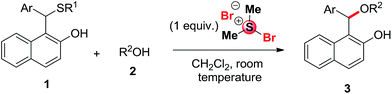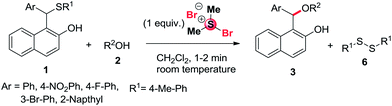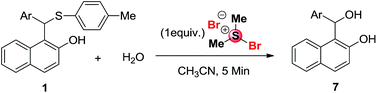A direct approach for the expedient synthesis of unsymmetrical ethers by employing bromodimethylsulfonium bromide (BDMS) mediated C–S bond cleavage of naphthalene-2-ol sulfides†
Kobirul Islama,
R. Sidick Basha‡
a,
Ajaz A. Dar‡a,
Deb K. Das‡a and
Abu T. Khan*ab
aDepartment of Chemistry, Indian Institute of Technology Guwahati, Guwahati 781 039, India. E-mail: atk@iitg.ernet.in; Fax: +91 361 2582349; Tel: +91 361 2582305
bAliah University, IIA/27, New Town, Kolkata 700 156, West Bengal, India
First published on 9th September 2015
Abstract
The unsymmetrical ether derivatives 1-(alkoxy(aryl)methyl)naphthalen-2-ols (3a–q) were synthesized from 1-(aryl(alkyl/arylthio)methyl)-naphthalene-2-ol derivatives (1) and alcohols (2) by cleavage of C–S bond using bromodimethylsulfonium bromide (BDMS) at room temperature. Moreover, 1-(hydroxy(aryl)methyl)naphthalen-2-ol derivatives (7a–d) were also synthesized from the corresponding sulfide derivatives (1) on reaction with water using one equivalent BDMS. The direct synthesis of 3a was also achieved from β-napthol 5 by tuning the reaction conditions with overall one-pot two-steps sequence. Interestingly, the desired products 3a are not accessible directly from 2-naphthol, aromatic aldehyde and alcohol in the presence of BDMS at room temperature. Some of the salient features of this protocol are mild reaction conditions, good yields, operational simplicity and with a wide substrate scope.
Introduction
The construction of an ether linkage adjacent to a sterically hindered carbon center is an important synthetic step for the synthesis of many biologically active compounds.1 Ethers are important structural motifs for life sciences and polymer industries.2 The conventional method for ether synthesis is the direct SN2-type O-alkylation also known as Williamson ether synthesis.3 However, this protocol is sometimes synthetically impractical owing to the strong basicity of the alkoxide anion, which may be incompatible with other functional groups present in the system.4 A variety of transition metal5 or Lewis acid6 catalyzed cross-coupling reactions of different alcohols have also been used for the synthesis of ethers. It is well-known that substituted 2-naphthoquinone-1-methide intermediates can be generated easily from 2-naphthol and aromatic aldehydes in the presence of a suitable catalyst and it has been utilized for the synthesis of carbamate derivatives using aza-Michael reaction,7a which is better known as Betti reaction.7b Recently we have demonstrated that synthesis of 1-[(alkyl/arylthio) (phenyl)methyl]-naphthalene-2-ol derivatives (1) from in situ generated 2-naphthoquinone-1-methide intermediate involving thia-Michael reaction.8 From this result, we inspired to investigate whether in situ generated substituted 2-naphthoquinone-1-methide9 intermediates used for the synthesis of ethers with alcohols by employing oxa-Michael reaction.10 In this paper we would like to disclose the synthesis of hitherto unreported ether derivatives (3) from 1-(aryl(alkyl/arylthio)methyl)-naphthalene-2-ol derivatives (1) by cleavage of C–S bond using one equivalent BDMS followed by oxa-Michael type reaction with alcohols (2) using alternate pathway as shown in Scheme 1.Results and discussion
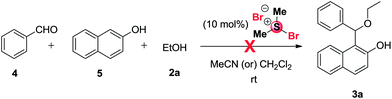
Initial attempts with a mixture of benzaldehyde (4, 1 mmol), 2-naphthol (5, 1 mmol) and ethyl alcohol (2a, 2 mmol) was examined in the presence of 10 mol% of BDMS in 2 mL of CH3CN or DCM at room temperature. Unfortunately, the attempt was unsuccessful as shown in Scheme 2 and the expected unsymmetrical ether (3a) is not formed instead of that we have isolated 1-bromo-2-naphthol in trace amount.From the results above, we turned our attention for devising an alternate strategy. Recently our research group demonstrated that organic ammonium tribromides (OATB) and in situ generated bromonium ion can be utilized for C–S bond cleavage in deprotection of dithioacetals11 and in hydrolysis of 1-thioglycosides.12 Therefore, we thought that it might be possible to synthesize 1-(alkoxy(aryl)methyl)naphthalen-2-ol derivatives (3) from 1-[aryl(alkyl/arylthio)methyl)-naphthalene-2-ol derivatives (1) by cleavage of C–S bond through activation with bromonium ion generated from BDMS followed by nucleophilic attack with alcohols (2).
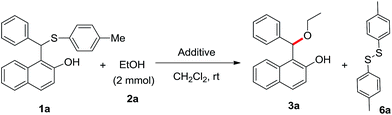
Bromodimethylsulfonium bromide (BDMS) is a useful brominating agent as well as highly efficient pre-catalyst, which has been used extensively by us13 and others14 for numerous organic transformations. We have prepared the unsymmetrical sulfide (1a) from benzaldehyde (4), 2-naphthol (5) and 4-methylthiophenol by following our earlier reported procedure.8a Then, various trial reactions were examined with unsymmetrical sulfide 1a using different amount of BDMS as shown in Table 1. We have isolated the desired product 3a along with unreacted starting material 1a (entries 1–3, Table 1) in the presence of 0.10, 0.50 and 0.75 equivalent of BDMS. We have noted the complete conversion with 1.0 equivalent of BDMS using 2 mmol of EtOH in DCM (2 mL) at room temperature and the desired product 3a was isolated in 94% (entry 4, Table 1). The required product 3a and the by-product 6a15 were characterized from 1H NMR and 13C NMR analysis, which encouraged us to further investigate the reaction. We have also noted that similar transformation is also possible using molecular bromine, NBS and TBATB (entries 5–7, Table 1) however, they provide the desired product in lower yield as compared to BDMS.
After optimizing the reaction conditions, we prepared various sulfide derivatives (1a–f) from aromatic aldehydes, 2-naphthol (5) and 4-methylthiophenol using 10 mol% BDMS as catalyst in acetonitrile at room temperature (entries 1–6, Table SI-S1, ESI†). Similarly, other sulfides (1g–i) were also prepared using different thiols such as 4-chlorothiophenol, ethane thiol and propanethiol under identical reaction conditions (entries 7–9, Table SI-S1, ESI†).
Next, several reactions were performed with compound 1a with various alcohols like methanol (2b), n-propanol (2c), iso-propanol (2d), n-butanol (2e), benzyl alcohol (2f) and the desired products 3b–f were obtained in good yields on treatment with one equivalent of BDMS at room temperature. The reactions completed instantaneously and the percentage yields of the products are shown in Table 2 (entries 2–6).
| a The reactions were carried out with 1 (1.0 mmol) and different aliphatic alcohols 2 (2.0 mmol) in the presence of one equiv. of BDMS in 2 mL of DCM at room temperature. |
|---|
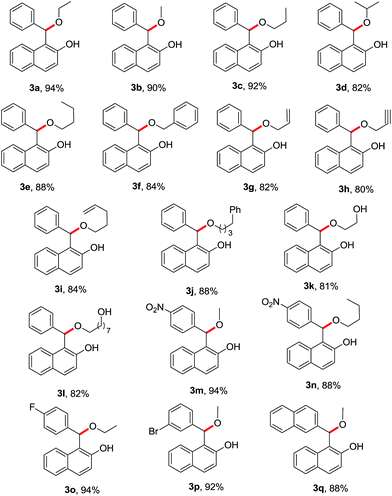 |
Subsequently, the reactions were carried out with different alcohols such as allyl alcohol (2g), propargyl alcohol (2h), 4-pentene-1-ol (2i), 4-phenylbutan-1-ol (2j), 1,2-ethanediol (2k) and 1,8-octanediol (2l) with 1a under identical reaction conditions and the desired products 3g–l were isolated in good yields (Table 2, entries 7–12). The product 3k was further confirmed by acetylation, which was characterized from 1H NMR and 13C NMR spectra.
To explore the synthetic scope and generality of the present protocol, other sulfides 1b–d containing different substituents in the aromatic ring such as NO2, F, and Br were treated with methanol (2b), ethanol (2a) and butanol (2b), in the presence of one equivalent BDMS and the desired products 3m–p were isolated in good yields as shown in Table 2 (entries 13–16). Similarly, 1-(naphthalen-2-yl(p-tolylthio)methyl)naphthalen-2-ol (1e) react with methanol under similar reaction condition and the expected products 3q obtained in 88% yield respectively.
It is worthwhile to mention that sulfides synthesized from aliphatic thiols such as 1h and 1i can be transformed into the product 3b and 3m with 88% and 90% in the presence of methanol using one equivalent of BDMS at room temperature. However, the expected product is not formed in case of phenol under similar reaction condition.
Encouraged by these successful results, we thought to explore the synthesis of 1-(hydroxy(aryl)methyl)naphthalen-2-ol (7) derivatives using unsymmetrical sulfide 1 with water as a nucleophile.16 Various other aromatic sulfide derivatives 1a–c and 1f on treatment with one equivalent of BDMS in acetonitrile solvent at room temperature provided the expected products 7a–d in good to excellent yield as shown in Table 3.
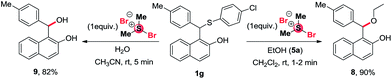
We have also carried out the reaction with different substituted sulphur moiety such as 1-(((4-chlorophenyl)thio)(p-tolyl)methyl)naphthalen-2-ol 1g with ethanol or water under identical reaction condition which afford the required product 8 and 9 with 90% and 82% yield as shown in Scheme 3.
Finally, the structures of the representative compounds 1-(alkoxy(aryl)methyl) naphthalen-2-ol such as (3p) and 1-(hydroxy(aryl)methyl)naphthalen-2-ol (7a) were also ascertained by single crystal X-ray diffraction data as shown in Fig. 1.
The compounds 3p and 7a were crystallized in P21/n monoclinic and P21/c space group, respectively. The selected bond lengths and angles are given in Table SI-S2 (see ESI†). The non-covalent strong and weak interactions such as O–H⋯O, C–H⋯O and C–H⋯π facilitate the orientation of molecules in lattice arrangement in both of crystals. The weak hydrogen bonding interactions C–H⋯O play the constructive role in stabilizing the solid-state structure of 3p. In 7a, intra- and inter-molecular O–H⋯O hydrogen bonding forms one dimensional (1D) zigzag chains which further undergo C–H⋯π interactions to furnish 2D sheets in the ac plane as shown in Fig. SI-S1 (ESI†). The hydrogen bond parameters for both the compounds are given in Table SI-S4 (see ESI†).
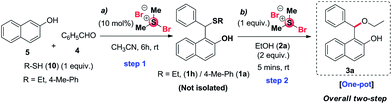
Next, we thought whether it is possible to achieve the transformation in a one-pot manner, by trapping 2-naphthoquinone-1-methide intermediate with thiol using 10 mol% BDMS followed by cleavage of it with another equivalent of BDMS. For this purpose, two set of reactions were conducted using benzaldehyde, 2-naphthol and ethane thiol or 4-methylthiophenol followed by addition of 2 equiv. of ethanol as shown in Scheme 4 and the desired product 3a was isolated in 64% and 70% yield, respectively. It is noteworthy to mention here that when similar reaction was carried out without consequent addition of BDMS (i.e.) 1.1 equivalent of BDMS at once, it gave exclusively 1-bromo-2-naphthol instead of the expected product 3a.
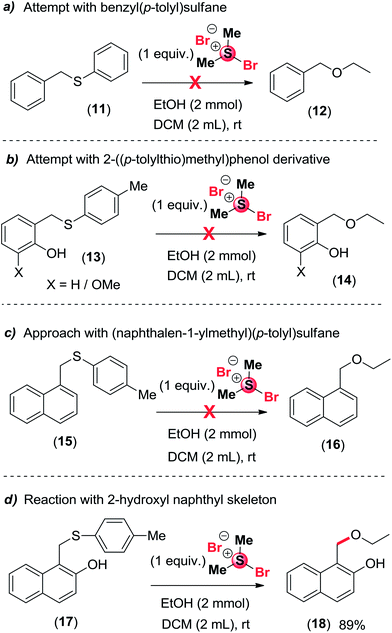
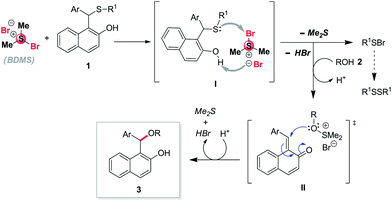
To understand the mechanism of the reaction, we have carried out four different type of reactions as shown in Scheme 5. At first, the substrate benzyl(p-tolyl)sulfane 11 was treated with treatment of 1 equiv. BDMS in the presence of 2 mmol of ethanol in 2 mL of DCM to obtain desired product 12 Unfortunately, we didn't get the expected benzyl ethyl ether. Next we thought by putting a hydroxyl group at the ortho-position of the phenyl ring, we may get the expected ether 14 from cleavage of thioether 13. In this case, the reaction failed. The failure of the reaction may be due to ortho-quinone-1-methide intermediate is very much less stable as compared to 2-naphthoquinone-1-methide intermediate. Subsequently, another reaction was carried out with fused aromatic ring of (naphthalen-1-ylmethyl)(p-tolyl)sulfane 15 under similar reaction condition and the product 16 was not formed as shown in Scheme 5. Interestingly, the installation of hydroxyl group in 2-position of (naphthalen-1-ylmethyl)(p-tolyl)sulfane 17 on treatment with 1 equivalent of BDMS in the presence of 2 mmol of ethanol in 2 mL of DCM which afford successfully 1-(ethoxymethyl)naphthalen-2-ol 18 in 89% yield as shown in Scheme 5. The obtained results indicated that presence of hydroxyl group in ortho-position of naphthalene ring in sulphide plays a crucial role for carrying out the transformations for the synthesis of unsymmetrical ether using alcohols.
The plausible mechanism for the formation of the product may be explained as follows: initially, BDMS can activate the sulphur atom on 1 for cleaving C–S bond and it tend to form an intermediate I, then it prefer to release dimethylsulfide, HBr and R1SBr. The incoming alcohol 2 may react with dimethyl sulphide and HBr to undergo oxa-Michael type reaction which proceed through the intermediate II. Subsequently, the incoming H+ ion intend to release dimethylsulfide and HBr which lead to the product 3 as shown in Scheme 6. The failure of the two reactions in Scheme 5(a and c) evidently discard the possibility of the reaction going through direct nucleophilic substitution. The presence of –OH group at the ortho-position of naphthalene ring in intermediate I plays a prime role to facilitate the cleavage of C–S bond.
Conclusions
In short, we have developed a new methodology for the synthesis of unreported unsymmetrical ether derivatives 1-(alkoxy(aryl)methyl)naphthalen-2-ols using 1-(aryl(alkyl/aryl thio)methyl)-naphthalene-2-ol and alcohols in the presence of BDMS. This protocol might be useful for the preparation of highly substituted ethers and in addition, the reaction is simple, fast and transformation is quite effective for a wide range of substrates. Moreover, we have also demonstrated that direct transformation of 3a can be achieved in a one-pot two-step manner through consequent addition of 1.1 equiv. amount of BDMS.General procedure
Synthesis of 1-(alkoxy(aryl)methyl)naphthalen-2-ol (3)
In a 25 mL round bottom flask a mixture of compound 1 (1.0 mmol) and alcohol (2.0 mmol) was taken in 2 mL of DCM. Then, 1 equivalent of BDMS was added to the reaction mixture and the reaction was completed instantaneously. After completion of reaction, the reaction mixture was extracted with DCM (1 × 25 mL) and washed with aqueous sodium bicarbonate. The organic layer was dried over Na2SO4 and it was concentrated in a rotatory evaporator. After purification through a silica gel (60–120 mesh) column chromatography, the pure product 3 was obtained in good yield. Similar reaction procedure was followed for the preparation of compounds 8.One-pot synthesis of 1-(ethoxy(phenyl)methyl)naphthalen-2-ol derivatives (3a)
Bromodimethylsulfonium bromide BDMS (0.2 mmol) was added to a mixture of benzaldehyde (2.0 mmol) and 2-naphthol (2.0 mmol) in 5 mL of acetonitrile and the reaction mixture was kept for stirring at room temperature. Then, 4-methylthiophenol (2.0 mmol) was added to it and the progress of the reaction was monitored by TLC. After 6 h of stirring, 2.0 mmol of ethanol (120 μL) and 1 equivalent BDMS were added into it and the reaction completed instantly. Then, acetonitrile was removed in a rotatory evaporator and the residue was extracted with dichloromethane (1 × 25 mL) and washed with aqueous sodium bicarbonate. The organic layer was washed with water and dried over anhydrous sodium sulfate. On concentration followed by purification through a silica gel column, the desired product 3a was obtained.Synthesis of 1-(hydroxy(aryl)methyl)naphthalen-2-ol (7)
Into a 25 mL round bottom flask was taken 1 mmol of 1 in 2 mL of CH3CN. To this reaction mixture, 40 μL of water and 1 equivalent of BDMS were added successively and the reaction mixture was stirred at room temperature for 5 minutes and the progress of the reaction was monitored by TLC. After completion of the reaction, the mixture was concentrated in a rotatory evaporator and the crude residue was extracted with DCM (1 × 25 mL) and washed with water. The organic layer was dried over Na2SO4 and it was concentrated in vacuo. The pure product 7 was obtained in good yield after purification through a silica gel (60–120 mesh) column chromatography. Similarly, 9 were obtained by following identical reaction procedure.Acknowledgements
KI is thankful to UGC for his research fellowship. RSB is grateful to CSIR for Research Associate Fellowship. AAD is thankful to IIT Guwahati for his research fellowship. DKD acknowledges to CSIR, New Delhi for SRF. ATK acknowledge CSIR, New Delhi for financial supports grant no. 02(0181)/14/EMR-II. We acknowledge SerajulHaqueFaizi and Mohammad Hedayetullah Mir for important suggestion for analyzing crystallographic data. The authors are grateful to the Director, IIT Guwahati for providing general facilities to carry out the research work. We are thankful to DST for providing XRD facility under the FIST programme.Notes and references
- (a) J. Buckingham, Dictionary of Natural Products, University: Cambridge, MA, 1994 Search PubMed; (b) T. F. Woiwode, C. Rose and T. J. Wandless, J. Org. Chem., 1998, 63, 9594 CrossRef CAS.
- (a) A. V. Ambade and A. Kumar, Prog. Polym. Sci., 2000, 25, 1141 CrossRef CAS; (b) M. Beldi, R. Medimagh, S. Chatti, S. Marque, D. Prim, A. Loupy and F. Delolme, Eur. Polym. J., 2007, 43, 3415 CrossRef CAS PubMed.
- (a) L. G. Wade, Organic Chemistry, Pearson Education, Inc, London, 5th edn, 2005 Search PubMed; (b) E. Fuhrmann and J. Talbiersky, Org. Process Res. Dev., 2005, 9, 206 CrossRef CAS.
- F. L. Lam, T. T.-L. Au-Yeung, F. Y. Kwong, Z. Zhou, K. Y. Wong and A. S. C. Chan, Angew. Chem., Int. Ed., 2008, 47, 1280 CrossRef CAS PubMed.
- (a) A. Prades, R. Corberán, M. Poyatos and E. Peris, Chem.–Eur. J., 2008, 14, 11474 CrossRef CAS PubMed; (b) T. Mitsudome, T. Matsuno, S. Sueoka, T. Mizugaki, K. Jitsukawa and K. Kaneda, Green Chem., 2012, 14, 610 RSC; (c) J. S. Yadav, D. C. Bhunia, K. V. Krishna and P. Srihari, Tetrahedron Lett., 2007, 48, 8306 CrossRef CAS PubMed.
- J.-L. Yu, H. Wang, K.-F. Zou, J.-R. Zhang, X. Gao, D.-W. Zhang and Z.-T. Li, Tetrahedron, 2013, 69, 310 CrossRef CAS PubMed.
- (a) X. J. Sun, J. F. Zhou and P. S. Zhao, J. Heterocycl. Chem., 2011, 48, 1347 CrossRef CAS PubMed; (b) J. M. Khurana, B. Nand and S. Sneha, J. Heterocycl. Chem., 2011, 48, 1388 CrossRef CAS PubMed.
- (a) A. T. Khan, S. Ali, A. A. Dar and M. Lal, Tetrahedron Lett., 2011, 52, 5157 CrossRef CAS PubMed; (b) A. Hassanabadi, J. Chem. Res., 2013, 152 CrossRef CAS.
- (a) A. R. Katritzky and X. Lan, Synthesis, 1992, 761 CrossRef CAS; (b) A. K. Shaikh, A. J. A. Cobb and G. Varvounis, Org. Lett., 2012, 14, 584 CrossRef CAS PubMed; (c) S. Arumugam and V. V. Popik, J. Am. Chem. Soc., 2011, 133, 15730 CrossRef CAS PubMed; (d) S. Arumugam and V. V. Popik, J. Am. Chem. Soc., 2011, 133, 5573 CrossRef CAS PubMed.
- (a) C. F. Nising and S. Brӓse, Chem. Soc. Rev., 2012, 41, 988 RSC; (b) C. F. Nising and S. Brӓse, Chem. Soc. Rev., 2008, 37, 1218 RSC.
- (a) E. Mondal, G. Bose and A. T. Khan, Synlett, 2001, 785 CrossRef CAS; (b) E. Mondal, G. Bose, P. R. Sahu and A. T. Khan, Chem. Lett., 2001, 1158 CrossRef CAS.
- (a) P. M. B. Barua, P. R. Sahu, E. Mondal, G. Bose and A. T. Khan, Synlett, 2002, 81 CrossRef CAS; (b) E. Mondal, P. M. B. Barua, G. Bose and A. T. Khan, Chem. Lett., 2002, 210 CrossRef CAS.
- (a) A. T. Khan, A. Choudhury, S. Ali and M. M. Khan, Tetrahedron Lett., 2012, 53, 4852 CrossRef CAS PubMed; (b) A. T. Khan, A. Ali, P. Goswami and L. H. Choudhury, J. Org. Chem., 2006, 71, 8961 CrossRef CAS PubMed; (c) S. Bhattacharjee, D. K. Das and A. T. Khan, Tetrahedron Lett., 2015, 56, 2412 CrossRef CAS PubMed; (d) A. T. Khan, R. S. Basha, M. Lal and M. H. Mir, RSC Adv., 2012, 2, 5506 RSC; (e) A. T. Khan, R. S. Basha and M. Lal, Tetrahedron Lett., 2012, 53, 2211 CrossRef CAS PubMed; (f) A. T. Khan and M. M. Khan, Carbohydr. Res., 2010, 345, 2139 CrossRef CAS PubMed; (g) L. H. Choudhury, T. Parvin and A. T. Khan, Tetrahedron, 2009, 65, 9513 CrossRef CAS PubMed; (h) A. T. Khan, T. Parvin and L. H. Choudhury, J. Org. Chem., 2008, 73, 8398 CrossRef CAS PubMed.
- (a) S. Gazi and R. Ananthakrishnan, RSC Adv, 2012, 2, 7781 RSC; (b) D. K. Yadav, R. Patel, V. P. Srivastava, G. Watal and L. D. S. Yadav, Tetrahedron Lett., 2010, 51, 5701 CrossRef CAS PubMed; (c) L. D. S. Yadav, R. Patel and V. P. Srivastava, Tetrahedron Lett., 2010, 51, 739 CrossRef CAS PubMed; (d) B. Jiang, Y. Dou, X. Xu and M. Xu, Org. Lett., 2008, 10, 593 CrossRef CAS PubMed; (e) D. S. Bhalerao and K. G. Akamanchi, Synlett, 2007, 2952 CAS; (f) B. Das, Y. Srinivas, H. Holla, K. Laxminarayana and R. Narender, Tetrahedron Lett., 2007, 48, 6681 CrossRef CAS PubMed.
- H. Loghmani-Khouzani, M. R. Poorheravi, M. M. M. Sadeghi, L. Caggiano and R. F. W. Jackson, Tetrahedron, 2008, 64, 7419 CrossRef CAS PubMed.
- (a) L.-Z. Dai, M.-J. Qi, Y.-L. Shi, X.-G. Liu and M. Shi, Org. Lett., 2007, 9, 3191 CrossRef CAS PubMed; (b) T. Hirabayashi, Y. Okimoto, A. Saito, M. Morita, S. Sakaguchi and Y. Ishii, Tetrahedron, 2006, 62, 2331 CrossRef PubMed; (c) M. Koskinen and K. Hemminki, Org. Lett., 1999, 1, 1233 CrossRef CAS.
Footnotes |
| † Electronic supplementary information (ESI) available: X-ray crystallographic data of 3p and 7a CCDC no. is 913509 and 913508 and copies of the 1H and 13C NMR spectra for all compounds. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c5ra14563e |
| ‡ These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2015 |