,
Yaxi Tian,
Guijia Cui and
Shiqiang Yan*
College of Chemistry and Chemical Engineering, Lanzhou University, Lanzhou, 730000, P.R. China. E-mail: yansq@lzu.edu.cn; Fax: +86 931 8912582; Tel: +86 931 8912582
First published on 14th September 2015
Abstract
This study presents four hybrid materials of CNT@MCo2O4 (M = Ni, Mn, Cu, Zn) which were prepared via a simple chemical bath deposition method at room temperature followed by a post-annealing treatment. The resulting materials were characterized by different techniques including SEM, EDS, XRD, FTIR, and BET surface area measurement and were first used as adsorbents for removal of methyl orange (MO). Experimental data suggested that the CNT@MCo2O4 samples especially CNTs@NiCo2O4 showed excellent adsorption properties (1188.3 mg g−1). The kinetic adsorption of different materials could be accurately described by the pseudo-second-order model and the overall rate process was apparently influenced by external mass transfer and intraparticle diffusion. Furthermore, Langmuir and Freundlich isotherms are employed and both of them fitted the experimental data with good correlation coefficients. Moreover, the thermodynamic parameters indicated that the adsorption process was spontaneous and physical adsorption mechanisms including electrostatic interaction might play a dominant role in the adsorption mechanism between MO and adsorbents. The results of this work are of great significance for environmental applications of CNT@MCo2O4 as promising adsorbents for organic pollutants from wastewater.
1. Introduction
With the development of technology and industry, aqueous dye pollution, commonly caused by wastewater discharged from textile, leather and paper industries, has emerged as one of most serious pollutions in water especially in developing countries.1 Unfortunately, the color of wastewater is rather difficult to treat because of the non-degradable synthetic origins and aromatic structures of the pollutants.2 Meanwhile, the hazardous pollutants pose ecological risks and are toxic to aquatic life and mankind if discharged directly into receiving waters, because they may be mutagenic and carcinogenic and can cause a severe health hazard to human beings, such as dysfunction of kidney, reproductive system, liver, brain and central nervous system.3,4 Thus, it is imperative to treat the dyes in their wastes to an acceptable level before discharging them into the environment.There are many physical, chemical and biological treatment techniques for removing dyes from industrial effluent, such as chemical oxidation, photochemical degradation, biological degradation, ultrasonic degradation, reverse osmosis, flotation and adsorption procedures.5–10 Among those methods, adsorption is recognized to be the most popular method because of its low cost, easy operation and high removal efficiency.11–17
Among various available adsorbents, transition metal oxides have potential applications in water treatment owing to their excellent characteristics such as environmentally friendly synthesis methods and low price.18 In the large family of metal oxides, spinel cobaltites, MCo2O4 (M = Ni, Mn, Cu, Zn) are very promising multifunctional materials and have been widely used in several applications such as catalysis, environmental remediation and electrochemical supercapacitors as well as adsorption.19–21 However, the low specific area of spinel cobaltites micrometer powder could greatly reduce the contact area of the materials and dye molecules, which could significantly decrease the adsorption capacity of spinel cobaltites. In addition, the pure spinel cobaltites are easy to agglomerate. In order to alleviate the above-mentioned problems, multi-walled CNTs are introduced due to their high specific surface area, very high strength, chemical stability and low density.22–24 The multi-walled CNTs could not only buffer the agglomeration of the active metal oxide nanostructures, but also enhance the specific area of spinel cobaltites. And the active sites of spinel cobaltites can be also further increased, which greatly increased the adsorption capacity for dyes. Thus far, many studies focussing on CNT-supported MCo2O4 have been reported,25–28 however, to the best of our knowledge, few of them are selected to be adsorbents for removing dyes.
In this work, we grew MCo2O4 (M = Ni, Mn, Cu, Zn) on individual CNTs via a simple and cost-effective chemical bath deposition method at room temperature followed by a post-annealing treatment. The chemical co-precipitation method has several advantages such as simplicity, low processing temperature and pressure, low cost, high product purity and the ability to control the morphology compared with most conventional methods. This prepared CNTs@MCo2O4 hybrids possess high specific surface area and display a remarkable adsorption performance for removal organic dyes. And their adsorption capability for organic pollutants were investigated using methyl orange (MO) as a model. The CNTs@MCo2O4 were characterized by field-emission scanning electron microscopy (FESEM), X-ray energy dispersive spectrometry (EDS), X-ray diffraction (XRD), Fourier-transform infrared spectroscopy (FTIR) analysis, Brunauer, Emmett, and Teller surface area measurement (BET) analyses. The adsorption capacities of different materials, effects of pH, contact time at different initial concentrations were examined in bath experiments. Moreover, the adsorption kinetics, rate-controlling mechanisms, and thermodynamics of the adsorbents were also comprehensively investigated.
2. Experimental section
2.1. Materials
MWCNTs having diameter 20–30 nm and length of 5–20 μm, were purchased from Alpha Nano Technology Co., Ltd (China). Polyvinylpyrrolidone (PVP), absolute ethanol and NH3·H2O (25–28 wt%) were purchased from the Sinopharm Group Chemical Reagent Co., Ltd. CoCl2·6H2O, NiCl2·6H2O, MnCl2·4H2O, CuCl2·2H2O and ZnCl2 were obtained from Kelong chemical company (Chengdu, China). All chemicals are of analytical grade and used without further purification.2.2. Preparation of CNT@MCo2O4
CNT@MCo2O4 nanomaterials were fabricated by a simple chemical bath deposition method at room temperature followed by a post-annealing treatment. And the detailed preparation procedure is illustrated in Scheme 1. Taking the preparation of CNT@NiCo2O4 as an example, briefly, 35 mg MWCNT powder was uniformly dispersed into 100 mL 7 mg mL−1 PVP aqueous solution with ultrasonication for 1.5 h. Then 0.9517 g (4 mmol) of CoCl2·6H2O and 0.4754 g (2 mmol) of NiCl2·6H2O were dissolved into the MWCNT dispersion with vigorous stirring for 0.5 h to form a black solution, followed by a drop-by-drop addition of 5% NH3·H2O solution until its pH reached 9. The mixture was kept still for 4 h at room temperature. The obtained precipitate was filtered, washed with water and ethanol several times to remove the surfactant and the residual ions, and dried at 60 °C for 12 h under vacuum. In order to obtain the CNT@NiCo2O4 nanostructure, the hybrid precursor was finally annealed in air at 300 °C for 3 h with a slow heating rate of 1 °C min−1. With the same procedure and amounts of CoCl2·6H2O and MWCNT, MnCl2·4H2O (0.396 g, 2 mmol), CuCl2·2H2O (0.341 g, 2 mmol) and ZnCl2 (0.273 g, 2 mmol) were used instead of NiCl2·6H2O for the syntheses of other CNT@MCo2O4 (M = Mn, Cu and Zn) hybrids, respectively.2.3. Material characterizations
The as-prepared CNT@MCo2O4 nanomaterials were characterized with field-emission scanning electron microscopy (FESEM, JSM-6701F) and X-ray energy dispersive spectrometry (EDS, Apollo40SDD). The crystallinity of the samples was investigated by X-ray diffraction (XRD, XRD-6000, Shimadzu, Japan). Fourier transform infrared spectra (FTIR) were carried out using a NEXUS 670 FTIR equipped with a pressed KBr pellets in wavenumber range of 400–4000 cm−1. The BET surface area, pore volume, and pore size were characterized using nitrogen adsorption at liquid nitrogen temperatures (Sorptomatic 1990, Thermo, USA).2.4. Adsorption experiments
Standard solutions of methyl orange (MO) in six different concentrations (100–600 mg L−1) were prepared by dissolving analytical grade methyl orange in distilled water.To determine the adsorption capacities of different materials, the CNT@MCo2O4 were prepared as adsorbents for MO removal. 5 mg sample of the different adsorbents was added to each 25 mL of MO solution with initial concentration of 100 mg L−1 to 600 mg L−1 (100 mg L−1 intervals). The mixture was sonicated for 5 minutes at room temperature and stirred for 120 minutes and then filtered using a 0.45 μm microporous membrane filter. The filtered MO solutions were diluted and measured by a spectrophotometric method at 465 nm.
The effect of pH on MO adsorption was studied by varying the solution pH from 3.0 to 11.0, with the initial MO concentration of 100 mg L−1. The pH of each solution was adjusted using 1 M HCl or NaOH solution.
Adsorption kinetic experiments were conducted by adding 300 mg of different CNT@MCo2O4 nanomaterials to each of 1500 mL MO solution, and the MO concentration was 600 mg L−1 at 298 K. Samples were withdrawn at predetermined time intervals and filtered through a 0.45 μm filter.
The amount of MO adsorption at equilibrium qe (mg g−1) was calculated using the following equation:
 | (1) |
3. Results and discussion
3.1. Characterization of the adsorbents
The morphology of the CNT@NiCo2O4, CNT@MnCo2O4, CNT@CuCo2O4, CNT@ZnCo2O4 and CNT were characterized by field-emission scanning electron microscopy (FESEM) observation and transmission electron microscope (TEM). Fig. 1(a–d) shows the representative FESEM images of the as-prepared CNT@MCo2O4 hybrids. It can be found that the surface of CNT@MCo2O4 is flake shaped and finegrained. And the surface of CNT (Fig. 1(e)) is relatively smooth compared with CNT@MCo2O4. Fig. 1(f) is the TEM of CNT@NiCo2O4. It can be seen that NiCo2O4 is uniformly coated on the CNT. However, the morphology of the CNT is not easy to distinguish. The possible reason is that CNT has only left a skeleton through annealing process in air in the process of our material preparation. In all cases, MCo2O4 nanomaterials were found well-grown around individual CNTs to form a conformal coating on the surface, showing that the method of chemical bath deposition at room temperature followed by a post-annealing treatment could be generally used in the synthesis of various CNT@MCo2O4 hybrids. | ||
| Fig. 1 SEM images of (a) CNT@NiCo2O4, (b) CNT@MnCo2O4, (c) CNT@CuCo2O4, (d) CNT@ZnCo2O4 and (e) CNT. TEM image of (f) CNT@NiCo2O4. | ||
To confirm the composition of the hybrids, the EDS attached to the FESEM was measured and the corresponding EDS spectra of the as-prepared CNT@MCo2O4 hybrids was displayed in Fig. 2(a–d). The EDS spectra of the CNT@MCo2O4 hybrids revealed the presence of M (Ni, Mn, Cu, and Zn), Co, C and O with the atomic ratio of M/Co close to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2. The C mainly comes from the CNTs, while O from both MCo2O4 and some oxygen-containing functional groups of CNTs. The results of EDS confirmed that the MCo2O4 were successfully synthesized.
2. The C mainly comes from the CNTs, while O from both MCo2O4 and some oxygen-containing functional groups of CNTs. The results of EDS confirmed that the MCo2O4 were successfully synthesized.
The crystallographic phase of CNT@NiCo2O4, CNT@MnCo2O4, CNT@CuCo2O4, CNT@ZnCo2O4 and CNT were identified by XRD, and the typical wide-angle diffraction patterns are shown in Fig. 3(a–e). It can be noticed that CNT@MCo2O4 consisted of seven well-defined diffraction peaks, which can be well indexed to the (220), (311), (222), (400), (422), (511), and (440) crystal planes of cubic spinel structural NiCo2O4 (JCPDS card no. 20-0781), MnCo2O4 (JCPDS card no. 23-1237), CuCo2O4 (JCPDS card no. 78-2177), and ZnCo2O4 (JCPDS card no. 23-1390), respectively.29–32 It is worth mentioning that no other peaks are observed, which effectively confirms the purity of the obtained MCo2O4 phase, which was supported on CNTs. For CNT, two characteristic peaks can be clearly observed, corresponding to the (002) and (100) crystal planes. However, the typical peaks (002) of CNT are not observed in CNT@MCo2O4. The possible reason is that the typical peaks of CNT are covered by the peaks of MCo2O4.
 | ||
| Fig. 3 XRD patterns of (a) CNT@NiCo2O4, (b) CNT@MnCo2O4, (c) CNT@CuCo2O4, (d) CNT@ZnCo2O4 and (e) CNT. | ||
The FTIR spectra of CNT@NiCo2O4, CNT@MnCo2O4, CNT@CuCo2O4, CNT@ZnCo2O4 and CNT are presented in Fig. 4(a–e). It is obvious that all spectra exhibit adsorption bands at about 3400, and 1625 cm−1, which can be associated to the –OH stretching and bending vibration for the free or physically absorbed water molecules.33,34 The corresponding metal oxide characteristic peaks positioned at 650 and 555 cm−1 for the samples (a–d) further reveals the formation of the spinel MCo2O4 nanostructure. These bands mainly correspond to the stretching vibrational modes of M–O (M = Ni, Mn, Cu, Zn) in the tetrahedral positions and Co–O bands in octahedral positions, respectively.34
 | ||
| Fig. 4 FTIR spectra of (a) CNT@NiCo2O4, (b) CNT@MnCo2O4, (c) CNT@CuCo2O4, (d) CNT@ZnCo2O4 and (e) CNT. | ||
As illustrated in Fig. 5, the adsorption and desorption isotherms of N2 for the CNT@NiCo2O4, CNT@MnCo2O4, CNT@CuCo2O4, CNT@ZnCo2O4 and CNT are clearly of type IV, according to the IUPAC classification of adsorption isotherms.35 Type IV isotherms characteristically show the simultaneous presence of micro- and mesopores.36 And the physical properties of the CNT@NiCo2O4, CNT@MnCo2O4, CNT@CuCo2O4, CNT@ZnCo2O4 and CNT, such as surface areas, total pore volumes, pore size are summarized in Table 1. It can be seen that the BET surface area, total pore volume and average pore size of CNT@NiCo2O4 are 144.0 m2 g−1, 0.8506 cm3 g−1 and 23.63 nm, respectively, which were higher than that of other CNT@MCo2O4. Generally, the BET surface area, total pore volume and average pore size have great influence on the material performance especially the adsorption capacity. Thus, the CNT@MCo2O4 may have excellent adsorption performance, and this conclusion will be confirmed in the part 3.2.1.
 | ||
| Fig. 5 Typical N2 adsorption–desorption isotherms of CNT@NiCo2O4, CNT@MnCo2O4, CNT@CuCo2O4, CNT@ZnCo2O4 and CNT. | ||
| Materials | Surface area (m2 g−1) | Pore volume (cm3 g−1) | Pore size (nm) |
|---|---|---|---|
| CNT@NiCo2O4 | 144.0 | 0.8506 | 23.63 |
| CNT@MnCo2O4 | 103.64 | 0.5877 | 22.68 |
| CNT@CuCo2O4 | 73.45 | 0.2630 | 14.32 |
| CNT@ZnCo2O4 | 120.58 | 1.0156 | 32.99 |
| CNT | 174.48 | 0.8532 | 19.56 |
3.2. Adsorption experiment studies
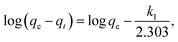 | (2) |
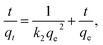 | (3) |
The values of qe and k1 for the pseudo-first-order kinetic model are determined from the intercepts and the slopes of the plots of log(qe − qt) versus t, and plots of t/qt versus t allows calculation of the rate constant k2 and qe for the pseudo-second-order kinetic model. The straight line plots of them are presented in Fig. S1 and S2,† respectively. The calculated kinetic parameters and correlation coefficients for the removal of MO with CNT@MCo2O4 are tabulated in Table 2. It is obvious that all the experimental data showed better compliance with the pseudo-second-order kinetic model in terms of higher correlation coefficient values (r2 > 0.99). Furthermore, the qe calculated from the pseudo-second-order kinetic model are rather approximate with those from the experimental results. These results suggest that the pseudo-second-order kinetic model is more suitable to describe the adsorption process.
| Adsorbents | qe,exp | Pseudo-first-order model | Pseudo-second-order model | ||||
|---|---|---|---|---|---|---|---|
| k1 | qe | r2 | k2 × 10−4 | qe | r2 | ||
| CNT@NiCo2O4 | 1211 | 0.01621 | 374.7 | 0.9027 | 2.809 | 1206.9 | 0.9939 |
| CNT@MnCo2O4 | 678.4 | 0.03871 | 291.6 | 0.9627 | 2.754 | 709.2 | 0.9996 |
| CNT@CuCo2O4 | 804 | 0.01911 | 381.4 | 0.9796 | 2.20 | 819.7 | 0.9907 |
| CNT@ZnCo2O4 | 906.9 | 0.02796 | 360.4 | 0.9152 | 2.018 | 925.9 | 0.9918 |
| qt = kdt1/2 + C | (4) |
The multilinear plots of intra-particle diffusion process of MO adsorption onto CNT@MCo2O4 are presented in Fig. 9. It suggests that two steps have taken place during the adsorption process. The first linear portion is related to external diffusion which is the diffusion of MO molecules from solution to the external surface of CNT@MCo2O4. And the second regions can be attributed to the internal diffusion of MO from the external surface to the internal pores of the adsorbent. According to the Weber–Morris kinetic model, if the regression of qt versus t1/2 is linear and passes through the origin, then intra-particle diffusion is the sole rate-limiting step. The regression is linear, but the plot does not pass through the origin, suggesting that adsorption involve intra-particle diffusion, but that is not the only rate-controlling step.42 From the Fig. 9, it can be noticed that the plots did not pass through the origin, indicating that both external diffusion and intra-particle diffusion are significant.
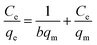 | (5) |
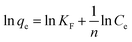 | (6) |
The separation factor (RL) related to Langmuir isotherm is also used to evaluate the feasibility of adsorption on adsorbent. It can be defined as follows:
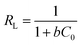 | (7) |
In this equation, C0 represents the initial MO concentration (mg L−1) and b (L mg−1) refers to the Langmuir constant. And the values of RL suggest that the types of the isotherm which can be irreversible (RL = 0), favorable (0 < RL < 1), linear (RL = 1) or unfavorable (RL > 1).
The Langmuir and Freundlich isotherms are shown in Fig. S3 and S4,† respectively. The parameters such as b, qm, KF, n, RL and R2 values determined from these two isotherms are displayed in Table 3. According to the determination parameters, both Langmuir and Freundlich models closely fitted the experimental data with good correlation coefficients (R2). And the RL values are lying in the 0–1 range, demonstrating that the isotherms are favorable for adsorption of MO onto CNT@MCo2O4. The computed maximum MO adsorption capacities are 1257, 840.3, 869.6 and 980.4 mg g−1 at 293 K for CNT@NiCo2O4, CNT@MnCo2O4, CNT@CuCo2O4 and CNT@ZnCo2O4, respectively. It is clear that the CNT@MCo2O4 have excellent adsorption performance.
| Adsorbents | Langmuir isotherm | Freundlich isotherm | ||||||
|---|---|---|---|---|---|---|---|---|
| qm | b | r2 | RL × 10 | 1/n | KF | r2 | ||
| CNT@NiCo2O4 | 1257 | 0.0300 | 0.9900 | 0.525–2.497 | 0.2587 | 261.20 | 0.9954 | |
| CNT@MnCo2O4 | 840.3 | 0.0265 | 0.9939 | 0.591–2.737 | 0.2287 | 200.25 | 0.9823 | |
| CNT@CuCo2O4 | 869.6 | 0.0308 | 0.9955 | 0.513–2.452 | 0.2142 | 229.05 | 0.9949 | |
| CNT@ZnCo2O4 | 980.4 | 0.0322 | 0.9889 | 0.491–2.367 | 0.1794 | 310.7 | 0.9507 | |
Thermodynamic parameters can improve our understanding regarding the inherent energetic changes involved during the sorption process. The free energy change (ΔG0) was calculated from the variation of the thermodynamic equilibrium constant (K0) to estimate the type of adsorption for the CNT@MCo2O4 toward MO, which was determined by the following equations:9
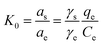 | (8) |
ΔG0 = −RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K0 K0
| (9) |
In the equations, K0 is the thermodynamic equilibrium constant; as is the activity of the adsorbed MO, ae is the activity of the MO in the solution at equilibrium, γs is the activity coefficient of the adsorbed MO, γe is the activity coefficient of MO in solution at equilibrium; R is the gas constant (8.314 J mol−1 K−1); T is the sorption temperature in Kelvin. As the MO concentration in the solution decreases and approaches zero, the activity coefficient approaches unity, and reducing eqn (8) to the following form:
 | (10) |
Therefore, the values of K0 can be acquired by plotting ln(qe/Ce) versus qe and by extrapolating qe to zero, as shown in Fig. S5.†
The acquired thermodynamic parameters such as ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K0, ΔG0 and R2 are shown in Table 4. The negative ΔG0 values confirm the feasibility of the process and the spontaneous nature of the adsorption. Generally ΔG0 for physisorption is less than that for chemisorption. The former is between −20 and 0 kJ mol−1 and the latter is between −80 and −400 kJ mol−1.45 Thus, the ΔG0 results implied that physisorption might dominate the adsorption of MO onto CNT@MCo2O4.
K0, ΔG0 and R2 are shown in Table 4. The negative ΔG0 values confirm the feasibility of the process and the spontaneous nature of the adsorption. Generally ΔG0 for physisorption is less than that for chemisorption. The former is between −20 and 0 kJ mol−1 and the latter is between −80 and −400 kJ mol−1.45 Thus, the ΔG0 results implied that physisorption might dominate the adsorption of MO onto CNT@MCo2O4.
| Adsorbents | ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K0 K0 |
r2 | ΔG0 (kJ mol−1) |
|---|---|---|---|
| CNT@NiCo2O4 | 5.4891 | 0.9724 | −13.378 |
| CNT@MnCo2O4 | 5.2259 | 0.9844 | −12.737 |
| CNT@CuCo2O4 | 5.7166 | 0.9995 | −13.933 |
| CNT@ZnCo2O4 | 6.5943 | 0.8801 | −16.072 |
3.3. Comparison of adsorbent performance with literature data
Previously some researchers investigated several adsorbents for the removal of MO from aqueous solutions. Table 5 shows a comparison of the adsorption capacities of MO onto various adsorbents. It clearly can be seen that the MO removal capacities of CNT@MCo2O4 (M = Ni, Mn, Cu, Zn) are higher than that of other adsorbents,38,39,46–51 indicating that CNT@MCo2O4 nanomaterials have great potential as dye adsorbents in wastewater treatment.| Adsorbates | Adsorbents | qmax (mg g−1) | Ref. |
|---|---|---|---|
| Methyl orange | Carbon nanotubes | 52.9 | 46 |
| CNTs-A | 149 | 38 | |
| NH2-MWCNTs | 185.5 | 47 | |
| Activated carbon/Fe3O4 nanocomposites | 303.0 | 48 | |
| Mesoporous MgO nanoplates | 370 | 49 | |
| SiO2–Al2O3 mixed oxides | 381 | 50 | |
| Powdered activated carbon modified by HNO3 | 384.6 | 48 | |
| H-δ-MnO2 nanoparticles | 427 | 51 | |
| Phragmites australis activated carbon | 437.9 | 39 | |
| CNT@NiCo2O4 | 1188.3 | This work | |
| CNT@MnCo2O4 | 790.5 | This work | |
| CNT@CuCo2O4 | 826.9 | This work | |
| CNT@ZnCo2O4 | 935.4 | This work |
4. Conclusions
In summary, CNT@MCo2O4 (M = Ni, Mn, Cu, Zn) nanomaterials were prepared via a simple chemical bath deposition method at room temperature followed by a post-annealing treatment. Their adsorption properties for the removal of MO from aqueous solution have been studied for the first time and the adsorption process were systematically researched under different conditions including pH, contact time, initial MO concentrations. The CNT@MCo2O4 samples especially CNTs@NiCo2O4 showed excellent adsorption properties. And the maximum adsorption capacity for CNT@MCo2O4 (M = Ni, Mn, Cu, Zn) are 1188.3, 790.5, 826.9 and 935.4 mg g−1, respectively. The high adsorption capacity of CNT@MCo2O4 may be ascribed to their high BET surface area. The kinetic data of the adsorption fitted well with the pseudo-second-order kinetic model and the results of Weber–Morris kinetic model indicated that overall process appeared to be jointly influenced by intraparticle diffusion and external mass transfer. Moreover, Langmuir and Freundlich isotherms are employed to describe the equilibrium adsorption and both of them fitted the experimental data with good correlation coefficients. The adsorption mechanism was also discussed, which could be attributed to physical adsorption including electrostatic interactions and hydrogen bond interaction between MO and the adsorbents. The results obtained in this work suggest that CNTs@MCo2O4 should be a promising adsorbent for the removal of MO from wastewater.References
- P. Luo, Y. Zhao, B. Zhang, J. Liu, Y. Yang and J. Liu, Water Res., 2010, 44, 1489–1497 CrossRef CAS PubMed.
- G. Crini, Bioresour. Technol., 2006, 97, 1061–1085 CrossRef CAS PubMed.
- Z. Ajji and A. M. Ali, Nucl. Instrum. Methods Phys. Res., Sect. B, 2007, 265, 362–365 CrossRef CAS.
- K. Kadirvelu, M. Kavipriya, C. Karthika, M. Radhika, N. Vennilamani and S. Pattabhi, Bioresour. Technol., 2003, 87, 129–132 CrossRef CAS PubMed.
- Y. Haldorai and J.-J. Shim, Appl. Surf. Sci., 2014, 292, 447–453 CrossRef CAS.
- P. Xu, G. M. Zeng, D. L. Huang, C. L. Feng, S. Hu, M. H. Zhao, C. Lai, Z. Wei, C. Huang, G. X. Xie and Z. F. Liu, Sci. Total Environ., 2012, 424, 1–10 CrossRef CAS PubMed.
- V. K. Gupta, S. Agarwal and T. A. Saleh, J. Hazard. Mater., 2011, 185, 17–23 CrossRef CAS PubMed.
- L. Zhang, H. Li, Y. Liu, Z. Tian, B. Yang, Z. Sun and S. Yan, RSC Adv., 2014, 4, 48703–48711 RSC.
- H. Li, L. Zhang, Z. Sun, Y. Liu, B. Yang and S. Yan, RSC Adv., 2015, 5, 31787–31797 RSC.
- Z. Tian, B. Yang, G. Cui, L. Zhang, Y. Guo and S. Yan, RSC Adv., 2015, 5, 2266–2275 RSC.
- B. Qiu, C. Xu, D. Sun, H. Yi, J. Guo, X. Zhang, H. Qu, M. Guerrero, X. Wang, N. Noel, Z. Luo, Z. Guo and S. Wei, ACS Sustainable Chem. Eng., 2014, 2, 2070–2080 CrossRef CAS.
- B. Qiu, C. Xu, D. Sun, Q. Wang, H. Gu, X. Zhang, B. L. Weeks, J. Hopper, T. C. Ho, Z. Guo and S. Wei, Appl. Surf. Sci., 2015, 334, 7–14 CrossRef CAS.
- B. Qiu, C. Xu, D. Sun, H. Wei, X. Zhang, J. Guo, Q. Wang, D. Rutman, Z. Guo and S. Wei, RSC Adv., 2014, 4, 29855 RSC.
- P. S. Guru and S. Dash, Adv. Colloid Interface Sci., 2014, 209, 49–67 CrossRef CAS PubMed.
- P. S. Guru and S. Dash, J. Dispersion Sci. Technol., 2012, 34, 1099–1112 CrossRef.
- P. S. Guru and S. Dash, J. Dispersion Sci. Technol., 2012, 34, 898–907 CrossRef.
- P. S. Guru and S. Dash, J. Dispersion Sci. Technol., 2011, 33, 1012–1020 CrossRef.
- M. Zhi, C. Xiang, J. Li, M. Li and N. Wu, Nanoscale, 2013, 5, 72–88 RSC.
- F. Cai, Y. Kang, H. Chen, M. Chen and Q. Li, J. Mater. Chem. A, 2014, 2, 11509 CAS.
- J. Cheng, H. Yan, Y. Lu, K. Qiu, X. Hou, J. Xu and Y. Luo, J. Mater. Chem. A, 2015, 3, 9769–9776 CAS.
- S. K. Bikkarolla and P. Papakonstantinou, J. Power Sources, 2015, 281, 243–251 CrossRef CAS.
- J. M. Schnorr and T. M. Swager, Chem. Mater., 2011, 23, 646–657 CrossRef CAS.
- I. Dumitrescu, P. R. Unwin and J. V. Macpherson, Chem. Commun., 2009, 6886–6901, 10.1039/b909734a.
- X. Chen, H. Zhu, Y.-C. Chen, Y. Shang, L. Hu and G. W. Rubloff, ACS Nano, 2012, 6, 7948–7955 CrossRef CAS PubMed.
- X. Wang, X. Han, M. Lim, N. Singh, C. L. Gan, M. Jan and P. S. Lee, J. Phys. Chem. C, 2012, 116, 12448–12454 CAS.
- F. Deng, L. Yu, G. Cheng, T. Lin, M. Sun, F. Ye and Y. Li, J. Power Sources, 2014, 251, 202–207 CrossRef CAS.
- G. Zhang and X. W. David Lou, Sci. Rep., 2013, 3, 1470 Search PubMed.
- W.-w. Liu, C. Lu, K. Liang and B. K. Tay, J. Mater. Chem. A, 2014, 2, 5100–5107 CAS.
- H. S. Jadhav, R. S. Kalubarme, C.-N. Park, J. Kim and C.-J. Park, Nanoscale, 2014, 6, 10071–10076 RSC.
- A. K. Mondal, D. Su, S. Chen, A. Ung, H. S. Kim and G. Wang, Chem.–Eur. J., 2015, 21, 1526–1532 CrossRef CAS PubMed.
- S. Sun, Z. Wen, J. Jin, Y. Cui and Y. Lu, Microporous Mesoporous Mater., 2013, 169, 242–247 CrossRef CAS.
- L. Guo, Q. Ru, X. Song, S. Hu and Y. Mo, J. Mater. Chem. A, 2015, 3, 8683–8692 CAS.
- V. Subramanian, Solid State Ionics, 2004, 175, 511–515 CrossRef CAS.
- Z. Liu, R. Ma, M. Osada, K. Takada and T. Sasaki, J. Am. Chem. Soc., 2005, 127, 13869–13874 CrossRef CAS PubMed.
- K. S. W. Sing, D. H. Everett, R. A. W. Haul, L. Moscou, R. A. Pierotti, J. Rouquerol and T. Siemieniewska, Pure Appl. Chem., 1985, 57, 603–619 CrossRef CAS.
- G. Jiang, Z. Lin, C. Chen, L. Zhu, Q. Chang, N. Wang, W. Wei and H. Tang, Carbon, 2011, 49, 2693–2701 CrossRef CAS.
- Y. Dong, H. Lin, Q. Jin, L. Li, D. Wang, D. Zhou and F. Qu, J. Mater. Chem. A, 2013, 1, 7391 CAS.
- J. Ma, F. Yu, L. Zhou, L. Jin, M. Yang, J. Luan, Y. Tang, H. Fan, Z. Yuan and J. Chen, ACS Appl. Mater. Interfaces, 2012, 4, 5749–5760 CAS.
- S. Chen, J. Zhang, C. Zhang, Q. Yue, Y. Li and C. Li, Desalination, 2010, 252, 149–156 CrossRef CAS.
- I. D. Mall, V. C. Srivastava and N. K. Agarwal, Dyes Pigm., 2006, 69, 210–223 CrossRef CAS.
- C. Luo, Z. Tian, B. Yang, L. Zhang and S. Yan, Chem.–Eur. J., 2013, 234, 256–265 CAS.
- S. Vasiliu, I. Bunia, S. Racovita and V. Neagu, Carbohydr. Polym., 2011, 85, 376–387 CrossRef CAS.
- H. Aydın and G. Baysal, Desalination, 2006, 196, 248–259 CrossRef.
- L. Chen and B. Bai, Ind. Eng. Chem. Res., 2013, 52, 15568–15577 CrossRef CAS.
- Y. Yu, Y.-Y. Zhuang, Z.-H. Wang and M.-Q. Qiu, Chemosphere, 2004, 54, 425–430 CrossRef CAS PubMed.
- Y. Yao, H. Bing, X. Feifei and C. Xiaofeng, Chem.–Eng. J., 2011, 170, 82–89 CrossRef CAS.
- Y. Liu, G. Cui, C. Luo, L. Zhang, Y. Guo and S. Yan, RSC Adv., 2014, 4, 55162–55172 RSC.
- M. H. Do, N. H. Phan, T. D. Nguyen, T. T. Pham, V. K. Nguyen, T. T. Vu and T. K. Nguyen, Chemosphere, 2011, 85, 1269–1276 CrossRef CAS PubMed.
- X. Li, W. Xiao, G. He, W. Zheng, N. Yu and M. Tan, Colloids Surf., A, 2012, 408, 79–86 CrossRef CAS.
- M. Arshadi, F. Salimi Vahid, J. W. L. Salvacion and M. Soleymanzadeh, Appl. Surf. Sci., 2013, 280, 726–736 CrossRef CAS.
- Y. Liu, C. Luo, J. Sun, H. Li, Z. Sun and S. Yan, J. Mater. Chem. A, 2015, 3, 5674–5682 CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra14748d |
| This journal is © The Royal Society of Chemistry 2015 |






